Abstract
Although it has been reported that uterine signal transducer and activator of transcription 3 (STAT3) is essential for embryo implantation, the exact roles of uterine epithelial and stromal STAT3 on embryo implantation have not been elucidated. To address this issue, we generated Stat3-floxed/Ltf-iCre (Stat3-eKO), Stat3-floxed/Amhr2-Cre (Stat3-sKO), and Stat3-floxed/Pgr-Cre (Stat3-uKO) mice to delete Stat3 in uterine epithelium, uterine stroma, and whole uterine layers, respectively. We found that both epithelial and stromal STAT3 have critical roles in embryo attachment because all the Stat3-eKO and Stat3-sKO female mice were infertile due to implantation failure without any embryo attachment sites. Stat3-eKO uteri showed indented structure of uterine lumen, indicating the role of epithelial STAT3 in slit-like lumen formation in the peri-implantation uterus. Stat3-sKO uteri exhibited hyper-estrogenic responses and persistent cell proliferation of the epithelium in the peri-implantation uterus, suggesting the role of stromal STAT3 in uterine receptivity. In addition, Stat3-uKO female mice possessed not only the characteristic of persistent epithelial proliferation but also that of indented structure of uterine lumen. These findings indicate that epithelial STAT3 controls the formation of slit-like structure in uterine lumen and stromal STAT3 suppresses epithelial estrogenic responses and cell proliferation. Thus, epithelial and stromal STAT3 cooperatively controls uterine receptivity and embryo attachment through their different pathways.
Subject terms: Disease model, Reproductive disorders, Experimental models of disease, Endocrine reproductive disorders, Infertility
Introduction
Implantation failure is a major problem of patients with infertility undergoing in vitro fertilization and embryo transfer (IVF-ET). Successful embryo implantation necessitates an intimate crosstalk between the receptive uterus and the implantation-competent blastocyst. These molecular and cellular interactions initiate when embryonic development to blastocyst stage is synchronized with the endometrium being receptive1–3. Although the molecular mechanism of uterine receptivity is not fully clarified, several molecules and pathways have been identified as key regulators of embryo implantation such as progesterone (P4) signaling, leukemia inhibitory factor (LIF), signal transducer and activator of transcription 3 (STAT3) signaling3–7.
STAT3 is a major transcription factor which transduces signals from interleukin (IL)-6 family cytokines including IL-6, IL-11, LIF, oncostatin M (OSM) and other growth factors8. LIF is crucial for embryo implantation and activates uterine STAT3 pathway5,9. Depending on the ligands and cell types, STAT3 exerts a variety of functions: for example, cell proliferation/differentiation and amplification/suppression of inflammatory responses8,10. In the process of acute and chronic inflammation, pro-inflammatory IL-6 produced by immune cells and fibroblasts activates STAT3 and induces a series of genes responsible for cell proliferation and inflammation11. At the same time, STAT3 also mediates the signal from anti-inflammatory IL-10 to suppress inflammation12. Since systemic Stat3 null mice show embryonic lethality13, a conditional knockout mouse model of STAT3 was recently established to investigate the function of STAT3 in the uterus14,15. Pgr-Cre mice show the specific expression of Cre recombinase in the whole uterine layers16, and Pgr-Cre mice were crossed with Stat3-floxed mice17 to create Stat3-deleted mice at the whole uterine layers (Stat3-floxed/Pgr-Cre (Stat3-uKO)). Stat3-uKO female mice showed infertility due to implantation failure with enhanced estrogenic responses, presenting increased expression of 17β-estradiol (E2) -responsive genes Ltf and Muc1 and persistent epithelial proliferation in the peri-implantation uteri. These findings indicate the role of uterine STAT3 in uterine receptivity by modulating E2 signaling, a critical determinant of uterine receptivity. In addition, our group recently demonstrated the key role of uterine STAT3 in uterine regeneration using Stat3-uKO mice15, suggesting that STAT3 acts on the physiological uterine reconstruction processes such as menstruation and postpartum. Thus, Stat3-uKO mice have been useful for the investigation of the role of uterine STAT3.
Uterine epithelium and stroma are two major components of the endometrium, and their interactions are involved in the process of the receptivity acquisition1,18,19. Uterine epithelium and stroma have independent and mutual functions to support uterine receptivity and embryo implantation1,18–20. Although the previous literature demonstrated the significance of uterine STAT3 in embryo implantation14, the role of epithelial and stromal STAT3 in the process of embryo implantation remains unclear. In this study, we generated Stat3-floxed/Ltf-iCre (Stat3-eKO) and Stat3-floxed/Amhr2-Cre (Stat3-sKO) mice to delete Stat3 in the uterine epithelium and stroma, respectively, and investigated the detailed functions of epithelial and stromal STAT3 during embryo implantation. Stat3-eKO and Stat3-sKO female mice were infertile due to implantation failure without any embryo attachment sites. Stat3-eKO mice compromised the formation of slit-like structure in uterine lumen which enables normal embryo attachment. In contrast, Stat3-sKO mice showed epithelial hyper-estrogenic responses and persistent proliferation which impair uterine receptivity. In addition, Stat3-uKO mice showed both compromised luminal structure and persistent epithelial proliferation. Taken together, epithelial and stromal STAT3 supports uterine receptivity and embryo attachment through the regulation of different pathways.
Results
STAT3 is activated both in the epithelium and stroma of the peri-implantation uterus
We examined spatiotemporal activation of STAT3 in the uteri of wild-type (WT) mice on days 1, 2, 3 and 4 of pregnancy. The expression of total STAT3 was observed in both uterine epithelium and stroma from days 1 to 4 of pregnancy (Supplemental Fig. S1). The immunostaining for pSTAT3 showed that the activation of STAT3 in both epithelium and stroma was higher on days 1 and 4 compared to days 2 and 3 (Fig. 1A), while the immunoreactivity of isotype control was negative (Fig. 1B). The activation patterns of STAT3 correlate the expression pattern of leukemia inhibitory factor (LIF)21, which is expressed in the uterine epitheliumon days 1 and 4 of pregnancy14,21,22 and activates uterine STAT3 pathway to induce embryo implantation in mice5,6,9.
Figure 1.
