Abstract
Subjects formerly born preterm subsequently develop arterial - particularly isolated systolic- hypertension more frequently than their peers born at term.
Numerous factors may influence this predisposition, including an incomplete nephrogenesis, implying the presence of kidneys with a reduced number of nephrons and consequent reduction in haematic filtration, increased sodium absorption and activation of renin-angiotensin-aldosterone system, increased arterial rigidity produced by an elastin deficiency previously observed in anatomic specimens of human immature aorta, and reduced endothelial nitric oxide excretion, due to high blood levels of ADMA, a strong direct inhibitor of nitric oxide that exerts a vasoconstrictor effect.
Other possible factors (i.e. excretion of neuroendocrine compounds) may also be implicated. The aim of this paper was to review all possible mechanisms involved in the observed increase in blood pressure in individuals who had been born preterm and/or with intrauterine growth restriction. The outlook for new and promising laboratory techniques capable of identifying alterations in the metabolic pathways regulating blood pressure levels, such as metabolomics, is also provided.
Keywords: Prematurity, preterm birth, low birth weight, blood pressure, hypertension, perinatal programming
1. Introduction
Prematurity at birth, i.e. birth before the 37th week of gestation, is a condition affecting approx. 10% of the general population [1]. It is frequently associated with low birth weight/intrauterine growth restriction and may lead to a series of consequences in adulthood, including early development of high blood pressure (BP) [2]. Strong scientific evidence based on large epidemiological studies support the association between preterm birth and hypertension (HTN) [3], with reports confirming how more than 50% of individuals born preterm are affected by HTN [4]. As a general rule, the lower the gestational age, the higher the BP will be. For instance, at 24-hour ambulatory blood pressure monitoring (24-h ABPM) subjects born extremely preterm show a rise in BP involving both systolic and diastolic values, as well as lack of the physiological dip of nightime values (non-dipper pattern). These BP abnormalities tend to soften in line with an increase in gestational age, leading to a normalization of diastolic BP, although systolic values may continue to increase. Nocturnal BP decline is usually still lacking. In subjects born late preterm, i.e. between 34 weeks and 0 days and 36 weeks and 6 days, systolic and diastolic values are generally within the normal range, with a persistence of non-dipper nocturnal pattern in spite of the size and weight of these subjects corresponding to measurements of some infants born at term [5]. It has been suggested that preterm birth may represent a higher risk factor for increased BP than intrauterine growth restriction [6]. When these two conditions are associated, the risk of HTN rises markedly [7].
2. Purpose of the review
This review was aimed at identifying the underlying relationship between prematurity at birth/intrauterine growth restriction and development of elevated BP later in life, as the latter is a well-known risk factor for ischaemic heart disease and stroke. The following topics were reviewed in this study:
the role of incomplete kidney formation in the pathogenesis of HTN;
the effect of preterm birth on the development of the elastic structures of the arterial walls;
the influence of increased blood levels of asymmetric dimethylarginine (ADMA), an endogenous vasoconstrictor compound, on BP levels;
the involvement of other still poorly understood factors;
the outlook for possible future therapeutic strategies.
3. Methods
3.1. Search Strategy
A systematic electronic search into Pubmed/Medline, Scopus, and Web of Science databases was performed from their inception up to March 31st, 2019. All searches were conducted using Boolean operators to combine MeSH terms and free text words. The following terms were searched: blood (arterial) pressure, hypertension, prematurity, preterm(s), birth weight, intrauterine growth restriction (or retardation), kidney(s), renal development, nephrogenesis, nephron(s), aortic stiffness, aortic rigidity, aortic elasticity, aortic compliance, elastin, asymmetric dimethylarginine (ADMA), corticosteroids, therapy, therapeutic strategy(-ies), metabolomics, stem cells.
3.2. Other Sources
Reference lists of the identified papers and relevant manuscripts were examined. Emails were sent to the Authors of the above articles for further information when needed. Four reviewers were calibrated for the identification of eligible studies (Bassareo PP, Calcaterra G, Crisafulli A, and Kelleher S) by a fifth reviewer (Mercuro G) through successive stages of quadruplicate independent screening among randomly selected titles and abstracts in groups of five, until a complete intra-examiner agreement was obtained (k scores from the first to the last calibration exercise: 0.85, 0.88, 0.92 and 1). A parallel, triple-blind screening procedure of all titles and abstracts retrieved by the electronic search was performed by four reviewers (Bassareo PP, Calcaterra G, Crisafulli A, and Kelleher S). The titles and abstracts were screened for subject importance. Studies that were not definitively excluded on the basis of abstract information were also selected for full-text screening. The reviewers examined the full text of all relevant studies to evaluate the possibility of inclusion. In the case of disagreement over study inclusion, a discussion was held with the fifth reviewer (Mercuro G) to reach an agreement. Studies were eligible for inclusion on meeting the following criterion: all studies relating to the development of elevated BP in subjects born preterm and/or with intrauterine growth retardation with no limitation on the period of publication. The exclusion criteria were as follows:
studies not focused on the topic selected; (b) papers in a language other than English; (c) duplicates, and (d) studies not available from libraries for full-text assessment.
3.3. Data Extraction
Four reviewers (Bassareo PP, Calcaterra G, Crisafulli A, and Kelleher S) independently extracted data using a structured form specifically developed for this review. The following information was extracted from each included study: authors, year of publication, type of paper, raise in BP in subjects born with low birth weight and/or intrauterine growth restriction, renal impairment, increased aortic stiffness, increased production and/or reduced excretion of ADMA, possible therapeutic strategies to predict and/or prevent and/or cure the above-stated increase.
Thus, five possible outcomes were recognized: (a) renal impairment as a possible cause of this increase in arterial BP; (b) increased aortic stiffness as another possible cause of high BP; (c) increased production and/or reduced excretion of ADMA associated with surge in BP; (d) other possible mechanisms related to HTN development in this specific setting; and (e) suggestions on strategies to avoid HTN development in these subjects.
