Abstract
A hypothesis concerning the potential utility of surfactant supplementation for the treatment of critically ill patients with COVID-19 is proposed, along with a brief summary of the data in the literature supporting this idea. It is thought that surfactant, which is already approved by the Food and Drug Administration for intratracheal administration to treat neonatal respiratory distress syndrome in pre-term infants, could benefit COVID-19-infected individuals by: (1) restoring surfactant damaged by lung infection and/or decreased due to the virus-induced death of the type II pneumocytes that produce it and (2) reducing surface tension to decrease the work of breathing and limit pulmonary edema. In addition, a constituent of surfactant, phosphatidylglycerol, could mitigate COVID-19-induced lung pathology by: (3) decreasing excessive innate immune system stimulation via its inhibition of toll-like receptor-2 and -4 activation by microbial components and cellular proteins released by damaged cells, thereby limiting inflammation and the resultant pulmonary edema, and (4) possibly blocking spread of the viral infection to non-infected cells in the lung. Therefore, it is suggested that surfactant preparations containing phosphatidylglycerol be tested for their ability to improve lung function in critically ill patients with COVID-19.
Abbreviations: ARDS, acute respiratory distress syndrome; DAMP, danger- or damage-associated molecular pattern; IL, interleukin; NFκB, nuclear factor kappa-light-chain-enhancer of activated B cells; PAMP, pathogen-associated molecular pattern; PG, phosphatidylglycerol; sPLA2, secretory phospholipase A2; TLR, toll-like receptor; TNFα, tumor necrosis factor-alpha
Keywords: COVID-19, Innate immunity, Phosphatidylglycerol, SARS-CoV-2, Surfactant
Introduction
COVID-19, caused by the novel coronavirus SARS-CoV-2, has resulted in massive morbidity and mortality, as well as profound economic difficulties due to the necessity for quarantining to contain and mitigate the pandemic. Although many people who become infected exhibit only mild or moderate symptoms, others develop severe symptoms, and COVID-19 appears to be more deadly than influenza, especially in older individuals and those with pre-existing conditions. Treatment to date is mainly symptomatic supportive care including invasive or non-invasive ventilation. In a recent retrospective study of 52 Chinese patients with COVID-19 requiring intensive care, more than 60% of the patients died [1]. Of the non-survivors about 80% of the patients developed acute respiratory distress syndrome (ARDS) [1], and respiratory failure associated with ARDS is the leading cause of COVID-19 mortality [2].
ARDS is characterized by lung inflammation and pulmonary edema, which reduces gas exchange and leads to hypoxemia and dyspnea, often requiring mechanical ventilation to provide sufficient oxygenation. ARDS is also accompanied by enhanced secretory phospholipase A2 (sPLA2) activity in the lungs [3], [4]; sPLA2 degrades the phospholipids that are components of surfactant, including phosphatidylglycerol (PG) (reviewed in [5]). Indeed, a recent study has demonstrated an increase in the activity of an sPLA2 that preferentially hydrolyzes PG, as well as a significant decrease in PG in the bronchoalveolar lavage fluid of ARDS patients versus normal control subjects [3]. The impairment of surfactant function not only can increase surface tension and reduce lung compliance but may also further exacerbate pulmonary edema, since surfactant helps to reduce fluid infiltration into the alveoli through its reduction of surface tension [6], [7]. Knowledge about this sequence of events has led to studies in humans testing the efficacy of exogenous surfactant in the treatment of ARDS, and some results have been promising [8], [9], [10], [11], although meta-analyses have largely failed to show an effect of exogenous surfactant administration on the survival of adult ARDS patients (e.g., [12], [13]). On the other hand, another meta-analysis determined a benefit of surfactant administration on oxygenation levels and mortality in those patients with severe ARDS caused by pneumonia or aspiration of gastric contents [14], suggesting that co-morbidities other than ARDS may potentially determine the effect (or lack thereof) of surfactant administration on survival.
In contrast, the pulmonary failure induced by COVID-19 seems to differ in many respects from other types of ARDS [15], [16]. For example, many COVID-19 patients initially present with hypoxemia with maintained lung compliance and low elastance, termed the L type presentation [17]. These L-type patients can often be treated with oxygen supplementation and prone positioning, or if they are intubated due to worsening hypoxia, mechanical ventilation at low positive end expiratory pressures (PEEP). However, L-type patients often transition into the second or H-type clinical presentation [17], in which they exhibit high elastance and low compliance and usually require mechanical ventilation at higher PEEP [18]; low pulmonary compliance portends worsening lung disease manifested by atelectasis and increasing hypoxia. Therefore, only the H-type mimics the lung parameters observed in pre-term infants that produce minimal surfactant, suggesting that at least in the early stages, COVID-19 patients exhibiting L-type disease may retain some pulmonary surfactant activity.