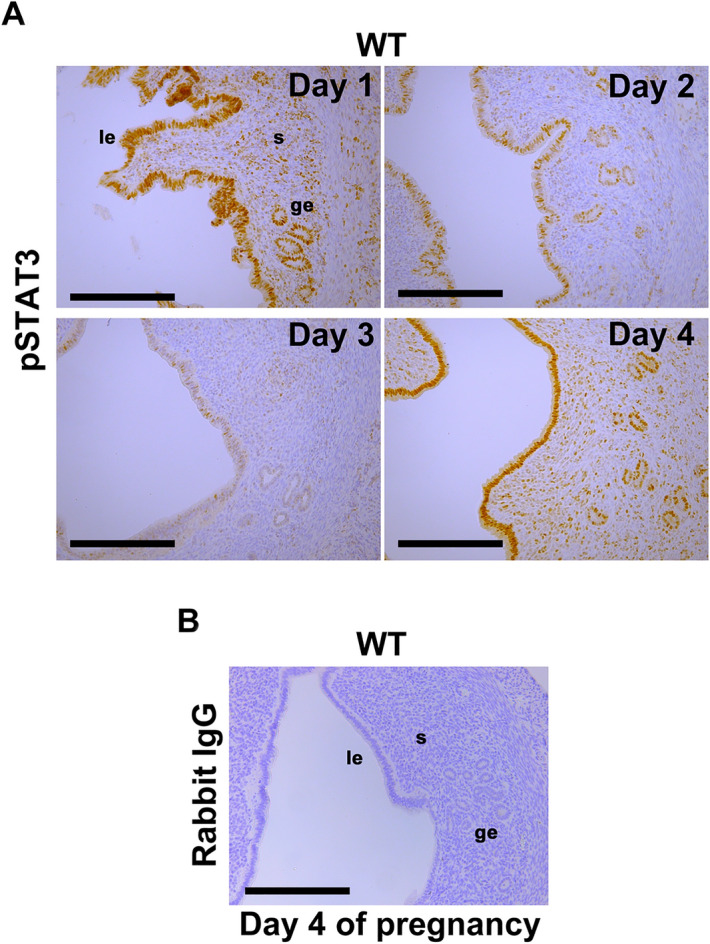
Spatiotemporal activation of STAT3 in the mouse uterus during preimplantation period. (A) Phosphorylated STAT3 (pSTAT3), the active form of STAT3, was localized in the epithelium and stroma of WT mouse uteri on days 1 and 4 of pregnancy, in contrast to the weaker immunoreactivity on day 2 and 3. (B) As an isotype control of pSTAT3 immunostaining, rabbit IgG was used. The IgG showed negative immunoreactivity in the WT mouse uterus on day 4 of pregnancy. All experiments were performed with more than three biological replicates. Scale bar = 200 μm. le, luminal epithelium; ge, glandular epithelium; s, stroma.
Mice with uterine epithelial deletion of STAT3 are infertile due to embryo attachment failure
Although the significance of STAT3 in the whole uterus in mouse embryo implantation has already been demonstrated in the former study using Stat3-uKO mice14, the specific functions of STAT3 in the epithelium and stroma is still unclear. Recently, various types of genetically-modified mice are available such as Ltf-iCre mice and Amhr2-Cre mice23,24, which have the specific expression of Cre recombinase in the luminal epithelium and the stroma. Crossing these mice with Stat3-floxed mice, we generated Stat3-deleted mice in the epithelium, stroma and all uterine layers (Stat3-eKO, Stat3-sKO and Stat3-uKO mice, respectively) to clarify the roles of epithelial and stromal STAT3 in embryo implantation.
We first investigated the reproductive phenotypes of Stat3-eKO mice. We performed immunohistochemistry for pSTAT3 and confirmed that the nuclear accumulation of activated STAT3 was clearly diminished in the epithelium of Stat3-eKO uteri (Fig. 2A, B). We analyzed the parturition phenotype of Stat3-eKO mice to examine the function of epithelial STAT3. Stat3-eKO female mice and their littermate controls (Stat3-floxed (Stat3-eCtrl) mice) were mated with WT fertile male mice. Although vaginal plugs were equally observed both in Stat3-eCtrl and Stat3-eKO mice, all Stat3-eKO mice did not delivered any pups (Fig. 2C). To identify the stage of pregnancy failure in Stat3-eKO females, both Stat3-eCtrl and Stat3-eKO females were sacrificed at the different periods during the early stages of pregnancy. Ovulation and fertilization occur on day 1, and fertilized embryo grows into blastocysts and enter the uterus on day 4 morning. Blastocysts attach to the uterine luminal epithelium from day 4 midnight to day 5 morning25. To assess the embryo development before implantation, pregnant Stat3-eCtrl and Stat3-eKO mice were sacrificed on day 4 morning and bilateral uterine horns were flushed, and the embryos retrieved from uterine lumens were observed with microscope. The numbers of normal-appearing good-quality blastocysts were comparable between Stat3-eCtrl and Stat3-eKO mice (Fig. 2D), suggesting that ovulation, fertilization and pre-implantation blastocyst growth were normal in Stat3-eKO mice. To examine whether these normal blastocysts can attach to the endometrium, Stat3-eCtrl and Stat3-eKO uteri were examined on day 5. The embryo attachment onto the luminal epithelium induces heightened stromal vascular permeability at the site of the blastocyst and can be visualized by clear blue bands along the uterus after intravenous injection of Chicago blue dye solution26. We found that while Stat3-eCtrl females had distinct blue bands, Stat3-eKO female mice did not have any blue bands (Fig. 2E, F). The unattached blastocysts were recovered from Stat3-eKO uteri without any attachment sites by uterine flushing (Fig. 2F). These results indicate that Stat3-eKO mice are infertile due to embryo attachment failure.
Figure 2.
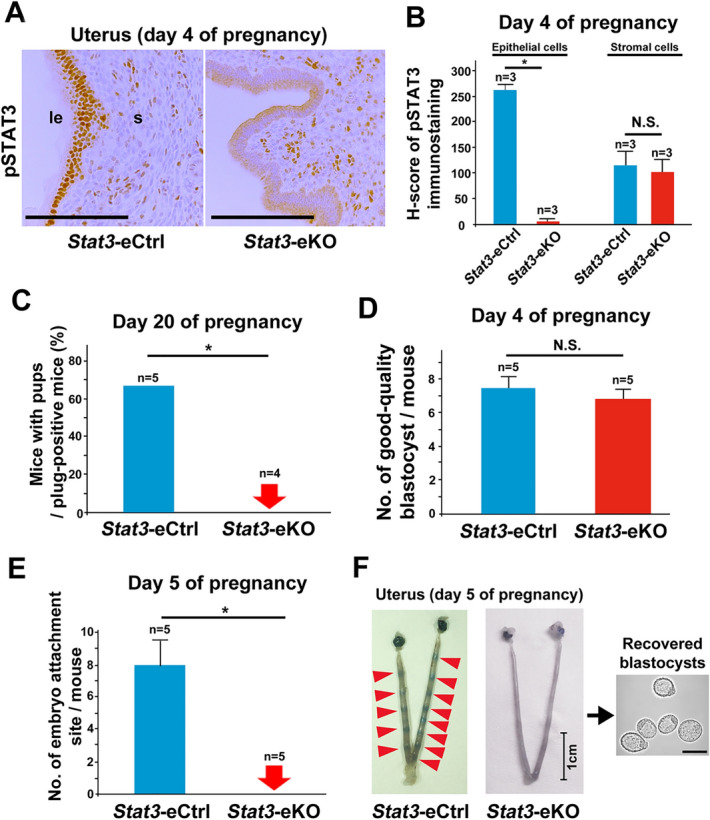
Epithelial deletion of STAT3 leads to embryo attachment failure. (A, B) pSTAT3 was efficiently deleted in the uterine epithelium of the mice with epithelial STAT3 deficiency (Stat3-eKO mice) on day 4 of pregnancy. Stat3-eCtrl means the littermate control mice. Scale bar = 100 μm. le, luminal epithelium; s, stroma. H-scores of pSTAT3 in uterine epithelium and stroma were demonstrated in (B). More than three samples obtained from different mice in each group were assessed. (C) Stat3-eKO females were infertile. Parturition of Stat3-eCtrl and Stat3-eKO mice were evaluated on day 20 of pregnancy (*, P < 0.05, Fisher’s exact probability test). (D) The number of good-quality blastocysts retrieved by uterine flushing was comparable between Stat3-eCtrl and Stat3-eKO mice on day 4 of pregnancy (P > 0.05, mean ± SEM, Student’s t test). (E, F) No embryo attachment sites were observed in Stat3-eKO uteri on day 5 of pregnancy (*, P < 0.05, mean ± SEM, Mann–Whitney U test.). The minimum, median, and maximum values in the numbers of embryo attachment sites in Stat3-eCtrl mice were 5, 7, and 13, respectively, and all the mice did not have any attachment sites in Stat3-eKO mice. In (F), the Stat3-eCtrl uterus with 13 attachment sites and the representative Stat3-eKO uterus without any attachment sites were presented. The unattached blastocysts were recovered from Stat3-eKO uteri by uterine flushing. Scale bar in the uterine pictures = 1 cm. Scale bar in the picture of the recovered embryos = 100 µm. Arrowhead, an embryo attachment site.
