Abstract
Background
Haemodialysis patients are at risk of developing severe forms of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: coronavirus disease 2019 (COVID-19). In March 2020, hydroxychloroquine (HCQ) and azithromycin (AZI) were proposed as potential treatments of COVID-19, but with warnings concerning their possible toxicity. No data are available regarding the toxicity of this treatment in haemodialysis patients.
Methods
We report the use of HCQ and AZI in a cohort of COVID-19 haemodialysis patients with focus on safety concerns.
Results
Twenty-one patients received 200 mg HCQ thrice daily during 10 days, and AZI 500 mg on Day 1, and 250 mg on the four following days. HCQ plasma concentrations were within the recommended range (0.1–1.0 µg/mL) in all patients except one, in which maximum concentration was 1.1 µg/mL. HCQ concentration raised until the third day and remained stable thereafter. No cardiac event occurred in spite of progressive lengthening of corrected QT interval (QTc) during the treatment. One patient experienced a long QTc syndrome (QTc >500 ms) without any arrhythmia episode, although HCQ concentration was in the target range. Five (23.8%) patients experienced hypoglycaemia, a well-known HCQ side-effect. SARS-CoV-2 RNA remained detectable in nasopharyngeal swabs for a long time in haemodialysis patients (mean time 21 days).
Conclusions
HCQ and AZI are safe in haemodialysis patients at these doses but can lead to long QTc syndrome and hypoglycaemia. HCQ concentrations were not correlated with side effects. We recommend monitoring of the QTc length throughout treatment, as well as glycaemia. SARS-CoV-2 could persist for longer in haemodialysis patients than in the general population.
Keywords: azithromycin, COVID-19, haemodialysis, hydroxychloroquine, SARS-CoV-2
KEY LEARNING POINTS
What is already known about this subject?
• Hydroxychloroquine (HCQ) and azithromycin (AZI) are candidate treatments for coronavirus disease 2019 (COVID-19). The dosage and side effects are well known in the general population, but no data are available in haemodialysis patients. There are no data on HCQ accumulation in haemodialysis patients.
What this study adds?
• HCQ during 10 days associated with AZI during 5 days for COVID-19 treatment is safe in haemodialysis patients. Corrected QT interval (QTc) increases during the treatment, but plasma HCQ concentrations are not correlated with QTc length. Hypoglycaemia is a frequent HCQ side effect in haemodialysis patients.
What impact this may have on practice or policy?
• Electrocardiography QTc monitoring must be performed during HCQ and AZI treatment, especially in the last days of the treatment course. Determination of plasma HCQ concentration is not useful in these patients. Blood glucose should be monitored and antidiabetic drugs reduced because of the risk of hypoglycaemia.
INTRODUCTION
In December 2019, the city of Wuhan in China was the starting point of an outbreak of pneumonia caused by a novel coronavirus later designated as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Despite protection barriers and quarantine of the population, the virus spread through the country and rapidly became pandemic at the world scale [1, 2].
Pulmonary symptoms and fever are the most frequent clinical presentation of the disease, but other symptoms such as headache, diarrhoea, vomiting, anosmia, ageusia and myocarditis, or even asymptomatic patients, have been described. The disease was called coronavirus disease 2019 (COVID-19). On hospital admission, kidney failure was described in 13.1% of patients and proteinuria in 43.9%, but renal pathogenesis of the disease is not well known at this point [3].
Although most patients exert mild forms of pneumonia, 14% of patients show severe presentation and almost 5% are critical with acute respiratory distress syndrome (ARDS) necessitating mechanical ventilation, leading to death in 2.3% of patients in China [2, 4]. Several risk factors for developing severe forms have been recognized: ageing, hypertension, diabetes mellitus, cardiovascular diseases, cancer and chronic kidney disease (CKD) [3, 5].
To date, no specific treatment has been clearly identified, but many molecules tested on the previous Middle East respiratory syndrome coronavirus and SARS-CoV epidemics have been proposed for COVID-19 treatment. Yao et al. showed that remdesivir and hydroxychloroquine (HCQ) inhibit virus replication in vitro [6, 7]. Gao et al. suggested that HCQ may have efficacy in the treatment of COVID-19-associated pneumonia [8].
In March 2020, a French team from Marseille conducted a small trial testing an association of HCQ and azithromycin (AZI) treatment in 24 patients with COVID-19, but describing mild or no symptoms [9]. At Day 6, 75% of treated patients had no SARS-CoV-2 RNA in nasal swabs detected by real-time quantitative polymerase chain reaction (RT-PCR). Authors compared their results with patients with few or no symptoms who did not receive treatment with HCQ-AZI from Avignon and Nice, nearby cities. At Day 6, 90% of these untreated patients were still positive for COVID-19 in the nasal swabs, suggesting an antiviral effect of this treatment. The same team published a larger study with the same trend in favour of the association HCQ-AZI [10]. Other trials were in favour of efficacy of HCQ, but some were not peer-reviewed [11].
