Abstract
Background and Aims
Treatment options for radiation-induced intestinal injury (RIII) are limited. Crocetin has been demonstrated to exert antioxidant, antiapoptotic, and anti-inflammatory effects on various diseases. Here, we investigate the effects of crocetin on RIII in vitro. Materials and Method. IEC-6 cells exposed to 10 Gy of radiation were treated with different doses of crocetin (0, 0.1, 1, 10, and 100 μM), and cell viability was assessed by CCK-8. The levels of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), malondialdehyde (MDA), myeloperoxidase (MPO), tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interferon-γ (IFN-γ) in culture supernatants were measured using colorimetric and ELISA kits, respectively. Cellular apoptosis was evaluated by Annexin V/PI double staining.
Results
Crocetin dose-dependently improved the survival of irradiated IEC-6 cells with the optimal dose of 10 μM, as indicated by the reduction of cellular apoptosis, decreased levels of MDA, MPO, and proinflammatory cytokines (TNF-α, IL-1β, and IFN-γ), and increased activities of antioxidative enzymes (SOD, CAT, and GPx).
Conclusion
Our findings demonstrated that crocetin alleviated radiation-induced injury in intestinal epithelial cells, offering a promising agent for radioprotection.
1. Introduction
Radiation-induced intestinal injury (RIII) is a common complication of radiation therapy in patients with abdominal or pelvic malignancies, which seriously affects the quality of life and even leads to substantial mortality [1, 2]. Exposure of the small intestine to radiation may produce a large amount of free radicals and epithelial cell apoptosis, which cause impaired barrier function, followed by inflammatory response and even septicemia [3, 4]. Although RIII seriously affects the efficacy of abdominopelvic radiotherapy, there are no therapeutic agents available to attenuate the intestinal toxicity of radiation [5].
Radioprotectors targeting oxidative damage and inflammatory reaction have been studied for decades with limited success, because of either the limited protective effect or inevitable toxicity [6]. In addition, previous studies have showed that some radioprotective agents had the risk of tumorigenesis, hampering their clinical application [6, 7].
Crocetin, an active constituent of saffron (Crocus sativus L) stigma, belongs to the large family of carotenoids [8]. Accumulated evidences have demonstrated that crocetin exerted beneficial effects on injured tissue [9, 10] and tumor cells [11, 12]. It has been reported that crocetin attenuated TNBS-induced colitis in mice by reducing inflammatory cytokines and lipid peroxidation [13]. A previous study has also proved that crocetin treatment protected against burn-induced intestinal injury via inhibiting oxidative stress and inflammatory response [14]. Additionally, crocetin could inhibit the growth and metastasis of tumor cells both in vitro and in vivo [15–17]. However, the potential role of crocetin on RIII has not been reported. In this study, we aimed to investigate whether and how crocetin protected against RIII.
2. Materials and Methods
2.1. Cell Culture
Rat intestinal epithelial IEC-6 cells were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cells were maintained in high-glucose Dulbecco's modified Eagle's medium (Sigma, St Louis, MO, USA; no. D5796) with 10% fetal bovine serum, 1% penicillin/streptomycin, and 0.1 U/mL recombinant human insulin at 37°C in a humidified atmosphere containing 5% CO2. The culture medium was changed every 2 or 3 d. The cells were passaged as they grew to 70~80% confluence, and cells before 20th passages were used for the experiments.
2.2. Irradiation and Crocetin Treatment
The radiation procedure was performed according to our previously described protocols [4]. Briefly, IEC-6 cells were exposed to 10 Gy doses of radiation using a linear accelerator (Siemens PRIMUS) at a dose-rate of 300 cGy/min. IEC-6 cells were seeded into 96-well plates at a density of 1 × 104 cells/well and grown to 70%~80% confluence prior to experiment. After 10 Gy radiation, IEC-6 cells were replaced with serum-free DMEM-F12 medium and subsequently treated with different doses of crocetin (0, 0.1, 1, 10, and 100 μM, MP Biomedicals, Santa Ana, CA, USA; CAS no.: 27876-94-4), then incubated for 24 h at 37°C. After 24 h incubation, the culture medium was collected for biochemical assay and ELISA and then replaced with new fresh serum-free medium for subsequent condition of IEC-6 cells. To determine the most effective concentration of crocetin in the following experiments, cell viability was assessed daily for the next 7 days after radiation. Further studies were performed at the most effective concentration to improve cell viability.