Mice with uterine stromal deletion of STAT3 are also infertile due to embryo attachment failure
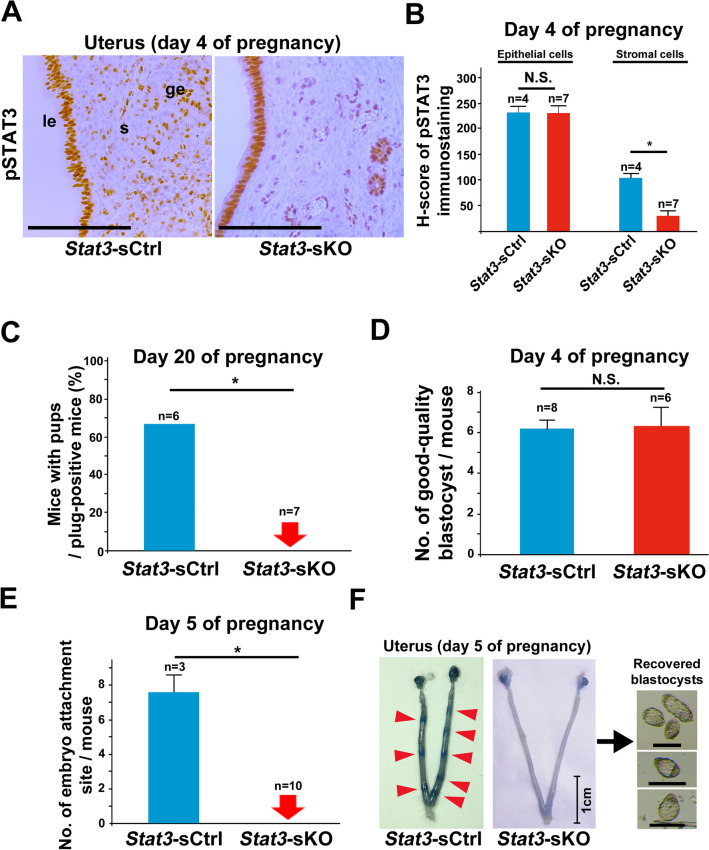
To clarify the role of uterine stromal STAT3 in pregnancy, we next examined Stat3-sKO female mice. We performed pSTAT3 immunohistochemistry and confirmed negative staining of pSTAT3 in the stroma of Stat3-sKO uteri (Fig. 3A). H-scores of pSTAT3 immunostaining showed significant inactivation of STAT3 in the stroma (Fig. 3B). We analyzed the parturition phenotype of Stat3-sKO mice. Stat3-sKO female mice and their littermate controls (Stat3-floxed (Stat3-sCtrl) mice) were mated with WT fertile males. As is the case with Stat3-eKO females, all Stat3-sKO females failed to produce offspring although vaginal plugs were normally observed (Fig. 3C). Normal-appearing good-quality blastocysts were recovered from Stat3-sKO uteri on day 4 of pregnancy (Fig. 3D), but no embryo attachment sites were detected on day 5 with the blue dye method (Fig. 3E, F). The unattached blastocysts were recovered from Stat3-sKO uteri without any attachment sites by uterine flushing (Fig. 3F). These findings indicate that Stat3-sKO mice are infertile due to embryo attachment failure. Taken together, not only epithelial but also stromal STAT3 is crucial for embryo attachment. However, uterine mRNA expressions of Lif, the inducer of embryo attachment through STAT3 activation, were normal in Stat3-eKO and Stat3-sKO mice (Supplemental Fig. S2), suggesting that the embryo attachment failure in Stat3-eKO and Stat3-sKO mice is due to the causes other than their uterine LIF expression levels.
Figure 3.
Stromal deletion of STAT3 leads to embryo attachment failure. (A, B) pSTAT3 was efficiently deleted in the uterine stroma of the mice with stromal STAT3 deficiency (Stat3-sKO mice) on day 4 of pregnancy. Stat3-sCtrl means the littermate control mice. Scale bar = 100 μm. le, luminal epithelium; ge, glandular epithelium; s, stroma. H-scores of pSTAT3 in uterine epithelium and stroma were demonstrated in (B). More than three samples obtained from different mice in each group were assessed. (C) Stat3-sKO females were infertile. Parturition of Stat3-sCtrl and Stat3-sKO mice were evaluated on day 20 of pregnancy (*, P < 0.05, Fisher’s exact probability test). (D) The number of good-quality blastocysts retrieved by uterine flushing was comparable between Stat3-sCtrl and Stat3-sKO mice on day 4 of pregnancy (P > 0.05, mean ± SEM, Student’s t test). (E, F) No embryo attachment sites were observed in Stat3-sKO uteri on day 5 of pregnancy (*, P < 0.05, mean ± SEM, Mann–Whitney U test). The minimum, median, and maximum values in the numbers of embryo attachment sites in Stat3-sCtrl mice were 6, 8, and 9, respectively, and all the mice did not have any attachment sites in Stat3-sKO mice. In Fig. 3F, the Stat3-sCtrl uterus with 8 attachment sites and the representative Stat3-sKO uterus without any attachment sites were presented. The unattached blastocysts were recovered from Stat3-sKO uteri by uterine flushing. Scale bar in the uterine pictures = 1 cm. Scale bar in the picture of the recovered embryos = 100 µm. Arrowhead, an embryo attachment site.
