Abstract
Background
Type 2 diabetes (T2D) is characterized by β-cell dysfunction and insulin resistance. Icariin (ICA), a flavonoid from Epimedium, possesses anti-diabetic and anti-inflammatory properties. However, it is unclear whether ICA acts on pancreatic β-cells. The present study was designed to explore the effects and latent mechanism of ICA on uric acid (UA)-stimulated pancreatic β-cell dysfunction.
Material/Methods
Min6 cells were exposed to various concentrations of ICA for 24 h, and cell viability was assessed by MTT assays. Min6 cells were treated with ICA for 2 h, followed by 5 mg/dl UA for 24 h, and cell viability, apoptosis, apoptosis-associated protein levels and insulin secretion were assessed by MTT, flow cytometry, western blotting and glucose-stimulated insulin secretion assays, respectively. The effects of ICA and UA on the PI3K/Akt pathway were also analyzed by western blotting, as were the effects of the specific PI3K/Akt inhibitor LY294002.
Results
ICA was not cytotoxic toward Min6 cells. UA decreased Min6 cell viability, enhanced cell apoptosis and levels of cleaved caspase-3, and reduced pro-caspase3 levels and insulin secretion, with all of these effects reversed by ICA in a dose-dependent manner. UA inhibited the PI3K/AKT pathway, an effect reversed by ICA treatment. The specific PI3K/Akt inhibitor LY294002, however, reversed these effects of ICA on UA-treated Min6 cells.
Conclusions
ICA protected Min6 cell function, an effect likely mediated by the PI3K pathway. ICA may inhibit the progression of diabetes.
MeSH Keywords: Diabetes Mellitus, Insulin-Secreting Cells, Phosphatidylinositol 3-Kinases
Background
Diabetes mellitus (DM), one of the most frequent endocrine diseases, is characterized by high blood sugar levels. The incidence of type 2 diabetes (T2D), the most frequent form of this disease, has increased worldwide, due to changes in diet and minimal exercise [1,2]. T2D is an immediate cause of pancreatic β-cell dysfunction, which may reduce insulin secretion and shorten lifespan [3]. T2D is characterized by insulin tolerance and pancreatic β-cell injury, emphasizing the importance of treatments that protect pancreatic β-cell function and/or block the progression of disease [4]. Increased uric acid (UA) levels may led to pancreatic β-cell dysfunction, reducing insulin release and enhancing the risk of T2D [5]. However, the mechanism underlying UA-induced pancreatic β-cell dysfunction has not been fully explored.
Epimedium is a common traditional herbal medicine, with icariin (ICA), the major constituent of Epimedii herbaa, being responsible for the pharmacological activities of epimedium [6]. ICA has been shown to prevent premature ovarian failure [7], as well as having anti-inflammatory [8] and anti-tumor [9] properties. Furthermore, ICA was shown to block cytokine-induced β-cell death by inactivation of NF-kappaB signaling [10] and to inhibit insulin resistance in C2C12 mouse muscle cells by activation of the AMPK pathway [11]. However, it is unclear whether ICA can suppress UA-induced pancreatic β-cell dysfunction or enhance β-cell function.
The PI3K/AKT pathway, the downstream target of the Wnt signaling pathway, has been reported to display vital roles in many diseases. For example, down-regulation of miRNA-26a-5p was found to promote endothelial cell apoptosis by blocking the PI3K/AKT pathway in coronary heart disease [12]. The PI3K/AKT pathway may also be involved in the development of insulin resistance, and ICA may affect this pathway.
The present study was designed to explore the effects of ICA on UA-treated pancreatic β-cells, as well as to evaluate the possible mechanisms of action of ICA in diabetes. We therefore tested the effects of ICA on cell viability, apoptosis and insulin release in UA-treated pancreatic β-cells. The involvement of the PI3K/AKT pathway was analyzed by testing the effects of LY294002, a specific inhibitor of the PI3K/AKT pathway. The results of this study suggest that ICA may be effective clinically in blocking the progression of T2D.
Material and Methods
Cell culture
The mouse insulinoma cell line Min6 was purchased from the American Type Culture Collection (ATCC; Manassas, VA, USA) and cultured in Dulbecco’s modified Eagle’s medium (DMEM; Hyclone; Marlborough, MA, USA), supplemented with 15% fetal bovine serum (FBS; Gibco, Waltham, MA, USA), 100 U/ml penicillin and streptomycin (Invitrogen, Carlsbad, CA, USA), in an incubator with 5% CO2 at 37°C. UA solutions were prepared in pre-warmed cell culture medium and passed through a 2.2 μm sterile filter. Min6 cells were pre-treated with various concentrations of ICA (0, 5, 10, 20 μM; dissolved in fresh medium) for 2 h, and 5 mg/dl UA for another 24 h. For PI3K/AKT pathway inhibition, cells were cultivated with 10 mM LY294002 (dissolved in DMSO; Cell Signaling Technology, Danvers, MA, USA) for 2 h before UA treatment.
