Abstract
This research analyses the performance of bacteria-assisted phytoremediation of aluminium (Al)-contaminated soil using native Indonesian plants namely, Scirpus grossus and Thypa angustifolia. A range finding test (RFT) was carried out for 14 days to obtain the tolerable Al concentration for both plants. A total of 2% and 5% (v/v) of Vibrio alginolyticus were bioaugmented during the 28-day phytoremediation test to enhance the overall Al removal. Result of the RFT showed that both plants can tolerate up to 500 mg/kg Al concentration. The addition of V. alginolyticus to the reactors resulted in a significant increment of Al removal from the contaminated soil (p < 0.05). Such addition of V. alginolyticus increased the Al removal by up to 14.0% compared with that without-bacteria addition. The highest Al removal was obtained for S. grossus with 5% V. alginolyticus with an efficiency of 35.1% from 500 mg/kg initial concertation. T. angustifolia with 500 mg/kg initial concentration showed the highest removal of 26.2% by the addition of 5% V. alginolyticus. The increase of Al removal by the bioaugmentation of V. alginolyticus was due to the interaction in the plant's rhizosphere. Exudates of both plants provided a good environment for bacteria to live in the root area. Meanwhile, the bacteria increased the bioavailability of Al to be further extracted by plants. Certain mechanisms, such as rhizostabilisation, phytostimulation and phytoextraction, were considered to be the main processes that occurred during the treatment. S. grossus and T. angustifolia displayed promising ability to act as Al hyperaccumulators with bioaccumulation factor values up to 5.308 and 3.068, respectively. Development of the design of the ex-situ soil phytoremediation reactors is suggested as a future research direction because it can significantly enhance the current obtained finding.
Keywords: Environmental science, Soil pollution, Environmental engineering, Waste treatment, Green engineering, Environmental hazard, Aluminum, Free-flow surface, Hyperaccumulator, Phytotechnology, Pollution, Remediation
Environmental science; Soil pollution; Environmental engineering; Waste treatment; Green engineering; Environmental hazard; Aluminum; Free-flow surface; Hyperaccumulator; Phytotechnology; Pollution; Remediation.
1. Introduction
Aluminium (Al) is widely used in kitchen equipment, building construction material, refining industry and recycling activities. Al refining produces various types of waste, namely, skimming wastewater, ash/dross, slag and various types of impurities (Tsakiridis, 2012). The extensive use of Al has resulted in the emergence of wastes with critically dangerous effects on the environment (Supriyanto, 2009). Al inhibits the growth of plant roots (Krstic et al., 2012) and causes various diseases, such as Alzheimer's disease, amyotrophic lateral sclerosis and Parkinson's dementia, in humans (Exley, 2017; Mirza et al., 2017; Pogue and Lukiw, 2016). One example of the use of Al with harmful effects to the environment occurs in Sumobito District, Jombang Regency, East Java, Indonesia. The Al recycling industry in Sumobito District, Jombang Regency has been processing metal waste for more than 30 years (BLH Jombang, 2016). This industrial area is located close to the paddy fields and residential areas, thereby causing negative effects in the form of health and environmental disturbances (Laksono and Muzayanah, 2012). This Al recycling industry recycles slag, ash/dross and foil into economically valuable Al plates/bars. On the basis of the nature of the material category, slag, ash/dross and foil are classified as hazardous and toxic waste (HW). Accordingly, Al recycling industrial activities are classified as HW utilisation activities or businesses.
The concentrations of Al in the soil of four different locations near the industrial area of Al recycling were 35,639, 49,967, 32,916 and 23,680 mg/kg (BLH Jombang, 2016). The sampling locations were former HW dumping sites in the form of ash/dross from the Al recycling industry. The recycling industry that uses slag, ash/dross and Al foil as raw materials must implement proper waste management to reduce its negative effect that will contribute to environmental pollution and health problems. The solid waste management of the HW recycling industry is regulated under Indonesian Government Regulation No. 101 of 2014 on Hazardous Waste Management (Purwanti et al., 2018a). The residual liquid waste from this industrial activity was directly discharged into the water body without any proper treatment. Impure materials were also openly dumped in areas surrounding the industrial location. Al in environment may cause potential damages, such as toxic effects on aquatic biota, decreased soil fertility and disruption of the enzymatic activity of organisms (Krstic et al., 2012; Setiadi and Anira, 2015; Zhang and Zhou, 2005). The pollution of soil requires human intervention with the existing technology to overcome pollution (Imron et al., 2020; Menon et al., 2019). Efforts were initiated to process Al-contaminated soils by using several methods, namely, precipitation (Kurniawan et al., 2018), evaporation, ion exchange and electrolysis (Manikandan et al., 2020; Rezaee et al., 2017b, 2017a). However, some of the above-mentioned technologies have caused further issues from the chemical residue and the used energy.
Phytoremediation is a technique used to restore polluted environmental media by degrading or stabilising contaminants using plants (Lae et al., 2019; Sarwoko and Samudro, 2006). This mechanism is a sustainable and inexpensive process and a quickly developing alternative; thus, it is highly suitable for applications to remediate contaminated medium (Ghosh et al., 2006; Ismail et al., 2015). Phytoremediation features many advantages, such as environmental friendliness, easy application and low-cost maintenance, good efficiency, high aesthetic value and applicability to a variety of pollutants. Moreover, phytoremediation can be easily implemented in situ, addition to its low cost (Aransiola et al., 2013; Imron et al., 2019b).
In the case of Al-contaminated soil, the plants of Scirpus grossus and Typha angustifolia can be used as remediating agents. S. grossus is a type of plant that shows excellent potential as a heavy metal hyperaccumulator (Ismail et al., 2019; Tangahu et al., 2010). T. angustifolia is a grass with high resistance and plays an active and passive role in eliminating pollutants (Ahmad et al., 2017; Khan et al., 2000). Ismail et al. (2017) reported that S. grossus can accumulate up to 8864 mg Al/kg plant weight with an Al concentration of 100–150 mg/L. Gallon et al. (2004) also reported that the genus of Thypa sp. had a potential in Al accumulation in their root area. Research on Al phytoremediation using Indonesian native plants is currently scarce. Bioaugmentation of bacteria to assist the phytoremediation of contaminated soil was proven to increase the pollutant removal efficiency (Ismail et al., 2019; Lebeau et al., 2011; Ojuederie and Babalola, 2017). Vibrio alginolyticus is a gram-negative bacteria and can remove Al up to 59.72% from 100 mg/L initial concentration (Purwanti et al., 2019b; Titah et al., 2019). However, the effect of the addition of this species to enhance the phytoremediation of Al by using S. grossus and T. angustifolia is yet to be explored. On the basis of this background, this research aims to analyse the performance of S. grossus and T. angustifolia in treating Al-contaminated soil. Moreover, this work aimed to study the effect of Al-resistant bacteria (V. alginolyticus) addition to the performance of Al removal. This paper covers the propagation of plants, growth rate measurement, range finding test (RFT) of contaminated soil to the used plants and bioaugmentation of V. alginolyticus in phytoremediation of Al-contaminated soil processes. The workflow is presented in Figure 1. The study result is expected to contribute in the field of alternative technology for Al-contaminated soil treatment.
