Abstract
Aims
Tablets may be subdivided for dose adaptations or to ease swallowing. The handling is common in older patients but can be difficult and inaccurate. Currently, it is not known which hand–eye functions determine the ability of older people to break tablets by hand and to do so with acceptable ease and accuracy. The aim of this study was to develop a test battery to assess the hand–eye functions relevant in predicting easy and accurate tablet subdivision in older people.
Methods
A mixed methods study was conducted including literature reviews and a pilot experiment. The reviews were conducted in Pubmed, Google Scholar, Dutch journals and professional standards. The first review tried to identify the hand–eye functions relevant to tablet subdivision and the second the associated measuring instruments, testing protocols and normative data. A test battery was empanelled. A pilot experiment was conducted in 30 adult volunteers to optimize and evaluate the test battery.
Results
Five domains were considered relevant: hand size, hand strength, flexibility/manual dexterity, vision and coordination. Hand size could best be measured by finger circumference, hand strength by pinch‐ and grip strength, flexibility by active range of joint motion, manual dexterity (and flexibility, coordination, cognition, vision) by pegboard function, vision by near visual acuity. Older people preferred the use of tablet splitters over hand breaking.
Conclusion
Easy and accurate tablet subdivision is essential to the good use of medicines. We developed a test battery for older people, but probably of value to all age groups.
Keywords: clinical pharmacology, medication errors, clinical pharmacology, medication safety, clinical pharmacology, drug information
What is already known about this subject
Older people may need to break tablets to lower the dose or to ease swallowing.
Older people are commonly facing difficulties breaking tablets by hand.
Subdividing tablets by hand or a tablet splitter may result in inaccurate dosages.
What this study adds
A test battery was developed to study the hand–eye functions determining the ease and accuracy of tablet breaking.
The study provides preliminary insights into the dependency of sensory functions on tablet breaking.
1. INTRODUCTION
Tablets are frequently divided to lower the dose, to ease swallowing or to save cost.1, 2, 3 For several reasons, tablet subdivision is more common in older patients. First of all, ageing increases the risk for multi‐ and comorbidities and therewith overall medication use.4, 5, 6 It also increases the risk for impaired renal and hepatic functioning, generally implying a need for dose reductions.1, 7 Moreover, ageing increases the risk that patients will suffer from reduced saliva, impaired oropharyngeal swallow response and loss of cognition, which in turn, increase the risk that older people will encounter difficulties swallowing tablets intact.8, 9 Despite the increasing need for tablet subdivision in older people, evidence suggests that the subdivision of tablets by hand (i.e. breaking) can be difficult and inaccurate, and that this is especially true for people with impaired manual handling capacities and reduced vision (i.e. older people). These people, are also more likely to encounter difficulties taking tablets from their packaging prior to use.1, 10, 11 Difficulties may not always be recognised by patients themselves, as patients' self‐reported medication management skills can differ from their observed performance.12 Thus, there is an urgent need to ensure that tablets can be subdivided with sufficiently ease and accuracy, especially for home‐dwelling older people.
Tablet subdivision is determined by the characteristics of the tablet and the patient.1 For a long time, the use of a tablet splitter was considered a practical coping strategy for patients who were unable to break tablets by hand.13 However, in 2014, we observed that the use of some commercially available splitters resulted in unequal tablet parts and crumbling, and that a best case tablet (paracetamol) was more accurately broken by the hands of a pharmacy student than by any of the 6 tablet splitters tested.14 Meanwhile, our finding has been confirmed by Habib et al15 and Somoby et al,16 whereas Ciavarella et al17 found that tablets may be accurately subdivided by hand as well as by some specific types of tablet splitters. Realizing that there are many different types of tablet splitters on the market, that their performance may depend on tablet type and that they are not classified as a medical device (meaning their performance and/or consistency is not ensured over time and may thus differ between batches of the same device), we consider that tablets can best be subdivided by hand and that the subdivision of tablets by hand should thus be possible for patients who are expected to manage medications themselves i.e. we consider that the majority of home‐dwelling older people should be able to subdivide tablets by hand.
Currently, it is not known which specific characteristics of the hands and eyes of older people determine their ability to subdivide tablets by hand and/or with the help of a tablet splitter and to do so with sufficiently ease and accuracy. Thus it is not known which hand–eye functions should be considered as a determinant for the selection of a test panel that would be able to predict the ease and accuracy of tablet breaking by older people during the drug product development studies. The aim of this study was to develop a test battery assessing the hand–eye functions relevant to easy and accurate tablet subdivision in older people. Once such method has been developed, it is hoped that advances in medication management will result in a robust system verifying that the right patient has taken the right dose at the right time and remains capable of self‐medication management.18
2. METHODS
The test battery was developed in 4 phases using a mixed method approach. First, the hand–eye domains and functions relevant to tablet subdivision were systematically identified from the literature. Subsequently, measurement instruments, protocols and normative data were retrieved from the same data sources. Then all this was empanelled into a test battery that was optimized and evaluated in the fourth and final phase.
2.1. Phase I: Identifying hand–eye functions relevant to tablet subdivision
2.1.1. Study design
Scoping literature.
2.1.2. Objective
To identify the hand–eye functions that are likely to be of relevance to the ease and accuracy of tablet subdivision.
2.1.3. Data sources and search profile
Pubmed, Google Scholar and 2 national journal databases (Pharmaceutisch Weekblad and Nederlands Tijdschrift voor Geneeskunde) were searched from their start for publications in English or Dutch identifying hand–eye functions possibly related to tablet subdivision or to associated handlings such as opening a medication package. The search profile used a snowball approach using keywords as tablet, pill, breaking, splitting, subdivision, functionality, acceptability, capability, problem, ease, adherence, satisfaction, usability, accuracy, pain, discomfort, swallowing, dysphagia, specific hand–eye function, ageing, medication management.
2.1.4. Data selection and analysis
The title and, where needed, the abstract and full text of the identified publications were evaluated by a student in drug innovation (L.D.). A second expert was involved in case of the slightest doubt and a third where needed to reach consensus (D.V.R.N. and A.N., pharmacists and quality assessors). Publications were searched for relevant domains and associated functions and the findings were summarized into a list. Evaluation of publications was discontinued when no new domains and/or functions were identified. Data saturation was verified by evaluating the references cited in the included publications. Face validity of the list was confirmed by other Dutch quality assessors and a Dutch physiotherapist.
2.2. Phase II: Selecting and evaluating the measuring instruments and relevant protocols
2.2.1. Study design
Second scoping literature review followed by an expert panel evaluating the findings.
