Abstract
The optimal duration of prophylactic antimicrobial usage in clean-contaminated elective oncological surgeries is not clear. This single-center randomized trial evaluated the effectiveness of single-dose antimicrobial prophylaxis in clean-contaminated surgeries for the reduction of surgical site infection (SSI). Between April 2018 and January 2019, 315 patients undergoing major oncological clean-contaminated surgeries where the gastrointestinal or genital tract was opened under controlled conditions were randomized into 2 groups i.e., single dose versus extended dose groups. The single dose group received a 1.5 g dose of cefuroxime immediately before surgery while the extended group received the same dose of cefuroxime thrice daily for 4 days from the day of surgery till postoperative day 3. In addition, patients undergoing esophageal and colorectal surgeries received metronidazole. The overall SSI rate of the single dose group was not significantly different from that of the extended group (11.3% vs. 14.7%, respectively, p 0.40), with absolute difference of 3.4% and relative risk of 0.85 (95% C.I, 0.59 to 1.22). The rate of remote site infection was also not different between the two groups (14.4% vs 10.2%, p 0.31) with absolute difference of 4.2% and relative risk 1.19 (95% C.I, 0.89 to 1.59). In univariate analysis, parameters like nodal dissection, colorectal surgery, smoking, and hospital stay were significantly associated with SSI. In multivariate analysis, age, smoking, nodal dissection, and hospital stay retained significance. Single-dose antimicrobial prophylaxis is as effective as extended usage for 4 days in the prevention of postoperative SSI in patients undergoing clean-contaminated major oncological surgeries. Trial was registered with the clinical trial registry of India (CTRI/2018/06/014344).
Keywords: Surgical site infection, Prophylaxis, Antibiotic, Perioperative, Surgery
Introduction
Surgical site infections (SSIs) contribute to about 20% of nosocomial infection and remain a substantial burden in terms of morbidity, prolonged hospitalization, and death [1, 2]. The SSIs after clean-contaminated oncological surgeries vary from 5 to 27% in various studies [2–6]. In clean-contaminated surgeries, the respiratory, alimentary, genital, or urinary tracts are breached under controlled conditions with no undue contamination [7]. Antibiotic prophylaxis can effectively reduce SSI in clean and contaminated surgery, but the optimal period of prophylaxis remains unclear. Present guidelines advocate single-dose first-generation or second-generation cephalosporins for SSI prophylaxis in clean-contaminated surgeries [8]. Though the trials have shown the effectiveness of single-dose antibiotic prophylaxis, extending antibiotic usage beyond 3–4 days remains a routine practice [9, 10]. Single-dose prophylaxis is a cheaper and cost-effective option than extended regimens, decreases bacterial resistance, and lowers the risk of antibiotic-related drug complications. Randomized trials have evaluated the role of single-dose versus extended antimicrobial prophylaxis in colorectal, gastric, hepatobiliary, and gynecological oncological surgeries, but none have evaluated taking the clean-contaminated oncological group as a single cohort. That is why the present study evaluates this group. This study is an open-labeled randomized clinical trial, evaluating the role of single-dose antimicrobial prophylaxis versus extended usage for 4 days in clean-contaminated oncological surgeries, i.e., gastrointestinal and gynecological surgeries.
Material and Methods
The study was conducted by the departments of Surgical Oncology, Anaesthesiology and Microbiology at Basavatarakam Indo American Cancer Hospital and Research Institute, Hyderabad, India, from April 2018 till January 2019. The study was an open-labeled randomized controlled trial, evaluating the role of single-dose prophylactic antibiotic versus extended usage for 4 days for the prevention of surgical site infection in clean-contaminated oncological surgeries, i.e., gastrointestinal and gynecological surgeries. Of 333 patients who were assessed for eligibility, 315 patients with gastrointestinal and gynecological malignancies undergoing surgeries with curative intent were randomly allocated in two groups, single dose or extended dose groups. The single dose group received a 1.5 g dose of cefuroxime immediately before surgery while the extended group received the same dose cefuroxime three times a day for 4 days from the day of surgery till postoperative day 3. After induction, 1.5 g of cefuroxime was given intravenously. An extra dose was considered when the duration of surgery was more than 3 hours. Patients undergoing colonic and esophageal surgeries additionally received metronidazole intravenously for the duration of antibiotic prophylaxis course considering the high anaerobic bacterial load at both these sites. All patients undergoing colorectal and major gynecological surgeries where bowel resection was anticipated underwent mechanical bowel preparation a day before surgery with polyethylene glycol preparation (PEGLEC, Til Health Care (India) Limited, Chennai, India).
Inclusion criteria included patients with clean-contaminated oncological gastrointestinal and gynecological surgeries done with a curative intent where gastrointestinal or genital tract was opened under controlled conditions, who gave written informed consent and were between the ages of 15 and 75 years. Exclusion criteria included the patients who refused to consent for the study, who were less than 15 years or ≥ 76 years, those with cephalosporins allergy, any infections in precedings 2 weeks, whose blood leucocyte count is less than 3000 or more than 12,000 cells/mm3, clean surgeries, and cases in which intraoperatively gross contamination with uncontrolled spill from gastrointestinal tract has occurred. The patients who developed an infection after the surgery were treated as per the standard hospital protocol. Urological surgeries, palliative procedures, emergency surgeries, and stoma reversals were excluded. The primary intent of the study was the measurement of surgical site infection. The secondary outcome was to analyze remote site infections. The ethics committee of the institute approved the study. The study was registered with the central trial registry of India (CTRI/2018/06/014344).