Figure 1.

Research workflow.
2. Materials and methods
2.1. Source of the plants
The plants used in this study were brought from the location where S. grossus and T. angustifolia naturally thrive in large numbers. Both plants grow in habitats with soil conditions submerged in water, such as in swamps and riverbanks. S. grossus was obtained from the riverside area in Semut Kali, Bongkaran, Surabaya, Indonesia. Meanwhile, T. angustifolia was obtained from the riverside area in Rungkut Kidul, Surabaya, Indonesia.
2.2. Plant propagation
Plant propagation was conducted to multiply the plants needed for research. In the propagation stage, S. grossus and T. angustifolia were planted in 40 L square-shaped plastic reactors in the garden soil and routinely watered twice a day with tap water (Al concentration of 0.0098 mg/L). The characteristics of the used soil were as follows: pH 6.5, dark brown colour, 1.55 g/cm3 of bulk density and 0.33 mL/g of water retention capacity. The propagation stage was performed under a greenhouse condition (temperature of 33 °C–35 °C) to obtain a second generation of S. grossus and T. angustifolia (Tong et al., 2004). This stage also aimed to determine the growth rate of S. grossus and T. angustifolia. The propagation phase was conducted for 1 month.
2.3. Growth rate measurement
The growth rates of S. grossus and T. angustifolia were monitored to determine the optimal plant age to be used in this study. The plant developments in terms of the length and the number of leaves were measured once every 2 days (Tangahu, 2016). Physical observation is a simple analysis. However, this mechanism is reliable when observing the growth of plants during experiments compared with other methods (Tangahu, 2016). Biochemical characterisation and pigment analyses can also be performed to observe the plant growth rate. However, such analyses are time consuming and require chemical reagents. The plant age used for the next stage was chosen by considering the generative phase to allow the maximum degradation or stabilisation of pollutants by the plants.
2.4. Plant acclimatisation
Plant acclimatisation aims to allow plants to adapt to the specific conditions and the medium used in the RFT and phytoremediation test (Anjum, 2015; Kozai, 1991). The acclimatisation phase was conducted for 7 days by using garden soil medium without additional pollutants and watered with tap water (Al concentration of 0.0098 mg/L). In this condition, the plants were expected to attain the characteristics of flourishing with healthy plants used in the RFT and phytoremediation test.
2.5. Range finding test (RFT)
The range finding test was performed to determine the ability of plants to survive at certain contaminated soil concentrations (Purwanti et al., 2018b; Tangahu et al., 2010). At this stage, S. grossus and T. angustifolia were exposed to various concentrations of Al. The reactor used was a 5 L plastic bucket containing 5 kg soil under a free-surface flow system planted with four plants. The plants used in the RFT stage were obtained from the acclimatisation stage. The concentrations of Al in soil used were 10,000, 5000, 500, 50 and 0 mg/kg (control) prepared using the spike method of AlCl3 (SAP, Indonesia) solution with soil medium. At each concentration, the plant morphology (plant height) was also observed daily. The RFT was carried out for 14 days. If the plants died or withered within this time span, then the concentration was considered high for plants to tolerate. Thus, the lethal concentrations were excluded in the next research phase (phytoremediation stage). The concentrations that can be tolerated by plants (showing no harmful effect to plants) were used in the following phytoremediation stage.
2.6. Phytoremediation of Al
The phytoremediation stage of the Al-polluted soil was carried out with a batch system. At this stage, 40 L plastic reactors filled with 10 kg garden soil were used. The garden soil used was dried and then sieved to obtain the uniform size. The different concentrations of Al-contaminated soil were prepared using the spike method of soil + Al solution. A 10,000 mg/L AlCl3 solution stock was mixed with soil medium under a certain ratio to obtain the desired concentration (Ismail et al., 2015). The mixture was then settled for 1 week to allow the Al to be completely spiked inside the soil matrix.
S. grossus and T. angustifolia, which had been acclimatised, were planted inside each reactor. Eight plants (single species) were used in the phytoremediation test for each reactor. The number of plants used in the phytoremediation test was calculated by upscaling the number of plants from the RFT stage. Hence, the load received by plants in the phytoremediation test was similar to that in the RFT stage. This research was conducted for 28 days with threefold replications to obtain data in triplicates. During the research, the pH was measured with a portable pH meter (OneMed, Indonesia). Approximately 1 g of soil sample was taken from the middle depth of the sand medium height (6 cm) from each reactor in the beginning and the end of the test period. Al was extracted from soil and plant with a strong acid mixture of Aqua Regia (1:3 of HNO3 and HCl) (Hseu et al., 2002). The Al measurements were conducted with an ICP-OES (Fisher, USA) in accordance with the USEPA 6010 standard.
2.7. Bioaugmentation of Al-resistant bacteria
Bacterial species used in this experiment were the gram-negative bacteria of V. alginolyticus. Titah et al. (2019) showed that this species can remove Al from wastewater via a biosorption mechanism. The inoculum of bacteria was prepared overnight (24 h) in a nutrient broth medium (Merck, Germany), harvested with 3000 rpm centrifuge for 15 min (Macario, 2012) and diluted with physiological solution (8.5% NaCl) before application until the initial optical density of 1 Å (approximate cell counts of 9 Log CFU/ml) (Purwanti et al., 2019b). The bioaugmentation of bacteria was performed in the beginning of the phytoremediation test using v/v ratio of 2% (initial cell counts of 6.5 ± 0.7 Log CFU/g) and 5% (initial cell counts of 7.0 ± 0.3 Log CFU/g) (Purwanti et al., 2019c).
