Abstract
Asthma is a chronic inflammatory disease associated with airway hyper-responsiveness, chronic inflammatory response, and excessive structural remodeling. The current therapeutic strategies in asthmatic patients are based on controlling the activity of type 2 T helper lymphocytes in the pulmonary tissue. However, most of the available therapies are symptomatic and expensive and with diverse side outcomes in which the interruption of these modalities contributes to the relapse of asthmatic symptoms. Up to date, different reports highlighted the advantages and beneficial outcomes regarding the transplantation of different stem cell sources, and relevant products from for the diseases’ alleviation and restoration of injured sites. However, efforts to better understand by which these cells elicit therapeutic effects are already underway. The precise understanding of these mechanisms will help us to translate stem cells into the clinical setting. In this review article, we described current knowledge and future perspectives related to the therapeutic application of stem cell-based therapy in animal models of asthma, with emphasis on the underlying therapeutic mechanisms.
Keywords: Stem cells, Asthma, Regeneration, Cellular and molecular mechanisms
Background
It has been estimated that pulmonary diseases are the third leading cause of death worldwide [1]. The pathological changes that occurred in asthma are complex and accompanied by prominent airway conduit inflammation and obstruction, bronchiole epithelial metaplasia, and overproduction of mucus [2]. Ultimately, the existence of such conditions leads to airway hyperactivity and exaggerated reactions in response to numerous endogenous and endogenous stimuli [3]. Several reports have shown that different immune cell types, predominantly eosinophils, and Th were recruited to the asthmatic niche coincided with abnormal formation of ECM [4]. Along with these changes, terminal alveoli and air sacs also show excessive pathological remodeling which is indicated by the thickening of the interstitial wall [5]. Ultimately, the persistence of asthmatic changes contributes to epithelial cell detachment and shedding, per-bronchiolar cuffing, type 1 collagen synthesis, and progressive loss of oxygen and carbon dioxide interchange [6]. From a mechanistic standpoint, asthma is categorized into both allergic and non-allergic forms which correlate with dynamic production IgE antibodies. In allergic asthma, the immune system is hyperactive and responds adversely to diverse stimuli [7]. Pathological examinations have revealed that Th2 cells actively were recruited into bronchioles and released different cytokines such as IL-4, IL-5, and IL-13. The cytokines shift the activity of resident pulmonary cells, such as epithelial cells, fibroblasts, and smooth muscle cells, and other immune cells mainly mast cells, eosinophils, and IgE-producing B lymphocytes [8, 9]. Due to the inefficiency of current treatment protocols, many researchers and clinicians try to find safe and effective modalities in the alleviation and restoration of the pulmonary system after asthmatic disease. This review aims to scrutinize experiments related to the application of stem cells and underlying cellular and molecular mechanisms participating in the alleviation of asthmatic changes.
To date, several methods have been developed for the induction of bronchial asthma using different animal models. For example, cotton dust, OVA, Ascaris lumbricoides antigens, cockroach extracts (Blatella germanica), house dust mite extract (HDM) (Dermatophagoides pteronyssinus, Dermatophagoides farina), fungi, and molds (Aspergillus fumigatus, Alternaria alternata), ragweed, latex (Hevea brasiliensis), and bacterial lipopolysaccharide (LPS) are allergens commonly implicated in asthma development and exacerbations [10–13]. Besides, the application of Alum via intraperitoneal route followed by airway challenges induces an acute allergic response that will mask immune events that are only partially contributing to inflammation and airway hyper-responsiveness [12, 14]. Considering the previously published experiments, it becomes clear that OVA, extracted from a chicken egg, is a widely used allergen for the sensitization of bronchioles in different animal models solely because of all the immune tools are available and recruited to the pulmonary niche after OVA challenge [13]. OVA can be produced on large scales at a lower cost. However, each sensitization method has strengths and weaknesses. It has been shown that repeated airway exposures to OVA may contribute to immune tolerance and the induced airway inflammation is not quite like what happens in the human asthmatic lungs. Distinct properties of HDM such as appropriate immunogenicity, direct activation of innate cells, and intrinsic enzymatic activity, make it suitable to mimic asthma-like conditions [13]. Commensurate with these descriptions, the selection of certain allergen correlates with the number of replicates and using alone or in combination with other allergens [15].
Application of stem cells in the asthmatic niche
Over the past decade, the use of stem cell-based therapies and bioengineering modalities has been extensively studied for the regeneration of lung diseases. There are a growing number of scientific reports conducted in terms of different stem cells application to promote either structural or functional pulmonary restoration with a focus on both differentiation capacity and paracrine activity [16]. In this regard, it seems that adult bone marrow stem cells, including different lineages such as hematopoietic stem cells (HSCs), mesenchymal stem cells (MSCs), and endothelial progenitor cells (EPCs) hold splendid promise for the healing of injured tissues. The bone marrow niche is an appropriate microenvironment for the dynamic growth of HSCs and EPCs, while MSCs could be isolated from different tissues. In keeping with this theme, the regenerative potential of multiple stem cell types has been previously assessed in different chronic pulmonary pathologies such as asthma, COPD, and broncho-pulmonary dysplasia [16] (Table 1). According to the released data, stem cells were administered via intravenous infusion, intraperitoneally, or transplanted directly into the pulmonary niche via intranasal and intra-tracheal routes [42, 43]. Delving a bit deeper, the best appropriate administration route and cell delivery methods have not been determined yet. Considering relative ease of doing the experiments, it is thought that cell delivery via the intra-tracheal pathway is logically preferred to the other routes. For instance, the possibility of cell bio-distribution and colonization to the non-pulmonary tissues will be decreased dramatically which in turn reduces the dose of transplant target cells. Also, the direct introduction of distinct cell types to the injury site could yield better regenerative outcomes [43]. Even specific anatomical regions in the pulmonary niche encompass specific stem cells that are generally inactive under the normal condition and further acquire regenerative potential during injuries to the epithelial layers. Several reports highlighted the existence of pluripotency and stemness features in bronchioalveolar stem cells, non-ciliated bronchiolar secretory cells (Clara cells), basal cells, alveolar type II pneumocytes, and submucosal gland stem cells [44, 45]. Inside the lungs, there are a fraction of stem cells, namely c-Kit+ cells, with highly regenerative potential and self-renewal capacity [46]. It is thought that a large amount of c-Kit+ cells from bone marrow and systemic circulation and notably massive recruitment of these cells into the pulmonary niche not only did not ameliorate the progression of pulmonary disease but also exacerbate the progression of pathological responses [20]. Overall, the results from different animal models of asthma confirmed the advantageous and potential benefits after transplantation of stem cells which coincided with a reduction of inflammatory reactions, immune cell recruitment, and regulation of Th1 to Th2 ratio, reduced collagen fiber deposition in the lung parenchyma, and changes in the dynamic synthesis of pro- and anti-inflammatory cytokines. Besides, structural changes and pathological remodeling of the blood-air barrier, epithelial metaplasia, and mucus-producing goblet cells are faded post-stem cell transplantation [47].
Table 1.