MTT assay
Min6 cell viability was determined using MTT assays. Briefly, cells (5×104 cells per well) were exposed to ICA, UA or LY294002. A 20 μl aliquot of MTT solution was added to each sample, and the samples were cultured for 4 h. After incubation, 100 μl DMSO was added to each well and the samples were incubated in the dark for 10 min. The absorbance of each well at 570 nm was determined using a micro-plate reader (Bio-Rad, Hercules, CA, USA). Each experiment was repeated three times, all on cells of the same passage number.
Flow cytometry analysis
To assess cell apoptosis, Min6 cells were incubated with ICA, UA or LY294002 for the indicated times. The cells were subsequently washed, centrifuged and re-suspended, and aliquots of 106 cells were exposed in Annexin V-FITC and PI solutions for 15 min in the dark, according to the manufacturer’s instructions. Apoptotic cells were quantified by flow cytometry (BD Biosciences, Franklin Lakes, NJ, USA), and the results were analyzed using FlowJo software. Q1 (AnnexinV-FITC)−/PI+ contained dead cells; Q2 (AnnexinV+FITC)+/PI+ contained late apoptotic cells; Q3 (AnnexinV-FITC)+/PI− contained early apoptotic cells; and Q4 (AnnexinV-FITC)−/PI− contained live cells. Each experiment was repeated three times, all on cells of the same passage number.
Glucose-stimulated insulin secretion (GSIS) assay
Min6 cells (5×104 cells per well) were incubated with various concentrations of ICA for 2 h, followed by incubation with 5 mg/dl UA for an additional 4 h. The cells were subsequently incubated with 0.1% BSA (Sigma Aldrich; St. Louis, MO, USA) in KRB buffer (Sigma Aldrich) for 1 h, followed by incubation with KRB buffer containing low or high concentrations of glucose for another 1 h. Insulin secretion was measured by radioimmunoassays, as described previously. Each experiment was repeated three times, all on cells of the same passage number.
qRT-PCR analysis
Min6 cells (5×104 cells per well) were incubated with ICA, UA or LY294002 for the indicated times. Total RNA was prepared using Trizol reagent (Invitrogen) and reverse transcribed to cDNA using PrimeScript® RT reagent kits (Vazyme Beyotime; Nanjing, China), according to the manufacturers’ instructions. qRT-PCR was performed on a Real-Time PCR System (Applied Biosystems, Foster City, CA, USA) with SYBR Green Master mix (TaKaRa; Tokyo, Japan) and primers for
-
PI3K (forward, 5′-AGTACCTTGTTCCAATCCCA-3′;
reverse, 5′-GTTCCTCTTTAGCACCCTTTC-3′),
-
AKT (forward, 5′-CGTCGGAGACTGACACCA-3′;
reverse, 5′-GCTGGCCGAGTAGGAGAA-3′) and
-
GAPDH (forward, 5′-AAGGTCGGAGTCAACGGA-3′;
reverse, 5′-TTAAAAGCAGCCCTGGTGA-3′).
The amplification protocol consisted of an initial denaturation for 10 min at 95°C, followed by 35 cycles of denaturation at 95°C for 60 sec, annealing at 60˚C for 60 sec, and extension at 72°C for 15 sec; and a final extension at 72°C for 10 min. Levels of expression were calculated by the 2−ΔΔCq method, with the expression of PI3K and AKT normalized to that of GAPDH. Each experiment was repeated three times, all on cells of the same passage number.
Western blot analysis
Total protein was collected from Min6 cells (5×104 cells per well) lysed with RIPA lysis buffer (Vazyme Beyotime), and protein concentrations determined using BCA™ Protein Assay Kits (Solarbio; Beijing, China). Aliquots of 40 μg of protein were loaded onto each lane, and the proteins separated by 10% SDS-PAGE and transferred to PVDF membranes. The membranes were incubated with 5% skim milk and immunoblotted with primary anti-p-PI3K, anti-PI3K, anti-p-AKT, anti-AKT, anti-cleaved caspase-3, anti-pro caspase-3 and anti-GAPDH (Abcam, Cambridge, UK) antibodies overnight at 4°C, following by incubation with the respective secondary antibodies (Abcam) for 1 h at room temperature. The protein bands were visualized using ECL Kits (BestBio, Shanghai, China) and quantified with Image J Software (Bio-Rad, Shanghai, China). Each experiment was performed three times, all on cells of the same passage number.
Statistical analysis
Data are presented as mean±SD and compared by Student’s t-test or one-way ANOVA followed by Student-Newman-Keuls tests. All statistical analyses were performed using GraphPad Prism 6.0 software, with P<0.05 defined as statistically significant.