Any discrepancy that occurred during data extraction was solved by consensus or discussion with the fifth reviewer (Mercuro G).
3.4. Data Synthesis
As this review included a large number of heterogeneous study designs and sources, the results and key information obtained were summarized by means of a narrative approach.
4. Results
4.1. Literature Search
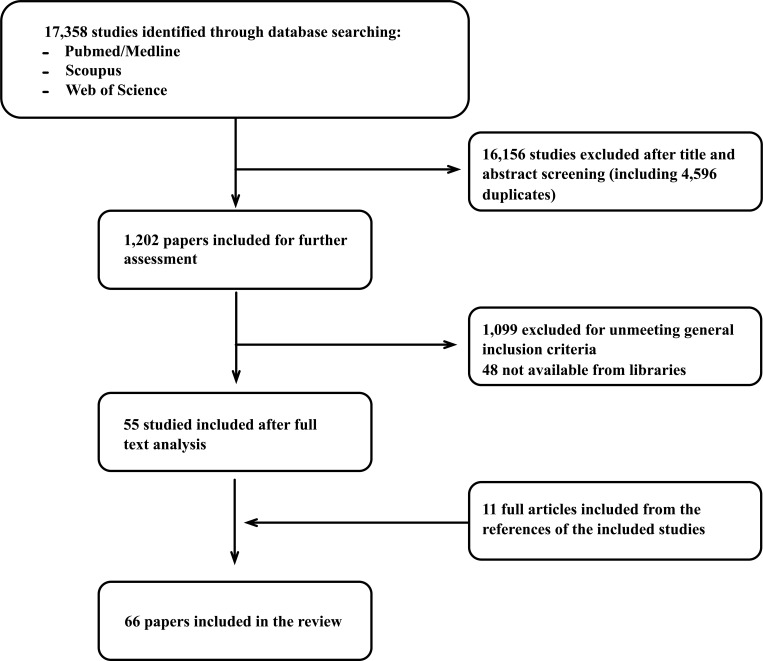
The literature search yielded a total of 17,358 primary studies, of which 16,156 (4,596 duplicates) were excluded following the screening of the title and abstract. Ultimately, 1,202 studies were included for full-text assessment based on a relevant title and abstract. A total of 1,099 were excluded as they failed to meet general inclusion criteria, and 48 were not available from online libraries. As a result, 55 studies met the inclusion criteria, and an additional 11 papers were included after checking the references of the relevant selected reviews and studies. A flow diagram of the study selection process is presented in Fig. 1. The characteristics of the selected studies are summarized in Table 1.
Fig. (1).
Flow chart of the study selection process.
Table 1.
Clinical characteristics of the enrolled studies.
| Authors | Main Characteristic(s) of the Research | |
|---|---|---|
| Luyckx VA [1] | Preterm birth epidemiology. Link between prematurity at birth and renal/cardiovascular diseases. | |
| Bassareo PP, et al. [2] | Correlation between preterm birth and high blood pressure. | |
| de Jong F, et al. [3] | Correlation between preterm birth and high blood pressure. | |
| Mercuro G, et al. [4] | Prematurity at birth and low birth weight as new cardiovascular risk factors. | |
| Sipola-Leppänen M, et al. [5] | 24-hours ambulatory blood pressure monitoring in former preterm subjects. | |
| Steen E, et al. [6] | Trade-off between prematurity at birth and low birth weight in leading to early high blood pressure onset. | |
| Juonala M, et al. [7] | Effect of birth weight on blood pressure. | |
| Richir MC, et al. [8] | Preterm birth and incomplete kidneys development. | |
| Hinchliffe SA, et al. [9] | The effect of intrauterine growth retardation on the development of renal nephrons. | |
| Koeda Y, et al. [10] | Serum C-reactive protein levels and cardiovascular events in chronic kidney disease. | |
| Böger RH, et al. [11] | The role of asymmetric dimethylarginine in the link between end-stage renal disease and adverse cardiovascular events. | |
| Rinat C, et al. [12] | Cardiovascular risk factors, cardiac function and vascular disease in children with chronic renal failure. | |
| Ritz E, et al. [13] | Renal involvement in blood pressure levels regulation. | |
| Tulassay T, et al. [14] | Relationship between birth weight and renal function. | |
| Kusano E. [15] | Relationship between renal diseases and cardiovascular diseases. | |
| Eriksson JG, et al. [16] | Relationship between prematuruty at birth and coronary artery disease. | |
| South AM, et al. [17] | Blood pressure and renal function impairments in adolescents who had been born preterm. | |
| Tauzin L. [18] | Relationship between preterm birth and altered arterial elastic properties. | |
| Rossi P, et al. [19] | Roles of preterm birth and fetal growth restriction in blood pressure and arterial stiffness. | |
| Berry CL, et al. [20] | Aorta development. | |
| Leung DY, et al. [21] | Aorta and pulmonary arteries postnatal growth. | |
| Bendek MP, et al. [22] | Arteries development. | |
| Resnick N, et al. [23] | Vascular endothelial cells development. | |
| Jensen DE, et al. [24] | Elastin synthesis in aortic smooth muscle cell. | |
| Berry CL, et al. [25] | Elastin synthesis in in aortic smooth muscle cells. | |
| Berry CL, et al. [26] | Aortic artery walls development. | |
| Martin CN, et al. [27] | Relationship between elastin synthesis impairment and hypertension development. | |
| Chemla D, et al. [28] | Arterial elasticity evaluation. | |
| Tauzin L, et al. [29] | Characteristics of arterial stiffness in very low birth weight premature infants. | |
| Bonami AK, et al. [30] | Relationship among preterm birth, increased vascular resistance, and high blood pressure. | |
| Roberts G, et al. [31] | 24-h ambulatory blood pressure monitoring in former extremely preterm newborns. | |
| Tauzin L, et al. [32] | Increased systemic blood pressure and arterial stiffness in young adults born prematurely. | |
| Oren A, et al. [33] | Influence of gestational age and birth weight on aortic stiffness. | |
| Bassareo PP, et al. [34] | Impaired central arterial elasticity in young adults born with intrauterine growth restriction. | |
| Tauzin L. [35] | Pathogenesis of hypertension in former preterm subjects. | |
| Franco MC, et al. [36] | Effects of low birth weight on endothelial function. | |
| Authors | Main Characteristic(s) of the Research | |
| Payne GA, et al. [37] | Animal model (impaired endothelial vascular relaxation in growth-restricted offspring of pregnant rats with reduced uterine perfusion). | |
| Bassareo PP, et al. [38] | Reduced brachial flow-mediated vasodilation in young adult ex extremely low birth weight preterm. | |
| Koenigsberger M, et al. [39] | Role of endothelium on arterial vasomotion. | |
| Bassareo PP, et al. [40] | Increased aymmetric dimethylarginine and early appearance of endothelial dysfunction in young adult ex extremely low birth weight preterm. | |