List of some in vivo and in vitro studies on the effects of stem cells or cell products on experimental induced asthma
| Stem cell type | Route of administration | Animal model and type of injury | Time of study | Outcome | Ref |
|---|---|---|---|---|---|
| Bone marrow-derived c-Kit+ cells | Intratracheally | Hyperoxia-induced lung injury in rats | 15 days | Angiogenesis and pro-angiogenic factors ↑, aveolarization↑, apoptosis↓ | [17] |
| Anti-c-Kit siRNA | Intravenously | OVA-induced allergic asthma in mouse | 72 h after siRNA injection | Pulmonary infiltration of inflammatory cells (eosinophils and lymphocytes)↓, IL-4 and IL-5↓ | [18] |
| Anti-c-Kit siRNA | Intranasal | OVA-induced allergic asthma in mouse | 72 h after siRNA injection | SCF, IL-4, and IL-5↓, eosinophil infiltration↓ | [19] |
| c-Kit+ cells | Intratracheally | OVA-induced allergic asthma in mouse | 10 days | Inflammation ↓, airway remodeling, and function↑ | [20] |
| Bone marrow MSCs | Intravenously, Intratracheally | OVA-induced allergic asthma in mouse | More than 10 days | Th2 and Th17 cytokines↓, IgE↓, eosinophilia↓ | [21] |
| Bone marrow MSCs and CM | Intratracheally | OVA-induced allergic asthma in rats | 14 days | MSC-treated rats: neutrophili and neutrophilia↓, CD3+/CD4+↓, IL-10↑, IL-4↓ | [22] |
| Bone marrow MSCs and CM | Intravenously | OVA-induced allergic asthma in rats | 14 days | CD3+/CD4+↑, CD3+/CD8+↓, immune cells infiltration↓ (the therapeutic effects were more higher than CM) | [23] |
| Adipose-derived MSCs | Intratracheally | OVA-induced allergic asthma in mouse | ND | Airway responsiveness↓, lymphocytes infiltration↓, lgE, IL-1β ↓, IL-4 ↓, IL-17F↓, IL-10↑, RORγ↑, CD4+CD25+Foxp3 Treg cells↑ | [11] |
| Adipose-derived MSC | Intravenously | Feline chronic allergic asthma | More than 4 months | Airway eosinophilia and responsiveness→, bronchial wall thickening ↓ | [24] |
| Bone marrow mononuclear cells | Intratracheally | OVA-induced allergic asthma in mouse | 7 days | Alveolar collapse↓; bronchoconstriction↓; fibrosis↓; IL-4, -5, and -13↓; IFN-γ↑; TGF-β↑ | [25] |
| iPSC-derived MSCs | Intratracheally | OVA-induced allergic asthma in mouse | 4 days | Connexin 43-mediated mitochondrial transfer↑, epithelial cells death↓ | [26] |
| Human umbilical cord blood-derived MSCs | Intravenously | OVA-induced allergic asthma in mouse | 29–31 days | IL-4, IL-5, and IL-13↓; IgE and IgG1↓; bronchial hyper-responsiveness↓; eosinophil infiltration↓ | [27] |
| iPSCs, and mesenchymoangioblast-derived MSCs | Intranasal and Intravenously | OVA-induced allergic asthma in mouse | 14 days | TGF-β1↓; airway/lung fibrosis↓; collagen-degrading gelatinase ↑ | [28] |
| Human ESC-MSCs | Intravenously | OVA-induced allergic asthma in mouse | 20 days | Th2 cells and eosinophils↓; Treg↑; periodic acid–Schiff positive cells↓; CD4+CD25+FOXP3+ cells↑; IL-4, IL-5, and IL-13↓; Ccl11, Ccl24, Il13, Il33, and Ear11 expression ↓ | [29] |
| Bone marrow, umbilical cord, and adipose-derived MSCs | Intravenously | OVA-induced allergic asthma in mouse | 7–10 days | Eosinophil↓; IL-4, IL-5, and IL-13↓; INF-γ↑; IL-10 producing macrophages↑ | [30] |
| MSC-derived exosomes | In vitro | Target cells: asthmatic peripheral mononuclear cells | 24 h | IL-10 and TGF-β↑, proliferation of CD4+CD25+FOXP3+ cells↑ | [31] |
| MSCs CM | In vitro | GM-CSF-induced asthmatic changes in 3 T3 murine airway fibroblast cells | 14 days | Collagen types I, III↓; hyaluronan↓ | [32] |
| MSCs | Retro-orbital | OVA-induced allergic asthma in mouse | 4 weeks | Hyaluronan↓, airway inflammation↓ | [32] |
| Adipose-derived MSCs | Intravenously | OVA-induced allergic asthma in mouse | 12 days | IDO, TGF-β, and PGE2↑ (IL-4, IL-5, and IL-13↓); IFN-γ↑; IL-10↑ | [33] |
| Human placenta MSCs | Intravenously | OVA-induced allergic asthma in rats | 22 days | Notch3 and delta-4↑; notch-1, -2 and jagged-1↓; IgE, Th2 cytokines↓ | [34] |
| iPSC-derived MSCs | Intravenously | OVA-induced allergic asthma in mouse | 55 days | Fibrosis and α-SMA↓, TGF-β1↓, phosphorylated Smad2/3 expression↓ | [35] |
| Adipose tissue MSC-derived extracellular vesicles | Intravenously | OVA-induced allergic asthma in mouse | 7 days | TGF-β↓, fibrosis↓, inflammation↓, bronchiolar Siglec-F+ eosinophils↓, eotaxin↓, CD3+ CD4+ cells↓, CD4+CD25+Foxp3+ cells↑ | [10] |
| Bone marrow MSCs | Intravenously | OVA-induced allergic asthma in mouse | 7 days | Pulmonary oxidative stress↓, and nitrotyrosine↓ | [36] |
| Adipose-derived MSCs and bone marrow-derived MSCs | Intratracheally | HDM-induced allergic asthma in mouse | 3–7 days | Bone marrow MSCs: IL-10↑, the influx of eosinophils and B cells ↓, alveolar macrophage inflammatory response↓, lung function, and remodeling→, adipose-derived MSCs were ineffective | [15] |
| Adipose-derived MSCs | Intravenously | HDM-induced allergic asthma in mouse | 3 days | Inflammation↓, Th1 cytokines↓, hyper-responsiveness →, contractile tissue→, cell integration, and differentiation → | [37] |
| Bone marrow-derived MSCs | Intravenously | HDM-induced allergic asthma in mouse | 8–10 days | Airway responsiveness↓, bronchial contraction ↓, inhibitory type 2 muscarinic receptor↑, phagocytosis of MSCs by local macrophages, macrophage M2 suppressive phenotype↑ | [38] |
| Human iPSC-MSCs | Intravenously | Neutrophilic airway inflammation induced by LPS and OVA in mouse | 4–48 h | Th cells (Th17)↓, Th cells-associated cytokines↓, neutrophilic airway inflammation↓, p-STAT3↓, GATA3↓, RORγt↓, iPSC-MSCs differentiation into Th cells↑ | [39] |
| Adipose-derived MSCs | Intravenously | HDM-induced allergic asthma in mouse | 7 days | IL-3 and IL-4↓, BALF CD4+ T cells, and Eosinophils↓, Fibrosis↓, TGF-β↓, α-actin↓ | [40] |
| Bone marrow-derived MSCs | Intravenously | Aspergillus fumigatus hyphal extract-induced asthma in mouse | 76–78 days | Th17-mediated airway inflammation↓, T regulatory cells →, airway hyper-responsiveness↓, BALF Th2, and Th17 soluble mediators↓ | [41] |
ND, non-determined; “↑”, increase; “↓”, decrease; “→”, ineffective
Application of MSCs in asthma
In a review of previously published experiments, MSCs have been extensively applied in the alleviation of asthma in different animal models more than other types of stem cells [48]. Many researchers showed that MSCs could proliferate for multiple passages which allow for large-scale production of these cells for different regenerative medicine applications in animal models of asthma. Based on a scientific document, it has been shown that MSCs are capable of suppressing inflammatory response and pathological remodeling in the asthmatic context [47, 49]. Based on conducted experiments, MSCs were transplanted to the asthmatic animals at the range from 1 × 106 to 5 × 107 [50, 51]. According to histological examination, these cells easily migrate toward inflammatory sites in response to cytokine concentration gradients after systemic or local administration. It can be claimed that the production of different factors and cytokines triggers MSCs activation. In vitro pre-treatment of bone marrow-derived mesenchymal stem cells with sera from asthmatic mice increase immunomodulatory properties in allergic asthma [52]. It seems that the positive therapeutic effects of MSCs are mainly done by releasing an array of factors in a paracrine manner which modulates the cell-based and humoral immune responses compared to differentiation potential and juxtacrine activity [43]. In support of this statement, several papers were published that the majority of transplant MSCs are cleared from the pulmonary niche after few days possibly through phagocytosis by alveolar macrophages or apoptosis pathways, raising the question of how they prompt such long-lasting immunosuppressive effects [53]. The activity of recipient immune cells, cytotoxic T cells, promotes MSCs apoptosis via perforin-dependent mechanism [54]. Although it may seem that the decrease of transplanted MSCs by immune rejection could diminish regenerative outcome this phenomenon is done in antibody- and MHC-free manner [54]. Surprisingly, the possible apoptotic death of transplanted MSCs in the asthmatic niche could in part, but not completely, regulated local cellular and humoral immunity via the regulation of phagocytes recruited to the pulmonary tissue [55]. Besides, an elevated ROS generation and enhanced pro-inflammatory cytokines could accelerate functional MSCs depletion at the site of inflammation by eliminating trans-differentiation capacity, self-renewal, and prompt aging [56]. Despite these limitations after the introduction of MSCs to the asthmatic niche, MSCs potentially possess magnificent immunomodulatory capacity without provoking immunogenic responses. MSC secretome harbors diverse factors and cytokine could regulate the functional activity of T and B lymphocytes, dendritic cells, and natural killer cells [57]. Even in the presence of TNF-α and IFN-γ, MSCs can acquire immunosuppression phenotype and immunomodulatory properties. It seems that the production of indoleamine 2,3-dioxygenase and prostaglandin E2 is actively involved in this phenomenon [58].
Several experiments revealed that MSCs exhibited a different restorative capacity based on isolation from various tissues. Also, the MSC subtype possesses a different multipotential capacity [59]. Bone marrow is a primary and well-known source for the isolation of MSCs. However, alternative sources for MSCs isolation in order of importance and number of conducted studies are adipose tissue, blood, umbilical cord blood, skeletal muscle, tendons, lung, etc. [60]. In addition to differences in the therapeutic capacity of MSC subtype, experiments have shown distinct immunomodulatory properties for multiple MSC types. For instance, it has been elucidated that MSCs isolated from adipose and placenta tissues elicited robust immunomodulation in experimental allergic asthma compared to bone marrow MSCs [61, 62]. Lung and umbilical cord blood MSCs have short-lasting persistence in inflamed sites compared to bone marrow MSCs administrated intravenously [63, 64]. Li et al. claimed that the introduction of placental-derived MSCs in OVA-sensitized rats, upregulated IL-10, reduced IL-17, and blunted Th17 to Treg ratio [65]. Recent data examining the anti-asthmatic properties of placental MSCs within the pulmonary niche showed the reduction of eosinophils in bronchoalveolar lavage fluid and suppression of IgE and IL-4. Along with these changes, the proliferation of goblet cells, and synthesis of mucus closed to near-normal levels and lymphocyte polarization toward Th2 was interrupted [34].
It is noteworthy that various MSC subtypes could exhibit diverse regenerative potential in the asthmatic pulmonary niche. As abovementioned, it seems that the source of MSCs could alter persistence in the inflammatory asthmatic niche correlated with the ability to express adhesion molecules, distinct integrins. MSC subtype isolated from the multiple tissues possess different global gene expression profiles and paracrine activity which may contribute to diverse regenerative outcomes [66]. In support of this statement, it has been shown that intra-tracheal administration of mouse MSCs from three different tissues such as adipose tissue, bone marrow, and lungs modulated inflammatory response and structural remodeling with different outcomes in OVA-induced asthma model, possibly via distinct secretome profile [66]. Abreu and colleagues highlighted a superior paracrine activity of bone marrow MSCs in comparison with adipose-derived counterparts [66]. Hence, it seems logical to select an appropriate MSC type with a special multipotentiality regarding the entity of pulmonary injury.