Results
Icariin does not affect Min6 cell viability
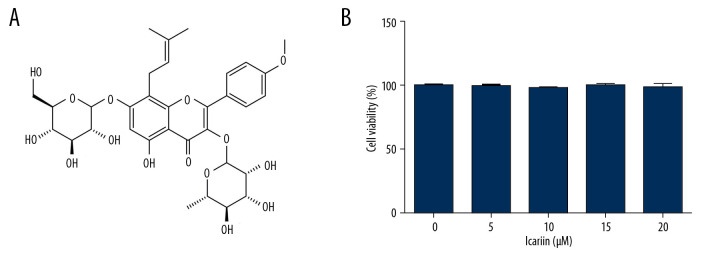
To evaluate the effects of ICA (Figure 1A) on pancreatic β-cells, Min6 cells were incubated with 0, 5, 10, 20, and 40 μM ICA for 24 h, and cell viability was assessed using MTT assays. ICA did not affect Min6 cell viability (Figure 1B), suggesting that ICA is not cytotoxic to Min6 cells.
Figure 1.
Effects of various concentration of icariin on the viability of Min6 cells. (A) Chemical structure of icariin. (B) Effect of icariin on cell viability. Min6 cells were treated with various concentration of icariin (0, 5, 10, 20, 40 μM) for 24 h, and their viability evaluated by MTT assays.
Icariin protects Min6 cells from UA-induced cell damage
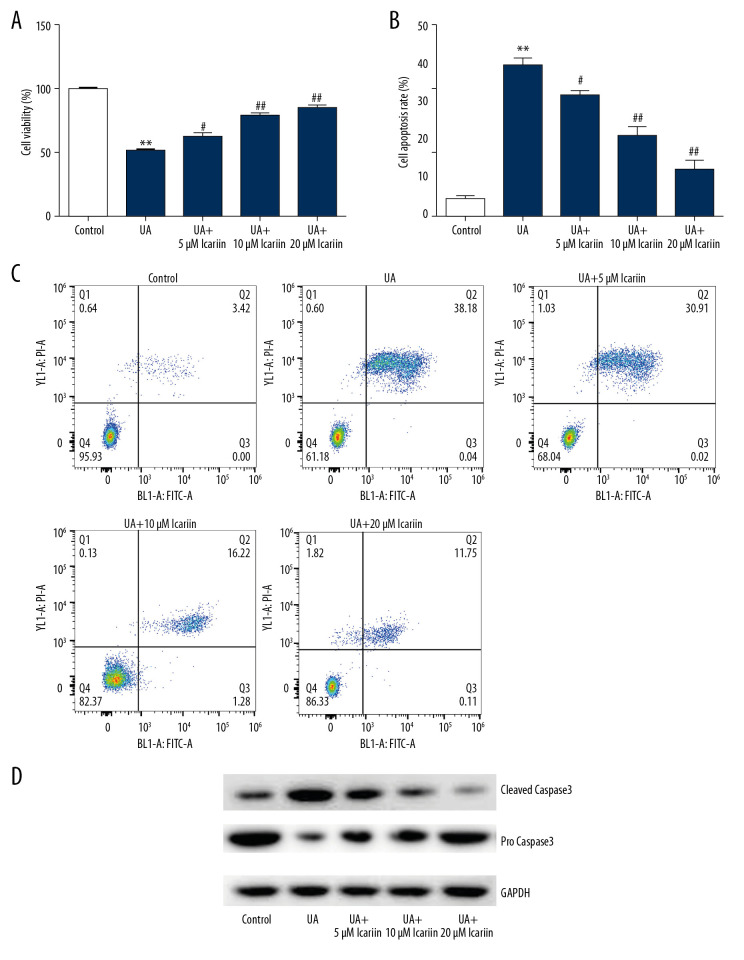
To evaluate the effects of ICA on UA-treated pancreatic β-cells, Min6 cells were incubated with 0, 5, 10, and 20 μM ICA for 2 h, and then with 5 mg/dl UA for an additional 24 h, and the effects of ICA and UA on cell viability, apoptosis and apoptosis-associated proteins were determined using MTT, flow cytometry and western blotting, respectively. UA treatment markedly reduced cell viability, as shown by MTT assays (Figure 2A), while increasing the numbers of apoptotic cells, as shown by flow cytometry (Figure 2B, 2C). Moreover, UA treatment enhanced the levels of cleaved caspase-3 protein while reducing the levels of pro-caspase-3 (Figure 2D). All of these effects, however, were reversed by ICA treatment in a dose-dependent manner.
Figure 2.
Icariin reversed the effects of UA on Min6 cell viability and apoptosis. Min6 cells were treated with various concentration of icariin (0, 5, 10, 20 μM) for 2 h, followed by treatment with 5 mg/dl UA for another 24 h. (A) Cell viability and (B) apoptosis were assessed using MTT and flow cytometry analysis, respectively. (C) Quantity of apoptotic cells. (D) Protein levels of cleaved caspase-3 and pro-caspase-3 were examined using western blotting. ** P<0.01 compared with the control group; # P<0.05, ## P <0.01 compared with the UA group.