| Sonmez A, et al. [41] | Apelin and ADMA levels in patients with hypertension. | |
| Curgunlu A, et al. [42] | Plasma apelin and ADMA levels in patients with hypertension. | |
| Bassareo PP, et al. [43] | Inverse correlation between gestational age and aymmetric dimethylarginine renal excretion. | |
| Bertagnolli M, et al. [44] | Endothelial colony-forming cells impaired function as a possible cause of increased cardiovascular disease in young adults born preterm. | |
| Bassareo PP, et al. [45] | Possible early development of cardio-renal syndrome in individuals who had been born preterm. | |
| Bassareo PP, et al. [46] | Possible early development of cardio-renal syndrome in individuals who had been born preterm. | |
| Nuyt AM, et al. [47] | Perinatal programming of hypertension. | |
| Godfrey KM, et al. [48] | Perinatal programming of hypertension. | |
| Johanson G, et al. [49] | Prematurity at birth and adrenergic hyperactivity. | |
| Cohen G, et al. [50] | Prematurity at birth and adrenergic hyperactivity. | |
| Drake AG, et al. [51] | Prematurity at birth and hypothalamus-pituitary-adrenal axis dysregulation. | |
| Hellström A, et al. [52] | Generalized abnormal microvascularisation as the underlying mechanism of high blood pressure in those born preterm. | |
| Lewandowski AJ, et al. [53] | Capillary rarefaction as the main cause of hypertension during adulthood in preterm born offspring. | |
| Parkinson JR, et al. [54] | Preterm birth and metabolic syndrome appearance. | |
| Hovi P, et al. [55] | Female gender and maternal pre-eclampsia as causes of hypertension during adulthood in preterm born offspring appearance. | |
| Bassareo PP, et al. [56] | Prematurity at birth and adrenergic hyperactivity. | |
| Raaijmakers A, et al. [57] | Preterm birth as a factor capable of influencing cardiovascular and renal function. | |
| Falkner B. [58] | Maternal and gestational influences on childhood blood pressure. | |
| Kumar RK, et al. [59] | Optimizing diet in preterm newborns as a tool to prevent high blood pressure development. | |
| Singhal A, et al. [60] | Early nutrition in preterm infants and its influence on later blood pressure. | |
| Singhal A, et al. [61] | Ensuring a not too fast weight gain in preterm babies as a preventive tool against high blood pressure development. | |
| Bassareo PP, et al. [62] | Stem cells therapy. | |
| Bertagnolli M, et al. [63] | Endothelial colony‐forming cells impairment in young adults born preterm. | |
| Pintus R, et al. [64] | Metabolomics as a new tool to understand the mechanisma underlying the perinatal programming of cardiovascular diseases. | |
| Barker DJ, et al. [65] | “Barker” hypothesis. | |
| Chatmethakul T, et al. [66] | Intrauterine growth restriction and prematurity as risk factors for hypertension. | |
Our search resulted in a list of 10 articles responding to outcome (a), 18 articles targeting outcome (b), 11 articles focused on outcome (c), 12 articles related to outcome (d), and 6 articles for the outcome (e).
4.2. Incomplete Kidney Formation
Preterm birth may cause an interruption in the renal parenchyma maturation process, known as nephrogenesis, which is normally completed by the 31st-32nd week of gestation [8]. Accordingly, many preterm newborns are delivered prior to the completion of nephrogenesis. The lower the gestational age and the earlier the birth, the more incomplete the nephrogenesis.
The latter likewise implies that kidneys have an overall reduced number of filtration units (nephrons) [9]. An inverse relationship between the number of nephrons and gestational age/birthweight has been shown in both animal and human studies [10, 11]. The consequences of an incomplete nephrogenesis result in reduced renal filtration surface area, hyperfiltration of each nephron, glomerular sclerosis, and increased apoptosis (programmed cell death) [12]. In a variant of this hypothesis, the onset of high BP is not caused by a reduced number of nephrons - which is balanced by compensatory hypertrophy and unchanged total filtration surface area - but is due to post-glomerular renal modifications with increased sodium reabsorption, activation of the renin-angiotensin-aldosterone system, and generalized arterial vasoconstriction [13].
The two immature kidneys play a pivotal role in predicting the future development of arterial HTN and cardiovascular diseases in adulthood; this is known as the Barker hypothesis or fetal origin hypothesis [14]. Increased BP is encountered in early adulthood in subjects born preterm [15]. Not only is this true, but Erikson et al, analyzing a wide cohort of men born in Helsinki over the decade 1934-1944, showed an inverse correlation between the onset of high BP and birthweight [16]. The strength of this association may be modified by overweight/obesity and sex [17].
4.3. Arterial Elastic Properties Impairment
Prematurity at birth significantly affects elastin synthesis and content and viscoelastic properties of the vascular extracellular matrix in human arterial vessels, thus causing a likely permanent increase in arterial stiffness and BP [18]. In this respect, in a study comparing a group of adolescents born with low birth weight as a result of preterm birth and a control group of small for gestational age born at term, the former displayed increased BP and carotid-radial pulse. This finding indicates that preterm birth, rather than weight at birth, causes a rise in BP and arterial stiffness in adolescents [19].