Standard procedures as per Centers for Disease Control (CDC) guidelines were followed in all patients [11]. Patients were advised for full-body bath with soap and 10% betadine scrub solution a night before surgery. The skin was prepared in the operation theater using an alcohol-based chlorhexidine solution. Plastic adhesive drapes to cover skin were not used. Nonabsorbable synthetic suture nylon was used to approximate fascia. Intra-abdominal drains were taken out via a separate stab incision discrete from the primary wound. Stainless steel staples or nylon sutures were used to approximate skin and then wound was mopped by normal saline. Intraoperative irrigation of wound was done with dilute betadine solution, but intraperitoneal lavage with an antiseptic solution was not done. Normothermia and euglycemia were strictly maintained perioperatively. A suction drain was kept in the wound only if the patient was obese. Intra-abdominal drains were taken out usually after 2 to 3 days or when the output was less than 100 ml and serous, and the staples after 7 days. The wound was assessed daily by the surgical team until the patient was discharged and at every visit in the outpatient clinics till 30 days post-surgery. The detection and diagnosis of SSIs (incision site infection and organ/space infection) were done according to the CDC definitions [7]. Anastomotic site disruption, as well as other organs/space infection, was established by clinical assessment with or without imaging guidance. Remote site infections are the infections that occurred at a location different from the surgical site, such as urinary tract infection, pneumonia, or bloodstream (catheter-related) infection [12].
Various parameters determining the risk of SSIs, like age, sex, antibiotic dose (single versus extended), American Society of Anesthesiologists–Physical Status (ASA-PS) score, body mass index (BMI), serum albumin levels, hemoglobin levels (<10 or > 10 g %), diabetes mellitus, duration of operation, blood loss, site of tumor, lymphadenectomy, surgical approach (open versus laparoscopic-assisted versus laparoscopic), and combined excision of other organ(s), surgical site infection, organ space infection, anastomosis disruption, remote site infection, and mortality rate were assessed using univariate logistic regression.
Statistics
The prime objective was to assess the effectiveness of single-dose antibiotic prophylaxis in an overall reduction of SSIs in comparison to extended usage. The trial was conceived to detect a 10% difference in the incidence of SSIs between the two groups, with 95% confidence interval (CI) and 80% power; assuming a 9% incidence of SSIs in the extended usage group based on earlier studies on the SSIs after clean-contaminated surgeries [2–5, 8, 9, 13–16]. Thereby, a sample size of 140 was required in both treatment arms (total 280). Considering a 10% attrition rate, 315 patients were randomized.
Continuous data were depicted as median and interquartile range or 95% CI. Statistical analyses were executed by GraphPad Prism 5 for Windows (GraphPad Software, La Jolla, CA, USA). The Fisher exact test was utilized to analyze categorical data. Categorical data from more than two groups were analyzed by the chi-square test. Mann-Whitney U test was utilized for continuous data. A p value of 0.05 or lesser was taken as statistically significant. Univariate and multivariate statistical analyses of various risk factors determining SSI were done by logistic regression model by the software Statistical Package for the Social Sciences (SPSS) 20th version for Windows (SPSS Inc., Chicago, IL, USA).
Randomization
Randomization was done after the surgery when the inclusion and exclusion criteria were met. Patients were allocated randomly to one of the two groups in a 1:1 enrolment ratio to either a single dose or extended dose antibiotics group by sequential, random numbers generated in blocks of 20 from the website www.random.org.
Results
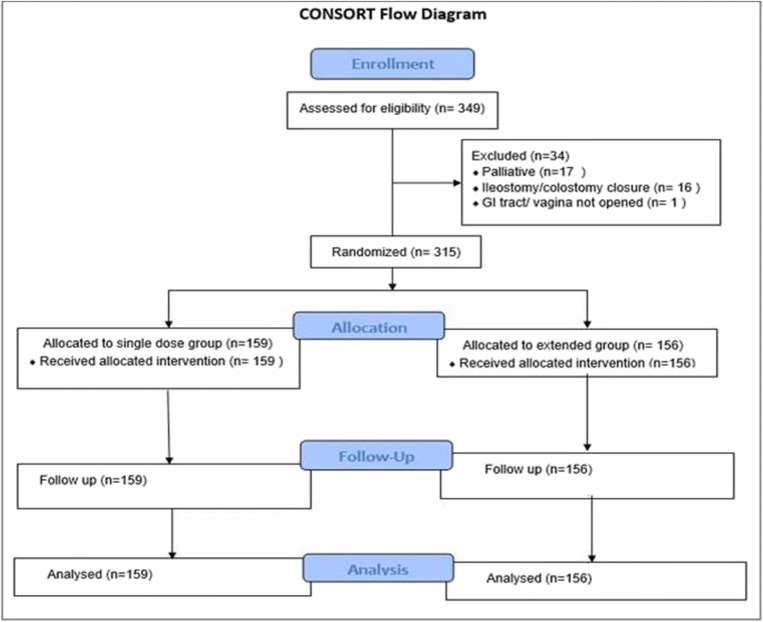
Among 333 patients who were assessed for eligibility, 18 patients were excluded as 17 patients underwent palliative procedure and in one patient, surgery was clean. In total, 315 patients were allocated randomly to get either single dose of cefuroxime (single dose group) or extended group (Fig. 1). All patients were followed till 1 month postoperatively. No patient was lost to follow-up.
