Abstract
Angiotensin-converting enzyme 2 (ACE2) is the receptor of the novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative agent of the coronavirus disease 2019 (COVID-19) pandemic. ACE2 has been shown to be down-regulated during coronaviral infection, with implications for circulatory homeostasis. In COVID-19, pulmonary vascular dysregulation has been observed resulting in ventilation perfusion mismatches in lung tissue, causing profound hypoxemia. Despite the loss of ACE2 and raised circulating vasoconstrictor angiotensin II (AngII), COVID-19 patients experience a vasodilative vasculopathy. This article discusses the interplay between the immune system and pulmonary vasculature and how SARS-CoV-2-mediated ACE2 disruption and AngII may contribute to the novel vascular pathophysiology of COVID-19.
Keywords: COVID-19, SARS-CoV-2, ACE2, Angiotensin II, Vasculopathy, Inflammation
Introduction
Angiotensin-converting enzyme 2 (ACE2) is the receptor of the novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), responsible for the coronavirus disease 2019 (COVID-19) pandemic (Hoffmann et al., 2020). ACE2 is a carboxypeptidase that cleaves angiotensin II (AngII) into angiotensin-(1–7), countering the vasoconstrictive effect of the angiotensin converting enzyme 1 (ACE)–AngII– angiotensin II receptor type 1 (AT1) axis of the renin–angiotensin system (RAS), an essential process in maintaining circulatory homeostasis (Alexandre et al., 2020). AngII is a vasoconstrictive peptide catalysed from the proteolytic cleavage of angiotensin I by ACE in the lung that increases blood pressure (Erdös, 1976). In coronavirus disease, ACE2 has been shown to be down-regulated in lung tissue, which has pathophysiological implications for infected patients. Although it has not been explicitly demonstrated for SARS-CoV-2, the mechanism of viral binding and internalization of ACE2 is similar to that of SARS-CoV and may even produce a more dramatic depletion given the increased affinity of SARS-CoV-2 spike protein for ACE2 (Verdecchia et al., 2020, Kuba et al., 2005, Imai et al., 2008, Wrapp et al., 2020). ACE2 is a well-characterized regulator of pulmonary vasodilation, postulated as a potential treatment for pulmonary hypertension (Li et al., 2013).
Interestingly, patients hospitalized with COVID-19 display features of pulmonary vascular dysregulation, and while ACE2 may be down-regulated in pulmonary tissue, patients experience increased vasodilatation as opposed to vasoconstriction. This would also appear discordant with the observed increase in circulating AngII. This article explores this phenomenon and postulates how despite the classical vasodilatory property of ACE2, its SARS-CoV-2-mediated down-regulation may have consequences beyond the RAS pathway that may shed light on this clinico-molecular discordance.
COVID-19 pneumonia and pulmonary vasculopathy
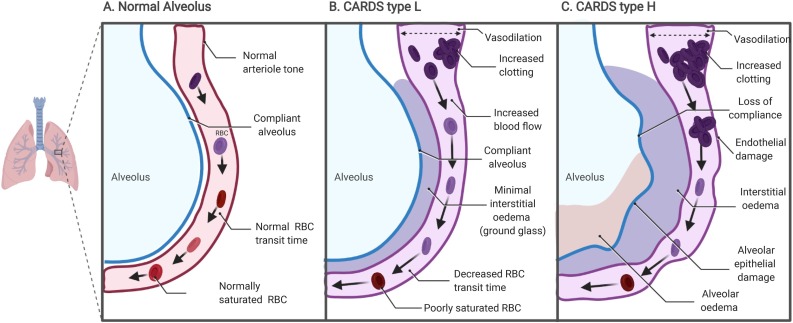
Thus far in the coronavirus pandemic, two distinct phases of COVID-19 acute respiratory distress syndrome (CARDS) have been observed: type H, a more classical acute respiratory distress syndrome picture, and type L (Gattinoni et al., 2020a, Gattinoni et al., 2020b). In CARDS type L, patients present with generally preserved lung mechanics with mild interstitial oedema evidenced by ground glass opacifications on computed tomography (CT) scan. COVID-19 patients may remain at type L and improve, or progress to type H (Gattinoni et al., 2020b). Type L patients present with profound hypoxaemia and show evidence of V/Q mismatch; a dilative pulmonary vasculopathy is observed, while alveolar ventilation is generally preserved with minimal alveolar oedema (Fig. 1) ( Gattinoni et al., 2020a, Gattinoni et al., 2020b, Marini and Gattinoni, 2020). In terms of pulmonary physiology, excessive pulmonary vasodilation increases blood flow across the alveoli, resulting in a decrease in the red blood cell transit time required for gas exchange, which may account for the hypoxia in these patients evidenced by increased right-to-left venous admixture of over 50% (normally 4–10%) (Gattinoni et al., 2020a). Pulmonary vasodilatation and congestion are also important risk factors for pulmonary emboli, both of which are observed in COVID-19 patient autopsies (Leisman et al., 2020).