A key role of pulmonary surfactant is to reduce surface tension and prevent alveolar atelectasis at end expiration. In the absence of active surfactant, high surface tension at the air-liquid interface in the alveoli creates collapsing forces [19]. In addition, since surface tension draws fluid from the capillaries into the alveolar spaces, surfactant decreases pulmonary fluid accumulation by reducing surface tension to maintain airway dryness [20]. Increased surface tension related to surfactant dysfunction also alters alveolar capillary shape and pulmonary blood flow to exacerbate hypoxemia [19]. On the other hand, during severe respiratory distress with mechanical ventilation, the role of higher PEEP is to keep the alveoli “recruited” or, in other words, to prevent end-expiratory alveolar collapse. However, mechanical ventilation and high PEEP represent a double-edged sword: maintaining or improving oxygenation while causing alveolar lung injury. Mechanical ventilation with high PEEP may also reduce the ability of surfactant to lower surface tension, since compression of surfactant to an area of less than 50% of its original surface area by higher pressures can result in rupturing of the film on re-expansion, resulting in compromised surface tension-reducing capacity [21], [22]. In addition, the stresses of mechanical ventilation also stimulate the inflammatory response [21].
Thus, it seems likely that another function of surfactant may be important: its ability to dampen the inflammatory response to microbial components. Thus, at least one of the surfactant phospholipids, PG, has been shown to inhibit activation of toll-like receptors (TLR) of the lung innate immune system by microbial components (reviewed in [23]). Voelker and colleagues have shown that PG inhibits TLR2 and TLR4 activation by microbial components, also known as pathogen-associated molecular patterns (PAMPs), such as acylated lipopeptides and lipopolysaccharide [23]. This inhibition then results in reduced production of inflammatory mediators and decreased lung inflammation and damage [24], [25], [26]. These results are also consistent with the results of Wu at al. [27], who demonstrated that PG inhibits endotoxin-stimulated activation of nuclear factor-kappaB (NFκB), a transcription factor associated with inflammation, to reduce Type IIA secretory phospholipase A2 levels/activity in macrophages. The mechanism of action of PG seems to be related to the ability of the TLR2 and TLR4 co-receptor CD14 to bind this phospholipid and somehow prevent TLR activation [26], [28]. Indeed, Martin et al. [29] have recently suggested blocking CD14, but with inhibitory antibodies, to control inflammation in COVID-19.
Of note, PAMPs are not the only molecules that can activate TLRs. TLR activation can also be induced by endogenous proteins that are released by damaged or stressed cells, the so-called danger- or damage-associated molecular patterns (DAMPs) (reviewed in [30]). Many such TLR-stimulating DAMPs have been identified (reviewed in [31]), including several heat shock proteins, high mobility group B1 (HMGB1) and fibrinogen [31], and these can be released extracellularly upon cell damage. We recently showed that PG can inhibit DAMP-induced inflammatory mediator production [32] and skin inflammation [32], [33]. Specifically, PG inhibits TLR2 and TLR4 activation by the DAMPs S100A9 and beta-defensin-2 [32]. PG can also inhibit TLR2 and TLR4 activation in response to PAMPs in several cell types and in TLR2 and TLR4 reporter cell lines [34], with minimal effects on the activation of, or stimulation of inflammatory mediator expression by, other pattern recognition receptors, such as the TLR7/8 that recognizes single-stranded RNA. In addition, supplementation of surfactant with additional PG (to a molar percentage of 6%) preserves lung function and prevents alveolar epithelial injury and the expression of pro-fibrotic mediators in a neonatal pig triple injury model of ARDS [35]. The ability of PG to protect against cell injury would be expected to be beneficial in COVID-19 ARDS. Furthermore, COVID-19 patients have been reported to exhibit elevated levels of tumor necrosis factor-alpha (TNFα) [2], and drugs targeting pro-inflammatory mediators, such as interleukin (IL)-1 and IL-6 have been proposed or are in use for the treatment of COVID-19 [36], [37]. PG has been shown to inhibit the expression of IL-1α, IL-1β, IL-6, and/or TNFα, as well as IL-8, interferon-gamma and/or macrophage inflammatory protein-2, in response to TLR activation by PAMPs and DAMPs [24], [25], [26], [28], [32], [34], [38], [39]. In turn, several of these inflammatory mediators (e.g., IL-6 and TNFα) are also known to increase the levels of certain sPLA2s, in particular that encoded by the gene PLA2G2A [3], which would decrease PG levels even further. Finally, excessive inflammation (e.g., markedly increased C-reactive protein and D-dimer levels) is associated with the hypercoagulopathy sometimes seen in COVID-19 patients [40]. Therefore, the ability of PG to inhibit PAMP- and DAMP-induced might also decrease these COVID-19 sequelae as well.
Hypothesis
Collectively, these results have led to the current hypothesis that PG, in the form of exogenous surfactant, might be efficacious in treating the symptoms of COVID-19. By analogy with SARS-CoV [41], [42], SARS-CoV-2 is thought to target alveolar type-II cells [43], [44], the lung cells that produce surfactant; the resulting release of endogenous molecules by these damaged cells would presumably activate TLRs and stimulate inflammatory mediator production and inflammation. These effects likely would, together with the gradual reduction in surfactant resulting from the death of these type II pneumocytes (and possibly the increased activity of sPLA2) [3], [4], [5], promote the pulmonary edema that is a hallmark of COVID-19. The pulmonary edema, in turn, further impairs gas exchange and leads to ARDS with further hypoxemia and dyspnea. However, the initial presentation might be expected to show differences from the respiratory distress seen in pre-term infants: phosphatidylcholine represents approximately two-thirds to three-quarters of pulmonary surfactant lipid content [45], [46] and thus provides the majority of its surfactant activity. Therefore, gradual loss of phosphatidylcholine would allow maintenance of compliance despite enhanced inflammation resulting from decreased levels of PG, which comprises only 9–12% of surfactant phospholipid [45], [46], and the resultant pulmonary edema. Presumably, loss of the anti-surface tension effects of surfactant would only occur once large numbers of Type II alveolar cells were destroyed and phosphatidylcholine was severely depleted. At this point, then, patients would transition to the H-type clinical presentation, with the low compliance more typical of neonatal respiratory distress syndrome.
