Abstract
Therapeutic plasma exchange is a blood purification technique designed for the removal of large molecular weight toxins such as pathogenic antibodies and lipoproteins. Plasma exchange can be performed either by membrane separation or centrifugation. Centrifugal plasma exchange is more common in the United States, while membrane separation is more popular in Germany and Japan. The membrane separation technique is similar to the ultrafiltration procedures performed with a standard dialysis machine but in which the membrane’s pores are large enough to allow removal of all circulating molecules while retaining the cellular components. The current availability of plasma separation membranes compatible with CRRT systems has dramatically increased the potential for almost all nephrologists to perform these treatments. This review describes the membrane separation techniques available in the United States, the practical aspects of ordering and operating a membrane separation plasma exchange procedure, and its possible complications.
Keywords: clinical nephrology, Plasmapheresis, Plasma Exchange, ultrafiltration, Nephrologists, Molecular Weight, Centrifugation, Kidneys, Artificial, Antibodies, Lipoproteins, renal dialysis
Introduction
Therapeutic plasma exchange is a procedure carried out for various life-threatening and debilitating diseases as a principal mode of treatment or as an adjunct with other therapies. In this procedure, plasma is separated from the cellular components of blood. The removed plasma is discarded and replaced with a colloid, a combination of crystalloid and colloid (1), or fresh frozen plasma. Separation of plasma from the cellular components of blood can be achieved by two different techniques: either by membrane separation or by centrifugation. Plasma separation by centrifugation is commonly performed by pathologists, transfusion medicine experts, and nephrologists. Alternatively, the concept of membrane separation of plasma using a filtration device is very similar to hemofiltration or ultrafiltration using a dialysis machine and, hence, mainly performed by nephrologists. Membrane filtration plasma exchange is also referred to as membrane plasma separation. Although very popular in Japan and Germany (2), in the United States, the membrane separation technique, although gaining popularity (3), is used less often compared with centrifugal plasma exchange. Nephrologists and dialysis nurses are naturally familiar with this technique because they are experts in using these machines. This review article will discuss the basic principles, mechanisms, instruments, complications, and practical aspects of prescribing therapeutic plasma exchange by membrane separation.
Principles of Blood Purification by Membrane Separation Plasma Exchange