Since then, several studies have shown conflicting results. A small randomized controlled trial showed no efficacy of HCQ in the rate of positive RT-PCR in nasal swabs at Day 7 of treatment [12]. Another small prospective trial found no evidence of benefit with HCQ, but this trial included only 11 patients [13]. In a wider randomized prospective study in 150 patients, Tang et al. found no benefit of daily 800 mg HCQ treatment in negative conversion of SARS-CoV-2 at Day 28 compared with standard care [14]. Finally, Geleris et al. in a large observational study in 1446 patients found no benefit of HCQ on COVID-19 clinical outcomes [15].
Several large randomized controlled trials are in progress to assess if this strategy is clinically relevant, but results will not be available for several weeks, and the debate on the benefit of HCQ is still ongoing. Unfortunately, the pandemic in Europe is a fast-growing outbreak with many deaths due to ARDS. Capacities of intensive care units (ICUs) cannot face the needs in some cluster regions, urging a treatment to emerge. In this context, the French government authorized the treatment of COVID-19 by HCQ in March 2020 in specific conditions (in hospital settings, after careful evaluation, collegial medical decision and patient information; www.legifrance.gouv.fr).
Haemodialysis patients are at high risk of ARDS, and many cases were diagnosed day after day in our haemodialysis facility. We thus decided to keep hospitalized all patients diagnosed with COVID-19, and to propose the treatment to these patients in compliance with the health authorities.
HCQ has been used for many years to avoid malaria or for lupus treatment, and AZI is frequently used in pulmonary infection, but these molecules warrant specific concerns in CKD, particularly in haemodialysis patients in which they may accumulate and cause more secondary effects (arrhythmia, myocarditis and retinopathy). Higher HCQ concentrations were observed in systemic lupus erythematosus patients with chronic renal insufficiency patients, but only three of them were dialysed and no impact was reported [16]. No impact was expected on AZI regarding its high molecular weight and hepatic metabolism associated with a biliary excretion. Hypokalaemia, a frequent condition at the end of haemodialysis sessions, might also increase cardiac arrhythmia. As no data are available on the use of this treatment, we describe in this study the use of the association HCQ-AZI in 21 haemodialysis patients, focusing on safety concerns.
MATERIALS AND METHODS
Population
All haemodialysis patients of the Phocean Institute of Nephrology with COVID-19 treated with HCQ and AZI at the Clinique Bouchard, Marseille, France, between 17 March and 10 April 2020 were included in this observational retrospective study. After the first patient was found positive in our centre, all haemodialysis patients were screened by SARS-CoV-2 RNA RT-PCR by nasopharyngeal swab, explaining why some patients were diagnosed without symptoms.
All patients had three conventional haemodialysis sessions per week with a polysulphone membrane (Polyflux 21, Baxter®). Time of sessions was reduced to 3 h (instead of 4 h usually) because of organizational constraints and the time needed for disinfection. All patients were on arteriovenous fistula or a polytetrafluoroethylene graft with a minimal blood flow of 300 mL/min.
COVID-19 diagnosis
Haemodialysis patients were considered as COVID-19 patients as soon as one nasopharyngeal swab tested positive for SARS-CoV-2. Two different RT-PCR assays were used: Bosphore Novel Coronavirus (2019-nCoV) Detection kit (Launchdiagnostics®) and, from 8 April 2020 Abbott RealTime SARS-CoV-2 (Abbott®). RT-PCR was considered as positive if Ct < 35, as recommended by the manufacturer.
PCR for SARS-CoV-2 RNA in nasopharyngeal swabs was performed every 7 days to check if patients were cured of the virus. Patients were considered cured if SARS-CoV-2 RNA was undetectable in nasopharyngeal swab (Ct > 35), but as some patients had again a positive RT-PCR after one negative swab, they were controlled for 48 h after a first negative swab.
Management of COVID-19 haemodialysis patients
All patients diagnosed with COVID-19 were hospitalized in a COVID-19-specific department. Medical and paramedical staff had specific barriers against virus transmission, including protective masks and glasses, waterproof disposable gowns and gloves.
Haemodialysis sessions were also performed in a specific COVID-19 department. The usual parameters preceding infection were maintained for all patients except dialysate potassium concentration, which was set at 3 mmol/L during HCQ treatment to avoid hypokalaemia at the end of the dialysis session. Medications which lengthen corrected QT interval (QTc) were avoided when possible (citalopram, escitalopram, hydroxyzine and domperidone).