2.3. Cell Viability Assay
The viability of IEC-6 cells was assessed by CCK-8 assay, and all the steps followed the manufacturer's instruction (Dojindo Laboratories, Kumamoto, Japan; no. CK04). IEC-6 cells were cultured in 96-well plates with a density of 1 × 104 cells/well for 24 h. After 10 Gy radiation and treatment with different doses of crocetin for 24 h, 10 μL of CCK-8 was added to each well and for incubation for another 1 h at 37°C. Cell viability was measured daily for 7 consecutive days after radiation. Absorbance of each well was determined at 450 nm using a Multiskan Spectrum (Thermo Fisher, CA, USA). The experiment was independently repeated at least three times.
2.4. Biochemical Measurements
Malondialdehyde (MDA, Beyotime Institute of Biotechnology, Shanghai, China; no. S0131) levels, superoxide dismutase (SOD, Abcam, Cambridge, MA, USA; no. ab65354) activities, catalase (CAT, Sigma, St. Louis, MO, USA; no. CAT100) activities, glutathione peroxidase (GPx, Beyotime Institute of Biotechnology, Shanghai, China; no. S0056) levels, and myeloperoxidase (MPO, Abcam, Cambridge, MA, USA; no. ab105136) activities in the cell culture supernatants were measured at 1, 3, 5, and 7 d after radiation using commercial assay kits, respectively, according to the manufacturer's protocols.
2.5. Cell Apoptosis Assay
Cell apoptosis was detected at 1, 3, 5, and 7 days after radiation using Annexin V-FITC/PI Apoptosis Detection Kit (BD Biosciences, San Diego, CA, USA) based on our previously described procedures [18]. Briefly, IEC-6 cells were plated in 6-well plates at a concentration of 1 × 105 cells/well. The cells in all groups were incubated and then harvested at 1, 3, 5, and 7 d after treatment of crocetin, washed with PBS twice, resuspended in binding buffer, and stained with Annexin V and propidium iodide (PI) for 10 min at room temperature in the dark. Annexin V fluorescence was measured using a flow cytometer (BD Biosciences), and the membrane integrity of the cells was simultaneously assessed by the PI exclusion method.
2.6. Cytokine Assay
Proinflammatory cytokines tumor necrosis factor-α (TNF-α, R&D Systems, Minneapolis, MN, USA; no. PMTA00B), interleukin-1β (IL-1β, RayBiotech, Peachtree Corners, GA, USA; no. ELM-IL1b-1), and interferon-γ (IFN-γ, R&D Systems, Minneapolis, MN, USA; no. PMIF00) levels were obtained from the cell culture supernatants at 1, 3, 5, and 7 days after radiation and were measured using ELISA kits according to the manufacturer's instructions.
2.7. Statistical Analysis
The differences of all measured parameters among groups were analyzed by one-way analysis of variance followed by Student-Newman-Keuls- (SNK-) q test and between two groups by Student t-test. All analyses were performed with SPSS statistics package (IBM SPSS, Chicago, IL, USA). Data were considered statistically significant for P < 0.05.
3. Results
3.1. Crocetin Improved the Survival of Irradiated IEC-6 Cells
To evaluate the therapeutic mechanisms of crocetin in radiation-induced intestinal injury (RIII), we established in vitro experimental systems (Figure 1). To determine the optimal concentration of crocetin on irradiated IEC-6 cells, the cell viability of each group was tested by CCK-8 assay. The cell viability of IEC-6 cells was significantly decreased after radiation (Figure 2(b)), whereas treatment with crocetin at concentrations of 0.1 μM, 1 μM, and 10 μM improved the survival of irradiated IEC-6 cells in a dose-dependent manner with the maximal effect achieved at 10 μM (Figures 2(a) and 2(b)). In contrast, 100 μM of crocetin showed a decrease on the cell viability of irradiated IEC-6 cells compared to that of the irradiated group (Figures 2(a) and 2(b)). According to the results, 10 μM was the most effective dose of crocetin to improve the viability of irradiated IEC-6 cells, which was used for subsequent experiments.
Figure 1.