The separation and removal of plasma from the patient by a semipermeable membrane and its simultaneous replacement by an equal volume of appropriate fluid purify the blood by removing the pathogenic molecules responsible for certain diseases. The substances removed by plasma exchange are abnormally produced in the body in various diseased states. These high molecular weight pathogenic molecules, such as Igs, proteins, or immune complexes (Table 1) (4,5), cannot be removed from the blood by conventional methods, such as dialysis or hemofiltration, due to their large size. Urgent removal of these molecules is necessary to prevent ongoing organ damage because these molecules have long half-lives; additionally, therapies aimed at suppression of their production take weeks or months to be effective. Rapid removal of these large molecules by plasma exchange reduces morbidity and mortality. The commonly removed molecules and their molecular weights are described in Table 1 (4,5).
Table 1.
Size and distribution of some of the proteins removed by therapeutic plasma exchange
| Protein | Concentration, mg/ml | M.W. × 103 D | Percentage Intravascular |
|---|---|---|---|
| IgG (except IgG3 subclass) | 12 | 150 | 45 |
| IgG3 | 0.7 | 150 | 64 |
| IgMa | 0.9 | 950 | 78 |
| IgA | 2.5 | 160 | 42 |
| IgD | 0.02 | 175 | 75 |
| IgE | 0.0001 | 190 | 45 |
| Albumin | 45 | 66 | 44 |
| C3 | 1.4 | 240 | 67 |
| C4 | 0.5 | 200 | 66 |
| Fibrinogen | 3–4 | 340 | 81 |
| Factor VIII | 0.1 | 100–340 | 71 |
| Antithrombin III | 0.2 | 56–58 | 45 |
| Lipoprotein cholesterol | 1.5–2.0 | 1300 | >90 |
Mechanism of Plasma Separation by Membrane Filtration Plasma Exchange
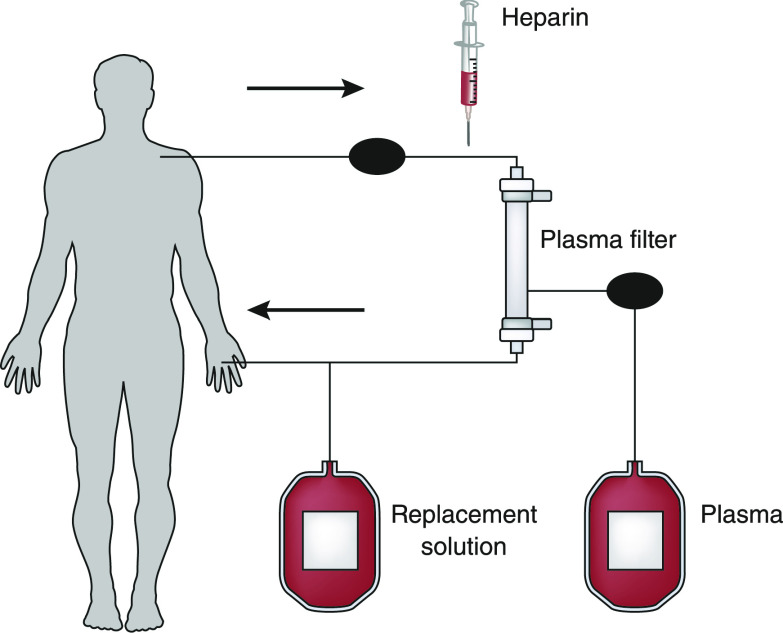
Separation of plasma from the blood’s cellular components can be accomplished by filtration through a highly permeable membrane. This methodology separates the blood into its cellular and noncellular components by subjecting it to sieving through a membrane with pores that allow the plasma proteins to pass but retain the larger cellular elements within the blood path (Figure 1). Twenty-five years after the first centrifugal plasma exchange procedure in a patient with multiple myeloma in 1952, the membrane separation technique was introduced in 1978 (6,7) as an alternative to the existing centrifugal technique. Membrane separation plasma exchange is very similar to the operation of a conventional dialysis machine running in ultrafiltration mode (Figure 2) or a continuous RRT (CRRT) in venovenous hemofiltration mode. In membrane separation plasma exchange, instead of a regular filter designed for the removal of small and “middle” molecules, a special filter with large pores (0.3–0.5 µm) is used. Pore sizes of these membranes are usually 0.5 µm or less, thus easily rejecting the smallest cellular component, the platelets (3 µm). Blood drawn from a central venous access, such as a dialysis catheter, is passed through this special plasma filter before it is returned to the patient (Figure 2). Because of the large pores, the ultrafiltrate or effluent produced in this manner contains all of the noncellular components of the patient’s plasma (Figure 1); as plasma is filtered out, it is replaced simultaneously with appropriate fluids (usually albumin or fresh frozen plasma [FFP]) in equal volume to the plasma removed to prevent hemodynamic compromise. The removal rate of any given plasma component depends on the plasma filtration rate and its sieving coefficient (ratio of a given plasma protein or solute concentration between the filtrate and the blood side of the membrane). With the currently available modern plasma filtration membranes, the sieving coefficient of even the largest molecules, weighing >1 million D (LDL cholesterol, etc.), is essentially “one.” Thus, the concentration of these larger proteins in the removed plasma (effluent) is essentially equal to that in the patient’s circulating plasma.
