Abstract
Background
Osteoporosis is a metabolic bone disease. Bisphosphonate (BP) and eldecalcitol (ELD) are two clinical first-line drugs for osteoporosis patients. However, the effect of ELD + BP vs. BP alone on osteoporosis treatment is still unclear. The present meta-analysis was conducted to evaluate the different therapeutic effect of BP + ELD vs. BP alone in osteoporosis treatment.
Methods
Eligible documents that selected from online databases including PubMed, Embase, and Cochrane Library were included in this study (updated to March 3, 2020). The quality assessment of the included studies was based on the guidelines of Cochrane. Meta-analysis was performed according to criteria such as intervention plan and outcome. The indicators including bone mineral density (BMD) in all enrolled studies were included in the current analysis. Pooled odds ratios (ORs) and weighted mean differences (WMDs) with 95% confidence intervals (CI) were calculated using fixed- or random-effects models. Then, heterogeneity analysis was performed based on Cochran’s Q test and I2 statistics.
Results
A total of 4 studies (456 cases) with high quality were enrolled in this study. The effect of ELD + BP was superior to BP alone based on indicators including femoral neck BMD (FN-BMD) and total hip BMD (TH-BMD) in patients with followed up ≤ 6 months. Moreover, the effect of ELD + BP was superior to BP alone based on lumbar spine BMD (LS-BMD) in patients with 12 months followed up.
Conclusion
Therapeutic effect of ELD + BP was superior to BP alone in osteoporotic patients based on the influence of BMD.
Keywords: Osteoporosis, Bisphosphonate, Eldecalcitol, Therapeutic effect, Bone mineral density, Meta-analysis
Background
Osteoporosis is a group of bone diseases characterized by bone pain and easy fracture [1]. This disease lead to a total of 8.9 million fractures in 1 year all over the world [2]. Although the monitoring therapy based on bone mineral density (BMD) measurement contributes to the fracture diagnose, there is still a high mortality in people with osteoporosis due to the complications of fracture [3, 4]. Thus, effective drug prevention and treatment is necessary for people with osteoporosis or suffering osteoporotic fracture [5].
Bisphosphonate (BP) is one of the most commonly used first-line drugs for the clinical therapy of osteoporosis [6]. It contributes to the decrease of future fracture occurrence in patients with osteoporosis via change in the expression of blood mRNA in the process of osteoporosis [7, 8]. In Japan, a previous 2-year multicenter study shows that weekly used BP treatment significantly increases the quality of life osteoporosis patients [9]. On the contrary, some studies prove that BP not only is unsuitable for secondary osteoporosis treatment in children, but also do not appear to change the overall risk of death [10, 11]. Furthermore, eldecalcitol (ELD) is an active analog of vitamin D commonly used for the clinical treatment of osteoporosis [12]. A clinical study shows that ELD can reduce the re-absorption of blood calcium into the bone, improve the absorption of calcium in the intestine, and then further increase the bone density in osteoporosis patients [13]. Compared with other drugs such as alfacalcidol, ELD is more effective in preventing vertebral and wrist fractures in osteoporotic patients [14]. Actually, ELD + BP is useful for the osteoporotic patients who undergo the long-time BP therapy [15]. Increase in BMD by BP + ELD-treated osteoporotic patients is associated with the serum calcium level within the reference interval [16]. However, some studies indicated that compared with the BP-associated combination therapy, the BP treatment alone significantly increased the therapy effect in osteoporotic patients [17–19]. The clinical advantages of BP alone in the treatment of osteonecrosis compared with combined therapy have also been confirmed by multi-center studies [20]. To sum up, although previous studies prove the effect of BP alone or BP + ELD in clinical treatment of osteoporosis patients, it is still not clear which treatment strategy is better due to the limited sample size in each study. Therefore, it is necessary to explore more comprehensively and objectively the therapeutic effect of BP alone and BP + ELD on osteoporosis patients based on a meta-analysis.
In this study, the related articles which meet the certain inclusion criteria in Embase, PubMed, and Cochrane Library databases were searched. The indicators including BMD, blood calcium, blood phosphorus, and TRACP-5b in all enrolled RCT and non-RCT studies were included in the current analysis. Meta-analysis was performed according to criteria such as different indicators and outcome. The current study was aimed to evaluate the therapeutic effect of BP + ELD vs. BP in osteoporosis treatment.
Methods
Data sources
Relevant studies were searched from electronic databases including Embase, PubMed, and Cochrane Library (updated to March 3, 2020). The main searching keywords included the following: (“Eldecalcitol” OR “Bisphosphonate”) AND (“alendronate” OR “pamidronate” OR “ibandronate” OR “risedronate” OR “clodronate” OR “minodronate” OR “osteoporosis”). The detail information for search steps and corresponding results were shown in Table 1. Additionally, references of paper documents were hand searched for additional information on the procedures. No language restrictions were imposed in the current meta-analysis.
Table 1.
