Abstract
Craniopharyngiomas (CP) are rare brain tumors managed primarily with surgery and radiotherapy. There are 2 phenotypes of CP, i.e., one with a rather good outcome without hypothalamic damage and another with hypothalamic damage. With hypothalamic damage, progressive disease with recurrent operations and additional cranial radiotherapy often result in hypothalamic obesity, an affected psychosocial life, and cognitive dysfunction. The morbidity and mortality are increased for particularly cerebrovascular diseases. Preoperative hypothalamic involvement to predict hypothalamic damage is important for decision making for hypothalamus-sparing surgery. Also a postoperative hypothalamic damage evaluation with the use of hypothalamus volume measurement can predict hypothalamic obesity, which is important for early treatment options. The morbidity of CP includes cognitive dysfunction with attention deficits and impaired episodic memory and processing speed. Again patients with hypothalamic damage are more affected. Treatment options of hypothalamic obesity in the chronic phase are scarce and not convincingly successful. The most optimal situation is to try to hinder or stop the evolution of hypothalamic obesity. Prevention of hypothalamic damage is recommended, with special regard to hypothalamus-sparing therapeutic approaches that respect the integrity of essential nuclei located in both the medial and the posterior hypothalamic areas.
Keywords: Craniopharyngioma, Hypothalamus, Neurosurgery, Hypothalamic obesity
Background
Etiology and Survival of Craniopharyngiomas
Craniopharyngiomas (CP) are rare brain tumors managed primarily with surgery and radiotherapy [1, 2]. CP constitute 1.2–4.6% of all intracranial tumors, accounting for 0.5–2.5 new cases per 1 million per year globally [3, 4]. Due to their origin from remnants of the craniopharyngeal duct epithelium, CP are located either in the sella turcica or above the sella turcica (suprasellar). About 50% originate at the level of the third ventricle floor, within infundibulum and tuber cinereuom, and expand into the third ventricle cavity.
There are 2 histological subtypes of CP, i.e., adamantinomatous (ACP) and papillary CP (PCP), which differ in their genesis and age distribution. ACP are driven by somatic mutations in CTNNB1(encoding β-catenin) that increase β-catenin stability, leading to activation of the WNT pathway, and have been proposed to be of embryonic origin based on molecular and histological features [5, 6, 7]. In the childhood and adolescent age groups, the APC histological type with cyst formation is the most common [8]. PCP frequently harbor somatic BRAFV600E mutations that result in activation of the mitogen-activated protein kinase (MAPK) signaling pathway [9, 10]. In contrast to ACP, PCP are more frequently noncalcified and “solid.” In a retrospective study with surgical data of 500 CP, PCP typically presented as solid masses with no or rare calcifications, had more “loose” adhesions, and predominantly had a sessile or pedicle attachment (63%) in the inner lining of the third ventricle compared to ACP, which were associated with the widest and strongest adherence [11].
The age at presentation of ACP is bimodal and peaks at 5–15 years and rarely at 45–60 years, whereas PCP are mainly restricted to adults at 45–60 years [3, 4]. In a mixed population of childhood and adult onset CP, the overall mortality was increased and showed a standardized mortality ratio (SMR) of 5.5–9.28. The cardiovascular mortality was high (SMR 3.21–3.5) and particularly the cerebrovascular mortality was increased (SMR 5.1–19.4), with a higher risk in women [12, 13, 14]. The low incidence of CP and the dissimilarity in presentation, location, and treatment limit our knowledge, expertise, and management options. Among patients with ACP tumors the mortality was particularly increased in those with hypothalamic involvement, and the 20-year overall survival of patients with CP with hypothalamus (HT) involvement versus no HT involvement was particularly impaired. Importantly, overall survival was not related to degree of resection, sex, age at diagnosis, or year of diagnosis [15]. Gross total removal of a CP will reduce the recurrence rate, but it increases the rate of loss of endocrine function to 85% [16]. Preservation of the stalk reduces the rate of GTR but increases the tumor progression and the need for radiation. However, subtotal resection is the goal as it will reduce the risk of hypothalamic damage [16]. After adjustment for age in a mixed population of childhood and adult onset CP, a protective effect of radiotherapy and an increased risk of death after recurrence were found (HR = 4.4; 95% CI 1.4–14), but no obvious effect of radicality at surgery was seen [12] (Table 1).
Table 1.
HR and 95% CI for death after surgery for CP, with respect to tumor recurrence, radicality at surgery, and radiotherapy, with and without age stratification
| Cox regression model | Without age stratification1 |
With age stratification1 |
||
| HR (95% CI) | p value | HR (95% CI) | p value | |
|---|---|---|---|---|
| All patients (n = 60) | ||||
| Recurrence (time dependent; yes vs. no) | 2.7 (1.0–7.3) | 0.05 | 4.4 (1.4–14) | 0.01 |
| Radicality at surgery (subtotal vs. total) | 2.0 (o.9–4.5) | 0.09 | 1.1 (0.5–2.5) | 0.9 |
| Radiotherapy (yes vs. no) | 0.5 (0.2–1.2) | 0.1 | 0.3 (0.1–0.8) | 0.01 |
| Patients surviving the first 6 months after surgery (n = 50) | ||||
| Recurrence (time dependent; yes vs. no) | 1.6 (0.5–4.7) | 0.4 | 2.2 (0.6–7.6) | 0.2 |
| Radicality at surgery (subtotal vs. total) | 1.6 (o.6–4.5) | 0.4 | 0.9 (0.3–2.5) | 0.8 |
| Radiotherapy (yes vs. no) | 1.4 (o.4–4.1) | 0.6 | 0.7 (0.2–2.4) | 0.6 |
Reproduced from Billow et al. [12] with permission from Oxford University Press.
Age <29 or ≥29 years at surgery.
Although the long-term survival is high, quality of life and neuropsychological function are frequently impaired due to the close anatomical proximity to the optic chiasm, the HT, and the pituitary gland. Indeed, hypothalamic involvement and treatment-related hypothalamic lesions frequently result in hypothalamic obesity (HO), psychosocial, and cognitive deficits [17].
Hypothalamic Involvement from CP
Preoperative Hypothalamic Involvement and Prediction of Hypothalamic Damage
There are several studies analyzing preoperative hypothalamic involvement from CP; for example, the German KRANIOPHARYNGIOM 2000 and 2007 used a classification of CP according to their contact or compression of different structures of the HT [18]. Preoperative MRI were used to identify hypothalamic involvement, and there were 3 grades, i.e., 0, no contact with the floor of the third ventricle; 1, contact with or compression of the HT anterior to the mammillary bodies; and 2, those resulting in dislocation, compression, or destruction of the HT, including the mammillary bodies or the area dorsal to them [18]. Müller et al. [18] suggested that grade 2 is the most important predictor of damage to the HT. The question is whether grade 1 is really less grave than grade 2 as the medial HT is damaged and this area is of the utmost importance for energy expenditure.
Puget et al. [19] also suggested 3 grades of HT involvement on preoperative MRI, i.e., on sagittal T2 MRI: grade 0, no HT involvement with third ventricle floor intact and CP below the sellar diaphragm; grade 1, compression of the HT that can still be identified (from the mammillary bodies); and grade 2, unidentifiable HT. This grading can be of help in decision making regarding the type of operative approach for a CP with the aim of sparing the HT function.
Postoperative Hypothalamic Damage Evaluation and Prediction of HO
Based on 2-dimencial MRI scans, Roth et al. [20] investigated CP children with HO retrospectively (12–30 months after operation) and suggested that damage to the post-HT (dorsomedial nucleus and dorsal hypothalamic area) was the best predictor of a postoperative BMI z score increase. Additionally, lesions affecting the floor of the third ventricle, the mamillary bodies, and the anterior and medial HT showed correlations with a BMI z score increase [20].