Figure 1.
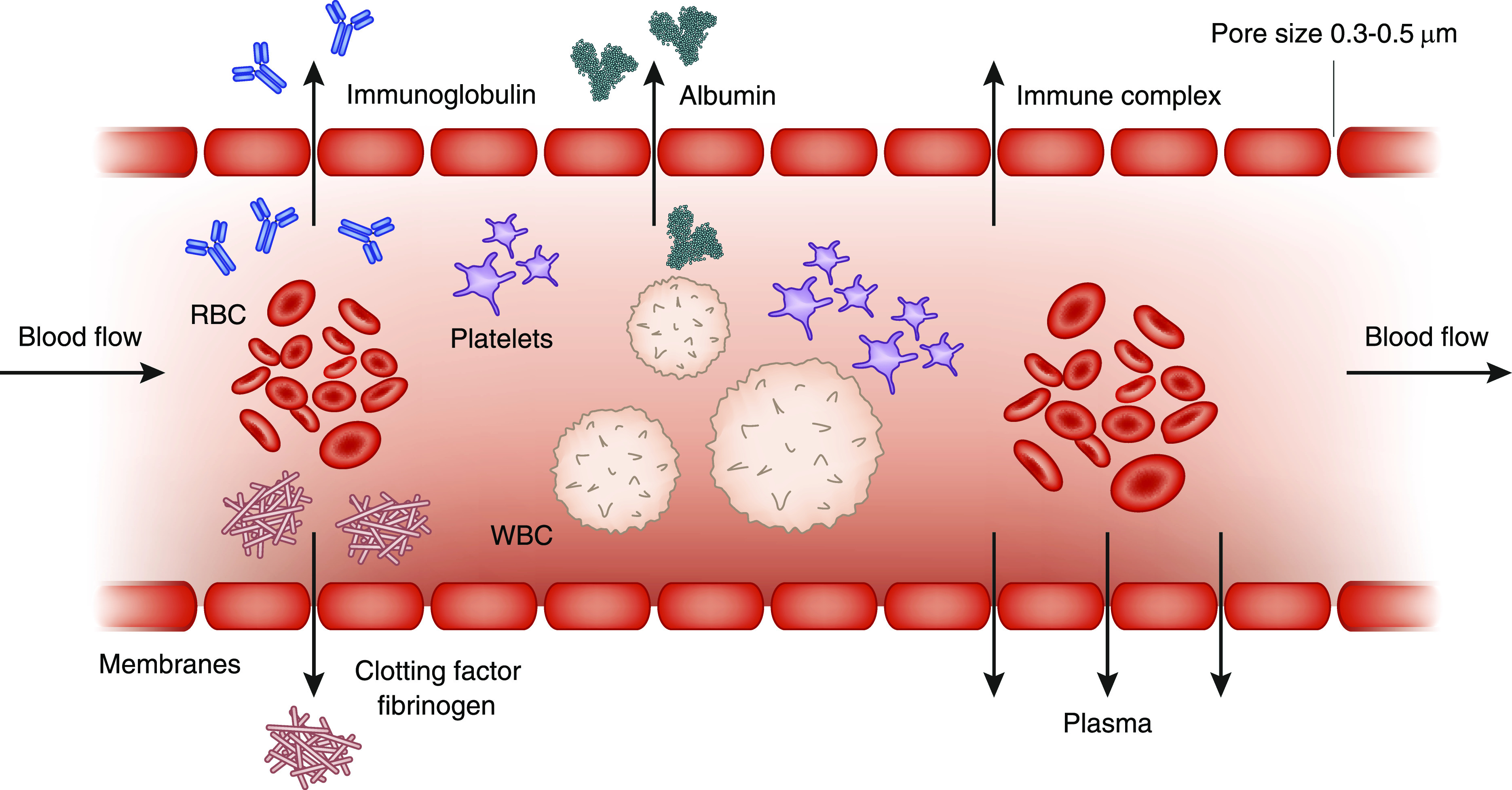
Schematic section of a hollow fiber for plasma exchange. Whole blood flows lengthwise along the interior of the fiber, whereas its plasma components, such as Igs, clotting factors, fibrinogen, and albumin, pass through the pores in the fiber wall and collect outside the fiber. The wall of the hollow fiber functions as the separating membrane with a pore size that allows penetration by plasma but not by the blood’s cellular components (red blood cells [RBCs], white blood cells [WBCs], and platelets).
Figure 2.
Blood flowing through the plasma filter, plasma separation and infusion of replacement solution, and return to the patient.
Membrane Separation Plasma Exchange Options Available in the United States
The flat parallel plate membrane (Centry TPE; Cobe) (8) was the first membrane separation plasma exchange filter approved in the United States by the Food and Drug Administration in 1982, and it was found to be equally effective in removing plasma as centrifugal exchange (6). Eventually, more advanced hollow fiber membranes evolved and replaced the parallel plates. Over the years, different types of plasma filters were marketed in the United States, but most of those were discontinued. Worldwide, there is a myriad of filters available for this purpose, but most are not available in the United States (2,8,9). Practitioners in the United States have only two choices of filters for membrane separation plasma exchange. They are the Prismaflex TPE-2000 plasma filter (Baxter) and the Plasmaflo OP-05W (Asahi Kasei Medical Company Ltd. Japan). The physical properties of these two filters are very similar but different from a conventional dialyzer, and they are compared in Table 2.
Table 2.
Comparison of available membrane separation therapeutic plasma exchange filters in the United States with a dialyzer for hemodialysis
| Specification | TPE 2000 Filter | Asahi Plasmaflo Filter | F200NR Dialyzer |
|---|---|---|---|
| Indication for use | Plasma exchange | Plasma exchange | Hemodialysis |
| Molecular mass cutoff, D | 3 million | NA but estimated close to 3 million | Estimated 15,000 |
| Pore size, µm | 0.5 | 0.3 | NA |
| Fiber material | Polypropylene | Polyethylene | Polysulfone |
| Hollow fibers | Yes | Yes | Yes |
| Surface area, m2 | 0.35 | 0.5 | 2 |
| Blood volume in filter, ml | 55 | 41 | 113 |
| TMP, mm Hg | 120–193 | 100 | 600 (maximum) |
| Anticoagulation | Heparin (citrate rare) | Heparin (citrate rare) | Heparin |
| Blood flow rate, ml/min | 100–250 | Up to 200 | Up to 600 |
| Sieving coefficient | |||
| Albumin | 0.97 | 0.99 | 0 |
| IgG | 1 | 1 | 0 |
| IgA | 1 | 1 | 0 |
| IgM | 0.92 | 1 | 0 |
| Sterilization | Ethylene oxide | γ-Ray | Ethylene oxide |
NA, not available; TMP, transmembrane pressure.
