Abstract
A 69-year-old lady with 2 renal cell carcinomas, one sited at the upper pole of her solitary right kidney, underwent percutaneous image-guided cryoablation and developed urinothorax as a complication. This was diagnosed from pleural fluid analysis and radiology imaging with computed tomography (CT). Management included image-guided chest drain and retrograde ureteric stent insertion to divert the urine from entering the pleural cavity. CT images demonstrated a fistula between the site of renal puncture and the pleural cavity, indicating that the cryoprobes traversed the diaphragm during the procedure. This case highlights urinothorax as an unusual complication of cryoablation of renal cell carcinoma. Prompt diagnosis by interventional radiologists is crucial to avert from this potentially life-threatening complication.
Keywords: Urinothorax, Cryoablation, Renal cell carcinoma, Solitary kidney, Complication
Introduction
Image-guided ablation with heat or cold-based energy, for example, cryoablation is now an accepted treatment for small (<3 cm) renal cell carcinoma (RCC) and is a conditional recommendation according to American Urological Association guideline [1]. The technique has seen increased use in treating renal tumours, especially in patients with impaired renal functions or a solitary kidney. Due to its minimal invasiveness, ability to preserve renal function and lower risk of requiring the dialysis post-procedure, cryoablation has comparable oncological durability as with partial nephrectomy [2]. Its primary advantage over other ablation techniques, such as radiofrequency ablation and microwave ablation, is the ability to monitor the ablation zone (ie, ability to visualise the ice ball) during the procedure [3].
Urinothorax is an extremely rare but important cause of pleural effusion. The phenomenon was first described by Corriere et al. in 1968 [4]. The condition is characterized accumulation of urine in the pleural space and can be life threatening in the absence of timely management. In a recent systematic review of urinothorax, there were only 88 cases reported in the published literature [5]. We present here an unusual case of urinothorax post-percutaneous image-guided renal cryoablation.
Case report
This case report has followed the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Clinical details
A 69-year-old lady was referred for consideration of image-guided renal ablation for 2 new renal tumours within her solitary right kidney, one sited at the upper pole measuring 2.7 cm (Figs. 1a and b) and one located in the lower pole measuring 2.1 cm. The patient had previous left RCC for which she underwent open left radical nephrectomy and splenectomy 21 years ago. She had a history of ulcerative colitis, primary sclerosing cholangitis, hiatus hernia and chronic airway disease with occasional infective exacerbations. The renal multidisciplinary team had recommended percutaneous image-guided cryoablation as the management of choice to preserve her renal function in the context of the solitary kidney status and her chronic renal impairment status with an estimated Glomerular Filtration Rate (eGFR) of 52 mL/min/1.73 m².
Fig. 1.
(a) Axial and (b) sagittal contrast-enhanced CT showed the right upper pole RCC (horizontal arrow) and its relation to the diaphragm (vertical arrow).
Technical details of image-guided cryoablation
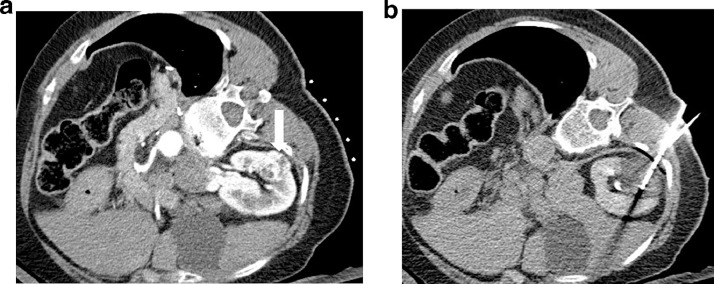
The patient underwent percutaneous image-guided renal cryoablation under general anaesthesia. Under computed tomography (CT) guidance, 3 IceRod and 2 IceSphere cryoprobes were inserted into the right upper pole RCC (Figs. 2a and b), and 4 IceSphere cryoprobes were inserted into the right lower pole RCC. Two complete freezing and thawing cycles were performed with a total treatment time of 32 minutes. The patient experienced no immediate complications and was monitored for as per standard care in the perioperative recovery ward. After recovering from general anaesthesia, she was transferred to the regular ward.
Fig. 2.