All patients were informed of the treatment. Patients received HCQ 200 mg three times daily (TID) for 10 days and AZI 500 mg on the first day and then 250 mg/day on the four following days as proposed by Gautret et al. [9]. HCQ plasma concentration was determined at both the beginning and at the end of each dialysis session during the whole course of treatment for the first patients treated in our centre. HCQ assay was developed using a previously validated liquid chromatography-tandem mass spectrometry method and validated according to the European Guidelines on Bioanalytical Method Validation [17]. However, as all observed plasma concentrations achieved rapidly the recommended range, considering the cost–effectiveness of the therapeutic drug monitoring, we decided to stop after the 13 first patients. Therapeutic drug monitoring results were obtained 24 h after blood sampling and the recommended range recommended was 0.1–1.0 µg/mL [Recommendations for Therapeutic Drug Monitoring of Lopinavir/ritonavir and HCQ in Patients Treated for SARS-CoV-2 (COVID-19) Infection, available on the website: sfpt-fr.org].
Electrocardiogram (ECG) analysis with monitoring of QTc was performed daily by a cardiologist. Correction of QT to calculate QTc was performed as recommended using Bazett formula and a QTc >500 ms was considered as requiring treatment interruption [18–20]. Delta QTc was the difference between the first measured QTc before treatment or on the first day of treatment and the daily QTc.
Data collection
Clinical, biological and haemodialysis data were retrospectively obtained from medical files. Patients had a whole blood test at each haemodialysis session (thrice weekly) including white blood cell (WBC) count, haemoglobin, platelets, potassium, C-reactive protein and procalcitonin, ferritin, hepatic enzymes and ultrasensitive troponin T. HCQ plasma concentrations, ECG and QTc length were collected as well.
Statistical analysis
A descriptive analysis was carried out on socio-demographic, clinical, biological and para-clinical bases to summarize the population data. Results are shown as their means ± standard deviation (SD) or percentages. Linear regression was performed to compare quantitative values. P-value was considered as significant if <0.05.
Ethical statement
This study has been registered in the public directory of the French National Institute of Health Data on 30 May 2020. In compliance with the European Regulation n°2016/679, the so-called General Data Protection Regulation, patients were informed, and gave their non-opposition to the use of the data for research.
RESULTS
Study population
Between 17 March and 10 April, among 270 patients of our haemodialysis centres, 24 (8.6%) patients had a positive nasopharyngeal swab for SARS-CoV-2. Among the 24 patients, 3 were excluded from the study: 2 patients died before receiving HCQ and 1 did not receive HCQ because he presented a myocarditis with elevated troponin T level. Baseline characteristics of the 21 patients of our cohort are reported in Table 1.
Table 1.
Baseline characteristics of patients at diagnosis
| Baseline characteristics | Total population (n = 21) |
|---|---|
| Age, years | 68.1 ± 15.7 |
| Male gender (%) | 57.1 |
| Dialysis vintage, months | 65.9 ± 78.7 |
| BMI, kg/m² | 26.2 ± 5.6 |
| Nephropathy, n (%) | |
| Vascular | 6 (28.6) |
| Diabetic | 5 (23.8) |
| Glomerular | 3 (14.3) |
| Interstitial nephritis | 1 (4.8) |
| Myeloma | 2 (9.5) |
| Undetermined | 4 (19.0) |
| Comorbidities, n (%) | |
| Obesity with BMI >30 kg/m² | 3 (14.3) |
| Diabetes mellitus | 9 (42.9) |
| Hypertension | 21 (100) |
| Immunosuppression | 5 (23.8) |
| Pulmonary disease | 5 (23.8) |
| Heart failure | 4 (19.0) |
| HIV infection | 1 (4.8) |
Immunosuppression was defined as active or past chemotherapy for cancer, immunosuppressive treatment for auto-immune or organ transplantation or HIV infection. Data are provided in number (percentages) or mean ± SD.
HIV, Human immunodeficiency virus; BMI, body mass index.
COVID-19 symptoms
COVID-19 symptoms at diagnosis and during follow-up are reported in Table 2. Within our cohort, five (23.8%) asymptomatic patients were diagnosed during screening, but four of them developed COVID-19 symptoms during the following days. Only one patient remained asymptomatic during the whole follow-up. Clinical presentation of COVID-19 in our patients did not differ from previously described cohorts [21]. Fever >38°C occurred in 13 (61.9%) patients and 12 (57.1%) had dyspnoea that required oxygen, but the most frequent symptom was cough, which occurred in 17 (81%) of the COVID-19 patients.
Table 2.
