Abstract
The COVID-19 outbreak emerged in December 2019 and has rapidly become a global pandemic. A great deal of effort has been made to find effective drugs against this disease. Chloroquine (CQ) and hydroxychloroquine (HCQ) were widely adopted in treating COVID-19, but the results were contradictive. CQ/HCQ have been used to prevent and treat malaria and are efficacious anti-inflammatory agents in rheumatoid arthritis and systemic lupus erythematosus. These drugs have potential broad-spectrum antiviral properties, but the underlying mechanisms are speculative. In this review, we re-evaluated the treatment outcomes and current hypothesis for the working mechanisms of CQ/HCQ as COVID-19 therapy with a special focus on disruption of Ca2+ signaling. In so doing, we attempt to show how the different hypotheses for CQ/HCQ action on coronavirus may interact and reinforce each other. The potential toxicity is also noted due to its action on Ca2+ and hyperpolarization-activated cyclic nucleotide-gated channels in cardiac myocytes and neuronal cells. We propose that intracellular calcium homeostasis is an alternative mechanism for CQ/HCQ pharmacology, which should be considered when evaluating the risks and benefits of therapy in these patients and other perspective applications.
Keywords: Chloroquine, Hydroxychloroquine, Calcium, HCN, SARS-CoV-2, Cardiology, Pathology, Infectious disease, Neurology, Pharmacology, Immunology, Clinical toxicology, Toxicology, Eye-ear-nose-throat, Internal medicine
Chloroquine, Hydroxychloroquine, Calcium, HCN, SARS-CoV-2; Cardiology; Pathology; Infectious disease; Neurology; Pharmacology; Immunology; Clinical toxicology; Toxicology; Eye-ear-nose-throat; Internal medicine
1. Introduction
The outbreak of COVID-19 at the end of 2019 has caused more than ten million confirmed infections and over half a million deaths. Despite strong mobilization and disease containment, the pandemic has spread worldwide. There have been many attempts to cure and mitigate the effects of SARS-CoV-2 infection, but an effective treatment is still highly desired. Among the treatments used so far, two structurally related quinoline drugs, chloroquine (CQ) and hydroxychloroquine (HCQ), have been widely adopted for prophylaxis, compassionate use or clinical trials (Arshad et al., 2020; Boulware et al., 2020; Chen et al., 2020b; Gao et al., 2020; Gautret et al., 2020; Huang et al., 2020; Molina et al., 2020; Principi and Esposito, 2020; Rosenberg et al., 2020; Yu et al., 2020), however, these studies have yielded conflicting reports. This prompted us to reconsider the proposed mechanisms of action of CQ/HCQ (MOA).
2. Clinical use and mechanisms
2.1. Current hypothesis
As a generic drug, quinine was initially employed to treat malaria but has since been succeeded by chloroquine and its derivative, hydroxychloroquine. These drugs were later used for rheumatoid arthritis and systemic lupus erythematosus (Rainsford et al., 2015). Their mechanisms of action and side effects are similar, and have been well reported (Frisk-Holmberg et al., 1983; Korinihona et al., 1992; Maxwell et al., 2015). More recently, CQ/HCQ was evolving to be deployed to treat several viral infections, such as Borna disease, hepatitis A and AIDS (Savarino et al., 2003). Encouraged by successful clinical use for these infectious diseases, Savarino and colleagues first proposed that CQ/HCQ may be repurposed to treat coronavirus during the severe acute respiratory syndrome (SARS) outbreak in 2003 (Savarino et al., 2003). This hypothesis was soon validated by in vitro studies which demonstrated inhibition of SARS-CoV (Keyaerts et al., 2004; Vincent et al., 2005). Clinical studies were not proceeded as SARS pandemic was put out quickly, but using animal models, CQ has been revealed to have antiviral activity against human coronavirus OC43 (Keyaerts et al., 2009). Wang and colleagues were amongst the first to perform In vitro studies demonstrating inhibition of SARS-CoV-2 (Wang et al., 2020), prompting widespread clinical interest in CQ/HCQ, but the efficacy and underlying MOA remains debatable (Cortegiani et al., 2020; Touret and de Lamballerie, 2020).
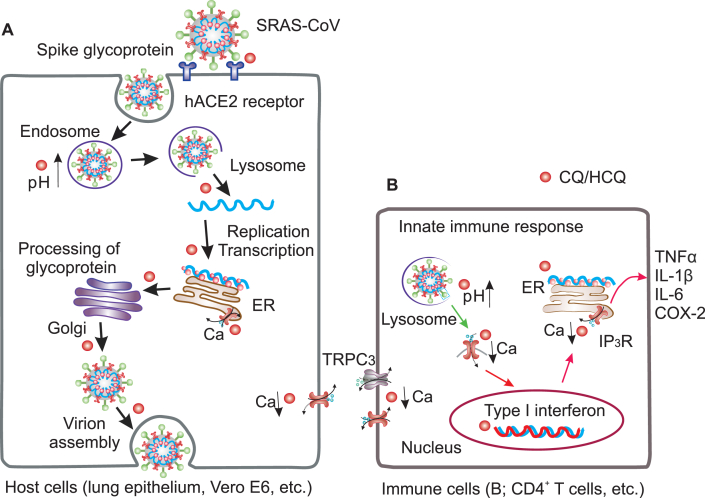
A long standing hypothesis suggests that CQ/HCQ may exhibit effects on coronavirus through two different mechanisms: pH elevation of endosomes/lysosomes and immunity modulation (Figure 1A,B) (Savarino et al., 2003; Tripathy et al., 2020; Zumla et al., 2016). The pH regulation may be a common mechanism for malaria and COVID-19. For antimalarial use, CQ/HCQ can be trapped in food vacuoles of the parasite, a lysosome-like organelle, where it inhibits formation of hemozoin (from the heme released by digestion of hemoglobin) and accumulates waste, leading to parasite death (Sullivan et al., 1996). Likewise, CQ/HCQ in its unprotonated form can diffuse passively across cellular membranes where it becomes protonated and trapped in the endosomes/lysosomes, alkalinizing these acidic compartments. This alkaline environment may block viral entry into vulnerable host cells (Figure 1A) as SARS-CoV-2 entry is facilitated by spike protein with the host cell factors (ACE2), and this process employs serine protease TMPRSS2 for spike protein priming, whereas the TMPRSS2 priming uses endosomal cysteine proteases cathepsin B/L that requires low pH (Hoffmann et al., 2020a). Furthermore, when endosomes fuse with lysosomes, the high pH can also prevent viral envelope from breaking and liberating RNAs into host cells. Additionally, it also inhibits viral RNA replication as relevant enzymic functions are compromised by an elevated pH. This hypothesis was supported by well-designed in vitro study: when the Vero E6 culture was inoculated with SARS-CoV in presence of CQ, potent anti-viral activity can be observed; whereas when CQ was added later, anti-viral activity diminished (Keyaerts et al., 2004). However, this result could not be duplicated in Calu-3 lung cells (Hoffmann et al., 2020b) and human airway epithelium (Maisonnasse et al., 2020). It should be noted that pH elevation by ammonium chloride can block infection of 293T and Caco-2 cell lines (Hoffmann et al., 2020a), and the failure in lung cells is likely attributed to cell types. The experiment on primary epithelial cells has not yet to be done, and more work is needed to validate this hypothesis. The cell line independent effects implicate that CQ/HCQ may be beneficial via some types of cells (such as kidney cells) rather than lung cells, as COVID-19 is not a typical pneumonia and it can be more broadly defined as system disease for multiple organs being infected. Besides, several other postulations were proposed as well. It should be noted that through regulating endosome/lysosome function on vasculature, CQ/HCQ may serve as an anti-thrombotic agent to improve hypoxia commonly found in acute respiratory distress syndrome (ARDS) (Ackermann et al., 2020; Jung et al., 2010; Maes et al., 2016). Though the evidence is limited, this is of interest as blood clotting is one of the major fatal processes in COVID-19.