Fig. 1.
CONSORT flow diagram
The two groups were identical with regards to age, gender, ASA-PS, BMI, serum albumin, hemoglobin, and prevalence of comorbidities (Table 1).
Table 1.
Baseline and demographic parameters
| Variable (expressed as median and range) | Duration of antimicrobial prophylaxis | P value | |
|---|---|---|---|
| Single dose group | Extended dose group | ||
| Number | 159 | 156 | |
| Age in years | 55 (19–83) | 52 (19–79) | 0.311 |
| Sex (male/female) | 49/110 | 45/111 | 0.7110 |
| BMI (kg/m2) | 24 (13–49) | 25 (13–44) | 0.3300 |
| ASA | 0.0978 | ||
| 1 | 63 | 52 | |
| 2 | 86 | 100 | |
| 3 | 10 | 4 | |
| 4 | 0 | 0 | |
| ASA 1:2 vs 3 | 63/96 | 52/104 | 0.2922 |
| Serum albumin (g %) | 4 (2.8–5.1) | 4.1 (2.1–4.9) | 0.1325 |
| Hemoglobin (g %) | 11.2 (8.1–15.1) | 11.7 (6.8–15.9) | 0.3034 |
| Comorbidities (no) | 74/159 | 84/156 | 0.2158 |
| Diabetes mellitus (no) | 37/159 | 37/156 | 1 |
| Smoking (no) | 10/159 | 9/156 | 1 |
| Operative approach | 0.0006 | ||
| Open | 45 | 75 | |
| Laparoscopic assisted | 70 | 56 | |
| Laparoscopic | 44 | 25 | |
| Nodal dissection (no) | 125 | 111 | 0.1527 |
| Combined organ resection (no) | 10/159 | 29/156 | 0.001 |
| Tumor site (no) | 0.0885 | ||
| Esophagus | 18 | 17 | |
| Stomach | 20 | 12 | |
| Rectum | 19 | 24 | |
| Colon | 34 | 28 | |
| Ovary | 26 | 40 | |
| Endometrium | 36 | 23 | |
| Cervix | 6 | 12 | |
| Operating time (min) | 270 (65–565) | 245 (90–630) | 0.8191 |
| Blood loss (ml) | 150 (50–800) | 150 (40–2000) | 0.7516 |
| Morbidity | 36/159 (22.6%) | 31/156 (19.8%) | 0.5835 |
| Mortality (no) | 0 | 1 | 0.4952 |
| Hospital stay in days | 7 (2–22) | 7 (3–25) | 0.6121 |
| Operative time | 0.5452 | ||
| <3 hours | 24 | 28 | |
| >3 hours | 135 | 128 | |
| Operative blood loss | 0.2605 | ||
| <200 ml | 140 | 129 | |
| >200 ml | 19 | 27 | |
no number of patients
The two groups were also alike in relation to smoking history, lymph node dissection, blood loss, operating time, and site of the tumor. Also, no significant differences were found with regard to the hospital stay (p 0.61) or mortality (p 0.49). The extended dose group had more patients who underwent open surgeries, whereas single dose group had more patients who had laparoscopic-assisted surgery (p 0.0006). The frequency of combined resection of other organs was also higher in the extended group (p 0.001) (Table 1).
The incidences of incisional SSI were 6.9% (11/159) in single dose group, 10.2% (16/156) in extended dose group which were not different (p 0.32), with relative risk of 0.79 (95% C.I. 0.49 to 1.26). Similarly, the incidences of organ/space SSI without anastomotic site dehiscence were 2% (3/159) and 1.2% (2/156) in single and extended dose groups respectively (Table 2). The incidences of anastomotic site dehiscence were 3.7% (6/159) and 4% (6/156) respectively in single and extended dose groups. Infections involving the superficial and deep incisional sites in contiguity were counted as deep incisional SSI. All SSIs secondary to anastomotic site dehiscence were taken as organ/space infection under anastomotic site dehiscence category. Those SSIs that were separate and not secondary to anastomotic site dehiscence or deep space/organ infections were counted as two separate events. Two patients who underwent low anterior resection for carcinoma rectosigmoid in the single-dose antibiotic group had anastomotic site dehiscence detected via feculent discharge through the drain with simultaneous surgical site infection of the abdominal wound. These were taken as two separate SSIs. A patient with carcinoma rectum in extended group underwent low anterior resection with inguinal nodal dissection had anastomotic site dehiscence and surgical site infection of the left inguinal area. These were counted as two separate events.
Table 2.