2.2.2. Objective review
To identify which measuring instruments and protocols exist to evaluate the functions identified in phase I and which can best be used for this study.
2.2.3. Data sources and search profile
The same literature was examined by the same experts by the same experts as indicated in phase I. A variety of search profiles were adopted to identify measuring instruments for the functions selected in phase I and to identify any related protocols and normative data. In addition, professional standards were considered.
2.2.4. Data selection and data analysis
The measuring instruments and protocols were selected and subsequently evaluated for use in the test battery by the same expert panel. Selection criteria included an evaluation of the general acceptability of the measuring instrument, the availability and content of a protocol for the instrument, the use of the instrument and/or protocols in earlier studies, cost, portability of the instrument to patients, need for specific operator training, patient burden and maintenance of patient integrity. Face validity of the selected instruments was evaluated by other Dutch quality assessors and by a physiotherapist.
2.3. Phase III: Development of the draft test battery
2.3.1. Study design
Expert panel.
2.3.2. Objective
To empanel a first draft of the test battery.
2.3.3. Data selection and analysis
The results from phase I and phase II were evaluated by the same expert panel. It was considered how the selected instruments could be ordered (empanelled) into a test battery that could be conducted in 1 h at the maximum, and that would follow the protocols for each of the measuring instruments to the best extent possible.
2.4. Phase IV: Optimization and evaluation of the (draft) test battery
2.4.1. Study design
Pilot experiment using a test panel to evaluate results.
2.4.2. Objective
To optimize the draft test battery derived from phase III through a step by step approach and subsequently, to evaluate the suitability of the optimized test battery.
2.4.3. Ethical approval
The authors considered that this study was not subject to ethical approval according to the Dutch law on medical‐scientific research in man. This opinion was confirmed by the medical research ethical committee of Arnhem–Nijmegen. The protocol for this pilot study was agreed by the chair of the Dutch Medicines Evaluation Board (MEB) Committee on Clinical Practice. Information about this pilot study was provided in verbal and written format. All participants were asked to sign for informed consent.
2.4.4. Participants
Participants were recruited via the network of the authors. To foster a wide spread in hand–eye capacities, not only older people (age ≥65 years), but also adults of younger (18–45 years) and middle (45–65 years) age were approached. The purpose of the pilot study was explained verbally, and written information was supplied. Participants were excluded when missing arms or digits or when they needed help to manage their medication. Informed consent was obtained. The study was conducted at a non‐public location indicated by the participant. Participants had to wear their glasses during the experiment.
To avoid selection bias, the authors tried to include participants who are sufficiently representative for all levels of society, education level and intelligence. However, information on the participant's highest level of education was not acquired as participants may feel uncomfortable indicating, as the level in older people may be more related to social class than intelligence and as cognition may have declined upon ageing. The same test panel was used as indicated above.
2.4.5. Tablets and splitters
Immediate release, round, uncoated, 500‐mg paracetamol tablets (13 mm) and 8 mg bromhexine tablets (7 mm) were selected as suitable medicines for this experiment as 1) these tablets are commonly used in older people and their handling does not result in safety issues; 2) they can be purchased with a break‐mark approved for dose adjustment and 3) the are commercially available in commonly used plastic/aluminium blisters in cardboard boxes. The portfolio of all tablet batches on the Dutch market (paracetamol n = 9 and bromhexine n = 4) was considered to select a batch/trademark that was sufficiently representative of all products; and that would show sufficient diversity upon breaking (no floor or ceiling effects). Paracetamol Etos (RVG 33987; batch 1599093; diameter 13.08 mm; thickness 4.37 mm; crushing strength 8.2 kPa) and Bromhexin Apotex (RVG 11295, diameter 7.10 mm, thickness 2.65 mm; crushing strength 5.5 kPa) were selected because of average characteristics with regards to these parameters.
Two tablet splitters that performed best in earlier work and that comprised a fundamentally different design were selected for this study: Pilltool (cylinder shaped container and cap, diameter 40 mm, thickness 60–65 mm (cap with bevelled top), subdivide by pushing the container cap down) and HealthCare Logistics (HCL; box shaped, length 83 mm container and 86 mm cap, total thickness 17 mm at edges and 25 mm at middle top, subdivide by pushing the container cap down over the length of the box; Figure 1).14
Figure 1.

Methods for tablet subdivision: Participant hands, HealthCare Logistics tablet splitter, Pilltool tablet splitter
2.4.6. Sample size
The first step of the pilot experiment was discontinued when saturation occurred and no new suggestions for optimization of the test battery were made in 3 participants in a row, e.g. the investigator did not have any problems adhering to the resting periods or writing down the results meanwhile, the patients understood the instructions, the time for the experiment was about 1 h, there were no new voluntary participant remarks etc. The optimized battery was evaluated in the same participants (additional data only where needed) and a similar number of new participants with a minimum of 30 participants in total.
2.4.7. Procedure
The aim of the study was explained to the participants verbally for the second time and any remaining questions were addressed. It was verified that the participants had signed informed consent, did not obtain assisted care for their medication management (other than for convenience reasons), and were not suffering from incidental hand–eye conditions such as sores, infections etc. The clock was started. The following data were collected in a standardized form: participant code, sex, date of birth, handedness (right, left, ambidextrous), living situation (alone, with an adult aged ≥65 or <65 years, residence). The hand–eye measurements were performed as indicated in Figure 2.
Figure 2.
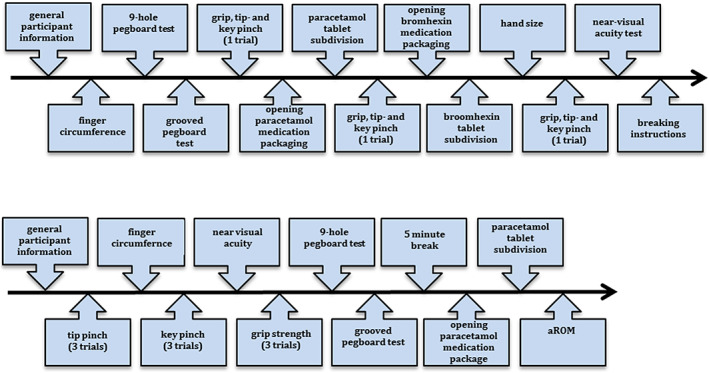
Order of the tests in the draft (top) and optimized (bottom) battery
Participants were asked to open the paracetamol and bromhexine cardboard boxes and indicate how easy/difficult this was and whether they experienced any discomfort/pain. Then participants were asked to push 3 paracetamol and 3 bromhexine tablets through their blister strip and to indicate the ease/difficulty and discomfort/pain for each tablet type. Outcomes for ease/difficulty and pain/discomfort needed to be reported on a 0–10 cm numeric rating scale (NRS), where 0 indicated “it goes poorly, it is difficult, much pain, much discomfort” (statements printed in red) and 10 “it goes well, easy, no pain, no discomfort” (statements printed in green). Patients were instructed to use the NRS as if the numbers were grades at school (1 is worst outcome possible, 10 best outcome possible and a 5.5. is rounded to a 6, which is the outcome that is considered sufficient to pass a test/exam.