Efficacy of Membrane Separation in Comparison with Centrifugal Plasma Exchange
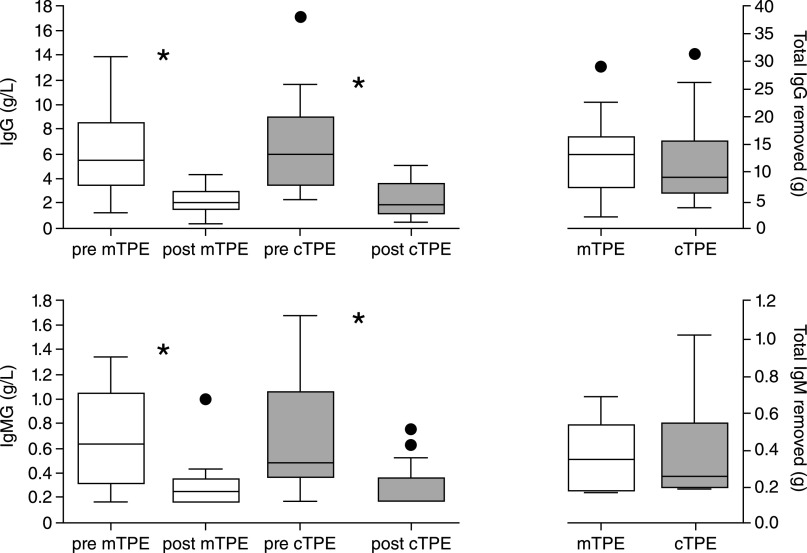
Membrane separation is limited to plasma exchange only and cannot separate the cellular components of blood. For this reason, it cannot be used for cellular apheresis procedures, such as red blood cell exchange, leukapheresis, platelet removal, and harvesting stem cells. The plasma exchanging efficiency of membrane separation is similar to the centrifugal technique due to a very high (close to 100%) sieving coefficient (ratio of the concentrations of the molecules between the filtrate and the blood) of the plasma proteins and the pathogenic molecules (10). Studies from the 1980s showed that flat-plate parallel membranes had similar efficacy to centrifugal plasma exchange (4). A recent prospective study compared membrane filtration by the Plasmaflo filter with the centrifugal separation by the Spectra Optia and found that both systems are equally effective in removing Igs (Figure 3) (11); however, the centrifugal procedure required less time (11,12). In a retrospective study, membrane separation was also found to be effective in the pediatric population (13).
Figure 3.
Comparison between efficacy of membrane separation therapeutic plasma exchange (mTPE) and centrifugation therapeutic plasma exchange (cTPE). IgG and IgM levels before and after showing similar removal with the two techniques. Reprinted from ref. 11, with permission.
Kinetics of Ig Removal with Plasma Exchange
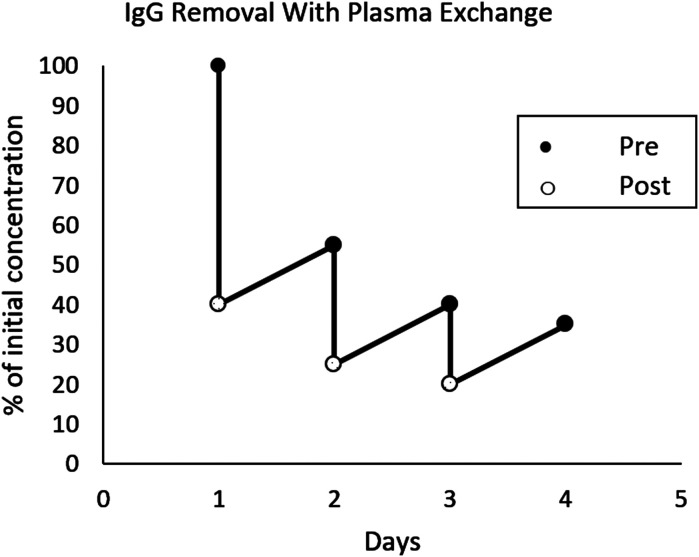
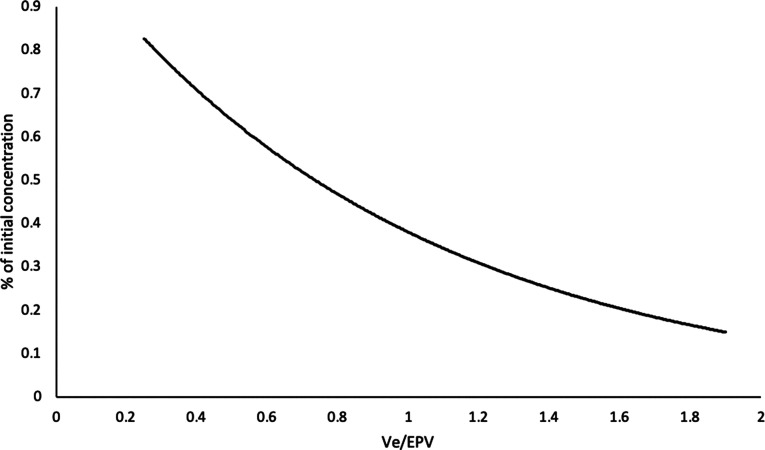
The volume of plasma exchanged determines how much of the pathogenic molecules is removed from the body. Removal of these molecules is independent of the separation technique used. The membrane separation procedure removes the pathogenic molecules from the intravascular space. The volume of distribution and the t1/2 of the molecules have a role in its removal. For example, IgM is largely (78%) (14) intravascular with a t1/2 of 5 days, and it can be removed effectively with a few daily treatments. However, IgG is <45% intravascular with a t1/2 of 21 days, and it needs more treatments with reasonable intervals for effective reduction of its level. Intervals of 24–48 hours after each treatment will allow the levels of the pathogenic target to increase due to new generation and equilibration between the extravascular and intravascular spaces (5) (Figure 4). Studies show that a plasma exchange equal to 1 estimated plasma volume will lower the pretreatment Ig levels approximately 63% and that a 1.4× estimated plasma volume exchange will lower the pretreatment levels by 75% (15) (Figure 5). Exchange of >1.5× estimated plasma volume during one single session provides only minimal additional removal of these large molecules but increases the treatment time and cost. In a prospective trial (16) on patients with acute liver failure, where antibody removal was not the focus, a higher volume of exchange was found to be effective, but the potential toxins involved with acute liver failure have a wide range of molecular weights; additionally, the plasma treatment was supplemented with high-volume CRRT, which can be very effective in removal of small and middle molecular weight toxins. Furthermore, in this study evaluating high-volume exchanges, the replacement fluid was FFP, and the higher exchange volumes allowed for the infusion of larger volumes of coagulopathy-correcting FFP without risk of fluid overload (17).
