Abstract
The Human Immunodeficiency Virus Type I (HIV-1) subtype B comprises approximately 10% of all HIV infections in the world. The HIV-1 subtype B epidemic comprehends a pandemic variant (named BPANDEMIC) disseminated worldwide and non-pandemic variants (named BCAR) that are mostly restricted to the Caribbean. The goal of this work was the identification of amino acid signatures (AAs) characteristic to the BCAR and BPANDEMIC variants. To this end, we analyzed HIV-1 subtype B full-length (n = 486) and partial (n = 814) genomic sequences from the Americas classified within the BCAR and BPANDEMIC clades and reconstructed the sequences of their most recent common ancestors (MRCA). Analysis of contemporary HIV-1 sequences revealed 13 AAs between BCAR and BPANDEMIC variants (four on Gag, three on Pol, three on Rev, and one in Vif, Vpu, and Tat) of which only two (one on Gag and one on Pol) were traced to the MRCA. All AAs correspond to polymorphic sites located outside essential functional proteins domains, except the AAs in Tat. The absence of stringent AAs inherited from their ancestors between modern BCAR and BPANDEMIC variants support that ecological factors, rather than viral determinants, were the main driving force behind the successful spread of the BPANDEMIC strain.
Introduction
The Human Immunodeficiency Virus Type I (HIV-1) is one of the most important human pathogens that have emerged in the 20th century and exhibits an extraordinary degree of genetic variability, organized in groups (M, N, O, and P), subtypes (A-D, F-H, and J-L), sub-subtypes, and many recombinant forms [1]. HIV-1 subtype B comprises approximately 10% of all HIV infections in the world, being one of the most globally disseminated HIV-1 variants and the most prevalent subtype in the Americas, Europe, Oceania, as well as some Asian countries [2].
The HIV-1 subtype B shares a common ancestor with subtype D that was present in Kinshasa, capital of Democratic Republic of Congo (DRC), by the early 1940s [3]. The currently accepted hypothesis about the emergence and worldwide spread of subtype B is punctuated by two major founder events. The first event occurred when the HIV-1 subtype B ancestor strain moved from the DRC into the Caribbean around the middle 1960s, establishing the first HIV epidemic outside the African region [4–7]. The second major event took place when one subtype B strain spread from the Caribbean to the U.S. around the late 1960s and got access to high-risk transmission networks globally connected that fueled the establishment of a pandemic clade (BPANDEMIC) responsible for most of the infections of this subtype worldwide [8, 9]. In contrast, other non-pandemic subtype B lineages (BCAR) spread and circulates at high prevalence in several Caribbean islands and the Northern South American region, but were not successfully disseminated worldwide [4, 6, 8–11].
The introduction of the BPANDEMIC ancestor in highly connected transmission networks in the U.S. during the very early phase of the epidemic probably accounts for the successful dissemination of this viral variant worldwide [4, 12, 13]. Differences in viral fitness, however, may also have shaped the uneven geographic distribution of distinct HIV-1 subtype B lineages. Viral transmissibility correlated with the plasma viremia during chronic infection [14], and some studies found that plasma viremia within subtype B is highly heritable, thus indicating that this trait depends strongly on the virus genotype [15–18]. Notably, a significant trend for higher viral loads among subjects infected with BPANDEMIC relative to BCAR strains was recently described in French Guiana [11] which may have played a role in the differential transmissibility of the two viral strains. Studies of molecular signatures in non-pandemic subtype B lineages, however, have been limited so far to the analysis of the env gene of BCAR strains circulating in Trinidad and Tobago [19, 20].
The objective of this work is to identify amino acid signatures (AAs) that can distinguish BCAR and BPANDEMIC strains by the analysis of full-length (FL) and partial genome subtype B sequences representative of different Caribbean islands and American countries and by reconstructing the sequences of the most recent common ancestors (MRCA) of BCAR and BPANDEMIC strains. These analyses may provide crucial information to understand the potential relevance of viral genetic determinants on the epidemic spread of different subtype B variants.
Materials and methods
HIV-1 subtypes B and D datasets
HIV-1 subtype B FL genome sequences from North America (n = 330), South America (n = 151), and the Caribbean (n = 25), as well as Sub-Saharan African subtype D FL genome sequences (n = 18) that were available at Los Alamos HIV Database (http://www.hiv.lanl.gov) by November 2018, were downloaded (S1 and S2 Tables). We also downloaded HIV-1 subtype B sequences from Caribbean islands with high prevalence of BCAR strains and that covered selected genomic regions of gag (HXB2 coordinates: 1,264 to 2,148), pol (HXB2 coordinates: 2,253 to 3,272), and env (HXB2 coordinates: 6,450 to 8,480) and HIV-1 pol sequences (HXB2 coordinates: 2,253 to 3,272) from drug-naïve individuals of Caribbean origin (S1 Table). Only one sequence for patient was selected.
Clade assignment of HIV-1 subtype B sequences
The HIV-1 subtype B sequences were aligned with the HIV Align online tool [21] and then manually curated. The presence of putative intra-subtype recombinant sequences was evaluated using the RDP4 software [22], being deemed as recombinant those sequences selected as such by three or more of the algorithms. The remaining FL and partial subtype B genome sequences were classified as either BCAR or BPANDEMIC based on their placement on a maximum likelihood (ML) phylogenetic tree inferred with the PhyML program [23] under the best nucleotide substitution model, selected using an online web server [24]. The heuristic tree search was performed using the SPR branch-swapping algorithm, and the reliability of the obtained topology was estimated with the approximate likelihood-ratio test [25] based on the Shimodaira–Hasegawa-like procedure. The ML phylogenetic trees were visualized using the FigTree v1.4.4 program [26].
Reconstruction of ancestral subtype B sequences
To reduce computation time while retaining most viral diversity information, we generate a “non-redundant” subtype B FL genome subset by removing very closely related BPANDEMIC sequences. To achieve this goal, BPANDEMIC sequences with identity above 91.5% were grouped with the CD-HIT program [27] using an online web server [28], and only one sequence per cluster (the oldest one) was selected. To map amino acid changes fixed during the evolution of subtype B, FL genome sequences of the MRCA of BCAR and BPANDEMIC strains were then reconstructed using a Bayesian Markov Chain Monte Carlo (MCMC) approach, as implemented in BEAST v1.8 [29, 30] with BEAGLE [31] to improve run-time. The Bayesian time-tree was reconstructed using the GTR+I+Г4 nucleotide substitution model [32], a relaxed uncorrelated lognormal molecular clock model [33], and a Bayesian Skyline coalescent tree prior [34] with non-informative default priors. MCMC chains were run for 100 × 106 generations, and convergence and uncertainty of parameter estimates were assessed by calculating the Effective Sample Size (ESS) and 95% Highest Probability Density (HPD) values, respectively, after excluding the initial 10% of each run with Tracer v1.7 [35]. The convergence of parameters was considered when ESS ≥ 200. After the exclusion of sequences corresponding to the burn-in phase, the remaining ones were utilized to generate an FL consensus sequence for each MRCA using the Seaview version 4 program [36].