2.8. Statistical analysis
All applicable data were presented as mean ± standard error of mean (SEM), and the statistical analysis in this research was performed using Minitab version 12.0 (Kurniawan and Imron, 2019). The analysis of correlation between the bacterial addition and the Al removal efficiency was performed using one-way analysis of variance (ANOVA) with null-hypothesis is as follows: the addition of bacteria did not significantly affect the Al removal. The alternative hypothesis is as follows: the addition of bacteria significantly affects the Al removal. Further significance analysis was conducted using Tukey HSD test. Determination of conclusion was carried out on the basis of the p-value with p < 0.05 indicating the significant difference between the results.
3. Results and discussion
3.1. Plant propagation and acclimatisation stage
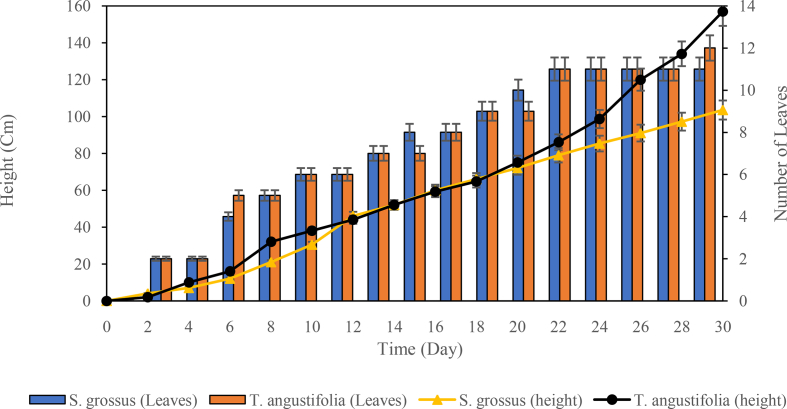
Growth rate monitoring was started during sprouting of the second-generation plants. The height and number of leaves of S. grossus and T. angustifolia were measured daily (Figure 2). Meanwhile, the height and number of leaves of S. grossus used for the next research stage were 20 cm and 5, respectively. The height and number of leaves of T. angustifolia considered for next research stage were 28 cm and 5, respectively. The determination of plant height and number of leaves were carried out on the basis of the observed growth parameter after 7 days of propagation. The obtained plants then underwent acclimatisation stage. The healthy plants after a 7-day period of acclimatisation will be used in the phytoremediation test.
Figure 2.
Initial growth measurement of S. grossus and T. angustifolia.
3.2. RFT of Al-contaminated soil
The Al concentration for phytoremediation test was determined through RFT. The results of 14-day observation (Figure 3) exhibited that S. grossus and T. angustifolia had symptoms of death at concentrations of 10,000 and 5000 mg/kg, respectively, after 7 days of exposure. This condition was marked by the physical changes (yellowing and withering leaves) in both plants. By contrast, the leaves, stems and roots of S. grossus and T. angustifolia turned yellow and dried on 14 days of exposure, thereby indicating that the plant cannot grow well and were about to die. The presence of these symptoms in plants was due to Al toxicity. Al causes root damage and hence inhibits the absorption of water and minerals needed by plants (Setiadi and Anira, 2015). Krstic et al. (2012) and Ismail et al. (2015) stated that the decreased growth of plants or the appearance of death symptoms are influenced by N absorption and P deficiency due to Al accumulation in plant tissues, thus decreasing the plant metabolic activity. This condition occurs because Al binds to P in the form of phosphate, which is unavailable to be used by plants, thereby causing nutrient deficiency.
Figure 3.
Observation of RFT on S. grossus and T. angustifolia.
The plants exposed to Al solution at concentrations of 500, 50 and 0 mg/kg at the beginning to the end of the RFT stage remained fresh with no physical change. This result indicated that both plants can survive at concentrations of 0–500 mg/kg. These observations also showed that the plants can survive without being affected by the pollutant concentrations under these concentrations. The leaves of plants remained green, and the stems were still fresh. Paczkowska et al. (2007) stated that certain plants can adapt to Al because they employ a certain mechanism to suppress the adverse effects of Al to prevent it from interfering with nutrient uptake and streamline it. The Al concentration that can be tolerated by both plants in this research was 500 mg/kg. Thus, the concentration of 500 mg/kg was used for S. grossus and T. angustifolia in the phytoremediation stage.
The plant height measurements showed identical results, thereby supporting the physical observation. The height of S. grossus increased from day 0 to day 14 at Al concentrations of 0–500 mg/kg. At Al concentrations of 10,000 and 5000 mg/kg, the height of S. grossus increased on days 0–7 and then decreased on days 8–14 of exposure. Such finding was attributed to the toxicity of Al, which impeded the plant growth (Setiadi and Anira, 2015). The presence of Al causes root damage, thereby inhibiting the absorption of water and minerals required by plants (Bojórquez-Quintal et al., 2017). The height of T. angustifolia showed a fluctuating increase in the concentration of 0–500 mg/kg Al. Reductions in plant height in Al concentrations of 5000–10,000 mg/kg observed on the 7th day of exposure were due to Al toxicity. This observation is supported by the finding of Watanabe et al. (2001), who reported that Al poisoning affects the availability of nutrients, and the ability of plants to absorb nutrients is limited under these conditions because Al can interfere the cation exchange.
All treated reactors (T1 and T2) showed pH values ranging between 5.5 and 6.5. The pH in control reactor (T0) was gradually increased from 5.5–6.0 to 7.5. These observations indicated that precipitation by pH did not occur in a high rate for all treated reactors; however, it can still occur through complexation mechanisms (performed by root exudates or bacterial metabolisms) (Li et al., 2018; Talebi et al., 2019). On the basis of the pH observation, the removal of Al in the control reactors were carried out due to the increasing pH (Balintova and Petrilakova, 2011; Purwanti et al., 2019a), and it will be deposited in the bottom of the reactor.