Figure 4.
Progressive decline in IgG levels after three consecutive plasma exchanges (1 plasma volume each). IgG increased between treatments due to the combination of extravascular to intravascular re-equilibration and new IgG synthesis. Reprinted from ref. 5, with permission.
Figure 5.
Relationship of plasma volume exchanged (Ve), estimated plasma volume (EPV), and percentage reduction in initial concentration for pathogenic molecules removed during therapeutic plasma exchange. Ve/EPV=1 (i.e., 1 plasma volume will remove 63% of the pretreatment concentration of the molecule). Ve/EPV=1.4, or 1.4 plasma volume will remove 75% of the substance. Reprinted from ref. 15, with permission.
Nephrology Consultations for Membrane Separation Plasma Exchange
There are indications for plasma exchange for various diseases, including but not limited to the kidneys. The nephrology service is consulted by other services, such as hematology-oncology, neurology, transplant medicine, and rheumatology, to perform plasma exchanges for various indications. The plasma exchange for nonkidney-related indications is planned together with the consulting services. This collaboration is essential for making sure that the indication for the procedure is valid, for placing appropriate vascular access, and for making decisions on the frequency and number of treatments for a particular patient. Over the last few years, the American Society for Apheresis has produced a detailed description and evaluation of the varied indications for plasma exchange. Their most recent set of guidelines was published in 2019 (18), and it is a valuable resource for nephrologists. The guideline is updated every 3 years and contains user-friendly individual fact sheets for each disease where therapeutic plasma exchange has been considered as a treatment option. The most recent review provides 84 fact sheets for 157 proposed indications (18). The fact sheet of a particular disease contains a brief review of pathogenesis, the rationale for plasma exchange, and a review of the relevant literature. The recommendations for plasma exchange for a particular disease are classified and graded. It also provides technical notes on the volume of plasma to be exchanged per treatment, the number and frequency of treatments, and the choice of replacement fluid for a particular indication.
Access for Membrane Separation Plasma Exchange
A temporary or tunneled dialysis catheter is inserted in a central vein for membrane separation because other smaller-caliber central lines or peripheral accesses used for centrifugal exchange with low-blood flow rates are not compatible with the hemodialysis or CRRT machines for membrane separation. A preexisting arteriovenous fistula or graft can be used for membrane separation, and there are cases where an arteriovenous fistula is created in certain diseases requiring long-term plasma exchanges. It should be noted, however, that the immediate removal of a large-bore central venous catheter can cause bleeding after an intensive run of plasma exchanges due to a substantial “depletion” coagulopathy (19,20).
Anticoagulation for Membrane Separation Plasma Exchange
Similar to hemodialysis treatments, unfractionated heparin is commonly used for systemic anticoagulation in membrane separation plasma exchange and given as a single bolus of 3000–5000 IU or 40–60 U/kg (21) followed by 1000 IU/h as needed. Citrate (anticoagulant citrate dextrose-A solution) is commonly used in centrifugal exchange for anticoagulation, but it is rarely used (22) in membrane separation due to higher risk of toxicity and hypocalcemia from increased citrate exposure because higher volumes of blood are processed in the membrane separation technique. Seventy to eighty percent of the citrate infused in membrane separation is retained by the patient due to reduced removal because plasma extraction ratio is only 20%–30% in membrane separation compared with 80% in centrifugal exchanges. Citrate toxicity is still possible in membrane separation even when heparin is used for anticoagulation if FFP (contains 14% citrate by volume) is used for replacement fluid in a patient with kidney or hepatic failure (23). Citrate-induced hypocalcemia can be avoided with prophylactic administration of calcium.
A recent report of nearly 1000 membrane separation plasma exchanges with the NxStage machine found that only 7% of treatments required anticoagulation (3).
Calculation of Plasma Volume
For the plasma exchange prescription, the patient’s plasma volume is estimated on the basis of the hematocrit and weight of the patient in kilograms. A formula to estimate plasma volume is (0.065× weight [kilograms]) × (1− hematocrit) (24). This formula is simple, quick, and reliable compared with other formulas (25–28). Plasma exchange volume is usually prescribed as 1–1.5× estimated plasma volume.