The detail information for search steps and corresponding results (retrieval time: March 3, 2020)
| Search | Query | Items found |
|---|---|---|
| #1 | “eldecalcitol”[Supplementary Concept] OR “eldecalcitol”[All Fields] | 177 |
| #2 | (“diphosphonates”[MeSH Terms] OR “diphosphonates”[All Fields] OR “bisphosphonate”[All Fields]) OR “diphosphonate”[All Fields]) OR “bisphosphonates”[All Fields]) OR Bisphosphate[All Fields] OR (“alendronate”[MeSH Terms] OR “alendronate”[All Fields]) OR (“pamidronate”[MeSH Terms] OR “pamidronate”[All Fields]) OR “zoledronic acid”[All Fields] OR (“ibandronic acid”[MeSH Terms] OR (“ibandronic”[All Fields] AND “acid”[All Fields]) OR “ibandronic acid”[All Fields] OR “ibandronate”[All Fields]) OR (“risedronic acid”[MeSH Terms] OR (“risedronic”[All Fields] AND “acid”[All Fields]) OR “risedronic acid”[All Fields] OR “risedronate”[All Fields]) OR (“clodronic acid”[MeSH Terms] OR (“clodronic”[All Fields] AND “acid”[All Fields]) OR “clodronic acid”[All Fields] OR “clodronate”[All Fields]) OR (“YM 529”[Supplementary Concept] OR “YM 529”[All Fields] OR “minodronate”[All Fields]) OR (“3-(2,2,2-trimethylhydrazine)propionate”[Supplementary Concept] OR “3-(2,2,2-trimethylhydrazine)propionate”[All Fields] OR “mildronate”[All Fields]) | 57,670 |
| #3 | “osteoporosis, postmenopausal”[MeSH Terms] OR (“osteoporosis”[All Fields] AND “postmenopausal”[All Fields]) OR “postmenopausal osteoporosis”[All Fields] OR “osteoporosis”[All Fields] OR “osteoporosis”[MeSH Terms] | 87,049 |
| #4 | #1 AND #2 AND 3 | 33 |
Inclusion and exclusion criteria
The inclusion criteria for articles were as follows: (i) the study subjects were patients with osteoporosis; (ii) the experimental group received ELD combined with BP treatment, and the control group was treated with BP alone (the types of BP (alendronate, ibandronate, risedronate, minodronate, etc.); (iii) the outcome of the study was based on indicators such as BMD (lumbar spine BMD (LS-BMD)), total hip BMD (TH-BMD), FN-BMD, blood calcium, blood phosphorus, and TRACP-5b; and (iv) research type was clinical research including RCT, non-RCT, and other clinical studies. The before-after studies were excluded from the analysis. If more than one study were published by the same sample, then only one study (the latest study or the completest study) was extracted. Moreover, the non-treatise literatures including reviews, letters, and comments were excluded.
Data extraction and quality assessment
A total of 2 independent researchers participate in the data extraction in current meta-analysis. The strategies including discussion and reexamination were used for the problems during the article research. The available data including name of first author, year of publication, country, type of research, age, gender, sample size, and intervention plan, as well as outcome information in each literature, were enrolled in this study. Cochrane was used for the quality assessment [21]. A panel discussion with a third member was used to deal with disputes arising from current data retrieval process.
Statistical analysis
The RevMan software (version: 5.3, Oxford, UK) was used for the statistical analysis in current study. Weighted mean difference (WMD) and 95% confidence interval (CI) were used as the effect size for the evaluation of continuous variable. The mean difference (MD) and standard deviation (SD) of each evaluation index before and after the intervention in the ELD + BP group and BP group were used as the effect values for difference comparison. Then, the meta-analysis was used to assess whether there was a significant difference in MD between the two groups.
Then, heterogeneity analysis was performed based on Cochran’s Q test and I2 statistics [22]. A random-effects model was used if heterogeneity was observed (P < 0.05 or I2 > 50%); otherwise, a fixed-effects model was applied. Furthermore, sensitivity analysis is used in the current research to analyze the impact of the combined outcomes after the literature was proposed one by one. Finally, the publication bias was evaluated by a funnel plot.
Results
Included studies
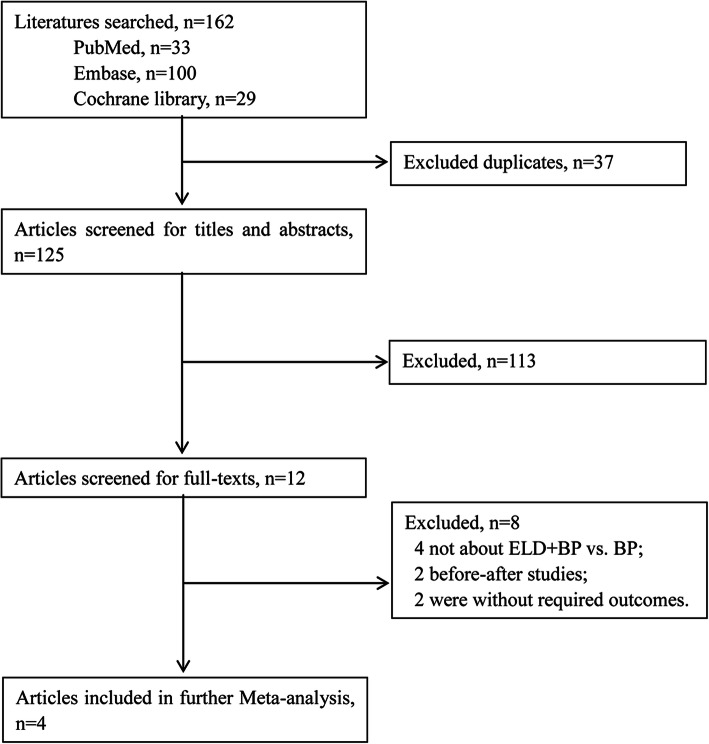
A total of 33, 100, and 29 studies in PubMed, Embase, and Cochrane library database, respectively, were included (Fig. 1). After deletion of the duplicate articles, a total of 125 studies were selected. After reading the title and abstract, a total of 113 articles were further excluded because these studies were not eligible for inclusion criteria. From the remaining 12 studies, 8 studies (4 studies without ELD + BP vs. BP; 2 before-after studies and 2 studies without required outcomes) were filtered out after reading the full text. Finally, totally, 4 studies with sufficient data including 2 RCT [23, 24] and 2 non-RCT [25, 26] were enrolled for the present meta-analysis (Table 2).