Pulmonary administration of exogenous surfactant would be expected to counter this sequence of events in multiple ways: (1) it would restore the levels of surfactant to protect against increased surface tension in the lung; (2) it would inhibit activation of the innate immune system by released DAMPs to reduce inflammation and inflammatory damage; and (3) it would decrease pulmonary edema through the combination of the first two effects. In addition, it is thought that in some individuals, COVID-19-related morbidity and mortality may be related to an over-reaction of the immune system and a “cytokine storm” [2], [37], [47], [48]. By inhibiting innate immune system activation and release of pro-inflammatory mediators that recruit and activate additional immune cells, including those of the adaptive immune system, PG would likely interrupt this process of immune system hyper-responsiveness, acting as a dampening mechanism, or rheostat, to regulate lung inflammation [23]. Finally, it is known that pulmonary surfactant can facilitate recruitment of collapsed airways and offer protection from mechanical ventilation-induced lung injury. Thus, exogenous surfactant therapy may restore or replenish insufficient or dysfunctional endogenous surfactant activity and improve outcomes in COVID-19. Thus, we are proposing that PG-containing surfactant medications that are already approved by the Food and Drug Administration for the treatment of neonatal respiratory distress syndrome (Table 1 ) be administered intratracheally via bronchoscopy to COVID-19 patients with severe acute respiratory distress syndrome. It should be noted that natural and second-generation synthetic surfactant preparations have been found to exhibit increased efficacy for improving neonatal respiratory distress syndrome relative to first-generation protein-free surfactant medications like Exosurf®. These results are consistent with data indicating the importance of certain surfactant proteins to improve the effect of surfactant on surface tension [49] and others to reduce microbial infection ([50], [51] and reviewed in [52]).
Table 1.
Surfactant Medications Approved by the Food and Drug Administration for Treatment of Neonatal Respiratory Distress Syndrome.
| Natural Surfactants | Synthetic Surfactants | ||||
|---|---|---|---|---|---|
| Generic Name | Beractant | Calfactant | Poractant alfa | Culfosceril palmitate* | Lucinactant |
| Brand Name | Survanta | Infasurf | Curosurf | Exosurf | Surfaxin |
| Company | Abbott | ONY Biotech | Chiesi | GSK† | Windtreeƒ |
| Source | Minced bovine lung extract | Calf lung lavage | Minced porcine lung extract | First-generation synthetic | Second generation synthetic |
| Protein/amount | SP-B and –C (1 to 21 mg/mM PL) | SP-B and –C (13.5 mg/mM PL | SP-B and –C (7 to 15 mg/mM PL) | N/A | SP-B-like sinapultide (0.9 mg/mL) |
| Phospholipid/amount | 30 mg/mL | 35 mg/mL | 80 mg/mL | 13.5 mg/mL | 30 mg/mL |
| PG/amount§ | 3.2% total PL | 4–6% total PL | 1.2% total PL | N/A | 25% total PL |
| FDA approval | July 1991 | July 1998 | November 1999 | August 1990 | March 2012 |
*Withdrawn from the market due to reduced efficacy relative to other natural surfactant medications.
Discovery Labs became Windtree Therapeutics in 2016.
Abbreviations: FDA, Food and Drug Administration; GSK, GlaxoSmithKline; N/A, not applicable; PG, phosphatidylglycerol; PL, phospholipid; SP-B, surfactant protein-B; SP-C, surfactant protein-C.
Additional considerations
The histologic description of COVID 19 pathology at autopsy shows diffuse alveolar damage with cellular fibromyxoid exudates, acute fibrinous, hyaline membrane formation, organizing pneumonia and desquamation of pneumocytes, all consistent with ARDS [53], [54]. Hyaline membrane formation has been observed in histological samples at both the early and later stages of the disease, suggesting early type II pneumocyte injury with surfactant dysfunction [53]. Although not all COVID-19 patients progress to a low-compliance phenotype, evidence in histological specimens highly suggests that there is surfactant dysfunction and hyaline membrane formation comparable to that observed in the non-COVID-19 ARDS-mediated alveolar damage described by Matthay and Zemans [55]. One approach to improve the dysfunctional surfactant in this disease is to treat with exogenous surfactant, thereby allowing maintenance of its function in the alveoli. Indeed, it seems likely that COVID-19-affected lungs will require functioning surfactant to fully recover. Exogenous bronchial surfactant instillation has been a feasible and safe approach in infants, although a higher dose and repeated administration may be required to restore dysfunctional alveoli impacted by COVID-19.