Fjalldal et al. [21] used an HT volume measurement from Gabery et al. [22]. A T1-weighted MRI was used. This a delineation procedure with the same anatomical landmarks used in histologically processed postmortem hypothalamic tissue from patients with Huntington disease. This is a 3-dimensial HT volume measurement based on MRI (Fig. 1). We performed this procedure postoperatively in 36 ACP CP patients (23 with HT damage) and 32 matched controls. HT volume correlated highly negatively with fat mass and leptin among CP patients (rs = −0.67, p < 0.001, and rs = −0.53, p = 0.001, respectively) and explained 39% of the variation in fat mass (Fig. 2a, b). For every 100-mm3 increase in HT volume, the fat mass decreased by 2.7 kg (95% CI 1.5–3.9; p < 0.001) Thus, this volume measurement is highly predictive of HO and can be included in a formal pituitary HT MRI and takes an additional 6–8 min to perform. Indeed, the interrater variability was very low (<4%), as was the interrater correlation (i.e., 0.947) for this procedure [21].
Fig. 1.
Delineation of the hypothalamic region in 3-T MRI. Representative overview of the delineation principles of the HT in T1-weighted MRI acquired at 3 T according to Gabery et al. [22]. The anatomical landmarks were based on histologically processed postmortem hypothalamic tissue derived from patients with Huntington disease [22]. Between 14 and 16 images were used for per case analysis. Images were preprocessed with cubic spine interpolation of the original voxel size to 0.5 × 0.5 × 0.5 mm. The images were taken from slices 1, 4, 8, 12, and 16 with 2-mm spacing. a, d, g, j, m In a rostral-to-caudal direction, the images are representative 3 T MR images on a coronal plane. b, e, h, k, n Schematic overview of the corresponding level modified from Tang-Christensen et al. [41]. c, f, i, l, o Overlap of the schematic overview on the MR image. The light blue lines illustrate how the HT region was delineated. Landmarks such as the HT sulcus (represented by a white asterisk) and the lateral edge of the optical tract (represented by a red asterisk) were identified for delineation and a straight line between these 2 points was drawn to set the superior/lateral border of the area in a reproducible fashion. The optical tract was excluded in all slides. d, dorsomedial hypothalamic nucleus; F, fornix; i, infundibular nucleus; l, lateral HT; MB, mammillary body; OT, optical tract; P, paraventricular nucleus; s, supra optic nucleus; v, ventromedial hypothalamic nucleus. Reproduced from Gabery et al. [22] with permission from Springer Nature.
Fig. 2.
a Correlation between leptin (ng/mL) and hypothalamic volume (mm3) among 35 CP patients and 31 controls (n = 35, rs = −0.53; p = 0.001). b Correlation between fat mass (kg) and hypothalamic volume (mm3) among 35 CP patients and 31 controls (n = 35, rs = −0.67; p < 0.001). TGTV, tumor growth into the third ventricle. Reproduced from Fjalldal et al. [21] with permission from Springer Nature.
Two Phenotypes of CP with or without Hypothalamic Damage
There are 2 phenotypes, i.e., one with a rather good outcome without HT damage and another with hypothalamic damage where progressive disease with recurrent operations with additional cranial radiotherapy resulting in HO, an affected psychosocial life, and cognitive dysfunction [23, 24]. Long-term morbidity is associated with tumor-related and/or treatment-related problems. Irrespectively of hypothalamic involvement by the CP, both genders show high BMI, fat mass, and leptin, but women with CP show the highest risk of lipid derangement [23]; this is in accordance with the higher cardiovascular mortality seen in CP females. Patients with hypothalamic involvement had significantly higher weight, BMI, waist circumference, fat mass, and lower fat-free mass values compared to patients without hypothalamic involvement (Table 2). Serum insulin levels and insulin per kilogram of fat mass were also significantly increased in patients with HT involvement, and 10 out of 11 patients receiving treatment for CVD and/or with metabolic syndrome belonged to the group with hypothalamic involvement [23].
Table 2.
Anthropometric measurements, cardiovascular risk factors, and serum and plasma hormone levels in patients with and without TGTV
| Patients with TGTV (n = 25; 11 females) | Patients without TGTV (n = 17; 9 females) | p value | |
|---|---|---|---|
| Anthropometric measurements and body composition (BIA) | |||
| Weight, kg | 96 (60–149) | 80 (53–110) | 0.001 |
| BMI | 32 (21–41) | 25 (19–35) | 0.001 |
| Waist circumference, cm | 105 (77–131) | 89 (72–109) | 0.002 |
| WHR | 0.94 (0.83–1.05) | 0.88 (0.76–0.99) | 0.02 |
| Fat mass, kg | 40 (9–64) | 20 (13–56) | 0.003 |
| Fat mass, % | 39 (16–51) | 30 (19–52) | 0.04 |
| Muscle mass, kg | 44 (19–59) | 25 (21–60) | 0.4 |
| Muscle mass, % | 35 (12–44) | 28 (15–44) | 0.8 |
| Fat-free mass, kg | 57 (41–86) | 51 (40–68) | 0.015 |
| Fat-free mass, % | 61 (49–84) | 70 (48–81) | 0.04 |
| Cardiovascular risk factors and serum and plasma hormone levels | |||
| P-leptin, ng/mL | 28 (4–215) | 16 (3–67) | 0.07 |
| Leptin/kg of fat mass (BIA) | 0.74 (0.26–4.51) | 0.95 (0.16–1.56) | >0.3 |
| S-insulin, mIU/L | 7 (2–38) | 4 (1–8) | <0.001 |
| Insulin/kg of fat mass (BIA) | 0.21 (0.05–0.59) | 0.15 (0.06–0.35) | 0.047 |
| P-glucose, mmol/L | 4.6 (3.6–11.0) | 4.4 (3.7–5,9) | >0.3 |
| P-LDL, mmol/L | 2.9 (0.5–5.4) | 3.2 (2.2–4.7) | 0.23 |
| P-HDL, mmol/L | 1.2 (0.7–8.5) | 1.3 (0.9–2.4) | 0.23 |
| ApoB/ApoA-I ratio | 0.68 (0.35–1.40) | 0.66 (0.37–1.09) | >0.3 |
| P-fibrinogen, g/L | 3 (2–5) | 3 (2–4) | >0.3 |
| P-hs-CRP, mg/L | 2.8 (0.4–23) | 0.4 (0.4–16) | 0.07 |
| Systolic blood pressure, mm Hg | 115 (102–155) | 120 (102–130) | >0.3 |
| Diastolic blood pressure, mm Hg | 78 (55–88) | 72 (60–80) | 0.25 |
| P-IGF-I, µg/L | 182 (34–526) | 196 (94–328) | >0.3 |
| P-TSH, mU/L | 0.01 (0.0–0.37) | 0.02 (0.1–3.2) | 0.002 |
| P-free T3, pmol/L | 4.8 (2.8–6.2) | 4.8 (2.9–3.7) | >0.3 |
| P-free T4, pmol/L | 20 (16–26) | 17 (13–31) | 0.010 |
Reproduced from Holmer et al. [23] with permission from Bio Scientifica. Values are presented as medians (range). TGTV, tumor growth into the third ventricle; P, plasma; S, serum; ApoA-I, apolipoprotein A-I; ApoB, apolipoprotein B; BIA, bioelectric impedance analysis; HDL-C, high-density lipoprotein cholesterol; hs-CRP, high-sensitivity C-reactive protein; IGF-I, insulin-like growth factor-I; LDL-C, low-density lipoprotein cholesterol; TSH, thyroid-stimulating hormone; WHR, waist/hip circumference ratio.
Hypothalamic Damage and Evolution of HO
Background of HO
The main hypothalamic areas involved in energy regulation are the ventromedial HT (VMH), the paraventricular nucleus (PVN), arcuate nucleus (ARC), and the lateral hypothalamic area (LH; Fig. 3). The ARC has 2 sets of neurons - one generates the orexigenic agouti-related protein (AgRP) and neuropeptide Y (NPY) and the other generates the anorexigenic proopiomelanocortin (POMC) and cocaine- and amphetamine-related transcript (CART) [25]. POMC is a precursor of α-MSH (α-melanocyte-stimulating hormone), whose main effect on weight regulation is through MC4R (melanocortin-4 receptor) [26].
