Abstract
Dolichoectasia of the distal internal carotid artery, posterior communication artery (PCoA) and posterior cerebral artery is an extremely rare abnormality. Dolichoectasia of the internal carotid artery, PCoA and the P1 segment of posterior cerebral artery can be postulated its pathogenesis by the embryological perspective basis from caudal ramus of the internal carotid artery terminus. The pathogenesis and treatment strategy are not well established. We reviewed and proposed embryological perspective, pathogenesis, clinical setting, radiological findings and management of this rare malformation.
Keywords: Dolichoectasia, aneurysm, embryology
Introduction
Intracranial arterial dolichoectasia (IADE) is defined as an increase in the length and diameter of at least one intracranial artery; the affected vessel is dilated, elongated and tortuous. The prevalence of the intracranial dolichoectasia ranges from 0.06 to 5.8% and usually involves posterior circulation, predominantly the basilar artery (BA), accounting for 80% of cases in people over 50 years old.1–4 Dolichoectasia can be categorized into two main types: congenital and atherosclerotic. The former is typically seen in younger patients without underlying atherosclerosis or other known causes of cerebral aneurysms. Some authors have proposed a third type called sequelae, from arterial dissection.5,6
In histopathological studies, IADE has revealed the fragmentation of the internal elastic lamina and rarefaction of the elastic tissue of the tunica media, whereas the lipid infiltration and inflammatory response at the arterial wall intima are significantly involved in atherosclerosis.7 The internal carotid artery (ICA), posterior communicating artery (PCoA), and posterior cerebral artery (PCA) dolichoectasia are considered exceedingly rare conditions, which can be grouped into the congenital type. Most of the literature involves sporadic case reports. The pathogenesis and predisposing factors are exceptionally different from classic IADE, particularly those described in the BA or diffuse intracranial dilative arteriopathy. It is our intention to specifically review dolichoectasia covering the embryological perspective, clinical presentation and pathogenesis.
Embryological and anatomical perspective
The peculiar luminal blood vessel has a strong linkage to embryological development. Certain diseases involve specific areas of the vascular segment, yet remarkably spare others. Such segmental specificities are beyond morphological analysis, creating an invisible discontinuity in a homogenous anatomical, histological and hemodynamic system. Pierre Lasjaunias calls this segmental property identity and vulnerability.8
At the 4th–5th week stage, the ICA branches divide into the cranial and caudal rami. Later, the cranial ramus of the ICA, which is a telencephalic brain artery, will give rise to the anterior cerebral artery (ACA), middle cerebral artery (MCA), and anterior choroidal artery (AchA), while the caudal division remains as PCoA and later annexes to the longitudinal neural axis derived BA distal to the trigeminal artery level (Figure 1). In summary, the caudal ICA ramus is the future PCoA, and future P1 segment of the PCA including the distal BA above the trigeminal artery level. The adult pattern PCA, with P2-3-4 segments, is considered the true PCA system.8–10
Figure 1.
Simplified illustration of the embryonic stage of vascular development. At the 4th–5th week stage, the ICA branches divide into the cranial and caudal rami (a). Later, the cranial ramus of the ICA, which is a telencephalic brain artery, will give rise to the ACA, MCA, and AchA, while the caudal division remains as PCoA and later annexes to the BA. The terminal caudal ICA ramus is the future PCoA and future P1 segment of the PCA (b). Source: reproduced with permission from Elsevier.10
We applied the seventh segment system proposed by Lasjaunias and Santoyo-Vazquez.11 They showed that the ICA derives from the distinct embryological segments. They proposed seven segments of the ICA separated by the branches of the embryonic vessels. All segments derive from the dorsal aorta, except the first segment, which derives from the third aortic arch. Those seven segments consist of the cervical ICA (first segment), the ascending petrous ICA (second segment), the horizontal petrous ICA (third segment), the ascending cavernous ICA (fourth segment), the horizontal cavernous ICA (fifth segment), the clinoid ICA (sixth segment), and the terminal ICA (seventh segment). In the adult configuration, the adult-type ophthalmic artery divides the sixth and seventh segments of the ICA. The seventh segment terminates at the origin of the PCoA. Literally speaking, dolichoectasia in our scope affects the seventh segment of ICA defined by Lasjaunias and Santoyo-Vazquez. However, the affected arteries cannot precisely designate the segment and demarcate from the adjacent part due to the variable tortuosity, elongation and disfiguration. The segments are subjectively designated by comparing the normal adult arrangement or normal contralateral side.