Clinical and biological COVID-19 symptoms at admission and during follow-up
| COVID-19 symptoms | At diagnosis | During follow-up |
|---|---|---|
| (n = 21) | (n = 21) | |
| Clinical symptoms, n (%) | ||
| No symptoms | 5 (23.8) | 1 (4.8) |
| Fever >38° | 9 (42.9) | 13 (61.9) |
| Cough | 13 (61.9) | 17 (81) |
| Dyspnoea requiring oxygen | 9 (42.9) | 12 (57.1) |
| Sore throat | 5 (23.8) | 8 (38.1) |
| Confusion | 0 (0) | 1 (4.8) |
| Headache | 3 (14.3) | 3 (14.3) |
| Myalgia | 3 (14.3) | 5 (23.8) |
| Diarrhoea | 3 (14.3) | 3 (14.3) |
| Nausea or vomiting | 1 (4.8) | 4 (19.0) |
| Anosmia or ageusia | 0 (0) | 2 (9.5) |
| Biological parameters, n (%) | ||
| Leucopaenia (WBC <4 g/L) | 14 (66.7) | 17 (81.0) |
| Anaemia (haemoglobin <100 g/L) | 5 (23.8) | 17 (81.0) |
| Lymphopaenia (lymphocytes <1.0 g/L) | 13 (61.9) | 17 (81.0) |
| Thrombopaenia (platelets <150 g/L) | 13 (61.9) | 14 (66.7) |
| Ferritin >800 ng/mL | 11 (55) | 16 (80.0) |
| C-reactive protein >10 mg/L | 18 (85.7) | 19 (90.5) |
| Procalcitonin >0.25 ng/mL | 18 (90.0) | 20 (95.2) |
| Abnormal hepatic enzymes (ASAT or ALAT >2× NV) | 1 (4.8) | 3 (14.3) |
| Myocarditis (troponin T >50 pg/mL) | 15 (71.4) | 18 (85.7) |
| Number of days with need for oxygen, mean ± SD | – | 4.0 ± 4.9 |
| Number of days with fever ≥38°C, mean ± SD | – | 1.5 ± 2.0 |
ASAT, aspartate aminotransferase; ALAT, alanine aminotransferase; NV, normal value.
Biological parameters are also reported in Table 2. They were also similar to what has been previously described in the literature. High ferritin levels in these patients have already been reported elsewhere [22]. Troponin T levels in our patients were high at diagnosis in 15 (71.4%) of patients, with mean concentration of 104 ± 98 pg/mL, but only three patients had levels >100 pg/mL corresponding to myocarditis.
HCQ plasma concentrations
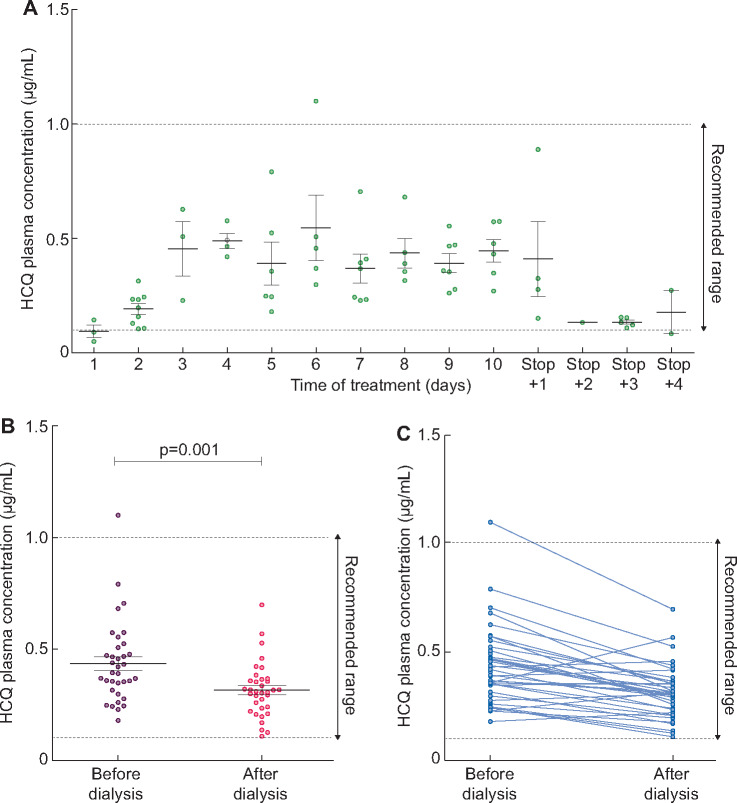
The first 13 COVID-19 patients had plasma HCQ concentration determination before and after each haemodialysis session of treatment and 10 of them were also monitored a few days after stopping the treatment. As described in Figure 1A, HCQ concentration increased during the first 3 days and then remained stable during the seven following days of treatment. At the end of treatment, HCQ concentrations reduced drastically within 48 h.