Amino acid signature (AAs) analyses
Nucleotide sequences were translated, and the software VESPA (Viral signature pattern analysis) [37] was used to identify positions in which the most common amino acid differs between BCAR and BPANDEMIC datasets as well as between subtype B and subtype D datasets. These positions were then selected, and for each a Chi-square test, as implement in R version 3.5.0 [38], was used to evaluate the statistical significance of their different amino acidic compositions. AAs were defined by positions in which both the most common amino acid was different, and the overall amino acid composition was significantly different between viral clades. For specific genomic regions corresponding to the structural gag, pol, and env genes, the number of BCAR sequences was expanded, and the process to identify AAs between BCAR and BPANDEMIC datasets previously detailed was applied. The false discovery rate (FDR) method was used to correct for multiple hypothesis testing and to reduce false positives. Statistical significance was defined as FDR < 0.05.
Phenotypic prediction
We determine the frequency of genetic polymorphisms in accessory (Vif, Vpr, Nef) and regulatory (Rev) HIV-1 proteins of BPANDEMIC and FL/expanded BCAR datasets that were previously associated with slow HIV-1 disease progression and differential function [39–50]. The Geno2Pheno algorithm was used to predict the chemokine receptor tropism of the BPANDEMIC and expanded BCAR env dataset sequences based on their V3 region [51]. V3 was studied through the 11/25 rule (R or K at position 11 and/or K at position 25) [52–54], and the combination of positively charged amino acids at position 25 and an increase in total net charge [55]. The frequency of surveillance drug-resistance mutations (SDRMs) was also explored in BCAR and BPANDEMIC pol sequences retrieved from drug-naïve individuals of Caribbean origin using the Calibrated Population Resistance (CPR) tool (http://cpr.stanford.edu/cpr.cgi) [56]. A Chi-square test, as implement in R version 3.5.0 [38] was used to evaluate the significance of the results in both cases. Statistical significance was defined as p-values < 0.05.
Results
Classification of HIV-1 BCAR and BPANDEMIC FL sequences
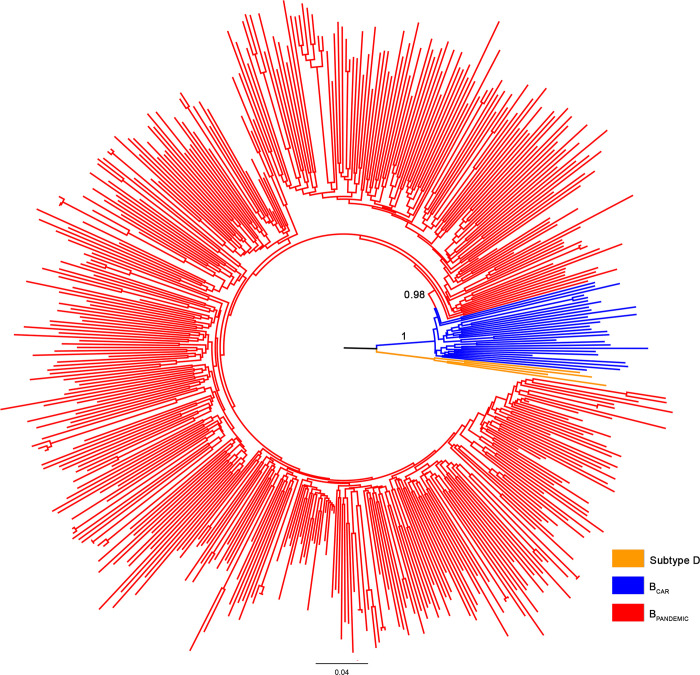
From the 506 HIV-1 subtype B FL genome sequences of American origin initially selected, 28 sequences (6%) were identified as putative intra-subtype B recombinants and removed from the final dataset (S1 Table). The ML phylogenetic analysis of the remaining 478 subtype B FL genome American sequences revealed that most Caribbean sequences (82%) branched as basal strains and were classified as BCAR strains, while most sequences from North (97%) and South (99%) America branched in a well-supported (SH-aLRT = 0.98) monophyletic sub-clade and were thus classified as BPANDEMIC strains (Fig 1 and S1 Table). Despite the low number of subtype B FL genome sequences available from the Americas, the estimated relative prevalence of the BCAR lineages in different countries was entirely consistent with previous estimates [6, 8, 9] based on much larger pol sequence datasets. Similarly, classification of additional subtype B Caribbean, covering specific regions of gag (n = 495), pol (n = 775), and env (n = 529) genes produced country ratios of BCAR/BPANDEMIC sequences akin to their counterparts based on the FL genome (S1 Fig and S1 Table).
Fig 1. ML phylogenetic tree of 478 HIV-1 subtype B FL genome American sequences.
Branches are colored according to their classification in pandemic (BPANDEMIC, n = 450) and non-pandemic (BCAR; n = 28) lineages as indicated in the legend at the bottom right. Node support (SH-aLRT) for subtype B and BPANDEMIC monophyletic groups are indicated. The tree was rooted using HIV-1 subtype D sequences. Branch lengths are drawn to scale with the bar at the bottom indicating nucleotide substitutions per site.
AAs in BCAR and BPANDEMIC modern sequences
In order to identify AAs of different subtype B clades, we compared the FL genome BPANDEMIC sequences (n = 450, sampled between 1978 and 2015) of American origin with FL genome (n = 18, sampled between 1983 and 2011), Gag (n = 28), Pol (n = 197), Env (n = 59) and Rev (n = 59, given the superposition of its CDS with Env) BCAR sequences of Caribbean origin (Table 1). Twenty-eight sequences were originally classified as BCAR, but 10 were removed for subsequent analyses because were sampled outside the Caribbean region. The same reasoning was used in the expanded dataset, where only BCAR sequences of Caribbean origin were considered. The analysis of translated FL genome sequences identified nine positions that displayed compositions significantly distinct between BCAR and BPANDEMIC datasets, covering structural (one in P6, one in PR and one in RT), regulatory (one in Tat and three in Rev) and accessory (one in Vif and one in Vpu) proteins (Table 2). Expanded BCAR datasets comprising partial regions of Gag, Pol, Env, and Rev encompass four out of the nine AAs previously identified. Three positions (one in PR, one in RT, and one in Rev) had their results endorsed by the additional sequences, while statistical significance in the P6 position was lost. Analyses of the expanded BCAR datasets also identified additional AAs not detected in the FL genome dataset (Table 2), four in Gag (three in P24, positions 27, 120 and 148; and one in P7, position 12) and another in the RT (position 211).
Table 1. Sequences used for identification of AAs signatures between BCAR and BPANDEMIC sequences.
| Country* | BPANDEMIC | BCAR | ||||||
|---|---|---|---|---|---|---|---|---|
| FL | Gag | Pol | Env/ Rev | Others | ||||
| FL | Expanded | FL | Expanded | FL | Expanded | FL | ||
| DO | - | 4 | - | 4 | 101 | 4 | - | 4 |
| HT | - | 5 | 4 | 5 | - | 5 | 6 | 5 |
| JM | 4 | 4 | 6 | 4 | 69 | 4 | - | 4 |
| TT | - | 5 | - | 5 | - | 5 | 35 | 5 |
| BR | 106 | - | - | - | - | - | - | - |
| US | 306 | - | - | - | - | - | - | - |
| Others | 34 | - | - | - | 9 | - | - | - |
| Total | 450 | 28 | 197 | 59 | 18 | |||
*Country codes are in accordance with ISO 3166–1. FL: Full length.