Gailloud et al. proposed a modified Lasjaunias–Vazquez pattern of segmentation by adding the eighth segment to describe their particular case: “segmental agenesis of the ICA distal to the PCoA”. The seventh and eighth segments were divided by PCoA, in which the eighth segment terminates at the ICA bifurcation. In this minor segment revision, they proposed that PCoA is simply another “embryonic bridged vessel” between the anterior and posterior circulations, comparable to the trigeminal artery and no longer regarded as coming from the caudal ramus of the ICA.11 Therefore, this interesting concept does not fit into the co-existing abnormalities found in ICA, PCoA, the P1 segment or distal BA.
As the matter of fact, some PCoA variants (circle of Willis variants) in the normal adult human regress to be so-called “hypoplasia” or “disappear” after the transfer of the PCA territory (embryonically speaking the cortical branches of the AchA) to the basilar system. This process seems to be obliged by the relatively large volume of the human brain compared to lower species.
The posterior circulation transfers from ICA to BA supply are due to the increasing demand for nutrients and the rapid growth of the posterior cerebrum (e.g. occipital lobes), whereby only ICA may not meet its consumption. If the hemodynamic shift does not take place, or ICA still forms a significant hemodynamic arrangement, the fetal pattern of PCA or the equal size of PCoA and P1 are persistent in the adult. Albeit in the absence of PCoA, the concomitant dolichoectasia found in distal ICA, the P1 segment and/or distal BA is able to link to the abnormality of the caudal ramus of ICA from the phylogenetic standpoint.
The BA is formed by the convergence of two combinations: one is the caudal ramus of the ICAs at the level above the trigeminal artery and the other is formed by the longitudinal neural plexus below the trigeminal artery. The trigeminal artery and carotid system determine the arrangement of the distal BA configuration by the reversal flow and vessel involution. In the circumstance when the carotid system is dominant, the more likely the disposition of caudal fusion of distal BA. Therefore, the dolichoectasia of BA might not be observed in this case.
Clinical manifestation
A dolichoectatic aneurysm may be discovered incidentally from neuroimaging without relevant symptoms related to the aneurysm. Some patients may present with a headache or other symptoms related to the compression of adjacent structures and associated cranial nerves. The mechanism of the headache is generally attributed to traction on the nearby pain-producing structures.12
Presentation with an aneurysm subarachnoid hemorrhage can be more complicated than berry aneurysm in terms of diagnosis and treatment due to the complex angioarchitecture of the branch vessels (Figure 2). Transient ischemic attacks and cerebral infarction may occur due to hemodynamic stress and abnormality/dysfunction of the vessel wall endothelium, diminished blood flow, stasis and turbulence resulting in arterial-to-arterial embolization or arterial in-situ thrombosis13,14 (Figure 3).
Figure 2.