It should also be noted that despite its ability to inhibit TLR activation and inflammation, PG in surfactant does not seem to be globally immunosuppressive. In fact, in animal models in vivo it protects against infection resulting from several viruses, including respiratory syncytial virus, influenza A (H3N2) and H1N1 [38], [39], [56], by inhibiting the interaction of these viruses with their receptors on host cells (Fig. 1 ). Although it is not known whether PG has a similar inhibitory effect on the infectious capacity of SARS-CoV-2, a positive-sense single-strand RNA virus, the minimal effect of PG on the activation of TLR7/8 [32], [34] would suggest that this phospholipid would likely not suppress innate immune system responses to the virus. Therefore, surfactant might be useful in preventing the spread of SARS-CoV-2 viral infection between infected and naïve cells within the lung without affecting the response to this infection, in addition to protecting against the damage caused by excessive inflammation and edema and the increased surface tension that eventually results from loss of surfactant. On the other hand, the surfactant lipids phosphatidylcholine, in particular disaturated phosphatidylcholine (dipalmitoyl-phosphatidylcholine), and phosphatidylserine are reported to potentially promote infection by viral pathogens [57]. However, the mechanisms are thought to involve facilitation of viral entry via the ability of the virus to bind lipid and co-opt reuptake/recycling pathways in the case of PC and promotion of viral fusion by mimicking of an apoptotic signal in the case of PS [57]. Since SARS-CoV-2 purportedly gains entry into cells through angiotensin-converting enzyme 2 (ACE2) [40], these mechanisms used by other viruses seem unlikely to be relevant to SARS-CoV-2 and COVID-19 pneumonia.
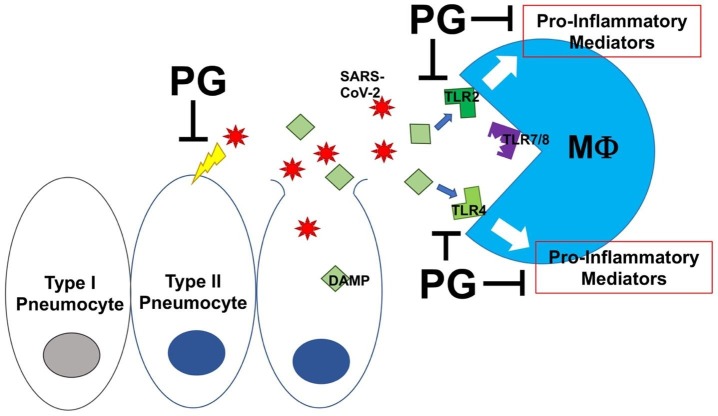
Fig. 1.
Phosphatidylglycerol in surfactant inhibits toll-like receptor-2 (TLR2) and TLR4 activation in response to damage-associated molecular patterns (DAMPs). SARS-CoV-2, the virus that causes COVID-19, is thought to infect type II pneumocytes, the cells in the lung responsible for surfactant production. Propagation of the virus results in pneumocyte death and the release of viral particles and endogenous intracellular molecules, some of which can serve as DAMPs. These DAMPs, as well as pathogen-associated molecular patterns (PAMPs) derived from microorganisms and viruses (reviewed in [74]), activate the innate immune system through TLR2 and TLR4 on alveolar macrophages (Mϕ), triggering pro-inflammatory mediator production and inflammation. Phosphatidylglycerol (PG) in surfactant inhibits PAMP- and DAMP-induced TLR2 and TLR4 activation thereby reducing inflammatory mediator production and inhibiting inflammation. PG also may inhibit SARS-CoV-2′s ability to infect naïve cells. It is hypothesized that restoration or supplementation of surfactant PG by administration of exogenous surfactant will improve the respiratory failure characteristic of COVID-19 pneumonia.
It should also be noted that certain conditions that increase the risk of a severe response to SARS-CoV-2 infection are also known to reduce surfactant and/or surfactant phospholipid levels or to impair surfactant function. Thus, phospholipid levels inversely correlate with age, at least in horses [58]. Smoking also reduces phospholipid levels in pulmonary surfactant [59] and is thought to increase the risk of adverse outcomes from COVID-19 [48]. Diabetes causes increased serum levels of high mobility group-B1 (HMGB1) [60], a known DAMP [31] that activates TLR4 [31], which would be expected to enhance inflammation. Similarly, in some cases hypertension has also been proposed to result from enhanced serum DAMP levels (reviewed in [61]), which again could possibly lead to a chronic low-level inflammation. Obesity is also thought to be accompanied by inflammation (reviewed in [62]). Indeed, serum levels of C-reactive peptide, a marker of inflammation, have been observed to correlate well with SARS-CoV-2 viral load and the Murray score, which assesses the severity of lung injury in individuals with ARDS [63]. Diabetes, hypertension and obesity have been suggested to predispose individuals to worse outcomes from COVID-19 [64]. In addition, serum levels of angiotensin II, which is also reported to increase inflammation through TLR4 [31], [65], are reported to be elevated in patients with COVID-19 [63]. Finally, a recent report has suggested that the corticosteroid dexamethasone may improve survival in patients with severe COVID-19 [66]. If confirmed, this result would be consistent with the hypothesis described here, since glucocorticoids are known not only to suppress inflammation but also to increase lung surfactant synthesis [67], [68]. By stimulating any remaining Type II alveolar cells to produce more surfactant phospholipids including PG, dexamethasone could both directly and indirectly decrease lung inflammation.