Mechanisms involved in the therapeutic effects of MSCs in experimental asthma models
Immunomodulation
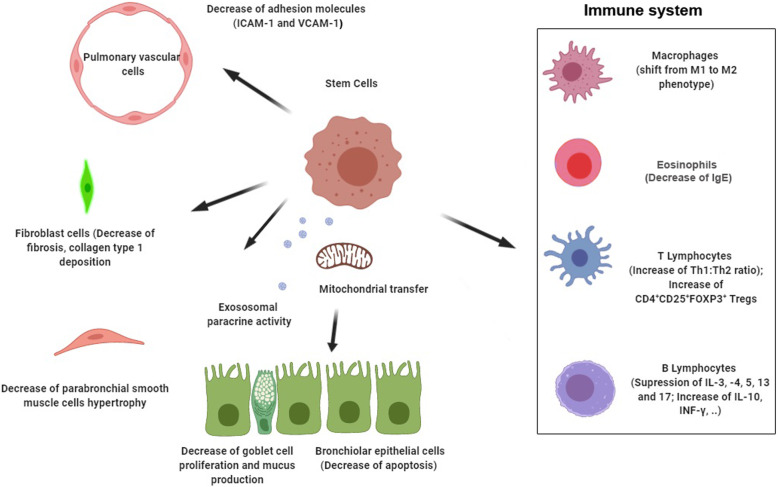
As abovementioned, MSCs have been extensively applied in several experimental studies in asthma. It seems that the therapeutic properties of MSCs are mainly correlated with the immune system regulation at the site of inflammation [43, 63, 64, 67]. MSCs are eligible to regulate Th2 to Th1 ratio, synthesis of interleukins such as 4, 5, and 13; IgE; and mucus after introduction into the asthmatic niche. By increasing TGF-β and IFN-γ, MSCs can abort untamed allergic responses [67–69]. These events coincide with the increase of CD4+CD25+ FoxP3+ Treg cells, IL-10 in bronchioalveolar discharge, a decrease of mast and goblet cells, suppression of nitrosative stress, and inhibition of phagocytic activity in alveolar macrophages and [38, 67, 70] (Fig. 1). There is a close reverse relationship between the eosinophil population in bronchoalveolar lavage and Treg cells which seem critical for the protective impact of MSCs [71]. Most experiments implied that the control of Th2 lymphocytes and relevant allergic reactions could be an efficient strategy for the control of asthmatic injuries [67–69]. Notably, different types of MSCs transplantation have different time-dependent therapeutic outcomes. Due to non-specific bio-distribution and the problem associated with the capacity to cross the blood-air sac barrier, intravenous administration possibly leads to inadequate MSCs recruitment to the asthmatic niche. Despite these limitations, this approach is recommended in unstable conditions [72]. Some authorities claimed that approximately 4- to 5-fold doses of MSCs is required to yield the same therapeutic outcome applicable to results by local administration [72]. Based on previously published data, direct intra-tracheal delivery of whole bone marrow mononuclear cells yielded more cells trapped inside the lung parenchyma in comparison with the systemic route. Both modalities resulted in a similar immunomodulatory capacity of MSCs [25]. It has been shown that intraperitoneal administration of bone marrow MSCs is the potential to modulate the allergic asthma reaction. Soon after injection into the peritoneal cavity, these cells easily migrate to the pulmonary niche and exert immunomodulatory capacity [73, 74].
Fig. 1.
Pleiotropic effects of stem cells on pulmonary asthmatic niche via different mechanisms
If we assume that the therapeutic capacity of transplant MSCs is mainly done via paracrine activity, thus, it is logical to transplant distinct cell types to the asthmatic niche locally rather than via a systemic route [28]. Of course, we must be aware that the local administration is per se invasive, expensive due to surgical procedure and postoperative care. Due to mechanical pressure and inflammatory tissue conditions, a fraction of transplant MSC dies soon after injection into inflamed sites. Despite these limitations, fewer MSCs are needed in local delivery to accomplish therapeutic efficacy. Calling attention, paracrine activity is just efficient in a short distance after close interaction of pulmonary resident cells with MSCs in proximity [75]. Considering these limitations, the repeated dose of MSCs could, if not completely, circumvent disadvantage related to systemic administration in the asthmatic niche [76]. Even though, a repeated dose of MSCs increases the risk of ectopic non-specific overgrowth in different tissues except the lungs [77]. It should be in mind that most studies in animal models reported the efficiency of MSCs in the alleviation of asthmatic changes soon after initial sensitization. Some researchers used episodic allergen exposure to the stabilized asthma-like condition [24]. However, there are few long-term follow-up studies related to the therapeutic effects of MSCs asthmatic animals. Commensurate with these descriptions, there was no basis for the statement that MSCs could completely restore or retrieve the asthmatic lung function. For example, Trizl and co-workers performed six intravenous administrations of MSCs bi-monthly in asthmatic cats and followed up for 1 year [24]. They declared that MSCs failed to suppress inflammation of airway conduits in terms of eosinophil number and bronchiolar hypersensitivity. In another study done by the same research group, five systemic administrations of MSCs showed an anti-inflammatory response at day 130 while data represented the lack of prominent inflammation suppression by month 9 [78]. The scientific rationale for these data could correlate with the fact that bona fide MSCs exert therapeutic effects only short periods after transplantation via differentiation, juxtacrine, and paracrine mechanisms before their death.
Multiple mechanisms action of MSCs in asthmatic niche
In addition to the immunomodulatory capacity of MSCs, growing evidence has proposed the existence of multiple mechanisms, such as trans-differentiation capacity, cell fusion, mitochondrial transfer, and paracrine activity done by microvesicles and exosomes, in these cells to alleviate asthma-related pathology [79] (Fig. 1). As aforementioned, multiple experiments confirmed the existence of factors and cytokines in MSCs secretome which are packed inside extracellular nano-sized vesicles, namely exosomes and transported to the target cells. Additionally, the direct differentiation potency of MSCs has been neglected previously by reports which are possibly due to the enhanced MSCs death at the site of transplantation [22, 80]. A study conducted by Spees and co-workers showed that the simultaneous culture of human MSCs with heat-injured pulmonary epithelial cells promoted differentiation toward epithelial-like lineage [81]. Based on the facts from experiments, it would not be an exaggeration to mention that paracrine activity is the main suggested therapeutic bioactivity of MSCs in the asthmatic niche [82]. These cells have inherent capacity to release 40–200 nm nanoscale exosomes which harbor multiple anti-inflammatory factors that could regulate the function of immune cells [83]. Interestingly, these nanoparticles easily spread in the bio-fluids, are stable, and survive in a harsh milieu when even the source cells could not survive [84]. On this basis, Cruz and co-workers claimed that systemic injection of conditioned media (CM) or extracellular vesicles harvested from mouse and human MSCs are equally effective in the regulation of Th2/Th17-associated asthma hypersensitivity and inflammation in a mouse model of mycosis [85]. Concerning the fact that some regenerative effects of MSCs are done via releasing soluble effectors, thus, MSC-free therapy such as CM and exosomes could be an alternative due to easy storing and handling. By using these approaches, it is less likely to see cellular emboli, tumorigenesis, and unwanted immune responses after transplantation [86]. According to data from a study conducted by Keyhanmanesh et al., they reported that three times the systemic injection of MSC CM inhibited hallmark of asthma indicated by suppression of IL-4 and increase of INF-γ. The results were accompanied by the upregulation of T-bet and downregulation of GATA-3 in the asthmatic rats [76], due to a large number of factors inside exosomes exhibiting pleiotropic effects. For instance, Rahbarghazi et al. demonstrated that the intra-tracheal administration of either rat bone marrow MSCs or CM could suppress the Th2-associated activity via suppression of IL-5 and activation of IL-12. They also found that the expression of adhesion molecules such as intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 significantly decreased which per se decreased immune cell recruitment to the pulmonary tissues [48]. In contrast, Ahmadi and colleagues reported the inefficiency of rat MSC CM on the modulation of inflammation in OVA-induced asthma [23]. The reason for contradictory results could correlate with the time and route of administration, and total dose [66, 87]. Additionally, short activity and rapid distribution of factors after transplantation compared to cell injection could be logic for transient therapeutic effects of CM and exosomes. Aside from the fact that CM and exosomes are integral to the paracrine activity of MSCs in injured sites, more investigations are highly demanded to address underlying mechanisms.
Some data showed that MSCs promote tissue regeneration via mitochondrial transfer is a response to external stimuli. The critical role of gap junctional channel, tunnel tube formation and Rho-GTPases such as Miro1 were previously confirmed by which mitochondrial mass was transferred to the damaged cells [88, 89]. In this regard, it has been shown that connexin-43 GJC+ MSCs retrieved epithelial cell bioactivity after mitochondrial transfer in lipopolysaccharide-associated acute pulmonary inflammation. Islam and co-workers found that the suppression of connexin-43 was interrupted by mitochondrial transfer from MSCs to epithelial cells [90]. It seems that the phenomenon of mitochondrial transfer is effective in the alleviation of other pulmonary injuries. For example, Li et al. confirmed the therapeutic effect of bone marrow MSCs against rat COPD induced by cigarette smoke [91]. In a recent study, it has been shown that the intra-tracheal administration of induced pluripotent stem cell-derived MSCs improved mitochondrial dysfunction in epithelial cells via mitochondrial transfer via connexin-43 [26]. Mitochondrial transfer is efficient to slow down the procedure of apoptotic changes in epithelial cells [92]. In addition to the mitochondrial donation, multiple factors released by MSCs could inhibit the apoptosis signaling pathway either in a non-mitochondrial or mitochondrial-dependent manner [93]. Besides, other cell-protective mechanisms, namely autophagy, actively could alter the development of asthmatic remodeling. For instance, it has been shown that intravenous administration of MSCs reduced inflammation in the pulmonary microvascular system via engaging autophagy-related effectors during ischemia/reperfusion in the model of mouse by inhibiting miR-142a-5p in endothelial cells [94, 95].