The period between the 25th and 42nd week of gestation is characterized by a significant arterial remodeling. Indeed, the elastin/collagen ratio, which plays a crucial role in arterial compliance, increases progressively during this interval. These data support the importance of the latter part of pregnancy in the development of arterial walls [20]. During foetal life, the collagen content in the aorta increases gradually from the 12th to 25th week, after which it tends to stabilize.
Conversely, elastin production increases much more significantly after the 25th week of gestation and ceases a few weeks after birth [21]. This is because elastin accumulation in vascular walls is usually stimulated by the increase in blood cortisol in late gestation [22]. Elastin is synthesized by smooth muscle cells through the release of endothelial factors such as platelet-derived growth factor and insulin like growth factors [22-24]. For all these reasons in preterm babies the elastin/collagen ratio is significantly lower than that in term infants [25]. This impairment in elastogenesis leads to permanent structural changes in vascular walls structure [26]. These findings are consistent with the theory that an early impairment in elastin synthesis during foetal life cannot subsequently be reversed, thus causing persistence of increased arterial rigidity [27]. A deficiency in elastin had indeed been observed previously in anatomic samples of human immature aorta [25].
Other scientific evidence suggests that the abdominal aorta in young adults born with a very low birthweight (<1,500 Kg) is narrower than normal, which may explain at least in part the upward trend in BP [28]. Indeed, a decrease in arterial compliance has been detected in preterm babies since their birth [29]. Again, the presence of a persistent increase in BP, pulse pressure, and vascular resistance has been reported in preterm girls after puberty [30]. An unfavourable trend has also been demonstrated, with systolic BP in childhood being related to an increased BP in adolescents born extremely preterm (<28 weeks) [27, 31, 32]. Compared to individuals born at term, those born preterm also display an increased peripheral arterial stiffness as assessed by pulse wave velocity measurement using a 24-h ABPM device [19]. An inverse correlation has been shown between low birthweight and peripheral increased arterial rigidity that is even stronger in young adults born prematurely. This relationship was purportedly not influenced by BP, gender, and age in young adults [33]. The same inverse correlation has been reported when considering central aortic elasticity. In this respect, former preterm subjects appear to have a much older “vascular age” than their healthy peers born at term [34, 35].
4.4. Impairment in Arterial Vasodilatation
There is wide epidemiologic and experimental evidence that preterm birth and low birth weight are linked with impaired endothelium-dependent vasodilatation at birth and in childhood [36, 37]. Furthermore, this shared abnormal condition also persists in early adulthood [38]. With regard to the still poorly understood reasons for this observation, it is well-known that arterial vasomotion, i.e. the variations in arterial diameter, is induced by calcium-dependent contractions of smooth muscle cells in the tunica media. However, the endothelium is also involved in this mechanism, releasing vasodilatory (nitric oxide, NO) and vasoconstrictive compounds [39]. The strongest of the latter is represented by the endogenous NO synthase inhibitor ADMA [40]. Elevated blood levels of ADMA contribute to endothelial dysfunction and exacerbated vasoconstriction. They have been demonstrated in patients with varying degrees of HTN [41, 42], as well as in young adults born preterm, in whom ADMA levels are inversely correlated with gestational age [40]. In addition, the lower the gestational age, the lower the renal excretion of ADMA, resulting in consequently high levels in the blood [43].
Endothelial progenitor cells play a crucial role in vascular development and repair. Cord blood endothelial progenitor cells of preterm-born infants, especially endothelial colony-forming cells, show enhanced susceptibility to prematurity-related pro-oxidant stress. In vitro studies carried out on preterm-born young adults demonstrated a marked dysfunction in these cells compared with full-term controls. In the former preterm-born group, this dysfunction was associated with vasoconstriction, and in turn with increased systolic BP [44].
A significant relationship between increased ADMA and impaired renal function was found in young adults who had been born preterm, thus suggesting that the two mechanisms together may contribute to the early development of a cardio-renal syndrome and subsequently elevated BP [45, 46].
4.5. Other Possible Mechanisms
The pathways by which prematurity at birth may affect the development of high BP are complicated and in part still poorly studied. Oxidative stress, inflammation, epigenetic changes, and neuroendocrine changes may be implicated [47]. For example, human and animal models have suggested that oxidative stress and inflammation linked with preterm birth or intrauterine growth restriction may cause vascular dysfunction and high BP [47]. Another hypothesized mechanism considers intrauterine growth restriction as an epigenetic factor capable of inducing a modification of the germline by DNA methylation [48]. Again, further possible aetiologies that should be investigated in greater depth relate to alterations in the autonomic nervous system and hypothalamus-pituitary-adrenal axis [48-51].
Generalized abnormal microvascularisation has also been proposed as the mechanism underlying the onset of high BP in those born preterm [52]. With regard to this possible aetiology, a recent study suggested that capillary rarefaction is the main cause of HTN during adulthood in preterm-born offspring regardless of other cardiovascular structural and functional abnormalities [53].
According to other Authors, the relationship between prematurity at birth/intrauterine growth restriction and HTN is mediated by the onset of metabolic syndrome [54].
Other possible concomitant causes of early HTN development in individuals born preterm include female gender, maternal pre-eclampsia, and corticosteroid administration. In the past, the latter were extensively administered to prevent or reduce the incidence and severity of chronic lung disease in preterm newborns, but may also induce a detrimental long-term effect on BP [55, 56].
Due to these frequently contrasting hypotheses, the PREMATCH (PREMATurity as a predictor of Cardiovascular-renal Health) study was recently set up to characterize in-depth micro and macrovascular structure/function in former preterm birth survivors (8-13 years) compared with gender- and age-matched controls. The aim of this ongoing study was to collect information on cardiovascular-renal function in this clinical setting [57].