Implications
Surfactant has already been used in studies to treat ARDS [8], [9], [10], [11] in adult patients, although with less than impressive results. It should be noted that Walmrath et al. [9] discussed the likelihood that higher doses and/or more frequent administration of surfactant might be necessary in the case of ARDS (versus neonatal respiratory distress syndrome) to overcome the ongoing surfactant-inactivating conditions (increased sPLA2 levels, inflammation and oxidative stress) often present in ARDS lungs. We would also like to point out that not all surfactant medications contain PG (for example, Exosurf® does not), which could potentially be another explanation, in addition to potentially inadequate dosing and inactivation of surfactant function by shearing [13], for why not all studies of surfactant administration in ARDS have found a benefit [12]. Despite the mixed results concerning exogenous surfactant medication in adult ARDS [12], [13], it is approved by the Food and Drug Administration for intratracheal administration to pre-term infants to treat neonatal respiratory distress syndrome. In infants there are few side effects, and infants who receive surfactant have shorter hospital stays and better survival [69]. Similarly, eleven clinical trials in adults have indicated that surfactant therapy is both feasible and safe, with no significant adverse effects reported [70]. Therefore, it is postulated that because SARS-CoV-2 is thought to target and damage/destroy surfactant-producing type II pneumocytes, COVID-19 may be more like neonatal ARDS than are other types of ARDS. Indeed, like pre-term infants, patients with COVID-19 have been found to exhibit hyaline membrane formation [53]. Therefore, it is proposed that investigative studies to administer PG-containing surfactant, either synthetic (e.g., Surfaxin®) or isolated from bovine (Alveofact®, Survanta®, Beraksurf® and Infasurf®) or porcine lungs (Curosurf®), to critically ill COVID-19 patients be initiated, particularly in view of the fact that few therapies for severe COVID-19 have been shown to be effective to date [37], and such treatments are actively being sought.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
The authors would like to acknowledge and thank Dr. William Davis (Augusta University) for helpful feedback on this work. This work received no specific funding. WBB was supported in part by Veterans Affairs Merit Award #CX001357 and in part by National Institutes of Health/National Eye Institute award #R01EY030576. These sponsors had no role in the research, the writing of the manuscript or the decision to publish. The contents of this article do not represent the official views of the Department of Veterans Affairs or the United States Government.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.mehy.2020.110277.
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020 Feb 24. Epub 2020/02/28. [DOI] [PMC free article] [PubMed]
- 2.Mehta P., McAuley D.F., Brown M. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020 Mar 28;395(10229):1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Seeds M.C., Grier B.L., Suckling B.N. Secretory phospholipase A2-mediated depletion of phosphatidylglycerol in early acute respiratory distress syndrome. Am J Med Sci. 2012 Jun;343(6):446–451. doi: 10.1097/MAJ.0b013e318239c96c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.De Luca D., Lopez-Rodriguez E., Minucci A. Clinical and biological role of secretory phospholipase A2 in acute respiratory distress syndrome infants. Crit Care. 2013 Jul 24;17(4):R163. doi: 10.1186/cc12842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kitsiouli E., Nakos G., Lekka M.E. Phospholipase A2 subclasses in acute respiratory distress syndrome. BBA. 2009 Oct;1792(10):941–953. doi: 10.1016/j.bbadis.2009.06.007. [DOI] [PubMed] [Google Scholar]
- 6.Albert R.K., Lakshminarayan S., Hildebrandt J., Kirk W., Butler J. Increased surface tension favors pulmonary edema formation in anesthetized dogs' lungs. J Clin Invest. 1979 May;63(5):1015–1018. doi: 10.1172/JCI109369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nugent K., Dobbe L., Rahman R., Elmassry M., Paz P. Lung morphology and surfactant function in cardiogenic pulmonary edema: a narrative review. J Thorac Dis. 2019 Sep;11(9):4031–4038. doi: 10.21037/jtd.2019.09.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Walmrath D., Gunther A., Ghofrani H.A. Bronchoscopic surfactant administration in patients with severe adult respiratory distress syndrome and sepsis. Am J Respir Crit Care Med. 1996 Jul;154(1):57–62. doi: 10.1164/ajrccm.154.1.8680699. [DOI] [PubMed] [Google Scholar]
- 9.Walmrath D., Grimminger F., Pappert D. Bronchoscopic administration of bovine natural surfactant in ARDS and septic shock: impact on gas exchange and haemodynamics. Eur Respir J. 2002 May;19(5):805–810. doi: 10.1183/09031936.02.00243402. [DOI] [PubMed] [Google Scholar]