Fig. 3.
Schematic overview of the arcuate nucleus, the ventromedial hypothalamus, and the lateral hypothalamus with its main nucleus and interrelations. The arcuate nucleus has 2 sets of neurons – one generates the orexigenic AgRP and NPY and the other generates the anorexigenic POMC and CART. POMC is a precursor of α-MSH, whose main effect on weight regulation is through MC4R.
The first to find an association between hypothalamic tumors and obesity were Babinski [27] and Frohlich [28]; it was called “adipogenital syndrome” because patients also suffered from a loss of sex drive. In 1940 Hetherington and Ranson [29] called the syndrome “VMH obesity” based on the damage to the ventromedial hypothalamic nuclei. It is clear that in the initial phase of hypothalamic damage there is an enormous immediate urge for food and sweets and this is due to damage of the VMH. The strongest increase in weight is within the first 6–12 months [30], with a suggested plateau at 12–30 months [20]. The questions remain, however, when the acute phase of hunger is reduced or eliminated and whether treatment is possible. Roth et al. [20] suggested that a reduction of cravings appears after 12–30 months postoperatively. However, no strictly performed follow-up on this issue has yet been published and assumedly it depends on when the studies were performed. Some have recorded reduced or normalized eating after 12 years [15] and others around 16 years after the operation [31].
In animal studies it has been shown that an increase in insulin occurs within minutes or in the first days after VMH lesions in rats and even without the occurrence of hyperphagia or obesity [32]. Frohman and Bernardis [33] showed that VMH-lesioned weanling rats remained normophagic and yet became obese. Further, if the HT is damaged the result is an altered autonomic tone with a decrease in sympathetic tone and an increase in parasympathetic tone, where the latter causes vagus stimulation resulting in an increase in insulin, which causes hyperphagia [34, 35]. Additionally, high insulin levels stimulate lipogenesis in the liver and in adipose tissue [32]. In the liver the result is an increase in the production of triglycerides, which are converted to very low-density lipoproteins. In the adipose tissue, insulin stimulates triglyceride production and in blood insulin it may also directly affect hormone-sensitive lipase, which induces the conversion of triglycerides to fatty acid + glycerol. In addition, the decrease in sympathetic tone results in a reduction of fat mass lipolysis [35], which further accumulates fat mass. Thus, the background of HO is clearly multifactorial.
Neuropeptide Signaling Pathways in HO
Leptin is produced by fat cells and thus serum leptin levels are highly correlated to the amount of fat mass accumulation. However, there is also an increase in leptin/kilogram of fat mass, which means that there is a leptin resistance [31, 36]. Hypothalamic leptin resistance leads to the misperception of a hunger state with low leptin levels, which promotes an increase in energy intake that, together with a reduced energy expenditure [31, 37], will increase weight. The end result is a vicious circle of further weight gain.
Studies have identified the VMH nucleus as the “satiety center” while the lateral HT area is termed the “hunger center” [38]. Several observations indicate that leptin has a more important role than insulin in the CNS control of energy homeostasis. For instance, leptin deficiency causes severe obesity with hyperphagia in spite of high insulin levels [39]. Low leptin and insulin in the brain during weight loss increase the activity of the anabolic neural pathways that stimulate eating and suppress energy expenditure, and decrease the activity of catabolic pathways that cause anorexia and weight loss [39].
The hypothesis that the ARC transduces information related to signaling by leptin into a neural response is supported by the anorexic response to local microinjection of leptin into this area [40] and the inability of intracerebroventricular leptin to reduce the food intake after that the ARC has been destroyed [41, 42]. The majority of both NPY/AgRP and POMC/CART are co-expressed with leptin receptors [43, 44] as both types of neurons are regulated by leptin, but in an opposing manner.
Several neuropeptides synthesized in PVN neurons reduce the food intake and body weight when administered centrally. This includes CRH, which causes anorexia, and activation of the sympathetic nervous system, in addition to its central role in activation of the hypothalamic-pituitary-adrenal axis. Furthermore, also thyrotropin-releasing hormone (TRH) reduces the food intake [45] and stimulates the thyroid axis and oxytocin reduces the food intake. Additional factors for maintenance of HO are reduced nocturnal melatonin levels [46], which cause daytime sleepiness, as patients' neurological defects and visual failure result in decreased physical activity [31, 47, 48].
Additional Causes to HO
Brain inflammation might also be of importance for rapid weight gain, as increased interleukin-6 expression is observed in CP cystic fluid and concentrations reach levels 50,000-fold higher than in spinal fluid, and there is an increase in tumor necrosis factor-α in CP cyst fluid [49]. Two inflammatory mechanisms can have an impact; the craniotomy per se causes an inflammatory reaction [50] and, second, there is an inflammatory response due to intake of a high-fat diet. The latter has been recorded both in animal studies and in human studies where intake of a high-fat diet has resulted in gliosis development in mediobasal HT of obese humans assessed by MRI. Thus, neural injury has been recorded in brain areas crucial for body weight control [51].
Also energy expenditure, i.e., the basal metabolic rate (BMR) is very important. In the acute phase of HO it is not known, however, whether the BMR is downregulated. However, in the chronic phase of HO, the energy expenditure is decreased, as shown by indirect calorimetry [31, 37]. About 16 years after operation we recorded a decrease in energy expenditure, with a lower BMR, i.e., by 90 kcal per 24 h, after adjustment for sex and weight [31] (Fig. 4). This means that the BMR is reduced by about 2,700 kcal/month and so it goes on every month.
Fig. 4.
Linear association between total body weight (kg) and BMR (kcal/day) among patients and controls. Patients had a significantly lower BMR compared to controls after adjustment for sex and total body weight in the linear regression analysis (mean difference: −90 kcal/24 h; 95% CI −160 to −10; p = 0.02). Reproduced from Holmer et al. [31] with permission from Oxford University Press.
Food Intake and Eating Behavior in the Chronic Phase of HO
When investigations were conducted with dieticians to predict the present food intake, using a semiquantitative questionnaire from the SOS (Swedish Obesity Study) [52], CP patients had a significantly higher intake of light meals and sweets (mainly males), but they had a significantly lower intake of alcohol. Interestingly, no differences in the intake of fat, proteins, fibers, or carbohydrates were recorded [31]. Furthermore, with the use of Three Factor Eating Questionnaire [53], the CP patients versus normal controls experienced more “cognitive restraint,” i.e., they were more often thinking of not eating. However, they did not have more of a hunger feeling or “disinhibition,” with the latter meaning loss of eating control. Interestingly, the sum energy intake was significantly lower among patients (1,778 [range 970–3,230] vs. 2,094 kcal/day [range 1,128–5,449]; p < 0.003) compared to normal-weight controls, even after adjustment for age, sex, and weight. This is in agreement with a previous study in which CP patients' eating habits were not different from those of BMI-matched controls, and they did not a higher energy intake compared to BMI-matched controls [48].
Treatment of HO
Endocrine Hormone Supplementation
Total pituitary deficiencies are present in the majority of the patients; at least 3 hormone deficiencies have been reported in 50–100% of patients after treatment of CP [54]. The prevalence of GH, gonadotropin, ACTH, and TSH deficiencies has been reported as 91, 93.5, 92, and 86%, respectively [54]. The prevalence of diabetes insipidus is 81%. Interestingly, there is no clear difference in hormone deficiencies between patients with and without hypothalamic involvement by the CP [23].
GH, thyroid hormones, and testosterone are important for the metabolic system [55]. A loss or reduction of these hormones will cause a weight increase and an increased cardiovascular risk. Testosterone treatment in men is important for reducing the fat mass and increasing the muscle mass and for sexual function and bone mineral density [55]. Estrogen in women reduces the cardiovascular risk and mortality in premenopausal women [55]. High cortisol levels cause a weight increase in CP children [56]. Regarding levothyroxine treatment, there is no convincing evidence on BMI, but in general free thyroxine levels are recommended at the upper normal range in patients with central hypothyroidism [55]. GH treatment in children [57] and adults is important for the body composition [55], and there seems to be no risk of recurrence in CP [58, 59]. An individualized desmopressin intake is recommended as an increased intake of liquids may favor liquids with sugar [55].