Lastly, recent, although limited, data seem to indicate that assisted reproductive technologies, often correlated with prematurity at birth, may also be associated with the early development of high BP despite an otherwise normal gestation and healthy newborn [58].
4.6. Possible Future Therapeutic Strategies
Although among preterm newborns the number of survivors shows an upward trend owing to the steady progress in neonatal care, conversely prematurity at birth per se and its linked comorbidities such as intrauterine growth restriction/low birth weight continue to represent a challenging scenario, resulting in undesirable complications even in the medium and long-term.
Planning strategies aimed at lowering the risk of complications in prematurity at birth survivors are crucial. A correct nutrition is essential for growth and development, and early administration of optimal nutrition decreases the incidence of adverse health outcomes and improves cognition in adulthood. Accordingly, a panel of neonatologists, pediatricians, and nutrition experts suggested a series of evidence-based suggestions for the optimization of diet in preterm newborns. With regard to the positive effect produced on BP, feeding with breast milk and control of weight gain were recommended [59].
Scientific evidence highlights how the consumption of breastmilk exerts a beneficial effect on BP in later life and contributes towards regulating weight gain [60, 61].
Tissue developmental arrest appears to be the hallmark of the preterm birth-related sequelae. Loss of stem/progenitor cells may be the underlying cause. These hypotheses constitute the basis for the application of stem cell-based therapeutical protocols following preterm birth. Transplantation of different types of stem cells, including endothelial progenitor cells in view of their role in vascular development and repair, may be beneficial in the prevention and/or treatment of HTN in subjects formerly born preterm. However, before this new knowledge can be translated into clinical practice, several issues still need to be addressed in vitro and in vivo models [62]. Indeed, for the first time, a recently published study displayed a dysfunction in endothelial progenitor cells isolated from peripheral blood in formerly preterm adults. This condition was strongly related to increased BP [63].
Metabolomics is a new technology which allows a picture to be taken of the metabolic state of an individual exposed to different environmental influences or who is affected by pathological conditions, with the aim of preventing the development of the latter and facilitate treatment.
Metabolomics is potentially capable of identifying the metabolic shifts underlying the progressive development of high BP in former preterm individuals even before clinical manifestation. Moreover, early predictive biomarkers of inborn errors may also be discovered. This technology appears to be so promising that several companies are attempting to establish kits capable of immediately assessing metabolic variations in order to provide a faster diagnosis and the best specific treatment for the patient, thus advancing the development of a personalized (tailored) medicine [64].
Conclusion
The well-established theory of foetal programming of cardiovascular diseases in adulthood relies on the occurrence of adaptive responses of the embryo or foetus to a pathological utero-placental environment. Early alteration in vascular development is likely the cornerstone of foetal programming of cardiovascular diseases in adulthood [65].
Preterm birth and intrauterine growth restriction represent risk factors not only for the survival of newborns, but also for their future cardiometabolic health. Unfortunately, new therapeutic discoveries have not been developed at the same speed as rapidly improving perinatal survival [66].
Overall, although in the trade-off between the above-described pathological mechanisms it is hard to choose one which outweighs the others, both incomplete kidney formation and increased arterial stiffness appear to be the the most widely implicated, particularly in view of the extensive scientific evidence supporting their involvement. In line with the steady progress in neonatal intensive care, a new population of subjects prone to developing HTN is on the horizon.
Acknowledgements
Declared none.
Consent for Publication
Not applicable.
Standard OF Reporting
PRISMA guidelines and methodologies were followed.
Funding
None.
Conflict of Interest
The authors declare no conflict of interest, financial or otherwise.
References
- 1.Luyckx V.A. Preterm birth and its impact on renal health. Semin. Nephrol. 2017;37(4):311–319. doi: 10.1016/j.semnephrol.2017.05.002. [DOI] [PubMed] [Google Scholar]
- 2.Bassareo P.P., Fanos V., Mercuro G. Response to ‘In adolescence, extreme prematurity is associated with significant changes in the microvasculature, elevated blood pressure and increased carotid intima-media thickness’. Arch. Dis. Child. 2015;100(5):508–509. doi: 10.1136/archdischild-2014-308155. [DOI] [PubMed] [Google Scholar]
- 3.de Jong F., Monuteaux M.C., van Elburg R.M., Gillman M.W., Belfort M.B. Systematic review and meta-analysis of preterm birth and later systolic blood pressure. Hypertension. 2012;59(2):226–234. doi: 10.1161/HYPERTENSIONAHA.111.181784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mercuro G., Bassareo P.P., Flore G., et al. Prematurity and low weight at birth as new conditions predisposing to an increased cardiovascular risk. Eur. J. Prev. Cardiol. 2013;20(2):357–367. doi: 10.1177/2047487312437058. [DOI] [PubMed] [Google Scholar]