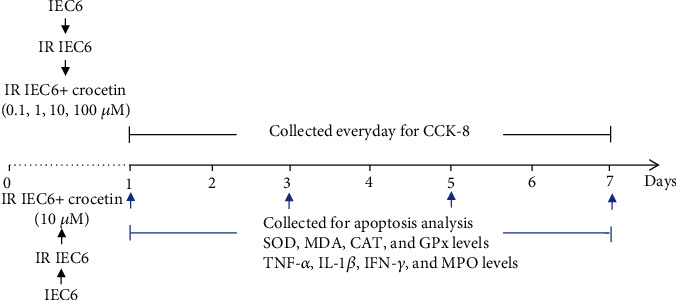
In vitro experiment design. IEC-6 cells were exposed to 10Gy of radiation, followed by treatment of different doses of crocetin (0.1 μM, 1 μM, 10 μM, and 100 μM) for 24 h, and culture supernatants were collected for CCK-8 assay from day 1 to day 7 after radiation. Apoptosis was detected on days 1, 3, 5, and 7 after radiation. The levels of SOD, CAT, GPx, MDA, MPO, TNF-α, IL-1β, and IFN-γ in culture supernatants were measured on days 1, 3, 5, and 7 after radiation. SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; MDA: malondialdehyde; MPO: myeloperoxidase; TNF-α: tumor necrosis factor-α, IL-1β: interleukin-1β, IFN-γ: interferon-γ; IR: irradiation group.
Figure 2.
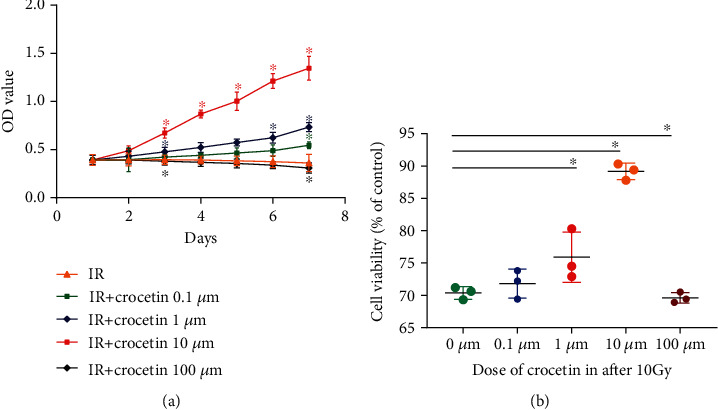
Crocetin improved the survival of irradiated IEC-6 cells in a dose-dependent manner (0.1 μM, 1 μM, and 10 μM). IEC-6 cells were treated with different doses of crocetin (0.1 μM, 1 μM, 10 μM, and 100 μM) after exposure of 10 Gy radiation. (a) Cell viability was detected by CCK-8 from day 1 to day 7 after radiation. (b) Cell viability was detected by CCK-8 on day 3 after radiation. Data represent mean ± SD of three independent experiments. ∗ represents P < 0.05 as compared to the IR group. IR group: irradiation group.
3.2. Crocetin Attenuated Oxidative Stress in Irradiated IEC-6 Cells
To investigate the effect of crocetin on oxidative stress, we examined the levels of SOD, GPx, CAT, and MDA in culture supernatants by colorimetric assays. While radiation led to increased level of MDA, this increase was alleviated by crocetin (Figure 3(b), P < 0.05). Conversely, treatment of irradiated IEC-6 cells with crocetin significantly elevated the activities of endogenous antioxidant enzymes (SOD, GPx, and CAT), compared to the irradiated group (Figures 3(a), 3(c), and 3(d), P < 0.05). These data suggested that crocetin exerted an antioxidant effect in irradiated IEC-6 cells.
Figure 3.
