Supplemental Digital Content is available in the text.
Keywords: human albumin, inflammation, mortality, prognosis
Abstract
Objectives:
Infection by severe acute respiratory syndrome coronavirus-2 can induce uncontrolled systemic inflammation and multiple organ failure. The aim of this study was to evaluate if plasma exchange, through the removal of circulating mediators, can be used as rescue therapy in these patients.
Design:
Single center case series.
Setting:
Local study.
Subjects:
Four critically ill adults with coronavirus disease 19 pneumonia that failed conventional interventions.
Interventions:
Plasma exchange. Two to six sessions (1.2 plasma volumes). Human albumin (5%) was used as the main replacement fluid. Fresh frozen plasma and immunoglobulins were administered after each session to avoid coagulopathy and hypogammaglobulinemia.
Measurements and Main Results:
Serum markers of inflammation and macrophage activation. All patients showed a dramatic reduction in inflammatory markers, including the main cytokines, and improved severity scores after plasma exchange. All survived to ICU admission.
Conclusions:
Plasma exchange mitigates cytokine storm, reverses organ failure, and could improve survival in critically ill patients with coronavirus disease 2019 infection.
Some patients affected by coronavirus disease 2019 (COVID-19) develop severe inflammation and progressive organ failure that threaten survival. Laboratory tests in these patients usually show elevation of inflammatory and coagulation markers (C-reactive protein [CRP], d-dimer) and data of macrophage activation (elevation of triglycerides, lactate dehydrogenase [LDH], and ferritin) (1–4). Corticosteroids and other immunosuppressive agents have been proposed in this setting (2–7). However, some patients do not respond to this therapy and develop multiple organ failure. Therapeutic plasma exchange removes endogenous and exogenous inducers of the systemic inflammatory response (pathogen-associated molecular pattern and damage-associated molecular pattern) and proinflammatory mediators (cytokines and reactive oxygen species) that are involved in the pathogenesis of organ failure (8). Sporadic case reports suggest that therapeutic plasma exchange can be an effective rescue therapy in patients with hemophagocytic lymphohistiocytosis (9) or severe influenza A (H1N1) infection (10). We report a case series of four critically ill patients infected by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) successfully treated with plasma exchange. Therapeutic plasma exchange, two to six sessions, was performed with 5% albumin as the main replacement fluid (2/3). Fresh frozen plasma (FFP) was used (1/3) at the end of the plasma exchange to avoid coagulopathy. IV immunoglobulin (IVIg) was administered after each session (100 mg/Kg) to prevent the hypogammaglobulinemia induced by the procedure.
CASE 1
A 49-year-old man with obesity, hypertension, and type 2 diabetes was admitted to the hospital with fever and cough for 1 week and progressive shortness of breath within the previous 48 hours. Oxygen saturation was 90% (Fio2 28%). Laboratory tests indicated the following: serum CRP 14.65 mg/dL, creatinine 1.44 mg/dL, LDH 449 U/L, triglycerides 752 mg/dL, ferritin 2,005 ng/mL, and d-dimer of 4,900 ng/mL. Apart from lymphopenia (0.6 × 109/L), blood cell counts were normal, as well as procalcitonin levels and coagulation variables (Supplementary Table 1, Supplemental Digital Content 2, http://links.lww.com/CCM/F804). Arterial blood gases showed a Pao2 of 61.8 mm Hg, and chest radiograph revealed bilateral lung infiltrates. Polymerase chain reaction (PCR) for SARS-CoV-2 in nasopharyngeal smear was positive. Lopinavir/ritonavir, hydroxychloroquine, interferon beta-1a, ceftriaxone, and linezolid were started. On day 2 of hospitalization, the patient was admitted to the ICU due to progressive respiratory failure and required orotracheal intubation and mechanical ventilation. After intubation, norepinephrine was started. Echocardiography disclosed mild ventricular dysfunction. Troponin levels were normal. Deep sedation and muscle relaxation were required in the first 48 hours after intubation due to severe hypoxemia (PaFio2: 68) and poor adaptation to the ventilator. Tocilizumab (400 mg) was administered on days 2 and 4 of ICU admission. Tracheotomy was performed 1 week after intubation. On day 9, he presented with high fever (41°C), sinus tachycardia with frequency-dependent left bundle branch block, and stage 2 acute kidney injury (AKI; creatinine: 3.65 mg/dL). Mild cardiac hypomotility persisted at echocardiography. Inflammatory and macrophage activation parameters increased markedly (Fig. 1) (Supplementary Table 2, Supplemental Digital Content 3, http://links.lww.com/CCM/F805). Cultures and body CT-scan ruled out bacterial or fungal infection or pulmonary thromboembolism. Upon suspicion of hyperinflammatory state due to cytokine storm and macrophage activation like syndrome, therapeutic plasma exchange was started on day 10. Four sessions were performed following an every other day schedule (days 10, 12, 14, and 16). The plasma volume exchanged by session was 4,900 mL.
