Abstract
Is it possible to develop a reliable, safe treatment for the widespread COVID-19 pandemic shortly? COVID-19 is characterized by a disruptive cytokine storm, quickly and often irreversibly damaging the patient’s lungs, as its main target organ, leading to lung failure and death. Actual experimental therapies are trying to reduce the activation of some specific cytokines, such as IL-6, somewhat reducing the burden for the patient. However, they are often unable to block the whole storm occurring at the cytokine level. In presence of the cytokine storm, especially in severe patients, antagomiRs, already demonstrated to be efficient and secure in cardiovascular disease, could represent a useful alternative to such treatment, customizable upon the disease specificities and applicable to other coronaviruses possibly associated with such clinical manifestations, while a reliable, efficient vaccine is being distributed.
Keywords: antagomiRs, ARDS, Coronavirus, COVID-19, Cytokine storm, microRNAs
1. Introduction
COVID-19, the novel coronavirus spreading worldwide since January 2020, accounts for more than 14 million cases in the world, with nearly 600,000 deaths as of July, 19th, 2020 (https://www.who.int/docs/default-source/coronaviruse/situation-reports/20,200,719-covid-19-sitrep-181.pdf?sfvrsn = 82352496_2). Until specific treatments are developed, strategies for quickly challenging the syndrome’s main symptoms are needed to reduce as much as possible the number of deaths and severe cases, as well as the consequences for public health and the health system in general.
From a clinical point of view, COVID-19, like other coronaviruses, including SARS and MERS, features severe acute respiratory problems, fast driving to pulmonary edema, and, to a larger extent, lung failure [1].
Mehta and colleagues’ [2] and other works highlighted that these symptoms are largely associated with a so-called “cytokine storm”, already displayed in other viruses, including H5N1 influenza, and associated with excessive levels of pro-inflammatory cytokines and widespread tissue damage. Such event features extremely high serum levels of several compounds, including IL-1β, IL-2, IL-6, IL-7, IL-8, IL-9, IL-10, IL-17, G-CSF, GMCSF, IFN-γ, TNF-α, IP10, MCP1, MIP1A, and MIP1B, some of which (IL-2, IL-7, IL-10, G-CSF, IP10, MCP1, MIP1A, and TNF-α) somewhat related to the severity of the condition, as being reported to be even higher in ICU patients [[1], [2], [3], [4]]. The cytokine storm leads to diffuse vascular injury, in turn causing thrombosis mainly in the lung, but also other target organs.
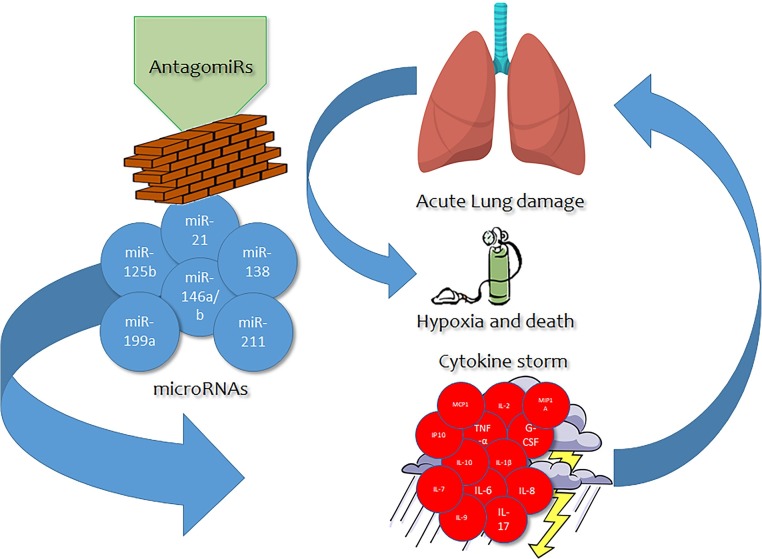
Indeed, one of the consequences of the extremely quick viral replication and strong pro-inflammatory response where cytokines play a major role is represented by the induction of apoptosis in lung endothelial and epithelial cells. As such, this event leads to damage at the level of the pulmonary microvascular and alveolar epithelial cell barriers, in turn driving to vascular leakage and alveolar edema. Taken together, such occurrence brings to significant hypoxia in the affected individual, that is a cornerstone for the survival of the patient itself given that Acute Respiratory Distress Syndrome (ARDS) is well recognized as the major cause of death in similar conditions, mainly including other coronaviruses (SARS-CoV and MERS-CoV) [5,6] (Fig. 1 ).