- 10.Gunther A., Schmidt R., Harodt J. Bronchoscopic administration of bovine natural surfactant in ARDS and septic shock: impact on biophysical and biochemical surfactant properties. Eur Respir J. 2002 May;19(5):797–804. doi: 10.1183/09031936.02.00243302. [DOI] [PubMed] [Google Scholar]
- 11.Anzueto A. Exogenous surfactant in acute respiratory distress syndrome: more is better. Eur Respir J. 2002 May;19(5):787–789. doi: 10.1183/09031936.02.00284902. [DOI] [PubMed] [Google Scholar]
- 12.Zhang L.N., Sun J.P., Xue X.Y., Wang J.X. Exogenous pulmonary surfactant for acute respiratory distress syndrome in adults: a systematic review and meta-analysis. Exp Ther Med. 2013 Jan;5(1):237–242. doi: 10.3892/etm.2012.746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Spragg R.G., Taut F.J., Lewis J.F. Recombinant surfactant protein C-based surfactant for patients with severe direct lung injury. Am J Respir Crit Care Med. 2011 Apr 15;183(8):1055–1061. doi: 10.1164/rccm.201009-1424OC. [DOI] [PubMed] [Google Scholar]
- 14.Taut F.J., Rippin G., Schenk P. A search for subgroups of patients with ARDS who may benefit from surfactant replacement therapy: a pooled analysis of five studies with recombinant surfactant protein-C surfactant (Venticute) Chest. 2008 Oct;134(4):724–732. doi: 10.1378/chest.08-0362. [DOI] [PubMed] [Google Scholar]
- 15.Gattinoni L., Chiumello D., Caironi P. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med. 2020 Jun;46(6):1099–1102. doi: 10.1007/s00134-020-06033-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gattinoni L., Coppola S., Cressoni M., Busana M., Rossi S., Chiumello D. COVID-19 does not lead to a “typical” acute respiratory distress syndrome. Am J Respir Crit Care Med. 2020 May 15;201(10):1299–1300. doi: 10.1164/rccm.202003-0817LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mohlenkamp S., Thiele H. Ventilation of COVID-19 patients in intensive care units. Herz. 2020 Jun;45(4):329–331. doi: 10.1007/s00059-020-04923-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Phua J., Weng L., Ling L. Intensive care management of coronavirus disease 2019 (COVID-19): challenges and recommendations. Lancet Respir Med. 2020 May;8(5):506–517. doi: 10.1016/S2213-2600(20)30161-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ikegami M., Weaver T.E., Grant S.N., Whitsett J.A. Pulmonary surfactant surface tension influences alveolar capillary shape and oxygenation. Am J Respir Cell Mol Biol. 2009 Oct;41(4):433–439. doi: 10.1165/rcmb.2008-0359OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schwartzstein R.M., Parker M.J. Lippincott, Williams and Wilkins; Philadelphia, PA: 2006. Respiratory Physiology: A Clinical Approach. [Google Scholar]
- 21.Albert R.K. The role of ventilation-induced surfactant dysfunction and atelectasis in causing acute respiratory distress syndrome. Am J Respir Crit Care Med. 2012 Apr 1;185(7):702–708. doi: 10.1164/rccm.201109-1667PP. [DOI] [PubMed] [Google Scholar]
- 22.Tierney D.F., Johnson R.P. Altered surface tension of lung extracts and lung mechanics. J Appl Physiol. 1965;20:1253–1260. [Google Scholar]
- 23.Voelker D.R., Numata M. Phospholipid regulation of innate immunity and respiratory viral infection. J Biol Chem. 2019 Mar 22;294(12):4282–4289. doi: 10.1074/jbc.AW118.003229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kandasamy P., Zarini S., Chan E.D., Leslie C.C., Murphy R.C., Voelker D.R. Pulmonary surfactant phosphatidylglycerol inhibits Mycoplasma pneumoniae-stimulated eicosanoid production from human and mouse macrophages. J Biol Chem. 2011 Mar 11;286(10):7841–7853. doi: 10.1074/jbc.M110.170241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Numata M., Chu H.W., Dakhama A., Voelker D.R. Pulmonary surfactant phosphatidylglycerol inhibits respiratory syncytial virus-induced inflammation and infection. PNAS. 2010 Jan 5;107(1):320–325. doi: 10.1073/pnas.0909361107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kuronuma K., Mitsuzawa H., Takeda K. Anionic pulmonary surfactant phospholipids inhibit inflammatory responses from alveolar macrophages and U937 cells by binding the lipopolysaccharide-interacting proteins CD14 and MD-2. J Biol Chem. 2009 Sep 18;284(38):25488–25500. doi: 10.1074/jbc.M109.040832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wu Y.Z., Medjane S., Chabot S. Surfactant protein-A and phosphatidylglycerol suppress type IIA phospholipase A2 synthesis via nuclear factor-kappaB. Am J Respir Crit Care Med. 2003 Sep 15;168(6):692–699. doi: 10.1164/rccm.200304-467OC. [DOI] [PubMed] [Google Scholar]
- 28.Kandasamy P., Numata M., Berry K.Z. Structural analogs of pulmonary surfactant phosphatidylglycerol inhibit toll-like receptor 2 and 4 signaling. J Lipid Res. 2016 Jun;57(6):993–1005. doi: 10.1194/jlr.M065201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Martin T.R., Wurfel M.M., Zanoni I., Ulevitch R. Targeting innate immunity by blocking CD14: novel approach to control inflammation and organ dysfunction in COVID-19 illness. EBioMedicine. 2020 Jun;20(57) doi: 10.1016/j.ebiom.2020.102836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schenck E.J., Ma K.C., Murthy S.B., Choi A.M.K. Danger signals in the ICU. Crit Care Med. 2018 May;46(5):791–798. doi: 10.1097/CCM.0000000000003007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Erridge C. Endogenous ligands of TLR2 and TLR4: agonists or assistants? J Leukoc Biol. 2010 Jun;87(6):989–999. doi: 10.1189/jlb.1209775. PubMed PMID: 20179153. [DOI] [PubMed] [Google Scholar]