Figure 1.
Highlights of SARRS-CoV-2 lifecycle and proposed mechanisms of CQ/HCQ interruption of virulence: 1) modulation of endosome or lysosome function by elevation of pH, thereby interrupting virus entry, as acidic pH is required for glycosyltransferases to modify envelope glycoprotein. CQ/HCQ impairs viral spike proteins binding with ACE2 receptors, thereby blocking virus-host cell fusion; Note that solid black arrows indicate the critical phases by which CQ/HCQ may disrupt the lifecycle of coronavirus (A). 2) Modulation of cytokine production (TNFα, etc.) and antigen presentation. The typical innate response to SARS-CoV-2 is marked by type I interferon. The anti-inflammatory property is proved effective for rheumatoid arthritis and lupus (B); 3) Intervention of intracellular calcium gradients, impairing RNA replication, spike glycoprotein production, and innate inflammatory response. Alternatively, attenuation of Ca2+ influx through viroporin (Green arrow) to mediate proinflammation (B). ER. Endoplasmic reticulum; hACE2, human angiotensin-converting enzyme 2; TNFα, tumor necrosis factor α.
The immunity hypothesis was derived from the successful use of CQ/HCQ in rheumatoid arthritis and lupus erythematosus to suppress inflammation by decreasing proinflammatory cytokine production (Fox, 1993; Rainsford et al., 2015). In this scenario, CQ/HCQ may play a role in COVID-19 as an immunomodulant to suppress fatal hyperinflammation (Mehta et al., 2020). However, despite suppression of the cytokine storm is beneficial in severe COVID-19 cases, this may also disrupt the ability to process antigen and the subsequent immune response, which may have adverse consequences in combatting viral infection. In addition, chloroquine might affect antigen-antibody interaction, thereby compromising immune neutralization to coronal virus when used for prolong time or under high dosages (Edwards et al., 1982). Therefore, the ultimate effects of CQ/HCQ on the innate immune response to SARS-CoV-2 remains to be determined (Meyerowitz et al., 2020).
2.2. Calcium homeostasis as an emerging antiviral target?
As virus exploits the machinery and intracellular environment of a host cell for viral lifecycles, there are several other likely targets critical for RNA duplication, transcription or budding (Clark and Eisenstein, 2013; Schoeman and Fielding, 2019). One potential target is calcium channels and intracellular calcium pool. CQ/HCQ appears to serve as an antagonistic agent to modulate slow calcium channels in atrial trabeculae (Filippov et al., 1989), calcium signaling in primary B lymphocytes (Wu et al., 2017) and murine CD4+ Thymocytes (Xu et al., 2015). Xu and colleagues further proved that chloroquine inhibits IP3R-mediated Ca2+ release from intracellular stores and TRPC3 channel mediated Ca2+ influx in CD4+ T cells (Figure 1B). Interestingly, our previous work demonstrated that quinine can reversibly block hyperpolarization-activated cyclic nucleotide-gated (HCN) channels in dopaminergic neurons (Zou et al., 2018), which is weakly permeable to Ca2+, implicating an alternative mode of action by quinolone drugs (Postea and Biel, 2011). Furthermore, our preliminary data showed that compartmental Ca2+ concentration in cortical neurons derived from human induced pluripotent stem cells (iPSCs) is differently attenuated by chloroquine. This data suggests that chloroquine can modulate intracellular Ca2+ by attenuation both extracellular Ca2+ entry and intracellular store-operated calcium (SOC) release. Therefore, CQ/HCQ may alternatively block virulence by interfering cytosolic Ca2+ required for viral lifecycles in vulnerable cells, such as airway epithelial cells, pulmonary capillary endothelial cells and other types of cells (Wiersinga et al., 2020; Zhang et al., 2020) (Figure 1A).
Targeting host store-operated Ca2+ release has been suggested a general strategy to attenuate viral infection (Clark and Eisenstein, 2013; Zhou et al., 2009). In fact, Ca2+ involvement in viral replication, budding or shell assembly has been demonstrated in a number of viruses, such as rotavirus, Dengue virus, polyomavirus, bovine herpesvirus and Hepatitis A or B virus (Bishop and Anderson, 1997; Cui et al., 2019; Dionicio et al., 2018; Geng et al., 2012; Han et al., 2015; Li et al., 2003; Lobeck et al., 2016; Sun et al., 2017; Zhang et al., 2016; Zhu et al., 2017), it is highly plausible for RNA replication in coronaviruses as well. In addition, spike protein of SARS-CoV was revealed to induce an influx of extracellular Ca2+, leading to activation of a Ca2+-dependent protein kinase C alpha (Liu et al., 2007). This process is critical for SARS-associated coronavirus activated cyclooxygenase-2 (COX-2) expression, as the PKC alpha modulates downstream ERK/NF-kappa B pathway, which is required to activate COX-2 protein, a putative marker of inflammatory response. Thus, CQ/HCQ may modulate inflammatory responses by blocking Ca2+ influx into host cells. Further, CQ/HCQ may directly modulate virulence by affecting coronavirus envelope (E) protein Ca2+ transport. SARS-CoV E protein is a viroporin which allows Ca2+ and other cations to pass through. This is a critical process to trigger proinflammation, such as IL-1β, a potent cytokine (Figure 1B). SARS E protein can even form a protein-lipid channel in Golgi membrane by which Ca2+ can mediate pH and E protein pore selectivity. Therefore, CQ/HCQ may alternatively modulate immune reaction by blocking Ca2+ influx through viroporin and suppression of SARS-CoV-2 derived, exacerbated pro-inflammation (Nieto-Torres et al., 2015; Schoeman and Fielding, 2019). Furthermore, CQ/HCQ may serve as an immunomodulant by blocking Ca2+-dependent calcineurin activity (Tanaka et al., 2013), as the inhibition of calcineurin blocks the translocation of nuclear factor of activated T cells, thereby prevent the transcription of cytokine. These lines of evidence suggests that cytosolic Ca2+ modulation may be an underlying mechanism through which CQ/HCQ serves as a broad-spectrum antiviral agent (Clark and Eisenstein, 2013; Keyaerts et al., 2004, 2009; Vincent et al., 2005; Yan et al., 2013). Given the above-mentioned considerations and the close phylogenic relationship between SARS-CoV and SARS-CoV-2, targeting on calcium is likely to be a part of CQ/HCQ effects for COVID-19.