3.3. Al content in roots and plant stems
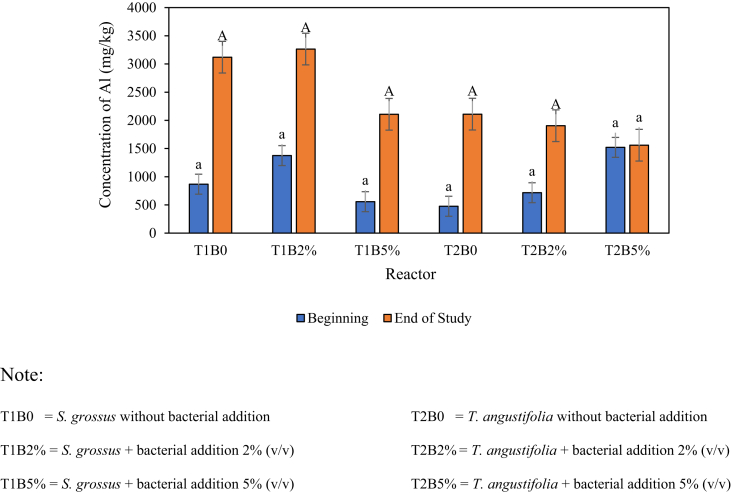
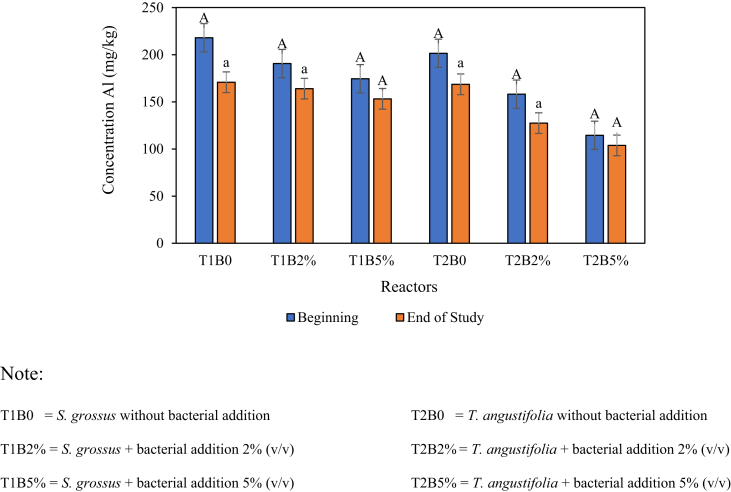
The Al content in roots and stems was analysed to determine the amount of Al absorbed by the roots and stems of S. grossus and T. angustifolia after 28 days of phytoremediation test. This analysis was performed at the beginning and end of the study. Figures 4 and 5 demonstrate the results of the Al test on the roots and stems of plants, respectively, in each test reactor during the observation.
Figure 4.
Al content in plant roots (values are presented in mean ± SEM). Letter a-A indicates the significant differences between the initial and the final concentrations.
Figure 5.
Al content in plant stems (values are presented in mean ± SEM). Letter a-A indicates the significant differences between the initial and the final concentrations.
The results of the initial Al content analysis of the roots and stems of S. grossus and T. angustifolia (Figures 4 and 5, respectively) show that the plants already contained Al. This finding was observed because the soil used in the propagation stage contained Al with a concentration of 1015 mg/kg. The presence of Al in the roots and stems of plants resulted from the absorption of Al from the propagation medium. The high Al content for roots was in the T1B2% reactor (S. grossus with 2% bacteria addition) with a value of 3265 mg/kg.
During the research, both plants healthily grow. The S. grossus height increased up to 120 ± 12 cm, and the number of added leaves was 10. By contrast, the T. angustifolia height increased up to 80 ± 8 cm, and the number of added leaves was 10. No sign of sclerosis or withering was observed until the end of the test period. The observed growth of plants was the main indicator that the tested species can withstand the given Al concentration and continuously perform phytoextraction (Abdullah et al., 2020; Frydenvang et al., 2015). The observed Al content for all plants was higher at the beginning of the study than that at the end due to the effect of stem growth accompanied by low Al transport from root to the stem area. Watanabe et al. (2001) reported that the accumulation of Al in stem occurred under a steady state or low rate; thus, the high growth of stem will decrease the concentration of total Al in stem (as it calculated as mg Al/kg stem). The high Al content in roots was due to their direct contact with Al-contaminated soil as the growing medium and the tendency of Al accumulation inside plants.
Al usually showed no/low accumulation in the stem and leaf parts of plants due to the tendency and the mechanism of Al accumulation inside the root area of plants (Shahandeh and Hossner, 2000). The obtained result in this study was in agreement with that of Ismail et al. (2020) who also obtained a significantly higher concentration of Al in root compared with the plant stem. Rhizostabilisation and phytoextraction played important roles in this observed result. Plant's exudates can directly stabilise/complexify the bioavailable Al in their root area (Ismail et al., 2019; Titah et al., 2018). Bioavailable Al was extracted by plants and accumulated in a certain part of plants referring to their tendency/fate (Lebeau et al., 2011). In the case of Al, the accumulation tendency is in root part.
The interaction between metals and plants facilitated by root exudates, such as amino acids, organic acids or other compounds that can accumulate metals and transport them to other plant parts, was expected to occur during this research. This finding is supported by the work of Dakora and Phillips (2002) who stated that plants that grow in soils with high Al content produce root exudates (in the form of anions of organic acids, sugars, vitamins, amino acids, purines, nucleotides and inorganic ions). These exudates help plant roots avoid the damaging effects of Al ions. Accordingly, roots can continually absorb nutrients and water. The high absorption of Al in the roots was influenced by the length during the observation period. This result is supported by the work of Sampanpanish et al. (2010) who reported that the Al accumulation increased with increasing metal concentration and time.