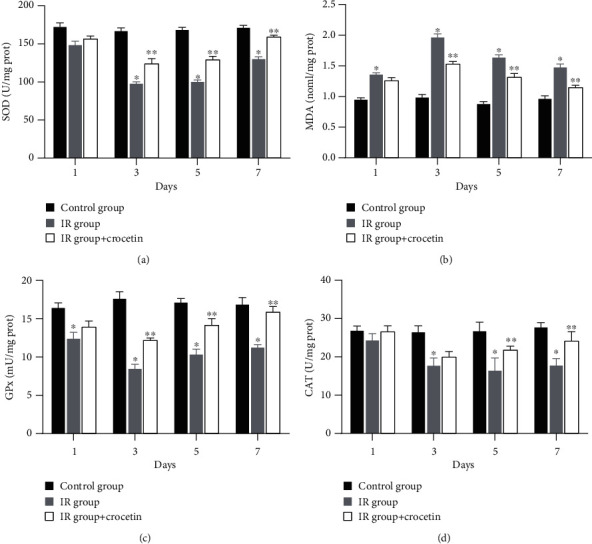
Crocetin attenuated oxidative stress in irradiated IEC-6 cells. The activities of (a) SOD, (b) MDA, (c) GPx, and (d) CAT in cell culture supernatants were detected on days 1, 3, 5, and 7 after radiation. Data were expressed as mean ± SD of three independent experiments. ∗P < 0.05 compared to the control group, ∗∗P < 0.05 compared to the IR group. SOD: superoxide dismutase; MDA: malondialdehyde; GPx: glutathione peroxidase; CAT: catalase; IR group: irradiation group, IEC-6 cells exposed to 10G y of radiation. Control group: IEC-6 cells without irradiation or crocetin.
3.3. Crocetin Ameliorated Apoptosis in Irradiated IEC-6 Cells
We further evaluated the effect of crocetin on apoptosis of irradiated IEC-6 cells by Annexin V/PI double staining. The percentage of apoptotic cells increased after radiation compared to the control group (Figure 4(b), P < 0.05), whereas crocetin dramatically reduced the apoptosis of irradiated IEC-6 cells on day 3 and day 5 (P < 0.05), with less effects on day 7 (Figures 4(a)–4(c)). These results indicated that crocetin reduced radiation-induced intestinal epithelial apoptosis.
Figure 4.
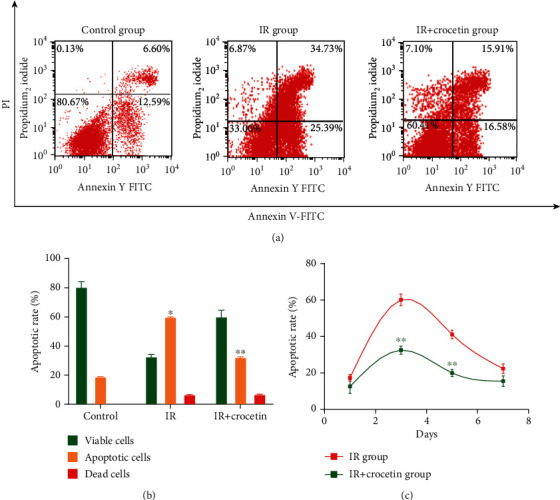
Crocetin ameliorated apoptosis in irradiated IEC-6 cells. (a) Apoptosis of IEC-6 cells was detected by flow cytometry after Annexin V/PI staining 3 days after radiation. The left upper quadrant contains necrotic cells (%); the upper right quadrant contains late apoptotic cells (%); the lower left quadrant contains live cells (%); and the lower right quadrant contains early apoptotic cells (%). (b) The percentage of total apoptotic cells and dead cells was calculated on day 3 after radiation. (c) Apoptotic ratio of IEC-6 cells was detected by Annexin V/PI double staining at 1, 3, 5, and 7 days after radiation. Data were expressed as mean ± SD of three independent experiments. ∗P < 0.05 compared to the control group, ∗∗P < 0.05 compared to the IR group. IR group: irradiation group, IEC-6 cells exposed to 10 Gy of radiation. Control group: IEC-6 cells without irradiation or crocetin.
3.4. Crocetin Inhibited Inflammation in Irradiated IEC-6 Cells
To explore the effect of crocetin on inflammatory response in irradiated IEC-6 cells, the levels of proinflammatory cytokines in culture supernatants were assessed. Exposure to radiation remarkably increased the levels of TNF-α, IL-1β, and IFN-γ, while administration of crocetin dramatically decreased this effect (Figures 5(a)–5(c), P < 0.05). Consistent with the results of proinflammatory cytokines, crocetin significantly suppressed MPO activity (Figure 5(d), P < 0.05), suggesting crocetin attenuated radiation-induced inflammation in IEC-6 cells.
Figure 5.