Fig. 1.
Possible action of antagomiRs blocking the miRNA-cytokine storm cascade related to acute lung damage.
Such lung acute inflammation and related damage processes, like those mediated by cytokines, occurring in COVID-19 patients, often feature a pivotal role for microRNAs, small (22 nucleotides on average) non-coding RNA molecules, present in plants, viruses, and animals, capable of modulating the expression of hundreds of genes in a synergistic or antagonistic fashion. Their role and their relationship with the cytokine storm occurring in COVID-19 could pave the way for novel, fast, effective treatment strategies alternative to biological approaches including antivirals or monoclonal antibodies.
2. Role for miRNA in respiratory syndromes
According to several works, some miRNAs are potential candidates for mediating this process in acute respiratory syndromes with inflammatory fingerprints.
Among them, miR-125b, well known for its involvement in carcinogenesis, was seen to be positively associated with the expression of TNF-α, IL-1β, and IL-8, cytokines found to be involved in the acute exacerbation of Chronic Obstructive Pulmonary Disease (COPD) [7] and among the main responsible for the cytokine storm occurring in the COVID-19.
Another evidence in favour of the association between miRNAs and inflammatory cytokines was seen for miR-21, an oncogene, profibrotic molecule that, at least in murine models, was seen to be associated with acute inflammatory processes at the lung level mediated by the classic “triad” of pro-inflammatory cytokines, IL-1β, IL-6, TNF-α [8,9]. Besides, it was also seen that its upregulation increases the expression of IL-8 and tumorigenesis in airway epithelial cells exposed to cigarette smoke [10].
The reduction of microRNA expression was already seen to be a possible way to relieve acute symptoms of lung inflammation and distress, as found in the case of the miR-138 downregulation and associated clinical improvement noticed after metformin treatment in mouse models of ARDS. In such a protocol, also the levels of TNF-α, IL-1β, IL-6, and IL-17 were significantly reduced after the treatment, confirming the manifold validity of the approach [11].
Under similar principles, also the specific downregulation of miR-199a brought to the inhibition of the upregulation of inflammatory cytokines (including TNF-α, IL-1β, IL-6) in alveolar macrophages and induced the remission of histopathologic changes, the reduction of proinflammatory cytokines, and the upregulation of apoptosis protein expression in an ARDS lung, in turn showing a role for miR-199a [12].
Finally, miR-146a and miR-146b are also involved. Indeed, in acute COPD, miR-146a was seen to be negatively associated with TNF- α, IL-6, IL-8, whereas miR-146b is negatively associated with IL-1β, possibly representing further compounds to be taken into account when promoting or inhibiting the activity of miRNAs [13].
3. Discussion: AntagomiRs as a novel therapeutic strategy for COVID-19 and future pandemics
In this framework, antagomiRs, a novel class of chemically engineered oligonucleotides, specific silencers of endogenous miRNAs, could act as the cornerstone of a more novel therapeutic approach than common treatment methods, featuring much higher customizability and a wider range of activity in terms of target compounds eventually silenced or downregulated at a larger extent. Such treatments could act with a good efficacy in those subjects presenting severe conditions due to the cytokine storm related to COVID-19 infection. AntagomiRs, possibly provided by systemic administration of polymer-based nanoparticles encapsulating the oligonucleotides [14,15], can be applied to specifically tailor the activity of key miRNAs involved in the inflammatory process, improving the clinical outcome of the patients affected [16].