3.4. Analysis of Al content in soil
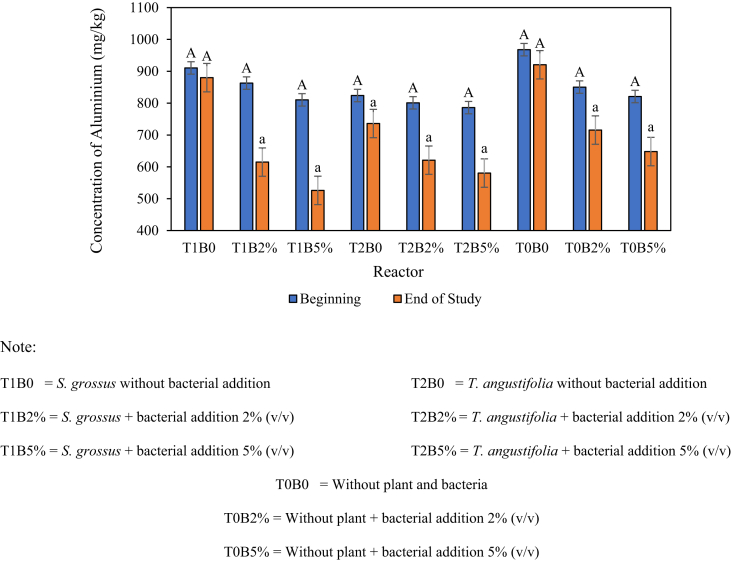
The analysis of the Al content in the soil was carried out to determine the efficiency of Al removal using S. grossus and T. angustifolia assisted by V. alginolyticus. The analysis was performed at the beginning and end of the study. The initial analysis was implemented to determine the initial Al content in the soil (day 0). By contrast, the final analysis was conducted to measure the Al content in the soil after 28 days of observation. Figure 6 shows the results of the Al content in the soil in each test reactor.
Figure 6.
Al content in the soil (values are presented in mean ± SEM). Letter a-A indicates the significant differences between the initial and the final concentrations.
Figure 6 shows that the soil in each reactor contained Al at the beginning of the study (day 0). The Al content in the soil was ranged between 786 mg/kg and 968 mg/kg. This result indicated that the soil medium used in this study already contained Al. Tsakiridis (2012) stated that Al is one of the chemical elements that constitute the earth's crust. Overall, the T1B5% reactor with S. grossus and the addition of 5% (v/v) bacteria during the 28 days of testing showed a low final Al content (526 mg/kg). This finding was due to the biochemical–physiological processes performed by plants and bacteria creating absorption and accumulation mechanism (Krstic et al., 2012; Ngo et al., 2014; Titah et al., 2019). The efficiency of Al removal in soil was calculated on the basis of the reduction of Al concentration in the soil medium. The efficiencies of Al removal in each reactor are shown on Figure 7.
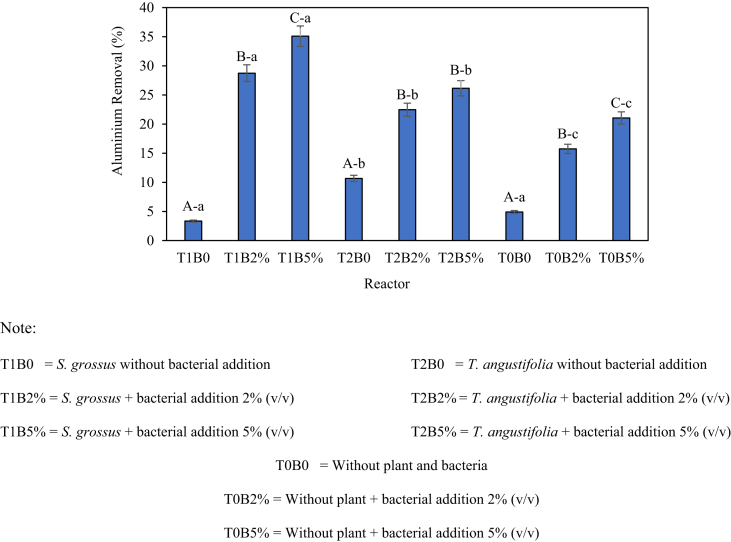
Figure 7.
Efficiency of Al removal (values are presented in mean ± SEM. Letter A-B-C above the graph indicates the significant difference of Al removal from soil medium between no bacterial addition, 2% bacterial addition and 5% bacterial addition; letter a-b-c above the graph indicates the significant difference of Al removal from the soil medium between S. grossus, T. angustifolia and control reactor.
Figure 7 shows that the high removal of Al occurred on the T1B5% reactor, which is S. grossus, with the addition of 5% V. alginolyticus reaching up to 35.1%. In comparison with T0B5% (without plants but with 5% bacteria addition), the addition of S. grossus significantly increased the Al removal capacity by 14.0% (p < 0.05). T. angustifolia showed a lower Al removal compared with S. grossus with a high efficiency of 26.2% achieved with the addition of 5% V. alginolyticus (T2B5%). The utilisation of T. angustifolia showed a significant increasement of 5.1% Al removal compared with the control (T0B5%). The addition of V. alginolyticus showed a significant increasement of Al removal compared with the no-bacterial addition reactor (p < 0.05) with the highest increasement up to 31.8% (T1B0 vs. T1B5%). This result indicated that the bacterial addition (especially pollutant-resistant/tolerant bacteria) will enhance the removal of the pollutant. Ismail et al. (2020) mentioned that the addition of bacteria promotes the growth of plants and enhance the Al removal by up to 19%. The proposed mass balances of Al for control and treated reactors are presented on Figure 8.
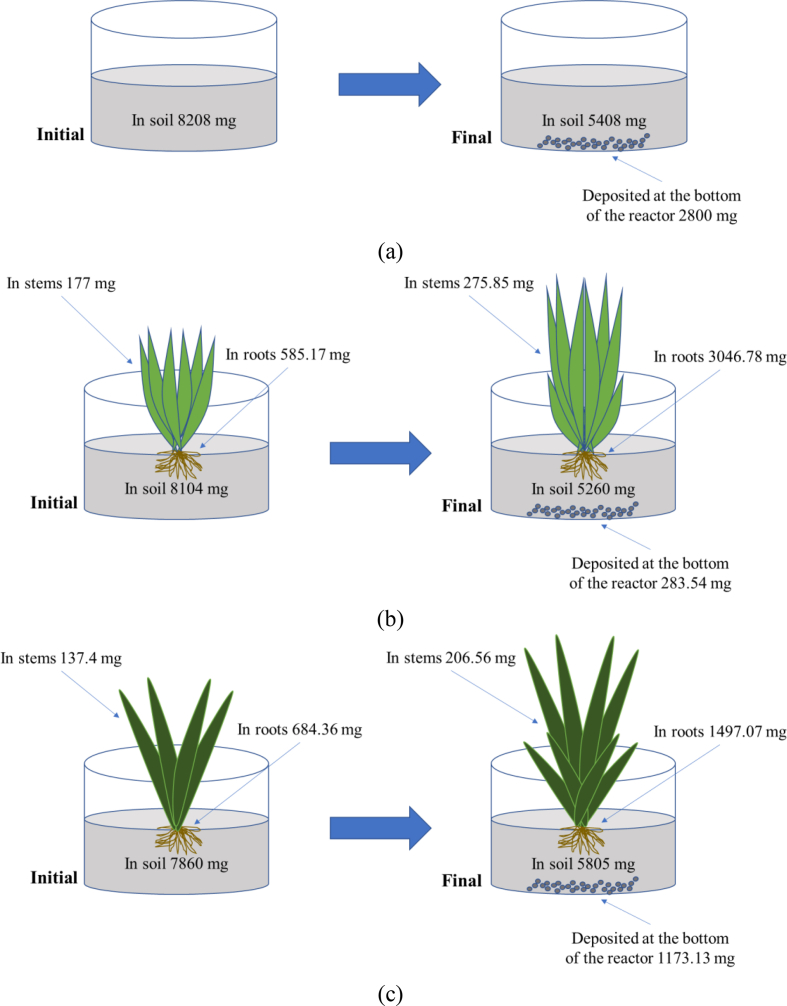
Figure 8.