Comparisons of SSIs between the two groups
| Single dose group | Extended dose | P value | RR | 95% C.I. | |
|---|---|---|---|---|---|
| Incisional site infection | 11 (6.9%) | 16 (10.2%) | 0.32 | 0.79 | 0.49 to 1.26 |
| Organ/space infection | |||||
| 1. Without anastomotic dehiscence | 3 (2%) | 2 (1.2%) | 1.0 | 1.19 | 0.57 to 2.46 |
| 2. Anastomotic site dehiscence | 6 (3.7%) € | 6 (4%) ¥ | 1.0 | 0.99 | 0.56 to 1.76 |
| Total SSI (incisional + organ/space infection) ¶ | 18/159 (11.3%) € | 23/156 (14.7%) ¥ | 0.40 | 0.85 | 0.59 to 1.22 |
| Remote site infection | 23 (14.4%) | 16 (10.2%) | 0.31 | 1.19 | 0.89 to 1.59 |
| Overall infective complications (SSI + remote site) | 37 (23.3%) | 31 (19.87%) | 0.49 | 1.10 | 0.85 to 1.41 |
RR relative risk, C.I. confidence interval, SSI surgical site infection. ¶ Includes the total number of patients with surgical site infections. € Two patients had anastomotic site dehiscence detected via drain with simultaneous SSI of abdominal wound unrelated to anastomotic dehiscence. ¥ One patient who underwent low anterior resection with left inguinal nodal dissection for carcinoma rectum had anastomotic site dehiscence and SSI of the left inguinal area
The cumulative incidence rates of all SSIs (incisional, organ/space infection and anastomotic site dehiscence) were 11.3% (18/159) and 14.7% (23/156) in single dose and extended dose groups respectively and were similar (p 0.4) with an absolute difference of 3.4% and relative risk of 0.85, (95% C.I., 0.59 to 1.22) (Table 2). Also, remote site infection rate incidence was 14.4% (23/159) and 10.2% (16/156) respectively in the single dose and extended dose groups and similar (p 0.31) with an absolute difference of 4.2% and a relative risk of 1.19 (95% C.I., 0.89 to 1.59). Patients with colorectal surgery in the single dose group had an SSI rate of 19%, whereas in the extended group 25% (p 0.48). Overall, SSI rates in colorectal surgery were 21% and 8.57% in non-colorectal surgery (p 0.0013).
In univariate analysis parameters like nodal dissection, colorectal surgery, smoking, and hospital stay were associated significantly with SSI, reaching the proportion of significance level in patients with hemoglobin less than 10 g % (p 0.051) and in those who underwent laparoscopic-assisted surgery (p 0.053) (Table 3). On multivariate logistic regression analysis, age [p 0.042 odd ratio 0.963 (95% C.I, 0.93–1)], nodal dissection [p 0.003 odd ratio 0.083 (95% C.I, 0.016–0.43)], smoking [p 0.021 odd ratio 0.18 (95% C.I, 0.042–0.776)], and hospital stay [p 0.00 odd ratio 1.37 (C.I, 1.22–1.546)] retained their significance.
Table 3.
Univariate logistic regression analysis
| Parameters | P value | Odds ratio | 95% C.I. | Standard error |
|---|---|---|---|---|
| Age | 0.776 | 1.004 | 0.979–1.029 | 0.013 |
| Sex male | 0.070 | 0.559 | 0.298–1.049 | 0.321 |
| Surgery lap assisted | 0.053 | 2.733 | 0.987–7.567 | 0.520 |
| Surgery laparoscopy | 0.094 | 2.400 | 0.861–6.693 | 0.523 |
| Nodal dissection | 0.003 | 0.163 | 0.049–0.540 | 0.611 |
| Organ resected | 0.171 | 0.565 | 0.250–1.279 | 0.417 |
| Neoadjuvant therapy | 0.229 | 0.687 | 0.373–1.266 | 0.312 |
| Colorectal surgery | 0.026 | 0.495 | 0.267–0.919 | 0.316 |
| ASA score 1 | 0.582 | 0.635 | 0.125–3.209 | 0.827 |
| ASA score 2 | 0.643 | 1.440 | 0.309–6.720 | 0.786 |
| BMI < 18.5 kg/m2 | 0.428 | 0.634 | 0.206–1.955 | 0.575 |
| BMI 18.5–24.9 kg/m2 | 0.360 | 1.360 | 0.711–2.558 | 0.327 |
| Albumin <4 g% | 0.409 | 1.294 | 0.702–2.387 | 0.312 |
| Hb <10 g% | 0.051 | 0.381 | 0.145–1.006 | 0.494 |
| DM present | 0.090 | 0.563 | 0.289–1.094 | 0.339 |
| Smoking | 0.012 | 0.283 | 0.106–0.761 | 0.504 |
| Blood loss | 0.376 | 0.696 | 0.311–1.554 | 0.410 |
| Hospital stay | 0.000 | 1.321 | 1.209–1.443 | 0.278 |
Lap laparoscopy, ASA American Society of Anesthesiologists, BMI body mass index, Hb hemoglobin, DM diabetic mellitus
Discussion
The current study found a single dose of cefuroxime was as efficacious as extended dose course for 4 days for the reduction of surgical site infection in clean-contaminated surgeries. The overall incidence of SSI for clean-contaminated surgeries was 13%, in this study similar to that reported in the literature [2–6, 8,9,12–14,16–18]. The incidences of SSI in the single dose and extended dose arms were not statistically different (11.3% versus 14.7%) (p 0.40), conforming to the various randomized studies, meta-analyses, and large retrospective studies which have shown similar SSI rates in both short-course prophylactic antibiotic group as well as long course group in clean-contaminated surgeries.