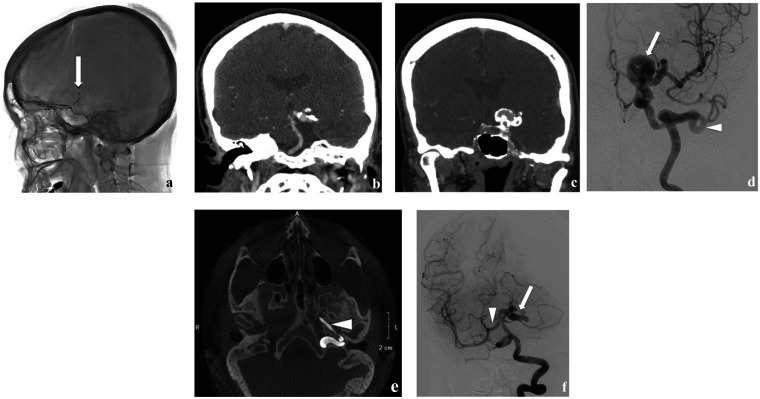
A case of calcified dolichoectasia in the right PCoA-PCA with acute subarachnoid hemorrhage. Axial (a) non-contrasted CT scan of the brain shows acute subarachnoid hemorrhage at the right ambient cistern and tubular-like shaped calcifications at the right-sided suprasellar cistern. Axial (b) T1-weighted MR with gadolinium image shows a multi-lobulated aneurysm of the P1 segment in the right PCA and tortuous vessels. Left vertebral angiogram (c) shows dilated, elongated and tortuous at the right PCoA (arrow), which contributed to the right MCA territories (double arrows) via the residual short segment of ICA. There is right ICA occlusion, which terminates at the ophthalmic artery (not shown). Note the large, multi-lobulated aneurysm at the P1 segment of the right PCA (arrowhead). Embolization was done at the outpouching portion, which was surrounded by the subarachnoid blood detected in the non-contrasted CT scan. Coil embolization (arrowhead) was meticulous to preserve the right PCA (arrow) (d).
Figure 3.
An illustrative case of dolichoectasia in the right ICA, P1 segment of the right PCA and distal BA presenting with acute left hemiparesis. Axial (a) diffusion-weighted MR image shows acute lacunar infarction at the left cerebral peduncle. Axial (b) proton density-weighted MR image shows the significant tortuous course of the supraclinoid right ICA and fetal-type AChA (arrow). Left vertebral angiogram anteroposterior (c) view shows irregular aneurysmal dilatation, and the elongated and tortuous P1 segment of the right PCA (arrowhead) and dysplasia of the distal BA (arrow). Notably, the SCA appears normal. Right internal carotid angiogram anteroposterior (d) views show a 360-degree loop of the right supraclinoid ICA and confirmed the fetal-type AchA (arrow). The fetal-type AChA gives the terminal temporal cortical branches. The fetal-type AChA variation is rare. In general, the telencephalic embryonic branches of the AchA will annex with the P1 and become the future P2, P3 and P4 in the typical adult human pattern. In this particular case, the abluminal P1 might fail to completely annex the embryonic AchA branches. The persistent embryonic arterial pattern is one piece of evidence that links segmental dolichoectasia back to a date in early life.
Pathogenesis
Several theories have been proposed to explain the pathogenesis of segmental vascular abnormalities. Congenital factors have been linked to the pathogenesis, including damage to or defects in the internal elastic lamina.8 Interestingly, there is a case series in which PHACE syndrome, a congenital disorder, can harbor segmental dolichoectasia of the distal ICA, PCoA, and the P1 segment of PCA with multiple aneurysmal formations.15 However, congenital factors are not always the instant answer; several diseases such as Ehlers–Danlos syndrome, Marfan’s syndrome and PHACE syndrome express their phenotypic features in other structures.
One of the particular curiosities of the congenital factors in pathogenesis is why it affects such a specific segment. Could it be an acquired etiology that has its effects in utero or unknowingly after birth? These acquired diseases have been implicated in infection, vascular inflammatory disease and trauma. These conditions are attributable to the direct mutilation of the endothelium or smooth muscles by pathogenic processes or indirectly through accelerated endothelial dysfunction. This still unknown trigger probably hits the patient, but the exposed, vulnerable segment response to these various stresses is variable and has different tolerability. Clinical expression, however, may vary in terms of time and clinical phenotypes.8,9,16
Vasculogenesis is the de novo formation of blood vessels from the mesodermal-derived endothelial cell (EC) precursors, the so-called angioblast, and the migration of the precursors of ECs and smooth muscle cells (SMC). Later, fusion, division and maturation in addition to complex signaling factors lead to the formation of simple tubes which become vessels, arteries and veins.17,18 Vascular SMCs can transform to osteo/chondrocyte-like cells that produce the extracellular matrix of collagen and non-collagenous proteins. The transcription factors including Runx-2, MSX2, Sox9 and osterix are involved in de-differentiating vascular SMC into osteochondrogenic phenotypes with upregulated expression of bone and chondrocyte proteins as well as the downregulation of the contractile proteins. This transformation process of vascular SMC has been evident during normal aging with associated cellular senescence. The calcified artery can occur in tunica intima and tunica media. In tunica intima, calcification can be apparent in atherosclerotic fibrofatty plaques. In the tunica media, the deposition of the extracellular matrix and thickening layer results in arteriosclerosis in smaller arteries, classically described as Monckeberg’s calcification or medial calcinosis.19 The vascular calcification in patients with chronic kidney disease can be regarded as the result of premature senescence.19,20 We proposed that the segmental dolichoectasia vessel wall was prone to atherosclerosis formation and calcification as a result of the premature aging of the vascular wall. This hypothesis is supported by several cases in the series of Jia et al. They reported the example case of vessel wall enhancement and calcification in a seven-year-old patient.15
In other words, its abnormal histologic segment is vulnerable and prone to decompensate to the exposure resulting from insufficient adaptation, vascular remodeling and fragility (Figure 4).