Fig. 1.
Alveolar changes in CARDS type L: (A) normal lung tissue; (B) lung tissue in CARDS type L; (C) lung tissue in CARDS type H.
ACE2, angiotensin II, and pulmonary vasculopathy
The clinical observation of pulmonary vasodilation in CARDS and the presence of significantly increased AngII in COVID-19 requires further investigation (Gattinoni et al., 2020a, Liu et al., 2020). A current theory explaining this phenomenon involves the loss of hypoxic pulmonary vasoconstriction (HPV) (Gattinoni et al., 2020a). The effect of AngII on HPV has been controversial; despite its vasoconstrictive activity, AngII does not influence HPV (Hubloue et al., 2004). Conversely, recombinant ACE2 has been shown to counter HPV, indicating that the enzyme’s down-regulation in a hypoxic patient should cause further vasoconstriction (Kleinsasser et al., 2012). Additionally, AngII is elevated in patients with pulmonary hypertension, which should, in theory, steer the pathophysiology of increased AngII signalling in COVID-19 towards a vasoconstrictive and pulmonary hypertensive phenotype (Maron and Leopold, 2014). Interestingly, the conversion of AngII to angiotensin-(1–7) may even be independent of ACE2 activity in mice, indicating that the effect of ACE2 down-regulation, AngII up-regulation, and pulmonary vasodilation in COVID-19 may require investigating these proteins beyond their counter-regulatory effects on vascular tone and even beyond vascular tissue (Serfozo et al., 2020).
The immunovascular effects of ACE2 down-regulation and angiotensin II signalling
While clinical data for COVID-19 are ever-increasing, one can extrapolate the immunovascular effects of ACE2 and AngII dysregulation from in vitro and in vivo data (Table 1 ).
Table 1.
Effects of angiotensin-converting enzyme 2 (ACE2) down-regulation and angiotensin II (AngII) signalling on endothelial cells and macrophages.
eNOS, endothelial nitric oxide synthase; IL-6, interleukin 6; iNOS, inducible nitric oxide synthase; MCP1, monocyte chemoattractant protein 1; MIF, macrophage inhibitory factor; MMP2/9, matrix metalloproteinase 2/9; NA, not available; TNFα, tumour necrosis factor alpha.