Participants were then asked to break 3 paracetamol and 3 bromhexine tablets by hand and by both types of tablet splitters in a randomized order. They were asked to indicate the ease/difficulty and pain/discomfort on 0–10 cm NRS scales each time a tablet was subdivided. Participants were asked to indicate their preference for the method of subdivision.
2.4.8. Tablet handling
The tablet parts were individually stored in small plastic zipper bags. The bags from 1 participant were stored in a larger plastic zipper bag. All zipper bags were stored in a plastic box. The weight of each tablet part was measured on a precision mass balance (Mettler Toledo AG64) at Utrecht University within 1 week after the experiment was conducted. In addition, 100 tablets were individually weighed by the investigator (L.D.) on the same balance.
2.4.9. Study outcomes and data analysis (optimization procedure)
The practicality of the test battery for use with the patient and operator was investigated after testing each participant (see section 2.4.6 sample size).
It was evaluated if the hand–eye functions and NRS scores showed diversity. In addition, it was evaluated if the accuracy of the subdivided tablets showed diversity and if any floor or ceiling effects exist. For this, the average weight of the intact tablets and their weight variability was calculated. The half of the average intact tablet weight was considered to reflect the theoretical weight of half a tablet. The variability of the weight of the subdivided tablet parts was determined. It was evaluated if the weight variability of the theoretical weight of a half tablet was sufficiently small compared to the weight variability of the subdivided tablet parts. Then, the accuracy of the subdivided tablet parts was determined on basis of the theoretical weight of half a tablet rather than half of the weight of a specific tablet prior to subdivision.The test battery was considered optimized when no further changes were made in 3 participants in a row.
2.4.10. Study outcomes and data analysis (evaluation procedure)
The practicality of the optimized test battery for use with the patient and operator was investigated.
The difficulty/ease of opening the cardboard boxes and the (lack of any) discomfort/pain when pushing the paracetamol and bromhexine tablets through the blister strip was evaluated.
The ability of the participants to break the tablets was evaluated.
The ease/difficulty and (lack of any) discomfort/pain when breaking the tablets by hand or with the help of each tablet splitter was evaluated.
The participant preference for the method of subdivision was evaluated.
The suitability of the test battery for evaluating the hand–eye functions relevant to tablet subdivision was evaluated. It was evaluated if the hand–eye functions were within the normative data. Then, it was evaluated if the accuracy of the tablet parts indicated sufficient diversity among the participants in their ability to subdivide tablets. A scatterplot was made for each of the identified functions versus the difficulty/ease and discomfort/pain (i.e. acceptability) and the accuracy of tablet subdivision respectively. All scatterplots were visually evaluated if the results showed sufficient diversity and if the results suggested a relationship.
3. RESULTS
The selection of the hand–eye domains/functions and measuring instruments is summarized in Table 1.
Table 1.
Development of the test battery
| Phase | Aim | Method | Result |
|---|---|---|---|
| I | Identifying hand–eye parameters that are considered most relevant to tablet subdivision | Review of the literature |
Identified functions • hand size • hand strength • manual dexterity • vision • coordination |
| II | Selection of suitable measuring instruments for the parameters identified in phase I | Review of the literature |
Hand size: • finger circumference Manual strength: • pinch strength • grip strength Manual dexterity: • active range of joint motion • pegboard function Vision: • near visual acuity Coordination: • pegboard function |
| III | Combination of the measuring instruments into a practical test battery. | Literature & Expert panel (L.D., D.V.R.N., A.N.) | See Figure 2 |
| IV | Pilot study to evaluate and further optimize the test battery |
Study in healthy adults ‐ step 1: n = 13 ‐ step 2: n = 30 (n = 11, 21–49 years; n = 11, 50–69 years; n = 8, ≥70 years) |
See Figure 2 |
3.1. Phase I: Identifying hand–eye functions relevant to tablet subdivision
On basis of the literature search, the following domains of the hand and eye were identified as likely to be related to tablet subdivision: hand size19; hand strength20; flexibility and/or manual dexterity21; vision.10, 21, 22 There was conflicting evidence for coordination.10, 21
The literature also suggested that besides the patient hand–eye function, age23, 24 and sex10 would also have an impact on the ease and accuracy of tablet breaking. However, other authors considered that such relationships did not exist19, 25 or were just a surrogate indicator e.g. sex for hand size.26
The following functions were considered to best reflect the identified domains: finger circumference for measuring hand size27, 28; pinch and grip strength for measuring hand strength29, 30, 31; pegboard tests for measuring flexibility, manual dexterity, coordination, vision (and also cognition)32; near visual acuity for measuring vision.22
3.2. Phase II: Selecting and evaluating the measuring instruments and relevant protocols
The selected instruments are described in Table 1 and Figure 3.
Figure 3.

Measuring instruments used for the draft and/or optimized test battery. (A) Jamar dynamometer for measuring grip strength; (B) Early Treatment Diabetic Retinopathy Study chart for measuring near‐visual acuity; (C) grooved pegboard test (left) and 9‐hole pegboard test (right) for measuring manual dexterity; (D) B&L Engineering pinch gauge for measuring tip and key pinch; (E) standard ring gauge for measuring finger circumference; (F) finger goniometer for measuring active range of joint motion
3.2.1. Finger circumference (for measuring hand size)
According to the literature, the most straightforward method to measure finger circumference is the use of a measuring tape.33 As this method requires close patient contact, the use of a self‐administered standard ring gauge was preferred.19, 28 As for McDevitt et al,19 it was considered appropriate to measure the size of the index finger and thumb. However, to control the duration of the overall test‐battery, it was considered sufficient to measure the size of the dominant hand only rather than both hands as done by McDevitt et al.
