Abstract
In recent years, many studies have been conducted to investigate the non-thermal effects of THz radiation on different organisms, but further studies are needed to fully elucidate the effects, especially on the molecular level. In this study, we explored the effects of at 3.1 THz radiation on protein expression in Escherichia coli (E. coli) using red fluorescent protein as a reporter molecule. After 8 hours of continuous THz irradiation of bacteria on LB (Luria-Bertani) solid plates at an average power of 33 mW/cm2 and 10 Hz pulse repetition frequency, we found that the plasmid copy number, protein expression and fluorescence intensity of bacteria from the irradiated area were 3.8-, 2.7-, and 3.3 times higher than in bacteria from the un-irradiated area, respectively. These findings suggest that plasmid replication changed significantly in bacteria exposed to 3.1 THz radiation, resulting in increased protein expression as evidenced by increased fluorescence intensity of the RFP reporter.
1. Introduction
Terahertz (THz) radiation denotes electromagnetic waves with a frequency ranging from 0.1 to 10 THz. Unlike X-rays, the quantum energy of THz radiation is relatively low and it is much less likely to damage living tissues. However, the frequency of terahertz waves matches the conformational oscillation of biomolecules. Therefore, it is possible that THz radiation may act upon the intra- or intermolecular interactions of biomolecules and consequently affect many physiological processes. Therefore, THz radiation is expected to have a wide range of potential biomedical and security-related applications [1,2], which has led to safety concerns and great interest in its non-thermal effects. In the past few years, many studies were conducted to investigate the non-thermal effects of THz radiation on different organisms as well as biomolecular interactions. Increasing evidences indicates that THz radiation may interfere with protein-protein interactions, homeostasis of cellular components and the stability of gene expression, which was reviewed in detail by Romanenko et al. [2]. Although no impact of terahertz radiation on primary DNA structure was found, potential genetic effects are still possible, since several studies revealed transcriptional alteration of certain genes in mouse mesenchymal stem cells, human embryonic stem cells [3], and human keratinocytes [4]. Furthermore, chromosomal perturbations such as asynchronous centromere replication and spindle disturbances were also observed in lymphocytes and human-hamster hybrid cells, respectively [5,6]. Most of these impacts of THz radiation are considered to be the result of non-thermal effects, but they are also a comprehensive outcome of THz radiation exposure since numerous protein-protein interaction, protein-DNA interaction, and DNA double-strain dynamics are involved in these processes. Therefore, the underlying mechanisms need further elucidation.
E. coli is one of the most popular model organisms in the field of genetics and molecular biology because of its short growth cycle and relatively simple and clear genetic background [7,8]. It is also a valuable model organism for the research on THz bio-effects, and several previous studies have reported the effects of THz radiation on E. coli. A very early study reported that 0.136 THz radiation inhibited the growth of E. coli in a time-dependent manner. Exposure of bacteria to the radiation for 150 and 240 minutes reduced cell growth two- and seven-fold, respectively [9]. In recent years, the impact of terahertz radiation on various E. coli stress systems was also reported [10–12]. It was found that 1.4 W/cm2 130 µm THz radiation activated the promoters of genes related to oxidative stress and copper ion homeostasis in E. coli, but did not affect the promoter of the emrR gene, which could be induced by antibiotics, protonophores, or superoxide anions. The research also demonstrated that the oxidative stress-induced katG gene promoter was most sensitive to THz radiation and the copper-ion-activated copA promoter was also activated due to THz radiation, but to a lesser extent [11]. The authors consequently concluded that THz radiation affects the homeostasis of oxidative processes. This stands to reason, since oxidative stress is commonly induced by various sources of electromagnetic radiation, but whether this is the only mechanism by which THz radiation promotes the transcription of certain genes still needs more investigation.
In this study, we used a recombinant E. coli strain harboring the pUC-rfp plasmid to investigate the effects of THz radiation on the expression of red fluorescent protein (RFP). The pUC-rfp plasmid contains a RFP reporter gene under the control of the J23100 promoter which is derived from J23119, the strongest constitutive promoter of the Anderson promoter family [13]. The results indicated that the expression of the RFP reporter gene was not influenced by oxidative stress, but changes occurred mainly at the level of transcription activity and plasmid copy number. It was therefore worth investigating whether THz radiation can affect the copy number of the pUC-rfp plasmid, since this phenomenon has never been studied.