Proposed mass balance for (a) T0B5% (b) T1B5% (c) T2B5%.
The addition of bacteria might assist the phytoremediation through some mechanisms, including phytostimulation and rhizostabilisation (Ebrahimipour et al., 2020; Ismail et al., 2019; Tangahu et al., 2011). Plant's exudates create a good environment for bacteria to live in the root area exhibiting the phytostimulation mechanism. Exudates also provide some essentials elements that bacteria need to perform enzymatic reactions (Dakora and Phillips, 2002). Bacteria assist the removal of pollutant by transforming the pollutant into a less toxic compound, which can then be extracted by plants (Mohan and Tippa, 2019). Moreover, bacteria can increase the bioavailability of pollutant (especially metal); thus, phytoextraction might occur in a high rate (Imron et al., 2020). Understanding how the Al removal occurred during the study, it is strongly suggested that the in-situ application of this method must be accompanied with underlayer/liner protection to prevent the infiltration of metal ion that might pollute the groundwater further (Imron et al., 2019a). Layer need to be placed below the area of the remediated soil (by excavating) to limit/block the percolation of water that carries soluble metal ion (Adar and Bilgili, 2015). Several layer types can be used on the application, including geomembrane and clay (Abd-Elhamid et al., 2019).
No standard for maximum/allowable concentration of Al in soil is available at present. Several studies also reported a high concentration of Al in soil because it is an abundant metal element in the earth's crust. Rasmussen et al. (2001) reported a concentration higher than 12,000 mg/kg of Al on the 160 sampling sites in the province of Canada. Zołotajkin et al. (2011) also reported the concentration of Al up to 4800 mg/kg in some sampling locations in Poland. The toxicity of Al depends on the pH condition. A concentration starting from 300 mg/kg in acidic condition of soil might cause toxicity to plants (Zołotajkin et al., 2011), thereby resulting in a withering of leaves by interfering the essential elemental uptake (Ismail et al., 2020). Sequential/multiple stages of phytoremediation can be performed (Said et al., 2020) by using the tested plants in this study to achieve the concentration below 300 mg/kg.
3.5. Bioaccumulation of Al by plants
The assessment of bioconcentration factor (BCF) aimed to determine the ability of plants to accumulate Al from the contaminated medium. BCF was calculated on the basis of the ratio of the metal concentration in plant parts with the metal concentration in the growing medium (Santiago et al., 2011). The BCF value more than unity indicated the capability of plants in performing hyperaccumulation of metal (Baker and Brooks, 1989; Bolan et al., 2011). The BCF was calculated for the final concentration of Al in plants (roots and stems). The soil medium is presented on Table 1.
Table 1.
BCF of Al by S. grossus and T. angustifolia.
| Reactor | Aluminium Concentration in Medium (mg/kg) | Aluminium Concentration in Plants (mg/kg) |
BCF Value | |
|---|---|---|---|---|
| Root | Stem | |||
| T1B0 | 880.1 | 3,120 | 218 | 3.545 |
| T1B2% | 615 | 3,265 | 190.6 | 5.308 |
| T1B5% | 526 | 2,108 | 174.5 | 4.007 |
| T2B0 | 736 | 2,110 | 201.5 | 2.866 |
| T2B2% | 621 | 1,905 | 158.2 | 3.068 |
| T2B5% | 580.5 | 1,559 | 114.5 | 2.686 |
Table 1 demonstrates that S. grossus and T. angustifolia can be categorised as hyperaccumulator plants. In accordance with the obtained result, Tangahu et al. (2010) also stated that S. grossus exhibits its capability as a hyperaccumulator plant on the basis of the bioaccumulation of pollutants. Tahira (2006) characterised T. angustifolia as a hyperaccumulator plant due to its high ratio of metal bioconcentrations in roots and other plant parts. All S. grossus reactors showed higher BCF values compared with all T. angustifolia reactors. This situation demonstrated that S. grossus has a good performance in the bioaccumulation of Al from the contaminated medium.
4. Conclusions
The RFT result showed that S. grossus and Thypa angustifolia can tolerate Al in soil up to 500 mg/kg. The addition of V. alginolyticus showed a significant increment in the Al removal from soil reaching up to 14.0% in the S. grossus reactor with 5% bacterial addition compared with the no-bacteria addition (control). The maximum Al removal was obtained in a reactor of S. grossus plant with 5% bacteria addition with the removal efficiency of 35.1%. Meanwhile, a removal efficiency of 26.2% was observed for T. angustifolia with 5% bacteria addition. Bioaugmentation of V. alginolyticus enhanced the overall Al removal due to the improvement of Al bioavailability, which can be further extracted from soil by plants. Rhizostabilisation, phytostimulation and phytoextraction mechanisms were subjected to be occurred during the treatment. Both plants have shown potential as Al hyperaccumulators with BCF values up to 5.308 and 3.068 for S. grossus and T. angustifolia, respectively. The presented result was limited to the indirect analysis of the enhancing effect of Al removal by bacteria addition; thus, a further study on the interaction of plants and bacteria in phytoremediation of Al is highly suggested. A further study on the design of the reactor for a real ex situ treatment application can be carried out to enrich the current findings. A direct in situ application can also be applied by planting the above-mentioned plant species and introducing the V. alginolyticus bacteria into the soil under several parameter control to maintain the optimal growth of plants and bacteria.