Unraveling the regenerative effects of MSCs and other stem cell types could be done in both protein and gene levels. The discovery of miRNAs and other factors function is a de novo strategy for the reduction of asthmatic changes [96]. As implied by previous experiments, molecular examination confirmed the alteration of miRNAs during asthmatic changes. Therefore, elucidation of miRNAs and relative changes could be a reliable tool for monitoring the progression of asthma [97, 98]. Further investigations in animal asthma models showed the potency of MSCs and induced pluripotent stem cell-derived MSCs to modulate the expression of pro-inflammatory miRNAs, such as miRNA-155, -133, mmu-miR-21a-3p, and mmu-miR-449c-5p coincided with the induction of miRNA21 and mmu-miR-496a-3p [97, 99, 100]. For example, previous studies showed that MSCs could alter the phenotype and bioactivity of different cells via horizontal transfer of genetic data such as miRNAs and mRNAs [101]. It has been shown that exosomes are the major players in the paracrine activity and transfer of genetic materials and factors from MSCs to the immune cells [101]. The exposure of M1 pro-inflammatory macrophages to the MSC-derived exosomes induced polarization toward M2 anti-inflammatory macrophages [102]. As previously mentioned, the overproduction of Th2-derived cytokines, such as IL-4, IL-5, IL-9, and IL-13, is associated with dysregulated immunity in asthma [103]. It has been shown that the application of MSCs in different asthmatic models could decrease inflammatory response by altering the levels of Th2-derived cytokines and miRNAs associated with the function of these cells [21]. The interaction of microbial pathogen-associated molecular patterns (PAMPs) with pulmonary epithelial cells and immune cells via toll-like ligand receptors in asthmatic niche leads to the production of cytokines and chemokines [104]. Toll-like ligand receptors were also expressed on the surface of different stem cells such as MSCs and endothelial progenitor cells [105, 106]. It seems that the simultaneous stimulation of MSCs and immune cells suggests the putative role of MSCs in controlling the activity of immune cells. The activation of toll-like ligand receptors by PAMPs could frustrate pulmonary macrophages and release a large content of chemokines such as CXCL8 and CXCL11 which in turn increase the migration of MSCs toward the site of asthmatic niche. Also, the presence of MSCs suppresses the activity of microbes by producing antibacterial proteins [107]. It has shown that MSCs can suppress the function of complement cascade by releasing complement inhibitors such as factor H, leading to the inhibition of C3 and C5 convertase [108]. According to different experiments, several miRNAs have critical roles in the inflammation of airway conduit, including miRNA-126, miRNA-let-7, and miRNA-155 [97]. In a study performed by Kuo and co-workers, the therapeutic potential of MSCs has been proved to reduce stretch-induced inflammatory miR-155 in pulmonary bronchiolar epithelial cells by downregulating miR-155 [109]. Data showed that MSCs are promising cell sources to alter the expression of miRNAs in immune cells and pulmonary tissue to reduce the inflammatory condition.
Despite numerous advantages of MSCs application, there are very few reports regarding MSC side effects in pulmonary disease. For instance, it was shown that allogenic transplantation of MSCs in patients with idiopathic pulmonary fibrosis did not cause serious clinically and laboratory abnormalities [110]. However, the long-term follow-up of these patients revealed a total number of 2 deaths per 9 MSC-treated cases because of disease exacerbation [110]. Clinical trials conducted already by local investigators in different countries showed that transplantation of MSCs was appropriately tolerated and only a limited number of side effects were observed due to uncontrolled suppression of immune cells. Besides trans-differentiation of transplanted to undesired cell types, the progression of tumor-like cells and possible metastasis to remote sites are the main challenging issues [110]. Attention should be made to interpret the immunomodulatory properties of MSCs after transplantation under acute and chronic inflammation. It has been shown that the administration of allogenic MSCs increased alveolar macrophage activity a few hours after transplantation via the intravenous route indicated by enhanced MCP-1, CXCL-1, and IL-6 production [111]. To increase the survival rate and modulatory effect of transplanted MSCs, the simultaneous administration of mycophenolate mofetil was performed from the time of cell administration onwards. This strategy inhibits the accumulation of reactive T cells and allogeneic rejection [112]. There is still a long way to go to confirm the therapeutic outcomes of MSCs in different pulmonary allergic diseases.
Xenogeneic transplantation of human MSCs into an animal model of asthma
Despite the existence of inherent species variation in MSC function, some experiments conducted xenogeneic lung transplantation models in animals using human MSCs [113–115]. Similar to animal MSCs, human MSCs exhibited potent immunomodulatory properties either in the acute or chronic asthma mouse model [114, 115]. It has been elucidated that typical hallmarks of asthma were mostly subsided after transplantation of human MSCs, isolated from bone marrow, adipose tissue, and umbilical cord, in the mouse model [30]. Systemic injection of human bone marrow MSCs via tail vein induced pulmonary macrophage polarization toward M2 type via the promotion of the TGF-β/Smad signaling pathway [116]. Interestingly, xenogeneic transplantation of human MSC retro-orbital administration in mice diminished the hyaluronan mucus in the chronic asthma model [32]. These findings support the assumption that autologous, allogeneic, and xenogeneic transplantation of MSCs could promote anti-inflammatory outcomes via engaging different mechanisms. Regarding massive genetic heterogeneity in allogeneic and xenogeneic models, these cells are, although not fully, able to exert regenerative outcomes.
Clinical trials
According to the promising results of animal studies, some efforts have been made to investigate the paracrine and juxtacrine effect of MSCs on the human counterpart of asthma. By March 2020, clinical trial results (conducted in URL: https://clinicaltrials.gov) represented about 9 clinical trials to deal with asthma in the patients (Table 2). Of the nine clinical trials, two ongoing examinations evaluated the efficacy of MSCs in asthmatic patients. In a clinical trial conducted by the University Of Miami Miller School Of Medicine, the therapeutic effects of allogeneic MSCs were examined intravenously and patients were followed for 12 weeks. In the second study performed by Punta Pacifica Hospital of Panama City, intranasal administration of human umbilical cord MSC-derived trophic factors was evaluated in adult asthmatic patients.
Table 2.
The list of clinical trials concerning asthma documented up to March 2020
| Status | Study title | Conditions | Interventions | Phase | Number enrolled |
|---|---|---|---|---|---|
| Active, not recruiting | Allogeneic human cells (hMSCs) via intravenous delivery in patients With Mild asthma | Asthma | Biological: hMSCs | 1 | 6 |
| Active, not recruiting | Safety and feasibility study of an intranasal mesenchymal trophic factor for the treatment of asthma | Asthma | Biological: trophic factors from umbilical cord mesenchymal stem cells | 1 and 2 | 20 |
| Completed | Study to evaluate the effect of benralizumab on allergen-induced inflammation in mild, atopic asthmatics | Asthma | Biological: benralizumab, other: placebo | Phase 3 | 46 |
| Completed | A pilot study for cell-based therapies in patients with asthma | Asthma, allergic rhinitis | Other: blood donation | Not applicable | 20 |
Conclusions and future perspectives
Overall, MSC delivery could diminish inflammation of lungs and airway conduits in the asthmatic animal models. The therapeutic effects of MSCs are done by involving different molecular and cellular pathways related to immunomodulation, mitochondrial donation, and protection against different pathways leading to cell death such as apoptosis and oxidative stress. It is highly recommended to establish diverse basic experiments and clinical trials to address the precise underlying mechanisms of MSCs therapy in asthmatic subjects. Long-term monitoring of asthmatic patients who received MSCs could carefully highlight the possible ineffectiveness and side effects before making a solid decision about cell-based therapies.
Defining the exact mechanism of MSC-therapy in the asthmatic condition is mandatory before the advent of cell therapy as an alternative modality in the clinical setting. The long-term outcomes and survival of locally or systemically administrated MSCs should be determined. The possible side effects of in vitro expansion of MSCs should be determined. We suggest future investigations need to find appropriate dosing and administration routes. Meanwhile, the exact bioactivity of MSCs is still unclear under acute and chronic inflammation.
Acknowledgements
The authors thank the personnel of Stem Cell Research Center and Drug Applied Research Center for guidance and support.
Abbreviations
- COPD
Chronic obstructive pulmonary disease
- CM
Conditioned media
- ESCs
Embryonic stem cells
- ECM
Extracellular matrix
- GJC
Gap junction
- HSCs
Hematopoietic stem cells
- HDM
House dust mite extract
- iPSCs
Induced pluripotent stem cells
- IFN-γ
Interferon-gamma
- IL
Interleukin
- LPS
Lipopolysaccharide
- MSCs
Mesenchymal stem cells
- EPCs
Endothelial progenitor cells
- miRNAs
MicroRNAs
- OVA
Ovalbumin
- siRNA
Small interference RNA
- SCF
Stem cell factor
- Th
T helper lymphocytes
- TGF-β
Transforming growth factor-beta
- TNF-α
Tumor necrosis factor-alpha
Authors’ contributions
F.M., M.A., A.R., and H.R. reviewed the articles and collected data. R.K. and R.R. edited the manuscript and supervised the study. The authors read and approved the final manuscript.
Funding
The manuscript is part of a Ph.D. thesis and is supported by a grant from Stem Cell Research Center of Tabriz University of Medical Sciences and Council for Development of Stem Cell Sciences and Technologies (No. 11.35730-1398.11.8) and approved by the Local Research Ethics Committee of Tabriz University of Medical Sciences (IR.TBZMED.VCR.REC.1397.404).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reza Rahbarghazi and Rana Keyhanmanesh contributed equally to this work.
Contributor Information
Reza Rahbarghazi, Email: rezarahbardvm@gmail.com, Email: rahbarghazir@tbzmed.ac.ir.
Rana Keyhanmanesh, Email: Rkeyhanmanesh@gmail.com.