Ishibashi et al. found short-course intravenous antimicrobial prophylaxis in conjugation with oral preoperative antibiotics as efficacious as 3 days extended antibiotic course in colon cancer surgery [19]. Similarly, Suzuki et al. also found a single dose of preoperative intravenous prophylactic antimicrobial along with chemical and mechanical bowel preparation to be as equally efficacious as extended intravenous antibiotics given for 4 days for elective colorectal cancer surgery [20]. In contrast, Fujita et al. had shown that 3 doses of cefmetazole to be more efficacious than a single dose in colorectal surgery [16].
A Cochrane meta-analysis of 34 studies evaluated the duration of antibiotic dosing for colorectal surgery and found no benefit with prolonged dosing over single-dose antibiotic (relative risk 1.10, 95% CI 0.93 to 1.30; P value 0.26) [4]. This review also showed benefit from the coverage of both aerobic and anaerobic microbes. Another Cochrane meta-analysis showed prolonged dosing in colorectal surgeries actually increases the incidence of Clostridium difficile colitis, and the emergence of resistant microbial clones [21]. Several randomized studies and a meta-analysis found a single antimicrobial dose to be as efficacious as extended antibiotic dosing in reducing surgical site infection after gastric cancer surgery [3, 22–26].
Even in gynecological surgeries, a single dose of antibiotic prophylaxis was shown to be sufficient. In a randomized clinical study involving 396 patients [27], a single dose of cefotaxime was demonstrated to be as efficacious as conventional multi-dose antibiotic therapy. It is also cost-effective and safe for both obstetric and gynecological surgeries. A single dose of cephazolin for antibiotic prophylaxis after hysterectomy is recommended by the American College of Obstetricians and Gynecologists [28]. Lastly, a randomised clinical trial involving biliary surgeries noted no remarkable difference in the incidences of wound infection between the single and multiple-dose regimens of cefotaxime [29].
The World Health Organization (WHO) meta-analysis of randomized clinical trials indicated that prolonging antibiotic prophylaxis dosing had no advantage over a single dose in decreasing SSI after surgery, including clean-contaminated. The meta-analysis also showed that antibiotic prolongation causes an increased risk of clostridial enterocolitis and antimicrobial resistance. There was a higher frequency of drug-related complications like erythema, skin rash, pruritus thrombophlebitis, hypotension, and gastrointestinal disturbances. It also reported cost reduction with shorter antibiotic prophylaxis regimens [30]. Another meta-analysis revealed no crystal benefit of multiple antibiotic dosing over a single dose in the prevention of SSI across various surgical fields [31].
Surgeries on colon and rectum are highly prone to develop wound infections owing to the high bacterial load in the lumen, risk of fecal contamination during bowel handling, and dissemination of microorganisms from bowel contents. The incidence of surgical site infection after colorectal surgery remains high ranging from 5 to 26% [18,32] and up to 36% without antibiotic prophylaxis [33]. A Cochrane meta-analysis demonstrated a 66% reduction in the surgical site infection rate with prophylaxis antibiotic administration in colorectal surgeries [4]. Wound infection rates are more common after abdominoperineal excisions with primary perineal skin closure performed for rectal cancer. With neoadjuvant radiation therapy, the total infective complications of perineum double from 23 to 47% [34].
In the present study, SSI rates were not different in both the groups in patients undergoing colorectal surgery, 19% in the single dose group versus 25% in the extended dose group (p 0.48), though higher but still in the range of reported incidence. Overall, SSI rates in colorectal surgery were significantly more than in non-colorectal surgery (21 vs 8.57%) (p 0.0013). In univariate analysis, colorectal surgery was significantly associated with SSI. All the patients undergoing colorectal surgeries underwent mechanical bowel preparation with polyethylene glycol preparation (PEGLEC) and received metronidazole for anaerobic cover along with second-generation cephalosporin for the entire duration of prophylaxis. Oral antibiotic preparation preoperatively was not given for this group owing to its debatable role.
There was an increased incidence of remote site infection rate in the single dose group of 14.4% (23/159) compared to 10.2% (16/156) in the extended dose group respectively although not statistically significant (p 0.31). This higher incidence is in concordance with few studies which showed higher remote site infection in the single-dose antibiotic arm compared to multiple-dose arm in clean-contaminated surgeries [20,23,24]. Short antibiotic prophylaxis course may slightly increase the incidence of remote site infection. It is to be noted that remote site infection is not associated with surgery directly as it occurs postoperatively and not during the surgery. Early ambulation, aggressive pulmonary rehabilitation, and early removal of the indwelling urinary catheter are the effective measures to curtail remote site infection such as pneumonia and urinary tract infection.
We used a laparoscopic-assisted approach for colorectal surgeries. After laparoscopic mobilization of the colon, a small abdominal incision was utilized for resection and hand-sewn colorectal anastomosis and also for the creation of covering stoma which itself independently increases the rate of SSI. The increased SSI in colorectal surgeries in the present study also includes perineal wound infection in patients undergoing abdominoperineal resection for carcinoma rectum post neoadjuvant chemoradiation, a risk factor for SSI [34].