Declarations
Author contribution statement
Ipung Fitri Purwanti, Siti Rozaimah Sheikh Abdullah: Conceived and designed the experiments; Contributed reagents, materials, analysis tools or data; Wrote the paper.
Adriana Obenu, Setyo Budi Kurniawan: Performed the experiments; Analyzed and interpreted the data; Wrote the paper.
Bieby Voijant Tangahu: Conceived and designed the experiments.
Muhammad Fauzul Imron: Analyzed and interpreted the data; Contributed reagents, materials, analysis tools or data.
Funding statement
This work was supported by Institut Teknologi Sepuluh Nopember (ITS), Universitas Airlangga (UNAIR) through a research work collaboration 443/UN3.14/PT/2020 with Universiti Kebangsaan Malaysia (UKM) and the Ministry of Higher Education Malaysia through DCP-2018-006/3 research grant.
Competing interest statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
References
- Abd-Elhamid H.F., Abdelaal G.M., Abd-Elaty I., Said A.M. Efficiency of using different lining materials to protect groundwater from leakage of polluted streams. J. Water Supply Res. Technol. 2019;68:448–459. [Google Scholar]
- Abdullah S.R.S., Al-Baldawi I.A., Almansoory A.F., Purwanti I.F., Al-Sbani N.H., Sharuddin S.S.N. Plant-assisted remediation of hydrocarbons in water and soil: application, mechanisms, challenges and opportunities. Chemosphere. 2020;247:125932. doi: 10.1016/j.chemosphere.2020.125932. [DOI] [PubMed] [Google Scholar]
- Adar E., Bilgili M.S. The performance of four different mineral liners on the transportation of chlorinated phenolic compounds to groundwater in landfills. Sci. World J. 2015;2015 doi: 10.1155/2015/171284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmad J., Abdullah S.R.S., Hassan H.A., Rahman R.A.A., Idris M. Screening of tropical native aquatic plants for polishing pulp and paper mill final effluent. Malaysian J. Anal. Sci. 2017;21:105–112. [Google Scholar]
- Anjum N.A. Plant acclimation to environmental stress: a critical appraisal. Front. Plant Sci. 2015;6 [Google Scholar]
- Aransiola S.A., Ijah U.J.J., Abioye O.P. Phytoremediation of lead polluted soil by Glycine max L. Appl. Environ. Soil Sci. 2013;2013:1–7. [Google Scholar]
- Baker A.J.M., Brooks R.R. Terrestrial higher plants which hyperaccumulate metallic elements - a review of their distribution, ecology and phytochemistry. Biorecovery. 1989 [Google Scholar]
- Balintova M., Petrilakova A. Study of pH influence on selective precipitation of heavy metals from acid mine drainage. Chem. Eng. Trans. 2011;25:345–350. [Google Scholar]
- Bojórquez-Quintal E., Escalante-Magaña C., Echevarría-Machado I., Martínez-Estévez M. Aluminum, a friend or foe of higher plants in acid soils. Front. Plant Sci. 2017;8:363–369. doi: 10.3389/fpls.2017.01767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolan N.S., Park J.H., Robinson B., Naidu R., Huh K.Y. Phytostabilization. A green approach to contaminant containment. Adv. Agron. 2011;112:145–204. [Google Scholar]
- Dakora F.D., Phillips D.A. Root exudates as mediators of mineral acquisition in low-nutrient environments. Plant Soil. 2002;245:35–47. [Google Scholar]
- Ebrahimipour G., Yaghoobi Avini M., Ghorbanmovahhed M. Isolation and characterization of glutaminase-free L-asparaginase produced by Staphylococcus sp. MGM1. SciMedicine J. 2020;2:46–55. [Google Scholar]
- Exley C. Aluminum should now be considered a primary etiological factor in Alzheimer’s disease. J. Alzheimer’s Dis. Reports. 2017;1:23–25. doi: 10.3233/ADR-170010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frydenvang J., van Maarschalkerweerd M., Carstensen A., Mundus S., Schmidt S.B., Pedas P.R., Laursen K.H., Schjoerring J.K., Husted S. Sensitive detection of phosphorus deficiency in plants using chlorophyll a fluorescence. Plant Physiol. 2015;169:353–361. doi: 10.1104/pp.15.00823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallon C., Munger C., Prémont S., Campbell P.G.C. Hydroponic study of aluminum accumulation by aquatic plants: effects of fluoride and pH. Water, air. Soil Pollut. 2004;153:135–155. [Google Scholar]
- Ghosh P., Mandal S., Pal S., Bandyopadhyaya G., Chattopadhyay B.D. Development of bioconcrete material using an enrichment culture of novel thermophilic anaerobic bacteria. Indian J. Exp. Biol. 2006;44:336–339. [PubMed] [Google Scholar]
- Hseu Z.Y., Chen Z.S., Tsai C.C., Tsui C.C., Cheng S.F., Liu C.L., Lin H.T. Digestion methods for total heavy metals in sediments and soils. Water Air Soil Pollut. 2002;141:189–205. [Google Scholar]
- Imron M.F., Kurniawan S.B., Soegianto A. Characterization of mercury-reducing potential bacteria isolated from Keputih non-active sanitary landfill leachate, Surabaya, Indonesia under different saline conditions. J. Environ. Manag. 2019;241:113–122. doi: 10.1016/j.jenvman.2019.04.017. [DOI] [PubMed] [Google Scholar]
- Imron M.F., Kurniawan S.B., Soegianto A., Wahyudianto F.E. Phytoremediation of methylene blue using duckweed (Lemna minor) Heliyon. 2019;5 doi: 10.1016/j.heliyon.2019.e02206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imron M.F., Kurniawan S.B., Ismail N. ‘Izzati, Abdullah S.R.S. Future challenges in diesel biodegradation by bacteria isolates: a review. J. Clean. Prod. 2020;251:119716. [Google Scholar]