References
- 1.Tarlo SM, Balmes J, Balkissoon R, Beach J, Beckett W, Bernstein D, Blanc PD, Brooks SM, Cowl CT, Daroowalla F. Diagnosis and management of work-related asthma: American College of Chest Physicians Consensus Statement. Chest. 2008;134(3):1S–41S. doi: 10.1378/chest.08-0201. [DOI] [PubMed] [Google Scholar]
- 2.Halwani R, Al-Muhsen S, Hamid Q. Airway remodeling in asthma. Curr Opin Pharmacol. 2010;10(3):236–245. doi: 10.1016/j.coph.2010.06.004. [DOI] [PubMed] [Google Scholar]
- 3.Ober C, Hoffjan S. Asthma genetics 2006: the long and winding road to gene discovery. Genes Immun. 2006;7(2):95. doi: 10.1038/sj.gene.6364284. [DOI] [PubMed] [Google Scholar]
- 4.Mohammadian M, Sadeghipour HR, Kashani IR, Jahromi GP, Omidi A, Nejad AK, Golchoobian R, Boskabady MH. Evaluation of simvastatin and bone marrow-derived mesenchymal stem cell combination therapy on airway remodeling in a mouse asthma model. Lung. 2016;194(5):777–785. doi: 10.1007/s00408-016-9884-1. [DOI] [PubMed] [Google Scholar]
- 5.Barnes PJ, Drazen JM (2002) Pathophysiology of asthma. In: Asthma and COPD. Elsevier, pp. 343–359.
- 6.Fehrenbach H, Wagner C, Wegmann M. Airway remodeling in asthma: what really matters. Cell Tissue Res. 2017;367(3):551–569. doi: 10.1007/s00441-016-2566-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lemanske RF, Jr, Busse WW. Asthma: clinical expression and molecular mechanisms. J Allergy Clin Immunol. 2010;125(2):S95–S102. doi: 10.1016/j.jaci.2009.10.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Holgate ST. A brief history of asthma and its mechanisms to modern concepts of disease pathogenesis. Allergy, Asthma Immunol Res. 2010;2(3):165–171. doi: 10.4168/aair.2010.2.3.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Y-h S, G-c S, Wan H-y, L-h J, X-y A, Zhu H-x, Wei T, X-y J, B-y Z. Coexistence of Th1/Th2 and Th17/Treg imbalances in patients with allergic asthma. Chin Med J. 2011;124(13):1951–1956. [PubMed] [Google Scholar]
- 10.de Castro LL, Xisto DG, Kitoko JZ, Cruz FF, Olsen PC, Redondo PAG, Ferreira TPT, Weiss DJ, Martins MA, Morales MM. Human adipose tissue mesenchymal stromal cells and their extracellular vesicles act differentially on lung mechanics and inflammation in experimental allergic asthma. Stem Cell Res Ther. 2017;8(1):151. doi: 10.1186/s13287-017-0600-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dai R, Yu Y, Yan G, Hou X, Ni Y, Shi G. Intratracheal administration of adipose derived mesenchymal stem cells alleviates chronic asthma in a mouse model. BMC Pulmonary Medicine. 2018;18(1):131. doi: 10.1186/s12890-018-0701-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Eisenbarth SC. Use and limitations of alum-based models of allergy. Clin Exp Allergy. 2008;38(10):1572–1575. doi: 10.1111/j.1365-2222.2008.03069.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Aun MV, Bonamichi-Santos R, Arantes-Costa FM, Kalil J, Giavina-Bianchi P. Animal models of asthma: utility and limitations. J Asthma Allergy. 2017;10:293–301. doi: 10.2147/jaa.s121092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yang Y-S, Yang M-J, Cho K-H, Lee K, Kim Y-B, Kim J-S, Kang M-G, Song C-W. Study of a BALB/c mouse model for allergic asthma. Toxicological Research. 2008;24(4):253–261. doi: 10.5487/TR.2008.24.4.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zosky GR, Sly PD. Animal models of asthma. Clin Exp Allergy. 2007;37(7):973–988. doi: 10.1111/j.1365-2222.2007.02740.x. [DOI] [PubMed] [Google Scholar]
- 16.Zhang L-B, He M. Effect of mesenchymal stromal (stem) cell (MSC) transplantation in asthmatic animal models: a systematic review and meta-analysis. Pulm Pharmacol Ther. 2019;54:39–52. doi: 10.1016/j.pupt.2018.11.007. [DOI] [PubMed] [Google Scholar]
- 17.Ramachandran S, Suguihara C, Drummond S, Chatzistergos K, Klim J, Torres E, Huang J, Hehre D, Rodrigues CO, McNiece IK, Hare JM, Young KC. Bone marrow-derived c-kit+ cells attenuate neonatal hyperoxia-induced lung injury. Cell Transplant. 2015;24(1):85–95. doi: 10.3727/096368913X667736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wu W, Wang T, Dong J-J, Liao Z-L, Wen F-Q. Silencing of c-kit with small interference RNA attenuates inflammation in a murine model of allergic asthma. Int J Mol Med. 2012;30(1):63–68. doi: 10.3892/ijmm.2012.963. [DOI] [PubMed] [Google Scholar]
- 19.Wu W, Chen H, Li Y-M, Wang S-Y, Diao X, Liu K-G. Intranasal sirna targeting c-kit reduces airway inflammation in experimental allergic asthma. Int J Clin Exp Pathol. 2014;7(9):5505–5514. [PMC free article] [PubMed] [Google Scholar]
- 20.Spaziano G, Cappetta D, Urbanek K, Piegari E, Esposito G, Matteis M, Sgambato M, Tartaglione G, Russo R, De Palma R. New role of adult lung c-kit+ cells in a mouse model of airway hyperresponsiveness. Mediat Inflamm. 2016;2016. [DOI] [PMC free article] [PubMed]
- 21.Habibian R, Delirezh N, Farshid AA (2018) The effects of bone marrow-derived mesenchymal stem cells on ovalbumin-induced allergic asthma and cytokine responses in mice. Iran J Basic Med Sci 21 (5):483-488. Doi:10.22038/ijbms.2018.26898.6575. [DOI] [PMC free article] [PubMed]
- 22.Ahmadi M, Rahbarghazi R, Aslani MR, Shahbazfar A-A, Kazemi M, Keyhanmanesh R. Bone marrow mesenchymal stem cells and their conditioned media could potentially ameliorate ovalbumin-induced asthmatic changes. Biomed Pharmacother. 2017;85:28–40. doi: 10.1016/j.biopha.2016.11.127. [DOI] [PubMed] [Google Scholar]
- 23.Ahmadi M, Rahbarghazi R, Soltani S, Aslani MR, Keyhanmanesh R. Contributory anti-inflammatory effects of mesenchymal stem cells, not conditioned media, on ovalbumin-induced asthmatic changes in male rats. Inflammation. 2016;39(6):1960–1971. doi: 10.1007/s10753-016-0431-2. [DOI] [PubMed] [Google Scholar]
- 24.Trzil JE, Masseau I, Webb TL, Chang CH, Dodam JR, Cohn LA, Liu H, Quimby JM, Dow SW, Reinero CR. Long-term evaluation of mesenchymal stem cell therapy in a feline model of chronic allergic asthma. Clin Exp Allergy. 2014;44(12):1546–1557. doi: 10.1111/cea.12411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Abreu SC, Antunes MA, Maron-Gutierrez T, Cruz FF, Ornellas DS, Silva AL, Diaz BL, Ab'Saber AM, Capelozzi VL, Xisto DG, Morales MM, Rocco PRM. Bone marrow mononuclear cell therapy in experimental allergic asthma: intratracheal versus intravenous administration. Respir Physiol Neurobiol. 2013;185(3):615–624. doi: 10.1016/j.resp.2012.11.005. [DOI] [PubMed] [Google Scholar]
- 26.Yao Y, Fan X-L, Jiang D, Zhang Y, Li X, Xu Z-B, Fang S-B, Chiu S, Tse H-F, Lian Q. Connexin 43-mediated mitochondrial transfer of iPSC-MSCs alleviates asthma inflammation. Stem Cell Reports. 2018;11(5):1120–1135. doi: 10.1016/j.stemcr.2018.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kang S-Y, Park D-E, Song W-J, Bae B-R, Lee J-W, Sohn K-H, Lee H-S, Kang H-R, Park H-W, Chang Y-S, Choi S-J, Oh W-I, Min K-U, Cho S-H. Immunologic regulatory effects of human umbilical cord blood-derived mesenchymal stem cells in a murine ovalbumin asthma model. Clin Exp Allergy. 2017;47(7):937–945. doi: 10.1111/cea.12920. [DOI] [PubMed] [Google Scholar]
- 28.Royce SG, Rele S, Broughton BR, Kelly K, Samuel CS. Intranasal administration of mesenchymoangioblast-derived mesenchymal stem cells abrogates airway fibrosis and airway hyperresponsiveness associated with chronic allergic airways disease. FASEB J. 2017;31(9):4168–4178. doi: 10.1096/fj.201700178R. [DOI] [PubMed] [Google Scholar]
- 29.Lin Y-D, Fan X-L, Zhang H, Fang S-B, Li C-L, Deng M-X, Qin Z-L, Peng Y-Q, Zhang H-Y, Fu Q-L. The genes involved in asthma with the treatment of human embryonic stem cell-derived mesenchymal stem cells. Mol Immunol. 2018;95:47–55. doi: 10.1016/j.molimm.2018.01.013. [DOI] [PubMed] [Google Scholar]
- 30.Mathias LJ, Khong SML, Spyroglou L, Payne NL, Siatskas C, Thorburn AN, Boyd RL, Heng TSP. Alveolar macrophages are critical for the inhibition of allergic asthma by mesenchymal stromal cells. J Immunol. 2013;191:5914–5924. doi: 10.4049/jimmunol.1300667. [DOI] [PubMed] [Google Scholar]
- 31.Y-m D, Y-x Z, Chen R, Lin L, Lin Y, J-g L. Mesenchymal stem cell exosomes promote immunosuppression of regulatory T cells in asthma. Exp Cell Res. 2018;363(1):114–120. doi: 10.1016/j.yexcr.2017.12.021. [DOI] [PubMed] [Google Scholar]