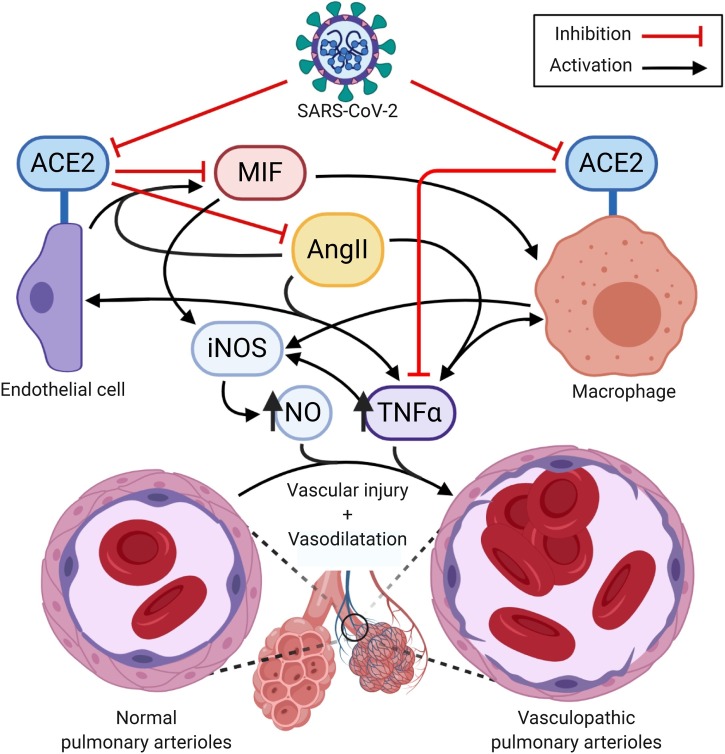
In cultured EAhy926 endothelial cells (ECs), ACE2 expression down-regulates macrophage migration inhibitory factor (MIF), a proinflammatory cytokine with implications for the pathogenesis of COVID-19 (Zhong et al., 2008). Interestingly, AngII has been shown to increase MIF expression through AT1 signalling in ECs, and is known to be proinflammatory, inducing M1 macrophages; thus both the loss of ACE2 and the observed increase in AngII may bolster cytokine expression and macrophage activity in SARS-CoV-2 infection (Zhong et al., 2008, Ruiz-Ortega et al., 2002, Nakamura et al., 1999). Moreover, ACE2 knockout mouse models have demonstrated significantly higher levels of interleukin 6 (IL-6), monocyte chemoattractant protein 1 (MCP1), matrix metalloproteinases 2 and 9 (MMP2/9), and tumour necrosis factor alpha (TNFα) in ECs, which are only further exacerbated by atherosclerosis, which is a risk factor for COVID-19 morbidity (Thomas et al., 2010, Bonow et al., 2020). Loss of ACE2 has also been shown to upregulate proinflammatory, vasodilative [Au?1] bradykinin signalling through the bradykinin B1 receptor (BKB1R), which also implicates the des-Arg bradykinin pathway in COVID-19 pulmonary vasodilation (Sodhi et al., 2018). ACE2 knockout ECs have also demonstrated increased responsiveness to TNFα, which leads to endothelial damage Thomas et al., 2010). Another knockout study in bone marrow resulted in increased expression of the macrophage markers F4/80, as well as TNFα (Thatcher et al., 2012). A SARS-CoV-2-mediated down-regulation of ACE2 in macrophages may produce a similar TNFα-mediated proinflammatory, vasodilatory effect, which is further exacerbated by both ACE2 disinhibition and AngII-mediated enhancement of vascular MIF and MCP1 expression and TNFα reactivity.
Interestingly, AngII, MIF, and TNFα are involved in vascular dysregulation and macrophage nitric oxide (NO) production, which implicates these molecules in CARDS pulmonary vasculopathy (Zhong et al., 2008, Riches et al., 1996, Wu et al., 2020, Nagareddy et al., 2005). ACE2 deficiency has also been shown to increase vascular smooth muscle reactivity to NO, and while TNFα overall supresses EC nitric oxide synthase (eNOS) activity, it remains an important inducer of NO production in Th1 responding macrophages. Moreover, TNFα and its induction of nitric oxide synthase (iNOS) and NO have pivotal roles in inducing vasodilatory shock (Fonseca et al., 2003, Rabelo et al., 2016, Vincent et al., 2000). This indicates that NO production in early COVID-19 disease by macrophages, combined with cytokine-mediated vascular injury potentiated by ACE2 down-regulation, increased AngII signalling, and viral EC lysis, may contribute to COVID-19 pulmonary vasculopathy (Fig. 2 ) (Kodukula et al., 1999, Merad and Martin, 2020). This immunovascular model of COVID-19 pathogenesis may also partially explain the effectiveness of immunosuppressive dexamethasone in COVID-19 patients requiring respiratory support (Horby et al., 2020).
Fig. 2.
Mechanism of angiotensin-converting enzyme 2 (ACE2) and angiotensin II (AngII) contribution to pulmonary vasculopathy.