FIGURE 1.
HCQ plasma concentrations in COVID-19 haemodialysis patients. (A) Daily HCQ plasma concentrations during and after treatment. Recommended range between 0.1 and 1.0 µg/mL. Data are provided as mean ± SD. (B) HCQ concentrations before (purple) and after (pink) haemodialysis session from Days 3 to 10 of treatment. Data are provided as mean ± SD. (C) HCQ concentrations before and after each haemodialysis session from Days 3 to 10 of treatment.
HCQ concentrations achieved the recommended range of 0.1–1 µg/mL in the majority of cases. Only one patient had once a high HCQ concentration at 1.1 µg/mL, but was once again within the recommended range 2 days later (performed before the treatment was modified because of the 48-h delay of laboratory results).
Mean pre-dialysis and post-dialysis HCQ concentration between Days 3 and 10 of treatment [time during which the HCQ concentrations remained relatively stable in our cohort (Figure 1A)] were, respectively, 0.43 [95% confidence interval (CI) 0.37–0.49] µg/mL and 0.32 (95% CI 0.28–0.36) µg/mL (Figure 1B and C). HCQ concentration was reduced to 0.12 (95% CI 0.08–0.16) µg/mL during haemodialysis sessions. Mean post-/pre-haemodialysis HCQ concentration ratio was 0.76 (95% CI 0.67–0.85). HCQ plasma concentrations were not influenced by age, gender or body mass index, but the small number of patients in our cohort should be considered (data not shown).
In one patient already receiving HCQ at 200 mg twice daily for several months for a lupus, the dose was raised to 200 mg TID for the 10 days after COVID-19 was diagnosed. This patient displayed similar plasma concentrations as the others.
Cardiac tolerance
A mean of 8.5 daily ECGs per patient was available for analysis. As reported in Figure 2, QTc raised slowly during the treatment and was the longest in the late days of treatment. QTc was not correlated with HCQ concentration (r = 0.01; P = 0.89), nor was the delta QTc (r = 0.002; P = 0.89).
FIGURE 2.
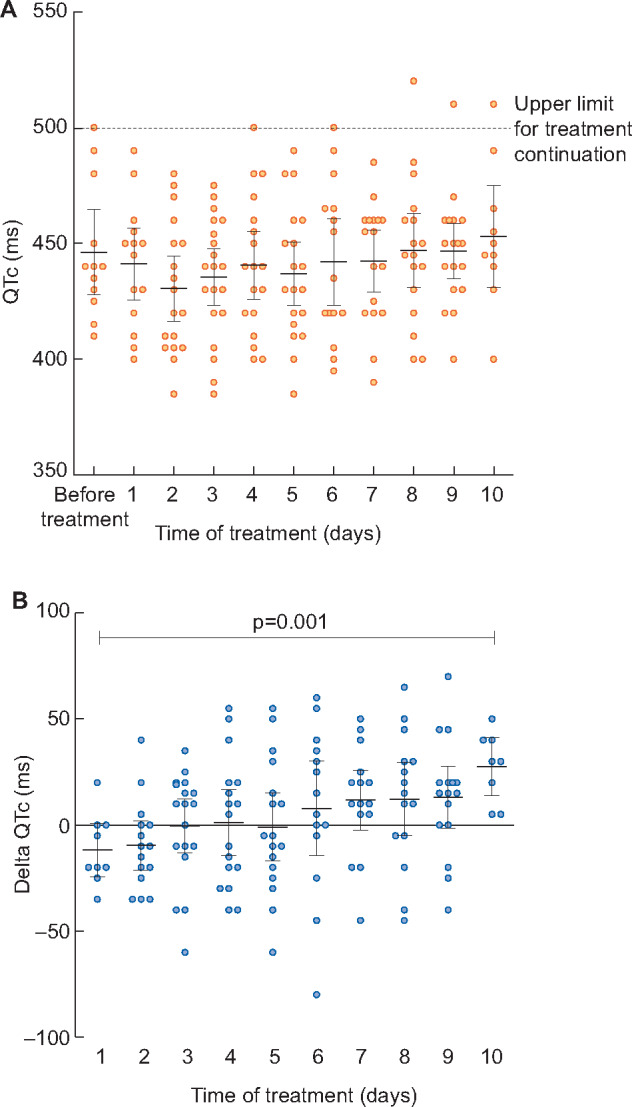
QTc interval in COVID-19 haemodialysis patients treated with HCQ and AZI. A. Daily QTc during treatment (mean and 95% CI). The long QTc syndrome is defined by a QTc >500 ms and requires treatment interruption because of the risk for ventricular arrhythmia. In part (A) the QTc >500 ms observed on Days 8–10 were in the same patient. (B) Daily changes in QTc during treatment (mean and 95% CI).