Stromal STAT3 suppresses excessive estrogenic responses in the peri-implantation uterus
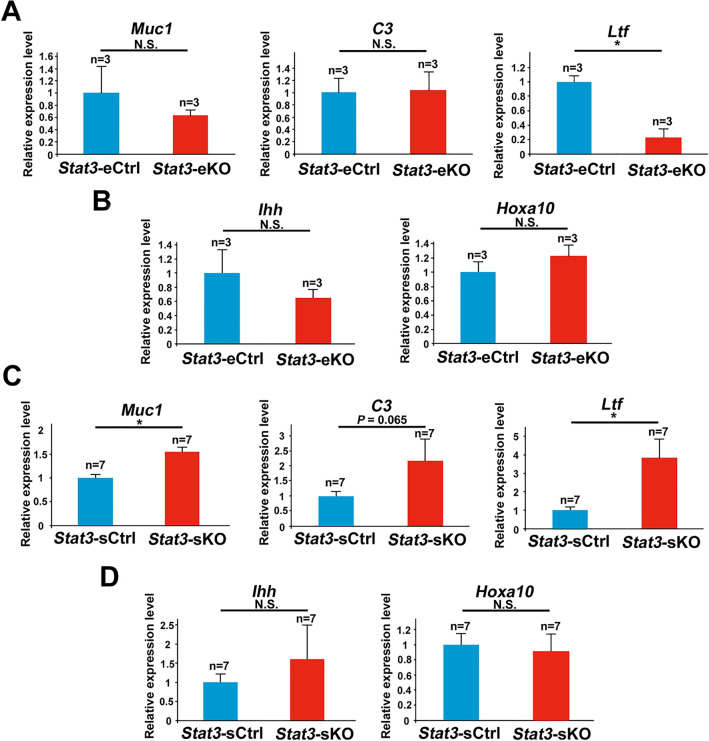
To elucidate why Stat3-eKO and Stat3-sKO females exhibit implantation failure, we examined the mRNA expression of genes regulated by 17β-estradiol (E2) and progesterone (P4). Former investigations have clarified that appropriate balance between E2 and P4 signaling is a crucial factor for uterine receptivity1–4,25,27,28. Therefore, we performed quantitative PCR and examined the expression of Muc1, C3, and Ltf as E2-responsive genes in the epithelium, Indian hedgehog (Ihh) and Hoxa10 as P4-responsive genes in the epithelium and stroma on day 4 of pregnancy. As a result, Stat3-eKO uteri did not show significant differences in the mRNA levels of neither E2-responsive genes nor P4-responsive genes compared to Stat3-eCtrl ones, except the significant suppression of Ltf in Stat3-eKO uteri. (Fig. 4A, B). This reduction of Ltf mRNA might be affected by Ltf promoter-driven Cre recombinase induction in Stat3-eKO uteri. On the other hand, Stat3-sKO uteri respectively exhibited 1.6, 2.2 and 3.9 times higher mRNA expressions of E2-responsive genes Muc1, C3, and Ltf than Stat3-sCtrl ones, while the expression levels of P4-responsive genes were unchanged in Stat3-sKO uteri compared to Stat3-sCtrl ones (Fig. 4C, D). As with Muc1 mRNA levels, the immunoreactivity of MUC1 was more intense in the luminal epithelium of Stat3-sKO mice than in that of Stat3-sCtrl mice (Supplemental Fig. S3). In addition, the immunoreactivity of E2 receptor ERα and P4 receptor PGR were normal in Stat3-eKO and Stat3-sKO uteri (Fig. 5A–D). These findings suggest that stromal STAT3 suppresses excessive estrogenic responses in the peri-implantation uterus to support uterine receptivity.
Figure 4.
Stromal deletion of STAT3 enhances estrogenic responses in the peri-implantation uterus. (A, B) The mRNA expression levels of E2 responsive genes Muc1, C3, and Ltf and P4 responsive genes Ihh and Hoxa10 in Stat3-eCtrl and Stat3-eKO uteri were examined by qPCR. (C, D) E2 responsive genes Muc1, C3 and Ltf and P4 responsive genes Ihh and Hoxa10 in Stat3-sCtrl and Stat3-sKO uteri were evaluated by qPCR (*, P < 0.05, mean ± SEM, Student’s t test). All assays were performed with more than three biological replicates derived from different mice.
Figure 5.
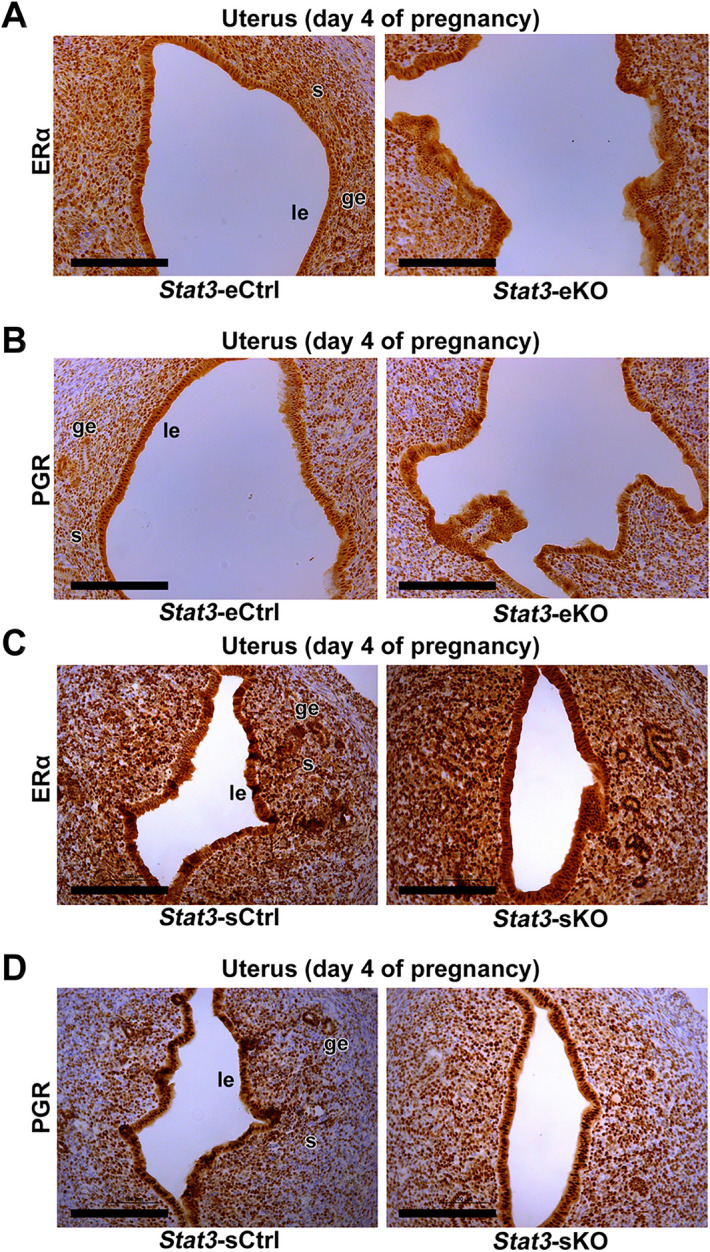
The expressions of uterine ERα and PGR are normal in Stat3-eKO and Stat3-sKO female mice. (A) The immunoreactivities of ERα were comparable in Stat3-eCtrl and Stat3-eKO uteri. (B) The immunoreactivities of PGR were comparable in Stat3-eCtrl and Stat3-eKO uteri. (C) The immunoreactivities of ERα were comparable in Stat3-sCtrl and Stat3-sKO uteri. (D) The immunoreactivities of PGR were comparable in Stat3-sCtrl and Stat3-sKO uteri. Scale bar = 200 μm. le, luminal epithelium; ge, glandular epithelium; s, stroma.