Table 2. Amino acid signatures of BCAR and BPANDEMIC datasets.
| Location | Gene | gag | pol | vif | vpu | tat | rev | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Protein | P24 | P24 | P24 | P7 | PR | RT | RT | Vif | Vpu | Tat | Rev | |||
| Position | 27 | 120 | 148 | 12 | 41 | 207 | 211 | 50 | 24 | 23 | 57 | 67 | 102 | |
| Ancestors | BCAR | V | S | V | I | K | E | R | R | T | T | A | S | V |
| BPANDEMIC | V | S | V | T | K | Q | R | R | T | T | A | S | V | |
| Datasets | D n = 18 | I89 V11 | S78 N17 | V100 | I56 T22 | K100 | E88 K6 | K61 R39 | K89 R11 | T94 S6 | N94 T6 | E88 A6 | S67 P33 | I78 V16 |
| BCAR n = 18 | I61 V39 | S50 N39 | V61 T22 | I39 T33 | K89 R11 | E50 A11 | K50 R50 | K56 R44 | T83 S6 | N44 T44 | A55 E39 | S83 P17 | V100 | |
| BCAR expanded | I71 V29 | S50 N25 | V68 T18 | I54 T25 | K74 R23 | E48 A25 | K54 R25 | - | - | - | A53 E32 | - | - | |
| n = 28 | n = 197 | n = 59 | ||||||||||||
| BPANDEMIC n = 450 | V69 I31 | N55 S26 | T66 V27 | T40 I28 | R75 K24 | Q82 E9 | R46 K44 | R76 K24 | S51 T45 | T76 N22 | G40 E27 | P59 S39 | I69 V28 | |
| p-values adj. | D/BCAR | - | - | - | - | - | - | 0.7563 | - | - | 0.00391 | 0.0039 | - | 0.0001 |
| D/BPANDEMIC | 0.0172 | 0.0461 | 0.0009 | 0.3137 | 0.0073 | 0.0002 | 0.7962 | 0.0004 | 0.0311 | 0.00229 | 0.0508 | 0.1355 | - | |
| BCAR/BPANDEMIC | 0.1084 | 0.3094 | 0.1084 | 0.0502 | 0.0200 | 0.0008 | 0.9670 | 0.0255 | 0.0015 | 0.0465 | 0.0300 | 0.0423 | 0.0300 | |
| BCAR exp/BPANDEMIC | 0.0091 | 0.0091 | 0.0376 | 0.0376 | 0.0001 | 0.0001 | 0.0001 | - | - | - | 0.0001 | - | - | |
The table details positions identified as bearing amino acidic compositions significantly divergent between BCAR and BPANDEMIC strains. For each position are supplied (1) the residues present in the reconstructed BCAR and BPANDEMIC ancestors; (2) the two most common amino acid observed in that position in Subtype D, BCAR, BCAR Expanded, and BPANDEMIC datasets accompanied by a number representing its frequency in each group; (3) the adjusted p-values. The sampling range of each sub dataset is indicated in the table. Ancestor sequences were reconstructed employing FL genomes of BCAR (n = 18) and BPANDEMIC (n = 69) lineages. G (Glycine), P (Proline), A (Alanine), V (Valine), L (Leucine), I (Isoleucine), M (Methionine), C (Cysteine), F (Phenylalanine), Y (Tyrosine), W (Tryptophan), H (Histidine), K (Lysine), R (Arginine), Q (Glutamine), N (Asparagine), E (Glutamic Acid), D (Aspartic Acid), S (Serine), T (Threonine). p-values < 0.05 are in bold.
By combining FL and partial genome BCAR datasets, 13 positions were considered as signature positions differentiating BCAR e BPANDEMIC clades: four located on Gag (three in P24, and one in P7); three on Pol (one in the PR and two in the RT); three in Rev; while Vif, Vpu, and Tat each contributed with one position (Table 2). No signature position was identified in Vpr, Env, or Nef. None of the 13 AAs that distinguished contemporary BCAR and BPANDEMIC sequences were found to be invariant sites, with the exception of position Rev 102 in the BCAR lineages. Furthermore, we also observed that for most AA positions, the most common amino acid found in a given subtype B clade corresponds to the second most frequent amino acid in the other clade (Table 2). The exceptions were position 207 in RT that displayed E48/A25 as the most frequent amino acids in BCAR strains and Q82/E9 in BPANDEMIC ones, and position 57 in Rev that displayed A53/E32 as the most frequent amino acids in BCAR strains and G40/E27 in BPANDEMIC ones.
AAs in BCAR and BPANDEMIC MRCA sequences
When the reconstructed MRCA sequences of BCAR and BPANDEMIC clades were compared, 21 amino acidic positions differentiated both ancestors (Table 3). Eight of them were located in Gag (one in P17, three in P24, two in P7, and two in P6); four in Pol (all of them in the RT); two in Vif, and four in Env (one in GP120, and three in GP41); while Tat, Rev, and Nef had one position each. Vpr and Vpu presented no difference in the comparisons. Of the 21 amino acidic positions that differentiated both ancestors, only four positions (three in Gag and one in Pol) displayed distinct majoritarian amino acids in the contemporary BCAR and BPANDEMIC datasets and only two of them (positions 12 in P7 and 207 in the RT) correspond to AAs between contemporary BCAR and BPANDEMIC sequences (Table 2). Thus, of the 21 amino acidic positions that differentiated the BCAR and BPANDEMIC ancestors only two continue to distinguish the contemporary descendant sequences. Furthermore, this analysis suggests that most (11/13) AAs that distinguished contemporary BCAR and BPANDEMIC sequences were probably not inherited from their ancestors. It is interesting to note, however, that the most common amino acid in 11 (including the two inherited from ancestors) out of 13 positions associated with the AAs was the same in BCAR and subtype D sequences; while subtype D and BPANDEMIC sequences coincide in only one position.
Table 3. Amino acid signatures of HIV-1 BCAR and 210 BPANDEMIC ancestors.