Figure 4.
An illustrative case of a woman who experienced chronic headache for years and developed left third cranial nerve paresis over a month. The skull radiography lateral view (a) shows serpentine, tram-track calcification at the suprasellar region (arrow). Coronal CT angiography of the brain (b) and (c) confirms the elongated, fusiform dilated vascular wall with calcification of the P1 segment of the left PCA and supraclinoid left ICA, respectively. Left internal carotid angiogram anteroposterior view (d) shows the elongated, fusiform dilated and tortuous nature of the left distal ICA (arrow). Axial reformation 3D digital subtraction angiography (e) shows the hyostapedial variant of the left ICA (arrowhead in d and e). Left vertebral artery angiogram anteroposterior (f) view shows elongated and fusiform dilatation of the P1 segment of the left PCA, including basilar tip (arrow). The P1 segment of the right PCA supplies the right MCA territory (arrowhead) due to aplastic right ICA (image not shown).
To the best of our knowledge, the pathogeneses of segmental dolichoectasia of the ICA, PCoA, P1 segment and distal BA are multifactorial mechanisms which are possibly provoked by unknown triggers in addition to intrinsic segmental dysfunction. All of this probably links to the early stages of life and development, the chronicity process before expressing its phenotypic feature, vascular elongation, tortuosity and calcification. Eventually, it results in clinical presentations in young adulthood.
Special attention should be addressed to the superior cerebellar artery (SCA). We observed no involvement of the SCA in our experience, which is similar to the observation in the Jia series.15 Although the SCA belongs to the caudal ramus ICA, it is a primitive vessel that holds the phylogenetics of the artery, in comparative anatomy, and supplies the cerebellum or posterior fossa content by the carotid system when the vertebral arterial system is not acting. The abluminal dolichoectatic vasculogenesis and the branching of the caudal ramus may have taken place after the establishment of the SCA.15,21 This is the explanation why the SCA remains intact (Figure 3).
Diagnostic and therapeutic overview
Generally, Computed Tomography Angiography (CTA) or Magnetic Resonance Angiography (MRA) is sufficient for the diagnosis of dolichoectasia. CTA can also illustrate the size of the aneurysm and is ideal for demonstrating the calcified vessel wall. Radiation exposure and contrast media allergies have to be taken into consideration by clinicians as well.
MRI scans of the intracranial vascular wall have gained increasing interest, mainly because they can directly visualize the features of the vascular wall behavior and pathology using high-resolution imaging. It has been applied in the broad spectrum of the vascular wall diseases from atherosclerotic plaque, intracranial arterial dissection and vasculitis to the prediction of an unruptured aneurysm.22 In the case of incidental findings or equivocal symptoms, this state-of-the-art imaging may play a crucial role in guiding treatments or the appropriate time of intervention. Besides, MRI of the intracranial vascular wall could also be used as a follow-up tool after providing treatment, such as for monitoring the activity of the disease through indirect visualization of the wall enhancement.
Catheter-based diagnosis is still necessary because it gives details for hemodynamics and a complete view of the complex arterial architecture. Digital Subtraction Angiography (DSA) is the gold standard for examining aneurysms and can also be used for surgical/interventional planning. While DSA gives details that may be the best for endoluminal imaging, it lacks vascular wall morphology, and procedural risk as well as interaction with adjacent structures.