Icariin enhances insulin secretion by glucose-treated Min6 cells
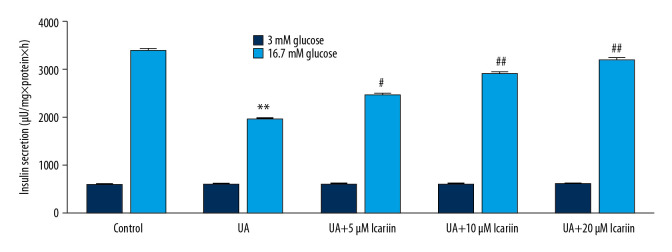
To further assess the effects of ICA on UA-induced Min6 cell dysfunction, Min6 cells were incubated with 0, 5, 10, and 20 μM ICA for 2 h, with 5 mg/dl UA for 24 h, and with 3 mM or 16.7 mM glucose for another 1 h. UA suppressed insulin production in response to stimulation with 16.7 mM glucose, whereas ICA significantly and dose-dependently increased high glucose-induced insulin release compared with UA alone (Figure 3). These findings suggested that ICA protected pancreatic β-cells from UA-induced injury.
Figure 3.
Icariin improved glucose-stimulated insulin secretion in Min6 cells. Min6 cells were treated with icariin for 2 h and 5 mg/dl UA for another 4 h, followed by stimulation with low or high concentrations of glucose for another 1 h. Cells were divided into six groups: control, UA, UA+5 μM icariin, UA+10 μM icariin, UA+20 μM icariin. Insulin concentrations were measured using the GSIS assay. ** P<0.01 compared with the control group; # P<0.05, ## P<0.01 compared with the UA group.
Icariin protects cells from UA-induced injury by regulating the PI3K/AKT signal pathway
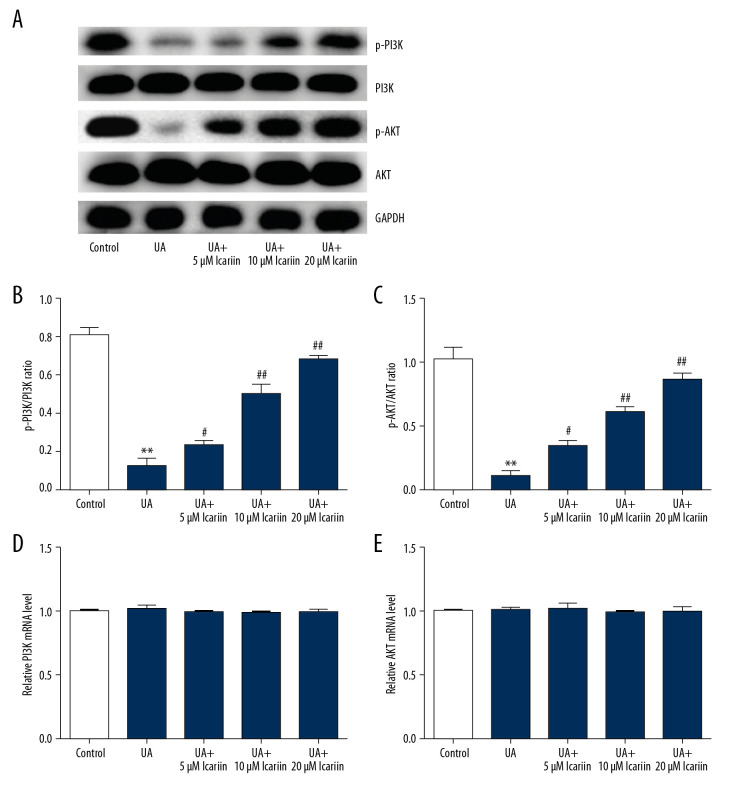
To assess the specific mechanism of action of ICA, its roles in the PI3K/AKT pathway were investigated. UA treatment markedly reduced p-PI3K and p-AKT levels, but this suppression was overcome by treatment with ICA (Figure 4A). In addition, the p-PI3K/PI3K (Figure 4B) and p-AKT/AKT (Figure 4C) ratios were markedly lower in cells treated with UA alone than in control cells, but were enhanced by ICA in a dose-dependent manner. In contrast, there were no differences in levels of PI3K (Figure 4D) and AKT (Figure 4E) mRNAs. These findings indicate that the PI3K/AKT signal pathway may participate in the regulation of ICA in UA-treated Min6 cells.
Figure 4.
