The global epidemic of obesity has led to the rise of nonalcoholic fatty liver disease (NAFLD) as a significant cause of cirrhosis, end-stage liver disease, and need for liver transplantation.(1) NAFLD is common, with a global estimate of 25% of adults(2); however, only a small proportion will progress to cirrhosis and develop liver-related morbidity. As with other chronic liver diseases, the severity of underlying liver fibrosis aids prediction of outcome, with patients with bridging fibrosis or cirrhosis being at greatest risk of future liver-related morbidity.(3) Nonetheless, key questions remain largely unanswered, including what proportion of subjects develop progressive disease, how fast this occurs, and how to identify and monitor these individuals. These questions are becoming increasingly relevant given that efficacious drug treatments are likely to become available within the next few years and will need to be targeted at those with greatest risk of disease-related morbidity.
In this context, Sanyal and colleagues explore in this issue of Hepatology the progression of liver disease and predictors of fibrosis progression and decompensation, as well as potential monitoring strategies in 475 patients with nonalcoholic steatohepatitis (NASH) and bridging fibrosis or compensated cirrhosis.(4) Notably, 68% of patients with cirrhosis had a baseline hepatic venous pressure gradient (HPVG) measurement of ≥10 mm Hg, demonstrating that the majority had clinically significant portal hypertension at study entry. The cohort originated from two phase 2 randomized clinical trials of simtuzumab, a humanized monoclonal antibody targeting lysyl oxidase-like 2 (LOXL2), which had negative results.(5) Patients were assessed with paired liver biopsies and HPVG measurements (if cirrhotic at baseline) and were followed for outcomes. The study has a number of strengths, including a relatively large number of well-defined patients with standardized follow-up and protocol biopsies that have been centrally scored by a single liver pathologist, thereby limiting selection bias and observer variability. Additional strengths include the use of HPVG to measure changes in portal hypertension and image morphometry to quantify liver fibrosis. The study design, however, also has some intrinsic limitations, with strict inclusion and exclusion criteria limiting generalizability, limited length of follow-up, and the possibility of altered lifestyle behavior of participants within the trial thereby changing the “natural history” of the disease.
The 20% Rule (Fig. 1)
FIG. 1.
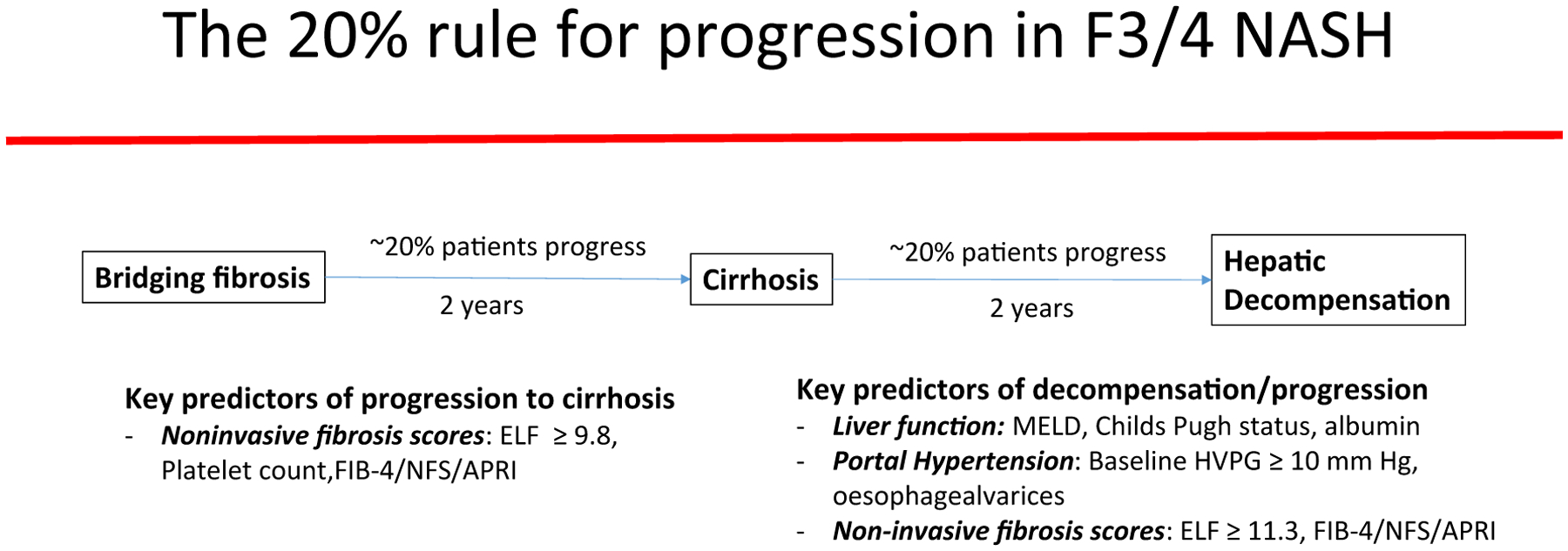
The natural history of disease progression in NASH with advanced fibrosis and cirrhosis: the 20% rule. Approximately 20% of patients with NASH with advanced NAFLD Fibrosis Score (NFS) (F3) fibrosis or compensated cirrhosis will progress to cirrhosis or develop decompensation, respectively, over a 2-year time period.