In this study, parameters like nodal dissection, smoking, anemia, and hospital stay in univariate analysis and age, smoking, nodal dissection, and hospital stay in multivariate analysis were associated with SSIs. There is a higher incidence of SSI with preoperative anemia, but it has not been recognized as an independent predictor [35,36]. Surgical site infections significantly increase the length of hospital stay, add to cost, and contribute to morbidity [37,38]. Across all sites and treatments, smoking has been consistently shown to increase complications from surgery. Former and current smokers with gastrointestinal malignancies have more likelihood to acquire surgical site infection [39,40]. Lymphadenectomy increases the duration, extent, and complexity of the surgery. Lymph leak and seroma formation lead to a longer retention of drains owing to prolonged drainage, leading to higher SSI [22,41].
To conclude, the present study found a single dose of cefuroxime as efficacious as multiple doses for reduction of surgical site infection during clean-contaminated surgery.
Acknowledgements
The authors like to acknowledge Dr. Hemant Nemade, Consultant Head and Neck Surgical Oncology, Basavatarakam Indo American Cancer Hospital and Research Institute, Hyderabad, in helping with the statistical analysis of our data.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Syed Nusrath, Email: dr.nusrath2008@gmail.com.
Abhijit Nair, Email: abhijitnair95@gmail.com.
Sridhar Dasu, Email: dasusridhar@gmail.com.
T. Subramanyeshwar Rao, Email: subramanyesh@gmail.com
K. V. V. N. Raju, Email: drkvvnraju2002@yahoo.co.in
Basanth Kumar Rayani, Email: basanthrayani@gmail.com.
Vibhavari Milind Naik, Email: vibnaik@gmail.com.
Sujit C. Patnaik, Email: drsujit888@gmail.com
R. Rajagopalan Iyer, Email: rajagopalan99@hotmail.com
Ajesh Raj Saksena, Email: ajeshraj@gmail.com.
M. Ramanuja Rao, Email: madiraju.r.r@gmail.com
Salman Saifuddin, Email: saifuddin_78@yahoo.com.
Harini Narayanan, Email: drharininarayanan@gmail.com.
Radha Rani Dandamudi, Email: dradharani@gmail.com.
Naresh Gupta, Email: nareshguptha555@gmail.com.
References
- 1.Burke JP. Infection control--a problem for patient safety. N Engl J Med. 2003;348:65–66. doi: 10.1056/NEJMhpr020557. [DOI] [PubMed] [Google Scholar]
- 2.Centers for Disease Control and Prevention National Nosocomial Infections Surveillance (NNIS) System report: data summary from January 1992–June 2001, issued August 2001. Am J Infect Control. 2001;29:404–421. doi: 10.1067/mic.2001.119952. [DOI] [PubMed] [Google Scholar]
- 3.Mohri Y, Tonouchi H, Kobayashi M, Nakai K, Kusunoki M, Mie Surgical Infection Research Group Randomized clinical trial of single- versus multiple-dose antimicrobial prophylaxis in gastric cancer surgery. Br J Surg. 2007;94:683–688. doi: 10.1002/bjs.5837. [DOI] [PubMed] [Google Scholar]
- 4.Nelson RL, Gladman E, Barbateskovic M (2014) Antimicrobial prophylaxis for colorectal surgery. Cochrane Database Syst Rev (5):CD001181 [DOI] [PMC free article] [PubMed]
- 5.Clarke-Pearson DL, Geller EJ. Complications of hysterectomy. Obstet Gynecol. 2013;121:654–673. doi: 10.1097/AOG.0b013e3182841594. [DOI] [PubMed] [Google Scholar]
- 6.Coppa GF, Eng K. Factors involved in antibiotic selection in elective colon and rectal surgery. Surgery. 1988;104:853–858. [PubMed] [Google Scholar]
- 7.Centers for Disease Control and Prevention. Procedure-associated Module. Surgical Site Infection (Internet). [Accessed 6th April 2019]. Available from https://www.cdc.gov/nhsn/pdfs/pscmanual/9pscssicurrent.pdf
- 8.Bratzler DW, Dellinger EP, Olsen KM, Perl TM, Perl TM, Auwaerter PG, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Surg Infect. 2013;14:73–156. doi: 10.1089/sur.2013.9999. [DOI] [PubMed] [Google Scholar]
- 9.Juul PZ, Klaaborg KE, Kronborg O. Single or multiple doses of metronidazole and ampicillin in elective colorectal surgery: a randomized trial. Dis Colon Rectum. 1987;30:526–528. doi: 10.1007/BF02554782. [DOI] [PubMed] [Google Scholar]
- 10.Göransson G, Nilsson-Ehle I, Olsson SA, Petersson BG, Bengmark S. Single-versus multiple-dose doxycycline prophylaxis in elective colorectal surgery. Acta Chir Scand. 1984;150:245–249. [PubMed] [Google Scholar]