Fig. 1.
Flow diagram of screening process for eligible articles
Table 2.
Characteristics of 4 included studies in this meta-analysis
| Study | Area | Design | Duration | Criteria for osteoporosis | Duration | Group | Intervention | n | Age, years | Women, n (%) | BMI |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ebina et al. [28] | Japan | Non-RCT | NR | JSBMR | 12 months | BP + ELD | MIN 50 mg/month, ELD 0.75 μg/day | 80 | 71.8 ± 0.9 | 74 (92.5%) | 21.3 ± 0.6 |
| BP | MIN 50 mg/month | 63 | 71.7 ± 1.6 | 58 (92.1%) | 21.5 ± 0.6 | ||||||
| Iwamoto and Sato [23] | Japan | RCT | April 2012–July 2012 | JDC | 6 months | BP + ELD | ALE 35 mg weekly or risedronate 17.5 mg weekly, ELD 0.75 μg/day | 50 | 72.3 ± 8.3 | 50 (100%) | 22.5 ± 3.3 |
| BP | ALE 35 mg weekly or risedronate 17.5 mg weekly | 46 | 69.1 ± 9.9 | 46 (100%) | 22.7 ± 2.5 | ||||||
| Suzuki et al. [29] | Japan | RCT | May 2016–August 2017 | JSBMR | 18 months | BP + ELD | MIN 50 mg/month, ELD 0.75 μg/day | 14 | 68.7 ± 3.1 | 14 (100%) | 20.4 ± 0.7 |
| BP | MIN 50 mg/month | 14 | 65.8 ± 4.0 | 14 (100%) | 20.1 ± 0.5 | ||||||
| Takeuchi et al. [27] | Japan | Non-RCT | March 2014–January 2017 | JDC | 12 months | BP + ELD | Ibandronate 1 mg injections, ELD NR | 94 | NR | NR | NR |
| BP | Ibandronate 1 mg injections | 95 | NR | NR | NR |
ALE alendronate, MIN minodronate, ELD eldecalcitol, BMI body mass index, BP bisphosphonate, JDC Japanese diagnostic criteria, JSBMR Japanese Society for Bone and Mineral Research, NR not reported, RCT randomized controlled trial
Information for all enrolled literatures
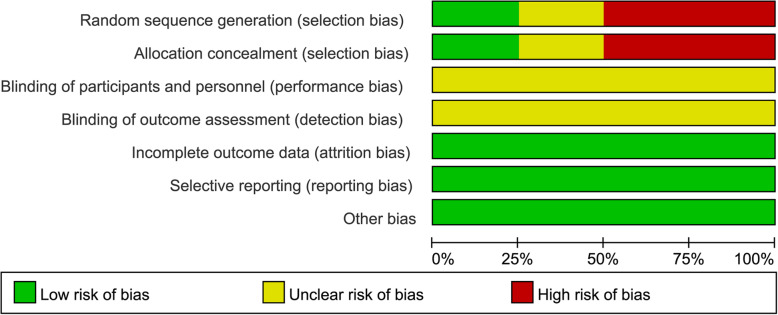
The publication year of the 4 literatures was from 2014 to 2019. The research areas mainly focused on Japan. A total of 456 cases were enrolled, including 238 in the ELD + BP group and 218 in the BP group. Except for the research by Takeuchi et al. [27], there was no significant difference in age and BMI between ELD + BP group and BP group in all studies. The intervention plan of Ebina et al. [28] was same with Suzuki et al. [29]. The follow-up period of each study was 6–18 months. The results of literature quality evaluation showed that the risk of selection bias in the included studies was relatively high. All studies had not reported the implementation of a blind method on measurement of implementers, research objects, and outcomes. Therefore, the evaluations of “blinding of participants and personnel” and “blinding of outcomes assessment” were defined as “unclear risk” (Fig. 2). However, the other evaluation items were defined as “low risk.” Overall, the methodological bias included in each literature was moderate (Fig. 3).
Fig. 2.
The publication bias evaluation based on enrolled literatures
Fig. 3.