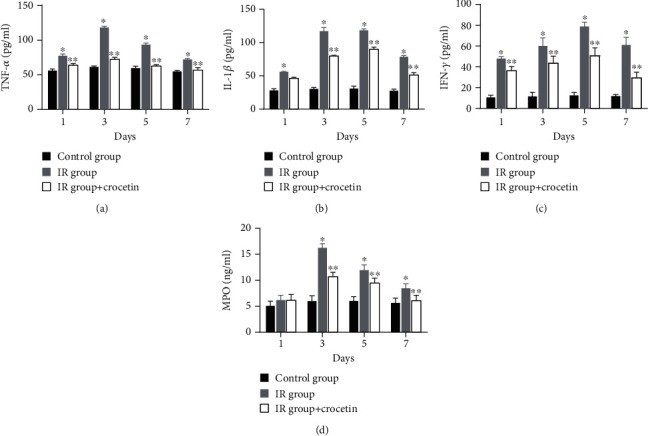
Crocetin inhibited inflammatory response in irradiated IEC-6 cells. The levels of (a) TNF-α, (b) IL-1β, (c) IFN-γ, and (d) MPO in cell culture supernatants were measured on days 1, 3, 5, and 7 after radiation. Data were expressed as mean ± SD of three independent experiments. ∗P < 0.05 compared to the control group, ∗∗P < 0.05 compared to the IR group. TNF-α: tumor necrosis factor-α, IL-1β: interleukin-1β, IFN-γ: interferon-γ; MPO: myeloperoxidase; IR group: irradiation group, IEC-6 cells exposed to 10 Gy of radiation. Control group: IEC-6 cells without irradiation or crocetin.
4. Discussion
Though agents ameliorating radiation-induced damage by reducing oxidants stress and inflammation may exert protective effects against RIII, the potential toxicity and tumorigenicity must be addressed before their clinical application [6]. In contrast, crocetin could be an alternative radioprotector for RIII with low toxicity [19, 20] and antitumor properties [11, 21]. In our study, we demonstrated the protective effects of crocetin against radiation-induced injury in intestinal epithelial cells and the underlying mechanisms could be attributed to inhibition of oxidative stress, cellular apoptosis, and inflammatory response, suggesting a safe and effective strategy for RIII.
There are some important discoveries in our work. First, the protective effects of crocetin in different concentrations on irradiated IEC-6 cells were investigated. In this study, we demonstrated that lower concentrations (0.1 μM, 1 μM, and 10 μM) of crocetin improved the survival of irradiated IEC-6 cells in a dose-dependent manner, showing the most pronounced effect at the dose of 10 μM. Consistent with our findings, Yoshino et al. found that crocetin at 1~10 μM protected HT22 cells against Aβ1-42-induced neuronal cell death [22]. Conversely, it was reported previously that high doses of crocetin exerted cytotoxic effects on healthy monocytes and Alzheimer's disease monocytes [23]. Our study also found that 100 μM of crocetin decreased cell viability whereas no cytotoxicity was observed at 0.1~10 μM, suggesting the safe concentration of crocetin should be lower than 100 μM. These findings suggested that 10 μM was relatively a safe and effective dose of crocetin to protect irradiated IEC-6 cells.
Second, our study demonstrated the mechanisms of crocetin on RIII. Previous studies showed that crocetin exerted beneficial effects on tissue regeneration by reducing oxidative stress, inhibiting cellular apoptosis, and attenuating inflammatory response [24–26]. Recently, a study further investigated that crocetin protected ultraviolet A radiation-induced skin damage by reducing oxygen species production and cellular apoptosis [27]. Similar with these studies, we observed that crocetin inhibited oxidative stress, the occurrence of apoptosis, and inflammation in irradiated IEC-6 cells, suggesting crocetin could attenuate intestinal toxicity induced by radiation.
Though some substances have shown variable degrees of radioprotective properties, the application of most agents is hindered by toxicity and narrow therapeutic time windows [28]. Crocetin has been reported to treat a wide range of diseases with low toxicity [19, 20]. Milajerdi et al. suggested that LD50 values of saffron stigma extracts containing crocetin could be very higher than the therapeutic dose [29]. A clinical study also reported that no adverse changes in volunteers were observed after crocetin was administrated at the dose of 37.5 mg/d for 4 weeks [30]. Moreover, crocetin could inhibit the proliferation and invasion of various tumor cells including intestinal cancer [31]. Kim et al. have demonstrated that crocetin could increase the death of HCT-116 colorectal cancer cells [11]. Ray et al. have also demonstrated that crocetin could induce p53-mediated cell death by p73-mediated FAS-FADD-caspase-8 activation and BID cleavage in colorectal cancer cells [32]. As RIII commonly occurs in patients with abdominopelvic malignancies receiving local radiation therapy, crocetin represents a promising therapy to attenuate radiation-induced injury of intestine and, at the same time, inhibit tumor growth. However, the possible optimal doses in vivo still need to be further studied.