Other Treatments for HO
There is very little data on successful weight loss in hypothalamic obese CP cases. There are 2 scenarios in the case of HT damage; the first and probably the most important is to take immediate action to try to stop the initial craving for food directly when it appears after the operation. The aim is to counteract the rapid rise in insulin, due to parasympathetic stimulation; to vagotomize is hardly possible, but octreotide has been used to lower insulin levels in children, with some good results [60]. Further, increasing the sympathetic tone is possible with the action of dextroamphetamin, which may improve impulse control and cravings. Methylfenidate has also been used in children to decrease the food intake [61, 62] and in a case report of a child with CP [63]. However, the indication is weak, possibly due to a lack of studies. The mechanism is suggested to increase the rewarding dopamine system and thus reduce the energy intake. The long-acting incretin glucagon-like peptide 1 receptor (GLP-1R) agonist is an option as it reduces stomach emptying and can bind to GLP-1R receptors in the HT, increasing satiety [64, 65]. The incretin liraglutide is registered for the indication of long-term use in general obesity and it has been used in CP with HO with some success (weight reduction of 9–22 kg) [66, 67, 68]. The second scenario is how to deal with the chronic phase of HO, which is reached about 12–24 months after treatment. The above mentioned drugs in the acute phase can be used, albeit with limited success, and they have not been used long-term. It seems clear that a patient with HO who tries to remain at a stable weight in the chronic phase of HO needs to keep up with daily physical exercise to increase their energy expenditure and particularly have a continuously lower energy intake compared to a person with normal weight. Which scenario for energy intake would be best? Not a state of starvation with rapid weight loss, because immediately the body will counteract to increase the energy intake together with a further lowering of the BMR. In addition, also thyroid hormones will be lowered among those patients with intact TRH-TSH axes, which also will counteract the weight loss [69]. The most reasonable way would be to slowly reduce the weight, but there is lack of data. GLP-1R agonists are promising and in favor of the more recent semaglutide [70] confounded with fewer side effects and a higher sense of satiety, but at present there are no published data on CP.
In addition, there is some experience with bariatric surgery in CP with HO. A positive effect on food intake has been reported in 3 studies; however, long-term data, i.e., more than 24 months, is lacking [71, 72, 73, 74]. The weight reduction was positive after sleeve gastrectomy and Roux-en-Y gastric binding but not after a laparoscopic adjustable gastric band. One year after bariatric surgery, 2 recent meta-analyses showed a weight increase after both a laparoscopic adjustable gastric band and sleeve gastrectomy [74, 75]. Except for Roux-en-Y gastric binding the total weight reduction during 2 years was lower compared to the result in normal controls [74, 75].
Consequences of Hypothalamic Damage on Psychosocial and Cognitive Function
A reduced quality of life and neuropsychological problems are prevalent in about 60% of CP cases [24, 76]. Quality of life was investigated in a mixed cohort of childhood onset and adult onset CP; it was shown to be significantly decreased after surgery, and 14% were dependent on others for daily activities [14]. This is in agreement with Karavitaki et al. [47], who reported that there was a 9% probability of complete dependency for basal daily activities at 10 years and 23% of patients were unable to work in their previous occupation. Others have shown, in a mixed cohort of childhood onset and adult onset CP, that an impaired quality of life is especially prominent in physical subscales with compromised energy levels [77]. Hypothalamic damage had a negative impact on the self-assessed quality of life among children [78]. Dextroamphetamin treatment may improve concentration and hyperactivity both in children and adults with CP [79, 80]. However, when we investigated a cohort of 42 patients (60% hypothalamic damage) surgically treated for childhood-onset CP, with a mean age of 28 years, the patients rated their quality of life as positively as their matched controls [24] (Table 3). This is, however, only the case in childhood onset disease and not in adult onset, and it mirrors the subject's adaptation. However, no severely disabled patients were included in the mentioned study [24].
The morbidity of CP includes cognitive dysfunction with attention deficits and impaired episodic memory and processing speed [81, 82]. Patients with hypothalamic damage are more affected (Table 4) [24]. Animal studies have shown connectivity between the hippocampus and the HT [83] as well as from the HT directly to the frontal cortex [84]. Thus, hypothalamic lesions might indirectly lead to frontal and medial temporal lobe dysfunction, causing deficits in memory, attention, and executive function [82]. Furthermore, the hippocampus is important for encoding and retrieving sequences of events that compose particularly the episodic (contemporary) memory [85].
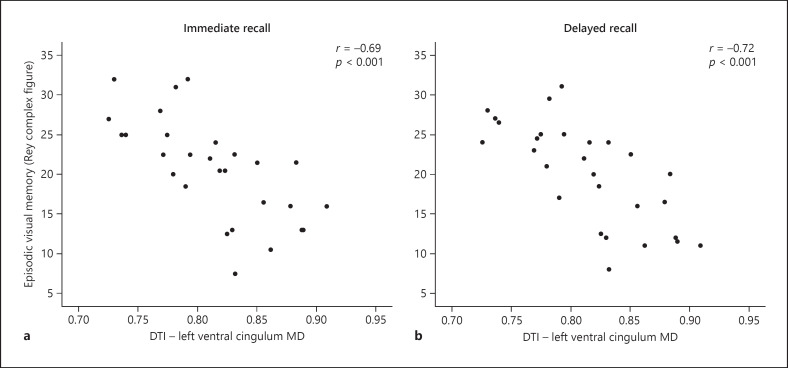
With the use of diffusion tensor imaging (DTI) we investigated how impairment in different cognitive domains would correlate to cognitive function and how hypothalamic lesions contribute to a worse outcome [81, 82]. DTI is a technique that allows noninvasive in vivo study of the white matter of the brain by assessing the motion of water molecules along and across neural axons [86, 87] (Fig. 5). The cingulum is an important WM tract with multiple reciprocal connections with the hippocampus and well-known importance for episodic memory [88, 89]. The uncinate fasciculus is also important for episodic memory and connects limbic regions in the temporal lobe to the frontal lobe [90]; the fornix connects the hippocampus to the prefrontal cortex [91]. We recorded significantly microstructural WM alterations in the right uncinate fasciculus (p ≤ 0.01) [81]. Further, WM alterations in the left ventral cingulum were significantly associated with a worse performance in visual episodic memory, explaining approximately 50% of the variation (Fig. 6a, b). Alterations in the dorsal cingulum were associated with a worse performance in immediate, delayed recall and recognition, explaining 26–38% of the variation, and for visuospatial ability and executive function it explained 19–29% of the variation. Patients had a smaller hippocampal volume compared to controls, and a smaller hippocampal volume was correlated with reduced general knowledge (p = 0.028). Moreover, microstructural WM alterations in the hippocampus were associated with a decline in general knowledge and episodic visual memory. Thus, a structure-to-function relationship is suggested between microstructural WM alterations in the cingulum and in the hippocampus with cognitive deficits in childhood onset CP [81].
Fig. 5.
Graphical renderings of directionally color-coded (red = right-left, green = anterior-posterior, and blue = superior-inferior) segmented tractographys of dorsal cingulum (a), ventral cingulum (b), fornix (c), and uncinate fasciculus (d) superimposed on a mid-sagittal FA map in a representative subject. Reproduced from Fjalldal et al. [81] with permission from Bio Scientifica.
Fig. 6.
Episodic visual memory (Rey complex figure test) with immediate recall (a) and delayed recall (b) in association with DTI in the left ventral cingulum (MD; mean diffusivity) among 32 CP patients. Reproduced from Fjalldal et al. [81] with permission from Bio Scientifica.