Prescribing Replacement Solutions
Replacement fluid is infused into the patient during plasma exchange to prevent hemodynamic collapse. The replacement fluid replaces the lost volume and exerts oncotic pressure to avoid hypotension. Replacement choices are 5% albumin or FFP; 5% albumin is isosmotic, and its oncotic pressure is close to plasma (29). Many centers use a combination of crystalloids and colloids, such as normal saline for 25%–30% of the exchange volume at the beginning followed by 5% albumin for the rest of the procedure. Some centers add 50 g of 25% albumin to a liter of normal saline or Ringers lactate to get a final concentration of 5% albumin for replacement solution. Increasing the saline infusion rate to 20% more than the plasma removal rate can compensate for third spacing of saline (30). Albumin derived from human plasma is highly purified with negligible risk of allergic reaction, but albumin replacement does not compensate for the depletion of clotting factors. FFP replacement is mandated in certain diseases, such as thrombotic thrombocytopenic purpura, to replace ADAMTS 13 (31) or if there is increased risk of bleeding because plasma exchange depletes the clotting factors. There is significant risk of anaphylactic reaction (20) with FFP, and this is the most serious complication of therapeutic plasma exchange, with uncommon reports of death due to these reactions (32–34). The patients getting FFP should be premedicated with intravenous diphenhydramine, and intravenous steroid and subcutaneous epinephrine should be kept ready at the bedside.
Machine Settings
In the United States, there are only two types of filters available for membrane separation plasma exchange, and they are used in three different settings as follows:
Prismaflex CRRT Machine with TPE-2000 Filter
The Prismaflex CRRT machines are used by simply changing the CRRT cartridge to the TPE 2000 cartridge. The tubing and the filter are preattached and come as a single kit to snap on to the Prismaflex machine. This filter cannot be used with any other machine. The standard blood flow rate for the procedure is usually 100–200 ml/min (machine range allows 100–250 ml/min). The rate of plasma replacement and loss can be set as high as 2000 ml/h (33 ml/min). The transmembrane pressure (TMP) is maintained at 100 mm Hg or less. A faster removal increases the filtration fraction and chance of clotting along with increase of TMP and higher risk of hemolysis. A 3- to 4-L exchange will take approximately 2 hours. The volume of plasma removal in this system is calculated by a weighing scale in the machine.
NxStage System One with Plasmaflo OP-05W Filters
The Plasmaflo OP-05W filter is used with the NxStage System One machine and can also be used with other hemodialysis machines. For the NxStage System One machine, the NxStage supplies a plasma exchange cartridge. The cartridge comes with a Plasmaflo OP-05W filter and a separate tubing set. The filter is manually attached to the tubing in the machine. The standard blood flow rate is 100–200 ml/min (machine range 10–200 ml/min). The plasma removal rate is also similar (i.e., up to 2000 ml/h; machine maximum is 4000 ml/h), with a TMP goal of 100 mm Hg or less. In this system, volume of plasma is not measured by the weighing scale but by the number of pulse strokes.
Hemodialysis Machine with Plasmaflo OP-05W Filter
The Plasmaflo OP-05W filter can also be used with a regular hemodialysis machine. The individual filters are sold by the Asahi Company. The filter is attached to a regular hemodialysis machine and connected to the blood pump, pressure monitoring system and the dialysis machine is put on “isolated” ultrafiltration mode, which bypasses the dialysate proportioning system. A dual-track PP O4A plasma pump (supplied by Asahi Kasei) is attached to the tubing to control the simultaneous plasma removal and replacement. Asahi stopped selling this pump in the United States market after 2017.
In all of the above membrane filtration procedures, circuit pressure, TMP, and blood flow rates are kept low (200 ml/min or lower) to avoid hemolysis.
Complications
Electrolyte disturbances, depletion coagulopathy, access-associated complications (20), and reaction to FFP (anaphylaxis, rigor, and hypotension) are some of the complications of this procedure. Use of the Plasmaflo OP-05W is associated with a potential for increased bradykinin levels, and it would be wise to discontinue angiotensin-converting enzyme inhibitors in any patient who is to be treated with this membrane (35). Symptomatic hypocalcemia is less likely in membrane separation plasma exchange because citrate is rarely used for anticoagulation, but it occurs more if FFP is used because it has citrate. Citrate in FFP can also lead to metabolic alkalosis with high-volume transfusion. Citrate toxicity is most likely to occur in a patient with hepatic or kidney failure (23) due to diminished citrate clearance. Hypomagnesemia and hypokalemia can occur due to bulk removal and citrate binding of magnesium because replacement fluid lacks these electrolytes. Muscle cramps can occur due to removal of plasma from the vascular space. Thrombocytopenia is less common but can occur in membrane separation plasma exchange, probably due to entrapment in the filter. Increased risk of bleeding due to removal of clotting factors and fibrinogen can occur with three or more exchanges per week with albumin infusion. To prevent this, the last 500 ml can be replaced with FFP instead of 5% albumin (36). Close monitoring of complete blood counts, coagulation status, and electrolytes (including magnesium and ionized calcium) are necessary for patients undergoing plasma exchanges. Administration of medications, such as IgG and therapeutic antibodies (rituximab, etc.), should be scheduled after the plasma exchange to avoid their removal.