- 5.Sipola-Leppänen M., Karvonen R., Tikanmäki M., et al. Ambulatory blood pressure and its variability in adults born preterm. Hypertension. 2015;65(3):615–621. doi: 10.1161/HYPERTENSIONAHA.114.04717. [DOI] [PubMed] [Google Scholar]
- 6.Steen E., Bonamy A.K., Norman M., Hellström-Westas L. Preterm birth may be a larger risk factor for increased blood pressure than intrauterine growth restriction. Acta Paediatr. 2015;104(11):1098–1103. doi: 10.1111/apa.13095. [DOI] [PubMed] [Google Scholar]
- 7.Juonala M., Cheung M.M., Sabin M.A., et al. Effect of birth weight on life-course blood pressure levels among children born premature: The cardiovascular risk in young finns study. J. Hypertens. 2015;33(8):1542–1548. doi: 10.1097/HJH.0000000000000612. [DOI] [PubMed] [Google Scholar]
- 8.Richir M.C., van Leeuwen P.A., van den Berg A., et al. Plasma ADMA concentrations at birth and mechanical ventilation in preterm infants: A prospective pilot study. Pediatr. Pulmonol. 2008;43(12):1161–1166. doi: 10.1002/ppul.20886. [DOI] [PubMed] [Google Scholar]
- 9.Hinchliffe S.A., Lynch M.R., Sargent P.H., Howard C.V., Van Velzen D. The effect of intrauterine growth retardation on the development of renal nephrons. Br. J. Obstet. Gynaecol. 1992;99(4):296–301. doi: 10.1111/j.1471-0528.1992.tb13726.x. [DOI] [PubMed] [Google Scholar]
- 10.Koeda Y., Nakamura M., Tanaka F., et al. Serum C-reactive protein levels and death and cardiovascular events in mild to moderate chronic kidney disease. Int. Heart J. 2011;52(3):180–184. doi: 10.1536/ihj.52.180. [DOI] [PubMed] [Google Scholar]
- 11.Böger R.H., Zoccali C. ADMA: A novel risk factor that explains excess cardiovascular event rate in patients with end-stage renal disease. Atheroscler. Suppl. 2003;4(4):23–28. doi: 10.1016/S1567-5688(03)00030-8. [DOI] [PubMed] [Google Scholar]
- 12.Rinat C., Becker-Cohen R., Nir A., et al. A comprehensive study of cardiovascular risk factors, cardiac function and vascular disease in children with chronic renal failure. Nephrol. Dial. Transplant. 2010;25(3):785–793. doi: 10.1093/ndt/gfp570. [DOI] [PubMed] [Google Scholar]
- 13.Ritz E. Hypertension: The kidney is the culprit even in the absence of kidney disease. Kidney Int. 2007;71(5):371–372. doi: 10.1038/sj.ki.5002142. [DOI] [PubMed] [Google Scholar]
- 14.Tulassay T., Vásárhelyi B. Birth weight and renal function. Curr. Opin. Nephrol. Hypertens. 2002;11(3):347–352. doi: 10.1097/00041552-200205000-00013. [DOI] [PubMed] [Google Scholar]
- 15.Kusano E. Mechanism by which chronic kidney disease causes cardiovascular disease and the measures to manage this phenomenon. Clin. Exp. Nephrol. 2011;15(5):627–633. doi: 10.1007/s10157-011-0461-x. [DOI] [PubMed] [Google Scholar]
- 16.Eriksson J.G., Forsén T., Tuomilehto J., Osmond C., Barker D.J. Early growth and coronary heart disease in later life: longitudinal study. BMJ. 2001;322(7292):949–953. doi: 10.1136/bmj.322.7292.949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.South A.M., Nixon P.A., Chappell M.C., et al. Renal function and blood pressure are altered in adolescents born preterm. Pediatr. Nephrol. 2019;34(1):137–144. doi: 10.1007/s00467-018-4050-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tauzin L. Alterations in viscoelastic properties following premature birth may lead to hypertension and cardiovascular disease development in later life. Acta Paediatr. 2015;104(1):19–26. doi: 10.1111/apa.12815. [DOI] [PubMed] [Google Scholar]
- 19.Rossi P., Tauzin L., Marchand E., Boussuges A., Gaudart J., Frances Y. Respective roles of preterm birth and fetal growth restriction in blood pressure and arterial stiffness in adolescence. J. Adolesc. Health. 2011;48(5):520–522. doi: 10.1016/j.jadohealth.2010.08.004. [DOI] [PubMed] [Google Scholar]
- 20.Berry C.L. The growth and development of large arteries. Exp. Embryol. Teratol. 1974;1(0):34–64. [PubMed] [Google Scholar]
- 21.Leung D.Y., Glagov S., Mathews M.B. Elastin and collagen accumulation in rabbit ascending aorta and pulmonary trunk during postnatal growth. Correlation of cellular synthetic response with medial tension. Circ. Res. 1977;41(3):316–323. doi: 10.1161/01.RES.41.3.316. [DOI] [PubMed] [Google Scholar]
- 22.Bendeck M.P., Keeley F.W., Langille B.L. Perinatal accumulation of arterial wall constituents: Relation to hemodynamic changes at birth. Am. J. Physiol. 1994;267(6 Pt 2):H2268–H2279. doi: 10.1152/ajpheart.1994.267.6.H2268. [DOI] [PubMed] [Google Scholar]
- 23.Resnick N., Gimbrone M.A., Jr Hemodynamic forces are complex regulators of endothelial gene expression. FASEB J. 1995;9(10):874–882. doi: 10.1096/fasebj.9.10.7615157. [DOI] [PubMed] [Google Scholar]
- 24.Jensen D.E., Rich C.B., Terpstra A.J., Farmer S.R., Foster J.A. Transcriptional regulation of the elastin gene by insulin-like growth factor-I involves disruption of Sp1 binding. Evidence for the role of Rb in mediating Sp1 binding in aortic smooth muscle cells. J. Biol. Chem. 1995;270(12):6555–6563. doi: 10.1074/jbc.270.12.6555. [DOI] [PubMed] [Google Scholar]
- 25.Berry C.L., Looker T., Germain J. Nucleic acid and scleroprotein content of the developing human aorta. J. Pathol. 1972;108(4):265–274. doi: 10.1002/path.1711080402. [DOI] [PubMed] [Google Scholar]