This study has potential limitations. First, the effect of crocetin on tumor cell lines after radiation was not studied because previous studies have reported the antitumor effects of crocetin on various tumor cells [11, 31]. Second, the solubility and the bioavailability of crocetin require optimization before being used as an effective radioprotective agent [12]. This problem may be solved with cyclodextrins or similar molecules. For example, Wong et al. suggested that crocetin-γ-cyclodextrin inclusion complex could enhance the solubility, bioavailability, and applicability of crocetin [33]. Puglia et al. showed that solid lipid nanoparticles containing crocetin improved its solubility, stability, and pharmacokinetic properties, offering an appropriated approach to resolve this issue [34]. Third, this is a study in cell model only providing preclinical clues for radioprotection of crocetin; more studies are needed to demonstrate its effects on RIII in animal models and further in clinical studies.
5. Conclusions
In conclusion, the present study suggests that crocetin could be an attractive agent for RIII not only attenuating intestinal injury induced by radiation via inhibiting oxidative stress, cellular apoptosis, and inflammatory response but also improving the efficacy of cancer cure with potential antitumor effects.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant Nos. 81300279 and 81741067), the Guangdong Province Natural Science Foundation (Grant Nos. 2016A030313815 and 2017A030313464), the Science and Technology Planning Project of Guangdong Province (Grant No. 2015A020212029), the Science and Technology Program of Guangzhou (Grant Nos. 201707010419 and 201804010050), the Medical Scientific Research Foundation of Guangdong Province (Grant Nos. A2018343 and A2019063), the Fundamental Research Funds for the Central Universities (Grant No. D2180600), the Scientific Research Project of Traditional Chinese Medicine Bureau of Guangdong Province (Grant Nos. 20201009 and 20201010), and the High-Level Hospital Construction Project (Grant Nos. DFJH201803, KJ012019099, and DFJH201923).
Contributor Information
Jun Liang, Email: guiandlong@163.com.
Weihong Sha, Email: shaweihong@gdph.org.cn.
Hao Chen, Email: chenhao@gdph.org.cn.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Authors' Contributions
Chen Zhang, Kequan Chen, Jinghua Wang, Zhongwen Zheng, and Yujun Luo contributed equally to this work.
References
- 1.Li Y., Dong J., Xiao H., et al. Gut commensal derived-valeric acid protects against radiation injuries. Gut Microbes. 2020;11(4):789–806. doi: 10.1080/19490976.2019.1709387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kim J.-S., Yang M., Lee C.-G., Kim S.-D., Kim J.-K., Yang K. In vitro and in vivo protective effects of granulocyte colony-stimulating factor against radiation-induced intestinal injury. Archives of Pharmacal Research. 2013;36(10):1252–1261. doi: 10.1007/s12272-013-0164-9. [DOI] [PubMed] [Google Scholar]
- 3.Moussa L., Usunier B., Demarquay C., et al. Bowel radiation injury: complexity of the pathophysiology and promises of cell and tissue engineering. Cell Transplantation. 2016;25(10):1723–1746. doi: 10.3727/096368916x691664. [DOI] [PubMed] [Google Scholar]
- 4.Chen H., Min X. H., Wang Q. Y., et al. Pre-activation of mesenchymal stem cells with TNF-α, IL-1β and nitric oxide enhances its paracrine effects on radiation-induced intestinal injury. Scientific Reports. 2015;5(1):p. 8718. doi: 10.1038/srep08718. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 5.Shadad A. K., Sullivan F. J., Martin J. D., Egan L. J. Gastrointestinal radiation injury: prevention and treatment. World Journal of Gastroenterology. 2013;19(2):199–208. doi: 10.3748/wjg.v19.i2.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Andreassen C. N., Grau C., Lindegaard J. C. Chemical radioprotection: a critical review of amifostine as a cytoprotector in radiotherapy. Seminars in Radiation Oncology. 2003;13(1):62–72. doi: 10.1053/srao.2003.50006. [DOI] [PubMed] [Google Scholar]