Figure 1.
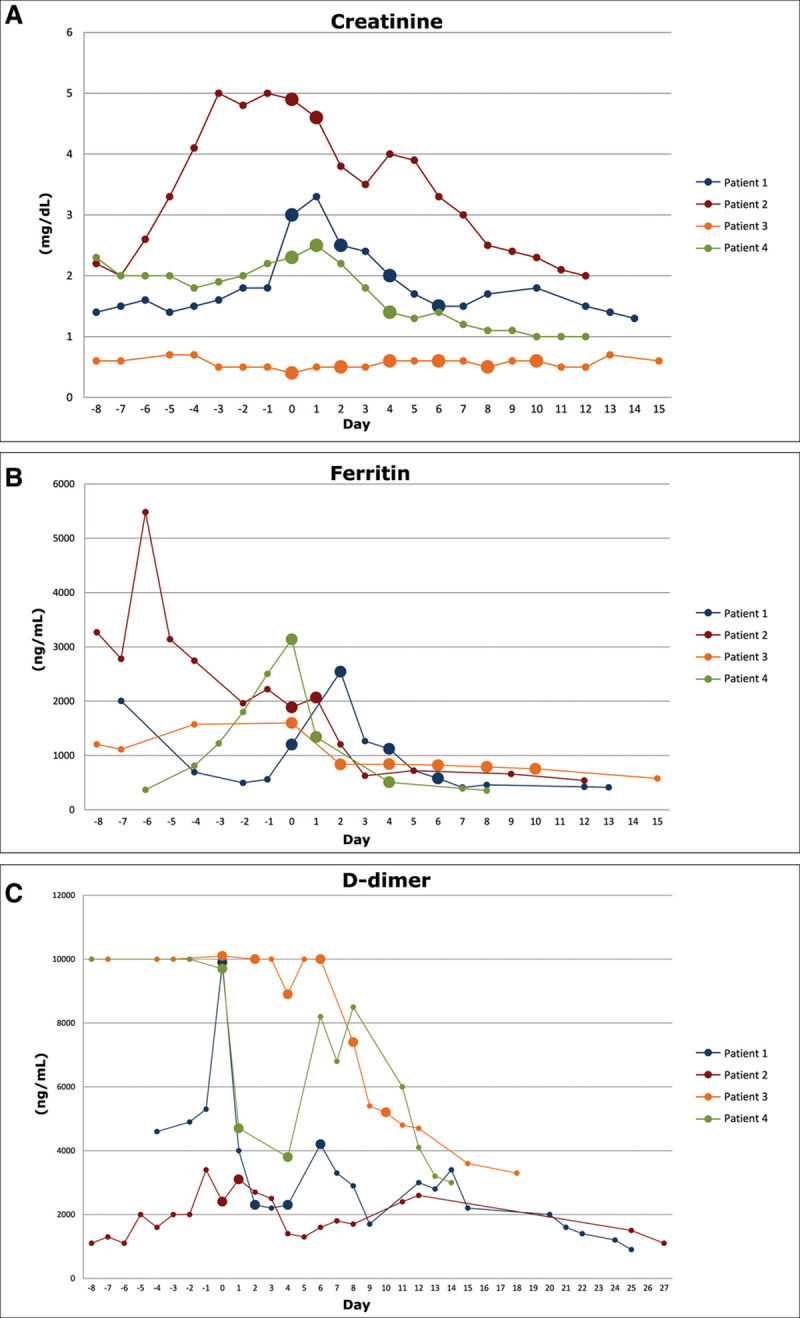
Effects of therapeutic plasma exchange on serum levels of creatinine (A), ferritin (B), and d-dimer (C) in the four patients included in the study. Day 0 is the day of first plasma exchange. Days in negative numbers correspond to the period prior to initiate plasma exchange. Large dots denote the days of plasma exchange in each patient. Therapeutic plasma exchange induced a marked and sustained decrease in the serum levels of the three variables analyzed in all four patients.