(a) Axial contrast-enhanced CT showed an avidly enhancing right upper pole RCC (vertical arrow) and (b) ice ball formation with water density Hounsfield Unit (HU) during image-guided Cryoablation (CRYO) treatment following cryoprobes insertion, where the cryoprobes have been shown to pass through the diaphragm and costophrenic space.
Post-image-guided cryoablation
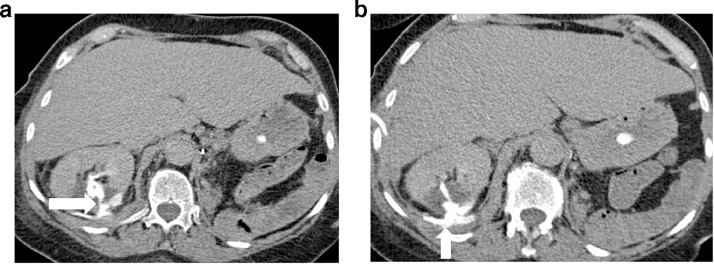
Day 2 post-cryoablation, the patient developed shortness of breath with raised temperature. Clinically, she was treated for acute chest infection with oral Augmentin. She unfortunately later developed respiratory failure and further deterioration of her renal function, thus was subsequently transferred to intensive care unit for close monitoring. The initial CT showed a small right perinephric haematoma, a small right-sided reactive pleural effusion and post-ablation changes (Fig. 3). On intensive care unit, continuous veno-venous haemodialysis was initiated for acute kidney injury and a chest drain was placed to drain the pleural effusion. Over the next 2 weeks, there were re-accumulations of pleural effusion when the chest drain was removed. On the second occasion of chest drain removal as the patient improved clinically, she became anuric over the course of a few days and increasingly unresponsive with respiratory failure requiring non-invasive ventilation. There was no improvement in urine output despite the use of diuretics.
Fig. 3.
Coronal non–contrast-enhanced CT chest and abdomen showed a small pleural effusion (horizontal arrow) and post-renal ablation changes, for example, perirenal haematoma (vertical arrow).
Diagnosing urinothorax
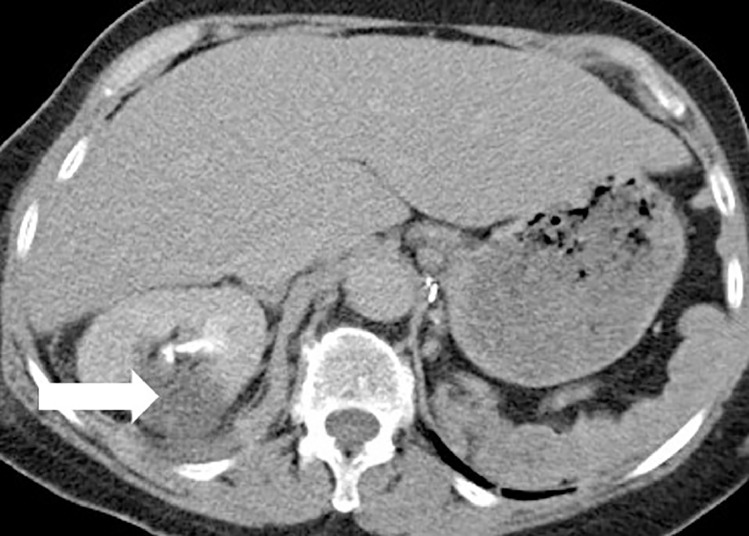
Following multidisciplinary discussion with consultant intensivists, consultant urologists and interventional radiologists, the suspicion of urine leaking into the pleural space was raised. The third bedside Ultrasound (USS) confirmed a right-sided pleural effusion for which a chest drain was inserted. The drain fluid showed elevated levels of urea (129 mmol/L), creatinine (1423 µmol/L) and no glucose. The culture of this fluid also grew Enterococcus faecium for which she was started on intravenous Teicoplanin. The consensus diagnosis of urinothorax was reached and believed to be due a fistula between the retroperitoneal space and pleural space, secondary to the initial cryoablation procedure. A right-sided retrograde ureteric stent was inserted under general anaesthesia to divert urine distally and allow the presumed fistula to heal. Four days after treating urinothorax, the patient recommenced passing urine, with good drainage of the remaining fluid inside the chest. Subsequent CT had confirmed the urinary leakage from the right upper pole calyx into the pleural cavity (Figs. 4a and b) which eventually healed 1 month after the retrograde ureteric stent insertion (Fig. 5). Unfortunately, her recovery was eventful and the patient developed empyema that required prolonged antibiotics treatment, which was later complicated by an atypical infection with Klebsiella oxytoca and Enterobacter cloacae. At week 5 after cryoablation, she underwent a right-sided video-assisted thoracic surgery washout and partial decortication to manage any further risks of infection. She was discharged home successfully a few days post-video-assisted thoracic surgery.