2. Materials and methods
2.1. Terahertz exposure system
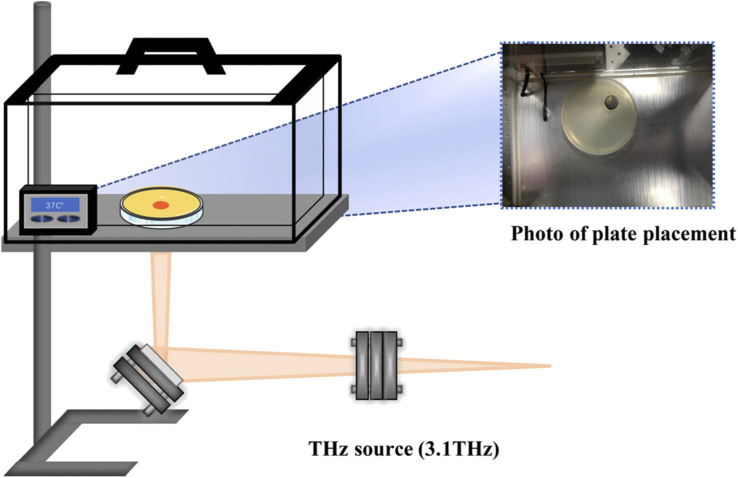
The exposure of E. coli to terahertz radiation was carried out using the terahertz free-electron laser device (CTFEL, China Academy of Engineering Physics Terahertz Free Electron Laser) which was developed by the Institute of Applied Electronics, China Academy of Engineering Physics [14]. After being emitted from the high-voltage DC GUN, the high average power high-brightness electron beam was accelerated by a CW RF superconducting accelerator. It generated a periodic oscillation using the periodic magnetic field of the undulator. With electron bunching during the oscillation process, we were able to produce coherent radiation. After that, the radiated light was reflected by the cavity mirrors and interacted with the subsequent electron beam. The electron beam delivered energy to the radiated light, which was amplified until it reached saturated output. The output THz wave was transported to the irradiation setup, which can irradiate the sample directly. All the experiments were conducted at a frequency of 3.1 THz, with a pulse width of 2 ms, a peak power per pulse of 1.6 W and a repetition rate of 10 Hz. The spot diameter was adjusted to 1 cm and the resulting average power density was 33 mW/cm2. A schematic diagram of the experimental setup is shown in Fig. 1.
Fig. 1.
Schematic diagram of the irradiation device. The exposure of E. coli DH5α (pUC-rfp) to terahertz radiation was carried out using a terahertz free electron laser device (CTFEL). The frequency was 3.1 THz, the spot diameter was 1 cm and the average power was 33mW/cm2. Terahertz radiation was released from the output hole coupled with the mirror downstream of the laser resonator, and then transmitted to the bunching collection system through the isolated vacuum environment. After bunching, terahertz radiation entered the atmospheric environment through the Mylar output window. The CTFEL radiation through each module was eventually directed with a rotary focus mirror to the sample in an incubator at 37 °C. The bacteria were exposed to continuous irradiation for 8 hours.
2.2. Cultivation of E. coli and the THz radiation exposure process
To construct the indicator strain, E. coli DH5α was transformed with the recombinant plasmid pUC-rfp which was kindly provided by Prof. Guoqiang Chen from Tsinghua University, China. The recombinant E. coli DH5α (pUC-rfp) was cultured in LB medium supplied with 100 mg/L ampicillin at 37 °C in an oscillating incubator until the OD600 reached 0.6. Then, 200 µL of culture broth was evenly spread on a solid LB agar plate according to the methodology outlined in USEPA Method 1600, and the plate was placed in a 37 °C incubator. There was a hole at the bottom of the incubator, through which the terahertz wave passed and directly touched the surface of the solid medium. The bacteria were allowed to grow for 8 hours with or without THz wave exposure, and three parallel plates were treated in the same way. An infrared camera (FLK-TEMP-62MAX-02, Fluke, U.S.) was used to monitor the temperature during the exposure. The irradiated and un-irradiated areas on the same plate were measured three times every 1h. All the chemical agents used in this study were purchased from Sinopharm (Sinopharm, China).