2.3. The CQ/HCQ efficacy in line with the pathogenesis of COVID-19
The discrepancy between the results of in vitro studies showing efficacy of CQ/HCQ and the results of clinical trials has been frustrating. While several reports have implied an improved efficacy administrated alone or with a concomitant use of azithromycin or zinc (Gautret et al., 2020; Yu et al., 2020), limited benefits for either prophylaxis or treatment have also been reported (Boulware et al., 2020; Magagnoli et al., 2020; Wiersinga et al., 2020). Presumably, Ca2+ dynamics is different in vitro and in vivo and Ca2+ homeostasis in COVID19 patients is likely to change during the disease progress from the prodromal, to the mild and severe phases. In these different phases, symptoms of COVID-19 become highly heterogenous. This involves respiratory and kidney failure, leading to hypoxia and dysfunctional homeostasis. On the other hand, in vitro study is well controlled for variables such as temperature, pH, oxygen and ion gradients, which may not be achieved when there is impaired pulmonary, renal and circulatory function (Takahashi and Suzuki, 2015). Of note, Low plasma pH is associated with low CQ/HCQ concentration in endosome/lysosome and high probability of blood coagulate (Crowell and Houston, 1961; Warhurst et al., 2007). With these consideration, Zn2+ and Azithromycin have been commonly used as adjuvants in combination with CQ/HCQ to achieve synergistic effect (Retallack et al., 2016; Schogler et al., 2015; te Velthuis et al., 2010; Xue et al., 2014; Zhu et al., 2013), as both agents have anti-viral properties. Specifically, CQ/HCQ is also suggested to be an Zn2+ ionophore to increase Zn2+ influx, contributing versatile actions within endosome/lysosome (Skalny et al., 2020; te Velthuis et al., 2010; Xue et al., 2014). We suggest that plasma Zn2+ test along with CQ/HCQ concentration assay is much needed in future clinical application. In contrast to the CQ/Zn2+ synergetic effect, there is no pharmacokinetic interaction between azithromycin and chloroquine (Cook et al., 2006). However, like CQ/HCQ, azithromycin can also act as anti-viral agent through impairing lysosome function or several other possible means (Damle et al., 2020; Nujic et al., 2012).
3. Adverse effects and mechanisms
3.1. Major adverse effects
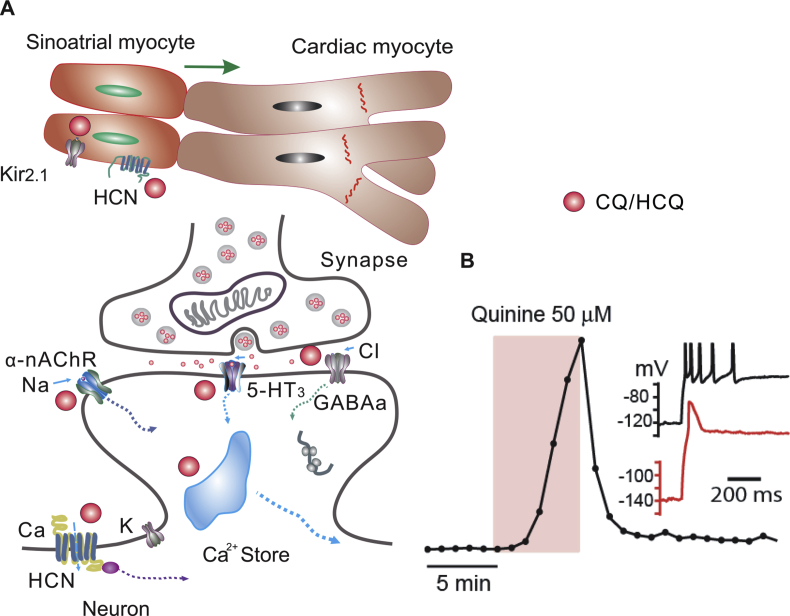
It is well established that quinoline drugs affect excitable cells, including cardiac myocytes and neurons, through modulation of cellular excitability and action potential (Wenzel and Kleoppel, 1980; White, 2007; Zou et al., 2018). Indeed, CQ/HCQ has broad effects on HCN channels and inwardly rectifying K+ channel 2.1 (Kir2.1), as both channels are phylogenetically related (Rodriguez-Menchaca et al., 2008). In addition, the HCN regulates rhythmic activity in both spontaneously spiking neurons (HCN1, 2, 4) or pace making sinoatrial myocytes (HCN3). Consequently, CQ/HCQ can mediate rhythmic activity of sinoatrial cardiac myocytes and affects heart contractility (Capel et al., 2015; Rodriguez-Menchaca et al., 2008) (Figure 2).
Figure 2.
Quinolines have major adverse effects through neurons and cardiac myocytes. A. HCN and potassium channels are expressed in both types of cells. Chloroquine diffuses into cells and binds the cytoplasmic pore domain of Kir2.1 (A). The effect on HCN and K channels on sinoatrial myocytes lead to slower pace-making activity, whereas the effect on cardiac myocytes leads to decreased excitability and aberrant electrical transduction from sinoatrial to cardiac myocytes (top panel in A; green arrow denotes the electric signal propagation direction from sinoatrial to cardiac myocytes), thereby causing QT prolongation and ventricular arrhythmias. CQ/HCQ also modulates neuronal function by intervention of intracellular calcium homeostasis and synaptic signaling by binding to α9α10-nAchR, 5-HT3 or GABAa receptors, inducing fast-onset neuropsychiatric effects (lower panel in A). B. The action potential is delayed by application of quinine in a human iPSC derived dopaminergic neuron (modified from Zou et al., 2018). The drug effect is fast and partially reversible. Inset, recording traces before (black) and after (red) quinine treatment (50 μM). GABA, γ-aminobutyric acid; 5-HT, 5-hydroxytryptamine.
The effects of CQ/HCQ on HCN is our major concern as it causes a delayed action potential in neurons and QT interval prolongation in cardiac myocytes (Figure 2) (Haeusler et al., 2018; Postea and Biel, 2011; Zou et al., 2018). The CQ/quinine adverse effects found in in vitro studies is quite similar with the clinical outcomes: both quinine and chloroquine appeared to cause a decrease of myocardial cell discharge and lead to aberrant cardiac electrical conduction and QT elongation (Figure 2). CQ also targets on nicotine receptors in neurons, thus it might affect intracardiac nerve-cardiac signaling mediated by nicotine receptors, thereby contribute to mediate the excitability of cardiac myocytes (Ballestero et al., 2005; Duraes Campos et al., 2018; Estes et al., 1987). In a small clinical trial, the mean QT prolongation was about 35 (28–43) ms and more than 20% percent of COVID-19 patients had a QT interval exceeding 500 ms during chloroquine treatment (van den Broek et al., 2020), but in general CQ/HCQ is safe even QT prolongation is commonly reported (Sarayani et al., 2020). In rare cases, this may lead to ventricular arrhythmias, insufficient ventricular blood reflux or ejection, and sudden death. Thus, it should be careful when treating COVID-19, as in severe cases of this disease, there is increased cardiac burden due to ARDS, and possibly direct cardiac injury (Chen et al., 2020a; Yang et al., 2020). As for those with comorbidities such as heart diseases, including tachycardia, bradycardia, cardiomegaly and arrhythmia (commonly found in older patients) (Xiong et al., 2020), special caution should be taken (medication may discontinue) as poor cardiac function may be significantly exacerbated by CQ/HCQ toxicity. Despite the potentially fatal effect on the QT interval, CQ/HCQ has been shown to have cardiovascular benefits by inhibiting cyclooxygenase-dependent contraction (McCarthy et al., 2016). CQ/HCQ also exerts beneficial effects in experimental pulmonary arterial hypertension (Long et al., 2013).
The sensitivity of quinolines on ion channels also raises concerns for patients with metabolic abnormalities such as hypokalemia, hypomagnesemia and hypocalcemia. Discussion of other issues of CQ/HCQ toxicity, such as those related to G-6-PD deficiency, hypoglycemia and interaction with other drugs are beyond the scope of this article (Choudhry et al., 1978; Jiang, 2020; Rajeshkumar et al., 2020; Rainsford et al., 2015).