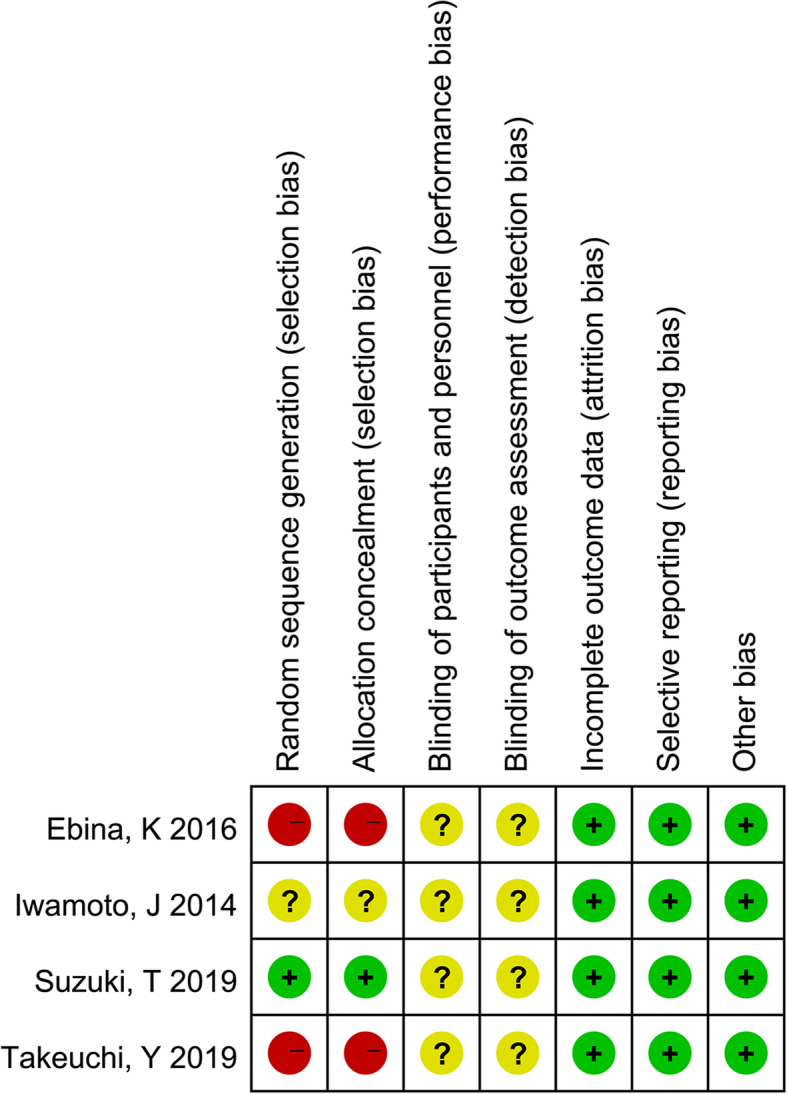
Summary of the risk of bias for each included study. “+” (green): low risk of bias; “?” (yellow): unclear risk of bias; “−” (red): high risk of bias
Meta-analysis
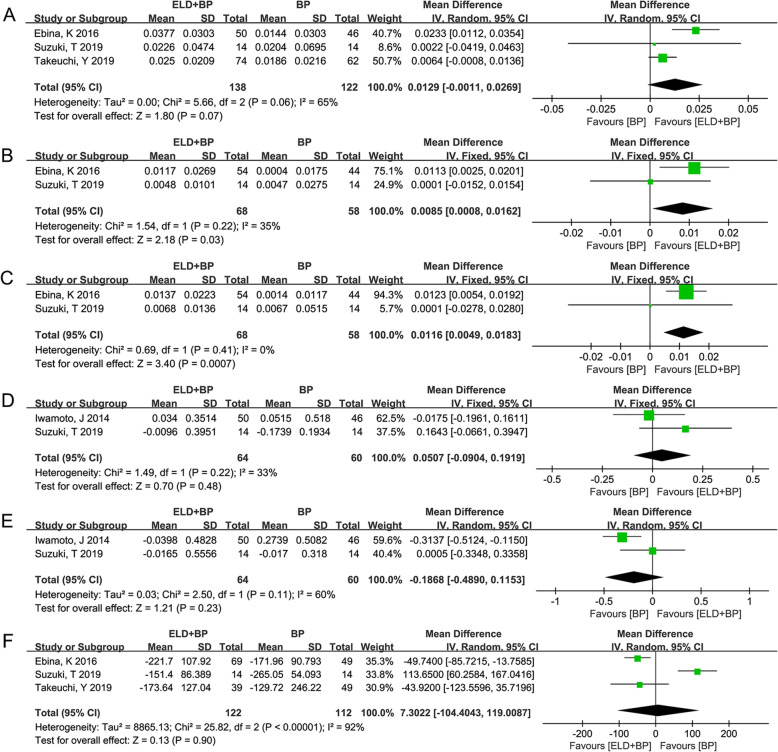
All events were divided according to the follow-up time (≤ 6 months and 12 months) in the subsequent analysis due to the different follow-up times in each study. A total of 3 studies [27–29] reported the outcomes of LS-BMD with follow-up time of less than 6 months. Since the heterogeneity test among these 3 studies [27–29] were statistically significant (P = 0.06, I2 = 65%), the random-effect model was used. The difference of LS-BMD between ELD + BP and BP (WMD (95%CI) = 0.0129 (− 0.0011, 0.0269) g/cm2, P = 0.07) (Fig. 4a) was not significant. Moreover, there were 2 studies [28, 29] that reported the outcomes of FN-BMD and TH-BMD with follow-up time less than 6 months. Since the heterogeneity test among these 2 studies [28, 29] were not statistically significant (P > 0.05, I2 < 50%), the fixed-effect model was used to calculate the WMD values and 95% CI. The difference between ELD + BP and BP for both FN-BMD (WMD (95%CI) = 0.0085 (0.0008, 0.0162) g/cm2) (Fig. 4b) and TH-BMD (WMD (95%CI) = 0.0116 (0.0049, 0.0183) g/cm2) (Fig. 4c) was significant, which further indicate the better effect of ELD + BP on increasing bone density. Furthermore, a total of 2 studies [29, 30] reported the outcomes of serum calcium and serum phosphorus with follow-up time less than 6 months. The results of meta-analysis for serum calcium (WMD (95% CI) = 0.0507 (− 0.0904, 0.1919) mg/dL, P = 0.48; heterogeneity: I2 = 33%, P = 0.22) and serum phosphorus (WMD (95%CI) = − 0.1868 (− 0.4890, 0.1153) mg/dL; heterogeneity: I2 = 60%, P = 0.23) showed that the difference of ELD + BP vs. BP was not significant in both serum calcium (Fig. 4d) and serum phosphorus (Fig. 4e). Additionally, a total of 3 studies [27–29] reported the outcomes of serum TRACP-5b with follow-up time less than 6 months. Since the heterogeneity test among these 3 studies [27–29] were statistically significant (I2 = 92%, P < 0.00001), the random-effect model was used. The difference of serum TRACP-5b between ELD + BP and BP (WMD (95%CI) = 7.3022 (− 104.4043, 119.0087) mU/dL, P = 0.90) (Fig. 4f) was not significant.