3.2.2. Pinch strength (for measuring hand strength)
Pinch strength is often measured by squeezing with the fingers in a pinch meter. Different strengths can be measured e.g. tip, key, palmar pinch29, 34 and various meters can be used.34, 35, 36 As many people break tablets with the help of their index finger and/or thumb, it was considered sufficient to measure tip and key pinch. The B&L Engineering pinch meter was selected because it is the most widely used37 and recommended instrument37 for which normative data exist.31
The American Society of Hand Therapists (ASHT) has developed a protocol for measuring pinch strength in the upper extremities.31 The ASHT recommends 3 trials and 15‐second intertrial resting. However, the protocol is subject to debate. A wealth of literature supports the use of only 1 or 2 trials. Trosmann et al38 found a significantly smaller decline in grip strength form the first to third trial if using 60‐ rather than 15‐second rest times. Watanabee et al39 found a statistically significant difference between a trial with 1‐minute rest time than continuous measurement. Some authors indicate that other aspects may be of relevance to the test.40, 41, 42 Taking all this into consideration, it was considered to use the ASHT protocol, but to conduct 3 separate trials in both hands with 30‐second intertrial rest periods (1 minute per hand), to further standardize the verbal instructions and to verify that the participants hands were at room temperature.
3.2.3. Grip strength (for measuring hand strength)
Grip strength can be measured with a range of measuring instruments, e.g. hydraulic,43 pneumatic,43, 44 mechanical or strain gauges.43 The Jamar dynometer (hydraulic) was selected as it is the most widely used45 and recommended instrument37, 44, 46 with excellent reliability34 for which normative data are available.31, 47 Grip strength is commonly measured in the same way as pinch strength, but with the handle of the meter in a second position. It was decided to also adopt this approach for the test battery.43, 46
3.2.4. Pegboard tests (for measuring manual dexterity, flexibility, coordination, vision, cognition)
The National Institute of Health Toolbox considers that the 9‐hole and grooved pegboard tests are the most suitable tests for flexibility/manual dexterity.48, 49 Both tests were selected respecting the importance of National Institute of Health recommendations and acknowledging that a ceiling effect may be found for the 9‐hole pegboard test in participants with normal hand function, and a floor effect for the grooved pegboard test in participants with impaired hand function. For the 9‐hole pegboard test, the standard protocol are described by Mathiowetz et al was used and for the grooved pegboard test the protocol as described by Lafayette.50 To align the instructions for both tests,51 a practice trial for the grooved pegboard test was added, which involved filling the first row.
3.2.5. Active range of joint motion (for measuring flexibility)
Active range of joint motion (aROM) was discussed in the literature as a potentially relevant measuring instrument for finger flexibility (manual dexterity), but at the same time it was indicated that pegboard function would be at better alternative. Following discussion among the expert panel and Dutch quality assessors, it was decided to add aROM to the draft test battery for the main reason that it is a very simple and quickly to run test, whereas pegboard tests are more difficult and time consuming. Thus, the results for aROM may either further support the need for inclusion of the pegboard tests into the battery, or they may indicate that these can be replaced by aROM. In addition, finger flexibility would be something that could be simply measured in clinical practice before prescribing or dispensing half a tablet.29
aROM of the hand can be measured in different ways e.g. torque range of motion, Ex‐it, goniometry, wire grams or visual examination.27, 52 The ASHT considers goniometry as most reliable and has developed a protocol indicating e.g. the position of the participant and goniometer and standard verbal instructions. Respecting the importance of ASHT, goniometry was selected for this experiment. aROM can be measured at both the proximal and distal interphalangeal joints of the finger. The proximal location was selected as this was considered more reliable by Ellis et al.27 Controversy exist on the number of trials to perform. Several authors consider that multiple trials are needed, but Boone et al53 indicated that 1 trial might be as reliable. As aROM was considered less important than pegboard testing in the literature, it was considered sufficient to limit aROM to 1 trial in this experiment.
3.2.6. Near visual acuity
Visual acuity (and to some extent visual perception) can be measured with different methods or controlling variables such as the size of and distance to the subject, time of exposure to the subject, illumination of the subject area and contrast between the subject and the background.22 Commonly used charts are those of Snellen and the Early Treatment Diabetic Retinopathy Study (ETDRS) charts. As the Snellen chart was reported to have multiple inconsistencies in its design e.g. too few letters of large size, difficulties to remember the letter sequences,22, 54, 55 the ETDRS chart was selected for this experiment.
3.3. Phase III: Development of the draft test battery
Taking the selected measuring instruments and protocols into consideration, a draft order of the tests in the battery was proposed. It was considered best practice to order the test from less to more demanding and then to make any further changes to ensure sufficient rest times between trials and variability in tasks (Figure 2).
3.4. Phase IV: Optimization and evaluation of the (draft) test battery
3.4.1. Optimization procedure
The draft test battery (Figure 2) was improved following evaluation in 13 participants (5 males; age 22–86 years; mean 52 years). The main modifications (Annex 1) included: (i) discontinuation of the use of bromhexine tablets in order to reduce time and patient burden and the time needed to run all experiments; (ii) measuring 3 trials of grip and pinch strength in 1 row to save time and increase practicality of the test battery.
It appeared necessary to select another paracetamol tablet with average characteristics for size and shape but higher resistance to crushing [Paracetamol Actavis, RVG 18565, batch IPL256, crushing strength 11.7 kPa; diameter 12.0 mm; thickness 4.29 mm; 10 tablets per blister; 5 blisters per cardboard box; box length × height × width = 115 × 38 × 46 mm)] as all participants could easily break the Paracetamol Etos tablet by hand. Otherwise, the variety in NRS scores was considered sufficient.
Because of these changes, the order of the tests in the battery had to be adapted to ensure sufficient rest times between trials.
3.4.2. Evaluation procedure
The optimized test battery was evaluated in the same 13 participants and 17 new participants i.e. 30 participants in total (14 males; age 21–90 years; mean age 55.9 years). The participants were all able to complete the test in about 40 min. All instructions were clear and no new findings regarding the set‐up of the battery were identified. The practicality of the optimized test battery was considered adequate.
For paracetamol, opening of the cardboard boxes and pushing the tablet through the blister strip was well accepted with average NRS scores for ease/difficulty between 7 and 10. The measurements for bromhexine tablets were discontinued as an outcome of the optimization phase.
Eighty‐three percent of all participants and 58% of the participants aged 65+ years were able to break the tablets by hand and all participants were able to subdivide the tablets with both types of tablet splitter.
Subdivision by hand received the lowest scores for difficulty/ease (5.0) and discomfort/pain (6.2).