3.2. The clinical use in consideration of CQ/HCQ toxicity
The adverse effect on different types of cells renders it a tricky drug to employ for clinical applications. A review for publications concerning CQ/HCQ toxicity prior to July 2017 has revealed that disrupted electrical conduction is the underlying mechanism for 85% of cases involved in CQ/HCQ heart toxicity (Chatre et al., 2018). These papers primarily address malaria, rheumatoid arthritis and lupus erythematosus, but their clinical effects with COVID-19 are vastly different, as the latter can be complicated by ARDS and myocarditis in the late phase, with resultant tissue hypoxia and cardiac stress (Xiong et al., 2020). As the CQ50 for anti-viral action is much higher than that for toxic effect on cardiac or neuronal cells (Liu et al., 2020; Rodriguez-Menchaca et al., 2008), the effect on excitable cells, whether neurons or cardiac cells, is more rapid than its effect on viral replication and inflammation. These side effects typically appear earlier (within hours) than anti-viral effects (days), however QT prolongation may be delayed for as long as three days due to plasma binding and reduced bioavailability (Chorin et al., 2020). The unique pharmacokinetics of these medications facilitates easy monitoring of the toxic effect on neuronal and cardiac cells.
There are two categories of drugs often associated with QT prolongation that need to be used with caution in patients treated with CQ/HCQ - psychotropic and antiarrhythmic drugs (see Table 1) (Jiang, 2020; Maxwell et al., 2015). More drugs involved in drug-drug interaction with chloroquine and QT prolongation can be found elsewhere (CredibleMeds, 2020). Interestingly, although azithromycin can cause QT prolongation, it has been safely used in combination with hydroxychloroquine with judicious monitoring, and there was no additional QT increase (Fossa et al., 2007; Gautret et al., 2020; Sagara et al., 2014).
Table 1.
Representative drugs with QT interval prolongation.
| Drug Class | Drugs |
|---|---|
| Antiarrhythmic drugs | Amiodarone, Cordarone, Dronedarone, Ibutilide, Procainamide, Sotalol; |
| Antimicrobial drugs | Astemizole, Azithromycin, Clarithromycin, Erythromycin, Fluconazole, Levofloxacin, Moxifloxacin, Quinolone, Terfenadine, Voriconazole |
| Somatostatin Analog | Octreotide |
| Psychotropic | Amitriptyline, Chlorpromazine, Citalopram, Clomipramine, Droperidol, Methadone, Haloperidol |
| Gastrointestinal | Dolasetron, Domperidone, Ondansetron |
The possible invasion of SARS-CoV-2 into the central nervous system has been highlighted by the presence of neurological symptoms such as nausea, anosmia and seizures (Le Guennec et al., 2020; Li et al., 2020; Mao et al., 2020). Of note, cholinergic system is implicated in anti-inflammatory modulation in COVID-19 (Farsalinos et al., 2020). Given all these likelihoods, the neurotoxic effects of CQ/HCQ on the nervous system should also be considered. The non-selective action on HCN and K+ channels renders quinolines capable of affecting the function of multiple types of neurons and synaptic transmission, including dopaminergic, GABAergic and α9α10-nAchR signaling (Figure 2A) (Ballestero et al., 2005; Thompson and Lummis, 2008; Zou et al., 2018). Rarely, this may lead to epileptic seizure that should prompt discontinuation of CQ/HCQ treatment (Krzeminski et al., 2018). There are also minor adverse effects including retinopathy, ototoxicity, itching and mood disturbance, and close monitoring of the effect of CQ/HCQ on neurological or neuropsychiatric, visual and auditory symptoms will also be warranted.
4. Management
CQ/HCQ can be binding with plasma and intracellular proteins, in vitro testing of quinoline pharmacology typically has a delayed washout effect, while it has a long elimination half-life (22–45 days) in administered in vivo (Cortegiani et al., 2020; McLachlan et al., 1993; Rainsford et al., 2015; Zou et al., 2018). Animal research has demonstrated a huge disparity of HQ/HCQ concentrations in different tissues or organs with the greatest accumulation in lungs (Meyerowitz et al., 2020). In this sense, the dosage for COVID-19 is still open to explore and is a challenge to current ongoing randomized placebo-controlled clinical trials and statistic interpretation. The drug bioavailability onsite changes from patients to patients due to binding capability, body weight, gender and sickness, etc., thus a more precise group concerning binding is needed, which has so far not been reported (Arshad et al., 2020; Gautret et al., 2020; Yu et al., 2020). One report revealed that twice daily of hydroxychloroquine at 200 mg, a relatively lower dosage for 7–10 days had achieved significant efficacy (Yu et al., 2020). However, there might be better benefit and less toxic side effects by following a lower dosing regimen (four or six-times daily) with close monitoring of plasma binding. More recently, a multi-hospital study of 2541 patients revealed promising results with reducing mortality by 66% with HCQ alone and 71% with HCQ/Azithromycin, respectively (Arshad et al., 2020). It is prominent that in the settings of this study, HCQ use was administered at a low dosage regimen (400 mg for first two days, following with 200 mg) in the earlier stage of COVID-19 (within 24 h confirmed by lab) and QT was closed monitored with no documented case of torsade de pointes. Interestingly, this dosage was equivalent to that recommended for rheumatoid arthritis and lupus.
5. Conclusion
The foregoing makes clear the complexity of CQ/HCQ action on SARS-CoV-2, we suggest CQ/HCQ has limited benefits for prodromal or mild COVID-19 subjects by following a definite protocol. Among other considerations, in these patients, cardiomyocytes may be healthy and resilient enough to recover from a brief toxic shock by HCQ/CQ. However, unexpected efficacy is possible due to complicated modes of action, adverse effects or patient specific conditions (CQ/HCQ bioavailability, Ca2+ concentration in the extracellular media and membrane permeability caused by pH fluctuation, oxygen saturation and fever). This is highly challenging to frontline doctors, a case-by-case strategy may be the most optional way to evaluate CQ/HCQ benefits for COVID-19 and adverse effects. A closer look at the effect of CQ/HCQ on Ca2+ homeostasis is warranted as we aim to eliminate coronavirus infection. It remains to be further validated whether cytosolic Ca2+ is a target for CQ/HCQ to suppress SARS-COVID-2 and other viral infections.
Declarations
Author contribution statement
All authors listed have significantly contributed to the development and the writing of this article.
Funding statement
This work was supported in part by HUCOM Bridge Grant, Hearing Health Foundation to X.Z. and by the National Institute on Minority Health and Health Disparities of the National Institutes of Health (2U54MD007597) to D.L.L.
Competing interest statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
Acknowledgements
We thank Drs. Celia Maxwell and Kebreten Manaye for generous support.