- 32.Goldstein BD, Lauer ME, Caplan AI, Bonfield TL. Chronic asthma and mesenchymal stem cells: hyaluronan and airway remodeling. J Inflammation (London, England) 2017;14:18. doi: 10.1186/s12950-017-0165-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cho K-S, Park M-K, Kang S, Park H-Y, Hong S-L, Park H-K, Yu H-S, Roh H-J. Adipose-derived stem cells ameliorate allergic airway inflammation by inducing regulatory T cells in a mouse model of asthma. Mediators of inflammation. Mediators Inflamm. 2014;2014:436476. [DOI] [PMC free article] [PubMed]
- 34.Li Y, Qu T, Tian L, Han T, Jin Y, Wang Y. Human placenta mesenchymal stem cells suppress airway inflammation in asthmatic rats by modulating notch signaling. Mol Med Rep. 2018;17(4):5336–5343. doi: 10.3892/mmr.2018.8462. [DOI] [PubMed] [Google Scholar]
- 35.Zhong H, Fan X-L, Fang S-B, Lin Y-D, Wen W, Fu Q-L. Human pluripotent stem cell-derived mesenchymal stem cells prevent chronic allergic airway inflammation via TGF-β1-Smad2/Smad3 signaling pathway in mice. Mol Immunol. 2019;109:51–57. doi: 10.1016/j.molimm.2019.02.017. [DOI] [PubMed] [Google Scholar]
- 36.Malaquias M, Oyama L, Jericó P, Costa I, Padilha G, Nagashima S, Lopes-Pacheco M, Rebelatto CLK, Michelotto P, Xisto D. Effects of mesenchymal stromal cells play a role the oxidant/antioxidant balance in a murine model of asthma. Allergol Immunopathol. 2018;46(2):136–143. doi: 10.1016/j.aller.2017.06.003. [DOI] [PubMed] [Google Scholar]
- 37.Mariñas-Pardo L, Mirones I, Amor-Carro O, Fraga-Iriso R, Lema-Costa B, Cubillo I, Rodríguez Milla MÁ, García-Castro J, Ramos-Barbón D. Mesenchymal stem cells regulate airway contractile tissue remodeling in murine experimental asthma. Allergy. 2014;69(6):730–740. doi: 10.1111/all.12392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Braza F, Dirou S, Forest V, Sauzeau V, Hassoun D, Chesné J, Cheminant-Muller MA, Sagan C, Magnan A, Lemarchand P. Mesenchymal stem cells induce suppressive macrophages through phagocytosis in a mouse model of asthma. Stem Cells. 2016;34(7):1836–1845. doi: 10.1002/stem.2344. [DOI] [PubMed] [Google Scholar]
- 39.Fang S-B, Zhang H-Y, Jiang A-Y, Fan X-L, Lin Y-D, Li C-L, Wang C, Meng X-C, Fu Q-L. Human iPSC-MSCs prevent steroid-resistant neutrophilic airway inflammation via modulating Th17 phenotypes. Stem Cell Res Ther. 2018;9(1):147. doi: 10.1186/s13287-018-0897-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Castro LL, Kitoko JZ, Xisto DG, Olsen PC, Guedes HLM, Morales MM, Lopes-Pacheco M, Cruz FF, Rocco PRM. Multiple doses of adipose tissue-derived mesenchymal stromal cells induce immunosuppression in experimental asthma. Stem Cells Transl Med. 2020;9(2):250–260. doi: 10.1002/sctm.19-0120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lathrop MJ, Brooks EM, Bonenfant NR, Sokocevic D, Borg ZD, Goodwin M, Loi R, Cruz F, Dunaway CW, Steele C, Weiss DJ. Mesenchymal stromal cells mediate aspergillus hyphal extract-induced allergic airway inflammation by inhibition of the Th17 signaling pathway. Stem Cells Transl Med. 2014;3(2):194–205. doi: 10.5966/sctm.2013-0061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Royce SG, Shen M, Patel KP, Huuskes BM, Ricardo SD, Samuel CS. Mesenchymal stem cells and serelaxin synergistically abrogate established airway fibrosis in an experimental model of chronic allergic airways disease. Stem Cell Res. 2015;15(3):495–505. doi: 10.1016/j.scr.2015.09.007. [DOI] [PubMed] [Google Scholar]
- 43.Urbanek K, De Angelis A, Spaziano G, Piegari E, Matteis M, Cappetta D, Esposito G, Russo R, Tartaglione G, De Palma R. Intratracheal administration of mesenchymal stem cells modulates tachykinin system, suppresses airway remodeling and reduces airway hyperresponsiveness in an animal model. PLoS One. 2016;11(7):e0158746. doi: 10.1371/journal.pone.0158746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Leibel S, Post M. Endogenous and exogenous stem/progenitor cells in the lung and their role in the pathogenesis and treatment of pediatric lung disease. Front Pediatr. 2016;4:36. doi: 10.3389/fped.2016.00036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Leeman KT, Fillmore CM, Kim CF. Lung stem and progenitor cells in tissue homeostasis and disease. Curr Top Dev Biol. 2014;107:207–233. doi: 10.1016/B978-0-12-416022-4.00008-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Suzuki T, Suzuki S, Fujino N, Ota C, Yamada M, Suzuki T, Yamaya M, Kondo T, Kubo H. c-kit immunoexpression delineates a putative endothelial progenitor cell population in developing human lungs. Am J Phys Lung Cell Mol Phys. 2014;306(9):L855–L865. doi: 10.1152/ajplung.00211.2013. [DOI] [PubMed] [Google Scholar]
- 47.Srour N, Thébaud B. Stem cells in animal asthma models: a systematic review. Cytotherapy. 2014;16(12):1629–1642. doi: 10.1016/j.jcyt.2014.08.008. [DOI] [PubMed] [Google Scholar]
- 48.Rahbarghazi R, Keyhanmanesh R, Aslani MR, Hassanpour M, Ahmadi M. Bone marrow mesenchymal stem cells and condition media diminish inflammatory adhesion molecules of pulmonary endothelial cells in an ovalbumin-induced asthmatic rat model. Microvasc Res. 2019;121:63–70. doi: 10.1016/j.mvr.2018.10.005. [DOI] [PubMed] [Google Scholar]
- 49.Prockop DJ, Oh JY. Medical therapies with adult stem/progenitor cells (MSCs): a backward journey from dramatic results in vivo to the cellular and molecular explanations. J Cell Biochem. 2012;113(5):1460–1469. doi: 10.1002/jcb.24046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Shigemura N, Okumura M, Mizuno S, Imanishi Y, Nakamura T, Sawa Y. Autologous transplantation of adipose tissue-derived stromal cells ameliorates pulmonary emphysema. Am J Transplant. 2006;6(11):2592–2600. doi: 10.1111/j.1600-6143.2006.01522.x. [DOI] [PubMed] [Google Scholar]
- 51.Luan Y, Zhang X, Kong F, Cheng G-H, Qi T-G, Zhang Z-H. Mesenchymal stem cell prevention of vascular remodeling in high flow-induced pulmonary hypertension through a paracrine mechanism. Int Immunopharmacol. 2012;14(4):432–437. doi: 10.1016/j.intimp.2012.08.001. [DOI] [PubMed] [Google Scholar]
- 52.Abreu SC, Xisto DG, de Oliveira TB, Blanco NG, de Castro LL, Kitoko JZ, Olsen PC, Lopes-Pacheco M, Morales MM, Weiss DJ, Rocco PRM. Serum from asthmatic mice potentiates the therapeutic effects of mesenchymal stromal cells in experimental allergic asthma. Stem Cells Transl Med. 2018;8(3):301–312. doi: 10.1002/sctm.18-0056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Prockop DJ. The exciting prospects of new therapies with mesenchymal stromal cells. Cytotherapy. 2017;19(1):1–8. doi: 10.1016/j.jcyt.2016.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Galleu A, Riffo-Vasquez Y, Trento C, Lomas C, Dolcetti L, Cheung TS, von Bonin M, Barbieri L, Halai K, Ward S. Apoptosis in mesenchymal stromal cells induces in vivo recipient-mediated immunomodulation. Sci Transl Med. 2017;9(416):eaam7828. doi: 10.1126/scitranslmed.aam7828. [DOI] [PubMed] [Google Scholar]
- 55.Thum T, Bauersachs J, Poole-Wilson PA, Volk H-D, Anker SD. The dying stem cell hypothesis: immune modulation as a novel mechanism for progenitor cell therapy in cardiac muscle. J Am Coll Cardiol. 2005;46(10):1799–1802. doi: 10.1016/j.jacc.2005.07.053. [DOI] [PubMed] [Google Scholar]
- 56.Chen F, Liu Y, Wong N-K, Xiao J, So K-F. Oxidative stress in stem cell aging. Cell Transplant. 2017;26(9):1483–1495. doi: 10.1177/0963689717735407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chen L, Zhang W, Yue H, Han Q, Chen B, Shi M, Li J, Li B, You S, Shi Y. Effects of human mesenchymal stem cells on the differentiation of dendritic cells from CD34+ cells. Stem Cells Dev. 2007;16(5):719–732. doi: 10.1089/scd.2007.0065. [DOI] [PubMed] [Google Scholar]
- 58.Bernardo Maria E, Fibbe Willem E. Mesenchymal stromal cells: sensors and switchers of inflammation. Cell Stem Cell. 2013;13(4):392–402. doi: 10.1016/j.stem.2013.09.006. [DOI] [PubMed] [Google Scholar]
- 59.Vizoso FJ, Eiro N, Costa L, Esparza P, Landin M, Diaz-Rodriguez P, Schneider J, Perez-Fernandez R. Mesenchymal stem cells in homeostasis and systemic diseases: hypothesis, evidences, and therapeutic opportunities. Int J Mol Sci. 2019;20(15):3738. doi: 10.3390/ijms20153738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mosna F, Sensebe L, Krampera M. Human bone marrow and adipose tissue mesenchymal stem cells: a user’s guide. Stem Cells Dev. 2010;19(10):1449–1470. doi: 10.1089/scd.2010.0140. [DOI] [PubMed] [Google Scholar]