ACE inhibitors and angiotensin receptor blockade
In the model presented, one cannot discuss the RAS system without addressing the effects of ACE inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) in COVID-19. Due to elevated circulating AngII in COVID-19, one would assume that modulators of the RAS system could be beneficial to limit the inflammatory effects of AngII and may increase the expression of cytoprotective ACE2 (Vaduganathan et al., 2020). In the immunovascular model presented, their use would be a double-edged sword, in that AT1 blockade or decreased AngII production would indeed decrease proinflammatory AngII signalling, however this may exacerbate the observed pulmonary vasodilation and worsen hypoxemia in CARDS type L. Therefore further studies assessing these drugs should distinguish these two types of coronaviral disease, as inflammation may play a larger role in CARDS type H (Gattinoni et al., 2020b). Lastly, there is both conflicting and insufficient evidence for the effects of ACEIs and ARBs on pulmonary ACE2 levels (Vaduganathan et al., 2020). Assuming these drugs increase ACE2, as several studies indicate, the theoretical increase in membrane-bound ACE2 would be protective against pulmonary and vascular injury, however it would also provide more binding sites for new virions and may facilitate the spread of the infection (Vaduganathan et al., 2020). Given the uncertainty in the literature and the possible risks and benefits of RAS modulation highlighted in this disease model, it is difficult to determine a precise role for RAS modulation in COVID-19 other than controlling important morbidity risk factors, namely hypertension and heart failure (Vaduganathan et al., 2020).
Conclusions
SARS-CoV-2 is a novel coronavirus displaying unprecedented clinical presentations with unique pathophysiology. Here, an immunovascular theory is postulated for pulmonary vasculopathy from SARS-CoV-2-mediated ACE2 down-regulation, resulting in vasodilatory signalling loops between ACE2-deficient macrophages, ACE2-deficient vessels, and enhanced AngII signalling, mediated by MIF, TNFα, and iNOS. While trials are underway targeting these pathways in COVID-19 patients (TNFα: NCT04425538; MIF: NCT04429555; dexamethasone: NCT04381936), we call for further investigation into this phenomenon in the hopes of increasing the understanding of COVID-19 and its pathophysiology.
Funding
None.
Ethical approval
None.
Conflict of interest
The author declares no conflict of interest.
Acknowledgements
Figures created with BioRender.com.
References
- Alexandre J., Cracowski J.L., Richard V., Bouhanick B., Drugs C-wgotFSoPT Renin-angiotensin-aldosterone system and COVID-19 infection. Ann Endocrinol (Paris) 2020;81(2-3):63–67. doi: 10.1016/j.ando.2020.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonow R.O., Fonarow G.C., O’Gara P.T., Yancy C.W. Association of Coronavirus Disease 2019 (COVID-19) With Myocardial Injury and Mortality. JAMA Cardiol. 2020;5(7):751–753. doi: 10.1001/jamacardio.2020.1105. [DOI] [PubMed] [Google Scholar]
- Erdös E.G. Conversion of angiotensin I to angiotensin II. Am J Med. 1976;60(6):749–759. doi: 10.1016/0002-9343(76)90889-5. [DOI] [PubMed] [Google Scholar]
- Fonseca S.G., Romão P.R., Figueiredo F., Morais R.H., Lima H.C., Ferreira S.H. TNF-alpha mediates the induction of nitric oxide synthase in macrophages but not in neutrophils in experimental cutaneous leishmaniasis. Eur J Immunol. 2003;33(8):2297–2306. doi: 10.1002/eji.200320335. [DOI] [PubMed] [Google Scholar]