The patient condition improved in the following days: fever resolved 3 days following the fourth session of plasma exchange (day 19), renal, cardiac, and respiratory function normalized, and laboratory findings showed sustained improvement (Fig. 1) (Supplementary Table 2, Supplemental Digital Content 3, http://links.lww.com/CCM/F805). Plasma levels of most of the main cytokines decreased markedly after therapeutic plasma exchange (Fig. 2). Severe myopathy and catheter-related infection by Klebsiella pneumoniae 8 days after finishing plasma exchange were the most relevant problems during the rest of ICU stay. On day 36, he was decannulated. The patient was discharged from the ICU and from the hospital on days 38 and 51, respectively.
Figure 2.
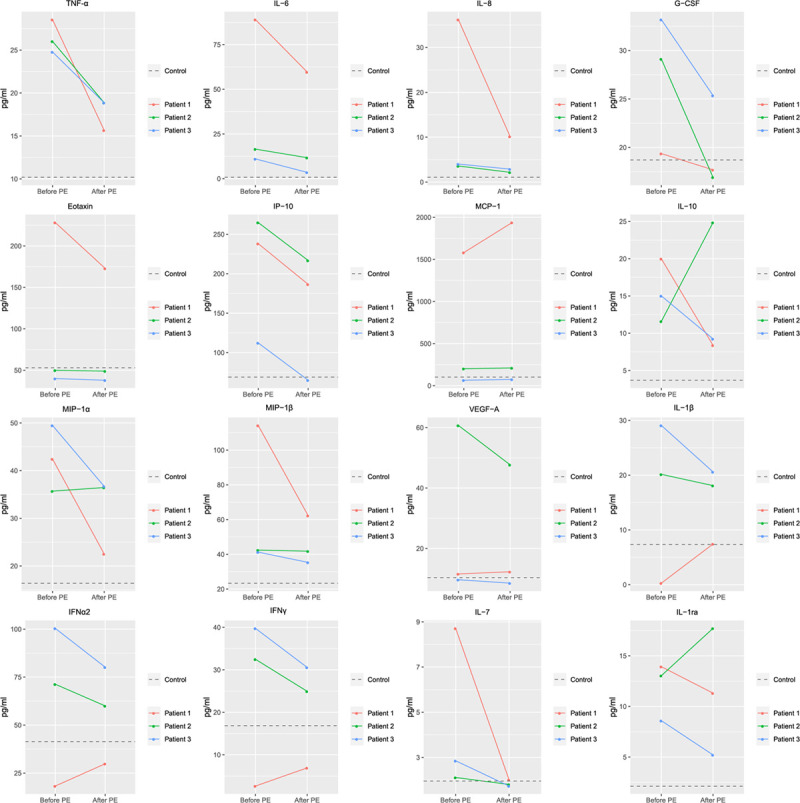
Effects of therapeutic plasma exchange on plasma levels of different cytokines and chemokines in the three patients in whom samples were obtained before and after the procedure. Baseline values are the median of the average of the first two samples. Values after treatment are the average of the rest of available values. Patients presented an hyperinflammatory state with extremely high plasma levels of tumor necrosis factor (TNF)-alpha, granulocyte colony- stimulating factor (G-CSF), interleukin (IL)-1ra, IL-10, and chemokines. Therapeutic plasma exchange induced a marked decrease in the plasma levels of the majority of cytokines and chemokines analyzed. IFN = interferon, IP-10 = interferon gamma-induced protein 10, MCP-1 = monocyte chemoattractant protein-1, MIP = macrophage inflammatory protein, PE = plasma exchange, VEGF = vascular endothelial growth factor.