2.3. Confocal laser scanning microscopy
The samples were prepared according to the standard procedure used for confocal laser scanning microscopy (CLSM) analysis. Briefly, E. coli colonies from the THz radiation-exposed area and unexposed area were collected from the same plate and dispersed evenly in 50% glycerol and PBS. The cell densities of different samples were adjusted to the same level by measuring the OD600 and the appropriate dilution. Then, 10 µL samples were dropped onto slides and CLSM (Leica TCS SP8 STED 3X, Lecia, Germany) was used to observe and capture the fluorescence signal in E. coli cells. CLSM images were used to quantify the RFP fluorescence intensities in E. coli, which were calculated using the free NIH software ImageJ (https://imagej.nih.gov/ij/index.html). For each sample, 3 slides were prepared. For each slide, at least 3 microscopical fields were selected randomly for CLSM analysis. For the ImageJ analysis, the fluorescence intensities of 8 fields in each photograph were measured and the Integrated Optical Density (IOD) of the field delineated by the threshold boundary was considered to be the average fluorescence density of bacteria in this field.
2.4. Western blot analysis
Total protein was extracted from E. coli cells using RIPA lysis buffer with PMSF. The protein concentration was quantified using the BCA assay. Protein samples were separated via 12% acrylamide sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), and then transferred onto a polyvinylidene fluoride (PVDF) membrane (Millipore, Merck KGaA, USA). The blots were blocked with 5% BSA for 1 h at room temperature and incubated with the primary antibody against RFP (1:1000, Affinity, Cat#T0055) overnight at 4 °C, followed by incubation with the HRP-conjugated secondary antibody (1:5000, Abcam, Cat#ab6728) for 1 h at room temperature. ClarityTM West ECL matrix (BIO-RAD, USA) and FlorChem FC2 System (Alpha Innotech, USA) were used to detect and visualize protein bands. The relative protein levels were quantified via densitometric analysis using Quantity One software (Bio-Rad Laboratories).
2.5. Quantification of the pUC-rfp plasmid copy number
The copy number of the pUC-rfp plasmid was quantified using the qRT-PCR method. The total DNA of E. coli cells collected from the THz radiation-exposed area and the unexposed area was extracted using the TIANprep Rapid Mini Plasmid kit (QIAGEN, Cat#DP105) according to the manufacture’s instruction. The 16S rDNA gene, a highly conserved house-keeping gene on the E. coli chromosome, was used as the internal reference gene. Two sets of primers specifically complementary to the 16S rDNA gene and the rfp gene were designed and denoted as the 16S- and rfp-set, respectively. The sequences of the primers are listed in Table 1. Real-time PCR was performed using the AceQ qPCR SYBY Green Master Mix (Vazyme, Cat#Q121). The copy number of the plasmid was calculated based on the threshold cycles Ct, and the relative copy number was determined using the 2−△△Ct method and normalized to the 16S gene.
Table 1. Sequences of primers for real-time QPCR.
| Target | Primer sequence (5’- 3’) |
|---|---|
| 16S-q | Forward: AGAGTTTGATCCTGGCTCAG |
| Reverse: ACGGCTACCTTGTTACGACT | |
|
| |
| rfp | Forward: TCCCACAACGAAGACTACACC |
| Reverse: TAAGCACCGGTGGAGTGAC | |
2.6. Statistical analysis
The statistical significance of differences was analyzed using Student’s t-test in Prism 5 software (GraphPad Software, Inc., San Diego, California, USA). The data were shown as means ± SD. Differences with p-values < 0.05 were considered statistically significant.