Fig. 4.
The results of meta-analysis for the effect of eldecalcitol (ELD) + bisphosphonate (BP) vs. BP on osteoporosis based on different indicators (intervention ≤ 6 months). a The pooled results from combing effect sizes for lumbar spine (LS) bone mineral density (BMD) using the random-effect model. b, c The pooled results from combing effect sizes for femoral neck (FN)-BMD and total hip (TH)-BMD using the fixed-effect model. d, e The meta-analysis for serum calcium and phosphorus. f The pooled results from combing effect sizes for serum TRACP-5b using the random-effect model
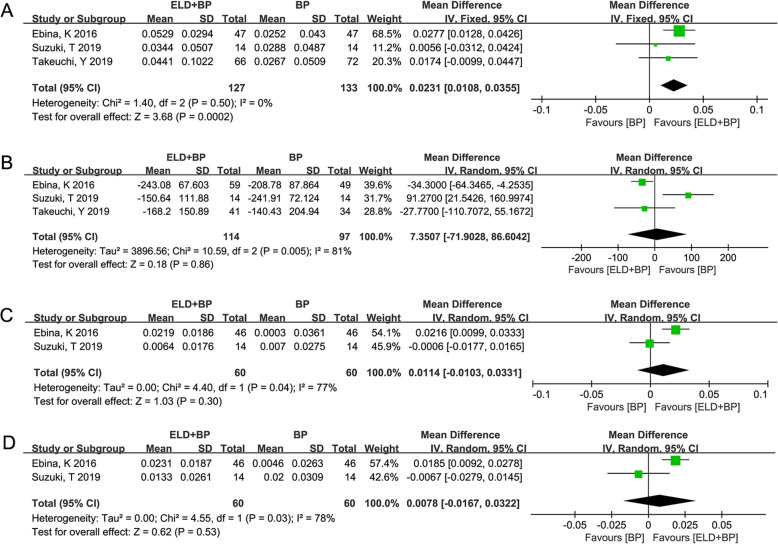
Three studies [27–29] reported the results of LS-BMD (Fig. 5a) and serum TRACP-5b (Fig. 5b) with 12-month follow-up. The result showed that there was no significant heterogeneity (I2 = 0%, P = 0.50) of LS-BMD between ELD + BP and BP; thus, the fixed-effect model analysis was performed (WMD (95% CI) = 0.0231 (0.0108, 0.0355) g/cm2, P = 0.0002). Meanwhile, there was a significant heterogeneity (I2 = 81%, P = 0.005) of serum TRACP-5b between ELD + BP and BP, and the combined result of the random-effects model was WMD (95% CI) = 7.3507 (− 71.9028, 86.6042) mU/dL, P = 0.86. Moreover, two studies [28, 29] reported the comparison of FN-BMD (Fig. 5c) and TH-BMD (Fig. 5d) with 12-month follow-up. The result of the meta-analysis for FN-BMD was WMD (95% CI) = 0.0114 (− 0.0103, 0.0331) g/cm2, P = 0.30 (heterogeneity test result: I2 = 77%, P = 0.04). The result of TH-BMD was WMD (95% CI) = 0.0078 (− 0.0167, 0.0322) g/cm2, P = 0.53 (heterogeneity test result: I2 = 78%, P = 0.03). However, the meta-analysis was not performed on the outcomes of serum calcium and serum phosphorus with 12-month follow-up due to the small sample size (only 1 study reported).
Fig. 5.
The results of meta-analysis for the effect of eldecalcitol (ELD) + bisphosphonate (BP) vs. BP on osteoporosis based on different indicators (intervention = 12 months). a The pooled results from combing effect sizes for lumbar spine (LS) bone mineral density (BMD) using the fixed-effect model. b The pooled results from combing effect sizes for serum TRACP-5b using the random-effect model. c, d The results of meta-analysis for femoral neck (FN)-BMD and total hip (TH)-BMD
Discussion
The result of current meta-analysis showed that the effect of ELD + BP was superior to BP alone based on indicators including FN-BMD and TH-BMD in patients with followed up ≤ 6 months. Meanwhile, the effect of ELD + BP was superior to BP alone based on LS-BMD in patients with 12 months follow-up. However, there was no significant difference between ELD + BP and BP with other indicators such as serum calcium in patients with follow-up ≤ 6 months or 12 months.
The prevalence of osteoporosis and low bone mass can be estimated by FN-BMD and TH-BMD in adults 50 years and older [31, 32]. The quality of postmenopausal osteoporosis patients is increased when receiving weekly BP treatment [9]. Based on a certain mechanism of cell regulation, BP can improve the bone density of patients and reduce the bone turnover, so as to reduce the possibility of fracture [33]. However, elderly, postmenopausal, osteoporotic obese women are resistant to long-term BP, especially in regions of the TH, FN, and forearm compared with the spine [34]. Although BP is indicated in the prevention and treatment during the process of osteoporosis, BMD continues to decline in up to 15% of BP users [35]. It is proved that with the increase of time, BP can effectively slow down the speed of bone gain [36]. As an active analog of vitamin D, ELD is commonly used in the treatment of osteoporosis [12]. ELD can reduce the re-absorption of blood calcium into the bone, improve the absorption of calcium in the intestine, and then increase the bone density in osteoporosis patients [13]. A previous study shows that ELD has a significant effect in additionally decreasing the level of the bone resorption, even in the osteoporosis patients who undergo the long-time therapy of BP [37]. Actually, ELD + BP has been proven to be effective for the treatment of osteoporotic patients in Japan [38]. Kamimura et al. indicated that ELD provided additive increasing in patients with long-term BP therapy based on the detection of indicators such as BMD and bone turnover [15]. In this meat-analysis, the result showed that the effect of ELD + BP was superior to BP alone based on indicators including FN-BMD and TH-BMD in patients with follow-up ≤ 6 months. Meanwhile, the effect of ELD + BP was superior to BP alone based on LS-BMD in patients with 12 months follow-up. Thus, we speculated that the therapeutic effect of ELD + BP was superior to BP alone in osteoporotic patients based on the influence of BMD.