| Location | Gene | gag | Pol | vif | tat | rev | env | nef | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Protein | P17 | P24 | P24 | P24 | P7 | P7 | P6 | P6 | RT | RT | RT | RT | Vif | Vif | Tat | Rev | GP120 | GP41 | GP41 | GP41 | Nef | |
| Position | 84 | 15 | 91 | 180 | 12 | 26 | 30 | 42 | 35 | 49 | 122 | 207 | 47 | 66 | 74 | 56 | 363 | 133 | 182 | 209 | 10 | |
| Ancestors | BCAR | T | L | I | D | I | K | Q | R | A | R | K | E | P | V | S | G | P | N | V | L | M |
| BPANDEMIC | V | I | V | E | T | R | P | K | V | K | E | Q | T | I | A | S | Q | T | I | R | V | |
| Datasets | D | T83* V17 | L50 I50 | V50 I28 | D61 E39 | I56 T22 | K72 R28 | Q100 | K100 | T100 | R72 K28 | E61 K39 | E89 K6 | P66 H28 | V89 I11 | S100 | G89 A11 | P83 S11 | N44 S44 | I100 | L100 | I89 L11 |
| BCAR | T61 V39 | L50 I50 | I72 V22 | D56 E44 | I39 T33 | K61 R39 | P50 Q28 | K72 R28 | V83 T11 | K89 R6 | K67 P11 | E50 A11 | T44 P22 | I67 V33 | T44 A39 | S78 G17 | Q56 P17 | T28 N22 | I56 V44 | H44 R39 | L22 V17 | |
| BPANDEMIC | T61 V39 | I71 L28 | I57 V37 | E68 D32 | T40 I28 | R55 K45 | P61 Q12 | R53 K47 | V79 I12 | K93 R7 | K62 E35 | Q82 E9 | T58 P15 | I64 V35 | T45 A41 | S96 G4 | Q49 H16 | T45 N26 | I58 V41 | H45 R42 | V18 L14 | |
The table summarizes the most common amino acid for positions in which the reconstructed ancestors of Subtype B and BPANDEMIC diverged, accompanied by a number representing its frequency in each group. G (Glycine), P (Proline), A (Alanine), V (Valine), L (Leucine), I (Isoleucine), M (Methionine), C (Cysteine), F (Phenylalanine), Y (Tyrosine), W (Tryptophan), H (Histidine), K (Lysine), R (Arginine), Q (Glutamine), N (Asparagine), E (Glutamic Acid), D (Aspartic Acid), S (Serine), T (Threonine).
Predicted phenotypic characteristics of BCAR and BPANDEMIC strains
Most AAs that distinguish the BCAR and BPANDEMIC strains were located outside domains or sites previously reported to be essential for protein function [40, 57–66] (Fig 2). The sole exception was position 23, located in Tat cysteine-rich domain (22–37), and reported as one of the three major sites of p53-derived restriction of Tat mediated by PKR phosphorylation [67]. We also evaluate the frequency of several polymorphisms in Vif, Vpr, Nef and Rev previously associated with long-term non-progressors (LTNPs) HIV-1 infected subjects and differential protein function in vitro and ex vivo [39–50] (Table 4). Analysis of amino acid composition at those positions failed to detect significant differences between BCAR and BPANDEMIC datasets, except for position 77 in Vpr that showed a significantly higher prevalence of the R77Q mutation in BCAR (83%) in comparison with BPANDEMIC (48%) sequences [45]. None of the methods here employed (Geno2Pheno algorithm, the 11/25 rule, or the combination of R at position 25 of V3 and a net charge of ≥ 5) pointed out to significant difference in the frequency of CXCR4 tropic viral variants, typically associated with a more rapid disease progression [68, 69], between the BPANDEMIC and BCAR env datasets (S3 Table). Finally, our analysis of HIV-1 pol sequences from drug naïve subjects from the Caribbean also failed to detect significant differences in the prevalence of SDRMs between BCAR and BPANDEMIC datasets (S4 Table).
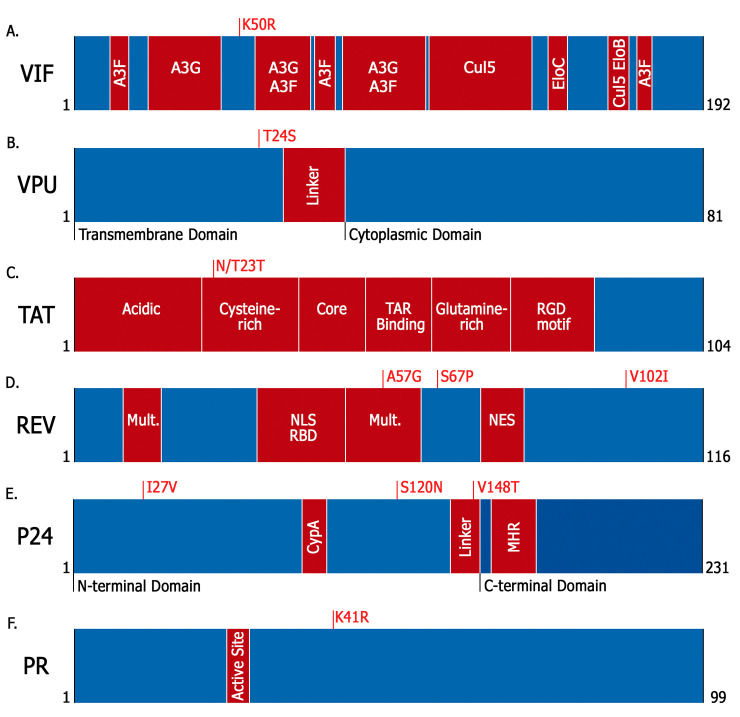
Fig 2. Mapping of the identified AAs between BCAR and BPANDEMIC on the accessory, regulatory and structural HIV-1 proteins.
For all proteins, their functional domains are represented; (A) Vif: regions responsible for the binding to APOBEC3G (A3G), APOBEC3F (A3F), Cullin 5 (Cul5), Elongin B (EloB), and Elongin C (EloC); (B) Vpu: the transmembrane domain, the cytoplasmic domain, and the linker region between them; (C) Tat: the N‐terminal acidic domain, the cysteine-rich domain, the hydrophobic core domain, the TAR binding domain, the glutamine-rich domain, and the RGD motif; (D) Rev: the RNA binding domain (RBD), that also functions as a nuclear localization signal (NLS), the nuclear exporting signal (NES), and sequences responsible for its multimerization (Mult.); (E) P24: the N-terminal domain and the C-terminal domain, connected by the inter-domain linker, the region mainly responsible for the interaction with cyclophilin A (CypA), and the major homology region (MHR); (F) Protease (PR): its active site.
Table 4. Prevalence of polymorphisms in BCAR and BPANDEMIC Vif, Vpr, Nef and Rev sequences associated with slow HIV-1 disease progression and differential function.
| Polymorphism | BCAR | BPANDEMIC | p-value | ||
|---|---|---|---|---|---|
| # seq. Analyzed |
# seq. with polymorphism (%) | # seq. analyzed |
# seq. with polymorphism (%) | ||
| RevE74P | 18 | 0 | 450 | 36 (8%) | - |
| RevL78I | 18 | 0 | 450 | 43 (10%) | - |
| RevV104L | 18 | 0 | 450 | 31 (7%) | - |
| RevS106P | 18 | 1 (6%) | 450 | 28 (6%) | 0.908 |
| VifI107T | 18 | 0 | 450 | 0 | - |
| VifR132S | 18 | 0 | 450 | 135 (30%) | - |
| VprQ3R | 18 | 3 (17%) | 450 | 42 (9%) | 0.300 |
| VprQ44R | 18 | 0 | 450 | 0 | - |
| VprF72L | 18 | 0 | 450 | 0 | - |
| VprR77Q | 18 | 15 (83%) | 450 | 218 (48%) | 0.003 |
| VprI83V | 18 | 0 | 450 | 8 (2%) | - |
| NefR22Q | 18 | 3 | 450 | 49 (11%) | |
| NefL58V | 18 | 3 | 450 | 45 (10%) | |
| NefK94E | 18 | 0 | 450 | 11 (2%) | - |
| NefH116N | 18 | 8 (44%) | 450 | 123 (27%) | 0.112 |
p-values < 0.05 are in bold.