3. Results
3.1. Exposure to terahertz radiation increased the RFP fluorescence intensity of recombinant E. coli
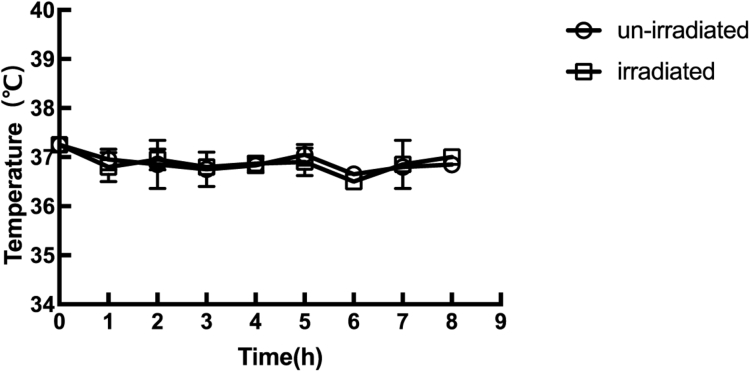
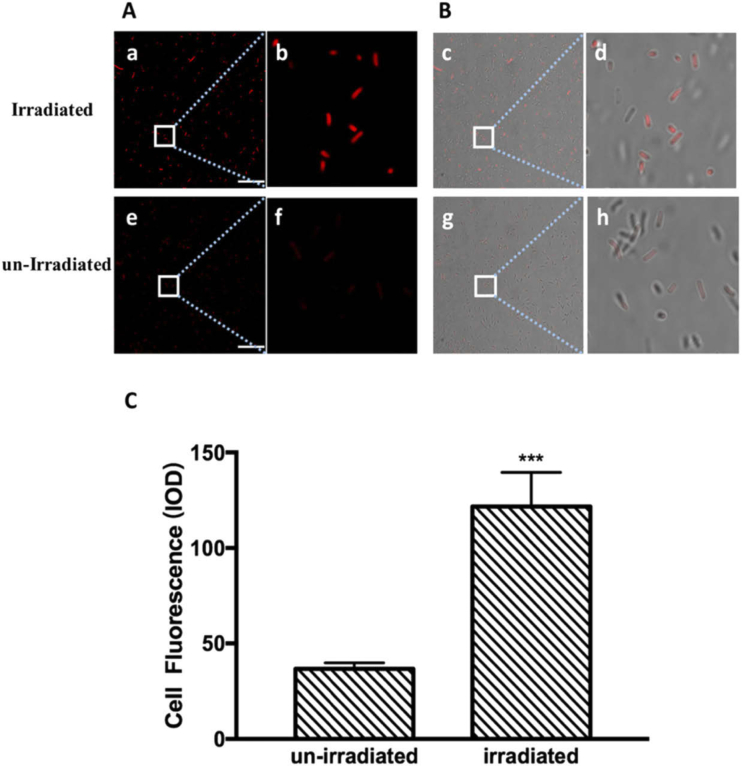
This study aimed to investigate if THz radiation has an effect on protein expression in E. coli. RFP, which was expressed under the control of the very strong constitutive J23100 promoter, was used as an indicator to differentiate the protein expression levels of irradiated and un-irradiated cells. To avoid the strong absorption of THz radiation by water, the E. coli cells were grown on solid LB plates, which were kept in an incubator. The 3.1 THz waves passed through a hole in the bottom of the incubator and directly irradiated the bacteria in the corresponding area (Fig. 1). The bacteria in the un-irradiated area served as the control sample. The bacteria were allowed to grow at 37 °C for 8 hours, and the E. coli colonies began to be visible after 6 hours of exposure both in the irradiated and un-irradiated area. During the process of irradiation, no significant temperature difference was found between the irradiated and the un-irradiated areas (Fig. 2). After the exposure, the bacteria in the irradiated area appeared more red than those in the un-irradiated area. The CLSM observation and quantitative image analysis results also indicated that the average red fluorescence intensity of individual bacterial cells in the irradiated area was about 3.3 times higher than that in the un-irradiated area (Fig. 3). This suggested that the 3.1 THz exposure increased the RFP levels in E. coli.
Fig. 2.
Temperature changes during exposure to terahertz radiation. The sample temperature was measured using an infrared camera. The hollow circles indicate the temperature of the un-irradiated area, and the hollow boxes indicate the temperature of the irradiated area.
Fig. 3.