Stromal STAT3 suppresses cell proliferation in the luminal epithelium and epithelial STAT3 forms slit-like structure of uterine lumen in the peri-implantation uterus
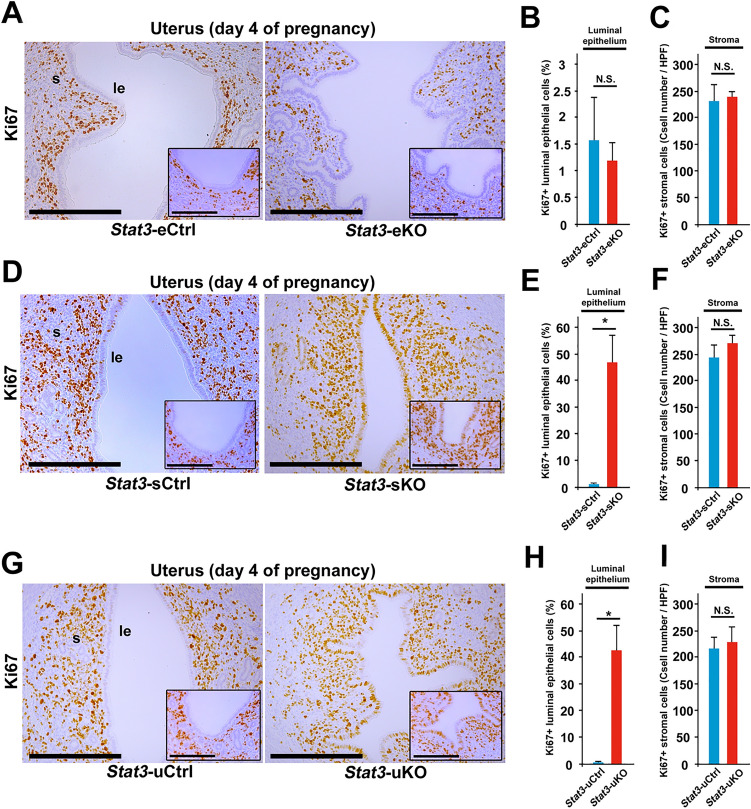
In the peri-implantation mouse uterus, the luminal epithelial cells undergo cessation of proliferation and the stromal cells have increased proliferative activity, as we call “proliferation-differentiation switching (PDS)” in embryo implantation1,3,4,6. PDS is mainly influenced by the balance of E2 and P4 signaling and is thought to be a critical determinant for uterine receptivity4,14,27,29. To investigate the detailed mechanisms of implantation failure in Stat3-eKO and Stat3-sKO mice, we next assessed PDS in Stat3-eKO and Stat3-sKO uteri by Ki67 immunostaining on day 4 of pregnancy. In Stat3-eKO uteri, most of the luminal epithelial cells were Ki67-negative, and many of the stromal cells were Ki67-posivite, which were comparable with Stat3-eCtrl uteri (Fig. 6A–C). However, in Stat3-sKO uteri, the numbers of Ki67-positive cells were significantly larger than those in Stat3-sCtrl ones (Fig. 6D–F), which might reflect the elevated E2 signaling in Stat3-sKO uteri (Fig. 4C), while the number of Ki67-positive stromal cells was unchanged between Stat3-sCtrl and Stat3-sKO uteri (Fig. 6D–F). In addition, aberrant PDS found in Stat3-sKO mice was similar to Stat3-uKO mice (Fig. 6G–I) which had hyper-estrogenic responses as described in the previous study14. Taken together, stromal STAT3 may suppress epithelial estrogenic responses and cease cell proliferation to acquire uterine receptivity and the subsequent embryo attachment.
Figure 6.
Stromal STAT3 suppresses cell proliferation of luminal epithelium and epithelial STAT3 forms slit-like structure of uterine lumen in the peri-implantation period. (A) Cessation of epithelial proliferation occurred normally but slit-like structure of uterine lumen was impaired in Stat3-eKO uteri on day 4 of pregnancy. (B, C) Percentage of Ki67-positive (Ki67 +) luminal epithelial cells and total number of Ki67 + stromal cells were unchanged between Stat3-eCtrl and Stat3-eKO uteri (P > 0.05, mean ± SEM, Student’s t test). (D) Slit-like structure of uterine lumen was normal, but persistent epithelial proliferation took place in Stat3-sKO uteri on day 4. (E) Percentage of Ki67 + luminal epithelial cells were significantly increased in Stat3-sKO uteri (*, P < 0.05, mean ± SEM, Student’s t test). (F) Total number of Ki67 + stromal cells were not significantly different between Stat3-eCtrl and Stat3-eKO uteri (P > 0.05, mean ± SEM, Student’s t test). (G) Neither slit-like structure of uterine lumen nor cessation of epithelial proliferation was observed in Stat3-uKO uteri on day 4. (H) Percentage of Ki67 + luminal epithelial cells were significantly increased in Stat3-uKO uteri (*, P < 0.05, mean ± SEM, Student’s t test). (I) Total number of Ki67 + stromal cells were not significantly different between Stat3-eCtrl and Stat3-eKO uteri (P > 0.05, mean ± SEM, Student’s t test). Scale bar = 200 μm. The antimesometrial edges of uterine lumen were shown in the inset. Scale bar in the inset = 100 µm. le, luminal epithelium; s, stroma.
To clarify the mechanisms of embryo attachment failure despite normal PDS in Stat3-eKO mice, we focused on the morphology of uterine lumen. Previous studies have revealed that the slit-like structure of uterine lumen before embryo attachment is critical for normal embryo attachment30–32. Histological sections on day 4 showed that the uterine lumen was complicated and indented in both Stat3-eKO and Stat3-uKO uteri but not in Stat3-sKO ones (Fig. 6A, D, G). These findings indicate that uterine epithelial STAT3 contributes to the formation of slit-like luminal structure, which may help successful embryo attachment. To get insight into the mechanism of inappropriate uterine luminal formation in Stat3-eKO uteri, we examined the mRNA expression of Cdh1, Wnt5a, Msx1 and Ror2, the genes associated with appropriate formation of slit-like structure in uterine lumen during peri-implantation period29,30. There were no significant differences of these gene expressions between Stat3-eCtrl and Stat3-eKO uteri (Supplemental Fig. S4), indicating the presence of unknown targets of epithelial STAT3 to control the formation of lumen structure for successful embryo implantation.
Discussion
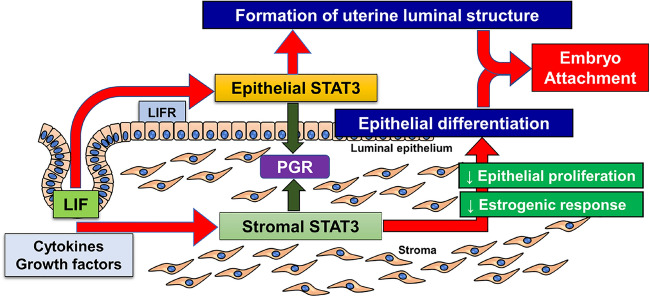
Although a previous study has reported that Stat3-uKO female mice show implantation failure due to hyper-estrogenic responses14, it has not been elucidated how uterine STAT3 is responsible for uterine receptivity. In the present study, we newly developed two lines of genetically modified mice with epithelial and stromal deletion of STAT3, and found that both epithelial and stromal STAT3 plays essential roles in uterine receptivity and embryo attachment through the different pathways. Epithelial STAT3 controls the formation of slit-like structure of the peri-implantation uterine lumen. On the other hand, stromal STAT3 suppresses E2 responsiveness and epithelial proliferation (Fig. 7).