Conclusions
Membrane-based plasma exchange technology is very similar to hemofiltration using an existing CRRT or dialysis machine. Considering that nephrologists are experts in the use of extracorporeal blood purification therapies, membrane-based plasma exchange can be performed by the nephrology service with minimal additional training, thus making this valuable technique available to a greater number of their patients.
Disclosures
All authors have nothing to disclose.
Funding
None.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.Adams WS, Blahd WH, Bassett SH: A method of human plasmapheresis. Proc Soc Exp Biol Med 80: 377–379, 1952 [DOI] [PubMed] [Google Scholar]
- 2.Córdoba JP, Larrarte C, Medina MC: Experience in therapeutic plasma exchange by membrane filtration at an academic center in Colombia: Registry of the first 500 sessions. J Clin Apher 30: 347–352, 2015 [DOI] [PubMed] [Google Scholar]
- 3.Gashti CN, Andreoli DC, Patel D: Membrane-based therapeutic plasma exchange (mTPE): Technical and clinical experience. J Clin Apher 33: 38–45, 2018 [DOI] [PubMed] [Google Scholar]
- 4.Kaplan AA: Therapeutic plasma exchange: A technical and operational review. J Clin Apher 28: 3–10, 2013 [DOI] [PubMed] [Google Scholar]
- 5.Kaplan AA: A Practical Guide to Therapeutic Plasma Exchange, Malden, MA, Blackwell Science, 1999 [Google Scholar]
- 6.Solomon BA, Castino F, Lysaght MJ, Colton CK, Friedman LI: Continuous flow membrane filtration of plasma from whole blood. Trans Am Soc Artif Intern Organs 24: 21–26, 1978 [PubMed] [Google Scholar]
- 7.Grossman L, Benny WB, Buchanan J, Erickson RR, Buffaloe GW: Clinical evaluation of a flat-plate membrane plasma exchange system. J Clin Apher 1: 225–237, 1983 [DOI] [PubMed] [Google Scholar]
- 8.Sawada K, Malchesky PS, Nose Y: Available removal systems: State of the art. In: Therapeutic Hemapheresis in the 1990s: Current Studies in Hematology and Blood Transfusion, Vol. 57, edited by Nydegger UE, Basel, Switzerland, Karger, 1990, pp 51–113 [DOI] [PubMed] [Google Scholar]
- 9.Gurland HJ, Lysaght MJ, Samtleben W, Schmidt B: Comparative evaluation of filters used in membrane plasmapheresis. Nephron 36: 173–182, 1984 [DOI] [PubMed] [Google Scholar]
- 10.Plasmaflo OP digital catalog: Available at: http://www.asahi-kasei.co.jp/medical/en/apheresis/product/plasma/op/performance.html. Accessed April 1, 2020
- 11.Hafer C, Golla P, Gericke M, Eden G, Beutel G, Schmidt JJ, Schmidt BM, De Reys S, Kielstein JT: Membrane versus centrifuge-based therapeutic plasma exchange: A randomized prospective crossover study. Int Urol Nephrol 48: 133–138, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Schmidt JJ, Asper F, Einecke G, Eden G, Hafer C, Kielstein JT: Therapeutic plasma exchange in a tertiary care center: 185 Patients undergoing 912 treatments—A one-year retrospective analysis. BMC Nephrol 19: 12, 2018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Reddy SK, Jahan A, Chaturvedi S, Agarwal I: Plasma exchange for paediatric kidney disease-indications and outcomes: A single-centre experience. Clin Kidney J 8: 702–707, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Okafor C, Ward DM, Mokrzycki MH, Weinstein R, Clark P, Balogun RA: Introduction and overview of therapeutic apheresis. J Clin Apher 25: 240–249, 2010 [DOI] [PubMed] [Google Scholar]
- 15.Kaplan AA: Toward the rational prescription of therapeutic plasma exchange: The kinetics of immunoglobulin removal. Semin Dial 5: 227–229, 2007 [Google Scholar]
- 16.Larsen FS, Schmidt LE, Bernsmeier C, Rasmussen A, Isoniemi H, Patel VC, Triantafyllou E, Bernal W, Auzinger G, Shawcross D, Eefsen M, Bjerring PN, Clemmesen JO, Hockerstedt K, Frederiksen HJ, Hansen BA, Antoniades CG, Wendon J: High-volume plasma exchange in patients with acute liver failure: An open randomised controlled trial. J Hepatol 64: 69–78, 2016 [DOI] [PubMed] [Google Scholar]