Confocal laser scanning (CLS) micrographs showing fluorescent bacteria after 8 hours of exposure to terahertz radiation. The scale bar is 20 µm. (A) CLS images showing the red fluorescence intensity of E. coli cells after being exposed to THz radiation for 8 h. The figures (e) and (f) are un-irradiated samples; (b) and (f) are enlarged views of (a) and (e), respectively. (B) Merged images with optical micrographs of E. coli cells after exposure to THz radiation and the untreated control; (d) and (h) are enlarged views of (c) and (g), respectively. (C) Quantification of fluorescence intensity of E. coli cells after exposure to THz radiation. The data represent the means ± SD of eight independent repeats. ***p<0.001
3.2. Exposure to terahertz radiation increased the accumulation of red fluorescent protein in E. coli
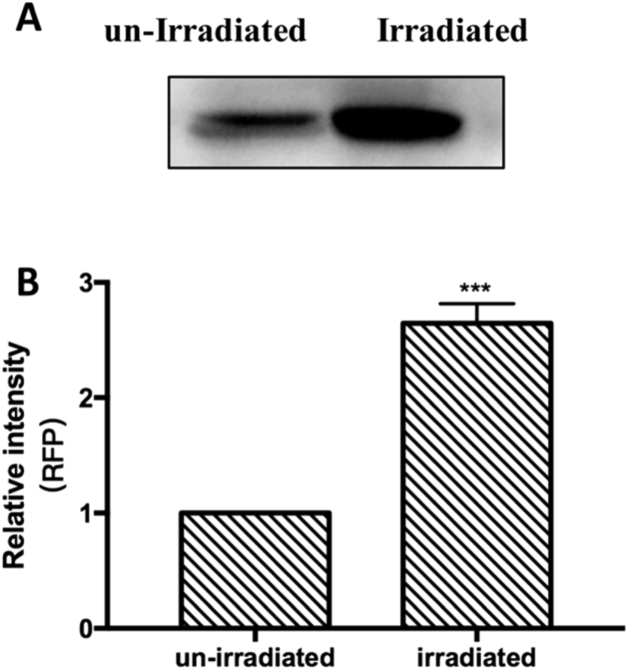
To further confirm the enhanced RFP levels in E. coli, western blot analysis was conducted to semi-quantitatively assess the amount of RFP. Whole-cell protein extracts were prepared and the concentration of each sample was measured. The same amount of total protein was loaded onto the SDS-PAGE gel and an RFP-specific primary antibody was used to identify the relative amount of RFP after electrophoresis and membrane transfer. As shown in Fig. 4, the amount of RFP in the irradiated bacteria was about 2.7 times of that in the un-irradiated bacteria, which clearly indicates that the 3.1 THz irradiation increased the total amount of RFP in transgenic E. coli. However, it was still unclear whether the enhanced accumulation of RFP was due to increased transcription from J23100 promoter, since the copy number of the RFP-coding sequence is also a crucial influencing factor.
Fig. 4.
Effects of exposure to terahertz radiation on the expression of red fluorescent protein in E. coli. (A) Protein was extracted and analyzed via western blotting with an RFP-specific antibody. The protein concentrations were measured using the BCA assay and normalized. (B) Quantitative analysis of the immunoblots. The data represent means ± SD (n = 3). *** p<0.001
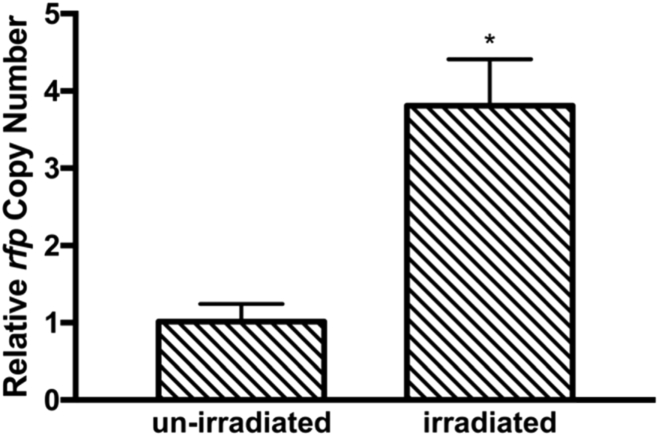
3.3. Exposure to terahertz radiation increased the copy number of the pUC-rfp plasmid in E. coli
Plasmids are often used as a vehicle to express various homologous or heterologous genes in bacterial host cells. We further analyzed the amount of pUC-rfp plasmid in E. coli with or without THz exposure by qRT-PCR, using 16S rDNA as an internal reference gene. The results demonstrated that the average copy number of the pUC-rfp plasmid in the irradiated group was 3.8 times that in the un-irradiated group (Fig. 5), which meant that there was more copies of the RFP-coding gene in bacteria exposed to the THz radiation. These results suggested that 3.1 THz irradiation might have accelerated the replication of the pUC-rfp plasmid and helped maintain its copy number at a relatively high level. This also at least largely explained the enhancement of RFP levels in the irradiated bacteria.