References
- Ackermann M., Verleden S.E., Kuehnel M., Haverich A., Welte T., Laenger F., Vanstapel A., Werlein C., Stark H., Tzankov A., Li W.W., Li V.W., Mentzer S.J., Jonigk D. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in covid-19. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arshad S., Kilgore P., Chaudhry Z.S., Jacobsen G., Wang D.D., Huitsing K., Brar I., Alangaden G.J., Ramesh M.S., McKinnon J.E., O’Neill W., Zervos M. Treatment with hydroxychloroquine, azithromycin, and combination in patients hospitalized with COVID-19. Int. J. Infect. Dis. 2020 doi: 10.1016/j.ijid.2020.06.099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballestero J.A., Plazas P.V., Kracun S., Gomez-Casati M.E., Taranda J., Rothlin C.V., Katz E., Millar N.S., Elgoyhen A.B. Effects of quinine, quinidine, and chloroquine on alpha9alpha10 nicotinic cholinergic receptors. Mol. Pharmacol. 2005;68:822–829. doi: 10.1124/mol.105.014431. [DOI] [PubMed] [Google Scholar]
- Bishop N.E., Anderson D.A. Early interactions of hepatitis A virus with cultured cells: viral elution and the effect of pH and calcium ions. Arch. Virol. 1997;142:2161–2178. doi: 10.1007/s007050050233. [DOI] [PubMed] [Google Scholar]
- Boulware D.R., Pullen M.F., Bangdiwala A.S., Pastick K.A., Lofgren S.M., Okafor E.C., Skipper C.P., Nascene A.A., Nicol M.R., Abassi M., Engen N.W., Cheng M.P., LaBar D., Lother S.A., MacKenzie L.J., Drobot G., Marten N., Zarychanski R., Kelly L.E., Schwartz I.S., McDonald E.G., Rajasingham R., Lee T.C., Hullsiek K.H. A randomized trial of hydroxychloroquine as postexposure prophylaxis for covid-19. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2016638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capel R.A., Herring N., Kalla M., Yavari A., Mirams G.R., Douglas G., Bub G., Channon K., Paterson D.J., Terrar D.A., Burton R.A. Hydroxychloroquine reduces heart rate by modulating the hyperpolarization-activated current If: novel electrophysiological insights and therapeutic potential. Heart Rhythm. 2015;12:2186–2194. doi: 10.1016/j.hrthm.2015.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatre C., Roubille F., Vernhet H., Jorgensen C., Pers Y.M. Cardiac complications attributed to chloroquine and hydroxychloroquine: a systematic review of the literature. Drug Saf. 2018;41:919–931. doi: 10.1007/s40264-018-0689-4. [DOI] [PubMed] [Google Scholar]
- Chen L., Li X., Chen M., Feng Y., Xiong C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovasc. Res. 2020;116:1097–1100. doi: 10.1093/cvr/cvaa078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z., Hu J., Zhang Z., Jiang S., Han S., Yan D., Zhuang R., Hu B., Zhang Z. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. MedRxiv. 2020 [Google Scholar]
- Chorin E., Dai M., Shulman E., Wadhwani L., Bar-Cohen R., Barbhaiya C., Aizer A., Holmes D., Bernstein S., Spinelli M., Park D.S., Chinitz L.A., Jankelson L. The QT interval in patients with COVID-19 treated with hydroxychloroquine and azithromycin. Nat. Med. 2020;26:808–809. doi: 10.1038/s41591-020-0888-2. [DOI] [PubMed] [Google Scholar]
- Choudhry V.P., Madan N., Sood S.K., Ghai O.P. Chloroquine induced haemolysis and acute renal failure in subjects with G-6-PD deficiency. Trop. Geogr. Med. 1978;30:331–335. [PubMed] [Google Scholar]
- Clark K.B., Eisenstein E.M. Targeting host store-operated Ca(2+) release to attenuate viral infections. Curr. Top. Med. Chem. 2013;13:1916–1932. doi: 10.2174/15680266113139990128. [DOI] [PubMed] [Google Scholar]
- Cook J.A., Randinitis E.J., Bramson C.R., Wesche D.L. Lack of a pharmacokinetic interaction between azithromycin and chloroquine. Am. J. Trop. Med. Hyg. 2006;74:407–412. [PubMed] [Google Scholar]
- Cortegiani A., Ingoglia G., Ippolito M., Giarratano A., Einav S. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J. Crit. Care. 2020;S0883–9441:30390–30397. doi: 10.1016/j.jcrc.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CredibleMeds . CredibleMeds; 2020. Combined list of drugs that prolong QT and/or cause Torsades De Pointes (TDP)http://www.CredibleMeds.org [Google Scholar]
- Crowell J.W., Houston B. Effect of acidity on blood coagulation. Am. J. Physiol. 1961;201:379–382. doi: 10.1152/ajplegacy.1961.201.2.379. [DOI] [PubMed] [Google Scholar]
- Cui T., Theuns S., Xie J., Nauwynck H.J. Porcine rotavirus mainly infects primary porcine enterocytes at the basolateral surface. Vet. Res. 2019;50:110. doi: 10.1186/s13567-019-0728-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Damle B., Vourvahis M., Wang E., Leaney J., Corrigan B. Clinical pharmacology perspectives on the antiviral activity of azithromycin and use in COVID-19. Clin. Pharmacol. Ther. 2020;108:201–211. doi: 10.1002/cpt.1857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dionicio C.L., Pena F., Constantino-Jonapa L.A., Vazquez C., Yocupicio-Monroy M., Rosales R., Zambrano J.L., Ruiz M.C., Del Angel R.M., Ludert J.E. Dengue virus induced changes in Ca(2+) homeostasis in human hepatic cells that favor the viral replicative cycle. Virus Res. 2018;245:17–28. doi: 10.1016/j.virusres.2017.11.029. [DOI] [PubMed] [Google Scholar]
- Duraes Campos I., Pinto V., Sousa N., Pereira V.H. A brain within the heart: a review on the intracardiac nervous system. J. Mol. Cell. Cardiol. 2018;119:1–9. doi: 10.1016/j.yjmcc.2018.04.005. [DOI] [PubMed] [Google Scholar]
- Edwards J.M., Moulds J.J., Judd W.J. Chloroquine dissociation of antigen-antibody complexes. A new technique for typing red blood cells with a positive direct antiglobulin test. Transfusion. 1982;22:59–61. doi: 10.1046/j.1537-2995.1982.22182154219.x. [DOI] [PubMed] [Google Scholar]
- Estes M.L., Ewing-Wilson D., Chou S.M., Mitsumoto H., Hanson M., Shirey E., Ratliff N.B. Chloroquine neuromyotoxicity. Clinical and pathologic perspective. Am. J. Med. 1987;82:447–455. doi: 10.1016/0002-9343(87)90444-x. [DOI] [PubMed] [Google Scholar]
- Farsalinos K., Niaura R., Le Houezec J., Barbouni A., Tsatsakis A., Kouretas D., Vantarakis A., Poulas K. Editorial: nicotine and SARS-CoV-2: COVID-19 may be a disease of the nicotinic cholinergic system. Toxicol. Rep. 2020 doi: 10.1016/j.toxrep.2020.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Filippov A., Skatova G., Porotikov V., Kobrinsky E., Saxon M. Ca2+-antagonistic properties of phospholipase A2 inhibitors, mepacrine and chloroquine. Gen. Physiol. Biophys. 1989;8:113–118. [PubMed] [Google Scholar]
- Fossa A.A., Wisialowski T., Duncan J.N., Deng S., Dunne M. Azithromycin/chloroquine combination does not increase cardiac instability despite an increase in monophasic action potential duration in the anesthetized Guinea pig. Am. J. Trop. Med. Hyg. 2007;77:929–938. [PubMed] [Google Scholar]
- Fox R.I. Mechanism of action of hydroxychloroquine as an antirheumatic drug. Semin. Arthritis Rheum. 1993;23:82–91. doi: 10.1016/s0049-0172(10)80012-5. [DOI] [PubMed] [Google Scholar]