As we all know, the calcium balance or calcium homeostasis is very important to human bone health, which is achieved by the continuous transformation between osteoblasts and osteoclasts [39]. In postmenopausal women, ELD increases urinary calcium, consistent with an increase in intestinal absorption, and reduces markers of bone turnover [40]. Compared to alfacalcidol, ELD can effectively inhibit bone resorption by urinary serum calcium excretion, which further indicates a better osteoprotective effect of ELD in osteoporotic patients [41]. Furthermore, the effect of BP on bone resorption, calcium balance, and BMD has already been proven in vivo [42]. A previous study shows that BP can be used to regulate the serum calcium in hypercalcemia [43]. In the current meta-analysis, there was no significant difference between ELD + BP and BP with indicators such as serum calcium in patients with followed up ≤ 6 months or 12 months. This result showed that the difference in the effects of ELD + BP and BP alone on blood calcium regulation was not significant, indicating a good reliability of ELD + BP in clinical use.
There were some limitations in the current study. First, the heterogeneity in this study is not ideal. The reason for heterogeneity might be the types of drugs included in BP which were not identical. Second, there was no publication bias test for the current study, since there were only four studies included (only from Japan), which is not suitable for publication bias test. Even so, this study might be the first meta-analysis on the efficacy of ELD + BP vs. BP in the treatment of osteoporosis. Meanwhile, there was no significant heterogeneity in the included literature, indicating that the results of meta-analysis were reliable.
Conclusion
In conclusion, the therapeutic effect of ELD + BP was superior to BP alone in osteoporotic patients based on the influence of BMD. However, further rigorous and high-quality validation studies based on a large sample size are needed in the future.
Acknowledgements
None.
Authors’ contributions
Conception and design of the research: ZZ; acquisition of data: ZZ; analysis and interpretation of data: JL; statistical analysis: JL; obtaining funding: ZZ; drafting the manuscript: ZZ; revision of manuscript for important intellectual content: ZZ. All authors read and approved the final manuscript.
Funding
This study was supported by the Zhejiang Public Technology and Policy Science (social science) Applied Research Project (No. 2020C35071) and Zhejiang Medical Science and Technology Project (No. 2020KY086).
Availability of data and materials
The data used to support the findings of this study are available from the corresponding author upon request.
Ethics approval and consent to participate
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kanis JA. Diagnosis and clinical aspects of osteoporosis. Pocket Reference to Osteoporosis: Springer; 2019. p. 11-20.
- 2.Sànchez-Riera L, Carnahan E, Vos T, Veerman L, Norman R, Lim S, et al. The global burden attributable to low bone mineral density. Ann Rheum Dis. 2014;73(9):1635–1645. doi: 10.1136/annrheumdis-2013-204320. [DOI] [PubMed] [Google Scholar]
- 3.Bonnick SL, Shulman L. Monitoring osteoporosis therapy: bone mineral density, bone turnover markers, or both? Am J Med. 2006;119(4):S25–S31. doi: 10.1016/j.amjmed.2005.12.020. [DOI] [PubMed] [Google Scholar]
- 4.Kaufman J-M, Orwoll E, Goemaere S, San Martin J, Hossain A, Dalsky G, et al. Teriparatide effects on vertebral fractures and bone mineral density in men with osteoporosis: treatment and discontinuation of therapy. Osteoporos Int. 2005;16(5):510–516. doi: 10.1007/s00198-004-1713-3. [DOI] [PubMed] [Google Scholar]
- 5.Fink HA, MacDonald R, Forte ML, Rosebush CE, Ensrud KE, Schousboe JT, et al. Long-term drug therapy and drug discontinuations and holidays for osteoporosis fracture prevention: a systematic review. Ann Intern Med. 2019;171(1):37–50. doi: 10.7326/M19-0533. [DOI] [PubMed] [Google Scholar]
- 6.Ensrud KE, Crandall CJ. Bisphosphonates for postmenopausal osteoporosis. JAMA. 2019;322(20):2017–2018. doi: 10.1001/jama.2019.15781. [DOI] [PubMed] [Google Scholar]
- 7.Naylor K, McCloskey E, Jacques R, Peel N, Paggiosi M, Gossiel F, et al. Clinical utility of bone turnover markers in monitoring the withdrawal of treatment with oral bisphosphonates in postmenopausal osteoporosis. Osteoporos Int. 2019;30(4):917–922. doi: 10.1007/s00198-018-04823-5. [DOI] [PubMed] [Google Scholar]
- 8.Kocijan R, Weigl M, Skalicky S, Geiger E, Ferguson J, Leinfellner G, et al. MicroRNA levels in bone and blood change during bisphosphonate and teriparatide therapy in an animal model of postmenopausal osteoporosis. Bone. 2020;131:115104. doi: 10.1016/j.bone.2019.115104. [DOI] [PubMed] [Google Scholar]
- 9.Hagino H, Soen S, Sugimoto T, Endo N, Okazaki R, Tanaka K, et al. Changes in quality of life in patients with postmenopausal osteoporosis receiving weekly bisphosphonate treatment: a 2-year multicenter study in Japan. J Bone Miner Metab. 2019;37(2):273–281. doi: 10.1007/s00774-018-0914-3. [DOI] [PubMed] [Google Scholar]
- 10.Fiscaletti M, Loucos R, Jamil KA, Biggins A, Munns C, Pacey V, editors. Does prior bisphosphonate therapy in children and adolescents with cerebral palsy alter surgical outcomes? 9th International Conference on Children; 2019: BioScientifica.