- 26.Berry C.L., Looker T. An alteration in the chemical structure of the aortic wall induced by a finite period of growth inhibition. J. Anat. 1973;114(Pt 1):83–94. [PMC free article] [PubMed] [Google Scholar]
- 27.Martyn C.N., Greenwald S.E. Impaired synthesis of elastin in walls of aorta and large conduit arteries during early development as an initiating event in pathogenesis of systemic hypertension. Lancet. 1997;350(9082):953–955. doi: 10.1016/S0140-6736(96)10508-0. [DOI] [PubMed] [Google Scholar]
- 28.Chemla D., Hébert J.L., Coirault C., et al. Total arterial compliance estimated by stroke volume-to-aortic pulse pressure ratio in humans. Am. J. Physiol. 1998;274(2):H500–H505. doi: 10.1152/ajpheart.1998.274.2.H500. [DOI] [PubMed] [Google Scholar]
- 29.Tauzin L., Rossi P., Giusano B., et al. Characteristics of arterial stiffness in very low birth weight premature infants. Pediatr. Res. 2006;60(5):592–596. doi: 10.1203/01.pdr.0000242264.68586.28. [DOI] [PubMed] [Google Scholar]
- 30.Bonamy A.K., Bendito A., Martin H., Andolf E., Sedin G., Norman M. Preterm birth contributes to increased vascular resistance and higher blood pressure in adolescent girls. Pediatr. Res. 2005;58(5):845–849. doi: 10.1203/01.PDR.0000181373.29290.80. [DOI] [PubMed] [Google Scholar]
- 31.Roberts G., Lee K.J., Cheong J.L., Doyle L.W., Victorian Infant Collaborative Study Group Higher ambulatory blood pressure at 18 years in adolescents born less than 28 weeks’ gestation in the 1990s compared with term controls. J. Hypertens. 2014;32(3):620–626. doi: 10.1097/HJH.0000000000000055. [DOI] [PubMed] [Google Scholar]
- 32.Tauzin L., Rossi P., Grosse C., et al. Increased systemic blood pressure and arterial stiffness in young adults born prematurely. J. Dev. Orig. Health Dis. 2014;5(6):448–452. doi: 10.1017/S2040174414000385. [DOI] [PubMed] [Google Scholar]
- 33.Oren A., Vos L.E., Bos W.J., et al. Gestational age and birth weight in relation to aortic stiffness in healthy young adults: Two separate mechanisms? Am. J. Hypertens. 2003;16(1):76–79. doi: 10.1016/S0895-7061(02)03151-5. [DOI] [PubMed] [Google Scholar]
- 34.Bassareo P.P., Saba L., Puddu M., Fanos V., Mercuro G. Impaired central arterial elasticity in young adults born with intrauterine growth restriction. Int. Angiol. 2017;36(4):362–367. doi: 10.23736/S0392-9590.16.03327-7. [DOI] [PubMed] [Google Scholar]
- 35.Tauzin L. Reply to letter by Bassareo regarding the article of Tauzin et al. ‘Increased systemic blood pressure and arterial stiffness in young adults born prematurely’. J. Dev. Orig. Health Dis. 2015;6(2):126. doi: 10.1017/S2040174415000070. [DOI] [PubMed] [Google Scholar]
- 36.Franco M.C., Christofalo D.M., Sawaya A.L., Ajzen S.A., Sesso R. Effects of low birth weight in 8- to 13-year-old children: implications in endothelial function and uric acid levels. Hypertension. 2006;48(1):45–50. doi: 10.1161/01.HYP.0000223446.49596.3a. [DOI] [PubMed] [Google Scholar]
- 37.Payne J.A., Alexander B.T., Khalil R.A. Reduced endothelial vascular relaxation in growth-restricted offspring of pregnant rats with reduced uterine perfusion. Hypertension. 2003;42(4):768–774. doi: 10.1161/01.HYP.0000084990.88147.0C. [DOI] [PubMed] [Google Scholar]
- 38.Bassareo P.P., Fanos V., Puddu M., et al. Reduced brachial flow-mediated vasodilation in young adult ex extremely low birth weight preterm: A condition predictive of increased cardiovascular risk? J. Matern. Fetal Neonatal Med. 2010;23(Suppl. 3):121–124. doi: 10.3109/14767058.2010.506811. [DOI] [PubMed] [Google Scholar]
- 39.Koenigsberger M., Sauser R., Bény J.L., Meister J.J. Role of the endothelium on arterial vasomotion. Biophys. J. 2005;88(6):3845–3854. doi: 10.1529/biophysj.104.054965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bassareo P.P., Puddu M., Flore G., et al. Could ADMA levels in young adults born preterm predict an early endothelial dysfunction? Int. J. Cardiol. 2012;159(3):217–219. doi: 10.1016/j.ijcard.2011.02.069. [DOI] [PubMed] [Google Scholar]
- 41.Sonmez A., Celebi G., Erdem G., et al. Plasma apelin and ADMA Levels in patients with essential hypertension. Clin. Exp. Hypertens. 2010;32(3):179–183. doi: 10.3109/10641960903254505. [DOI] [PubMed] [Google Scholar]
- 42.Curgunlu A., Uzun H., Bavunoğlu I., Karter Y., Genç H., Vehid S. Increased circulating concentrations of asymmetric dimethylarginine (ADMA) in white coat hypertension. J. Hum. Hypertens. 2005;19(8):629–633. doi: 10.1038/sj.jhh.1001867. [DOI] [PubMed] [Google Scholar]
- 43.Bassareo P.P., Fanos V., Puddu M., Flore G., Mercuro G. Advanced intrauterine growth restriction is associated with reduced excretion of asymmetric dimethylarginine. Early Hum. Dev. 2014;90(4):173–176. doi: 10.1016/j.earlhumdev.2014.01.010. [DOI] [PubMed] [Google Scholar]
- 44.Bertagnolli M., Xie L.F., Paquette K., et al. Endothelial colony- forming cells in young adults born preterm: A novel link between neonatal complications and adult risks for cardiovascular disease. J. Am. Heart Assoc. 2018;7(14):e009720. doi: 10.1161/JAHA.118.009720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bassareo P.P., Fanos V., Noto A., et al. Clinical metabolomics and hematic ADMA predict the future onset of cardiorenal syndrome in young grown-up subjects who were born preterm. Clin. Biochem. 2014;47(6):423–426. doi: 10.1016/j.clinbiochem.2013.11.018. [DOI] [PubMed] [Google Scholar]