Figure 7.
Schematic illustration of the role of uterine epithelial and stromal STAT3 in uterine receptivity and embryo attachment. Epithelial STAT3 regulates the formation of slit-like structure in uterine lumen, and stromal STAT3 suppresses proliferative activity and estrogenic response in the epithelium, thus controlling uterine receptivity and embryo attachment. PGR, progesterone receptor; LIF, leukemia inhibitory factor; LIFR, LIF receptor.
As shown in this study, STAT3 is activated in both epithelium and stroma on days 1 and 4 of pregnancy, LIF is expressed in the luminal and glandular epithelium on day 1 and expressed in the glandular epithelium on day 4, and Lif null female mice exhibit infertility due to implantation failure5. LIF receptor is localized in the luminal epithelium on day 4 of pregnancy9,14. These findings indicate that LIF is a major ligand to trigger epithelial STAT3 activation, although the precise molecular mechanisms how LIF induces embryo implantation remains to be elucidated. In contrast to epithelial STAT3 activation, the previous study showed that the stromal expression of LIF receptor (LIFR) looks faint14, implying that LIF-LIFR pathway is not responsible for the stromal STAT3 activation. This speculation may account for the current findings of different functions of epithelial and stromal STAT3.
LIFR is strongly expressed in the epithelium on day 4 of pregnancy14, suggesting that dysfunction of uterine LIF-LIFR-STAT3 signaling pathway may affect the phenotypes of Stat3-eKO mice such as indented uterine lumen and poor formation of slit-like structure. The previous studies reported that the indented luminal structure is involved in defective implantation29,30,33. These findings indicate that LIF-LIFR-STAT3 signaling in the uterine epithelium may control appropriate formation of luminal structure for successful embryo implantation.
A previous study has revealed that entire uterine deletion of Stat3 using Stat3-uKO mice causes the reduction of stromal PGR expression in the peri-implantation uterus7. However, stromal PGR expression was not decreased in both Stat3-eKO and Stat3-sKO mice in our study. These findings make us speculate that both epithelial and stromal STAT3 play a role in stromal PGR expression, and that even if either epithelial or stromal STAT3 is inactivated, stromal PGR is sufficiently maintained. In another previous study of uterine epithelial Stat3-deleted mouse model, Stat3-floxed Wnt7a-Cre (SWd/d) mice show the increased expression of E-cadherin encoded by Cdh134. Our study demonstrated that both stromal proliferation and uterine Cdh1 expression are normal in Stat3-floxed Ltf-iCre (Stat3-eKO) mice, another mouse model of uterine epithelial deletion of Stat3. There are temporal differences in epithelial Cre recombinase induction between Ltf-iCre and Wnt7a-Cre mice. Wnt7a is expressed in the uterine epithelium from fetal and neonatal stages35, while Ltf is expressed from the postpubertal stage23. These findings indicate that epithelial STAT3 in the developmental stage may suppress E-cadherin expression.
Also, a previous study has reported that Stat3-floxed/Amhr2-Cre female mice were subfertile due to resorption, although embryo implantation was normal36. In the present study, Stat3-floxed/Amhr2-Cre mice showed implantation failure. The discrepancy of the phenotypes between the previous and present studies might be derived from different Stat3-floxed mouse lines in which loxP-sites are inserted into the different loci. Therefore, it is possible that different efficiency of Stat3 deletion might cause the inconsistent phenotypes between these studies.
The present study showed that Stat3-sKO uteri have the enhanced estrogenic responses in the epithelium, suggesting the stromal-epithelial interactions through stromal STAT3. There is accumulated evidence showing that stromal transcriptional factors such as Hand2 and Hif2a affect epithelial functions to support embryo implantation37,38. In this sense, STAT3 could be another stromal transcriptional factor regulating epithelial conditions during embryo implantation.
The interactions between the uterus and embryos provide blastocyst growth and competency to embryo implantation39,40. Our study showed that neither epithelial nor stromal Stat3 deficiency affects blastocyst growth until day 4 of pregnancy. Since epithelial and stromal STAT3 may affect the intrauterine blastocyst growth and the blastocyst competency to embryo attachment from day 4 midnight to day 5, further investigations are needed to elucidate this issue.
The present study revealed that both epithelial and stromal STAT3 are critical for uterine receptivity and embryo attachment in different manners. However, it remains elusive how epithelial STAT3 regulates the formation of slit-like structure in uterine lumen, and how stromal STAT3 controls estrogen responsiveness in the epithelium. Although uterine STAT3 is involved in the uterine regeneration process15, it is also unclear whether regeneration activity of uterine STAT3 is associated with embryo implantation. Further investigations are needed to answer these questions and to clarify the association of STAT3 with recurrent implantation failure in humans.
Methods
Mice
WT (C57BL/6 N), Stat3-floxed mice (Oriental Bio Service, Kyoto, Japan)17, Ltf-iCre mice23, Amhr2-Cre mice24, and Pgr-Cre mice16 were used in this study. Ltf, Amhr2 and Pgr are expressed in the luminal epithelium, the stroma, and all layers of the endometrium, respectively. Therefore, Ltf-iCre, Amhr2-Cre and Pgr-Cre mice have the specific expression of Cre recombinase in the luminal epithelium, the stroma, and the endometrium, respectively. We generated Stat3-floxed/Ltf-iCre (Stat3-eKO), Stat3-floxed/Amhr2-Cre (Stat3-sKO), and Stat3-floxed/Pgr-Cre (Stat3-uKO) mice to deplete Stat3 specifically in the luminal epithelium, the stroma, and the endometrium, respectively. For the experiments of pregnancy, female mice were mated with fertile WT males. Day 1 of pregnancy was defined as the day when we recognized vaginal plug. These female mice were sacrificed on days 1, 2, 3, 4 and 5 for the evaluation of embryo implantation and sample collection. Embryo attachment sites were identified as blue bands under the intravenous injection of Chicago blue dye solution26. When no embryo attachment sites were detected, uterine horns were cut and flushed with normal saline to obtain embryos to confirm whether embryo development and transportation from the oviduct into the uterus were normal. All mice were housed in the University of Tokyo Animal Care Facility according to the institutional guidelines for the use of laboratory animals. All animal experiments were approved by the Institutional Animal Experiment Committee of the University of Tokyo Graduate School of Medicine (Approval number P16-066).
Immunohistochemistry
Immunostaining was performed in 10% formalin-fixed paraffin-embedded sections (6 µm), using antibodies to Ki67 (Thermo Fisher Scientific, SP6), MUC1 (Abcam, ab15481), ERα (Abcam, ab810922), PGR (Abcam, ab63605), total STAT3 (Santa Cruz, sc-8019) and phosphorylated STAT3 (phospho Y705, Abcam, ab76315). As an isotype control, rabbit IgG (Dako, IS600) was used.