In cardiovascular diseases, the use of antagomiRs was already demonstrated to provide optimal results within in-vivo and ex-vivo experiments. Indeed, several clinical trials demonstrated that such compounds could be considered highly safe and efficient in different conditions, with positive results obtained both in animals and human models, justifying their application in large-scale studies and clinical trials [17].
Indeed, the use of antagomiRs would not specifically inhibit the action of an only cytokine as occurring, for example, with tocilizumab, the immunosuppressor commonly used in Rheumatoid Arthritis selectively targeting the IL-6 receptor, therefore blocking a portion of the whole cytokine storm occurring in COVID-19 patients. AntagomiRs, easy to be produced in the laboratory, and cheaper than traditional biological approaches - especially when produced on a large scale - could be customized upon the immunological hallmarks of a given condition to block the activation of a range of compounds, effectively tailoring the whole cytokine storm, to prevent the patient from having serious, life-threatening complications associated to the COVID-19. Specifically, within the COVID-19 framework, such compounds should be designed to block the activity of one or more of the following cytokines, identified in previous literature works: IL-2, IL-4, IL-6, IL-7, IL-10, IP-10, MCP-1, MIP1A, G-CSF, IFN-γ, TNF-α (for a complete outline about their specific role in COVID-19, see [18]).
More importantly, the application of antagomiRs would not be limited to this novel coronavirus, actually representing a pandemic, according to the WHO, but can be extended to future pandemics, featuring cytokine storm. Indeed, the development and application of a set of antagomiRs, depending on the hallmarks of a given disorder, could potentially challenge the symptoms associated with such a virus. This could eventually bring to a decrease in the overall mortality caused by coronaviruses and lung inflammatory diseases and, to a much larger extent, to considerable savings for the national health services.
Declaration of Competing Interest
The authors report no declarations of interest.
Biographies

Sebastiano Gangemi was born in Messina, Italy, in 1960. MD, he obtained the PhD in Experimental Medicine in 2001. Actually, he is Directorof the Division of Allergy and Clinical Immunologyatthe School of Medicine,University of Messina, Italy, where he isalsoAssociate Professor of Medicine. His main researchover the years has covered the study of inflammatory mediators, the cytokine network (interleukins, chemokines, adhesion molecules, lipoxines) and the oxidative stress in various areas of clinical immunology, allergy and oncology. Actually, he is Section Editor (Clinical Immunology) of the journal Clinical and Molecular Allergy,and of the journal Current Molecular Medicine. He is author of more than 300 scientific articles, with H-index of 33 and around4200citations.

Alessandro Tonacci was born in Massa, Italy, in 1986. He is a Bioengineer,and obtained hisPhD in Automation, Robotics and Bioengineeringat the University of Pisa, School of Engineering. He is researcher at the Clinical Physiology Institute of the National Research Council of Italy (IFC-CNR). He is currently working on ICT solutionswithin the field of health and care, but is also involved in the study of biological mechanisms related to several diseases, including neurological ones, allergy, immune-mediated disorders. He is actually author of more than 80 scientific publications (h-index: 13, around 550 citations) andwithin the Editorial Board of several Journals, including Frontiers in Physiology and International Journal of Environmental Research and Public Health.