Fig. 5.
Effects of exposure to terahertz radiation on the plasmid copy number. Colonies were picked from the irradiated and the un-irradiated areas, respectively. The plasmid copy numbers were quantified by qPCR with corresponding primers. The data represent the means ± SD (n = 3). *p<0.05
4. Discussion
In this study, the effect of 3.1 THz radiation on the expression of RFP protein in recombinant E. coli harboring the pUC-rfp plasmid was investigated. Following irradiation for 8h, enhanced RFP fluorescence intensity and increased protein levels compared to un-irradiated cells were revealed by CLSM and Western blot analysis, respectively. The results therefore demonstrated that exposure to 3.1 THz radiation increased the expression of RFP in E. coli. The enhanced expression of RFP was mostly due to the increased copy number of the pUC-rfp plasmid, since the increase of plasmid copy number was somewhat greater than that of RFP protein levels. Whether the transcription of the rfp gene under the control of the J23100 promoter is affected still needs further study, but this study did not find evidence that the transcription activity of the J23100 promoter was enhanced. In fact, we chose the J23100 promoter because it has been reported to be the strongest constitutive promoter in the Anderson promoter family [13]. However, the effect of THz waves on the growth of E. coli is also an interesting topic. We did not observe much difference in colony formation during the exposure period, but precise quantification is still needed to fully illustrate the effects of THz waves on bacterial growth.
The copy number of a plasmid is controlled by its origin of replication (ori). The pUC-rfp plasmid used in this study was derived from the pUC plasmid, which contains a ColEl-like pMB1* ori [15] and has a high copy number of ∼75-300 copies per cell [16]. Replication of the pUC-rfp plasmid starts with the synthesis of an RNA primer precursor (RNA II), which must hybridize with its template DNA ∼555 nt upstream from the ori to become an active primer [17]. Once hybridized, it is cleaved by RNase H to produce a free 3’-OH, which is then used by DNA polymerase to initiate replication. The oligonucleotide RNA I acts as an antisense repressor of replication from this origin which folds into three adjacent hairpins, which can recognize the RNA II primer [18]. Consequently, RNA I prevents the formation of the RNA–DNA hybrid required to initiate replication [19]. Several studies have reported that terahertz radiation might affect the dynamic process and the function of DNA by perturbing the hydrogen bonds between nucleotide bases [20], producing partially unwound regions in the DNA double helix [21], and impairing the assembly of complementary DNA strands [22]. Therefore, we speculated that terahertz radiation might also promote plasmid replication by affecting the RNA-DNA and DNA-DNA interaction at the origin of replication. Bykhovski et al. analyzed the THz resonance spectrum of DNA and tRNA, and they concluded that the liquid cellular content might have greater effects on the partially paired single-strand RNA than on the fully complementary DNA, because the partially paired arrangement of RNA is more exposed to interactions with surrounding water molecules [23]. In the case of the ColE1-like origin, the inhibitory RNA I folds into three adjacent hairpin structures, while the RNA II primer hybridizes with the original DNA template strand through extensive complementary interactions. Thus, we speculated that THz radiation might attenuate the inhibitory activity of RNA I by affecting the surrounding environment as well as the structural dynamics of RNA I. By this line of reasoning, the RNA II-DNA double-stranded complex was less affected, which promoted the replication of the plasmid.
Since both transcriptional elongation and translation are required for the expression of a protein, it is also worth investigating whether THz radiation can also affect these processes. But even if the 3.1 THz radiation could affect the transcriptional and translational process, it would not only influence the expression of RFP, and is therefore beyond the scope of this study. We look forward to integrating terahertz equipment with bioanalytical equipment to facilitate in-depth research in the future and providing more powerful evidence for biological effects related to terahertz radiation. Although many issues are still awaiting further study, our results are exciting and encouraging. This study also indicates that there is a potential use for THz radiation in the bioengineering area, and provides a novel strategy to enhance both the copy number of plasmids and the expression of heterologous proteins.