H-score
The immunoreactivity of pSTAT3 and MUC1 staining was evaluated by a semiquantitative H-score41. The intensity of each cell was graded with a value of 0, 1+, 2+, or 3+ (negative, weak, moderate, or strong, respectively) in a high-power field, and then, the percentage of cells at each staining intensity level was calculated. H-score was obtained by the following equation: H-score = 1 × (% cells 1 +)+ 2 × (% cells 2 +) + 3 × (% cells 3 +) Five high-powered fields per respective section were analyzed, and the average value was assigned as the H-score for each section from different mice.
Evaluation of PDS
Ki67-positive (Ki67 +) epithelial and stromal cell number on five selected high-power fields in the uterine sections obtained from more than three different mice in each group were manually counted. PDS was assessed by the percentage of Ki67 + luminal epithelial cells and the total number of Ki67 + stromal cells.
RNA extraction quantitative PCR (qPCR)
Total RNA extraction was performed as described previously4,6,38,42,43. The complementary DNA were synthesized from the extracted RNA using ReverTra Ace qPCR RT Master Mix (TOYOBO), and, and qPCR was performed using SYBR Green PCR Master Mix (Thermo Fisher Scientific). A housekeeping gene Actb was used as an internal standard for normalizing the relative mRNA expression. Sequences of qPCR primers which were used to detect each gene are shown in Table 1.
Table 1.
Sequences of qPCR primers.
| Gene | Forward | Reverse |
|---|---|---|
| Actb | TGTTACCAACTGGGACGACA | GGGGTGTTGAAGGTCTCAAA |
| C3 | TGTTACCAACTGGGACGACA | GGGGTGTTGAAGGTCTCAAA |
| Cdh1 | AGGTTTTCGGGCACCACTTA | TGATGTTGCTGTCCCCAAGT |
| Hoxa10 | TAACTTAGCCGGAGCCTTAGGTC | CCTGATTAAACACAGCCCAGCA |
| Ihh | GAGAACACGGGTGCCGACCG | CAGCGGCCGAATGCTCAGACT |
| Lif | GCCCTGTAAATGCCACCTGT | CGACCATCCGATACAGCTCC |
| Ltf | GGAGCCTTGAGGTGTCTGAG | CCAGGTGGCACTCCTTGTAT |
| Msx1 | CCAGCCAGACGGCTGAGTC | GGACCGCCAAGAGGAAAAGA |
| Muc1 | GTGCCAGTGCCGCCGAAAGA | CCGCCAAAGCTGCCCCAAGT |
| Ror2 | GACCGGTTTGGCAAGGTCTA | GACCAGGAACTCATGGAGGT |
| Wnt5a | CATCGACTATGGCTACCGCTTC | CACTCCATGACACTTACAGGCTACA |
Statistical analysis
Statistical analyses were performed using two-tailed Student’s t test, Mann–Whitney U test, and Fisher’s exact probability test as appropriate. P values less than 0.05 were considered statistically significant.
Supplementary information
Acknowledgements
We thank Dr. Francesco J. DeMayo (National Institute of Environmental Health Sciences, Research Triangle Park, NC, USA) and Dr. John P. Lydon (Baylor College of Medicine, Houston, TX, USA) for providing Pgr-Cre mice, Dr. Sudhansu K. Dey (Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, USA) for providing Ltf-iCre mice, Dr. Richard Behringer (The University of Texas MD Anderson Cancer Center, Houston, TX, USA) for providing Amhr2-Cre mice, and Ms. Atsumi Miura for technical assistance. This work was supported by JSPS KAKENHI (Grant Nos. 19H03144, 18K19601, 19H03796, 18K19600, 18H02943, 19K16022, 19K18631, 19K18630, 20K08894) and AMED-Force (20gm4010010h0001), AMED-Wise (20gk0210021h0002), Takeda Science Foundation, and the fund of joint research with NIPRO corporation.
Author contributions
T.H. and Y.H. designed the study. T.H. and Y.H. performed experiments and collected the data. Y.F., M.G., T.K., S.A., T.H., S.A., M.M., H.H., M.S-K and Y.O. provided technical assistance for the experiments. T.H. and Y.H. analyzed the data. R.S.-H., N.T., Y.O., T.F. and Y.O. discussed and interpreted the results. T.H. and Y.H. wrote the manuscript. R.S.-H. critically reviewed the manuscript. Y.H. supervised the study.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-020-72640-0.
References
- 1.Fukui Y, et al. Uterine receptivity, embryo attachment, and embryo invasion: multistep processes in embryo implantation. Reprod. Med. Biol. 2019;18:234–240. doi: 10.1002/rmb2.12280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dey SK, et al. Molecular cues to implantation. Endocr. Rev. 2004;25:341–373. doi: 10.1210/er.2003-0020. [DOI] [PubMed] [Google Scholar]
- 3.Hirota Y. Progesterone governs endometrial proliferation–differentiation switching and blastocyst implantation. Endocr. J. 2019;66:199–206. doi: 10.1507/endocrj.EJ18-0431. [DOI] [PubMed] [Google Scholar]
- 4.Haraguchi H, et al. MicroRNA-200a locally attenuates progesterone signaling in the cervix, preventing embryo implantation. Mol. Endocrinol. 2014;28:1108–1117. doi: 10.1210/me.2014-1097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stewart CL, et al. Blastocyst implantation depends on maternal expression of leukaemia inhibitory factor. Nature. 1992;359:76–79. doi: 10.1038/359076a0. [DOI] [PubMed] [Google Scholar]
- 6.Matsuo M, et al. Levonorgestrel inhibits embryo attachment by eliminating uterine induction of leukemia inhibitory factor. Endocrinology. 2019;161:bqz005. doi: 10.1210/endocr/bqz005. [DOI] [PubMed] [Google Scholar]
- 7.Lee JH, et al. Signal transducer and activator of transcription-3 (Stat3) plays a critical role in implantation via progesterone receptor in uterus. FASEB J. 2013;27:2553–2563. doi: 10.1096/fj.12-225664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hillmer EJ, Zhang H, Li HS, Watowich SS. STAT3 signaling in immunity. Cytokine Growth Factor Rev. 2016;31:1–15. doi: 10.1016/j.cytogfr.2016.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cheng JG, Chen JR, Hernandez L, Alvord WG, Stewart CL. Dual control of LIF expression and LIF receptor function regulate Stat3 activation at the onset of uterine receptivity and embryo implantation. Proc. Natl. Acad. Sci. U S A. 2001;98:8680–8685. doi: 10.1073/pnas.151180898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Taniguchi K, et al. A gp130-Src-YAP module links inflammation to epithelial regeneration. Nature. 2015;519:57–62. doi: 10.1038/nature14228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kaur S, Bansal Y, Kumar R, Bansal G. A panoramic review of IL-6: structure, pathophysiological roles and inhibitors. Bioorg. Med. Chem. 2020;28:115327. doi: 10.1016/j.bmc.2020.115327. [DOI] [PubMed] [Google Scholar]
- 12.Schmetterer KG, Pickl WF. The IL-10/STAT3 axis: contributions to immune tolerance by thymus and peripherally derived regulatory T-cells. Eur. J. Immunol. 2017;47:1256–1265. doi: 10.1002/eji.201646710. [DOI] [PubMed] [Google Scholar]
- 13.Takeda K, et al. Targeted disruption of the mouse Stat3 gene leads to early embryonic lethality. Proc. Natl. Acad. Sci. U S A. 1997;94:3801–3804. doi: 10.1073/pnas.94.8.3801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sun X, Bartos A, Whitsett JA, Dey SK. Uterine deletion of Gp130 or Stat3 shows implantation failure with increased estrogenic responses. Mol. Endocrinol. 2013;27:1492–1501. doi: 10.1210/me.2013-1086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hiraoka, T. et al. STAT3 accelerates uterine epithelial regeneration in a mouse model of decellularized uterine matrix transplantation. JCI Insight1 (2016). [DOI] [PMC free article] [PubMed]