Discussion
The current work suggests that the hypothesis that viral genetic determinants shaped the remarkable differences in the geographic dissemination pattern of the HIV-1 BPANDEMIC and BCAR strains is highly unlikely. Among over 3,000 positions analyzed across nine genes coded by the HIV-1 genome, we detected only 13 AAs distinguishing the BCAR and BPANDEMIC clades. All AAs that did differentiate the BCAR and BPANDEMIC clades correspond to sites with distinct degrees of polymorphism and not to invariant (or highly conserved) sites. Furthermore, for 11 out of 13 AAs positions, the most common amino acid found in a given subtype B variant corresponds to the second most frequent amino acid in the other variant. Our study also suggests that 11 out of 13 AAs that distinguished contemporary BCAR and BPANDEMIC sequences were probably not inherited from their ancestors and that most (19/21) amino acid differences inferred between the BCAR and BPANDEMIC ancestors evolved into polymorphic sites with quite comparable compositions in modern descendant sequences. Finally, nearly all AAs identified were located outside functionally relevant protein domains.
Our data supports that BCAR and BPANDEMIC strains are probably not distinguished by functionally relevant AAs in structural genes. Analyzes of both FL and expanded partial env sequences failed to detect AAs in this variable genomic region. Furthermore, no significant differences in the frequency of CCR5/CXCR4 tropic variants were detected between BCAR and BPANDEMIC sequences, supporting not great variation in the chemokine receptor usage between pandemic and non-pandemic subtype B strains. These results are fully consistent with a previous study that demonstrate that the env V3 consensus sequence of BCAR strains from Trinidad and Tobago differs by few amino acids from the BPANDEMIC V3 consensus and that no phenotypic features, including syncytium induction, neutralization profiles, and chemokine receptor usage, distinguish both subtype B lineages [19]. Furthermore, all AAs in Gag and Pol that distinguishing BCAR and BPANDEMIC sequences were located outside known conserved protein functional domains.
By contrasting, a few interesting differences between BCAR and BPANDEMIC strains were observed in non-structural genes. The single AAs in position 23 of Tat is located in a cysteine-rich domain and has been reported as one of the three major sites of p53-derived restriction of Tat mediated by PKR phosphorylation [67]. The presence of an N residue in that position, that is the most prevalent amino acid in subtypes A, C, D and BCAR (44%) strains, but not in BPANDEMIC ones (22%), have been associated with increased Tat transactivation, probably through enhanced P-TEFb binding [67, 70]. We also detect much higher frequency of the naturally occurring variation Vpr R77Q in BCAR (83%) respect to BPANDEMIC (48%) sequences. That mutation, that also predominates in subtypes A, C, D, G, and H, reduces apoptosis and CD4 T-cell depletion in ex vivo-infected cells and is much more prevalent in subtype B-infected LTNPs individuals (75–90%) than in subjects with progressive HIV disease (33–42%) [45]. The similar genetic composition of BCAR and several pandemic HIV-1 clades (subtypes A, C and D) at positions Tat23 and Vpr77 argued against the hypothesis that differences at such positions resulted in a more restricted dispersion of BCAR compared with the BPANDEMIC strains.
Despite the very small size (n = 18) of the BCAR FL genome dataset here used, some evidences support that the BCAR genetic variability was not severely underestimated in this dataset and that the BCAR consensus sequence obtained was probably not biased by the low number of FL genomic sequences available. First, the most common amino acid recovered in most positions from extended datasets was fully coincident with the one detected in the FL dataset. Expanded and FL datasets converged in 99.7% (297/298) of Gag amino acid positions, 99.1% (336/339) of Pol positions, and 97.8% (683/698) of Env positions analyzed. Second, the degree of polymorphism of the 13 AAs positions that distinguished BCAR and BPANDEMIC sequences was roughly comparable in FL and expanded BCAR datasets. The paucity of FL BCAR strains, however, might have restricted our ability to detect some additional AAs between BCAR and BPANDEMIC sequences. By increasing the number of BCAR sequences we failed to recover new AAs between BCAR and BPANDEMIC env sequences, but we detected four additional AAs in Gag and one in Pol, increasing the overall number of AAs from three to eight in those genomic regions.
The low number of FL BCAR sequences used might have also introduced some bias on the reconstruction of the MRCA sequences. According to our analysis, of the 13 AAs detected in modern BCAR and BPANDEMIC sequences, only two matched with divergent sites between the BCAR and BPANDEMIC ancestors. This observation support that most genetic differences between the BCAR and BPANDEMIC ancestors evolved toward positions with similar amino acid composition in modern subtype B sequences and that most AAs in modern BCAR and BPANDEMIC sequences arose during subsequent evolution and diversification of subtype B lineages. An inspection of the amino acid composition at those 13 positions in the related subtype D clade, however, supports a different scenario. We observed that BCAR and subtype D sequences displayed the same prevalent amino acid in most (11/13) AAs positions, consistent with genetic identity inherited from the common B/D ancestor. In sharp contrast, the BPANDEMIC and subtype D sequences displayed the same prevalent amino acid in only one AAs position. Therefore, our reconstruction of the MRCA sequences may have underestimated the number of BCAR/BPANDEMIC AAs inherited from the ancestors.
In summary, albeit some mutations fixed in the HIV-1 BPANDEMIC ancestral strain could potentially have some phenotypic impact on viral transmissibility, the absence of stringent AAs distinguishing modern BCAR and BPANDEMIC variants and the similar amino acid composition between BCAR and other group M subtype pandemic variants at key sites indicates that viral genetic determinants were probably not the main factor shaping the divergent pattern of geographic spread of BCAR and BPANDEMIC variants. The successful dissemination of BCAR strains in some Caribbean countries that exhibit the highest HIV-1 prevalence rates outside of Africa [6, 11] also argues against the hypothesis of a reduced BCAR viral fitness. These results support that stochastic events leading to the introduction of BPANDEMIC ancestor into globally connected populations were the most probable driving force behind its pandemic dissemination and substantiate the crucial need for continued molecular surveillance of HIV-1 transmission on key populations worldwide.
Supporting information
Partial HIV-1 sequences covering (A) gag (1,264 to 2,148), (B) env (6,450 to 8,480), (C) pol (2,253 to 3,272), and (D) pol from drug naïve individuals (2,253 to 3,272) were classified into BPANDEMIC (red branches) and BCAR (blue branches) lineages according to the topology obtained in each tree. Node support (SH-aLRT) for subtype B and BPANDEMIC monophyletic groups are indicated. The trees were rooted using HIV-1 subtype D sequences. Branch lengths are drawn to scale with the bar at the bottom indicating nucleotide substitutions per site.
(PDF)
The table summarizes the results of the classification of the full-length (FL) and partial HIV-1 subtype B sequences in the BPANDEMIC or BCAR based on their placement on ML phylogenetic trees displayed in Fig 1 and S1 Fig. All sub-datasets are accompanied by their sampling range. *Country codes are in accordance with ISO 3166–1.
(PDF)
The table summarize the full-length (FL) HIV-1 Subtype D sequences used in our study and their sampling range. * Country codes are in accordance with ISO 3166–1.
(PDF)
The table summarizes the predicted usage of chemokines receptors CCR5 and CXCR4 based on different criteria: 1) the Geno2Pheno algorithm, which classifies the sequences between R5 variants or X4 and R5X4 dual-tropic variants; 2) the 11/25 Rule, which asses the presence of arginine (R) or lysine (K) at position 11 of env V3 sequences and/or K at position 25; 3) the combination of R at position 25 of V3 and a net charge of ≥ 5.