Conclusions
There are 2 phenotypes of CP; one has no HT involvement, resulting in a good outcome regarding quality of life and cognitive function and a lower cardiovascular risk. The other has HT involvement, resulting in a low cognitive function and an increased cerebrovascular risk with particularly HO. Immediately after HT damage due the tumor and/or its treatment, there is an increase in parasympathetic stimulation causing a rapid increase in insulin, which directly affects the liver and adipose tissue with lipogenesis. In addition, there is a downregulation of sympathetic tone, which causes an accumulation of triglycerides. Treatment options for HO in the chronic phase are very few and not convincingly successful. The most optimal situation is to try to hinder or stop the evolution of HO. Prevention of HO is recommended, with special regard to HT-sparing therapeutic approaches that respect the integrity of essential nuclei located in both the medial and the posterior hypothalamic areas.
Statement of Ethics
The responsible ethics committee approved the protocol for this study (DNR 2011/769). All of the participants gave written informed consent.
Conflict of Interest Statement
The authors have no conflict of interests to declare.
Funding Sources
This work was supported by the Swedish Children's Cancer Foundation and the Medical Faculty of Lund University (Sweden).
Author Contributions
Only E.-M.E. wrote this review and revised this work for resubmission.
Acknowledgement
We thank all of the patients and controls who participated in this study.
References
- 1.Müller HL, Merchant TE, Warmuth-Metz M, Martinez-Barbera JP, Puget S. Craniopharyngioma. Nat Rev Dis Primers. 2019 Nov;5((1)):75. doi: 10.1038/s41572-019-0125-9. [DOI] [PubMed] [Google Scholar]
- 2.Müller HL, Merchant TE, Puget S, Martinez-Barbera JP. New outlook on the diagnosis, treatment and follow-up of childhood-onset craniopharyngioma. Nat Rev Endocrinol. 2017 May;13((5)):299–312. doi: 10.1038/nrendo.2016.217. [DOI] [PubMed] [Google Scholar]
- 3.Bunin GR, Surawicz TS, Witman PA, Preston-Martin S, Davis F, Bruner JM. The descriptive epidemiology of craniopharyngioma. J Neurosurg. 1998 Oct;89((4)):547–51. doi: 10.3171/jns.1998.89.4.0547. [DOI] [PubMed] [Google Scholar]
- 4.Olsson DS, Andersson E, Bryngelsson IL, Nilsson AG, Johannsson G. Excess mortality and morbidity in patients with craniopharyngioma, especially in patients with childhood onset: a population-based study in Sweden. J Clin Endocrinol Metab. 2015 Feb;100((2)):467–74. doi: 10.1210/jc.2014-3525. [DOI] [PubMed] [Google Scholar]
- 5.Apps JR, Carreno G, Gonzalez-Meljem JM, Haston S, Guiho R, Cooper JE, et al. Tumour compartment transcriptomics demonstrates the activation of inflammatory and odontogenic programmes in human adamantinomatous craniopharyngioma and identifies the MAPK/ERK pathway as a novel therapeutic target. Acta Neuropathol. 2018 May;135((5)):757–77. doi: 10.1007/s00401-018-1830-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sekine S, Shibata T, Kokubu A, Morishita Y, Noguchi M, Nakanishi Y, et al. Craniopharyngiomas of adamantinomatous type harbor beta-catenin gene mutations. Am J Pathol. 2002 Dec;161((6)):1997–2001. doi: 10.1016/s0002-9440(10)64477-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Buslei R, Nolde M, Hofmann B, Meissner S, Eyupoglu IY, Siebzehnrübl F, et al. Common mutations of β-catenin in adamantinomatous craniopharyngiomas but not in other tumours originating from the sellar region. Acta Neuropathol. 2005 Jun;109((6)):589–97. doi: 10.1007/s00401-005-1004-x. [DOI] [PubMed] [Google Scholar]
- 8.Larkin SJ, Ansorge O. Pathology and pathogenesis of craniopharyngiomas. Pituitary. 2013 Mar;16((1)):9–17. doi: 10.1007/s11102-012-0418-4. [DOI] [PubMed] [Google Scholar]
- 9.Brastianos PK, Taylor-Weiner A, Manley PE, Jones RT, Dias-Santagata D, Thorner AR, et al. Exome sequencing identifies BRAF mutations in papillary craniopharyngiomas. Nat Genet. 2014 Feb;46((2)):161–5. doi: 10.1038/ng.2868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Haston S, Pozzi S, Carreno G, Manshaei S, Panousopoulos L, Gonzalez-Meljem JM, et al. MAPK pathway control of stem cell proliferation and differentiation in the embryonic pituitary provides insights into the pathogenesis of papillary craniopharyngioma. Development. 2017 Jun;144((12)):2141–52. doi: 10.1242/dev.150490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Prieto R, Pascual JM, Rosdolsky M, Castro-Dufourny I, Carrasco R, Strauss S, et al. Craniopharyngioma adherence: a comprehensive topographical categorization and outcome-related risk stratification model based on the methodical examination of 500 tumors. Neurosurg Focus. 2016 Dec;41((6)):E13. doi: 10.3171/2016.9.FOCUS16304. [DOI] [PubMed] [Google Scholar]
- 12.Bülow B, Attewell R, Hagmar L, Malmström P, Nordström CH, Erfurth EM. Postoperative prognosis in craniopharyngioma with respect to cardiovascular mortality, survival, and tumor recurrence. J Clin Endocrinol Metab. 1998 Nov;83((11)):3897–904. doi: 10.1210/jcem.83.11.5240. [DOI] [PubMed] [Google Scholar]
- 13.Tomlinson JW, Holden N, Hills RK, Wheatley K, Clayton RN, Bates AS, et al. West Midlands Prospective Hypopituitary Study Group Association between premature mortality and hypopituitarism. Lancet. 2001 Feb;357((9254)):425–31. doi: 10.1016/s0140-6736(00)04006-x. [DOI] [PubMed] [Google Scholar]
- 14.Pereira AM, Schmid EM, Schutte PJ, Voormolen JH, Biermasz NR, van Thiel SW, et al. High prevalence of long-term cardiovascular, neurological and psychosocial morbidity after treatment for craniopharyngioma. Clin Endocrinol (Oxf) 2005 Feb;62((2)):197–204. doi: 10.1111/j.1365-2265.2004.02196.x. [DOI] [PubMed] [Google Scholar]
- 15.Sterkenburg AS, Hoffmann A, Gebhardt U, Warmuth-Metz M, Daubenbüchel AM, Müller HL. Survival, hypothalamic obesity, and neuropsychological/psychosocial status after childhood-onset craniopharyngioma: newly reported long-term outcomes. Neuro-oncol. 2015 Jul;17((7)):1029–38. doi: 10.1093/neuonc/nov044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ordóñez-Rubiano EG, Forbes JA, Morgenstern PF, Arko L, Dobri GA, Greenfield JP, et al. Preserve or sacrifice the stalk? Endocrinological outcomes, extent of resection, and recurrence rates following endoscopic endonasal resection of craniopharyngiomas. J Neurosurg. 2018 Nov;:1–9. doi: 10.3171/2018.6.JNS18901. [DOI] [PubMed] [Google Scholar]