- 16.Soyal SM, et al. Cre-mediated recombination in cell lineages that express the progesterone receptor. Genesis. 2005;41:58–66. doi: 10.1002/gene.20098. [DOI] [PubMed] [Google Scholar]
- 17.Takeda K, et al. Stat3 activation is responsible for IL-6-dependent T cell proliferation through preventing apoptosis: generation and characterization of T cell-specific Stat3-deficient mice. J. Immunol. 1998;161:4652–4660. [PubMed] [Google Scholar]
- 18.Kelleher AM, et al. Forkhead box a2 (FOXA2) is essential for uterine function and fertility. Proc. Natl. Acad. Sci. U S A. 2017;114:E1018–E1026. doi: 10.1073/pnas.1618433114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang X, et al. SOX17 regulates uterine epithelial-stromal cross-talk acting via a distal enhancer upstream of Ihh. Nat. Commun. 2018;9:4421. doi: 10.1038/s41467-018-06652-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yuan J, et al. Primary decidual zone formation requires Scribble for pregnancy success in mice. Nat. Commun. 2019;10:5425. doi: 10.1038/s41467-019-13489-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Song H, Lim H, Das SK, Paria BC, Dey SK. Dysregulation of EGF family of growth factors and COX-2 in the uterus during the preattachment and attachment reactions of the blastocyst with the luminal epithelium correlates with implantation failure in LIF-deficient mice. Mol. Endocrinol. 2000;14:1147–1161. doi: 10.1210/mend.14.8.0498. [DOI] [PubMed] [Google Scholar]
- 22.Bhatt H, Brunet LJ, Stewart CL. Uterine expression of leukemia inhibitory factor coincides with the onset of blastocyst implantation. Proc. Natl. Acad. Sci. U S A. 1991;88:11408–11412. doi: 10.1073/pnas.88.24.11408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Daikoku T, et al. Lactoferrin-iCre: a new mouse line to study uterine epithelial gene function. Endocrinology. 2014;155:2718–2724. doi: 10.1210/en.2014-1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jamin SP, Arango NA, Mishina Y, Hanks MC, Behringer RR. Requirement of Bmpr1a for Mullerian duct regression during male sexual development. Nat. Genet. 2002;32:408–410. doi: 10.1038/ng1003. [DOI] [PubMed] [Google Scholar]
- 25.Wang H, Dey SK. Roadmap to embryo implantation: clues from mouse models. Nat. Rev. Genet. 2006;7:185–199. doi: 10.1038/nrg1808. [DOI] [PubMed] [Google Scholar]
- 26.Das SK, et al. Heparin-binding EGF-like growth factor gene is induced in the mouse uterus temporally by the blastocyst solely at the site of its apposition: a possible ligand for interaction with blastocyst EGF-receptor in implantation. Development. 1994;120:1071–1083. doi: 10.1242/dev.120.5.1071. [DOI] [PubMed] [Google Scholar]
- 27.Tranguch S, et al. FKBP52 deficiency-conferred uterine progesterone resistance is genetic background and pregnancy stage specific. J. Clin. Invest. 2007;117:1824–1834. doi: 10.1172/JCI31622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hirota Y, et al. Uterine FK506-binding protein 52 (FKBP52)-peroxiredoxin-6 (PRDX6) signaling protects pregnancy from overt oxidative stress. Proc. Natl. Acad. Sci. U S A. 2010;107:15577–15582. doi: 10.1073/pnas.1009324107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Daikoku T, et al. Conditional deletion of Msx homeobox genes in the uterus inhibits blastocyst implantation by altering uterine receptivity. Dev. Cell. 2011;21:1014–1025. doi: 10.1016/j.devcel.2011.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cha J, et al. Appropriate crypt formation in the uterus for embryo homing and implantation requires Wnt5a-ROR signaling. Cell Rep. 2014;8:382–392. doi: 10.1016/j.celrep.2014.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Arora R, et al. Insights from imaging the implanting embryo and the uterine environment in three dimensions. Development. 2016;143:4749–4754. doi: 10.1242/dev.144386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yuan J, et al. Tridimensional visualization reveals direct communication between the embryo and glands critical for implantation. Nat. Commun. 2018;9:603. doi: 10.1038/s41467-018-03092-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yuan J, et al. Planar cell polarity signaling in the uterus directs appropriate positioning of the crypt for embryo implantation. Proc. Natl. Acad. Sci. U S A. 2016;113:E8079–E8088. doi: 10.1073/pnas.1614946113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pawar S, et al. STAT3 regulates uterine epithelial remodeling and epithelial–stromal crosstalk during implantation. Mol. Endocrinol. 2013;27:1996–2012. doi: 10.1210/me.2013-1206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Parr BA, McMahon AP. Sexually dimorphic development of the mammalian reproductive tract requires Wnt-7a. Nature. 1998;395:707–710. doi: 10.1038/27221. [DOI] [PubMed] [Google Scholar]
- 36.Robker RL, et al. Identification of sites of STAT3 action in the female reproductive tract through conditional gene deletion. PLoS ONE. 2014;9:e101182. doi: 10.1371/journal.pone.0101182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li Q, et al. The antiproliferative action of progesterone in uterine epithelium is mediated by Hand2. Science. 2011;331:912–916. doi: 10.1126/science.1197454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Matsumoto L, et al. HIF2alpha in the uterine stroma permits embryo invasion and luminal epithelium detachment. J. Clin. Invest. 2018;128:3186–3197. doi: 10.1172/JCI98931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Paria BC, Huet-Hudson YM, Dey SK. Blastocyst's state of activity determines the "window" of implantation in the receptive mouse uterus. Proc. Natl. Acad. Sci. U S A. 1993;90:10159–10162. doi: 10.1073/pnas.90.21.10159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hamatani T, et al. Global gene expression analysis identifies molecular pathways distinguishing blastocyst dormancy and activation. Proc. Natl. Acad. Sci. U S A. 2004;101:10326–10331. doi: 10.1073/pnas.0402597101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hirota Y, et al. Deficiency of immunophilin FKBP52 promotes endometriosis. Am. J. Pathol. 2008;173:1747–1757. doi: 10.2353/ajpath.2008.080527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Egashira M, et al. F4/80+ macrophages contribute to clearance of senescent cells in the mouse postpartum uterus. Endocrinology. 2017;158:2344–2353. doi: 10.1210/en.2016-1886. [DOI] [PubMed] [Google Scholar]
- 43.Haraguchi H, et al. Mdm2-p53-SF1 pathway in ovarian granulosa cells directs ovulation and fertilization by conditioning oocyte quality. FASEB J. 2019;33:2610–2620. doi: 10.1096/fj.201801401R. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.