- 11.Cummings SR, Lui L-Y, Eastell R, Allen IE. Association between drug treatments for patients with osteoporosis and overall mortality rates: a meta-analysis. JAMA Intern Med. 2019;179(11):1491–1500. doi: 10.1001/jamainternmed.2019.2779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang W, Gao Y, Liu H, Feng W, Li X, Guo J, et al. Eldecalcitol, an active vitamin D analog, effectively prevents cyclophosphamide-induced osteoporosis in rats. Experimental and Therapeutic Medicine. 2019;18(3):1571–1580. doi: 10.3892/etm.2019.7759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jiang Y, Tang H, Ma X, Cheng Q, Lin H, Jin X, et al. Eldecalcitol increases bone mineral density in Chinese osteoporotic patients without vitamin D or calcium supplementation. J Bone Miner Metab. 2019;37(6):1036–1047. doi: 10.1007/s00774-019-01009-9. [DOI] [PubMed] [Google Scholar]
- 14.Matsumoto T, Yamamoto K, Takeuchi T, Tanaka Y, Tanaka S, Nakano T, et al. Eldecalcitol is superior to alfacalcidol in maintaining bone mineral density in glucocorticoid-induced osteoporosis patients (e-GLORIA). J Bone Min Metab. 2020:1–11. [DOI] [PubMed]
- 15.Kamimura M, Ikegami S, Mukaiyama K, Koiwai H, Nakamura Y, Taguchi A, et al. Additive effects of eldecalcitol in poorly responding long-term bisphosphonate treatment for osteoporosis. Osteoporosis and Sarcopenia. 2019;5(2):57–61. doi: 10.1016/j.afos.2019.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kinoshita M, Ishijima M, Kaneko H, Liu L, Nagao M, Sadatsuki R, et al. The increase in bone mineral density by bisphosphonate with active vitamin D analog is associated with the serum calcium level within the reference interval in postmenopausal osteoporosis. Mod Rheumatol. 2019;29(1):157–164. doi: 10.1080/14397595.2018.1442671. [DOI] [PubMed] [Google Scholar]
- 17.Wang J, Li H. Treatment of glucocorticoid-induced osteoporosis with bisphosphonates alone, vitamin D alone or a combination treatment in eastern Asians: a meta-analysis. Curr Pharm Des. 2019;25(14):1653–1662. doi: 10.2174/1381612825666190619125426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ebina K, Hashimoto J, Kashii M, Hirao M, Kaneshiro S, Noguchi T, et al. The effects of switching daily teriparatide to oral bisphosphonates or denosumab in patients with primary osteoporosis. J Bone Miner Metab. 2017;35(1):91–98. doi: 10.1007/s00774-015-0731-x. [DOI] [PubMed] [Google Scholar]
- 19.Watts NB, Diab DL. Long-term use of bisphosphonates in osteoporosis. The Journal of Clinical Endocrinology & Metabolism. 2010;95(4):1555–1565. doi: 10.1210/jc.2009-1947. [DOI] [PubMed] [Google Scholar]
- 20.Otto S, Abu-Id MH, Fedele S, Warnke PH, Becker ST, Kolk A, et al. Osteoporosis and bisphosphonates-related osteonecrosis of the jaw: not just a sporadic coincidence–a multi-Centre study. J Cranio-Maxillofac Surg. 2011;39(4):272–277. doi: 10.1016/j.jcms.2010.05.009. [DOI] [PubMed] [Google Scholar]
- 21.Higgins JP, Green S. Cochrane handbook for systematic reviews of interventions: Wiley online library. 2008. [Google Scholar]
- 22.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ (Clinical research ed). 2003 Sep 6;327(7414):557-60. PubMed PMID: 12958120. PMCID: 192859. Epub 2003/09/06. eng. [DOI] [PMC free article] [PubMed]
- 23.Iwamoto J, Sato Y. Eldecalcitol improves chair-rising time in postmenopausal osteoporotic women treated with bisphosphonates. Therapeutics and clinical risk management. 2014;10:51-9. PubMed PMID: 24476669. PMCID: Pmc3897513. Epub 2014/01/31. eng. [DOI] [PMC free article] [PubMed]
- 24.Suzuki T, Nakamura Y, Kato H. Effects of monthly minodronate with or without eldecalcitol addition in osteoporosis patients with rheumatoid arthritis: an 18-month prospective study. Osteoporosis and sarcopenia. 2019 Dec;5(4):122-7. PubMed PMID: 31938731. PMCID: Pmc6953524. Epub 2020/01/16. eng. [DOI] [PMC free article] [PubMed]
- 25.Ebina K, Noguchi T, Hirao M, Kaneshiro S, Tsukamoto Y, Yoshikawa H. Comparison of the effects of 12 months of monthly minodronate monotherapy and monthly minodronate combination therapy with vitamin K2 or eldecalcitol in patients with primary osteoporosis. J Bone Miner Metab 2016 May;34(3):243-250. PubMed PMID: 26303222. Epub 2015/08/26. eng. [DOI] [PubMed]
- 26.Takeuchi Y, Hashimoto J, Kakihata H, Nishida Y, Kumagai M, Yamagiwa C. Effectiveness of monthly intravenous ibandronate injections in a real-world setting: subgroup analysis of a postmarketing observational study. Osteoporosis and sarcopenia. 2019 Mar;5(1):11-8. PubMed PMID: 31008373. PMCID: Pmc6452926. Epub 2019/04/23. eng. [DOI] [PMC free article] [PubMed]