CASE 2
A 59-year-old man was admitted to the hospital for a 10-day course of cough, fever, asthenia, and diarrhea. His medical history revealed a liver transplantation in 2007 treated with mycophenolate until current hospitalization, hypertension, insulin-dependent diabetes, and chronic kidney disease (creatinine 2.0 mg/dL). Physical examination at admission was unremarkable except for lung basal crackles. Oxygen saturation at that time was 85% at Fio2 21%. Arterial blood gases demonstrated a Pao2 of 68.1 mm Hg, and chest radiograph showed bilateral lung infiltrates. Laboratory findings were as follows: CRP 8.65 mg/dL, creatinine 2.01 mg/dL, LDH 429 U/L, triglycerides 344 mg/dL, ferritin 2,780 ng/mL, d-dimer 1,300 ng/mL. Lymphopenia (0.4 × 109/L) was also present (Supplementary Table 1, Supplemental Digital Content 2, http://links.lww.com/CCM/F804). PCR for SARS-CoV-2 in nasopharyngeal smear was positive. He was started on lopinavir/ritonavir, hydroxychloroquine, azithromycin, ceftriaxone, and teicoplanin. Twenty-four hours after admission, the patient developed progressive respiratory failure. In this setting, he was admitted to the ICU and required invasive mechanical ventilation. After intubation, norepinephrine was started. An echocardiography disclosed moderate ventricular dysfunction suggestive of myocarditis. Troponin levels were 6,963 ng/L. Anakinra and stress dose hydrocortisone were started.
Although respiratory requirements improved over the next days, his overall condition worsened. He had persistent high fever without microbiological isolations and developed stage 3 AKI in the setting of persistent cardiac dysfunction. Serum markers of inflammation and macrophage activation remained high or worsened (Fig. 1) (Supplementary Table 2, Supplemental Digital Content 3, http://links.lww.com/CCM/F805). Tracheotomy was performed 9 days after ICU admission. In this setting, therapeutic plasma exchange was started on day 10 of ICU stay. Only two sessions were performed due to moderate FFP transfusion reaction at the end of the second session (tachycardia, hypotension requiring an increase in norepinephrine doses, maculopapular rash, and increased leukocyte count). These two sessions were performed in consecutive days. The plasma volume exchanged by session was 5,000 mL. Patient clinical condition improved 48 hours after the last session. Fever resolved, cardiac and renal function normalized in few days, and regular laboratory tests (Fig. 1) (Supplementary Table 2, Supplemental Digital Content 3, http://links.lww.com/CCM/F805) and plasma cytokines also improved (Fig. 2). The patient was decannulated 29 days after ICU admission and was discharged from the ICU and from the hospital on days 31 and 40, respectively.
CASE 3
A 56-year-old man with obesity and hypertension was admitted to the hospital with bilateral lumbar pain for 4 days and diarrhea and loss of sensitivity and strength in the legs within the last 24 hours. At admission, he had fever (38°C) and was diaphoretic, tachycardic, tachypneic, and hypotensive. Oxygen saturation at that time was 96% at Fio2 80%. Laboratory test showed CRP 22.76 mg/dL, procalcitonin 2.93 ng/mL, creatinine 2.36 mg/dL, LDH 682 U/L, ferritin 1,396 ng/mL, troponin 224.7 ng/L, lactate 28.8 mg/dL, and d-dimer greater than 10.000 ng/mL (Supplementary Table 1, Supplemental Digital Content 2, http://links.lww.com/CCM/F804). Blood cell counts disclosed leukocytosis and thrombocytopenia (25.93 and 52.0 × 109/L, respectively). Echocardiogram was normal. An angio-CT-scan showed bilateral infiltrates suggestive of COVID-19 pneumonia, lobar and segmental acute right pulmonary thromboembolism, and multiple nonocclusive arterial thrombosis in distal aortic arch, splenic artery, aortoiliac bifurcation, iliac arteries, and right femoral artery. PCR for SARS-CoV-2 in nasopharyngeal swab was positive. The patient was admitted to the ICU for respiratory (high flow nasal cannula) and vasopressor support (norepinephrine: 0.56 ug/Kg/min). He was started on lopinavir/ritonavir, hydroxychloroquine, azithromycin, and piperacillin-tazobactam. IV dexamethasone and heparin sodium infusion were also initiated. After initial improvement, the patient presented symptoms of acute limb ischemia on day 14 and required urgent bilateral transpopliteal embolectomy. Screen for thrombophilia factors identified positive immunoglobulin G (IgG) anticardiolipin serum antibodies. On day 15, catastrophic antiphospholipid syndrome was diagnosed and methylprednisolone (30 mg/d) and therapeutic plasma exchange were started. Sessions were performed every other day. The plasma volume exchanged by session was 5,000 mL. After completing six sessions, serum ferritin and d-dimer (Fig. 1) (Supplementary Table 2, Supplemental Digital Content 3, http://links.lww.com/CCM/F805) and plasma cytokines levels (Fig. 2) decreased markedly. The patient was discharged from hospital 33 days after admission on steroid and low molecular weight heparin therapy. No more thrombotic events were observed.