Fig. 4.
(a) Axial contrast-enhanced CT during delay excretory phase showed contrast-enhanced urine leaking from the right upper pole calyx (horizontal arrow) into the (b) pleural cavity (vertical arrow).
Fig. 5.
Axial contrast-enhanced CT during excretory phase showed that the reno-pleural fistula had healed (horizontal arrow), 1 month after retrograde ureteric stent insertion.
During her first follow-up outpatient visit at 2 months post-cryoablation, she developed an episode of recurrent pleural effusion which was drained without further event. The retrograde ureteric stent was removed uneventfully at 2.5 months post-cryoablation with no further evidence of urine leakage. To date, she remains cancer free at 2 years post-renal cryoablation with preservation of the renal function stabilising at eGFR of 37 mL/min/1.73 m² and no need for permanent renal dialysis.
Discussion
Urinothorax can be attributed to traumatic and obstructive origin [6]. Obstructive causes are usually associated with a bilateral or a common distal obstruction in the urinary tract system (eg, prostate disease bladder cancer and urethral valves). Traumatic causes can be secondary to accidents or iatrogenic, which includes surgical injury, blunt trauma, percutaneous nephrostomy and renal calculi lithotripsy. Urine can reach the pleural space via multiple routes, for example, urine passage through the retroperitoneal space and its lymphatic system then reaching the pleural space due to lymphatic drainage from an increased pressure from an urinoma. Intra-abdominal fluid can also drain into the pleural space through an anatomical defect in the diaphragm, which is a common cause for hydrothorax in cirrhotic and ascitic patients [5,7]. Currently, there is no reported case of urinothorax post-image-guided renal cryoablation, but there have been 2 isolated case reports of reno-pleural fistula after percutaneous image-guided ablation with heat-based energy (microwave ablation and radiofrequency ablation each) of renal tumour for 2 patients with Von Hippel-Lindau syndrome [8,9].
The most common presentation of urinothorax includes dyspnoea, chest pain, abdominal pain, decreased urine output, fever and sometimes ascites [5]. Once there is sufficient clinical suspicion of urinothorax, it can be diagnosed from the analysis of pleural effusion fluid based on 3 criteria: (1) the fluid is transudate, (2) pH is less than 7.3 and (3) pleural fluid/creatinine ratio is greater than 1 [10]. However, it has been reported that leaked urine progressively bears more resemblance to serum as the condition persists and thus the pleural fluid/creatinine ratio may not be always elevated [7]. Surprisingly, urinothorax can present with exudate fluid as well [5]. Therefore, early thoracentesis and analysis is crucial in accurate diagnosis. Other biochemical features of this fluid may include low glucose, low protein and high Lactate Dehydrogenase (LDH) level, though these findings are inconsistent across reported cases [5,6]. To aid diagnosis, contrast-enhanced CT or renal nuclear medicine scan such as 99mTc-Mercaptoacetyltriglycine (MAG-3) or 99mTc-DTPA (diethylenetriaminepentacetate) can help to demonstrate migration of urine into the pleural space [7,8].
Urinothorax is managed with chest drainage, clearance of obstructive uropathy and diversional procedure such as nephrostomy, ureteral catheter or bladder catheter to prevent further urine leakage into the pleural space [11]. Decortication, like the management in this case, may also be necessary to manage the complication from the long-standing pleural effusion [5].
The aim to avoid renal dialysis was achieved in this case; however, there is >25% deterioration of the patient's renal function post-cryoablation. The outcome corresponds to Weisbrod et al., where 50% of their patient population (n = 25) with solitary kidney had statistically significant decrease in eGFR post-renal cryoablation [2]. On reflection, the technical lesson learnt when treating the upper pole RCC with image-guided ablation, care should be taken to ensure the pleural had not been breached by the treatment probe. However, often the extreme caudal extent of the pleura can be hard to identify on CT and so inadvertent plural breach can still happen often without complication with the image-guided ablation of upper pole renal tumours. Therefore, it is crucial to have a low index of suspicion to consider the diagnosis of urinothorax caused by a pleural fistula in the context of renal failure and a persisting ipsilateral pleural effusion.