(PDF)
The table summarizes the surveillance drug-resistance mutations (SDRM) identified in PR/RT BCAR and BPANDEMIC sequences retrieved from drug naïve subjects. PI (protease inhibitor), NRTI (nucleoside analog reverse-transcriptase inhibitors), NNRTI (non-nucleoside analog reverse-transcriptase inhibitor). The p-values obtained in chi-squared tests are listed in the last column.
(PDF)
(PDF)
Acknowledgments
We thank Dra Ana Carolina Paulo Vicente for logistic support. We are grateful for support from the Coordination for the Improvement of Higher Education Personnel (CAPES).
Data Availability
The data underlying the results presented in the study are available from Los Alamos HIV Database (http://www.hiv.lanl.gov). The authors confirm they had no special access privileges to the data. A supplementary file with the accession codes of all sequences used in this study has been provided.
Funding Statement
I.A. is funded by a fellowship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). G.B. is funded by fellowships from the Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq (Grant number 302317/2017-1) and the Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro – FAPERJ (Grant number E-26/202.896/2018). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Tebit DM, Arts EJ. Tracking a century of global expansion and evolution of HIV to drive understanding and to combat disease. Lancet Infect Dis. 2011;11(1):45–56. [DOI] [PubMed] [Google Scholar]
- 2.Hemelaar J, Elangovan R, Yun J, Dickson-Tetteh L, Fleminger I, Kirtley S, et al. Global and regional molecular epidemiology of HIV-1, 1990–2015: a systematic review, global survey, and trend analysis. Lancet Infect Dis. 2019;19(2):143–55. 10.1016/S1473-3099(18)30647-9 [DOI] [PubMed] [Google Scholar]
- 3.Faria NR, Rambaut A, Suchard MA, Baele G, Bedford T, Ward MJ, et al. HIV epidemiology. The early spread and epidemic ignition of HIV-1 in human populations. Science. 2014;346(6205):56–61. 10.1126/science.1256739 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gilbert MT, Rambaut A, Wlasiuk G, Spira TJ, Pitchenik AE, Worobey M. The emergence of HIV/AIDS in the Americas and beyond. Proc Natl Acad Sci U S A. 2007;104(47):18566–70. 10.1073/pnas.0705329104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Junqueira DM, de Medeiros RM, Matte MC, Araújo LA, Chies JA, Ashton-Prolla P, et al. Reviewing the history of HIV-1: spread of subtype B in the Americas. PLoS One. 2011;6(11):e27489 10.1371/journal.pone.0027489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cabello M, Mendoza Y, Bello G. Spatiotemporal dynamics of dissemination of non-pandemic HIV-1 subtype B clades in the Caribbean region. PLoS One. 2014;9(8):e106045 10.1371/journal.pone.0106045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bello G, Arantes I, Lacoste V, Ouka M, Boncy J, Césaire R, et al. Phylogeographic Analyses Reveal the Early Expansion and Frequent Bidirectional Cross-Border Transmissions of Non-pandemic HIV-1 Subtype B Strains in Hispaniola. Front Microbiol. 2019;10:1340 10.3389/fmicb.2019.01340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cabello M, Junqueira DM, Bello G. Dissemination of nonpandemic Caribbean HIV-1 subtype B clades in Latin America. AIDS. 2015;29(4):483–92. 10.1097/QAD.0000000000000552 [DOI] [PubMed] [Google Scholar]
- 9.Cabello M, Romero H, Bello G. Multiple introductions and onward transmission of non-pandemic HIV-1 subtype B strains in North America and Europe. Sci Rep. 2016;6:33971 10.1038/srep33971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Divino F, de Lima Guerra Corado A, Gomes Naveca F, Stefani MM, Bello G. High Prevalence and Onward Transmission of Non-Pandemic HIV-1 Subtype B Clades in Northern and Northeastern Brazilian Regions. PLoS One. 2016;11(9):e0162112 10.1371/journal.pone.0162112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bello G, Nacher M, Divino F, Darcissac E, Mir D, Lacoste V. The HIV-1 Subtype B Epidemic in French Guiana and Suriname Is Driven by Ongoing Transmissions of Pandemic and Non-pandemic Lineages. Front Microbiol. 2018;9:1738 10.3389/fmicb.2018.01738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jaffe HW, Darrow WW, Echenberg DF, O'Malley PM, Getchell JP, Kalyanaraman VS, et al. The acquired immunodeficiency syndrome in a cohort of homosexual men. A six-year follow-up study. Ann Intern Med. 1985;103(2):210–4. 10.7326/0003-4819-103-2-210 [DOI] [PubMed] [Google Scholar]
- 13.Stevens CE, Taylor PE, Zang EA, Morrison JM, Harley EJ, Rodriguez de Cordoba S, et al. Human T-cell lymphotropic virus type III infection in a cohort of homosexual men in New York City. JAMA. 1986;255(16):2167–72. [PubMed] [Google Scholar]
- 14.Quinn TC, Wawer MJ, Sewankambo N, Serwadda D, Li C, Wabwire-Mangen F, et al. Viral load and heterosexual transmission of human immunodeficiency virus type 1. Rakai Project Study Group. N Engl J Med. 2000;342(13):921–9. 10.1056/NEJM200003303421303 [DOI] [PubMed] [Google Scholar]
- 15.Alizon S, von Wyl V, Stadler T, Kouyos RD, Yerly S, Hirschel B, et al. Phylogenetic approach reveals that virus genotype largely determines HIV set-point viral load. PLoS Pathog. 2010;6(9):e1001123 10.1371/journal.ppat.1001123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Blanquart F, Wymant C, Cornelissen M, Gall A, Bakker M, Bezemer D, et al. Viral genetic variation accounts for a third of variability in HIV-1 set-point viral load in Europe. PLoS Biol. 2017;15(6):e2001855 10.1371/journal.pbio.2001855 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bertels F, Marzel A, Leventhal G, Mitov V, Fellay J, Günthard HF, et al. Dissecting HIV Virulence: Heritability of Setpoint Viral Load, CD4+ T-Cell Decline, and Per-Parasite Pathogenicity. Mol Biol Evol. 2018;35(1):27–37. 10.1093/molbev/msx246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mitov V, Stadler T. A Practical Guide to Estimating the Heritability of Pathogen Traits. Mol Biol Evol. 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cleghorn FR, Jack N, Carr JK, Edwards J, Mahabir B, Sill A, et al. A distinctive clade B HIV type 1 is heterosexually transmitted in Trinidad and Tobago. Proc Natl Acad Sci U S A. 2000;97(19):10532–7. 10.1073/pnas.97.19.10532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Collins-Fairclough AM, Charurat M, Nadai Y, Pando M, Avila MM, Blattner WA, et al. Significantly longer envelope V2 loops are characteristic of heterosexually transmitted subtype B HIV-1 in Trinidad. PLoS One. 2011;6(6):e19995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gaschen B, Kuiken C, Korber B, Foley B. Retrieval and on-the-fly alignment of sequence fragments from the HIV database. Bioinformatics. 2001;17(5):415–8. 10.1093/bioinformatics/17.5.415 [DOI] [PubMed] [Google Scholar]
- 22.Martin DP, Murrell B, Golden M, Khoosal A, Muhire B. RDP4: Detection and analysis of recombination patterns in virus genomes. Virus Evol. 2015;1(1):vev003 10.1093/ve/vev003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O. New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol. 2010;59(3):307–21. 10.1093/sysbio/syq010 [DOI] [PubMed] [Google Scholar]
- 24.Guindon S, Lethiec F, Duroux P, Gascuel O. PHYML Online—a web server for fast maximum likelihood-based phylogenetic inference. Nucleic Acids Res. 2005;33(Web Server issue):W557–9. 10.1093/nar/gki352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Anisimova M, Gascuel O. Approximate likelihood-ratio test for branches: A fast, accurate, and powerful alternative. Syst Biol. 2006;55(4):539–52. 10.1080/10635150600755453 [DOI] [PubMed] [Google Scholar]
- 26.A R. FigTree v1.4: Tree Figure Drawing Tool. Available from: http://treebioedacuk/software/figtree/. 2009.