- 11.Berríos-Torres SI, Umscheid CA, Bratzler DW, Leas B, Stone EC, Kelz RR, Reinke CE, Morgan S, Solomkin JS, Mazuski JE, Dellinger EP, Itani KMF, Berbari EF, Segreti J, Parvizi J, Blanchard J, Allen G, Kluytmans JAJW, Donlan R, Schecter WP, for the Healthcare Infection Control Practices Advisory Committee Centers for disease control and prevention guideline for the prevention of surgical site infection, 2017. JAMA Surg. 2017;152:784–791. doi: 10.1001/jamasurg.2017.0904. [DOI] [PubMed] [Google Scholar]
- 12.Garner JS, Jarvis WR, Emori TG, Horan TC, Hughes JM. CDC definitions for nosocomial infections, 1988 [published correction appears in am J infect control 1988 Aug;16(4):177] Am J Infect Control. 1988;16(3):128–140. doi: 10.1016/0196-6553(88)90053-3. [DOI] [PubMed] [Google Scholar]
- 13.National Nosocomial Infections Surveillance System National Nosocomial Infections Surveillance (NNIS) system report, data summary from January 1992 through June 2004, issued October 2004. Am J Infect Control. 2004;32:470–485. doi: 10.1016/j.ajic.2004.10.001. [DOI] [PubMed] [Google Scholar]
- 14.Lilani SP, Jangale N, Chowdhary A, Daver GB. Surgical site infection in clean and clean-contaminated cases. Indian J Med Microbiol. 2005;23:249. [PubMed] [Google Scholar]
- 15.Haga N, Ishida H, Ishiguro T, Kumamoto K, Ishibashi K, Tsuji Y, Miyazaki T. A prospective randomized study to assess the optimal duration of intravenous antimicrobial prophylaxis in elective gastric cancer surgery. Int Surg. 2012;97:169–176. doi: 10.9738/CC91.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fujita S, Saito N, Yamada T, Takii Y, Kondo K, Ohue M, Ikeda E, Moriya Y. Randomized, multicenter trial of antibiotic prophylaxis in elective colorectal surgery: single dose vs 3 doses of a second-generation cephalosporin without metronidazole and oral antibiotics. Arch Surg. 2007;142:657–661. doi: 10.1001/archsurg.142.7.657. [DOI] [PubMed] [Google Scholar]
- 17.Imai E, Ueda M, Kanao K, Miyaki K, Kubota T, Kitajima M. Surgical site infection surveillance after open gastrectomy and risk factors for surgical site infection. J Infect Chemother. 2005;11:141–145. doi: 10.1007/s10156-005-0379-X. [DOI] [PubMed] [Google Scholar]
- 18.Tang R, Chen HH, Wang YL, Changchien CR, Chen J-S, Hsu K-C, Chiang JM, Wang JY. Risk factors for surgical site infection after elective resection of the colon and rectum: a single-center prospective study of 2,809 consecutive patients. Ann Surg. 2001;234:181–189. doi: 10.1097/00000658-200108000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ishibashi K, Kuwabara K, Ishiguro T, Ohsawa T, Okada N, Miyazaki T, Yokoyama M, Ishida H. Short-term intravenous antimicrobial prophylaxis in combination with preoperative oral antibiotics on surgical site infection and methicillin-resistant Staphylococcus aureus infection in elective colon cancer surgery: results of a prospective randomized trial. Surg Today. 2009;39:1032–1039. doi: 10.1007/s00595-009-3994-9. [DOI] [PubMed] [Google Scholar]
- 20.Suzuki T, Sadahiro S, Maeda Y, Tanaka A, Okada K, Kamijo A. Optimal duration of prophylactic antibiotic administration for elective colon cancer surgery: a randomized, clinical trial. Surgery. 2011;149:171–178. doi: 10.1016/j.surg.2010.06.007. [DOI] [PubMed] [Google Scholar]
- 21.Nelson RL, Suda KJ, Evans CT. Antibiotic treatment for Clostridium difficile-associated diarrhoea in adults. Cochrane Database Syst Rev. 2017;3:CD004610. doi: 10.1002/14651858.CD004610.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Imamura H, Kurokawa Y, Tsujinaka T, Inoue K, Kimura Y, Iijima S, Shimokawa T, Furukawa H. Intraoperative versus extended antimicrobial prophylaxis after gastric cancer surgery: a phase 3, open-label, randomized controlled, non-inferiority trial. Lancet Infect Dis. 2012;12:381–387. doi: 10.1016/S1473-3099(11)70370-X. [DOI] [PubMed] [Google Scholar]
- 23.Takagane A, Mohri Y, Konishi T, Fukushima R, Noie T, Sueyoshi S, Omura K, Ono S, Kusunoki M, Mochizuki H, Sumiyama Y. A randomized clinical trial of 24 versus 72 h antimicrobial prophylaxis in patients undergoing open total gastrectomy for gastric cancer. Br J Surg. 2017;104:e158–e164. doi: 10.1002/bjs.10439. [DOI] [PubMed] [Google Scholar]
- 24.Han JH, Jeong O, Ryu SY, Jung MR, Park YK. Efficacy of single-dose antimicrobial prophylaxis for preventing surgical site infection in radical gastrectomy for gastric carcinoma. J Gastric Cancer. 2014;14:156–163. doi: 10.5230/jgc.2014.14.3.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ohashi M, Saka M, Katayama H, Okinaka K, Morita S, Fukagawa T, Katai H. A prospective cohort study to evaluate the feasibility of intraoperative antimicrobial prophylaxis in open gastrectomy for gastric cancer. Surg Infect. 2015;16:833–839. doi: 10.1089/sur.2015.021. [DOI] [PubMed] [Google Scholar]