Effects of icariin on PI3K/AKT signal pathway in UA-induced cell injury. Min6 cells were treated with icariin, UA and/or glucose for the indicated times. Cells were divided into six groups: control, UA, UA+5 μM icariin, UA+10 μM icariin, UA+20 μM icariin. (A) P-PI3K and p-AKT levels measured by western blotting. (B) p-PI3K/PI3K and (C) p-AKT/AKT ratios. Levels of (D) PI3K and (E) AKT mRNAs determined by qRT-PCR. ** P<0.01 compared with the control group; # P<0.05, ## P<0.01 compared with the UA group.
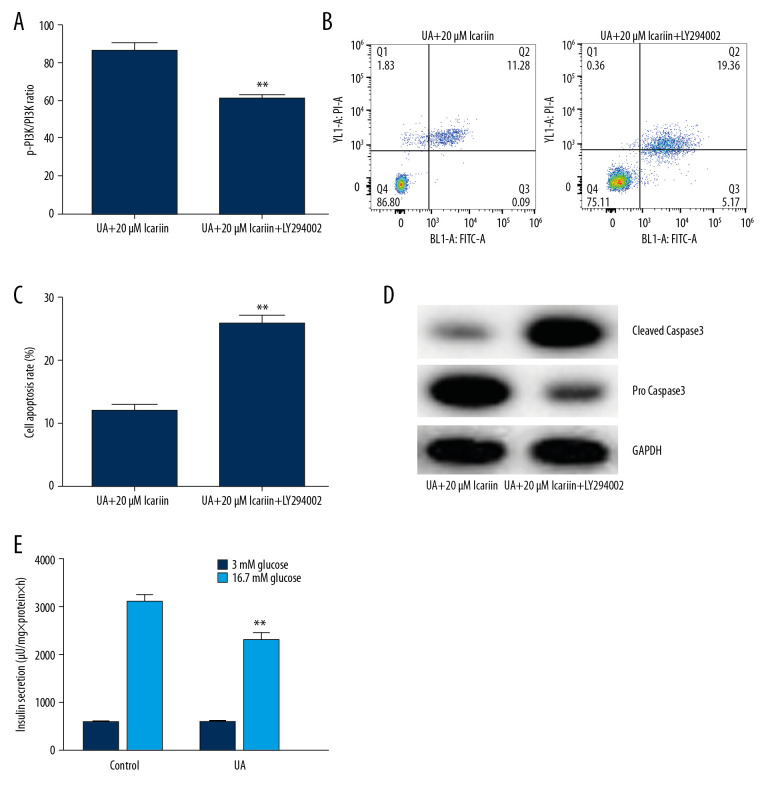
Effects of LY294002 on the viability, apoptosis and insulin secretion of Min6 cells treated with UA and ICA
To determine the specific roles of the PI3K/AKT signal pathway in UA-induced apoptosis, Min6 cells were treated with the PI3K/Akt inhibitor LY294002, followed by treatment with 20 μM ICA for 2 h and 5 mg/dl UA for an additional 24 h. Cell viability, apoptosis and apoptosis-related proteins were measured using MTT assays, flow cytometry and western blotting, respectively. LY294002 reversed the effects of ICA on UA-induced cytotoxicity (Figure 5A) and apoptosis (Figure 5B, 5C), as well as enhancing the expression of cleaved caspase-3 and reducing the expression of pro-caspase-3 (Figure 5D). Moreover, GSIS assays showed that LY294002 significantly reduced the secretion of insulin by glucose-treated Min6 cells (Figure 5E). Taken together, these findings indicated that LY294002 reversed the protective effects of ICA on UA-treated Min6 cells, suggesting that the anti-apoptotic activity of ICA may be regulated by the PI3K/Akt pathway.
Figure 5.
LY294002 reversed the protective effect of icariin in UA-induced Min6 cell injury. Min6 cells were treated with LY294002, UA and/or icariin for the indicated times. Cells were divided into two groups: UA+20 μM icariin and UA+20 μM icariin+LY294002. (A) Cell viability and (B) apoptosis were assessed using MTT and flow cytometry analysis, respectively. (C) Cell apoptosis. (D) Protein levels of cleaved caspase-3 and pro-caspase-3 measured by western blotting. (E) Secretion of insulin quantified by GSIS. ** P<0.01 compared with the control group; # P<0.05, ## P<0.01 compared with the UA group.