- 32.Choudhary V., Uaratanawong R., Patel R.R. Phosphatidylglycerol inhibits toll-like receptor-mediated inflammation by danger-associated molecular patterns. J Invest Dermatol. 2019 Apr;139(4):868–877. doi: 10.1016/j.jid.2018.10.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Xie D., Choudhary V., Seremwe M. Soy phosphatidylglycerol reduces inflammation in a contact irritant ear edema mouse model in vivo. J Pharmacol Exp Ther. 2018 Jul;366(1):1–8. doi: 10.1124/jpet.117.244756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Choudhary V., Griffith S., Chen X., Bollag W.B. Pathogen-associated molecular pattern-induced TLR2 and TLR4 activation increases keratinocyte production of inflammatory mediators and is inhibited by phosphatidylglycerol. Mol Pharmacol. 2020 May;97(5):324–335. doi: 10.1124/mol.119.118166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Preuss S., Scheiermann J., Stadelmann S. 18:1/18:1-Dioleoyl-phosphatidylglycerol prevents alveolar epithelial apoptosis and profibrotic stimulus in a neonatal piglet model of acute respiratory distress syndrome. Pulm Pharmacol Ther. 2014 Jun;28(1):25–34. doi: 10.1016/j.pupt.2013.10.002. [DOI] [PubMed] [Google Scholar]
- 36.Ceribelli A., Motta F., De Santis M. Recommendations for coronavirus infection in rheumatic diseases treated with biologic therapy. J Autoimmun. 2020 May;109 doi: 10.1016/j.jaut.2020.102442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sanders J.M., Monogue M.L., Jodlowski T.Z., Cutrell J.B. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA. 2020 Apr 13 doi: 10.1001/jama.2020.6019. Epub 2020/04/14. [DOI] [PubMed] [Google Scholar]
- 38.Numata M., Kandasamy P., Nagashima Y. Phosphatidylglycerol suppresses influenza A virus infection. Am J Respir Cell Mol Biol. 2012 Apr;46(4):479–487. doi: 10.1165/rcmb.2011-0194OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Numata M., Nagashima Y., Moore M.L. Phosphatidylglycerol provides short-term prophylaxis against respiratory syncytial virus infection. J Lipid Res. 2013 Aug;54(8):2133–2143. doi: 10.1194/jlr.M037077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cao W., Li T. COVID-19: towards understanding of pathogenesis. Cell Res. 2020 May;30(5):367–369. doi: 10.1038/s41422-020-0327-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mossel E.C., Wang J., Jeffers S. SARS-CoV replicates in primary human alveolar type II cell cultures but not in type I-like cells. Virology. 2008 Mar 1;372(1):127–135. doi: 10.1016/j.virol.2007.09.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Qian Z., Travanty E.A., Oko L. Innate immune response of human alveolar type II cells infected with severe acute respiratory syndrome-coronavirus. Am J Respir Cell Mol Biol. 2013 Jun;48(6):742–748. doi: 10.1165/rcmb.2012-0339OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mason R.J. Pathogenesis of COVID-19 from a cell biology perspective. Eur Respir J. 2020 Apr;55(4) doi: 10.1183/13993003.00607-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.https://www.biorxiv.org/content/10.1101/2020.03.17.995639v1.full.pdf; accessed August 7, 2020.
- 45.Christmann U., Buechner-Maxwell V.A., Witonsky S.G., Hite R.D. Role of lung surfactant in respiratory disease: current knowledge in large animal medicine. J Vet Intern Med. 2009;23(2):227–242. doi: 10.1111/j.1939-1676.2008.0269.x. [DOI] [PubMed] [Google Scholar]
- 46.Autilio C., Perez-Gil J. Understanding the principle biophysics concepts of pulmonary surfactant in health and disease. Arch Dis Child Fetal Neonatal Ed. 2019 Jul;104(4):F443–F451. doi: 10.1136/archdischild-2018-315413. [DOI] [PubMed] [Google Scholar]
- 47.Henderson LA, Canna SW, Schulert GS, et al. On the alert for cytokine storm: Immunopathology in COVID-19. Arthritis Rheumatol. 2020 Apr 15. Epub 2020/04/16. [DOI] [PMC free article] [PubMed]
- 48.Contentti E.C., Correa J. Immunosuppression during the COVID-19 pandemic in neuromyelitis optica spectrum disorders patients: a new challenge. Mult Scler Relat Disord. 2020;41:1–2. doi: 10.1016/j.msard.2020.102097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Jeon G.W. Surfactant preparations for preterm infants with respiratory distress syndrome: past, present, and future. Korean J Pediatr. 2019 May;62(5):155–161. doi: 10.3345/kjp.2018.07185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chiba H., Piboonpocanun S., Mitsuzawa H., Kuronuma K., Murphy R.C., Voelker D.R. Pulmonary surfactant proteins and lipids as modulators of inflammation and innate immunity. Respirology. 2006 Jan;11(Suppl):S2–S6. doi: 10.1111/j.1440-1843.2006.00797.x. [DOI] [PubMed] [Google Scholar]