- Frisk-Holmberg M., Bergqvist Y., Englund U. Chloroquine intoxication. Br. J. Clin. Pharmacol. 1983;15:502–503. doi: 10.1111/j.1365-2125.1983.tb01540.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao J., Tian Z., Yang X. Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci. Trends. 2020 doi: 10.5582/bst.2020.01047. [DOI] [PubMed] [Google Scholar]
- Gautret P., Lagier J., Parola P., Hoang V.T., Meddeb L., Mailhe M., Doudier B., Courjon J., Giordanengo V., Vieira V.E., Dupont H.T., Honoré S., Colson P., Chabrière E., Scola B.L., Rolain J.M., Brouqui P., Raoult D. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int. J. Antimicrob. Agents. 2020 doi: 10.1016/j.ijantimicag.2020.105949. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Geng X., Huang C., Qin Y., McCombs J.E., Yuan Q., Harry B.L., Palmer A.E., Xia N.S., Xue D. Hepatitis B virus X protein targets Bcl-2 proteins to increase intracellular calcium, required for virus replication and cell death induction. Proc. Natl. Acad. Sci. U. S. A. 2012;109:18471–18476. doi: 10.1073/pnas.1204668109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haeusler I.L., Chan X.H.S., Guerin P.J., White N.J. The arrhythmogenic cardiotoxicity of the quinoline and structurally related antimalarial drugs: a systematic review. BMC Med. 2018;16:200. doi: 10.1186/s12916-018-1188-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han Z., Madara J.J., Herbert A., Prugar L.I., Ruthel G., Lu J., Liu Y., Liu W., Liu X., Wrobel J.E., Reitz A.B., Dye J.M., Harty R.N., Freedman B.D. Calcium regulation of hemorrhagic fever virus budding: mechanistic implications for host-oriented therapeutic intervention. PLoS Pathog. 2015;11 doi: 10.1371/journal.ppat.1005220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Muller M.A., Drosten C., Pohlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. e278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Mösbauer K., Hofmann-Winkler H., Kaul A., Kleine-Weber H., Krüger N., Gassen N.C., Müller M.A., Drosten C., Pöhlmann S. Chloroquine does not inhibit infection of human lung cells with SARS-CoV-2. Nature. 2020 doi: 10.1038/s41586-020-2575-3. [DOI] [PubMed] [Google Scholar]
- Huang M., Tang T., Pang P., Li M., Ma R., Lu J., Shu J., You Y., Chen B., Liang J., Hong Z., Chen H., Kong L., Qin D., Pei D., Xia J., Jiang S., Shan H. Treating COVID-19 with chloroquine. J. Mol. Cell Biol. 2020:1–4. doi: 10.1093/jmcb/mjaa014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang S. Expert consensus on chloroquine phosphate for the treatment of novel coronavirus pneumonia. Chin. J. Tuberc. Respir. Dis. 2020;43:E019. doi: 10.3760/cma.j.issn.1001-0939.2020.0019. E019. [DOI] [PubMed] [Google Scholar]
- Jung H., Bobba R., Su J., Shariati-Sarabi Z., Gladman D.D., Urowitz M., Lou W., Fortin P.R. The protective effect of antimalarial drugs on thrombovascular events in systemic lupus erythematosus. Arthritis Rheum. 2010;62:863–868. doi: 10.1002/art.27289. [DOI] [PubMed] [Google Scholar]
- Keyaerts E., Vijgen L., Maes P., Neyts J., Van Ranst M. In vitro inhibition of severe acute respiratory syndrome coronavirus by chloroquine. Biochem. Biophys. Res. Commun. 2004;323:264–268. doi: 10.1016/j.bbrc.2004.08.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keyaerts E., Li S., Vijgen L., Rysman E., Verbeeck J., Van Ranst M., Maes P. Antiviral activity of chloroquine against human coronavirus OC43 infection in newborn mice. Antimicrob. Agents Chemother. 2009;53:3416–3421. doi: 10.1128/AAC.01509-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korinihona A., Laurenson I.F., Naraqi S. Chloroquine overdose in adults: a practical approach to management. P N G Med. J. 1992;35:311–318. [PubMed] [Google Scholar]
- Krzeminski P., Lesiak A., Narbutt J. Seizures as a rare adverse effect of chloroquine therapy in systemic lupus erythematosus patients: a case report and literature survey. Postepy Dermatol. Alergol. 2018;35:429–430. doi: 10.5114/ada.2018.77675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Guennec L., Devianne J., Jalin L., Cao A., Galanaud D., Navarro V., Boutolleau D., Rohaut B., Weiss N., Demeret S. Orbitofrontal involvement in a neuroCOVID-19 patient. Epilepsia. 2020 doi: 10.1111/epi.16612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li P.P., Naknanishi A., Tran M.A., Ishizu K., Kawano M., Phillips M., Handa H., Liddington R.C., Kasamatsu H. Importance of Vp1 calcium-binding residues in assembly, cell entry, and nuclear entry of simian virus 40. J. Virol. 2003;77:7527–7538. doi: 10.1128/JVI.77.13.7527-7538.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y.C., Bai W.Z., Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020 doi: 10.1002/jmv.25728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu M., Yang Y., Gu C., Yue Y., Wu K.K., Wu J., Zhu Y. Spike protein of SARS-CoV stimulates cyclooxygenase-2 expression via both calcium-dependent and calcium-independent protein kinase C pathways. Faseb. J. 2007;21:1586–1596. doi: 10.1096/fj.06-6589com. [DOI] [PubMed] [Google Scholar]
- Liu J., Cao R., Xu M., Wang X., Zhang H., Hu H., Li Y., Hu Z., Zhong W., Wang M. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov. 2020;6:16. doi: 10.1038/s41421-020-0156-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lobeck I., Donnelly B., Dupree P., Mahe M.M., McNeal M., Mohanty S.K., Tiao G. Rhesus rotavirus VP6 regulates ERK-dependent calcium influx in cholangiocytes. Virology. 2016;499:185–195. doi: 10.1016/j.virol.2016.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long L., Yang X., Southwood M., Lu J., Marciniak S.J., Dunmore B.J., Morrell N.W. Chloroquine prevents progression of experimental pulmonary hypertension via inhibition of autophagy and lysosomal bone morphogenetic protein type II receptor degradation. Circ. Res. 2013;112:1159–1170. doi: 10.1161/CIRCRESAHA.111.300483. [DOI] [PubMed] [Google Scholar]
- Maes H., Kuchnio A., Carmeliet P., Agostinis P. Chloroquine anticancer activity is mediated by autophagy-independent effects on the tumor vasculature. Mol. Cell Oncol. 2016;3 doi: 10.4161/23723548.2014.970097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magagnoli J., Narendran S., Pereira F., Cummings T., Hardin J.W., Sutton S.S., Ambati J. Outcomes of hydroxychloroquine usage in United States veterans hospitalized with Covid-19. medRxiv. 2020 doi: 10.1016/j.medj.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maisonnasse P., Guedj J., Contreras V., Behillil S., Solas C., Marlin R., Naninck T., Pizzorno A., Lemaitre J., Gonçalves A., Kahlaoui N., Terrier O., Fang R., Enouf V., Dereuddre-Bosquet N., Brisebarre A., Touret F., Chapon C., Hoen B., Lina B., Calatrava M.R., van der Werf S., de Lamballerie X., Le Grand R. Hydroxychloroquine use against SARS-CoV-2 infection in non-human primates. Nature. 2020 doi: 10.1038/s41586-020-2558-4. [DOI] [PubMed] [Google Scholar]