- 61.Melief SM, Zwaginga JJ, Fibbe WE, Roelofs H. Adipose tissue-derived multipotent stromal cells have a higher immunomodulatory capacity than their bone marrow-derived counterparts. Stem Cells Transl Med. 2013;2(6):455–463. doi: 10.5966/sctm.2012-0184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Li X, Bai J, Ji X, Li R, Xuan Y, Wang Y. Comprehensive characterization of four different populations of human mesenchymal stem cells as regards their immune properties, proliferation and differentiation. Int J Mol Med. 2014;34(3):695–704. doi: 10.3892/ijmm.2014.1821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hoffman AM, Paxson JA, Mazan MR, Davis AM, Tyagi S, Murthy S, Ingenito EP. Lung-derived mesenchymal stromal cell post-transplantation survival, persistence, paracrine expression, and repair of elastase-injured lung. Stem Cells Dev. 2011;20(10):1779–1792. doi: 10.1089/scd.2011.0105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nystedt J, Anderson H, Tikkanen J, Pietilä M, Hirvonen T, Takalo R, Heiskanen A, Satomaa T, Natunen S, Lehtonen S. Cell surface structures influence lung clearance rate of systemically infused mesenchymal stromal cells. Stem Cells. 2013;31(2):317–326. doi: 10.1002/stem.1271. [DOI] [PubMed] [Google Scholar]
- 65.Li Y, Li H, Cao Y, Wu F, Ma W, Wang Y, Sun S. Placenta-derived mesenchymal stem cells improve airway hyperresponsiveness and inflammation in asthmatic rats by modulating the Th17/Treg balance. Mol Med Rep. 2017;16(6):8137–8145. doi: 10.3892/mmr.2017.7605. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 66.Abreu SC, Antunes MA, Xisto DG, Cruz FF, Branco VC, Bandeira E, Zola Kitoko J, de Araújo AF, Dellatorre-Texeira L, Olsen PC, Weiss DJ, Diaz BL, Morales MM, Rocco PRM. Bone marrow, adipose, and lung tissue-derived murine mesenchymal stromal cells release different mediators and differentially affect airway and lung parenchyma in experimental asthma. Stem Cells Transl Med. 2017;6(6):1557–1567. doi: 10.1002/sctm.16-0398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Nemeth K, Keane-Myers A, Brown JM, Metcalfe DD, Gorham JD, Bundoc VG, Hodges MG, Jelinek I, Madala S, Karpati S, Mezey E. Bone marrow stromal cells use TGF-beta to suppress allergic responses in a mouse model of ragweed-induced asthma. Proc Natl Acad Sci U S A. 2010;107(12):5652–5657. doi: 10.1073/pnas.0910720107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Goodwin M, Sueblinvong V, Eisenhauer P, Ziats NP, LeClair L, Poynter ME, Steele C, Rincon M, Weiss DJ. Bone marrow-derived mesenchymal stromal cells inhibit Th2-mediated allergic airways inflammation in mice. Stem Cells. 2011;29(7):1137–1148. doi: 10.1002/stem.656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Park HK, Cho KS, Park HY, Shin DH, Kim YK, Jung JS, Park SK, Roh HJ. Adipose-derived stromal cells inhibit allergic airway inflammation in mice. Stem Cells Dev. 2010;19:1811–1818. doi: 10.1089/scd.2009.0513. [DOI] [PubMed] [Google Scholar]
- 70.Firinci F, Karaman M, Baran Y, Bagriyanik A, Ayyildiz ZA, Kiray M, Kozanoglu I, Yilmaz O, Uzuner N, Karaman O. Mesenchymal stem cells ameliorate the histopathological changes in a murine model of chronic asthma. Int Immunopharmacol. 2011;11(8):1120–1126. doi: 10.1016/j.intimp.2011.03.009. [DOI] [PubMed] [Google Scholar]
- 71.Kavanagh H, Mahon BP. Allogeneic mesenchymal stem cells prevent allergic airway inflammation by inducing murine regulatory T cells. Allergy. 2011;66(4):523–531. doi: 10.1111/j.1398-9995.2010.02509.x. [DOI] [PubMed] [Google Scholar]
- 72.Park WS, Ahn SY, Sung SI, Ahn J-Y, Chang YS. Strategies to enhance paracrine potency of transplanted mesenchymal stem cells in intractable neonatal disorders. Pediatr Res. 2018;83(1):214–222. doi: 10.1038/pr.2017.249. [DOI] [PubMed] [Google Scholar]
- 73.Işık S, Karaman M, Adan A, Kıray M, Bağrıyanık HA, Sözmen ŞÇ, Kozanoğlu İ, Karaman Ö, Baran Y, Uzuner N. Intraperitoneal mesenchymal stem cell administration ameliorates allergic rhinitis in the murine model. Eur Arch Otorhinolaryngol. 2017;274(1):197–207. doi: 10.1007/s00405-016-4166-3. [DOI] [PubMed] [Google Scholar]
- 74.Sakine I, Nevin U, Meral K, Özkan K, Müge K, İlknur K, Hüsnü Alper B, Zeynep A-A, Melis Kartal Y, Yusuf B. Effects of intraperitoneal injection of allogeneic bone marrow-derived mesenchymal stem cells on bronchiolitis obliterans in mice model. Iran J Allergy Asthma Immunol. 2017;16(3):205–18. [PubMed]
- 75.Brooke G, Cook M, Blair C, Han R, Heazlewood C, Jones B, Kambouris M, Kollar K, McTaggart S, Pelekanos R Therapeutic applications of mesenchymal stromal cells. In: Seminars in cell & developmental biology, 2007. Elsevier, pp 846–858. [DOI] [PubMed]
- 76.Keyhanmanesh R, Rahbarghazi R, Aslani MR, Hassanpour M, Ahmadi M. Systemic delivery of mesenchymal stem cells condition media in repeated doses acts as magic bullets in restoring IFN-γ/IL-4 balance in asthmatic rats. Life Sci. 2018;212:30–36. doi: 10.1016/j.lfs.2018.09.049. [DOI] [PubMed] [Google Scholar]
- 77.Kan C, Chen L, Hu Y, Lu H, Li Y, Kessler JA, Kan L. Microenvironmental factors that regulate mesenchymal stem cells: lessons learned from the study of heterotopic ossification. Histol Histopathol. 2017;32(10):977. doi: 10.14670/HH-11-890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Trzil JE, Masseau I, Webb TL, Chang C-H, Dodam JR, Liu H, Quimby JM, Dow SW, Reinero CR. Intravenous adipose-derived mesenchymal stem cell therapy for the treatment of feline asthma: a pilot study. J Feline Med Surg. 2016;18(12):981–990. doi: 10.1177/1098612X15604351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Liang X, Ding Y, Zhang Y, Tse H-F, Lian Q. Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell Transplant. 2014;23(9):1045–1059. doi: 10.3727/096368913x667709. [DOI] [PubMed] [Google Scholar]
- 80.Horie M, Choi H, Lee RH, Reger RL, Ylostalo J, Muneta T, Sekiya I, Prockop DJ. Intra-articular injection of human mesenchymal stem cells (MSCs) promote rat meniscal regeneration by being activated to express Indian hedgehog that enhances expression of type II collagen. Osteoarthr Cartil. 2012;20(10):1197–1207. doi: 10.1016/j.joca.2012.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Spees JL, Olson SD, Ylostalo J, Lynch PJ, Smith J, Perry A, Peister A, Wang MY, Prockop DJ. Differentiation, cell fusion, and nuclear fusion during ex vivo repair of epithelium by human adult stem cells from bone marrow stroma. Proc Natl Acad Sci U S A. 2003;100(5):2397–2402. doi: 10.1073/pnas.0437997100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hocking AM, Gibran NS. Mesenchymal stem cells: paracrine signaling and differentiation during cutaneous wound repair. Exp Cell Res. 2010;316(14):2213–2219. doi: 10.1016/j.yexcr.2010.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Harrell CR, Jovicic N, Djonov V, Arsenijevic N, Volarevic V. Mesenchymal stem cell-derived exosomes and other extracellular vesicles as new remedies in the therapy of inflammatory diseases. Cells. 2019;8(12):1605. doi: 10.3390/cells8121605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wang T, Nasser MI, Shen J, Qu S, He Q, Zhao M. Functions of exosomes in the triangular relationship between the tumor, inflammation, and immunity in the tumor microenvironment. J Immunol Res. 2019;2019. [DOI] [PMC free article] [PubMed]
- 85.Cruz FF, Borg ZD, Goodwin M, Sokocevic D, Wagner DE, Coffey A, Antunes M, Robinson KL, Mitsialis SA, Kourembanas S. Systemic administration of human bone marrow-derived mesenchymal stromal cell extracellular vesicles ameliorates aspergillus hyphal extract-induced allergic airway inflammation in immunocompetent mice. Stem Cells Transl Med. 2015;4(11):1302–1316. doi: 10.5966/sctm.2014-0280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Vizoso F, Eiro N, Cid S, Schneider J, Perez-Fernandez R. Mesenchymal stem cell secretome: toward cell-free therapeutic strategies in regenerative medicine. Int J Mol Sci. 2017;18(9):1852. doi: 10.3390/ijms18091852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gazdic M, Volarevic V, Arsenijevic N, Stojkovic M. Mesenchymal stem cells: a friend or foe in immune-mediated diseases. Stem Cell Rev Rep. 2015;11(2):280–287. doi: 10.1007/s12015-014-9583-3. [DOI] [PubMed] [Google Scholar]