- 51.Funk C.J., Wang J., Ito Y. Infection of human alveolar macrophages by human coronavirus strain 229E. J Gen Virol. 2012 Mar;93(Pt 3):494–503. doi: 10.1099/vir.0.038414-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Casals C., Campanero-Rhodes M.A., Garcia-Fojeda B., Solis D. The role of collectins and galectins in lung innate immune defense. Front Immunol. 2018;9:1998. doi: 10.3389/fimmu.2018.01998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Xu Z., Shi L., Wang Y. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020 Apr;8(4):420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Fox SE, Akmatbekov A, Harbert JL, Li G, Quincy Brown J, Vander Heide RS. Pulmonary and cardiac pathology in African American patients with COVID-19: an autopsy series from New Orleans. Lancet Respir Med. 2020 May 27. Epub 2020/05/31. [DOI] [PMC free article] [PubMed]
- 55.Matthay M.A., Zemans R.L. The acute respiratory distress syndrome: pathogenesis and treatment. Annu Rev Pathol. 2011;6:147–163. doi: 10.1146/annurev-pathol-011110-130158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Numata M., Mitchell J.R., Tipper J.L. Pulmonary surfactant lipids inhibit infections with the pandemic H1N1 influenza virus in several animal models. J Biol Chem. 2020 Feb 7;295(6):1704–1715. doi: 10.1074/jbc.RA119.012053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Glasser J.R., Mallampalli R.K. Surfactant and its role in the pathobiology of pulmonary infection. Microbes Infect. 2012 Jan;14(1):17–25. doi: 10.1016/j.micinf.2011.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Christmann U., Hite R.D., Witonsky S.G. Influence of age on surfactant isolated from healthy horses maintained on pasture. J Vet Intern Med. 2009;23(3):612–618. doi: 10.1111/j.1939-1676.2009.0298.x. [DOI] [PubMed] [Google Scholar]
- 59.More J.M., Voelker D.R., Silveira L.J., Edwards M.G., Chan E.D., Bowler R.P. Smoking reduces surfactant protein D and phospholipids in patients with and without chronic obstructive pulmonary disease. BMC Pulm Med. 2010 Oct;25(10):53. doi: 10.1186/1471-2466-10-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wang Y., Zhong J., Zhang X. The role of HMGB1 in the pathogenesis of type 2 diabetes. J Diabetes Res. 2016;2016:2543268. doi: 10.1155/2016/2543268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.McCarthy C.G., Webb R.C. The toll of the gridiron: damage-associated molecular patterns and hypertension in American football. FASEB J. 2016 Jan;30(1):34–40. doi: 10.1096/fj.15-279588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gregor M.F., Hotamisligil G.S. Inflammatory mechanisms in obesity. Annu Rev Immunol. 2011;29:415–445. doi: 10.1146/annurev-immunol-031210-101322. [DOI] [PubMed] [Google Scholar]
- 63.Liu Y., Yang Y., Zhang C. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020 Mar;63(3):364–374. doi: 10.1007/s11427-020-1643-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Fneed-extra-precautions%2Fgroups-at-higher-risk.html; accessed August 8, 2020.
- 65.Ji Y., Liu J., Wang Z., Liu N. Angiotensin II induces inflammatory response partly via toll-like receptor 4-dependent signaling pathway in vascular smooth muscle cells. Cell Physiol Biochem. 2009;23(4–6):265–276. doi: 10.1159/000218173. [DOI] [PubMed] [Google Scholar]
- 66.https://www.who.int/news-room/detail/16-06-2020-who-welcomes-preliminary-results-about-dexamethasone-use-in-treating-critically-ill-covid-19-patients; accessed June 18, 2020.
- 67.Rooney S.A. Lung surfactant. Environ Health Perspect. 1984 Apr;55:205–226. doi: 10.1289/ehp.8455205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Warburton D. Chronic hyperglycemia with secondary hyperinsulinemia inhibits the maturational response of fetal lamb lungs to cortisol. J Clin Invest. 1983 Aug;72(2):433–440. doi: 10.1172/JCI110991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Canadian Paediatric Society Recommendations for neonatal surfactant therapy. Paediatr Child Health. 2005 Feb;10(2):109–116. [PMC free article] [PubMed] [Google Scholar]
- 70.Lewis J.F., Brackenbury A. Role of exogenous surfactant in acute lung injury. Crit Care Med. 2003 Apr;31(4 Suppl):S324–S328. doi: 10.1097/01.CCM.0000057911.19145.9F. [DOI] [PubMed] [Google Scholar]
- 71.Schenck D.M., Fiegel J. Tensiometric and phase domain behavior of lung surfactant on mucus-like viscoelastic hydrogels. ACS Appl Mater Interfaces. 2016 Mar 9;8(9):5917–5928. doi: 10.1021/acsami.6b00294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Seeger W., Grube C., Gunther A., Schmidt R. Surfactant inhibition by plasma proteins: differential sensitivity of various surfactant preparations. Eur Respir J. 1993 Jul;6(7):971–977. [PubMed] [Google Scholar]
- 73.https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/021746s000lbl.pdf; accessed August 9, 2020.
- 74.Lester S.N., Li K. Toll-like receptors in antiviral innate immunity. J Mol Biol. 2014 Mar 20;426(6):1246–1264. doi: 10.1016/j.jmb.2013.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.