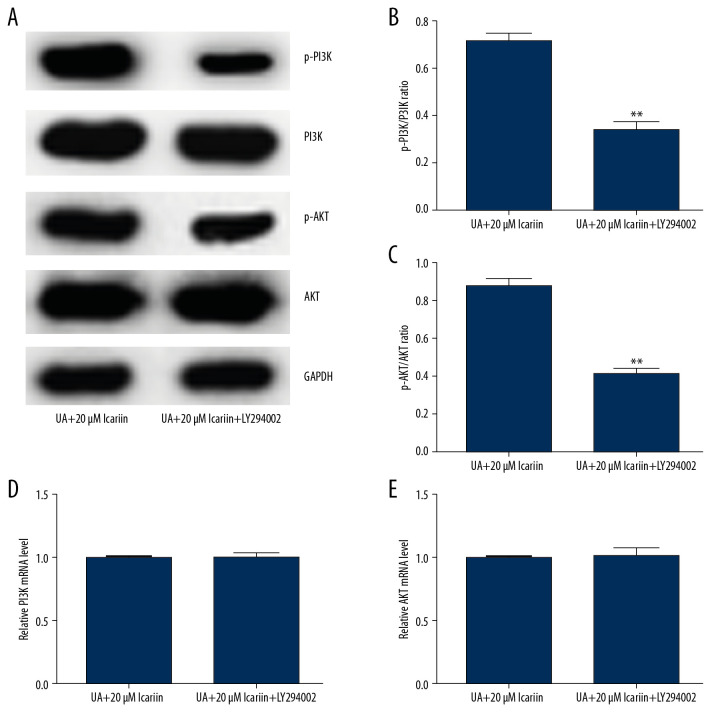
Icariin protects pancreatic β-cell function by activating the PI3K/AKT pathway in UA-treated Min6 cells
Because the PI3K/Akt pathway plays a vital role in regulating insulin secretion by pancreatic β-cells, we assessed whether ICA protected pancreatic β-cells via the PI3K/Akt pathway. Western blotting showed that LY294002 markedly reduced the levels of p-PI3K and p-AKT in Min6 cells treated with 20 μM ICA and UA (Figure 6A), while also reducing the p-PI3K/PI3K and p-AKT/AKT ratios (Figure 6B, 6C). In contrast, PI3K (Figure 6D) and AKT (Figure 6E) mRNA levels did not differ among these groups of cells. These results suggested that ICA protected pancreatic β-cell function by activating the PI3K/AKT pathway in UA-stimulated Min6 cells.
Figure 6.
LY294002 reversed the effect of icariin on the PI3K/AKT pathway in UA-treated Min6 cells. Min6 cells were treated with LY294002, UA and/or icariin for the indicated times. Cells were divided into two groups: UA+20 μM icariin and UA+20 μM icariin+LY294002. (A) Expression of P-PI3K and p-AKT proteins by western blotting. (B) p-PI3K/PI3K and (C) p-AKT/AKT ratios. Levels of (D) PI3K and (E) AKT mRNAs determined by qRT-PCR. ** P<0.01 compared with the UA+20 μM icariin group.
Discussion
T2D is a common disease, which results from injury to β-cells, impairing insulin release [13]. ICA, a compound in Epimedii herbaa, has been shown to prevent estrogen deficiency-induced alveolar bone loss via the STAT3 pathway [14]. Moreover, ICA was shown to affect a perimenopausal depression-like rat model by activating the PI3K-AKT signaling pathway [15] and to protect PC12 cells from oxygen-glucose deprivation-induced injury [16]. Based on these findings, we investigated whether ICA could effectively protect Min6 cells against UA induced injury.
To assess the function of ICA in pancreatic β-cells, Min6 cells were cultured in the presence of various concentrations of ICA (0, 5, 10, 20, 40 μM) for 24 h. We found that ICA did not affect Min6 cell viability, indicating that ICA did not have a cytotoxic effect on β-cells. This assay, however, did not include a positive control, making this a limitation of this study. UA, a product of purine metabolism, is highly associated with diabetes mellitus [17]. Over-expression of UA was shown to be related to metabolic syndrome, which, in turn, is associated with the development and progression of T2D [18]. UA was found to stimulate oxidative stress and inhibit the proliferation of pancreatic β-cells by activating adenosine monophosphate-activated protein kinase [19]. To explore the activity of ICA in UA-treated pancreatic β-cells, Min6 cells were treated with various concentration of ICA (0, 5, 10, 20, 40 μM) for 2 h, and then with 5 mg/dl UA for another 24 h to induce cell injury and dysfunction. We found that UA markedly inhibited Min6 cell viability and increased the numbers of apoptotic cells, indicating that UA promoted Min6 cell injury and dysfunction. Because cleaved caspase-3 and pro-caspase-3 are regulators of cell apoptosis [20], we assessed their levels of expression in UA-treated Min6 cells. We found that UA increased the level of cleaved caspase-3 while reducing the level of pro-caspase-3. All of these effects, however, were reversed dose-dependently by treatment with ICA.