CASE 4
A 64-year-old man with alcoholic liver cirrhosis, hypertension, and type 2 diabetes was admitted to the hospital with fever (38.5°C) for 2 days. PCR for SARS-CoV-2 in nasopharyngeal swab was positive. Treatment with lopinavir/ritonavir, hydroxychloroquine, azithromycin, interferon beta-1a, ceftriaxone, and teicoplanin was started. Four days after hospital admission, he presented progressive respiratory failure (oxygen saturation of 90% at Fio2 80%) and was admitted to the ICU for invasive mechanical ventilation. Chest radiograph showed bilateral lung infiltrates. At that time, laboratory test disclosed CRP 2.34 mg/dL, creatinine 2.41 mg/dL, LDH 426 U/L, ferritin 464 ng/mL, and d-dimer 900 ng/mL (Supplementary Table 1, Supplemental Digital Content 2, http://links.lww.com/CCM/F804). In this setting, methylprednisolone was started (60 mg/d for 5 d). Nine days after ICU admission, a tracheostomy was performed. However, 10 days later, the patient developed grade 2 hepatic encephalopathy and stage 2 AKI. Laboratory tests also showed an increase in inflammatory and coagulation markers (WBC, CRP, ferritin, and d-dimer) and deterioration of liver function (serum bilirubin 5.4 mg/dL, international normalized ratio 1.35). Abdominal Doppler ultrasonography ruled out vascular complications. Test for hepatotropic virus were negative except for herpes 6 (PCR in blood was weakly positive). With the orientation of an acute on chronic liver failure triggered by SARS-CoV-2, therapeutic plasma exchange was started 26 days after ICU admission. The plasma volume exchanged by session was 3,800 mL. After completing three sessions performed on days 26, 27, and 29, liver (bilirubin 1.8 mg/dL, INR 1.18) and renal function improved, hepatic encephalopathy resolved, and inflammatory variables normalized (Fig. 1) (Supplementary Table 2, Supplemental Digital Content 3, http://links.lww.com/CCM/F805). The patient was decannulated on day 41 of ICU stay and was discharged to an intermediate care unit 2 days later.
DISCUSSION
SARS-CoV-2 infection can induce uncontrolled inflammatory innate and adaptative responses that may lead to tissue damage and multiple organ failure (1, 3–5, 11). High serum levels of proinflammatory cytokines (tumor necrosis factor [TNF]-alpha, interleukin [IL]-1, IL-2, IL-6, IL-17, granulocyte colony-stimulating factor, granulocyte-macrophage colony-stimulating factor) and chemokines (monocyte chemoattractant protein-1, macrophage inflammatory protein 1-alpha, IL-8, interferon gamma-induced protein 10) have been reported in patients with severe acute respiratory syndrome (12), Middle East respiratory syndrome (MERS) (13), and also in severe COVID-19 (1, 11). Macrophage activation syndrome may also occur (3). Therapeutic plasma exchange removes inflammatory mediators from the systemic circulation and could ameliorate this COVID-19–related cytokine storm and immunopathology, a hypothesis that has not been evaluated so far. Our small case series supports this contention and suggests that plasma exchange combined to IV IgG is an effective salvage therapy in patients with COVID-19 pneumonia requiring critical care. Three of our patients presented multiple organ failure despite having received conventional therapies that included antiviral and anti-inflammatory drugs. Furthermore, two of them had analytical and clinical data of macrophage activation like syndrome. The initiation of therapeutic plasma exchange was temporarily associated with a marked clinical improvement with resolution of fever, improvement of renal and vascular function, decrease in Sequential Organ Failure Assessment and Acute Physiology and Chronic Health Evaluation scores, and amelioration of inflammatory markers including variables of macrophage activation such as serum ferritin and triglyceride. A fourth patient was treated with therapeutic plasma exchange due to a catastrophic antiphospholipid syndrome induced by SARS-CoV-2 that was refractory to anticoagulation. Importantly, systemic inflammatory markers decreased, and thrombotic events definitively resolved.