- 17.Erfurth EM, Holmer H, Fjalldal SB. Mortality and morbidity in adult craniopharyngioma. Pituitary. 2013 Mar;16((1)):46–55. doi: 10.1007/s11102-012-0428-2. [DOI] [PubMed] [Google Scholar]
- 18.Müller HL, Gebhardt U, Teske C, Faldum A, Zwiener I, Warmuth-Metz M, et al. Study Committee of KRANIOPHARYNGEOM 2000 Post-operative hypothalamic lesions and obesity in childhood craniopharyngioma: results of the multinational prospective trial KRANIOPHARYNGEOM 2000 after 3-year follow-up. Eur J Endocrinol. 2011 Jul;165((1)):17–24. doi: 10.1530/EJE-11-0158. [DOI] [PubMed] [Google Scholar]
- 19.Puget S, Garnett M, Wray A, Grill J, Habrand JL, Bodaert N, et al. Pediatric craniopharyngiomas: classification and treatment according to the degree of hypothalamic involvement. J Neurosurg. 2007 Jan;106((1 Suppl)):3–12. doi: 10.3171/ped.2007.106.1.3. [DOI] [PubMed] [Google Scholar]
- 20.Roth CL, Eslamy H, Werny D, Elfers C, Shaffer ML, Pihoker C, et al. Semiquantitative analysis of hypothalamic damage on MRI predicts risk for hypothalamic obesity. Obesity (Silver Spring) 2015 Jun;23((6)):1226–33. doi: 10.1002/oby.21067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fjalldal S, Follin C, Gabery S, Sundgren PC, Björkman-Burtscher IM, Lätt J, et al. Detailed assessment of hypothalamic damage in craniopharyngioma patients with obesity. Int J Obes. 2019 Mar;43((3)):533–44. doi: 10.1038/s41366-018-0185-z. [DOI] [PubMed] [Google Scholar]
- 22.Gabery S, Georgiou-Karistianis N, Lundh SH, Cheong RY, Churchyard A, Chua P, et al. Volumetric analysis of the hypothalamus in Huntington Disease using 3T MRI: the IMAGE-HD Study. PLoS One. 2015 Feb;10((2)):e0117593. doi: 10.1371/journal.pone.0117593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Holmer H, Ekman B, Björk J, Nordstöm CH, Popovic V, Siversson A, et al. Hypothalamic involvement predicts cardiovascular risk in adults with childhood onset craniopharyngioma on long-term GH therapy. Eur J Endocrinol. 2009 Nov;161((5)):671–9. doi: 10.1530/EJE-09-0449. [DOI] [PubMed] [Google Scholar]
- 24.Fjalldal S, Holmer H, Rylander L, Elfving M, Ekman B, Osterberg K, et al. Hypothalamic involvement predicts cognitive performance and psychosocial health in long-term survivors of childhood craniopharyngioma. J Clin Endocrinol Metab. 2013 Aug;98((8)):3253–62. doi: 10.1210/jc.2013-2000. [DOI] [PubMed] [Google Scholar]
- 25.Hochberg I, Hochberg Z. Expanding the definition of hypothalamic obesity [Review] Obes Rev. 2010 Oct;11((10)):709–21. doi: 10.1111/j.1467-789X.2010.00727.x. [DOI] [PubMed] [Google Scholar]
- 26.Coll AP, Farooqi IS, Challis BG, Yeo GS, O'Rahilly S. Proopiomelanocortin and energy balance: insights from human and murine genetics. J Clin Endocrinol Metab. 2004 Jun;89((6)):2557–62. doi: 10.1210/jc.2004-0428. [DOI] [PubMed] [Google Scholar]
- 27.Babinski J. Tumeur du corps pituitaire sans acromégalie et avec arrêt de développement des organes génitaux. Rev Neurol (Paris) 1900;8:531–3. [Google Scholar]
- 28.Frohlich A. Ein fall von tumor der hypophysis cerebri ohne akromegalie. Wiener Klin Rdsch. 1901;15:883–6. [Google Scholar]
- 29.Hetherington A, Ranson S. Hypothalamic lesions and adiposity in the rat. Anat Rec. 1940;78((2)):149–72. [Google Scholar]
- 30.Ahmet A, Blaser S, Stephens D, Guger S, Rutkas JT, Hamilton J. Weight gain in craniopharyngioma -a model for hypothalamic obesity. J Pediatr Endocrinol Metab. 2006 Feb;19((2)):121–7. doi: 10.1515/jpem.2006.19.2.121. [DOI] [PubMed] [Google Scholar]
- 31.Holmer H, Pozarek G, Wirfält E, Popovic V, Ekman B, Björk J, et al. Reduced energy expenditure and impaired feeding-related signals but not high energy intake reinforces hypothalamic obesity in adults with childhood onset craniopharyngioma. J Clin Endocrinol Metab. 2010 Dec;95((12)):5395–402. doi: 10.1210/jc.2010-0993. [DOI] [PubMed] [Google Scholar]
- 32.Bray GA, Inoue S, Nishizawa Y. Hypothalamic obesity: the autonomic hypothesis and the lateral hypothalamus. Diabetologia. 1981 Mar;20((S1 Suppl 1)):366–77. doi: 10.1007/BF00254505. [DOI] [PubMed] [Google Scholar]
- 33.Frohman LA, Bernardis LL. Growth hormone and insulin levels in weanling rats with ventromedial hypothalamic lesions. Endocrinology. 1968 Jun;82((6)):1125–32. doi: 10.1210/endo-82-6-1125. [DOI] [PubMed] [Google Scholar]
- 34.Lustig RH. Autonomic dysfunction of the beta-cell and the pathogenesis of obesity. Rev Endocr Metab Disord. 2003 Mar;4((1)):23–32. doi: 10.1023/a:1021819318484. [DOI] [PubMed] [Google Scholar]
- 35.King BM. The rise, fall, and resurrection of the ventromedial hypothalamus in the regulation of feeding behavior and body weight. Physiol Behav. 2006 Feb;87((2)):221–44. doi: 10.1016/j.physbeh.2005.10.007. [DOI] [PubMed] [Google Scholar]
- 36.Considine RV, Sinha MK, Heiman ML, Kriauciunas A, Stephens TW, Nyce MR, et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N Engl J Med. 1996 Feb;334((5)):292–5. doi: 10.1056/NEJM199602013340503. [DOI] [PubMed] [Google Scholar]
- 37.Shaikh MG, Grundy RG, Kirk JM. Reductions in basal metabolic rate and physical activity contribute to hypothalamic obesity. J Clin Endocrinol Metab. 2008 Jul;93((7)):2588–93. doi: 10.1210/jc.2007-2672. [DOI] [PubMed] [Google Scholar]
- 38.Stellar E. The physiology of motivation. Psychol Rev. 1954 Jan;61((1)):5–22. doi: 10.1037/h0060347. [DOI] [PubMed] [Google Scholar]
- 39.Schwartz MW, Woods SC, Porte D, Jr, Seeley RJ, Baskin DG. Central nervous system control of food intake. Nature. 2000 Apr;404((6778)):661–71. doi: 10.1038/35007534. [DOI] [PubMed] [Google Scholar]
- 40.Satoh N, Ogawa Y, Katsuura G, Hayase M, Tsuji T, Imagawa K, et al. The arcuate nucleus as a primary site of satiety effect of leptin in rats. Neurosci Lett. 1997 Mar;224((3)):149–52. doi: 10.1016/S0304-3940(97)00163-8. [DOI] [PubMed] [Google Scholar]
- 41.Tang-Christensen M, Holst JJ, Hartmann B, Vrang N. The arcuate nucleus is pivotal in mediating the anorectic effects of centrally administered leptin. Neuroreport. 1999 Apr;10((6)):1183–7. doi: 10.1097/00001756-199904260-00005. [DOI] [PubMed] [Google Scholar]
- 42.Dawson R, Pelleymounter MA, Millard WJ, Liu S, Eppler B. Attenuation of leptin-mediated effects by monosodium glutamate-induced arcuate nucleus damage. Am J Physiol. 1997 Jul;273((1 Pt 1)):E202–6. doi: 10.1152/ajpendo.1997.273.1.E202. [DOI] [PubMed] [Google Scholar]
- 43.Baskin DG, Breininger JF, Schwartz MW. Leptin receptor mRNA identifies a subpopulation of neuropeptide Y neurons activated by fasting in rat hypothalamus. Diabetes. 1999 Apr;48((4)):828–33. doi: 10.2337/diabetes.48.4.828. [DOI] [PubMed] [Google Scholar]