Breaking by hand was preferred by 10% of older patients with average NRS score for difficulty/ease of 3.6 for 3.4 for discomfort/pain) and 30% of young adults (with NRS score for difficulty/ease of 7.3 and 5.8 for discomfort/pain).
The suitability of the test battery for evaluating the hand–eye functions relevant to tablet breaking was determined. The tip pinch, key pinch, grip strength and pegboard times were within normative data.31, 47 There was also high agreement for finger circumference, near‐visual acuity and a‐ROM of the measured functions with population means. The participants could well be differentiated on basis of their hand–eye functions. Male and female participants could generally be differentiated on basis of their pinch and grip strength, finger circumference and manual dexterity. All hand–eye functions declined with age, except for aROM in the dominant hand of males.
The accuracy of tablet subdivision varied among participants. Breaking was the most accurate method (average weight deviation of theoretical half tablet weight 4.4%; 4.7% of the tablets parts deviated >10% and 3.4% >15% of the theoretical half tablet weight). Generally, the HCL splitter provided higher weight variability than Pilltool (>10/15% deviation: HCL 17.2/7.2% and Pilltool 15.0/5.6%). Accuracy of tablet subdivision with Pilltool was lower for older adults than for those who were younger (21–49 years (n = 11) 3.8%; 50–69 years (n = 11) 3.3%; 70+ years (n = 8) 6.8% but similar for HCL (21–49 years 5.3%; 50–69 years 3.4%; 70+ years 4.5%).
The scatterplots indicated that sufficient diversity between the participants hand–eye functions and the ease/difficulty, discomfort/pain (i.e. acceptability) and accuracy of tablet breaking respectively. The relationships between the hand–eye function and the acceptability of tablet subdivision seemed stronger for pinch and grip strength, followed by manual dexterity and weaker for finger circumference, near visual acuity and aROM. However, a greater number of participants is needed to support these findings. The results for the relationships between hand–eye functions and accuracy were more difficult to interpret. Generally, the highest deviations in weight were found for participants with specific characteristics, e.g. participants with the highest tip and key pinch, and for those with the lowest grip strength per sex. More research is needed. The relationships between grip strength and the accuracy and acceptability of tablet subdivision are depicted in Figure 4.
Figure 4.

A scatterplot indicating the relationships between 1 of the evaluated hand–eye functions (grip strength) and the accuracy and acceptability of tablet subdivision for hand broken tablets (A, B) and those subdivided by the Pilltool (PT; C, D) and HealthCare Logistics tablet splitter (E, F)
4. DISCUSSION
This study describes the development of a test battery to assess essential hand–eye functions for easy and accurate tablet subdivision by older people. Such a test battery is needed to determine which hand–eye characteristics can be considered as a determinant for the selection of the participants in a test panel that would be able to predict the ease and accuracy of tablet breaking in home‐dwelling older people during drug product development by the pharmaceutical industry or upon any postmarketing changes to the tablet design. On the basis of a review of the literature, hand size, hand strength, flexibility/manual dexterity, vision and coordination were considered as the 5 most relevant domains for hand–eye performance. Finger circumference (for hand size), pinch and grip strength (for hand strength), aROM (for flexibility), pegboard function (for flexibility, manual dexterity, coordination, vision and cognition) and near visual acuity (for vision) were selected for inclusion into the test battery.
One of the strengths of this study is its relevance to real‐world clinical practice. It is well known that older people may require assisted living when they are seriously ill, frail or otherwise unable to cope with normal daily activities and that frailty requires urgent attention in drug development and dosing.56 However, it is evident that people who cannot cope with normal daily activities will probably also not be able to subdivide tablets by hand, or even by a tablet splitter and that this has to be accepted i.e. these patients may need caregiver support for tablet subdivision. Therefore, participants who were receiving help for managing their medications were excluded from this study. However, the authors consider that, conversely, it is reasonable to expect that the majority of their caregivers and the remaining subset of self‐supportive home‐dwelling (older) adults should be able to subdivide tablets by hand or in any case by a well‐functioning tablet splitter. Respecting that the gold‐standard approach that any home‐dwelling older patient would always be able to break a tablet accurately by hand and with acceptable ease is unlikely to be reached, the study did not only evaluate tablets subdivision by hand but also by 2 well‐known tablet splitters that had proven to function best when splitting paracetamol tablets. This study has also value for other patient populations that older people who also encounter difficulties breaking tablets by hand, e.g. rheumatic patients.
To the best of our knowledge, the relationship between hand–eye functions and the ease and accuracy of tablet subdivision (hand, tablet splitter) has not been investigated. However, the relationship between the tablet characteristics and the ease and accuracy of tablet subdivision has already gained some attention. For example, Teixeira et al57 studied 15 different types of tablets that are commonly used in older people. They concluded that the accuracy of tablet subdivision by an undefined operator (probably a laboratory technician) with a kitchen knife or splitter related to the tablet excipient composition and that generally tablets that were oblong, coated and having a break‐mark performed better than tablets that were round, uncoated or not having a break‐mark.57 Pereira et al58 studied the relationship between the accuracy of tablet splitting with a commercial tablet splitter from Brazil (Incontem). They concluded that the use of the filler dicalcium phosphate dehydrate resulted in better accuracy and superior mechanical properties of the tablet parts than the use of the filler microcrystalline cellulose, but that accuracy was not affected by the use of the binder hydroxypropylcellulose or polyvinylpyrrolidone.58 Also, Gupta et al59 found that the accuracy of the division dicalcium phosphate dehydrate tablets was not affected by the binder type. Van der Steen et al60 studied round and oblong tablets in older, healthy volunteers. They concluded that the ease of breaking oblong tablets could be ensured by certain criteria for diameter, diameter/width ratio, depth of break‐mark and resistance to crushing. They also found that the ease of breaking round tablets could be predicted from a model.60 All this shows that industry has sufficient opportunities to change the tablet design to better support hand breaking tablets.
The methodological design of the study shows some flaws. The literature search was not conducted in a systematic manner but as a scoping review. The results of the test battery indicate that the review was sufficiently thorough. The study only evaluated 1 tablet type, paracetamol. As other authors have proved that tablet size, shape, excipient composition and other formulation aspects can influence tablet subdivision, the suitability of the test battery for other tablet types remains to be confirmed. However, it can be expected that no relevant changes to the test battery need to be made as the use of the paracetamol tablet resulted in a sufficient spread in data for difficulty/ease, discomfort/pain and accuracy. The sample size was based on a practical number of participants and no statistical analysis was performed other than a visual inspection of scatterplots. However, the results of the test battery indicate that these were sufficiently diverse to enable meaningful evaluation in this pilot phase.