Although the study was relatively short (96 weeks), a surprisingly high proportion of patients progressed or developed liver-related complications: 22% (48/217) patients with baseline F3 fibrosis progressed to cirrhosis. Although sampling error cannot be excluded as a reason for apparent fibrosis progression, those patients with F3 fibrosis who progressed had lower NAFLD fibrosis scores and Fibrosis-4 score (FIB-4) and aspartate aminotransferase-to-platelet ratio index (APRI) levels than patients with cirrhosis at baseline, suggesting that these were different groups. In addition, 19% of patients with cirrhosis (50/258) had a liver-related event. Similarly, Hui et al. found a 23% incidence of decompensation at 3 years in a small cohort of patients with NASH cirrhosis.(6) Although NASH is thought to be a relatively indolent and slowly progressive disease, this suggests heterogeneity in disease progression with a subset of patients having an accelerated disease course. This is supported by a meta-analysis of patients with NAFLD undergoing paired liver biopsies that found a similar proportion (21%) of patients were “rapid progressors” who developed F3/4 fibrosis from a baseline of no/minimal fibrosis over a mean of 5.9 years.(7)
Can we predict progression to cirrhosis or future decompensation? In the Sanyal study, simple demographics, such as age, sex, and body mass index, did not predict progression, although a trend was detected regarding type 2 diabetes mellitus and liver decompensation (hazard ratio 1.90; P = 0.07). Liver biopsy added limited guidance: although severe hepatocellular ballooning was associated with fibrosis progression, supporting its inclusion within the histological definition of NASH, neither baseline or change in steatosis, lobular inflammation, or the NAFLD activity score were predictive of disease progression or regression in patients with advanced fibrosis. Thus, as pointed out by the study authors, treatment endpoints for patients with advanced fibrosis/cirrhosis should focus on fibrosis and clinical outcomes.
Surprisingly, pathologist staging (Ishak stage 4 [marked bridging] versus Ishak stage 3 [occasional bridging]) did not predict a greater risk of progression to cirrhosis, nor did stage 6 versus 5 predict future clinical decompensation. In contrast, computerized quantification of collagen using image morphometry predicted cirrhosis progression and decompensation, and alpha smooth muscle staining (a marker of hepatic stellate cell activation) was also predictive of cirrhosis progression. Nonetheless, baseline image morphometry had poor sensitivity and specificity (60% and 62%, respectively), suggesting it is not a useful in isolation.
The most notable parameters predicting cirrhosis progression and future liver-related events were serum markers (Fig. 1). These included simple noninvasive fibrosis algorithms (FIB-4, APRI, NAFLD Fibrosis Score) and proprietary algorithms (Enhanced Liver Fibrosis [ELF] score and FibroTest/FibroSure) as well as levels of serum LOXL2 (sLOXL2), a protein involved in collagen cross-linking and fibrosis matrix stabilization. On multivariable analysis, only ELF and platelet count remained predictive of progression to cirrhosis, albeit with modest accuracy (C statistic 0.76). The inclusion of liver biopsy parameters (alpha smooth muscle expression, fibrosis stage) to ELF and FIB-4 marginally increased the accuracy to 0.78. For the prediction of liver decompensation in patients with cirrhosis, Model for End-Stage Liver Disease, alkaline phosphatase, and albumin but not noninvasive fibrosis algorithms were significant on multivariable analysis. HPVG was predictive of decompensation, with 8% of patients with cirrhosis with an HVPG < 10 mm Hg developing decompensation compared with 25% with a reading ≥10. However, HPVG added marginal benefit over blood tests alone (C statistic 0.76 versus 0.75).