Because reduced insulin release has been reported to be the main symptom during the pathogenesis of T2D [21], we assessed the effects of ICA on levels of insulin in glucose-treated Min6 cells. We found that ICA significantly and dose-dependently increased high glucose-induced insulin secretion when compared with cells treated with UA alone. ICA prevented injury to pancreatic β-cells by reducing pancreatic β-cell apoptosis and promoting insulin release. The PI3K/AKT signal pathway has been reported vital to many diseases, and activation of this pathway may have anti-diabetic properties. Loganin was found to have anti-diabetic effects on INS-1 cells by inhibiting FOXO1 nuclear translocation via the PI3K/Akt signaling pathway [22]. Moreover, the in vitro anti-diabetic effects of several new norditerpenoid alkaloids were shown to involve the PI3K/Akt signaling pathway [23]. Interestingly, we found that UA treatment of Min6 cells markedly reduced the levels of p-PI3K and p-AKT, and that these effects were reversed by treatment with ICA. Taken together, these findings showed that ICA was able to activate the PI3K/AKT pathway in UA-treated Min6 cells, suggesting that this signal pathway may be an important regulator in protecting Min6 cells from UA-induced dysfunction.
To further analyze the specific mechanisms underlying the protective effects of ICA, UA-treated Min6 cells were treated with LY294002 and/or 20 μM ICA, and cell viability, cell apoptosis and PI3K/Akt signaling pathway-related proteins were analyzed. ICA suppressed UA-induced cytotoxicity, whereas LY294002 significantly overcame the protective effects of ICA and induced cell apoptosis, indicating that the anti-apoptotic effects of ICA involve the stimulation of the PI3K/Akt pathway. Similarly, LY294002 enhanced cleaved caspase-3 expression while reducing pro-caspase-3 levels. Moreover, results from the GSIS assay showed that LY294002 significantly reduced insulin release in glucose-treated Min6 cells. Similar findings were observed when measuring the levels of expression of genes and proteins associated with the PI3K/Akt pathway. The levels of p-PI3K and p-AKT and the ratios of p-PI3K/PI3K and p-AKT/AKT were significantly lower in Min6 cells treated with UA, ICA, and LY294002 than in cells treated with UA and ICA. However, there were no differences in PI3K and AKT mRNA levels. These results further suggested that the PI3K/Akt pathway was activated after ICA exposure, and that LY294002 suppressed this activation.
This study had several limitations. First, we did not determine cell inhibitory effects by assessing the IC50 of various treatments, nor did we quantify the protein levels of cleaved caspase-3 and pro-caspase-3. Moreover, LY294002 is not a non-selective inhibitor of PI3K/AKT, and therefore should not be used to uniquely target PI3K. A second inhibitor, such as wortmannin, should be tested.
Conclusions
These findings indicate that ICA restored UA-stimulated injury in Min6 cells and that this effect was likely mediated through the PI3K pathway. These investigations may suggest a novel agent for the treatment of patients with T2D.
Footnotes
Conflict of interests
None.
Source of support: The present study was supported by 2018 Guangxi first class discipline construction project key topics of Guangxi University of Chinese Medicine (grant no. 2018XK036)
References
- 1.Campbell MR. Review of current status of molecular diagnosis and characterization of monogenic diabetes mellitus: A focus on next-generation sequencing. Expert Rev Mol Diagn. 2020;20:413–20. doi: 10.1080/14737159.2020.1730179. [DOI] [PubMed] [Google Scholar]
- 2.Watanabe Y, Kuribayashi N, Uchida D, et al. Correction to: Study protocol for the effects of formula diet with dapagliflozin on metabolic improvement and body composition in type 2 diabetes mellitus. Diabetes Ther. 2020;11:753. doi: 10.1007/s13300-020-00757-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lee EM, Lee YE, Lee E, et al. Protective effect of heme oxygenase-1 on high glucose-induced pancreatic β-cell injury. Diabetes Metab J. 2011;35:469–79. doi: 10.4093/dmj.2011.35.5.469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wen D, Li P, Li X, et al. The feature extraction of resting-state EEG signal from amnestic mild cognitive impairment with type 2 diabetes mellitus based on feature-fusion multispectral image method. Neural Netw. 2020;124:373–82. doi: 10.1016/j.neunet.2020.01.025. [DOI] [PubMed] [Google Scholar]
- 5.Shao Y, Shao H, Sawhney MS, Shi L. Serum uric acid as a risk factor of all-cause mortality and cardiovascular events among type 2 diabetes population: Meta-analysis of correlational evidence. J Diabetes Complications. 2019;33:107409. doi: 10.1016/j.jdiacomp.2019.07.006. [DOI] [PubMed] [Google Scholar]