- 26.Zhang CD, Zeng YJ, Li Z, Chen J, Li H-W, Zhang J-K, Dai DQ. Extended antimicrobial prophylaxis after gastric cancer surgery: a systematic review and meta-analysis. World J Gastroenterol. 2013;19:2104–2109. doi: 10.3748/wjg.v19.i13.2104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Boodt PJ, Snijders WP, Janknegt R. Single-dose prophylaxis in hysterectomies. An interim analysis. Pharm Weekbl Sci. 1990;12(6A):280–283. doi: 10.1007/BF01967833. [DOI] [PubMed] [Google Scholar]
- 28.American College of Obstetricians and Gynecologists Antibiotic prophylaxis for gynecologic procedures. ACOG practice bulletin no. 104. Obstet Gynecol. 2009;113:1180–1189. doi: 10.1097/AOG.0b013e3181a6d011. [DOI] [PubMed] [Google Scholar]
- 29.Castoldi R, Ferrari G, Di Palo S, Orsenigo E, Bartucci F, DiCarlo V. Prophylactic use of cefotaxime in biliary surgery. Comparison of single-dose versus multiple-dose schedule. Drugs. 1988;35(Suppl 2):151–153. doi: 10.2165/00003495-198800352-00031. [DOI] [PubMed] [Google Scholar]
- 30.World health organization. Infection prevention and control. Global guidelines on prevention of surgical site infection. Appendix 25. Summary of the systematic review of surgical antibiotic prophylaxis prolongation (Internet). [Accessed 6th April 2019]. Available from https://www.who.int/gpsc/appendix25.pdf
- 31.McDonald M, Grabsch E, Marshall C, Forbes A. Single- versus multiple-dose antimicrobial prophylaxis for major surgery: a systematic review. Aust N Z J Surg. 1998;68:388–396. doi: 10.1111/j.1445-2197.1998.tb04785.x. [DOI] [PubMed] [Google Scholar]
- 32.Smith RL, Bohl JK, McElearney ST, Friel CM, Barclay MM, Sawyer RG, et al. Wound infection after elective colorectal resection. Ann Surg. 2004;239:599–607. doi: 10.1097/01.sla.0000124292.21605.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Baum ML, Anish DS, Chalmers TC, Sacks HS, Smith H, Jr, Fagerstrom RM. A survey of clinical trials of antibiotic prophylaxis in colon surgery: evidence against further use of no-treatment controls. N Engl J Med. 1981;305:795–799. doi: 10.1056/NEJM198110013051404. [DOI] [PubMed] [Google Scholar]
- 34.Bullard KM, Trudel JL, Baxter NN, Rothenberger DA. Primary perineal wound closure after preoperative radiotherapy and abdominoperineal resection has a high incidence of wound failure. Dis Colon Rectum. 2005;48:438–443. doi: 10.1007/s10350-004-0827-1. [DOI] [PubMed] [Google Scholar]
- 35.Weber WP, Zwahlen M, Reck S, Misteli H, Rosenthal R. Buser AS et al the association of preoperative anemia and perioperative allogeneic blood transfusion with the risk of surgical site infection. Transfusion. 2009;49:1964–1970. doi: 10.1111/j.1537-2995.2009.02204.x. [DOI] [PubMed] [Google Scholar]
- 36.Mujagic E, Marti WR, Coslovsky M, Zeindler J, Staubli S, Marti R, Mechera R, Soysal SD, Gürke L, Weber WP. The role of preoperative blood parameters to predict the risk of surgical site infection. Am J Surg. 2018;215:651–657. doi: 10.1016/j.amjsurg.2017.08.021. [DOI] [PubMed] [Google Scholar]
- 37.Mahmoud NN, Turpin RS, Yang G, Saunders WB. Impact of surgical site infections on length of stay and costs in selected colorectal procedures. Surg Infect. 2009;10:539–544. doi: 10.1089/sur.2009.006. [DOI] [PubMed] [Google Scholar]
- 38.Mujagic E, Marti WR, Coslovsky M, Soysal SD, Mechera R. Strauss Mv et al. Associations of hospital length of stay with surgical site infections. World J Surg. 2018;42:3888–3896. doi: 10.1007/s00268-018-4733-4. [DOI] [PubMed] [Google Scholar]
- 39.Kelly KJ, Greenblatt DY, Wan Y, Rettammel RJ, Winslow E, Cho CS, Weber SM. Risk stratification for distal pancreatectomy utilizing ACS-NSQIP: preoperative factors predict morbidity and mortality. J Gastrointest Surg. 2011;15:250–261. doi: 10.1007/s11605-010-1390-9. [DOI] [PubMed] [Google Scholar]
- 40.Gajdos C, Hawn MT, Campagna EJ, Henderson WG, Singh JA, Houston T. Adverse effects of smoking on postoperative outcomes in cancer patients. Ann Surg Oncol. 2012;19:1430–1438. doi: 10.1245/s10434-011-2128-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jørgensen MG, Toyserkani NM, Thomsen JB, Sørensen JA. Surgical-site infection following lymph node excision indicates susceptibility for lymphedema: a retrospective cohort study of malignant melanoma patients. J Plast Reconstr Aesthet Surg. 2018;71:590–596. doi: 10.1016/j.bjps.2017.11.026. [DOI] [PubMed] [Google Scholar]