- 27.Fu L, Niu B, Zhu Z, Wu S, Li W. CD-HIT: accelerated for clustering the next-generation sequencing data. Bioinformatics. 2012;28(23):3150–2. 10.1093/bioinformatics/bts565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Huang Y, Niu B, Gao Y, Fu L, Li W. CD-HIT Suite: a web server for clustering and comparing biological sequences. Bioinformatics. 2010;26(5):680–2. 10.1093/bioinformatics/btq003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Drummond AJ, Nicholls GK, Rodrigo AG, Solomon W. Estimating mutation parameters, population history and genealogy simultaneously from temporally spaced sequence data. Genetics. 2002;161(3):1307–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Drummond AJ, Rambaut A. BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol Biol. 2007;7:214 10.1186/1471-2148-7-214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Suchard MA, Rambaut A. Many-core algorithms for statistical phylogenetics. Bioinformatics. 2009;25(11):1370–6. 10.1093/bioinformatics/btp244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rodríguez F, Oliver JL, Marín A, Medina JR. The general stochastic model of nucleotide substitution. J Theor Biol. 1990;142(4):485–501. 10.1016/s0022-5193(05)80104-3 [DOI] [PubMed] [Google Scholar]
- 33.Drummond AJ, Ho SY, Phillips MJ, Rambaut A. Relaxed phylogenetics and dating with confidence. PLoS Biol. 2006;4(5):e88 10.1371/journal.pbio.0040088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Drummond AJ, Rambaut A, Shapiro B, Pybus OG. Bayesian coalescent inference of past population dynamics from molecular sequences. Mol Biol Evol. 2005;22(5):1185–92. 10.1093/molbev/msi103 [DOI] [PubMed] [Google Scholar]
- 35.A R, MA S, D X, AJ D. Tracer v1.6, Available from http://tree.bio.ed.ac.uk/software/tracer/ 2014.
- 36.Gouy M, Guindon S, Gascuel O. SeaView version 4: A multiplatform graphical user interface for sequence alignment and phylogenetic tree building. Mol Biol Evol. 2010;27(2):221–4. [DOI] [PubMed] [Google Scholar]
- 37.Korber B, Myers G. Signature pattern analysis: a method for assessing viral sequence relatedness. AIDS Res Hum Retroviruses. 1992;8(9):1549–60. [DOI] [PubMed] [Google Scholar]
- 38.Team RC. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; 2018. [Google Scholar]
- 39.Iversen AK, Shpaer EG, Rodrigo AG, Hirsch MS, Walker BD, Sheppard HW, et al. Persistence of attenuated rev genes in a human immunodeficiency virus type 1-infected asymptomatic individual. J Virol. 1995;69(9):5743–53. 10.1128/JVI.69.9.5743-5753.1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Churchill MJ, Chiavaroli L, Wesselingh SL, Gorry PR. Persistence of attenuated HIV-1 rev alleles in an epidemiologically linked cohort of long-term survivors infected with nef-deleted virus. Retrovirology. 2007;4:43 10.1186/1742-4690-4-43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hassaïne G, Agostini I, Candotti D, Bessou G, Caballero M, Agut H, et al. Characterization of human immunodeficiency virus type 1 vif gene in long-term asymptomatic individuals. Virology. 2000;276(1):169–80. 10.1006/viro.2000.0543 [DOI] [PubMed] [Google Scholar]
- 42.Peng J, Ao Z, Matthews C, Wang X, Ramdahin S, Chen X, et al. A naturally occurring Vif mutant (I107T) attenuates anti-APOBEC3G activity and HIV-1 replication. J Mol Biol. 2013;425(16):2840–52. 10.1016/j.jmb.2013.05.015 [DOI] [PubMed] [Google Scholar]
- 43.Somasundaran M, Sharkey M, Brichacek B, Luzuriaga K, Emerman M, Sullivan JL, et al. Evidence for a cytopathogenicity determinant in HIV-1 Vpr. Proc Natl Acad Sci U S A. 2002;99(14):9503–8. 10.1073/pnas.142313699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhao Y, Chen M, Wang B, Yang J, Elder RT, Song XQ, et al. Functional conservation of HIV-1 Vpr and variability in a mother-child pair of long-term non-progressors. Virus Res. 2002;89(1):103–21. 10.1016/s0168-1702(02)00127-2 [DOI] [PubMed] [Google Scholar]
- 45.Lum JJ, Cohen OJ, Nie Z, Weaver JG, Gomez TS, Yao XJ, et al. Vpr R77Q is associated with long-term nonprogressive HIV infection and impaired induction of apoptosis. J Clin Invest. 2003;111(10):1547–54. 10.1172/JCI16233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mologni D, Citterio P, Menzaghi B, Zanone Poma B, Riva C, Broggini V, et al. Vpr and HIV-1 disease progression: R77Q mutation is associated with long-term control of HIV-1 infection in different groups of patients. AIDS. 2006;20(4):567–74. 10.1097/01.aids.0000210611.60459.0e [DOI] [PubMed] [Google Scholar]
- 47.Caly L, Saksena NK, Piller SC, Jans DA. Impaired nuclear import and viral incorporation of Vpr derived from a HIV long-term non-progressor. Retrovirology. 2008;5:67 10.1186/1742-4690-5-67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jin SW, Alsahafi N, Kuang XT, Swann SA, Toyoda M, Göttlinger H, et al. Natural HIV-1 Nef Polymorphisms Impair SERINC5 Downregulation Activity. Cell Rep. 2019;29(6):1449–57.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Corró G, Rocco CA, De Candia C, Catano G, Turk G, Mangano A, et al. Genetic and functional analysis of HIV type 1 nef gene derived from long-term nonprogressor children: association of attenuated variants with slow progression to pediatric AIDS. AIDS Res Hum Retroviruses. 2012;28(12):1617–26. [DOI] [PubMed] [Google Scholar]
- 50.Premkumar DR, Ma XZ, Maitra RK, Chakrabarti BK, Salkowitz J, Yen-Lieberman B, et al. The nef gene from a long-term HIV type 1 nonprogressor. AIDS Res Hum Retroviruses. 1996;12(4):337–45. [DOI] [PubMed] [Google Scholar]
- 51.Lengauer T, Sander O, Sierra S, Thielen A, Kaiser R. Bioinformatics prediction of HIV coreceptor usage. Nat Biotechnol. 2007;25(12):1407–10. [DOI] [PubMed] [Google Scholar]