- 27.Takeuchi Y, Hashimoto J, Kakihata H, Nishida Y, Kumagai M, Yamagiwa C. Effectiveness of monthly intravenous ibandronate injections in a real-world setting: subgroup analysis of a postmarketing observational study. Osteoporosis and Sarcopenia. 2019;5(1):11–18. doi: 10.1016/j.afos.2019.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ebina K, Noguchi T, Hirao M, Kaneshiro S, Tsukamoto Y, Yoshikawa H. Comparison of the effects of 12 months of monthly minodronate monotherapy and monthly minodronate combination therapy with vitamin K 2 or eldecalcitol in patients with primary osteoporosis. J Bone Miner Metab. 2016;34(3):243–250. doi: 10.1007/s00774-015-0710-2. [DOI] [PubMed] [Google Scholar]
- 29.Suzuki T, Nakamura Y, Kato H. Effects of monthly minodronate with or without eldecalcitol addition in osteoporosis patients with rheumatoid arthritis: an 18-month prospective study. Osteoporosis and Sarcopenia. 2019;5(4):122–127. doi: 10.1016/j.afos.2019.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Iwamoto J, Sato Y. Eldecalcitol improves chair-rising time in postmenopausal osteoporotic women treated with bisphosphonates. Ther Clin Risk Manag. 2014;10:51. doi: 10.2147/TCRM.S54772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wright NC, Looker AC, Saag KG, Curtis JR, Delzell ES, Randall S, et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner Res. 2014;29(11):2520–2526. doi: 10.1002/jbmr.2269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shin CS, Choi HJ, Kim MJ, Kim JT, Yu SH, Koo BK, et al. Prevalence and risk factors of osteoporosis in Korea: a community-based cohort study with lumbar spine and hip bone mineral density. Bone. 2010;47(2):378–387. doi: 10.1016/j.bone.2010.03.017. [DOI] [PubMed] [Google Scholar]
- 33.Bourrin S, Ammann P, Bonjour J, Rizzoli R. Recovery of proximal tibia bone mineral density and strength, but not cancellous bone architecture, after long-term bisphosphonate or selective estrogen receptor modulator therapy in aged rats. Bone. 2002;30(1):195–200. doi: 10.1016/s8756-3282(01)00661-5. [DOI] [PubMed] [Google Scholar]
- 34.Dagdelen S, Sener D, Bayraktar M. Influence of type 2 diabetes mellitus on bone mineral density response to bisphosphonates in late postmenopausal osteoporosis. Adv Ther. 2007;24(6):1314–1320. doi: 10.1007/BF02877778. [DOI] [PubMed] [Google Scholar]
- 35.Heckman GA, Papaioannou A, Sebaldt RJ, Ioannidis G, Petrie A, Goldsmith C, et al. Effect of vitamin D on bone mineral density of elderly patients with osteoporosis responding poorly to bisphosphonates. BMC Musculoskelet Disord. 2002;3(1):6. doi: 10.1186/1471-2474-3-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Orr-Walker B, Wattie DJ, Evans MC, Reid IR. Effects of prolonged bisphosphonate therapy and its discontinuation on bone mineral density in post-menopausal osteoporosis. Clin Endocrinol. 1997;46(1):87–92. doi: 10.1046/j.1365-2265.1997.d01-1741.x. [DOI] [PubMed] [Google Scholar]
- 37.Iba K, Sonoda T, Takada J, Dohke T, Yamashita T. Further significant effects of eldecalcitol on bone resorption markers and bone mineral density in postmenopausal osteoporosis patients having undergone long-term bisphosphonate treatment. J Bone Miner Metab. 2017;35(2):171–176. doi: 10.1007/s00774-016-0738-y. [DOI] [PubMed] [Google Scholar]
- 38.Mukaiyama K, Uchiyama S, Nakamura Y, Ikegami S, Taguchi A, Kamimura M, et al. Eldecalcitol, in combination with bisphosphonate, is effective for treatment of Japanese osteoporotic patients. Tohoku J Exp Med. 2015;237(4):339–343. doi: 10.1620/tjem.237.339. [DOI] [PubMed] [Google Scholar]
- 39.Cashman K. Calcium intake, calcium bioavailability and bone health. Br J Nutr. 2002;87(S2):S169–SS77. doi: 10.1079/BJNBJN/2002534. [DOI] [PubMed] [Google Scholar]
- 40.Sanford M, McCormack PL. Eldecalcitol. Drugs. 2011;71(13):1755-70. [DOI] [PubMed]
- 41.Matsumoto T, Takano T, Yamakido S, Takahashi F, Tsuji N. Comparison of the effects of eldecalcitol and alfacalcidol on bone and calcium metabolism. J Steroid Biochem Mol Biol. 2010;121(1-2):261–264. doi: 10.1016/j.jsbmb.2010.03.035. [DOI] [PubMed] [Google Scholar]
- 42.Ammann P, Rizzoli R, Caverzasio J, Shigematsu T, Slosman D, Bonjour JP. Effects of the bisphosphonate tiludronate on bone resorption, calcium balance, and bone mineral density. J Bone Miner Res. 1993;8(12):1491–1498. doi: 10.1002/jbmr.5650081212. [DOI] [PubMed] [Google Scholar]
- 43.van Breukelen FJ, Bijvoet OL, Frijlink WB, Sleeboom HP, Mulder H, van Oosterom AT. Efficacy of amino-hydroxypropylidene bisphosphonate in hypercalcemia: observations on regulation of serum calcium. Calcif Tissue Int. 1982;34(1):321–327. doi: 10.1007/BF02411261. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.