Only one patient had a QTc >500 ms, which is the upper tolerated limit requiring treatment stop, but he did not exert any cardiac rhythm disturbance, or conduction trouble. Plasma concentration was within the recommended range (0.55 µg/mL) in this patient, pre-dialysis potassium concentration at that time was between 4.2 and 4.8 mmol/L and magnesium was in the normal range.
Other side effects
Side effects of treatment are reported in Table 3. Hypoglycaemia occurred in five patients with diabetes mellitus. Within them, one had no treatment (neither insulin nor oral antidiabetic treatment), involving a specific role for HCQ. Four patients had nausea or vomiting during the treatment but it is not clear if these symptoms are attributable to treatment or COVID-19. No patient complained of visual impairment, and we did not conduct an examination by an ophthalmologist.
Table 3.
Side effects of therapy
| Side effects | Total population |
|---|---|
| (n = 21), n (%) | |
| Nausea or vomiting | 4 (19) |
| Allergy or dermatitis | 0 (0) |
| Visual impairment | 0 (0) |
| Cardiac | |
| Long QTc syndrome (>500 ms) | 1 (4.8) |
| Cardiac rhythm disturbances | 0 (0) |
| Hypoglycaemia | |
| Insulin treated | 3 (14.3) |
| Oral antidiabetic | 1 (4.8) |
| No treatment | 1 (4.8) |
| Other | 0 (0) |
Outcomes
The study was not designed to estimate the efficacy of the treatment. Considering the 24 COVID-19 patients of our centre, 4 (16.7%) died, 1 (4.2%) needed mechanical ventilation in ICU and then recovered, and the 19 (79.2%) others had favourable outcome without mechanical ventilation.
All deceased patients were old (mean age 84.6 ± 2.8 years), had severe respiratory symptoms of COVID-19, and, according to age and comorbidities, were not transferred in our ICU. Mean delay between diagnosis and death was short. One patient had received no HCQ treatment, while the others had received, respectively, 2, 4 and 5 days of treatment before death.
The patient who was transferred in the ICU was aged 51 years old. He had received 48 h of HCQ before he was put on mechanical ventilation, but treatment was continued in ICU and he recovered.
Among the 18 patients for whom the regular follow-up of RT-PCR was available, 3 still had positive nasopharyngeal swabs at the end of the study (respectively, on Days 21, 37 and 39). Fifteen patients became RT-PCR negative at a mean delay of 21.1 ± 8.5 days. Within them, one was negative at Day 7 but was positive again at Day 20, and finally negative on Day 23.
DISCUSSION
Our study is the first reporting tolerance of treatment with AZI and HCQ prescribed for COVID-19 treatment in haemodialysis patients.
Haemodialysis patients are at high risk of COVID-19: patients meet each other three times a week during several hours, need a medicated shuttle each time and often caregivers nursing at home [23]. Scarpioni et al. described that 16% of their centre had COVID-19 [24]. At the time of this study, 24 (8.8%) out of 270 haemodialysis patients had COVID-19, but the epidemic is still ongoing.
As haemodialysis patients are often aged and comorbid individuals, the COVID-19-associated rate of mortality is high: some units reported a mortality rate up to 41% [24]. In France, on 20 April 2020, the REIN registry described 209 (19.2%) deaths in 1089 patients (https://www.agence-biomedecine.fr/IMG/pdf/bulletin_no4_version1.pdf). Treatments with HCQ and AZI were provided to our patients to try to reduce these very high mortality rates, as these molecules were proposed as a treatment for COVID-19 by experts [9].
Our main objective was to describe the safety of this treatment, especially the cardiac tolerance in haemodialysis patients with potassium homoeostasis disturbances and cardiac abnormalities. AZI and HCQ are known to elongate QTc. Chorin et al. have reported recently an 11% rate of QTc >500 ms (long QTc syndrome) after treatment with HCQ 200 mg twice daily and AZI [20]. In their cohort of 84 non-CKD patients, the maximal average QTc occurred at 3.6 days of treatment, which is the time at which the HCQ concentrations became relatively stable in our cohort (Figure 1A). In our population, QTc increased during the whole duration of treatment. Although it is not possible to differentiate the respective role of each of the drugs, this may be more in favour of HCQ being the culprit, as AZI was only prescribed for 5 days.
As in the study from Chorin et al., we performed a daily ECG monitoring, and had no cardiac arrhythmia event. Although HCQ dose was higher in our patients (200 mg TID), we only noticed one long QTc syndrome, which occurred at the seventh day of treatment, raising concerns about tissue accumulation. This effect was not associated with an elevated HCQ concentration and the patient had no hypokalaemia.