5. CONCLUSION
On basis of a snowball review of the literature and a pilot experiment, a test battery was developed to assess the hand–eye functions relevant to easy and accurate tablet subdivision. As a next step, a formal validation study is needed. The very preliminary results of this pilot study suggest that pinch and grip strength are most important to easy and accurate tablet subdivision, followed by manual dexterity. It is likely that the outcomes of this study are also relevant to people younger than 65 years suffering from similar hand–eye impairments as those prevalent at older age.
COMPETING INTERESTS
There is no conflict to disclose according to BJCP statements. However, for transparency reasons please note the following. Anthonius de Boer, Agnes Nicia and Diana van Riet‐Nales are experts of the European Medicines Agency (EMA). Diana van Riet and Bart van den Bemt are both a member of the MEB's Committee of Clinical Practice. Anthonius de Boer is the chair of the MEB.
CONTRIBUTORS
Diana van Riet‐Nales initiated this study in close collaboration with Bart van den Bemt. Diana van Riet‐Nales, Linda Donkerbroek and Agnes Nicia conducted the literature review. Diana van Riet‐Nales and Agnes Nicia supported Linda Donkerbroek in the collection of the participant data and Christien Oussoren supported Linda in the weight measurements and the determination of the other tablet characteristics. The development of the test battery was regularly discussed in team discussions attended by all authors, except Anthonius de Boer. This publication was written by Diana van Riet‐Nales with support from Bart van den Bemt and Anthonius de Boer.
APPENDIX A.
Optimization of the draft test battery
| Sex | Age (y) | Hand | Study duration (min) | Modification test battery and/or procedure | |
|---|---|---|---|---|---|
| 1 | Female | 84 | Right | 75 |
• Pegboard tests: a standard instruction was added indicating that it was not necessary to take the pegs out of the boards • Near visual acuity: a standard instruction was added indicating that a guess should be made when reading was not clear |
| 2 | Male | 85 | Right | 60 |
• Order of tests: a 5‐min rest period was introduced before subdivision of the paracetamol and bromhexine tablets • The participant considered the study too long |
| 3 | Female | 57 | Left/ambidextrous | 55 |
• Participant data: it was decided that participants can also be registered as ambidextrous on basis of the study outcomes and participant information (recall in early days left‐handed people were obliged to write with their right hand) • Tablet breaking: it was decided not to pay attention to the way the participants broke the tablets by hand i.e. To accept the use of fingernails |
| 4 | Female | 29 | Right | 55 | • Grip strength: an instruction was added to the use of the Jamar dynometer, indicating that the handle will not move upon squeezing |
| 5 | Female | 24 | Right | Not known |
• The test was conducted at 2 moments. So, no reliable information on the duration was obtained • All tests: an instruction was added that participants will first be informed about the study details and that only thereafter questions can be asked • The next participants will be given 2 different breaking instructions. It will be evaluated with pinch orientation provides more ease and less pain during tablet breaking |
| 6 | Female | 22 | Right | Not known |
• The test was conducted at 2 times. So, no reliable information on the duration was obtained • It was decided that the strength measurements should be performed after 10.00 am • It was decided to ask participants for their order of preference (first, middle, last choice) • It was decided to use a new tablet splitter for every 3 participants |
| 7 | Female | 28 | Left | Not known |
• The participant became inpatient at the end of the study when the bromhexine tablet had to be broken. It considered the study too long • It was decided that 1 grip strength trial is not enough for accurate measurements • The use of a breaking instruction did not result in any relevant details and will be omitted in the next participants |
| 8 | Male | 59 | Right | 55 min | • None |
| 9 | Male | 66 | Right | 75 min |
• The duration of the study was reduced by deleting the subdivision of bromhexine tablets • The grip and tip pinch showed much variation among trials without clear reasons. Therefore, the average of 3 subsequent trials will be considered as indicated in the standard protocol • Expert discussion of the results up to now indicated that finger flexibility may be an issue. The literature was search and the measurement of active range of joint motion was added to the test battery as this could be done within 1 min • All this implied that the order of the procedures had to be changed to ensure sufficient rest periods between strength measurements |
| 10 | Male | 63 | Right | 45–50 |
• The modified test battery could be conducted in less time • It was considered to only state once that the pegboard tests should be conducted as fast as the participant was able |
| 11 | Female | 86 | Right | 50 | • No further modifications |
| 12 | Female | 54 | Right | 40 | • No further modifications |
| 13 | Male | 26 | Right | 55 | • No further modifications |
van Riet–Nales D, Donkerbroek L, Nicia A, Oussoren C, de Boer A, van den Bemt B. The development of a test battery to assess the hand–eye functions relevant in predicting easy and accurate tablet subdivision in older people: A pilot study. Br J Clin Pharmacol. 2020;86:1969–1981. 10.1111/bcp.14201
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Mehuys E, Dupond L, Petrovic M, et al. Medication management among home‐dwelling older patients with chronic diseases: possible roles for community pharmacists. The J of Nutr J Nutri, Healt Ag. 2012;16(8):721‐726. [DOI] [PubMed] [Google Scholar]
- 2. Dormuth CR, Schneeweiss S, Brookhart AM, et al. Frequency and predictors of tablet splitting in statin prescriptions: a population‐based analysis. Op MediOpen medicine: a peer‐reviewed, independent, open‐access journal. 2008;2(3):e74‐e82. [PMC free article] [PubMed] [Google Scholar]