- 88.Paliwal S, Chaudhuri R, Agrawal A, Mohanty S. Regenerative abilities of mesenchymal stem cells through mitochondrial transfer. J Biomed Sci. 2018;25(1):31. doi: 10.1186/s12929-018-0429-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ahmad T, Mukherjee S, Pattnaik BR, Kumar M, Singh S, Rehman R, Jha A, Wani M, Mabalirajan U, Ghosh B. Miro 1 knockdown in stem cells inhibits mitochondrial donation mediated rescue of bronchial epithelial injury. Biophysical J. 2013;104(2):659a. [Google Scholar]
- 90.Islam MN, Das SR, Emin MT, Wei M, Sun L, Westphalen K, Rowlands DJ, Quadri SK, Bhattacharya S, Bhattacharya J. Mitochondrial transfer from bone-marrow–derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat Med. 2012;18(5):759. doi: 10.1038/nm.2736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Li X, Zhang Y, Yeung SC, Liang Y, Liang X, Ding Y, Ip MS, Tse H-F, Mak JC, Lian Q. Mitochondrial transfer of induced pluripotent stem cell–derived mesenchymal stem cells to airway epithelial cells attenuates cigarette smoke–induced damage. Am J Respir Cell Mol Biol. 2014;51(3):455–465. doi: 10.1165/rcmb.2013-0529OC. [DOI] [PubMed] [Google Scholar]
- 92.Chan TK, Tan WD, Peh HY, Wong WF. Aeroallergens induce reactive oxygen species production and DNA damage and dampen antioxidant responses in bronchial epithelial cells. J Immunol. 2017;199(1):39–47. doi: 10.4049/jimmunol.1600657. [DOI] [PubMed] [Google Scholar]
- 93.Okazaki T, Magaki T, Takeda M, Kajiwara Y, Hanaya R, Sugiyama K, Arita K, Nishimura M, Kato Y, Kurisu K. Intravenous administration of bone marrow stromal cells increases survivin and Bcl-2 protein expression and improves sensorimotor function following ischemia in rats. Neurosci Lett. 2008;430(2):109–114. doi: 10.1016/j.neulet.2007.10.046. [DOI] [PubMed] [Google Scholar]
- 94.Li J, Zhou J, Zhang D, Song Y, She J, Bai C. Bone marrow-derived mesenchymal stem cells enhance autophagy via PI3K/AKT signalling to reduce the severity of ischaemia/reperfusion-induced lung injury. J Cell Mol Med. 2015;19(10):2341–2351. doi: 10.1111/jcmm.12638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Zhou Z, You Z. Mesenchymal stem cells alleviate LPS-induced acute lung injury in mice by MiR-142a-5p-controlled pulmonary endothelial cell autophagy. Cell Physiol Biochem. 2016;38(1):258–266. doi: 10.1159/000438627. [DOI] [PubMed] [Google Scholar]
- 96.Specjalski K, Jassem E. MicroRNAs: potential biomarkers and targets of therapy in allergic diseases? Arch Immunol Ther Exp. 2019;67(4):213–223. doi: 10.1007/s00005-019-00547-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Tang G-N, Li C-L, Yao Y, Xu Z-B, Deng M-X, Wang S-Y, Sun Y-Q, Shi J-B, Fu Q-L. MicroRNAs involved in asthma after mesenchymal stem cells treatment. Stem Cells Dev. 2016;25(12):883–896. doi: 10.1089/scd.2015.0339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mattes J, Collison A, Plank M, Phipps S, Foster PS. Antagonism of microRNA-126 suppresses the effector function of TH2 cells and the development of allergic airways disease. Proc Natl Acad Sci. 2009;106(44):18704–18709. doi: 10.1073/pnas.0905063106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ahmadi M, Rahbarghazi R, Shahbazfar A-A, Baghban H, Keyhanmanesh R. Bone marrow mesenchymal stem cells modified pathological changes and immunological responses in ovalbumin-induced asthmatic rats possibly by the modulation of miRNA155 and miRNA133. Gen Physiol Biophys. 2018;37(3):263–274. doi: 10.4149/gpb_2017052. [DOI] [PubMed] [Google Scholar]
- 100.Li C-L, Xu Z-B, Fan X-L, Chen H-X, Yu Q-N, Fang S-B, Wang S-Y, Lin Y-D, Fu Q-L. microRNA-21 mediates the protective effects of mesenchymal stem cells derived from iPSCs to human bronchial epithelial cell injury under hypoxia. Cell Transplant. 2018;27(3):571–583. doi: 10.1177/0963689718767159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Camussi G, Deregibus MC, Cantaluppi V. Role of stem-cell-derived microvesicles in the paracrine action of stem cells. Biochem Soc Trans. 2013;41(1):283–7. [DOI] [PubMed]
- 102.Zhang B, Yin Y, Lai RC, Tan SS, Choo ABH, Lim SK. Mesenchymal stem cells secrete immunologically active exosomes. Stem Cells Dev. 2014;23(11):1233–1244. doi: 10.1089/scd.2013.0479. [DOI] [PubMed] [Google Scholar]
- 103.Coyle AJ, Le Gros G, Bertrand C, Tsuyuki S, Heusser CH, Kopf M, Anderson GP. Interleukin-4 is required for the induction of lung Th2 mucosal immunity. Am J Respir Cell Mol Biol. 1995;13(1):54–59. doi: 10.1165/ajrcmb.13.1.7598937. [DOI] [PubMed] [Google Scholar]
- 104.Harrell CR, Sadikot R, Pascual J, Fellabaum C, Jankovic MG, Jovicic N, Djonov V, Arsenijevic N, Volarevic V. Mesenchymal stem cell-based therapy of inflammatory lung diseases: current understanding and future perspectives. Stem Cells Int. 2019;2019:4236973. doi: 10.1155/2019/4236973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Heidarzadeh M, Roodbari F, Hassanpour M, Ahmadi M, Saberianpour S, Rahbarghazi R. Toll-like receptor bioactivity in endothelial progenitor cells. Cell Tissue Res. 2020;379(2):223–230. doi: 10.1007/s00441-019-03119-2. [DOI] [PubMed] [Google Scholar]
- 106.Le Blanc K, Davies LC. Mesenchymal stromal cells and the innate immune response. Immunol Lett. 2015;168(2):140–146. doi: 10.1016/j.imlet.2015.05.004. [DOI] [PubMed] [Google Scholar]
- 107.Monsel A, Zhu YG, Gennai S, Hao Q, Hu S, Rouby JJ, Rosenzwajg M, Matthay MA, Lee JW. Therapeutic effects of human mesenchymal stem cell-derived microvesicles in severe pneumonia in mice. Am J Respir Crit Care Med. 2015;192(3):324–336. doi: 10.1164/rccm.201410-1765OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Tu Z, Li Q, Bu H, Lin F. Mesenchymal stem cells inhibit complement activation by secreting factor H. Stem Cells Dev. 2010;19(11):1803–1809. doi: 10.1089/scd.2009.0418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Kuo Y-C, Li Y-SJ, Zhou J, Shih Y-RV, Miller M, Broide D, Lee OK-S, Chien S. Human mesenchymal stem cells suppress the stretch-induced inflammatory miR-155 and cytokines in bronchial epithelial cells. PLoS One. 2013;8(8):e71342. doi: 10.1371/journal.pone.0071342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Tzouvelekis A, Paspaliaris V, Koliakos G, Ntolios P, Bouros E, Oikonomou A, Zissimopoulos A, Boussios N, Dardzinski B, Gritzalis D. A prospective, non-randomized, no placebo-controlled, phase Ib clinical trial to study the safety of the adipose derived stromal cells-stromal vascular fraction in idiopathic pulmonary fibrosis. J Transl Med. 2013;11(1):171. doi: 10.1186/1479-5876-11-171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Popp F, Eggenhofer E, Renner P, Slowik P, Lang S, Kaspar H, Geissler E, Piso P, Schlitt H, Dahlke M. Mesenchymal stem cells can induce long-term acceptance of solid organ allografts in synergy with low-dose mycophenolate. Transpl Immunol. 2008;20(1–2):55–60. doi: 10.1016/j.trim.2008.08.004. [DOI] [PubMed] [Google Scholar]
- 112.Eggenhofer E, Steinmann J, Renner P, Slowik P, Piso P, Geissler E, Schlitt H, Dahlke M, Popp F. Mesenchymal stem cells together with mycophenolate mofetil inhibit antigen presenting cell and T cell infiltration into allogeneic heart grafts. Transpl Immunol. 2011;24(3):157–163. doi: 10.1016/j.trim.2010.12.002. [DOI] [PubMed] [Google Scholar]
- 113.Ren G, Su J, Zhang X, Zhao LX, Ling W, L’huillie A, Zhang J, Lu Y, Roberts AI, Ji W. Species variation in the mechanisms of mes enchymal stem cell-mediated immunosuppression. Stem Cells. 2009;27:1954–1962. doi: 10.1002/stem.118. [DOI] [PubMed] [Google Scholar]
- 114.Bonfield TL, Koloze M, Lennon DP, Zuchowski B, Yang SE, Caplan AI. Human mesenchymal stem cells suppress chronic airway inflammation in the murine ovalbumin asthma model. Mol Physiol : Am J Physiol Lung Cell. 2010;299:L760–L770. doi: 10.1152/ajplung.00182.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Bonfield TL, Nolan Koloze MT, Lennon DP, Caplan AI. Defining human mesenchymal stem cell efficacy in vivo. J Inflamm (Lond) 2010;7:51. doi: 10.1186/1476-9255-7-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Song X, Xie S, Lu K, Wang C. Mesenchymal stem cells alleviate experimental asthma by inducing polarization of alveolar macrophages. Inflammation. 2015;38(2):485–492. doi: 10.1007/s10753-014-9954-6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.