The lack of correlation between plasma HCQ concentrations and QTc in our population could be explained by an accumulation of the drug in the whole blood or tissue compartments, which might explain the increase in QTc length observed during the treatment, but we did not determine HCQ concentration in these compartments. A role for AZI in QTc elongation could also be hypothesized.
HCQ plasma concentrations were determined in only 13 out of 21 patients, which is a limit of our study. Since almost no patient had high HCQ plasma concentration and these concentrations were not correlated with QTc length, we believe that HCQ plasma concentration monitoring is not necessary to prevent cardiac toxicity in this population. We rather recommend ECG and potassium monitoring, throughout treatment.
Five patients experienced hypoglycaemia during the treatment. Because of COVID-19, many patients had mild anorexia, ate less and lost weight, which led to lessening of their dry weight during haemodialysis. Poor intakes might have favoured hypoglycaemia, but the occurrence of hypoglycaemia in a patient with no antidiabetic treatment favours a side effect of HCQ. This has already been reported in the diabetic population and even in peritoneal dialysis [25, 26]. HCQ improves insulin sensitivity and reduces insulin degradation [27]. Thus, blood glucose level should be monitored during treatment and anti-diabetic treatment dosing might need to be reduced during the treatment. No patients had a visual complaint, which was not surprising given the very short course of treatment: visual impairment has been described after treatment of several months.
We did not determine HCQ metabolites concentrations, which might accumulate in haemodialysis patients; so far, no HCQ metabolites have been clearly identified and considered as more accurate in order to evaluate the efficacy or toxicity of the drug [28].
Our study was not designed to analyse the efficiency of HCQ and AZI in COVID-19 and we did not have any control group. The effect of HCQ-AZI is based on its ability to avoid membrane–cell fusion, therefore avoiding the entry of the virus into the cell [29]. One can hypothesize that the best effect of the treatment would be obtained if given early after diagnosis. We cannot conclude on the efficacy of HCQ-AZI in this situation, but given these crude data, the treatment could be more efficient if started early. Interestingly, the long delay between diagnosis and COVID-19 cure is in contrast to the substantially shorter delay of 6 days previously described [9] . This could be related to the different type of population (haemodialysis versus general population) and the immunocompromised status of haemodialysis patients. Haemodialysis patients should thus remain in quarantine longer than the general population.
In conclusion, HCQ 200 mg TID during 10 days associated with AZI during 5 days for COVID-19 treatment in haemodialysis patients was safe. We recommend electrocardiography monitoring because QTc increases during the treatment. Blood glucose should also be monitored because of the risk of hypoglycaemia. HCQ plasma monitoring seems not to be useful since side effects were not associated with HCQ plasma concentrations and HCQ remained within the recommended range.
AUTHORS’ CONTRIBUTIONS
P.G. designed the study, collected data and wrote the paper. M.G. carried out ECG analysis. N.P. collected data, and was responsible for regulatory and ethics compliance. H.N. was involved in hypoglycaemia data analysis and interpretation. J.-P.B, C.S. and R.G. made dosages, helped with interpretation of the biological data and helped to write the manuscript. J.S., J.O., H.S., M.S. and F.B. were involved in data collection and helped to design the study. S.B. designed the study, collected data and wrote the article.
CONFLICT OF INTEREST STATEMENT
The authors declare they have no conflict of interest regarding this study.