- 52.De Jong JJ, De Ronde A, Keulen W, Tersmette M, Goudsmit J. Minimal requirements for the human immunodeficiency virus type 1 V3 domain to support the syncytium-inducing phenotype: analysis by single amino acid substitution. J Virol. 1992;66(11):6777–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fouchier RA, Brouwer M, Broersen SM, Schuitemaker H. Simple determination of human immunodeficiency virus type 1 syncytium-inducing V3 genotype by PCR. J Clin Microbiol. 1995;33(4):906–11. 10.1128/JCM.33.4.906-911.1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hoffman NG, Seillier-Moiseiwitsch F, Ahn J, Walker JM, Swanstrom R. Variability in the human immunodeficiency virus type 1 gp120 Env protein linked to phenotype-associated changes in the V3 loop. J Virol. 2002;76(8):3852–64. 10.1128/JVI.76.8.3852-3864.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Fouchier RA, Groenink M, Kootstra NA, Tersmette M, Huisman HG, Miedema F, et al. Phenotype-associated sequence variation in the third variable domain of the human immunodeficiency virus type 1 gp120 molecule. J Virol. 1992;66(5):3183–7. 10.1128/JVI.66.5.3183-3187.1992 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Rhee SY, Gonzales MJ, Kantor R, Betts BJ, Ravela J, Shafer RW. Human immunodeficiency virus reverse transcriptase and protease sequence database. Nucleic Acids Res. 2003;31(1):298–303. 10.1093/nar/gkg100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cruz NV, Amorim R, Oliveira FE, Speranza FA, Costa LJ. Mutations in the nef and vif genes associated with progression to AIDS in elite controller and slow-progressor patients. J Med Virol. 2013;85(4):563–74. [DOI] [PubMed] [Google Scholar]
- 58.Kikuchi T, Iwabu Y, Tada T, Kawana-Tachikawa A, Koga M, Hosoya N, et al. Anti-APOBEC3G activity of HIV-1 Vif protein is attenuated in elite controllers. J Virol. 2015;89(9):4992–5001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Andrew A, Strebel K. HIV-1 Vpu targets cell surface markers CD4 and BST-2 through distinct mechanisms. Mol Aspects Med. 2010;31(5):407–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Le Noury DA, Mosebi S, Papathanasopoulos MA, Hewer R. Functional roles of HIV-1 Vpu and CD74: Details and implications of the Vpu-CD74 interaction. Cell Immunol. 2015;298(1–2):25–32. [DOI] [PubMed] [Google Scholar]
- 61.Vercruysse T, Daelemans D. HIV-1 Rev multimerization: mechanism and insights. Curr HIV Res. 2013;11(8):623–34. 10.2174/1570162x12666140307094603 [DOI] [PubMed] [Google Scholar]
- 62.Chen JH, Wong KH, Chan KC, To SW, Chen Z, Yam WC. Phylodynamics of HIV-1 subtype B among the men-having-sex-with-men (MSM) population in Hong Kong. PLoS One. 2011;6(9):e25286 10.1371/journal.pone.0025286 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Das K, Arnold E. HIV-1 reverse transcriptase and antiviral drug resistance. Part 1. Curr Opin Virol. 2013;3(2):111–8. 10.1016/j.coviro.2013.03.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Das K, Arnold E. HIV-1 reverse transcriptase and antiviral drug resistance. Part 2. Curr Opin Virol. 2013;3(2):119–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Su CT, Koh DW, Gan SK. Reviewing HIV-1 Gag Mutations in Protease Inhibitors Resistance: Insights for Possible Novel Gag Inhibitor Designs. Molecules. 2019;24(18). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Voshavar C. Protease Inhibitors for the Treatment of HIV/AIDS: Recent Advances and Future Challenges. Curr Top Med Chem. 2019;19(18):1571–98. [DOI] [PubMed] [Google Scholar]
- 67.Yoon CH, Kim SY, Byeon SE, Jeong Y, Lee J, Kim KP, et al. p53-derived host restriction of HIV-1 replication by protein kinase R-mediated Tat phosphorylation and inactivation. J Virol. 2015;89(8):4262–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Connor RI, Sheridan KE, Ceradini D, Choe S, Landau NR. Change in coreceptor use correlates with disease progression in HIV-1—infected individuals. J Exp Med. 1997;185(4):621–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Regoes RR, Bonhoeffer S. The HIV coreceptor switch: a population dynamical perspective. Trends Microbiol. 2005;13(6):269–77. 10.1016/j.tim.2005.04.005 [DOI] [PubMed] [Google Scholar]
- 70.Reza SM, Shen LM, Mukhopadhyay R, Rosetti M, Pe'ery T, Mathews MB. A naturally occurring substitution in human immunodeficiency virus Tat increases expression of the viral genome. J Virol. 2003;77(15):8602–6. 10.1128/jvi.77.15.8602-8606.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Partial HIV-1 sequences covering (A) gag (1,264 to 2,148), (B) env (6,450 to 8,480), (C) pol (2,253 to 3,272), and (D) pol from drug naïve individuals (2,253 to 3,272) were classified into BPANDEMIC (red branches) and BCAR (blue branches) lineages according to the topology obtained in each tree. Node support (SH-aLRT) for subtype B and BPANDEMIC monophyletic groups are indicated. The trees were rooted using HIV-1 subtype D sequences. Branch lengths are drawn to scale with the bar at the bottom indicating nucleotide substitutions per site.
(PDF)
The table summarizes the results of the classification of the full-length (FL) and partial HIV-1 subtype B sequences in the BPANDEMIC or BCAR based on their placement on ML phylogenetic trees displayed in Fig 1 and S1 Fig. All sub-datasets are accompanied by their sampling range. *Country codes are in accordance with ISO 3166–1.
(PDF)
The table summarize the full-length (FL) HIV-1 Subtype D sequences used in our study and their sampling range. * Country codes are in accordance with ISO 3166–1.
(PDF)
The table summarizes the predicted usage of chemokines receptors CCR5 and CXCR4 based on different criteria: 1) the Geno2Pheno algorithm, which classifies the sequences between R5 variants or X4 and R5X4 dual-tropic variants; 2) the 11/25 Rule, which asses the presence of arginine (R) or lysine (K) at position 11 of env V3 sequences and/or K at position 25; 3) the combination of R at position 25 of V3 and a net charge of ≥ 5.
(PDF)
The table summarizes the surveillance drug-resistance mutations (SDRM) identified in PR/RT BCAR and BPANDEMIC sequences retrieved from drug naïve subjects. PI (protease inhibitor), NRTI (nucleoside analog reverse-transcriptase inhibitors), NNRTI (non-nucleoside analog reverse-transcriptase inhibitor). The p-values obtained in chi-squared tests are listed in the last column.
(PDF)
(PDF)
Data Availability Statement
The data underlying the results presented in the study are available from Los Alamos HIV Database (http://www.hiv.lanl.gov). The authors confirm they had no special access privileges to the data. A supplementary file with the accession codes of all sequences used in this study has been provided.