The therapeutic management of this rare disease remains inconclusive at present. Current therapeutic options include observation, endovascular embolization, surgical trapping and bypass (Figure 5). Antiplatelet therapy is prescribed for patients presenting with transient ischemic attack or stroke.
Figure 5.
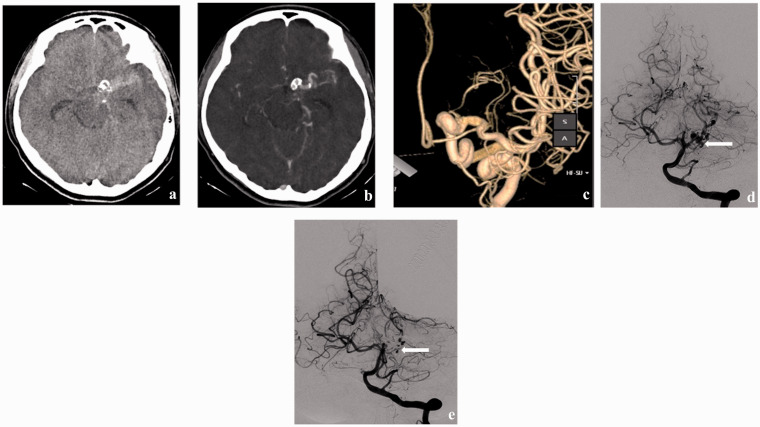
A case of dolichoectasia at the left ICA and P1 segment of the left PCA presented with acute subarachnoid hemorrhage. Axial non-contrasted CT scan (a) and axial CT angiography (b) of the brain show acute subarachnoid hemorrhage at the left-sided basal cistern and calcified left supraclinoid ICA. 3D-digital subtraction angiography reformation of the left internal carotid artery (c) shows fusiform dilatation, oblong and redundant. Left vertebral angiogram (d) shows multi-lobulated dilatation of the P1 segment of the left PCA (arrow). Post-surgical left vertebral angiogram (e) shows successful clipping (arrow). The left occipital artery–left PCA bypass is also successful (image not shown).
In incidental findings or with irrelevant symptoms, observation with imaging follow-up is proposed for the detection of evolutional morphological change, de novo or expansion of the aneurysmal formation, as well as mass effect or brain parenchyma.
In conclusion, the natural history of this condition is restrictive in terms of data due to its rarity. The vulnerability of its segmental embryological legacy is enhanced when considering that acquired constraint, healing and premature aging may possibly play a role. The management of these circumstances is challenging and strictly dependent on clinical presentation. Therapeutic opportunities offered depend on the clinical manifestation and resources available, though treatment should be carried out at centers with high volume and experience.
Authors’ contribution
Chai Kobkitsuksakul: Project development, review the article, manuscript drafting and revision. Kittiphop Somboonnitipon: Manuscript revision. Mungkorn Apirakkan: Manuscript revision. Peerapong Luengapapong: Manuscript revision. Ekachat Chanthanaphak: Review article, manuscript drafting and revision.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
- 1.Pico F, Labreuche J, Amarenco P. Pathophysiology, presentation, prognosis, and management of intracranial arterial dolichoectasia. Lancet Neurol 2015; 14: 833–845. [DOI] [PubMed] [Google Scholar]
- 2.Pico F, Labreuche J, Touboul PJ, et al. Intracranial arterial dolichoectasia and its relation with atherosclerosis and stroke subtype. Neurology 2003; 61: 1736–1742. [DOI] [PubMed] [Google Scholar]
- 3.Yu YL, Moseley IF, Pullicino P, et al. The clinical picture of ectasia of the intracerebral arteries. J Neurol Neurosurg Psychiatry 1982; 45: 29–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Flemming KD, Wiebers DO, Brown RD, Jr, et al. The natural history of radiographically defined vertebrobasilar nonsaccular intracranial aneurysms. Cerebrovasc Dis 2005; 20: 270–279. [DOI] [PubMed] [Google Scholar]
- 5.Chao KH, Riina HA, Heier L, et al. Endovascular management of dolichoectasia of the posterior cerebral artery report. AJNR Am J Neuroradiol 2004; 25: 1790–1791. [PMC free article] [PubMed] [Google Scholar]