5. Conclusions
In this study, we investigated the effects of 3.1 THz radiation on recombinant E. coli harboring the pUC-rfp plasmid by adopting solid medium-based bacteria cultivation which minimized the absorption of THz by water. Our results demonstrated that exposure to 3.1 THz radiation significantly enhanced the protein levels of RFP in recombinant E. coli by increasing the copy number of the pUC-rfp plasmid.
Acknowledgments
CTFEL terahertz technical equipment was provided by the Chinese Academy of Terahertz Free Electron Laser Research Team. We appreciate the THz technical support provided by Xuming Shen, Yong Xu, et al. of the China Academy of Engineering Physics. We are also grateful to Professor Guoqiang Chen and Dr. Chen Ling of Tsinghua University for providing the bacteria containing the red fluorescence plasmid. In addition, we thank Miss Ying Hao at the Instrumental Analysis Center of Xi’an Jiaotong University for her assistance with confocal laser scanning microscopy.
Funding
National Natural Science Foundation of China10.13039/501100001809 (11975218).
Disclosures
The authors declare that there are no conflicts of interest related to this article.
References
- 1.Frohlich H., “The extraordinary dielectric properties of biological materials and the action of enzymes,” Proc. Natl. Acad. Sci. U. S. A. 72(11), 4211–4215 (1975). 10.1073/pnas.72.11.4211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Romanenko S., Begley R., Harvey A. R., Hool L., Wallace V. P., “The interaction between electromagnetic fields at megahertz, gigahertz and terahertz frequencies with cells, tissues and organisms: risks and potential,” J. R. Soc., Interface 14(137), 20170585 (2017). 10.1098/rsif.2017.0585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bogomazova A. N., Vassina E. M., Goryachkovskaya T. N., Popik V. M., Sokolov A. S., Kolchanov N. A., Lagarkova M. A., Kiselev S. L., Peltek S. E., “No DNA damage response and negligible genome-wide transcriptional changes in human embryonic stem cells exposed to terahertz radiation,” Sci. Rep. 5(1), 7749 (2015). 10.1038/srep07749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Echchgadda I., Cerna C. Z., Sloan M. A., Elam D. P., Ibey B. L., “Effects of Different Terahertz Frequencies on Gene Expression in Human Keratinocytes,” Proc. SPIE 9321, 93210Q (2015). 10.1117/12.2082542 [DOI] [Google Scholar]
- 5.Korenstein-Ilan A., Barbul A., Hasin P., Eliran A., Gover A., Korenstein R., “Terahertz radiation increases genomic instability in human lymphocytes,” Radiat. Res. 170(2), 224–234 (2008). 10.1667/RR0944.1 [DOI] [PubMed] [Google Scholar]
- 6.Schrader T., Kleine-Ostmann T., Munter K., Jastrow C., Schmid E., “Spindle disturbances in human-hamster hybrid (A(L)) cells induced by the electrical component of the mobile communication frequency range signal,” Bioelectromagnetics 32(4), 291–301 (2011). 10.1002/bem.20634 [DOI] [PubMed] [Google Scholar]
- 7.Park G.-S., Convergence of Terahertz Sciences in Biomedical Systems (Springer, 2012), pp. xiii. [Google Scholar]
- 8.Wilmink G. J., Grundt J. E., “Invited Review Article: Current State of Research on Biological Effects of Terahertz Radiation,” J. Infrared, Millimeter, Terahertz Waves 32(10), 1074–1122 (2011). 10.1007/s10762-011-9794-5 [DOI] [Google Scholar]
- 9.Webb S. J., Dodds D. D., “Inhibition of bacterial cell growth by 136 gc microwaves,” Nature 218(5139), 374–375 (1968). 10.1038/218374a0 [DOI] [PubMed] [Google Scholar]