- 7.Hauerjensen M., Wang J., Denham J. Bowel injury: current and evolving management strategies. Seminars in Radiation Oncology. 2003;13(3):358–371. doi: 10.1016/s1053-4296(03)00032-8. [DOI] [PubMed] [Google Scholar]
- 8.Giaccio M. Crocetin from saffron: an active component of an ancient spice. Critical Reviews in Food Science and Nutrition. 2004;44(3):155–172. doi: 10.1080/10408690490441433. [DOI] [PubMed] [Google Scholar]
- 9.Ishizuka F., Shimazawa M., Umigai N., et al. Crocetin, a carotenoid derivative, inhibits retinal ischemic damage in mice. European Journal of Pharmacology. 2013;703(1-3):1–10. doi: 10.1016/j.ejphar.2013.02.007. [DOI] [PubMed] [Google Scholar]
- 10.Wang Y., Sun J., Liu C., Fang C. Protective effects of crocetin pretreatment on myocardial injury in an ischemia/reperfusion rat model. European Journal of Pharmacology. 2014;741:290–296. doi: 10.1016/j.ejphar.2014.07.052. [DOI] [PubMed] [Google Scholar]
- 11.Kim S. H., Lee J. M., Kim S. C., Park C. B., Lee P. C. Proposed cytotoxic mechanisms of the saffron carotenoids crocin and crocetin on cancer cell lines. Biochemistry and Cell Biology. 2014;92(2):105–111. doi: 10.1139/bcb-2013-0091. [DOI] [PubMed] [Google Scholar]
- 12.Colapietro A., Mancini A., D'Alessandro A. M., Festuccia C. Crocetin and crocin from saffron in cancer chemotherapy and chemoprevention. Anti-Cancer Agents in Medicinal Chemistry. 2019;19(1):38–47. doi: 10.2174/1871520619666181231112453. [DOI] [PubMed] [Google Scholar]
- 13.Qian Z., Kazi H. A. Crocetin reduces TNBS-induced experimental colitis in mice by downregulation of NFkB. Saudi Journal of Gastroenterology. 2009;15(3):181–187. doi: 10.4103/1319-3767.54750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhou C., Bai W., Chen Q., et al. Protective effect of crocetin against burn-induced intestinal injury. Journal of Surgical Research. 2015;198(1):99–107. doi: 10.1016/j.jss.2015.05.052. [DOI] [PubMed] [Google Scholar]
- 15.Bathaie S. Z., Hoshyar R., Miri H., Sadeghizadeh M. Anticancer effects of crocetin in both human adenocarcinoma gastric cancer cells and rat model of gastric cancer. Biochemistry and Cell Biology. 2013;91(6):397–403. doi: 10.1139/bcb-2013-0014. [DOI] [PubMed] [Google Scholar]
- 16.Dhar A., Mehta S., Dhar G., et al. Crocetin inhibits pancreatic cancer cell proliferation and tumor progression in a xenograft mouse model. Molecular Cancer Therapeutics. 2009;8(2):315–323. doi: 10.1158/1535-7163.mct-08-0762. [DOI] [PubMed] [Google Scholar]
- 17.Chen B., Hou Z. H., Dong Z., Li C. D. Crocetin downregulates the proinflammatory cytokines in methylcholanthrene-induced rodent tumor model and inhibits COX-2 expression in cervical cancer cells. BioMed Research International. 2015;2015:5. doi: 10.1155/2015/829513.829513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chen H., Zhang H., Zheng Y., et al. Prolyl hydroxylase 2 silencing enhances the paracrine effects of mesenchymal stem cells on necrotizing enterocolitis in an NF-κB-dependent mechanism. Cell Death & Disease. 2020;11(3):p. 188. doi: 10.1038/s41419-020-2378-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mohamadpour A. H., Ayati Z., Parizadeh M. R., Rajbai O., Hosseinzadeh H. Safety evaluation of crocin (a constituent of saffron) tablets in healthy volunteers. Iranian Journal of Basic Medical Sciences. 2013;16(1):39–46. [PMC free article] [PubMed] [Google Scholar]
- 20.Hashemi M., Hosseinzadeh H. A comprehensive review on biological activities and toxicology of crocetin. Food and Chemical Toxicology. 2019;130:44–60. doi: 10.1016/j.fct.2019.05.017. [DOI] [PubMed] [Google Scholar]