- 6.Findlay JM, Hao C, Emery D. Non-atherosclerotic fusiform cerebral aneurysms. Can J Neurol Sci 2002; 29: 41–48. [DOI] [PubMed] [Google Scholar]
- 7.Caplan LR. Dilatative arteriopathy (dolichoectasia): what is known and not known. Annals Neurol 2005; 57: 469–471. [DOI] [PubMed] [Google Scholar]
- 8.Lasjaunias PL. Segmental identity and vulnerability in cerebral arteries. Interv Neuroradiol 2000; 6: 113–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bang OY, Toyoda K, Arenillas JF, et al. Intracranial large artery disease of non-atherosclerotic origin: recent progress and clinical implications. J Stroke 2018; 20: 208–217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Raybaud C. Normal and abnormal embryology and development of the intracranial vascular system. Neurosurg Clin N Am 2010; 21: 399–426. [DOI] [PubMed] [Google Scholar]
- 11.Gailloud P, Clatterbuck RE, Fasel JH, et al. Segmental agenesis of the internal carotid artery distal to the posterior communicating artery leading to the definition of a new embryologic segment. AJNR Am J Neuroradiol 2004; 25: 1189–1193. [PMC free article] [PubMed] [Google Scholar]
- 12.Little JR, St Louis P, Weinstein M, et al. Giant fusiform aneurysm of the cerebral arteries. Stroke 1981; 12: 183–188. [DOI] [PubMed] [Google Scholar]
- 13.Steel JG, Thomas HA, Strollo PJ. Fusiform basilar aneurysm as a cause of embolic stroke. Stroke 1982; 13: 712–716. [DOI] [PubMed] [Google Scholar]
- 14.Watanabe T, Sato K, Yoshimoto T. Basilar artery occlusion caused by thrombosis of atherosclerotic fusiform aneurysm of the basilar artery. Stroke 1994; 25: 1068–1070. [DOI] [PubMed] [Google Scholar]
- 15.Jia ZY, Zhao LB, Lee DH. Localized marked elongation of the distal internal carotid artery with or without PHACE syndrome: segmental dolichoectasia of the distal internal carotid artery. AJNR Am J Neuroradiol 2018; 39: 817–823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lasjaunias P, Berenstein A, TerBrugge KG. Surgical neuroangiography. Part 1. Clinical vascular anatomy and variations, 2nd ed Berlin: Springer-Verlag, 2001. [Google Scholar]
- 17.Kubis N, Levy BI. Vasculogenesis and angiogenesis: molecular and cellular controls. Part I: growth factors. Morphologie 2003; 87: 23–30. [PubMed] [Google Scholar]
- 18.Kubis N, Levy BI. Vasculogenesis and angiogenesis: molecular and cellular controls. Part 2: interactions between cell and extracellular environment. Interv Neuroradiol 2003; 9: 239–248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chen NX, Moe SM. Pathophysiology of vascular calcification. Curr Osteoporos Rep 2015; 13: 372–380. [DOI] [PubMed] [Google Scholar]
- 20.Monk BA, George SJ. The effect of ageing on vascular smooth muscle cell behaviour – a mini-review. Gerontology 2015; 61: 416–426. [DOI] [PubMed] [Google Scholar]
- 21.Lasjaunias P, Santoyo-Vazquez A. Segmental agenesis of the internal carotid artery: angiographic aspects with embryological discussion. Anat Clin 1984; 6: 133–141. [DOI] [PubMed] [Google Scholar]
- 22.Mandell DM, Mossa-Basha M, Qiao Y. al e and Vessel Wall Imaging Study Group of the American Society of N. Intracranial vessel wall MRI: principles and expert consensus recommendations of the American Society of Neuroradiology. AJNR Am J Neuroradiol 2017; 38: 218–229. [DOI] [PMC free article] [PubMed] [Google Scholar]