- 3. Freeman MK, White W, Iranikhah M. Tablet splitting: a review of the clinical and economic outcomes and patient acceptance. Second of a 2‐part series. Part 1 was published in may 2012 (consult pharm 2012;27:239‐53). Consu Phar: J Ame Soc Cons Pharm. 2012;27(6):421‐430. [DOI] [PubMed] [Google Scholar]
- 4. Beard JR, Officer AM, Cassels AK. The world report on ageing and health. Gerontologist. 2016;56(Suppl 2):S163‐S166. [DOI] [PubMed] [Google Scholar]
- 5. Cerreta F, Eichler HG, Rasi G. Drug policy for an aging population‐‐the European medicines Agency's geriatric medicines strategy. N Engl J Med. 2012;367(21):1972‐1974. [DOI] [PubMed] [Google Scholar]
- 6. Rodenhuis N, De Smet PA, Barends DM. The rationale of scored tablets as dosage form. Eur J Pharm Sci. 2004;21(2–3):305‐308. [DOI] [PubMed] [Google Scholar]
- 7. Akhtar S. Pharmacological considerations in the elderly. Curr Opin Anaesthesiol. 2018. Feb;31(1):11‐18. [DOI] [PubMed] [Google Scholar]
- 8. Baijens LW, Clave P, Cras P, et al. European Society for Swallowing Disorders ‐ European Union geriatric medicine society white paper: oropharyngeal dysphagia as a geriatric syndrome. Clin Interv Aging. 2016;11:1403‐1428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Liu F, Ghaffur A, Bains J, Hamdy S. Acceptability of oral solid medicines in older adults with and without dysphagia: a nested pilot validation questionnaire based observational study. Int J Pharm. 2016;512(2):374‐381. [DOI] [PubMed] [Google Scholar]
- 10. Atkin PA, Finnegan TP, Ogle SJ, Shenfield GM. Functional ability of patients to manage medication packaging: a survey of geriatric inpatients. Age Ageing. 1994;23(2):113‐116. [DOI] [PubMed] [Google Scholar]
- 11. Beckman A, Bernsten C, Parker MG, Thorslund M, Fastbom J. The difficulty of opening medicine containers in old age: a population‐based study. Phar Wor Scie: PWS. 2005;27(5):393‐398. [DOI] [PubMed] [Google Scholar]
- 12. Schenk A, Eckardt‐Felmberg R, Steinhagen‐Thiessen E, Stegemann S. Patient behaviour in medication management: findings from a patient usability study that may impact clinical outcomes. Br J Clin Pharmacol. 2019;1 10.1111/bcp.13946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Quinzler R, Haefeli WE. [Tablet splitting]. Therapeutische Umschau Revue therapeutique. 2006. 63(6):441‐7. [DOI] [PubMed]
- 14. van Riet‐Nales DA, Doeve ME, Nicia AE, et al. The accuracy, precision and sustainability of different techniques for tablet subdivision: breaking by hand and the use of tablet splitters or a kitchen knife. Int J Pharm. 2014. May 15;466(1–2):44‐51. [DOI] [PubMed] [Google Scholar]
- 15. Habib WA, Alanizi AS, Abdelhamid MM, Alanizi FK. Accuracy of tablet splitting: comparison study between hand splitting and tablet cutter. Saudi Pharm JSPJ: the official publication of the Saudi Pharmaceutical Society. 2014;22(5):454‐459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Somogyi O, Mesko A, Csorba L, Szabo P, Zelko R. Pharmaceutical counselling about different types of tablet‐splitting methods based on the results of weighing tests and mechanical development of splitting devices. Eur J Pharm Sci. 2017;106:262‐273. [DOI] [PubMed] [Google Scholar]
- 17. Ciavarella AB, Khan MA, Gupta A, Faustino PJ. Dose uniformity of scored and unscored tablets: application of the FDA tablet scoring guidance for industry. PDA J Pharm Sci Technol. 2016;70(6):523‐532. [DOI] [PubMed] [Google Scholar]
- 18. Eggerth A, Hayn D, Schreier G. Medication management needs information and communications technology‐based approaches, including telehealth and artificial intelligence. Br J Clin Pharmacol. 2019;4 10.1111/bcp.14045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. McDevitt JT, Gurst AH, Chen Y. Accuracy of tablet splitting. Pharmacotherapy. 1998;18(1):193‐197. [PubMed] [Google Scholar]
- 20. Van Vooren L, De Spiegeleer B, Thonissen T, Joye P, Van Durme J, Slegers G. Statistical analysis of tablet breakability methods. J Phar & Pharm Sci: Pub Can Soc Pharma Sci, Soc Canadi Des Sci Pharma. 2002;5(2):190‐198. [PubMed] [Google Scholar]
- 21. Nikolaus T, Kruse W, Bach M, Specht‐Leible N, Oster P, Schlierf G. Elderly patients' problems with medication. An in‐hospital and follow‐up study. Eur J Clin Pharmacol. 1996;49(4):255‐259. [DOI] [PubMed] [Google Scholar]
- 22. Gittings NS, Fozard JL. Age related changes in visual acuity. Exp Gerontol. 1986;21(4–5):423‐433. [DOI] [PubMed] [Google Scholar]
- 23. Notenboom K, Vromans H, Schipper M, Leufkens HG, Bouvy ML. Relationship between age and the ability to break scored tablets. Front Pharmacol. 2016;7:222 10.3389/fphar.2016.00222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Barends DM, Groot DW, Frijlink HW, Rodenhuis N, van der Steen JC. Development of an in vivo test procedure for the ease of breaking of scored tablets. Pharmeur Sci Notes. 2005;2005(1):27‐30. [PubMed] [Google Scholar]
- 25. Abu‐Geras D, Hadziomerovic D, Leau A, et al. Accuracy of tablet splitting and liquid measurements: an examination of who, what and how. J Pharm Pharmacol. 2017;69(5):603‐612. [DOI] [PubMed] [Google Scholar]
- 26. Peters M, Servos P, Day R. Marked sex differences on a fine motor skill task disappear when finger size is used as covariate. J Appl Psychol. 1990;75(1):87‐90. [DOI] [PubMed] [Google Scholar]
- 27. Ellis B, Bruton A. A study to compare the reliability of composite finger flexion with goniometry for measurement of range of motion in the hand. Clin Rehabil. 2002;16(5):562‐570. [DOI] [PubMed] [Google Scholar]
- 28. Serup J. Ring size measurement of the digits in females suffering from generalized scleroderma (acrosclerosis). A simple method to quantify skin and soft tissue affection. Dermatologica. 1985;171(1):41‐44. [DOI] [PubMed] [Google Scholar]
- 29. Walker PS, Davidson W, Erkman MJ. An apparatus to assess function of the hand. J Hand Surg am. 1978;3(2):189‐193. [DOI] [PubMed] [Google Scholar]
- 30. Jones LA. The assessment of hand function: a critical review of techniques. J Hand Surg am. 1989;14(2 Pt 1):221‐228. [DOI] [PubMed] [Google Scholar]