- 10.Demidova E. V., Goryachkovskaya T. N., Mescheryakova I. A., Malup T. K., Semenov A. I., Vinokurov N. A., Kolchanov N. A., Popik V. M., Peltek S. E., “Impact of Terahertz Radiation on Stress-Sensitive Genes of E.Coli Cell,” IEEE Trans. Terahertz Sci. Technol. 6(3), 435–441 (2016). 10.1109/TTHZ.2016.2532344 [DOI] [Google Scholar]
- 11.Pelteka S. E., Demidova E. V., Popikb V. M., Goryachkovskayaa T. N., “Stress-Induced Systems in Escherichia coli and Their Response to Terahertz Radiation,” Russ. J. Genet.: Appl. Res. 7(8), 858–868 (2017). 10.1134/S2079059717080019 [DOI] [Google Scholar]
- 12.Demidova E. V., Goryachkovskaya T. N., Malup T. K., Bannikova S. V., Semenov A. I., Vinokurov N. A., Kolchanov N. A., Popik V. M., Peltek S. E., “Studying the non-thermal effects of terahertz radiation on E. coli/pKatG-GFP biosensor cells,” Bioelectromagnetics 34(1), 15–21 (2013). 10.1002/bem.21736 [DOI] [PubMed] [Google Scholar]
- 13.Liow L. T., Go M. D. K., Yew W. S., “Characterisation of Constitutive Promoters from the Anderson library in Chromobacterium violaceum ATCC 12472,” Eng. Biol. 3(3), 57–66 (2019). 10.1049/enb.2018.5007 [DOI] [Google Scholar]
- 14.Li M., Yang X., Xu Z., Wang H., Xiao D., Shu X., Lu X., Huang W., Dou Y., “China’s First Tera-Hertz Free Electron Laser Oscillator,” in 2018 International Conference on Microwave and Millimeter Wave Technology (ICMMT), (IEEE, 2018), 1–3. [Google Scholar]
- 15.del Solar G., Giraldo R., Ruiz-Echevarria M. J., Espinosa M., Diaz-Orejas R., “Replication and control of circular bacterial plasmids,” Microbiol. Mol. Biol. Rev. 62(2), 434–464 (1998). 10.1128/MMBR.62.2.434-464.1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mhatre S., “Construction of a Modular Vector Library for varied gene expression in Escherichia coli,” University of the Sciences in Philadelphia; (2010). [Google Scholar]
- 17.Cesareni G., Cornelissen M., Lacatena R. M., Castagnoli L., “Control of Pmb1 Replication - Inhibition of Primer Formation by Rop Requires Rna1,” Embo J. 3(6), 1365–1369 (1984). 10.1002/j.1460-2075.1984.tb01978.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lin-Chao S., Chen W.-T., Wong T.-T., “High copy number of the pUC plasmid results from a Rom/Rop-suppressible point mutation in RNA II,” Mol. Microbiol. 6(22), 3385–3393 (1992). 10.1111/j.1365-2958.1992.tb02206.x [DOI] [PubMed] [Google Scholar]
- 19.Di Primo C., “Real time analysis of the RNAI-RNAII-Rop complex by surface plasmon resonance: from a decaying surface to a standard kinetic analysis,” J. Mol. Recognit. 21(1), 37–45 (2008). 10.1002/jmr.860 [DOI] [PubMed] [Google Scholar]
- 20.Fischer B. M., Walther M., Uhd J. P., “Far-infrared vibrational modes of DNA components studied by terahertz time-domain spectroscopy,” Phys. Med. Biol. 47(21), 3807–3814 (2002). 10.1088/0031-9155/47/21/319 [DOI] [PubMed] [Google Scholar]
- 21.Alexandrov B. S., Gelev V., Bishop A. R., Usheva A., Rasmussen KØ, “DNA Breathing Dynamics in the Presence of a Terahertz Field,” Phys. Lett. A 374(10), 1214–1217 (2010). 10.1016/j.physleta.2009.12.077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Greschner A. A., Ropagnol X., Kort M., Zuberi N., Perreault J., Razzari L., Ozaki T., Gauthier M. A., “Room-Temperature and Selective Triggering of Supramolecular DNA Assembly/Disassembly by Nonionizing Radiation,” J. Am. Chem. Soc. 141(8), 3456–3469 (2019). 10.1021/jacs.8b10355 [DOI] [PubMed] [Google Scholar]
- 23.Bykhovski A., Globus T., Khromova T., Gelmont B., Woolard D., Bykhovskaia M., “An analysis of the THz frequency signatures in the cellular components of biological agents,” Terahertz for Military and Security Applications IV 6212, 62120H (2006). 10.1117/12.665272 [DOI] [Google Scholar]