- Mao L., Jin H., Wang M., Hu Y., Chen S., He Q., Chang J., Hong C., Zhou Y., Wang D., Miao X., Li Y., Hu B. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in wuhan, China. JAMA Neurol. 2020 doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxwell N.M., Nevin R.L., Stahl S., Block J., Shugarts S., Wu A.H., Dominy S., Solano-Blanco M.A., Kappelman-Culver S., Lee-Messer C., Maldonado J., Maxwell A.J. Prolonged neuropsychiatric effects following management of chloroquine intoxication with psychotropic polypharmacy. Clin. Case Rep. 2015;3:379–387. doi: 10.1002/ccr3.238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCarthy C.G., Wenceslau C.F., Goulopoulou S., Ogbi S., Matsumoto T., Webb R.C. Autoimmune therapeutic chloroquine lowers blood pressure and improves endothelial function in spontaneously hypertensive rats. Pharmacol. Res. 2016;113:384–394. doi: 10.1016/j.phrs.2016.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLachlan A.J., Cutler D.J., Tett S.E. Plasma protein binding of the enantiomers of hydroxychloroquine and metabolites. Eur. J. Clin. Pharmacol. 1993;44:481–484. doi: 10.1007/BF00315548. [DOI] [PubMed] [Google Scholar]
- Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J., Hlh Across Speciality Collaboration, U.K. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020 doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyerowitz E.A., Vannier A.G.L., Friesen M.G.N., Schoenfeld S., Gelfand J.A., Callahan M.V., Kim A.Y., Reeves P.M., Poznansky M.C. Rethinking the role of hydroxychloroquine in the treatment of COVID-19. Faseb. J. 2020;34:6027–6037. doi: 10.1096/fj.202000919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molina J.M., Delaugerre C., Le Goff J., Mela-Lima B., Ponscarme D., Goldwirt L., de Castro N. No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients. Med. Maladies Infect. 2020 doi: 10.1016/j.medmal.2020.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieto-Torres J.L., Verdia-Baguena C., Jimenez-Guardeno J.M., Regla-Nava J.A., Castano-Rodriguez C., Fernandez-Delgado R., Torres J., Aguilella V.M., Enjuanes L. Severe acute respiratory syndrome coronavirus E protein transports calcium ions and activates the NLRP3 inflammasome. Virology. 2015;485:330–339. doi: 10.1016/j.virol.2015.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nujic K., Banjanac M., Munic V., Polancec D., Erakovic Haber V. Impairment of lysosomal functions by azithromycin and chloroquine contributes to anti-inflammatory phenotype. Cell. Immunol. 2012;279:78–86. doi: 10.1016/j.cellimm.2012.09.007. [DOI] [PubMed] [Google Scholar]
- Postea O., Biel M. Exploring HCN channels as novel drug targets. Nat. Rev. Drug Discov. 2011;10:903–914. doi: 10.1038/nrd3576. [DOI] [PubMed] [Google Scholar]
- Principi N., Esposito S. Chloroquine or hydroxychloroquine for prophylaxis of COVID-19. Lancet Infect. Dis. 2020 doi: 10.1016/S1473-3099(20)30296-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rainsford K.D., Parke A.L., Clifford-Rashotte M., Kean W.F. Therapy and pharmacological properties of hydroxychloroquine and chloroquine in treatment of systemic lupus erythematosus, rheumatoid arthritis and related diseases. Inflammopharmacology. 2015;23:231–269. doi: 10.1007/s10787-015-0239-y. [DOI] [PubMed] [Google Scholar]
- Rajeshkumar N.V., Y S., Pai Shweta G., Maitra Anirban, Hidalgo Manuel, Dang Chi V. Fatal toxicity of chloroquine or hydroxychloroquine with metformin in mice. BioRxiv. 2020 [Google Scholar]
- Retallack H., Di Lullo E., Arias C., Knopp K.A., Laurie M.T., Sandoval-Espinosa C., Mancia Leon W.R., Krencik R., Ullian E.M., Spatazza J., Pollen A.A., Mandel-Brehm C., Nowakowski T.J., Kriegstein A.R., DeRisi J.L. Zika virus cell tropism in the developing human brain and inhibition by azithromycin. Proc. Natl. Acad. Sci. U. S. A. 2016;113:14408–14413. doi: 10.1073/pnas.1618029113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Menchaca A.A., Navarro-Polanco R.A., Ferrer-Villada T., Rupp J., Sachse F.B., Tristani-Firouzi M., Sanchez-Chapula J.A. The molecular basis of chloroquine block of the inward rectifier Kir2.1 channel. Proc. Natl. Acad. Sci. U. S. A. 2008;105:1364–1368. doi: 10.1073/pnas.0708153105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg E.S., Dufort E.M., Udo T., Wilberschied L.A., Kumar J., Tesoriero J., Weinberg P., Kirkwood J., Muse A., DeHovitz J., Blog D.S., Hutton B., Holtgrave D.R., Zucker H.A. Association of treatment with hydroxychloroquine or azithromycin with in-hospital mortality in patients with COVID-19 in New York state. JAMA. 2020 doi: 10.1001/jama.2020.8630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sagara I., Oduro A.R., Mulenga M., Dieng Y., Ogutu B., Tiono A.B., Mugyenyi P., Sie A., Wasunna M., Kain K.C., Djimde A.A., Sarkar S., Chandra R., Robbins J., Dunne M.W. Efficacy and safety of a combination of azithromycin and chloroquine for the treatment of uncomplicated Plasmodium falciparum malaria in two multi-country randomised clinical trials in African adults. Malar. J. 2014;13:458. doi: 10.1186/1475-2875-13-458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarayani A., Cicali B., Henriksen C.H., Brown J.D. Safety signals for QT prolongation or Torsades de Pointes associated with azithromycin with or without chloroquine or hydroxychloroquine. Res. Soc. Adm. Pharm. 2020 doi: 10.1016/j.sapharm.2020.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savarino A., Boelaert J.R., Cassone A., Majori G., Cauda R. Effects of chloroquine on viral infections: an old drug against today's diseases? Lancet Infect. Dis. 2003;3:722–727. doi: 10.1016/S1473-3099(03)00806-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoeman D., Fielding B.C. Coronavirus envelope protein: current knowledge. Virol. J. 2019;16:69. doi: 10.1186/s12985-019-1182-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schogler A., Kopf B.S., Edwards M.R., Johnston S.L., Casaulta C., Kieninger E., Jung A., Moeller A., Geiser T., Regamey N., Alves M.P. Novel antiviral properties of azithromycin in cystic fibrosis airway epithelial cells. Eur. Respir. J. 2015;45:428–439. doi: 10.1183/09031936.00102014. [DOI] [PubMed] [Google Scholar]