REFERENCES
- 1. Phelan AL, Katz R, Gostin LO.. The novel coronavirus originating in Wuhan, China: challenges for global health governance. JAMA 2020; 323: 709. [DOI] [PubMed] [Google Scholar]
- 2. Chan J, Ng C, Chan Y. et al. Short term outcome and risk factors for adverse clinical outcomes in adults with severe acute respiratory syndrome (SARS). Thorax Août 2003; 58: 686–689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Cheng Y, Luo R, Wang K. et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int 2020; 97: 829–838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Wu Z, McGoogan JM.. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese center for disease control and prevention. JAMA 2020. [cited 28 March 2020]; available at: https://jamanetwork.com/journals/jama/fullarticle/2762130 [DOI] [PubMed] [Google Scholar]
- 5. Shi Y, Yu X, Zhao H. et al. Host susceptibility to severe COVID-19 and establishment of a host risk score: findings of 487 cases outside Wuhan. Crit Care 2020; 24: 108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Yao X, Ye F, Zhang M. et al. In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis Publ Infect Dis Soc Am 2020; doi: 10.1093/cid/ciaa237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Wang M, Cao R, Zhang L. et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res 2020; 30: 269–271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Gao J, Tian Z, Yang X.. Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends 2020; 14: 72–73 [DOI] [PubMed] [Google Scholar]
- 9. Gautret P, Lagier JC, Parola P. et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents 2020; 105949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Gautret P, Lagier JC, Parola P. et al. Clinical and microbiological effect of a combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six-day follow up: a pilot observational study. Travel Med Infect Dis 2020; 34: 101663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Chen Z, Hu J, Zhang Z. et al. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. medRxiv 2020; Doi: 10.1101/2020.03.22.20040758 [DOI]
- 12. Chen J, Liu D, Liu L. et al. A pilot study of hydroxychloroquine in treatment of patients with moderate COVID-19. Zhejiang Xue Xue Bao Yi Xue Ban. J Zhejiang Univ Med Sci 2020; 49: 215–219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Molina JM, Delaugerre C, Le Goff J. et al. No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection. Med Mal Infect 2020; 50: 384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Tang W, Cao Z, Han M. et al. Hydroxychloroquine in patients with mainly mild to moderate coronavirus disease 2019: open label, randomised controlled trial. BMJ 14 2020; 369: m1849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Geleris J, Sun Y, Platt J. et al. Observational study of hydroxychloroquine in hospitalized patients with Covid-19. N Engl J Med 2020; 382: 2411–2418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Jallouli M, Galicier L, Zahr N. et al. ; the Plaquenil Lupus Systemic Study Group. Determinants of hydroxychloroquine blood concentration variations in systemic lupus erythematosus. Arthritis Rheumatol 2015; 67: 2176–2184 [DOI] [PubMed] [Google Scholar]
- 17. Chhonker YS, Sleightholm RL, Li J. et al. Simultaneous quantitation of hydroxychloroquine and its metabolites in mouse blood and tissues using LC-ESI-MS/MS: an application for pharmacokinetic studies. J Chromatogr B Analyt Technol Biomed Life Sci 2018; 1072: 320–327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Postema PG, De Jong J, Van der Bilt IAC. et al. Accurate electrocardiographic assessment of the QT interval: teach the tangent. Heart Rhythm 2008; 5: 1015–1018 [DOI] [PubMed] [Google Scholar]
- 19. Mason JW, Ramseth DJ, Chanter DO. et al. Electrocardiographic reference ranges derived from 79,743 ambulatory subjects. J Electrocardiol 2007; 40: 228–234 [DOI] [PubMed] [Google Scholar]
- 20. Chorin E, Dai M, Shulman E. et al. The QT interval in patients with COVID-19 treated with hydroxychloroquine and azithromycin. Nat Med 2020; 26: 808–809 [DOI] [PubMed] [Google Scholar]
- 21. Grasselli G, Zangrillo A, Zanella A. et al. ; for the COVID-19 Lombardy ICU Network. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy region. JAMA 2020; 323: 1574 [ 10.1001/jama.2020.5394] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Bataille S, Pedinielli N, Bergounioux JP.. Could ferritin help the screening for COVID-19 in hemodialysis patients? Kidney Int 2020; 98: 235–236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Su K, Ma Y, Wang Y. et al. How we mitigate and contain COVID-19 outbreak in hemodialysis center (HD): lessons and experiences. Infect Control Hosp Epidemiol 2020;1–6; doi: 10.1017/ice.2020.161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Scarpioni R, Manini A, Valsania T. et al. Covid-19 and its impact on nephropathic patients: the experience at Ospedale Guglielmo da Saliceto in Piacenza. G Ital Nefrol 2020; 37: 2020-vol2 [PubMed] [Google Scholar]
- 25. El-Solia A, Al-Otaibi K, Ai-Hwiesh AK.. Hydroxychloroquine-induced hypoglycaemia in non-diabetic renal patient on peritoneal dialysis. BMJ Case Rep 2018; 2018: bcr2017223639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Shojania K, Koehler BE, Elliott T.. Hypoglycemia induced by hydroxychloroquine in a type II diabetic treated for polyarthritis. J Rheumatol 1999; 26: 195–196 [PubMed] [Google Scholar]
- 27. Winter EM, Schrander-van der Meer A, Eustatia-Rutten C. et al. Hydroxychloroquine as a glucose lowering drug. BMJ Case Rep 2011; 2011: bcr0620114393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Masson E. P58: monitoring of hydroxychloroquine and its metabolites after a massive ingestion: are there contributive to poisoned patient management in the intensive care unit? [Internet]. EM-Consulte [cited 24 April 2020]. https://www.em-consulte.com/article/905860/article/p58-monitoring-of-hydroxychloroquine-and-its-metab (31 April 2020, date last accessed)
- 29. Sanders JM, Monogue ML, Jodlowski TZ. et al. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA 2020; doi: 10.1001/jama.2020.6019 [DOI] [PubMed] [Google Scholar]