- 21.Festuccia C., Mancini A., Gravina G. L., et al. Antitumor effects of saffron-derived carotenoids in prostate cancer cell models. BioMed Research International. 2014;2014:12. doi: 10.1155/2014/135048.135048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yoshino Y., Ishisaka M., Umigai N., Shimazawa M., Tsuruma K., Hara H. Crocetin prevents amyloid β1-42-induced cell death in murine hippocampal cells. Pharmacology & Pharmacy. 2014;5(1):37–42. doi: 10.4236/pp.2014.51007. [DOI] [Google Scholar]
- 23.Tiribuzi R., Crispoltoni L., Chiurchiù V., et al. Trans-crocetin improves amyloid-β degradation in monocytes from Alzheimer’s disease patients. Journal of the Neurological Sciences. 2017;372:408–412. doi: 10.1016/j.jns.2016.11.004. [DOI] [PubMed] [Google Scholar]
- 24.Nam K. N., Park Y. M., Jung H. J., et al. Anti-inflammatory effects of crocin and crocetin in rat brain microglial cells. European Journal of Pharmacology. 2010;648(1-3):110–116. doi: 10.1016/j.ejphar.2010.09.003. [DOI] [PubMed] [Google Scholar]
- 25.Niska K., Santos-Martinez M. J., Radomski M. W., Inkielewicz-Stepniak I. CuO nanoparticles induce apoptosis by impairing the antioxidant defense and detoxification systems in the mouse hippocampal HT22 cell line: protective effect of crocetin. Toxicology in Vitro. 2015;29(4):663–671. doi: 10.1016/j.tiv.2015.02.004. [DOI] [PubMed] [Google Scholar]
- 26.Tseng T. H., Chu C. Y., Huang J. M., Shiow S. J., Wang C. J. Crocetin protects against oxidative damage in rat primary hepatocytes. Cancer Letters. 1995;97(1):61–67. doi: 10.1016/0304-3835(95)03964-X. [DOI] [PubMed] [Google Scholar]
- 27.Ohba T., Ishisaka M., Tsujii S., et al. Crocetin protects ultraviolet A-induced oxidative stress and cell death in skin in vitro and in vivo. European Journal of Pharmacology. 2016;789:244–253. doi: 10.1016/j.ejphar.2016.07.036. [DOI] [PubMed] [Google Scholar]
- 28.Hauer-Jensen M., Denham J. W., Andreyev H. J. N. Radiation enteropathy—pathogenesis, treatment and prevention. Nature Reviews Gastroenterology & Hepatology. 2014;11(8):470–479. doi: 10.1038/nrgastro.2014.46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Milajerdi A., Djafarian K., Hosseini B. The toxicity of saffron (Crocus sativus L.) and its constituents against normal and cancer cells. Journal of Nutrition & Intermediary Metabolism. 2016;3:23–32. doi: 10.1016/j.jnim.2015.12.332. [DOI] [Google Scholar]
- 30.Yamashita S., Kakinuma T., Umigai N., Takara T. Safety evaluation of excessive intake of crocetin in healthy adult volunteers: - a randomized, double-blind, placebo-controlled, parallel-group comparison trial. Japanese Pharmacology and Therapeutics. 2018;46(3):393–401. [Google Scholar]
- 31.Gutheil W. G., Reed G., Ray A., Anant S., Dhar A. Crocetin: an agent derived from saffron for prevention and therapy for cancer. Current Pharmaceutical Biotechnology. 2012;13(1):173–179. doi: 10.2174/138920112798868566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ray P., Guha D., Chakraborty J., et al. Crocetin exploits p53-induced death domain (PIDD) and FAS-associated death domain (FADD) proteins to induce apoptosis in colorectal cancer. Scientific Reports. 2016;6(1, article 32979) doi: 10.1038/srep32979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wong K. H., Xie Y., Huang X., et al. Delivering crocetin across the blood-brain barrier by using γ-cyclodextrin to treat Alzheimer’s disease. Scientific Reports. 2020;10(1):p. 3654. doi: 10.1038/s41598-020-60293-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Puglia C., Santonocito D., Musumeci T., et al. Nanotechnological approach to increase the antioxidant and cytotoxic efficacy of crocin and crocetin. Planta Medica. 2019;85(3):258–265. doi: 10.1055/a-0732-5757. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon reasonable request.