Our study also suggests that critically ill COVID-19 patients show an hyperinflammatory state that can be mitigated by therapeutic plasma exchange. Plasma levels of TNF-alpha and of other proinflammatory cytokines and chemokines were extremely high in samples taken before plasma exchange. This treatment effectively decreased levels of the great majority of cytokines and chemokines, therefore attenuating cytokine storm.
It is important to remark that therapeutic plasma exchange was performed in our four patients using 5% albumin as the main replacement fluid. Albumin is the main transporter and the main antioxidant and free-radical scavenger of human plasma (14). FFP was administered at the end of each session to prevent coagulopathy. IVIgs were also administered after each session to prevent the development of hypogammaglobulinemia. This apheresis approach was safe with just one patient developing a moderate transfusion reaction related with FFP infusion.
In summary, our small case series suggests that therapeutic plasma exchange is an effective recue therapy in critically ill patients with COVID-19 infection who do not respond to conventional therapies. This treatment was safe, ameliorated cytokine storm, reversed organ failure, and improved survival in very severe COVID-19 patients. A randomized controlled trial, the recambio plasmatico (REP)-COVID (ClinicalTrials.gov Identifier: NCT04374539), is currently ongoing to confirm or reject our hypothesis.
Supplementary Material
Footnotes
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal’s website (http://journals.lww.com/ccmjournal).
Members of the Covid Clinic Critical Care (CCCC) Group are listed in the Supplementary Appendix (Supplemental Digital Content 1, http://links.lww.com/CCM/F803).
Dr. Sanchez disclosed work for hire. Dr. Reverter disclosed off-label product use of plasma exchange. Dr. Arroyo received funding from Grifols. Dr. Lozano’s institution received funding from Terumo BCT and received funding from Grifols and Cerus. The remaining authors have disclosed that they do not have any potential conflicts of interest.
REFERENCES
- 1.Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;180:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McGonagle D, Sharif K, O’Regan A, et al. The role of cytokines including interleukin-6 in COVID-19 induced pneumonia and macrophage activation syndrome-like disease. Autoimmun Rev. 2020;19:102537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cao X. COVID-19: Immunopathology and its implications for therapy. Nat Rev Immunol. 2020;20:269–270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mehta P, McAuley DF, Brown M, et al. ; HLH Across Speciality Collaboration, UK: COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chinese Clinical Trial Registry 2020. Available at: http://www.chictr.org.cn/showprojen.aspx?proj=49409. Accessed March 6, 2020.
- 7.Shakoory B, Carcillo JA, Chatham WW, et al. Interleukin-1 receptor blockade is associated with reduced mortality in sepsis patients with features of macrophage activation syndrome: Reanalysis of a prior phase III trial. Crit Care Med. 2016;44:275–281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Reeves HM, Winters JL. The mechanisms of action of plasma exchange. Br J Haematol. 2014;164:342–351. [DOI] [PubMed] [Google Scholar]
- 9.Kinjo N, Hamada K, Hirayama C, et al. Role of plasma exchange, leukocytapheresis, and plasma diafiltration in management of refractory macrophage activation syndrome. J Clin Apher. 2018;33:117–120. [DOI] [PubMed] [Google Scholar]
- 10.Patel P, Nandwani V, Vanchiere J, et al. Use of therapeutic plasma exchange as a rescue therapy in 2009 pH1N1 influenza A–an associated respiratory failure and hemodynamic shock. Pediatr Crit Care Med. 2011;12:e87–e89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Qin C, Zhou L, Hu Z, et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis. 2020;71:792–768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wong CK, Lam CW, Wu AK, et al. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin Exp Immunol. 2004;136:95–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Min CK, Cheon S, Ha NY, et al. Comparative and kinetic analysis of viral shedding and immunological responses in MERS patients representing a broad spectrum of disease severity. Sci Rep. 2016;6:25359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Arroyo V, García-Martinez R, Salvatella X. Human serum albumin, systemic inflammation, and cirrhosis. J Hepatol. 2014;61:396–407. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