- 44.Cheung CC, Clifton DK, Steiner RA. Proopiomelanocortin neurons are direct targets for leptin in the hypothalamus. Endocrinology. 1997 Oct;138((10)):4489–92. doi: 10.1210/endo.138.10.5570. [DOI] [PubMed] [Google Scholar]
- 45.Kow LM, Pfaff DW. The effects of the TRH metabolite cyclo(His-Pro) and its analogs on feeding. Pharmacol Biochem Behav. 1991 Feb;38((2)):359–64. doi: 10.1016/0091-3057(91)90291-9. [DOI] [PubMed] [Google Scholar]
- 46.Müller HL, Handwerker G, Wollny B, Faldum A, Sörensen N. Melatonin secretion and increased daytime sleepiness in childhood craniopharyngioma patients. J Clin Endocrinol Metab. 2002 Aug;87((8)):3993–6. doi: 10.1210/jcem.87.8.8751. [DOI] [PubMed] [Google Scholar]
- 47.Karavitaki N, Cudlip S, Adams CB, Wass JA. Craniopharyngiomas. Endocr Rev. 2006 Jun;27((4)):371–97. doi: 10.1210/er.2006-0002. [DOI] [PubMed] [Google Scholar]
- 48.Harz KJ, Müller HL, Waldeck E, Pudel V, Roth C. Obesity in patients with craniopharyngioma: assessment of food intake and movement counts indicating physical activity. J Clin Endocrinol Metab. 2003 Nov;88((11)):5227–31. doi: 10.1210/jc.2002-021797. [DOI] [PubMed] [Google Scholar]
- 49.Mori M, Takeshima H, Kuratsu J. Expression of interleukin-6 in human craniopharyngiomas: a possible inducer of tumor-associated inflammation. Int J Mol Med. 2004 Oct;14((4)):505–9. [PubMed] [Google Scholar]
- 50.Mutlu LK, Woiciechowsky C, Bechmann I. Inflammatory response after neurosurgery. Best Pract Res Clin Anaesthesiol. 2004 Sep;18((3)):407–24. doi: 10.1016/j.bpa.2003.12.003. [DOI] [PubMed] [Google Scholar]
- 51.Thaler JP, Yi CX, Schur EA, Guyenet SJ, Hwang BH, Dietrich MO, et al. Obesity is associated with hypothalamic injury in rodents and humans. J Clin Invest. 2012 Jan;122((1)):153–62. doi: 10.1172/JCI59660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lindroos AK, Lissner L, Sjöström L. Validity and reproducibility of a self-administered dietary questionnaire in obese and non-obese subjects. Eur J Clin Nutr. 1993 Jul;47((7)):461–81. [PubMed] [Google Scholar]
- 53.Stunkard AJ, Messick S. The three-factor eating questionnaire to measure dietary restraint, disinhibition and hunger. J Psychosom Res. 1985;29((1)):71–83. doi: 10.1016/0022-3999(85)90010-8. [DOI] [PubMed] [Google Scholar]
- 54.Crowley RK, Hamnvik OP, O'Sullivan EP, Behan LA, Smith D, Agha A, et al. Morbidity and mortality in patients with craniopharyngioma after surgery. Clin Endocrinol (Oxf) 2010 Oct;73((4)):516–21. doi: 10.1111/j.1365-2265.2010.03838.x. [DOI] [PubMed] [Google Scholar]
- 55.Fleseriu M, Hashim IA, Karavitaki N, Melmed S, Murad MH, Salvatori R, et al. Hormonal Replacement in Hypopituitarism in Adults: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2016 Nov;101((11)):3888–921. doi: 10.1210/jc.2016-2118. [DOI] [PubMed] [Google Scholar]
- 56.Müller HL, Heinrich M, Bueb K, Etavard-Gorris N, Gebhardt U, Kolb R, et al. Perioperative dexamethasone treatment in childhood craniopharyngioma -influence on short-term and long-term weight gain. Exp Clin Endocrinol Diabetes. 2003 Sep;111((6)):330–4. doi: 10.1055/s-2003-42722. [DOI] [PubMed] [Google Scholar]
- 57.Geffner M, Lundberg M, Koltowska-Häggström M, Abs R, Verhelst J, Erfurth EM, et al. Changes in height, weight, and body mass index in children with craniopharyngioma after three years of growth hormone therapy: analysis of KIGS (Pfizer International Growth Database) J Clin Endocrinol Metab. 2004 Nov;89((11)):5435–40. doi: 10.1210/jc.2004-0667. [DOI] [PubMed] [Google Scholar]
- 58.Karavitaki N, Warner JT, Marland A, Shine B, Ryan F, Arnold J, et al. GH replacement does not increase the risk of recurrence in patients with craniopharyngioma. Clin Endocrinol (Oxf) 2006 May;64((5)):556–60. doi: 10.1111/j.1365-2265.2006.02508.x. [DOI] [PubMed] [Google Scholar]
- 59.Olsson DS, Buchfelder M, Wiendieck K, Kremenevskaja N, Bengtsson BÅ, Jakobsson KE, et al. Tumour recurrence and enlargement in patients with craniopharyngioma with and without GH replacement therapy during more than 10 years of follow-up. Eur J Endocrinol. 2012 Jun;166((6)):1061–8. doi: 10.1530/EJE-12-0077. [DOI] [PubMed] [Google Scholar]
- 60.Lustig RH, Hinds PS, Ringwald-Smith K, Christensen RK, Kaste SC, Schreiber RE, et al. Octreotide therapy of pediatric hypothalamic obesity: a double-blind, placebo-controlled trial. J Clin Endocrinol Metab. 2003 Jun;88((6)):2586–92. doi: 10.1210/jc.2002-030003. [DOI] [PubMed] [Google Scholar]
- 61.Goldfield GS, Lorello C, Doucet E. Methylphenidate reduces energy intake and dietary fat intake in adults: a mechanism of reduced reinforcing value of food? Am J Clin Nutr. 2007 Aug;86((2)):308–15. doi: 10.1093/ajcn/86.2.308. [DOI] [PubMed] [Google Scholar]
- 62.Sokol MS, Gray NS, Goldstein A, Kaye WH. Methylphenidate treatment for bulimia nervosa associated with a cluster B personality disorder. Int J Eat Disord. 1999 Mar;25((2)):233–7. doi: 10.1002/(sici)1098-108x(199903)25:2<233::aid-eat14>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 63.Elfers CT, Roth CL, Elfers CT. Effects of methylphenidate on weight gain and food intake in hypothalamic obesity. Front Endocrinol (Lausanne) 2011 Dec;2:78. doi: 10.3389/fendo.2011.00078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kanoski SE, Hayes MR, Skibicka KP. GLP-1 and weight loss: unraveling the diverse neural circuitry. Am J Physiol Regul Integr Comp Physiol. 2016 May;310((10)):R885–95. doi: 10.1152/ajpregu.00520.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Secher A, Jelsing J, Baquero AF, Hecksher-Sørensen J, Cowley MA, Dalbøge LS, et al. The arcuate nucleus mediates GLP-1 receptor agonist liraglutide-dependent weight loss. J Clin Invest. 2014 Oct;124((10)):4473–88. doi: 10.1172/JCI75276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Thondam SK, Cuthbertson DJ, Aditya BS, Macfarlane IA, Wilding JP, Daousi C. A glucagon-like peptide-1 (GLP-1) receptor agonist in the treatment for hypothalamic obesity complicated by type 2 diabetes mellitus. Clin Endocrinol (Oxf) 2012 Oct;77((4)):635–7. doi: 10.1111/j.1365-2265.2012.04368.x. [DOI] [PubMed] [Google Scholar]
- 67.Simmons JH, Shoemaker AH, Roth CL. Treatment with glucagon-like Peptide-1 agonist exendin-4 in a patient with hypothalamic obesity secondary to intracranial tumor. Horm Res Paediatr. 2012;78((1)):54–8. doi: 10.1159/000339469. [DOI] [PubMed] [Google Scholar]
- 68.Zoicas F, Droste M, Mayr B, Buchfelder M, Schöfl C. GLP-1 analogues as a new treatment option for hypothalamic obesity in adults: report of nine cases. Eur J Endocrinol. 2013 Apr;168((5)):699–706. doi: 10.1530/EJE-12-0997. [DOI] [PubMed] [Google Scholar]