- 6.Zong N, Li F, Deng Y, et al. Icariin, a major constituent from Epimedium brevicornum, attenuates ibotenic acid-induced excitotoxicity in rat hippocampus. Behav Brain Res. 2016;313:111–19. doi: 10.1016/j.bbr.2016.06.055. [DOI] [PubMed] [Google Scholar]
- 7.Li N, Wang J, Wang X, et al. Icariin exerts a protective effect against d-galactose induced premature ovarian failure via promoting DNA damage repair. Biomed Pharmacother. 2019;118:109218. doi: 10.1016/j.biopha.2019.109218. [DOI] [PubMed] [Google Scholar]
- 8.Xiong W, Ma H, Zhang Z, et al. The protective effect of icariin and phosphorylated icariin against LPS-induced intestinal epithelial cells injury. Biomed Pharmacother. 2019;118:109246. doi: 10.1016/j.biopha.2019.109246. [DOI] [PubMed] [Google Scholar]
- 9.Ren Y, Zhu F, Liu Z. Inhibitory effect of icariin on osteosarcoma cell proliferation via the Wnt/beta-catenin signaling pathway. Oncol Lett. 2018;16:1405–10. doi: 10.3892/ol.2018.8821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhong S, Ge J, Yu JY. Icariin prevents cytokine-induced beta-cell death by inhibiting NF-kappaB signaling. Exp Ther Med. 2018;16:2756–62. doi: 10.3892/etm.2018.6502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Han Y, Jung HW, Park YK. Effects of icariin on insulin resistance via the activation of AMPK pathway in C2C12 mouse muscle cells. Eur J Pharmacol. 2015;758:60–63. doi: 10.1016/j.ejphar.2015.03.059. [DOI] [PubMed] [Google Scholar]
- 12.Jing R, Zhong QQ, Long TY, et al. Down-regulated miRNA-26a-5p induces the apoptosis of endothelial cells in coronary heart disease by inhibiting PI3K/AKT pathway. Eur Rev Med Pharmacol Sci. 2019;23:4940–47. doi: 10.26355/eurrev_201906_18084. [DOI] [PubMed] [Google Scholar]
- 13.Yano K, Seko Y, Takahashi A, et al. Effect of sodium glucose cotransporter 2 inhibitors on renal function in patients with nonalcoholic fatty liver disease and type 2 diabetes in Japan. Diagnostics (Basel) 2020;10:86. doi: 10.3390/diagnostics10020086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xu H, Zhou S, Qu R, et al. Icariin prevents oestrogen deficiency-induced alveolar bone loss through promoting osteogenesis via STAT3. Cell Prolif. 2020;53:e12743. doi: 10.1111/cpr.12743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cao LH, Qiao JY, Huang HY, et al. PI3K-AKT signaling activation and icariin: the potential effects on the perimenopausal depression-like rat model. Molecules. 2019;24:3700. doi: 10.3390/molecules24203700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mo ZT, Li WN, Zhai YR, Gao SY. The effects of icariin on the expression of HIF-1alpha, HSP-60 and HSP-70 in PC12 cells suffered from oxygen-glucose deprivation-induced injury. Pharm Biol. 2017;55:848–52. doi: 10.1080/13880209.2017.1281968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kawada T. Serum uric acid and metabolic components with special reference to diabetes mellitus. Clin Chim Acta. 2018;484:304. doi: 10.1016/j.cca.2018.06.013. [DOI] [PubMed] [Google Scholar]
- 18.Rafiullah M, Siddiqui K, Al-Rubeaan K. Association between serum uric acid levels and metabolic markers in patients with type 2 diabetes from a community with high diabetes prevalence. Int J Clin Pract. 2019;74:e13466. doi: 10.1111/ijcp.13466. [DOI] [PubMed] [Google Scholar]
- 19.Zhang Y, Yamamoto T, Hisatome I, et al. Uric acid induces oxidative stress and growth inhibition by activating adenosine monophosphate-activated protein kinase and extracellular signal-regulated kinase signal pathways in pancreatic beta cells. Mol Cell Endocrinol. 2013;375:89–96. doi: 10.1016/j.mce.2013.04.027. [DOI] [PubMed] [Google Scholar]
- 20.Yan X, Zhang L, Guo J, et al. Processing of kansui roots stir-baked with vinegar reduces kansui-induced hepatocyte cytotoxicity by decreasing the contents of toxic terpenoids and regulating the cell apoptosis pathway. Molecules. 2014;19:7237–54. doi: 10.3390/molecules19067237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu CZ, Ou SH, Chang LC, et al. Deficiency of urokinase plasminogen activator may impair beta cells regeneration and insulin secretion in type 2 diabetes mellitus. Molecules. 2019;24:4208. doi: 10.3390/molecules24234208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mo FF, Liu HX, Zhang Y, et al. Anti-diabetic effect of loganin by inhibiting FOXO1 nuclear translocation via PI3K/Akt signaling pathway in INS-1 cell. Iran J Basic Med Sci. 2019;22:262–66. doi: 10.22038/ijbms.2019.30246.7294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tang D, Chen QB, Xin XL, Aisa HA. Anti-diabetic effect of three new norditerpenoid alkaloids in vitro and potential mechanism via PI3K/Akt signaling pathway. Biomed Pharmacother. 2017;87:145–52. doi: 10.1016/j.biopha.2016.12.058. [DOI] [PubMed] [Google Scholar]