- Skalny A.V., Rink L., Ajsuvakova O.P., Aschner M., Gritsenko V.A., Alekseenko S.I., Svistunov A.A., Petrakis D., Spandidos D.A., Aaseth J., Tsatsakis A., Tinkov A.A. Zinc and respiratory tract infections: perspectives for COVID19. Int. J. Mol. Med. 2020;46:17–26. doi: 10.3892/ijmm.2020.4575. (Review) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan D.J., Jr., Gluzman I.Y., Russell D.G., Goldberg D.E. On the molecular mechanism of chloroquine's antimalarial action. Proc. Natl. Acad. Sci. U. S. A. 1996;93:11865–11870. doi: 10.1073/pnas.93.21.11865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Z., El Omari K., Sun X., Ilca S.L., Kotecha A., Stuart D.I., Poranen M.M., Huiskonen J.T. Double-stranded RNA virus outer shell assembly by bona fide domain-swapping. Nat. Commun. 2017;8:14814. doi: 10.1038/ncomms14814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi T., Suzuki T. Low-pH stability of influenza A virus sialidase contributing to virus replication and pandemic. Biol. Pharm. Bull. 2015;38:817–826. doi: 10.1248/bpb.b15-00120. [DOI] [PubMed] [Google Scholar]
- Tanaka Y., Sato Y., Sasaki T. Suppression of coronavirus replication by cyclophilin inhibitors. Viruses. 2013;5:1250–1260. doi: 10.3390/v5051250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- te Velthuis A.J., van den Worm S.H., Sims A.C., Baric R.S., Snijder E.J., van Hemert M.J. Zn(2+) inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture. PLoS Pathog. 2010;6 doi: 10.1371/journal.ppat.1001176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson A.J., Lummis S.C. Antimalarial drugs inhibit human 5-HT(3) and GABA(A) but not GABA(C) receptors. Br. J. Pharmacol. 2008;153:1686–1696. doi: 10.1038/bjp.2008.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Touret F., de Lamballerie X. Of chloroquine and COVID-19. Antivir. Res. 2020;177:104762. doi: 10.1016/j.antiviral.2020.104762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tripathy S., Dassarma B., Roy S., Chabalala H., Matsabisa M.G. A review on possible modes of action of chloroquine/hydroxychloroquine: repurposing against SAR-CoV-2 (COVID-19) pandemic. Int. J. Antimicrob. Agents. 2020:106028. doi: 10.1016/j.ijantimicag.2020.106028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Broek M.P.H., Mohlmann J.E., Abeln B.G.S., Liebregts M., van Dijk V.F., van de Garde E.M.W. Chloroquine-induced QTc prolongation in COVID-19 patients. Neth. Heart J. 2020 doi: 10.1007/s12471-020-01429-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vincent M.J., Bergeron E., Benjannet S., Erickson B.R., Rollin P.E., Ksiazek T.G., Seidah N.G., Nichol S.T. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol. J. 2005;2:69. doi: 10.1186/1743-422X-2-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang M., Cao R., Zhang L., Yang X., Liu J., Xu M., Shi Z., Hu Z., Zhong W., Xiao G. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269–271. doi: 10.1038/s41422-020-0282-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warhurst D.C., Craig J.C., Adagu I.S., Guy R.K., Madrid P.B., Fivelman Q.L. Activity of piperaquine and other 4-aminoquinoline antiplasmodial drugs against chloroquine-sensitive and resistant blood-stages of Plasmodium falciparum. Role of beta-haematin inhibition and drug concentration in vacuolar water- and lipid-phases. Biochem. Pharmacol. 2007;73:1910–1926. doi: 10.1016/j.bcp.2007.03.011. [DOI] [PubMed] [Google Scholar]
- Wenzel D.G., Kleoppel J.W. Arrhythmias of cultured heart cells and their reversal by antiarrhythmic and antimalarial drugs. Pharmacol. Res. Commun. 1980;12:863–871. doi: 10.1016/s0031-6989(80)80049-x. [DOI] [PubMed] [Google Scholar]
- White N.J. Cardiotoxicity of antimalarial drugs. Lancet Infect. Dis. 2007;7:549–558. doi: 10.1016/S1473-3099(07)70187-1. [DOI] [PubMed] [Google Scholar]
- Wiersinga W.J., Rhodes A., Cheng A.C., Peacock S.J., Prescott H.C. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19) JAMA. 2020 doi: 10.1001/jama.2020.12839. [DOI] [PubMed] [Google Scholar]
- Wu Y.F., Zhao P., Luo X., Xu J.C., Xue L., Zhou Q., Xiong M., Shen J., Peng Y.B., Yu M.F., Chen W., Ma L., Liu Q.H. Chloroquine inhibits Ca(2+) permeable ion channels-mediated Ca(2+) signaling in primary B lymphocytes. Cell Biosci. 2017;7:28. doi: 10.1186/s13578-017-0155-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong T.Y., Redwood S., Prendergast B., Chen M. Coronaviruses and the cardiovascular system: acute and long-term implications. Eur. Heart J. 2020 doi: 10.1093/eurheartj/ehaa231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J.C., Peng Y.B., Wei M.Y., Wu Y.F., Guo D., Qin G., Ji G., Shen J., Liu Q.H. Chloroquine inhibits Ca(2+) signaling in murine CD4(+) Thymocytes. Cell. Physiol. Biochem. 2015;36:133–140. doi: 10.1159/000374058. [DOI] [PubMed] [Google Scholar]
- Xue J., Moyer A., Peng B., Wu J., Hannafon B.N., Ding W.Q. Chloroquine is a zinc ionophore. PloS One. 2014;9 doi: 10.1371/journal.pone.0109180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan Y., Zou Z., Sun Y., Li X., Xu K.F., Wei Y., Jin N., Jiang C. Anti-malaria drug chloroquine is highly effective in treating avian influenza A H5N1 virus infection in an animal model. Cell Res. 2013;23:300–302. doi: 10.1038/cr.2012.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X., Yu Y., Xu J., Shu H., Xia J., Liu H., Wu Y., Zhang L., Yu Z., Fang M., Yu T., Wang Y., Pan S., Zou X., Yuan S., Shang Y. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir. Med. 2020;8:7. doi: 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu B., Li C., Chen P., Zhou N., Wang L., Li J., Jiang H., Wang D.W. Low dose of hydroxychloroquine reduces fatality of critically ill patients with COVID-19. Sci. China Life Sci. 2020 doi: 10.1007/s11427-020-1732-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L., Liu C., Xiao Y., Chen X. Oxethazaine inhibits hepatitis B virus capsid assembly by blocking the cytosolic calcium-signalling pathway. J. Gen. Virol. 2016;97:1198–1209. doi: 10.1099/jgv.0.000417. [DOI] [PubMed] [Google Scholar]
- Zhang H., Penninger J.M., Li Y., Zhong N., Slutsky A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46:586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y., Frey T.K., Yang J.J. Viral calciomics: interplays between Ca2+ and virus. Cell Calcium. 2009;46:1–17. doi: 10.1016/j.ceca.2009.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu C., Lei W., Huang J. Azithromycin inhibits double-stranded RNA-induced thymic stromal lymphopoietin release from human airway epithelial cells. Pharmazie. 2013;68:899–903. [PubMed] [Google Scholar]
- Zhu L., Huang L., Zhu Y., Ding X., Zhu G. Calcium signaling involved in bovine herpesvirus 1 replication in MDBK cells. Acta Virol. 2017;61:487–491. doi: 10.4149/av_2017_412. [DOI] [PubMed] [Google Scholar]
- Zou L., Xue Y., Jones M., Heinbockel T., Ying M., Zhan X. The effects of quinine on neurophysiological properties of dopaminergic neurons. Neurotox. Res. 2018;34:62–73. doi: 10.1007/s12640-017-9855-1. [DOI] [PubMed] [Google Scholar]
- Zumla A., Chan J.F., Azhar E.I., Hui D.S., Yuen K.Y. Coronaviruses - drug discovery and therapeutic options. Nat. Rev. Drug Discov. 2016;15:327–347. doi: 10.1038/nrd.2015.37. [DOI] [PMC free article] [PubMed] [Google Scholar]