- Gattinoni L., Chiumello D., Rossi S. COVID-19 pneumonia: ARDS or not? Critical Care. 2020;24(1):154. doi: 10.1186/s13054-020-02880-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gattinoni L., Chiumello D., Caironi P., Busana M., Romitti F., Brazzi L. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med. 2020;46(6):1099–1102. doi: 10.1007/s00134-020-06033-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181(2) doi: 10.1016/j.cell.2020.02.052. 271-80 e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horby P., Lim W.S., Emberson J., Mafham M., Bell J., Linsell L. Effect of Dexamethasone in Hospitalized Patients with COVID-19: Preliminary Report. medRxiv. 2020 2020.06.22.20137273. [Google Scholar]
- Hubloue I., Rondelet B., Kerbaul F., Biarent D., Milani G.M., Staroukine M. Endogenous angiotensin II in the regulation of hypoxic pulmonary vasoconstriction in anaesthetized dogs. Crit Care. 2004;8(4) doi: 10.1186/cc2860. R163-R71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imai Y., Kuba K., Penninger J.M. The discovery of angiotensin-converting enzyme 2 and its role in acute lung injury in mice. Exp Physiol. 2008;93(5):543–548. doi: 10.1113/expphysiol.2007.040048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleinsasser A., Pircher I., Treml B., Schwienbacher M., Schuster M., Janzek E. Recombinant angiotensin-converting enzyme 2 suppresses pulmonary vasoconstriction in acute hypoxia. Wilderness Environ Med. 2012;23(1):24–30. doi: 10.1016/j.wem.2011.09.002. [DOI] [PubMed] [Google Scholar]
- Kodukula P., Liu T., Rooijen N.V., Jager M.J., Hendricks R.L. Macrophage control of herpes simplex virus type 1 replication in the peripheral nervous system. J Immunol (Baltimore, Md: 1950) 1999;162(5):2895–2905. [PubMed] [Google Scholar]
- Kuba K., Imai Y., Rao S., Gao H., Guo F., Guan B. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus–induced lung injury. Nature Med. 2005;11(8):875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leisman D.E., Deutschman C.S., Legrand M. Facing COVID-19 in the ICU: vascular dysfunction, thrombosis, and dysregulated inflammation. Intensive Care Med. 2020;46(6):1105–1108. doi: 10.1007/s00134-020-06059-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li G., Liu Y., Zhu Y., Liu A., Xu Y., Li X. ACE2 activation confers endothelial protection and attenuates neointimal lesions in prevention of severe pulmonary arterial hypertension in rats. Lung. 2013;191(4):327–336. doi: 10.1007/s00408-013-9470-8. [DOI] [PubMed] [Google Scholar]
- Liu Y., Yang Y., Zhang C., Huang F., Wang F., Yuan J. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63(3):364–374. doi: 10.1007/s11427-020-1643-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marini J.J., Gattinoni L. Management of COVID-19 Respiratory Distress. JAMA. 2020 doi: 10.1001/jama.2020.6825. [DOI] [PubMed] [Google Scholar]
- Maron B.A., Leopold J.A. The role of the renin-angiotensin-aldosterone system in the pathobiology of pulmonary arterial hypertension (2013 Grover Conference series) Pulm Circ. 2014;4(2):200–210. doi: 10.1086/675984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merad M., Martin J.C. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol. 2020;20(6):355–362. doi: 10.1038/s41577-020-0331-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagareddy P.R., Xia Z., McNeill J.H., MacLeod K.M. Increased expression of iNOS is associated with endothelial dysfunction and impaired pressor responsiveness in streptozotocin-induced diabetes. Am J Physiol Heart Circulatory Physiol. 2005;289(5) doi: 10.1152/ajpheart.00591.2005. H2144-52. [DOI] [PubMed] [Google Scholar]
- Nakamura A., Johns E.J., Imaizumi A., Yanagawa Y., Kohsaka T. EFFECT OF β2-ADRENOCEPTOR ACTIVATION AND ANGIOTENSIN II ON TUMOUR NECROSIS FACTOR AND INTERLEUKIN 6 GENE TRANSCRIPTION IN THE RAT RENAL RESIDENT MACROPHAGE CELLS. Cytokine. 1999;11(10):759–765. doi: 10.1006/cyto.1999.0488. [DOI] [PubMed] [Google Scholar]