Conclusion
Urinothorax caused by pleural fistula should be considered in the presence of renal failure and a persisting ipsilateral pleural effusion after percutaneous image-guided renal ablation of an upper pole renal tumour. Prompt management of the cause of urinothorax offers the best outcome to minimize empyema formation and long-term renal damage.
Footnotes
Author contributions: The authors made the following contributions to this study: guarantor of integrity of entire study: T.M. Wah; literature search: H. Ng, K. Wang, T.M. Wah; case acquisition: T.M. Wah; manuscript preparation and editing: H. Ng, K. Wang, T.M. Wah; and manuscript review: all authors.
Funding: This study was not supported by any funding.
Competing interests: Professor Tze Min Wah receives educational grant from Angiodynamics and research grant from Boston Scientific.
Ethical approval: No ethical approval was required as the study was not classified as research under the United Kingdom National Health Service Health Research Authority. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent: Informed consent was obtained from all individual participants included in the study.
Consent for publication: Consent for publication was obtained for every individual person's data included in the study.
References
- 1.Campbell S, Uzzo RG, Allaf ME, Bass EB, Cadeddu JA, Chang A. Renal mass and localized renal cancer: AUA guideline. J Urol. 2017;198(3):520–529. doi: 10.1016/j.juro.2017.04.100. [DOI] [PubMed] [Google Scholar]
- 2.Weisbrod AJ, Atwell TD, Frank I, Callstrom MR, Farrell MA, Mandrekar JN. Percutaneous cryoablation of masses in a solitary kidney. Am J Roentgenol. 2010;194(6):1620–1625. doi: 10.2214/AJR.09.2978. [DOI] [PubMed] [Google Scholar]
- 3.Garnon J, Van Strijen MJ, Nielsen TK, King AJ, Montauban Van Swijndregt AD, Cazzato RL. Safety of percutaneous renal cryoablation: an international multicentre experience from the EuRECA retrospective percutaneous database. Eur Radiol. 2019;29(11):6293–6299. doi: 10.1007/s00330-019-06191-y. [DOI] [PubMed] [Google Scholar]
- 4.Corriere J, Miller W, Murphy J. Hydronephrosis as a cause of pleural effusion. Radiology. 1968;90(1):79–84. doi: 10.1148/90.1.79. [DOI] [PubMed] [Google Scholar]
- 5.Toubes ME, Lama A, Ferreiro L, Golpe A, Alvaroz-Dobano JM, Gonzalez-Barcala FJ. Urinothorax: a systematic review. J Thorac Dis. 2017;9(5):1209–1218. doi: 10.21037/jtd.2017.04.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Garcia-Pachon E, Padilla-Navas I. Urinothorax: case report and review of the literature with emphasis on biochemical diagnosis. Respiration. 2004;71(5):533–536. doi: 10.1159/000080642. [DOI] [PubMed] [Google Scholar]
- 7.Lee TW, Jang HN, Cho HS, Choi SM, Choi BH, Bae E. Urinothorax after ultrasonography-guided renal biopsy: a case report. BMC Nephrol. 2018;19(1):104. doi: 10.1186/s12882-018-0903-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yaqub K, Sakata K, Viggiano R. A unique case of chest and abdominal pain. Chest. 2015;148(4):452A. [Google Scholar]
- 9.Nilsen-Nunn A, Beckley I, Wah TM, Cartledge J, Joyce A. An unusual presentation of a reno-pleural fistula. J Clin Urol. 2013;7:66–68. [Google Scholar]
- 10.Ray K, Rattan S, Yohannes T. Urinothorax: unexpected cause of a pleural effusion. Mayo Clin Proc. 2003;78(11):1433–1434. doi: 10.4065/78.11.1433. [DOI] [PubMed] [Google Scholar]
- 11.Hendriks J, Michielsen D, Van Schil P, Wyndaele JJ. Urinothorax: a rare pleural effusion. Acta Chir Belg. 2002;102(4):274–275. doi: 10.1080/00015458.2002.11679314. [DOI] [PubMed] [Google Scholar]