- 46.Bassareo P.P., Fanos V., Mussap M., et al. Urinary NGAL and hematic ADMA levels: An early sign of cardio-renal syndrome in young adults born preterm? J. Matern. Fetal Neonatal Med. 2013;26(Suppl. 2):80–83. doi: 10.3109/14767058.2013.829698. [DOI] [PubMed] [Google Scholar]
- 47.Nuyt A.M., Alexander B.T. Developmental programming and hypertension. Curr. Opin. Nephrol. Hypertens. 2009;18(2):144–152. doi: 10.1097/MNH.0b013e328326092c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Godfrey K.M., Lillycrop K.A., Burdge G.C., Gluckman P.D., Hanson M.A. Epigenetic mechanisms and the mismatch concept of the developmental origins of health and disease. Pediatr. Res. 2007;61(5 Pt 2):5R–10R. doi: 10.1203/pdr.0b013e318045bedb. [DOI] [PubMed] [Google Scholar]
- 49.Johansson S., Norman M., Legnevall L., Dalmaz Y., Lagercrantz H., Vanpée M. Increased catecholamines and heart rate in children with low birth weight: Perinatal contributions to sympathoadrenal overactivity. J. Intern. Med. 2007;261(5):480–487. doi: 10.1111/j.1365-2796.2007.01776.x. [DOI] [PubMed] [Google Scholar]
- 50.Cohen G., Vella S., Jeffery H., Lagercrantz H., Katz-Salamon M. Cardiovascular stress hyperreactivity in babies of smokers and in babies born preterm. Circulation. 2008;118(18):1848–1853. doi: 10.1161/CIRCULATIONAHA.108.783902. [DOI] [PubMed] [Google Scholar]
- 51.Drake A.J., Tang J.I., Nyirenda M.J. Mechanisms underlying the role of glucocorticoids in the early life programming of adult disease. Clin. Sci. (Lond.) 2007;113(5):219–232. doi: 10.1042/CS20070107. [DOI] [PubMed] [Google Scholar]
- 52.Hellström A., Hård A.L., Niklasson A., Svensson E., Jacobsson B. Abnormal retinal vascularisation in preterm children as a general vascular phenomenon. Lancet. 1998;352(9143):1827. doi: 10.1016/S0140-6736(05)79889-5. [DOI] [PubMed] [Google Scholar]
- 53.Lewandowski A.J., Davis E.F., Yu G., et al. Elevated blood pressure in preterm-born offspring associates with a distinct antiangiogenic state and microvascular abnormalities in adult life. Hypertension. 2015;65(3):607–614. doi: 10.1161/HYPERTENSIONAHA.114.04662. [DOI] [PubMed] [Google Scholar]
- 54.Parkinson J.R., Hyde M.J., Gale C., Santhakumaran S., Modi N. Preterm birth and the metabolic syndrome in adult life: A systematic review and meta-analysis. Pediatrics. 2013;131(4):e1240–e1263. doi: 10.1542/peds.2012-2177. [DOI] [PubMed] [Google Scholar]
- 55.Hovi P., Vohr B., Ment L.R., et al. APIC Adults born preterm international collaboration. blood pressure in young adults born at very low birth weight: Adults born preterm international collaboration. Hypertension. 2016;68(4):880–887. doi: 10.1161/HYPERTENSIONAHA.116.08167. [DOI] [PubMed] [Google Scholar]
- 56.Bassareo P.P., Abella R., Fanos V., Mercuro G. Biomarkers of corticosteroid-induced hypertrophic cardiomyopathy in preterm babies. Front. Biosci. (Elite Ed.) 2010;2:1460–1471. doi: 10.2741/e205. [DOI] [PubMed] [Google Scholar]
- 57.Raaijmakers A., Petit T., Gu Y., et al. Design and feasibility of “PREMATurity as predictor of children’s Cardiovascular-renal Health” (PREMATCH): A pilot study. Blood Press. 2015;24(5):275–283. doi: 10.3109/08037051.2015.1053220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Falkner B. Maternal and gestational influences on childhood blood pressure. Pediatr. Nephrol. 2020;35:1409–1418. doi: 10.1007/s00467-019-4201-x. [DOI] [PubMed] [Google Scholar]
- 59.Kumar R.K., Singhal A., Vaidya U., Banerjee S., Anwar F., Rao S. Optimizing nutrition in preterm low birth weight infants-consensus summary. Front. Nutr. 2017;4:20. doi: 10.3389/fnut.2017.00020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Singhal A., Cole T.J., Lucas A. Early nutrition in preterm infants and later blood pressure: Two cohorts after randomised trials. Lancet. 2001;357(9254):413–419. doi: 10.1016/S0140-6736(00)04004-6. [DOI] [PubMed] [Google Scholar]
- 61.Singhal A., Cole T.J., Fewtrell M., Deanfield J., Lucas A. Is slower early growth beneficial for long-term cardiovascular health? Circulation. 2004;109(9):1108–1113. doi: 10.1161/01.CIR.0000118500.23649.DF. [DOI] [PubMed] [Google Scholar]
- 62.Bassareo P.P., Mercuro G. Stem cells and heart: An open future or a mirage? J Pediatr Neonat Individual Med. 2016;5(1):e050102. doi: 10.7363/050102. [DOI] [Google Scholar]
- 63.Bertagnolli M., Nuyt A.M., Thébaud B., Luu T.M.M. Endothelial progenitor cells as prognostic markers of preterm birth-associated complications. Stem Cells Transl. Med. 2017;6(1):7–13. doi: 10.5966/sctm.2016-0085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Pintus R., Bassareo P.P., Dessì A., Deidda M., Mercuro G., Fanos V. Metabolomics and cardiology: Toward the path of perinatal programming and personalized medicine. BioMed Res. Int. 2017;2017:6970631. doi: 10.1155/2017/6970631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Barker D.J., Osmond C., Golding J., Kuh D., Wadsworth M.E. Growth in utero, blood pressure in childhood and adult life, and mortality from cardiovascular disease. BMJ. 1989;298(6673):564–567. doi: 10.1136/bmj.298.6673.564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chatmethakul T., Roghair R.D. Risk of hypertension following perinatal adversity: IUGR and prematurity. J. Endocrinol. 2019;242(1):T21–T32. doi: 10.1530/JOE-18-0687. [DOI] [PMC free article] [PubMed] [Google Scholar]