- 17.Kaplan AA: Moderator’s view: High-volume plasma exchange: Pro, con and consensus. Nephrol Dial Transplant 32: 1464–1467, 2017 [DOI] [PubMed] [Google Scholar]
- 18.Padmanabhan A, Connelly-Smith L, Aqui N, Balogun RA, Klingel R, Meyer E, Pham HP, Schneiderman J, Witt V, Wu Y, Zantek ND, Dunbar NM, Schwartz GEJ: Guidelines on the use of therapeutic apheresis in clinical practice - evidence-based approach from the writing committee of the American Society for Apheresis: The eighth special issue. J Clin Apher 34: 171–354, 2019 [DOI] [PubMed] [Google Scholar]
- 19.Price CA: Therapeutic plasma exchange in a dialysis unit. ANNA J 14: 103–108, 1987 [PubMed] [Google Scholar]
- 20.Mokrzycki MH, Kaplan AA: Therapeutic plasma exchange: Complications and management. Am J Kidney Dis 23: 817–827, 1994 [DOI] [PubMed] [Google Scholar]
- 21.Gerhardt RE, Ntoso KA, Koethe JD, Lodge S, Wolf CJ: Acute plasma separation with hemodialysis equipment. J Am Soc Nephrol 2: 1455–1458, 1992 [DOI] [PubMed] [Google Scholar]
- 22.Ilamathi E, Kirsch M, Moore B, Finger M: Citrate anticoagulation during plasmapheresis using standard hemodialysis equipment. Semin Dial 6: 268, 2007 [Google Scholar]
- 23.Pearl RG, Rosenthal MH: Metabolic alkalosis due to plasmapheresis. Am J Med 79: 391–393, 1985 [DOI] [PubMed] [Google Scholar]
- 24.Kaplan AA: A simple and accurate method for prescribing plasma exchange. ASAIO Trans 36: M597–M599, 1990 [PubMed] [Google Scholar]
- 25.Inkley SR, Brooks L, Krieger H: A study of methods for the prediction of plasma volume. J Lab Clin Med 45: 841–850, 1955 [PubMed] [Google Scholar]
- 26.Retzlaff JA, Tauxe WN, Kiely JM, Stroebel CF: Erythrocyte volume, plasma volume, and lean body mass in adult men and women. Blood 33: 649–667, 1969 [PubMed] [Google Scholar]
- 27.Feldschuh J, Enson Y: Prediction of the normal blood volume. Relation of blood volume to body habitus. Circulation 56: 605–612, 1977 [DOI] [PubMed] [Google Scholar]
- 28.Sprenger KBG, Huber K, Kratz W, Henze E, Franz H-E: Prediction of patient’s plasma volume in plasma exchange therapy. In: Immune and Metabolic Aspects of Therapeutic Blood Purification Systems, edited by Smeby LC, Jørstad S, Widerøe T-E, Basel, Switzerland, Karger, 1986, pp 394–402 [Google Scholar]
- 29.Lasky L, Finnerty E, Genis L, Polesky H: Protein and colloid osmotic pressure changes with albumin and/or saline replacement during plasma exchange. Transfusion 24: 256–259, 1984 [DOI] [PubMed] [Google Scholar]
- 30.Ward DM: Therapeutic plasmapheresis and related apheresis techniques. In: Updates to Harrison’s Principles of Internal Medicine, Vol. 5, edited by Petersdorf R, Adams R, Braunwald E, Isselbacher K, Martin J, Wilson J, Philadelphia, Williams and Wilkins, 1984, pp 67–95 [Google Scholar]
- 31.Rock GA, Shumak KH, Buskard NA, Blanchette VS, Kelton JG, Nair RC, Spasoff RA: Comparison of plasma exchange with plasma infusion in the treatment of thrombotic thrombocytopenic purpura. Canadian Apheresis Study Group. N Engl J Med 325: 393–397, 1991 [DOI] [PubMed] [Google Scholar]
- 32.Huestis DW: Mortality in therapeutic haemapheresis. Lancet 1: 1043, 1983 [DOI] [PubMed] [Google Scholar]
- 33.Aufeuvre JP, Mortin-Hertel F, Lefloch A, Baudelot J: Clinical tolerance and hazards of plasma exchanges: A study of 6200 plasma exchanges in 1033 patients. In: Plasmapheresis in Immunology and Oncology, Vol. 10, edited by Beyer JH, Borberg H, Fuchs C, Basel, Switzerland, Karger, 1982, pp 65–77 [Google Scholar]
- 34.Sutton D, Nair R, Rock G: Complications of plasma exchange. Transfusion 29: 124–127, 1989 [DOI] [PubMed] [Google Scholar]
- 35.Nagano T, Nakazono K, Iwamoto H, Sato T, Shizume Y, Takenaka Y, Munekata K, Koga N: Comparative study of polyethylene plasma separator sterilized by ethylene oxide and gamma rays. Japan J Apheresis 16: 297, 1997 [Google Scholar]
- 36.Ward DM: Conventional apheresis therapies: A review. J Clin Apher 26: 230–238, 2011 [DOI] [PubMed] [Google Scholar]