A related question is whether serum markers can be used to monitor disease progression or regression. Standard liver function tests (alanine aminotransferase, aspartate aminotransferase, gamma glutamyltransferase) were not useful. Increasing ELF, APRI, and sLOXL2 levels correlated with fibrosis progression; however, only the latter improved with fibrosis regression. Changes in FIB-4, NAFLD fibrosis score, and FibroTest/FibroSure were not associated with change in fibrosis. Recent data from the NASH Clinical Research Network suggest that incorporation of these scores with baseline fibrosis stage (determined by liver biopsy) may be useful to predict the development of advanced fibrosis but not progression of any fibrosis stage or resolution of fibrosis.(8,9)
Clinical Implications
The study by Sanyal suggests that 1 in 5 patients with NASH and advanced fibrosis or cirrhosis will develop cirrhosis or decompensation within a short time frame. Heightened levels of hepatic fibrogenesis, evidenced by increased hepatic stellate cell activation (alpha smooth muscle actin staining), collagen deposition on biopsy, and increased serum fibrosis markers, are associated with a greater risk of progression to cirrhosis and can aid in prioritizing patients for more intensive lifestyle management, monitoring, and clinical trials. Nonetheless, noninvasive serum markers only have modest accuracy to guide clinical management decisions (C statistic 0.76–0.78), and the optimal combination of parameters to accurately predict outcomes or reflect changes in disease state remains to be determined.
The Future
The importance of fibrosis in predicting outcomes in NAFLD has been highlighted by several studies,(3,10) with discrete fibrosis categories dramatically influencing future liver-related outcomes. The study by Sanyal and colleagues goes beyond the pathologist fibrosis stage to highlight the biological process of fibrogenesis. Future endeavors will need to focus on how best to capture this in a noninvasive fashion to enable accurate prediction and monitoring of patients with NAFLD without the need for liver biopsy.
Acknowledgments
Supported by NIEHS (5P42ES010337), NCATS (5UL1TR001442), NIDDK (R01DK106419), and DOD PRCRP (CA170674P2) to R.L.
Abbreviations:
- APRI
aspartate aminotransferase-to-platelet ratio index
- ELF
Enhanced Liver Fibrosis
- HPVG
hepatic venous pressure gradient
- NAFLD
nonalcoholic fatty liver disease
- NASH
nonalcoholic steatohepatitis
Footnotes
Potential conflict of interest: LA holds patents for Hepascore.
REFERENCES
- 1).Noureddin M, Vipani A, Bresee C, Todo T, Kim IK, Alkhouri N, et al. NASH leading cause of liver transplant in women: updated analysis of indications for liver transplant and ethnic and gender variances. Am J Gastroenterol 2018;113:1649–1659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2).Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease—meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016;64:73–84. [DOI] [PubMed] [Google Scholar]
- 3).Angulo P, Kleiner DE, Dam-Larsen S, Adams LA, Bjornsson ES, Charatcharoenwitthaya P, et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology. 2015;149:389–397.e310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4).Sanyal AJ, Harrison SA, Ratziu V, Abdelmalek M, Diehl AM, Caldwell S, et al. The natural history of advanced fibrosis due to nonalcoholic steatohepatitis: data from the simtuzumab trials. Hepatology 2019;70:1913–1927. [DOI] [PubMed] [Google Scholar]
- 5).Harrison SA, Abdelmalek MF, Caldwell S, Shiffman ML, Diehl AM, Ghalib R, et al. ; GS-US-321–0105 and GS-US-321–0106. Simtuzumab is ineffective for patients with bridging fibrosis or compensated cirrhosis caused by nonalcoholic steatohepatitis. Gastroenterology 2018;155:1140–1153. [DOI] [PubMed] [Google Scholar]
- 6).Hui JM, Kench JG, Chitturi S, Sud A, Farrell GC, Byth K, et al. Long-term outcomes of cirrhosis in nonalcoholic steatohepatitis compared with hepatitis C. Hepatology 2003;38:420–427. [DOI] [PubMed] [Google Scholar]
- 7).Singh S, Allen AM, Wang Z, Prokop LJ, Murad MH, Loomba R. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin Gastroenterol Hepatol 2015;13:643–654.e1–9; quiz e39–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8).Siddiqui MS, Yamada G, Vuppalanchi R, Van Natta M, Loomba R, Guy C et al. ; NASH Clinical Research Network. Diagnostic accuracy of noninvasive fibrosis models to detect change in fibrosis stage. Clin Gastroenterol Hepatol 2019;17:1877–1885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9).Chalasani N, Abdelmalek MF, Loomba R, Kowdley KV, McCullough AJ, Dasarathy S, et al. Relationship between three commonly used non-invasive fibrosis biomarkers and improvement in fibrosis stage in patients with non-alcoholic steatohepatitis. Liver Int 2018;39:924–932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10).Vilar-Gomez E, Calzadilla-Bertot L, Wai-Sun Wong V, Castellanos M, Aller-de la Fuente R, Metwally M, et al. Fibrosis severity as a determinant of cause-specific mortality in patients with advanced nonalcoholic fatty liver disease: a multi-national cohort study. Gastroenterology 2018;155:443–457.e417. [DOI] [PubMed] [Google Scholar]