References
- 1.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. HLH Across Speciality Collaboration, UK, COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wu D., Yang X.O. TH17 responses in cytokine storm of COVID-19: an emerging target of JAK2 inhibitor Fedratinib. J. Microbiol. Immunol. Infect. 2020 doi: 10.1016/j.jmii.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Guo Y.R., Cao Q.D., Hong Z.S., Tan Y.Y., Chen S.D., Jin H.J., et al. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak - an update on the status. Mil. Med. Res. 2020;7:11. doi: 10.1186/s40779-020-00240-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Drosten C., Seilmaier M., Corman V.M., Hartmann W., Scheible G., Sack S. Clinical features and virological analysis of a case of Middle East respiratory syndrome coronavirus infection. Lancet Infect Diseases. 2013;13:745–751. doi: 10.1016/S1473-3099(13)70154-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lew T.W.K., Kwek T.K., Tai D., Earnest A., Loo S., Singh K. Acute respiratory distress syndrome in critically ill patients with severe acute respiratory syndrome. JAMA. 2003;290:374–380. doi: 10.1001/jama.290.3.374. [DOI] [PubMed] [Google Scholar]
- 7.Hu H.L., Nie Z.Q., Lu Y., Yang X., Song C., Chen H., et al. Circulating miR-125b but not miR-125a correlates with acute exacerbations of chronic obstructive pulmonary disease and the expressions of inflammatory cytokines. Medicine. 2017;96:e9059. doi: 10.1097/MD.0000000000009059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhu W.D., Xu J., Zhang M., Zhu T.M., Zhang Y.H., Sun K. MicroRNA-21 inhibits lipopolysaccharide-induced acute lung injury by targeting nuclear factor-κB. Exp. Ther. Med. 2018;16:4616–4622. doi: 10.3892/etm.2018.6789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jansing J.C., Fiedler J., Pich A., Viereck J., Thum T., Mühlfeld C., et al. miR-21-KO alleviates alveolar structural remodeling and inflammatory signaling in acute lung injury. Int. J. Mol. Sci. 2020;21 doi: 10.3390/ijms21030822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pace E., Di Vincenzo S., Di Salvo E., Genovese S., Dino P., Sangiorgi C., et al. MiR-21 upregulation increases IL-8 expression and tumorigenesis program in airway epithelial cells exposed to cigarette smoke. J. Cell. Physiol. 2019;234:22183–22194. doi: 10.1002/jcp.28786. [DOI] [PubMed] [Google Scholar]
- 11.Yu L.L., Zhu M., Huang Y., Zhao Y.M., Wen J.J., Yang X.J., et al. Metformin relieves acute respiratory distress syndrome by reducing miR-138 expression. Eur. Rev. Med. Pharmacol. Sci. 2018;22:5355–5363. doi: 10.26355/eurrev_201808_15737. [DOI] [PubMed] [Google Scholar]
- 12.Liu Y., Guan H., Zhang J.L., Zheng Z., Wang H.T., Tao K., et al. Acute downregulation of miR-199a attenuates sepsis-induced acute lung injury by targeting SIRT1. Am. J. Physiol., Cell Physiol. 2018;314:C449–C455. doi: 10.1152/ajpcell.00173.2017. [DOI] [PubMed] [Google Scholar]
- 13.Chen B.B., Li Z.H., Gao S. Circulating miR-146a/b correlates with inflammatory cytokines in COPD and could predict the risk of acute exacerbation COPD. Medicine. 2018;97:e9820. doi: 10.1097/MD.0000000000009820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Smith C.I.E., Zain R. Therapeutic Oligonucleotides: State of the Art. Annu. Rev. Pharmacol. Toxicol. 2019;59:605–630. doi: 10.1146/annurev-pharmtox-010818-021050. [DOI] [PubMed] [Google Scholar]
- 15.Malhotra M., Sekar T.V., Ananta J.S., Devulapally R., Afjei R., Babikir H.A., et al. Targeted nanoparticle delivery of therapeutic antisense microRNAs presensitizes glioblastoma cells to lower effective doses of temozolomide in vitro and in a mouse model. Oncotarget. 2018;9(30):21478–21494. doi: 10.18632/oncotarget.25135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Murdaca G., Tonacci A., Negrini S., Greco M., Borro M., Puppo F., et al. Effects of AntagomiRs on different lung diseases in human, cellular, and animal models. Int. J. Mol. Sci. 2019;20 doi: 10.3390/ijms20163938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Huang C.K., Kafert-Kasting S., Thum T. Preclinical and clinical development of noncoding RNA therapeutics for cardiovascular disease. Circ. Res. 2020;126:663–678. doi: 10.1161/CIRCRESAHA.119.315856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hu B., Huang S., Yin L. The cytokine storm and COVID-19. J. Med. Virol. 2020 doi: 10.1002/jmv.26232. [DOI] [PMC free article] [PubMed] [Google Scholar]