- 69.Fothergill E, Guo J, Howard L, Kerns JC, Knuth ND, Brychta R, et al. Persistent metabolic adaptation 6 years after “The Biggest Loser” competition. Obesity (Silver Spring) 2016 Aug;24((8)):1612–9. doi: 10.1002/oby.21538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.O'Neil PM, Birkenfeld AL, McGowan B, Mosenzon O, Pedersen SD, Wharton S, et al. Efficacy and safety of semaglutide compared with liraglutide and placebo for weight loss in patients with obesity: a randomised, double-blind, placebo and active controlled, dose-ranging, phase 2 trial. Lancet. 2018 Aug;392((10148)):637–49. doi: 10.1016/S0140-6736(18)31773-2. [DOI] [PubMed] [Google Scholar]
- 71.Inge TH, Pfluger P, Zeller M, Rose SR, Burget L, Sundararajan S, et al. Gastric bypass surgery for treatment of hypothalamic obesity after craniopharyngioma therapy. Nat Clin Pract Endocrinol Metab. 2007 Aug;3((8)):606–9. doi: 10.1038/ncpendmet0579. [DOI] [PubMed] [Google Scholar]
- 72.Müller HL, Gebhardt U, Maroske J, Hanisch E. Long-term follow-up of morbidly obese patients with childhood craniopharyngioma after laparoscopic adjustable gastric banding (LAGB) Klin Padiatr. 2011 Nov;223((6)):372–3. doi: 10.1055/s-0031-1284420. [DOI] [PubMed] [Google Scholar]
- 73.Bretault M, Laroche S, Lacorte JM, Barsamian C, Polak M, Raffin-Sanson ML, et al. Postprandial GLP-1 Secretion After Bariatric Surgery in Three Cases of Severe Obesity Related to Craniopharyngiomas. Obes Surg. 2016 May;26((5)):1133–7. doi: 10.1007/s11695-015-1977-z. [DOI] [PubMed] [Google Scholar]
- 74.Bretault M, Boillot A, Muzard L, Poitou C, Oppert JM, Barsamian C, et al. Clinical review: Bariatric surgery following treatment for craniopharyngioma: a systematic review and individual-level data meta-analysis. J Clin Endocrinol Metab. 2013 Jun;98((6)):2239–46. doi: 10.1210/jc.2012-4184. [DOI] [PubMed] [Google Scholar]
- 75.Wijnen M, Olsson DS, van den Heuvel-Eibrink MM, Wallenius V, Janssen JA, Delhanty PJ, et al. Efficacy and safety of bariatric surgery for craniopharyngioma-related hypothalamic obesity: a matched case-control study with 2 years of follow-up. Int J Obes. 2017 Feb;41((2)):210–6. doi: 10.1038/ijo.2016.195. [DOI] [PubMed] [Google Scholar]
- 76.Zada G, Kintz N, Pulido M, Amezcua L. Prevalence of neurobehavioral, social, and emotional dysfunction in patients treated for childhood craniopharyngioma: a systematic literature review. PLoS One. 2013 Nov;8((11)):e76562. doi: 10.1371/journal.pone.0076562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Dekkers OM, Biermasz NR, Smit JW, Groot LE, Roelfsema F, Romijn JA, et al. Quality of life in treated adult craniopharyngioma patients. Eur J Endocrinol. 2006 Mar;154((3)):483–9. doi: 10.1530/eje.1.02114. [DOI] [PubMed] [Google Scholar]
- 78.Müller HL, Bruhnken G, Emser A, Faldum A, Etavard-Gorris N, Gebhardt U, et al. Longitudinal study on quality of life in 102 survivors of childhood craniopharyngioma. Childs Nerv Syst. 2005 Nov;21((11)):975–80. doi: 10.1007/s00381-004-1124-y. [DOI] [PubMed] [Google Scholar]
- 79.Mason PW, Krawiecki N, Meacham LR. The use of dextroamphetamine to treat obesity and hyperphagia in children treated for craniopharyngioma. Arch Pediatr Adolesc Med. 2002 Sep;156((9)):887–92. doi: 10.1001/archpedi.156.9.887. [DOI] [PubMed] [Google Scholar]
- 80.Ismail D, O'Connell MA, Zacharin MR. Dexamphetamine use for management of obesity and hypersomnolence following hypothalamic injury. J Pediatr Endocrinol Metab. 2006 Feb;19((2)):129–34. doi: 10.1515/jpem.2006.19.2.129. [DOI] [PubMed] [Google Scholar]
- 81.Fjalldal S, Follin C, Svärd D, Rylander L, Gabery S, Petersén Å, et al. Microstructural white matter alterations and hippocampal volumes are associated with cognitive deficits in craniopharyngioma. Eur J Endocrinol. 2018 Jun;178((6)):577–87. doi: 10.1530/EJE-18-0081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Özyurt J, Müller HL, Thiel CM. A systematic review of cognitive performance in patients with childhood craniopharyngioma. J Neurooncol. 2015 Oct;125((1)):9–21. doi: 10.1007/s11060-015-1885-z. [DOI] [PubMed] [Google Scholar]
- 83.Aggleton JP. Multiple anatomical systems embedded within the primate medial temporal lobe: implications for hippocampal function. Neurosci Biobehav Rev. 2012 Aug;36((7)):1579–96. doi: 10.1016/j.neubiorev.2011.09.005. [DOI] [PubMed] [Google Scholar]
- 84.Preston AR, Eichenbaum H. Interplay of hippocampus and prefrontal cortex in memory. Curr Biol. 2013 Sep;23((17)):R764–73. doi: 10.1016/j.cub.2013.05.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Vargha-Khadem F, Gadian DG, Watkins KE, Connelly A, Van Paesschen W, Mishkin M. Differential effects of early hippocampal pathology on episodic and semantic memory. Science. 1997 Jul;277((5324)):376–80. doi: 10.1126/science.277.5324.376. [DOI] [PubMed] [Google Scholar]
- 86.Johansen-Berg H, Rushworth MF. Using diffusion imaging to study human connectional anatomy. Annu Rev Neurosci. 2009;32((1)):75–94. doi: 10.1146/annurev.neuro.051508.135735. [DOI] [PubMed] [Google Scholar]
- 87.Pierpaoli C, Jezzard P, Basser PJ, Barnett A, Di Chiro G. Diffusion tensor MR imaging of the human brain. Radiology. 1996 Dec;201((3)):637–48. doi: 10.1148/radiology.201.3.8939209. [DOI] [PubMed] [Google Scholar]
- 88.Koenig KA, Sakaie KE, Lowe MJ, Lin J, Stone L, Bermel RA, et al. The relationship between cognitive function and high-resolution diffusion tensor MRI of the cingulum bundle in multiple sclerosis. Mult Scler. 2015 Dec;21((14)):1794–801. doi: 10.1177/1352458515576983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Lin YC, Shih YC, Tseng WY, Chu YH, Wu MT, Chen TF, et al. Cingulum correlates of cognitive functions in patients with mild cognitive impairment and early Alzheimer's disease: a diffusion spectrum imaging study. Brain Topogr. 2014 May;27((3)):393–402. doi: 10.1007/s10548-013-0346-2. [DOI] [PubMed] [Google Scholar]
- 90.Olson IR, Von Der Heide RJ, Alm KH, Vyas G. Development of the uncinate fasciculus: implications for theory and developmental disorders. Dev Cogn Neurosci. 2015 Aug;14:50–61. doi: 10.1016/j.dcn.2015.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Douet V, Chang L. Fornix as an imaging marker for episodic memory deficits in healthy aging and in various neurological disorders. Front Aging Neurosci. 2015 Jan;6:343. doi: 10.3389/fnagi.2014.00343. [DOI] [PMC free article] [PubMed] [Google Scholar]