- Rabelo L.A., Todiras M., Nunes-Souza V., Qadri F., Szijártó I.A., Gollasch M. Genetic Deletion of ACE2 Induces Vascular Dysfunction in C57BL/6 Mice: Role of Nitric Oxide Imbalance and Oxidative Stress. PloS one. 2016;11(4) doi: 10.1371/journal.pone.0150255. e0150255-e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riches D.W., Chan E.D., Winston B.W. TNF-alpha-induced regulation and signalling in macrophages. Immunobiology. 1996;195(4-5):477–490. doi: 10.1016/s0171-2985(96)80017-9. [DOI] [PubMed] [Google Scholar]
- Ruiz-Ortega M., Ruperez M., Lorenzo O., Esteban V., Blanco J., Mezzano S. Angiotensin II regulates the synthesis of proinflammatory cytokines and chemokines in the kidney. Kidney Int. 2002;62:S12–S22. doi: 10.1046/j.1523-1755.62.s82.4.x. [DOI] [PubMed] [Google Scholar]
- Serfozo P., Wysocki J., Gulua G., Schulze A., Ye M., Liu P. Ang II (Angiotensin II) Conversion to Angiotensin-(1-7) in the Circulation Is POP (Prolyloligopeptidase)-Dependent and ACE2 (Angiotensin-Converting Enzyme 2)-Independent. Hypertension (Dallas, Tex: 1979) 2020;75(1):173–182. doi: 10.1161/HYPERTENSIONAHA.119.14071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sodhi C.P., Wohlford-Lenane C., Yamaguchi Y., Prindle T., Fulton W.B., Wang S. Attenuation of pulmonary ACE2 activity impairs inactivation of des-Arg(9) bradykinin/BKB1R axis and facilitates LPS-induced neutrophil infiltration. Am J Physiol Lung Cell Mol Physiol. 2018;314(1):L17–l31. doi: 10.1152/ajplung.00498.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thatcher S.E., Gupte M., Hatch N., Cassis L.A. Deficiency of ACE2 in Bone-Marrow-Derived Cells Increases Expression of TNF-α in Adipose Stromal Cells and Augments Glucose Intolerance in Obese C57BL/6 Mice. Int J Hypertens. 2012;2012 doi: 10.1155/2012/762094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas M.C., Pickering R.J., Tsorotes D., Koitka A., Sheehy K., Bernardi S. Genetic Ace2 Deficiency Accentuates Vascular Inflammation and Atherosclerosis in the ApoE Knockout Mouse. Circulation Res. 2010;107(7):888–897. doi: 10.1161/CIRCRESAHA.110.219279. [DOI] [PubMed] [Google Scholar]
- Vaduganathan M., Vardeny O., Michel T., McMurray J.J.V., Pfeffer M.A., Solomon S.D. Renin–Angiotensin–Aldosterone System Inhibitors in Patients with Covid-19. N Engl J Med. 2020;382(17):1653–1659. doi: 10.1056/NEJMsr2005760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verdecchia P., Cavallini C., Spanevello A., Angeli F. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur J Intern Med. 2020;76:14–20. doi: 10.1016/j.ejim.2020.04.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vincent J.L., Zhang H., Szabo C., Preiser J.C. Effects of nitric oxide in septic shock. Am J Respir Crit Care Med. 2000;161(6):1781–1785. doi: 10.1164/ajrccm.161.6.9812004. [DOI] [PubMed] [Google Scholar]
- Wrapp D., Wang N., Corbett K.S., Goldsmith J.A., Hsieh C.-L., Abiona O. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367(6483):1260. doi: 10.1126/science.abb2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu L., Chen K., Xiao J., Xin J., Zhang L., Li X., Ma K. Angiotensin II induces RAW264.7 macrophage polarization to the M1‑type through the connexin 43/NF‑κB pathway. Mol Med Rep. 2020;21:2103–2112. doi: 10.3892/mmr.2020.11023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong J.C., Yu X.Y., Lin Q.X., Li X.H., Huang X.Z., Xiao D.Z. Enhanced angiotensin converting enzyme 2 regulates the insulin/Akt signalling pathway by blockade of macrophage migration inhibitory factor expression. Br J Pharmacol. 2008;153(1):66–74. doi: 10.1038/sj.bjp.0707482. [DOI] [PMC free article] [PubMed] [Google Scholar]