- 31. Werle S, Goldhahn J, Drerup S, Simmen BR, Sprott H, Herren DB. Age‐ and gender‐specific normative data of grip and pinch strength in a healthy adult Swiss population. J Hand Surg Eur Vol. 2009;34(1):76‐84. [DOI] [PubMed] [Google Scholar]
- 32. Yancosek KE, Howell D. A narrative review of dexterity assessments. J the: Offi J am Soc Ther. 2009;22(3):258‐269. [DOI] [PubMed] [Google Scholar]
- 33. Yamazaki H, Sakurai T, Uchiyama S, Kato H. Reliability and concurrent validity of the ring gauge method and the metacarpal head tape measure method of measuring hand size. J Hand Sur Asi‐Pac Vol. 2018;23(3):382‐387. [DOI] [PubMed] [Google Scholar]
- 34. Mathiowetz V, Weber K, Volland G, Kashman N. Reliability and validity of grip and pinch strength evaluations. J Hand Surg am. 1984;9(2):222‐226. [DOI] [PubMed] [Google Scholar]
- 35. King TI 2nd. Interinstrument reliability of the Jamar electronic dynamometer and pinch gauge compared with the Jamar hydraulic dynamometer and B&L Engineering mechanical pinch gauge. Am J Occup Ther. 2013;67(4):480‐483. [DOI] [PubMed] [Google Scholar]
- 36. Simard J, Chalifoux M, Fortin V, Archambault MJ, St‐Cerny‐Gosselin A, Desrosiers J. Could questions on activities of daily living estimate grip strength of older adults living independently in the community? J ag Res. 2012;2012:427109 10.1155/2012/427109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Mathiowetz V, Vizenor L, Melander D. Comparison of baseline instruments to the Jamar dynamometer and the B&L Engineering Pinch Gauge. Occu the J Res. 2000;20(3):147‐162. [Google Scholar]
- 38. Trossman PB, Li P‐W. The effect of the duration of intertrial rest periods on isometric grip strength performance in young adults. Am Occup Ther Fou; Ame Occu the Assoc. 1989;9(6):362‐378. [Google Scholar]
- 39. Watanabe T, Owashi K, Kanauchi Y, Mura N, Takahara M, Ogino T. The short‐term reliability of grip strength measurement and the effects of posture and grip span. J Hand Surg am. 2005;30(3):603‐609. [DOI] [PubMed] [Google Scholar]
- 40. Bohannon RSN. Hand‐held dynamometry: a single trial may be adequate for measuring muscle strenght in healthy individuals. Psycho Can. 1990;42(1):6‐9. [Google Scholar]
- 41. Coldham F, Lewis J, Lee H. The reliability of one vs. three grip trials in symptomatic and asymptomatic subjects. J the: Offi J am Soc Ther. 2006;19(3):318‐326. [DOI] [PubMed] [Google Scholar]
- 42. Hamilton A, Balnave R, Adams R. Grip strength testing reliability. J Hand the: Off J Ame SocHand the. 1994;7(3):163‐170. [DOI] [PubMed] [Google Scholar]
- 43. Innes E. Handgrip strength testing: a review of the literature. Aust Occup Ther J. 1999;46:120‐140. [Google Scholar]
- 44. Roberts HC, Denison HJ, Martin HJ, et al. A review of the measurement of grip strength in clinical and epidemiological studies: towards a standardised approach. Age Ageing. 2011;40(4):423‐429. [DOI] [PubMed] [Google Scholar]
- 45. Smith RO, Benge MW. Pinch and grasp strength: standardization of terminology and protocol. Am J Occup Ther. 1985;39(8):531‐535. [DOI] [PubMed] [Google Scholar]
- 46. Fess EMC. Clinical assessment recommendations. Amer Soc Hand Ther. 1981;6‐8. [Google Scholar]
- 47. Massy‐Westropp NM, Gill TK, Taylor AW, Bohannon RW, Hill CL. Hand grip strength: age and gender stratified normative data in a population‐based study. BMC Res Notes. 2011;4:127 10.1186/1756-0500-4-127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Reuben DB, Magasi S, McCreath HE, et al. Motor assessment using the NIH toolbox. Neurology. 2013;80(11 Suppl 3):S65‐S75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Wang YC, Magasi SR, Bohannon RW, et al. Assessing dexterity function: a comparison of two alternatives for the NIH toolbox. J Hand the: Off J Ame SocHand the. 2011;24(4):313‐320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Lafayette I. Grooved pegboard test user's manual. 2002.
- 51. MCRJ, Nelles Wendy B, Haase Richard F. Practice effects in repeated neuropsychological assessments. Clin Neupsycho. 1992;6(1):32‐42. [Google Scholar]
- 52. Connolly J, Condell J, Curran K, Gardiner P. A new method to determine joint range of movement and stiffness in rheumatoid arthritic patients. Conf Proce: An Intern Conf IEEE Eng Med Bio Soc IEEE Eng MedBio Soc an Conf. 2012;2012:6386‐6389. [DOI] [PubMed] [Google Scholar]
- 53. Boone DC, Azen SP, Lin CM, Spence C, Baron C, Lee L. Reliability of goniometric measurements. Phys Ther. 1978. Nov;58(11):1355‐1360. [DOI] [PubMed] [Google Scholar]
- 54. Bailey IL, Lovie‐Kitchin JE. Visual acuity testing. From the laboratory to the clinic. Vision Res. 2013;90:2‐9. [DOI] [PubMed] [Google Scholar]
- 55. Lim LA, Frost NA, Powell RJ, Hewson P. Comparison of the ETDRS logMAR, 'compact reduced logMar' and Snellen charts in routine clinical practice. Eye. 2010;24(4):673‐677. [DOI] [PubMed] [Google Scholar]
- 56. Cerreta F. New harmonized considerations on the evaluation instruments for baseline characterisation of frailty in the European Union. Br J Clin Pharmacol. 2019;5 10.1111/bcp.14044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Teixeira MT, Sa‐Barreto LCL, Gratieri T, Gelfuso GM, Silva ICR, Cunha‐Filho MSS. Key technical aspects influencing the accuracy of tablet subdivision. AAPS PharmSciTech. 2017;18(4):1393‐1401. [DOI] [PubMed] [Google Scholar]
- 58. Pereira GRS, Taveira SF, Cunha‐Filho M, Marreto RN. The effects of fillers and binders on the accuracy of tablet subdivision. AAPS PharmSciTech. 2018;19(7):2929‐2933. [DOI] [PubMed] [Google Scholar]
- 59. Gupta A, Hunt RL, Khan MA. Influence of tablet characteristics on weight variability and weight loss in split tablets. Amer J he‐Sys Phar: AJHP: Off J Ame Soc Hea‐Sys Pharm. 2008;65(24):2326‐2328. 10.2146/ajhp080371 [DOI] [PubMed] [Google Scholar]
- 60. van der Steen KC, Frijlink HW, Schipper CM, Barends DM. Prediction of the ease of subdivision of scored tablets from their physical parameters. AAPS PharmSciTech. 2010;11(1):126‐132. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