- Ismail N.I., Sheikh Abdullah S.R., Idris M., Hasan H.A., Al Sbani N.H., Jehawi O.H. Tolerance and survival of scirpus grossus and lepironia articulata in synthetic mining wastewater. J. Environ. Sci. Technol. 2015;8:232–237. [Google Scholar]
- Ismail N. ‘Izzati, Halmi M.I.E., AL Sbani N.H., Idris M., Hasan H.A., Hashim M.H., Abdullah S.R.S., Jehawi O.H., Sanusi S.N.A., Sheikh Abdullah S.R., Idris M., Abu Hasan H., Halmi M.I.E., Hussin AL Sbani N., Hamed Jehawi O., Sanusi S.N.A., Hashim M.H. Accumulation of Fe-Al by Scirpus grossus grown in synthetic bauxite mining wastewater and identification of resistant rhizobacteria. Environ. Eng. Sci. 2017;34:367–375. [Google Scholar]
- Ismail N. ‘Izzati, Abdullah S.R.S., Idris M., Hasan H.A., Halmi M.I.E., Al Sbani N.H., Jehawi O.H. Simultaneous bioaccumulation and translocation of iron and aluminium from mining wastewater by Scirpus grossus. Desalin. Water Treat. 2019;163:133–142. [Google Scholar]
- Ismail N. ‘Izzati, Abdullah S.R.S., Idris M., Kurniawan S.B., Effendi Halmi M.I., AL Sbani N.H., Jehawi O.H., Hasan H.A. Applying rhizobacteria consortium for the enhancement of Scirpus grossus growth and phytoaccumulation of Fe and Al in pilot constructed wetlands. J. Environ. Manag. 2020;267 doi: 10.1016/j.jenvman.2020.110643. [DOI] [PubMed] [Google Scholar]
- Jombang B.L.H. Jombang; 2016. Laporan Informasi Kinerja Pengelolaan Lingkungan Hidup. [Google Scholar]
- Khan A.G., Kuek C., Chaudhry T.M., Khoo C.S., Hayes W.J. Role of plants, mycorrhizae and phytochelators in heavy metal contaminated land remediation. Chemosphere. 2000 doi: 10.1016/s0045-6535(99)00412-9. [DOI] [PubMed] [Google Scholar]
- Kozai T. 1991. Acclimatization of Micropropagated Plants; pp. 127–141. [Google Scholar]
- Krstic D., Djalovic I., Nikezic D., Bjelic D. Food Production - Approaches, Challenges and Tasks. 2012. Aluminium in acid soils: chemistry, toxicity and impact on maize plants. [Google Scholar]
- Kurniawan S.B., Imron M.F. Seasonal variation of plastic debris accumulation in the estuary of Wonorejo River, Surabaya, Indonesia. Environ. Technol. Innov. 2019;16:100490. [Google Scholar]
- Kurniawan S.B., Purwanti I.F., Titah H.S. The effect of pH and aluminium to bacteria isolated from aluminium recycling industry. J. Ecol. Eng. 2018;19:154–161. [Google Scholar]
- Lae K.Z.W., Su S.S., Win N.N., Than N.N., Ngwe H. Isolation of lasiodiplodin and evaluation of some biological activities of the stem barks of Phyllanthus Albizzioides (Kurz) Hook.f. Sci. Med. J. 2019;1:199–216. [Google Scholar]
- Laksono D.I., Muzayanah . Universitas Negeri Surabaya; 2012. Identifikasi Keluhan Masyarakat Akibat Industri Daur Ulang Aluminium di Kecamatan Sumobito Kabupaten Jombang. [Google Scholar]
- Lebeau T., Jézéquel K., Braud A. Microbes and Microbial Technology. Springer New York; New York, NY: 2011. Bioaugmentation-assisted phytoextraction applied to metal-contaminated soils: state of the art and future prospects; pp. 229–266. [Google Scholar]
- Li X., Li D., Yan Z., Ao Y. Adsorption of cadmium by live and dead biomass of plant growth-promoting rhizobacteria. RSC Adv. 2018;8:33523–33533. doi: 10.1039/c8ra06758a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macario A. Elsevier; 2012. Gene Probes for Bacteria. [Google Scholar]
- Manikandan G., Yuvashree M., Sangeetha A., Bhuvana K.P., Nayak S.K. Liver tissue regeneration using nano silver impregnated sodium alginate/PVA composite nanofibres. Sci. Med. J. 2020;2:16–21. [Google Scholar]
- Menon R.R., Luo J., Chen X., Zhou H., Liu Z., Zhou G., Zhang N., Jin C. Screening of fungi for potential application of self-healing concrete. Sci. Rep. 2019;9 doi: 10.1038/s41598-019-39156-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirza A., King A., Troakes C., Exley C. Aluminium in brain tissue in familial Alzheimer’s disease. J. Trace Elem. Med. Biol. 2017;40:30–36. doi: 10.1016/j.jtemb.2016.12.001. [DOI] [PubMed] [Google Scholar]
- Mohan S., Tippa A. Role of wetland soil bacteria in enhancing the phytoremediation process through bioavailability phenomenon. World Environ. Water Resour. Congr. 2019;2019:1–10. [Google Scholar]
- Ngo T.T.C., Le V.T., Shinjiro K. Identification and characterization of acidity-tolerant and aluminum-resistant bacterium isolated from tea soil. Afr. J. Biotechnol. 2014;13:2715–2726. [Google Scholar]
- Ojuederie O., Babalola O. Microbial and plant-assisted bioremediation of heavy metal polluted environments: a review. Int. J. Environ. Res. Publ. Health. 2017;14:1504. doi: 10.3390/ijerph14121504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paczkowska M., Kozłowska M., Goliński P. Oxidative stress enzyme activity in Lemna minor L. exposed to cadmium and lead. Acta Biol. Cracov. Bot. 2007 [Google Scholar]
- Pogue A.I., Lukiw W.J. Aluminum, the genetic apparatus of the human CNS and Alzheimer’s disease (AD) Morphologie. 2016;100:56–64. doi: 10.1016/j.morpho.2016.01.001. [DOI] [PubMed] [Google Scholar]
- Purwanti I.F., Kurniawan S.B., Titah H.S., Tangahu B.V. Identification of acid and aluminium resistant bacteria isolated from aluminium recycling area. Int. J. Civ. Eng. Technol. 2018;9:945–954. [Google Scholar]
- Purwanti I.F., Simamora D., Kurniawan S.B. Toxicity test of tempe industrial wastewater on cyperus rotundus and scirpus grossus. Int. J. Civ. Eng. Technol. 2018;9:1162–1172. [Google Scholar]
- Purwanti I.F., Kurniawan S.B., Imron M.F. Potential of Pseudomonas aeruginosa isolated from aluminium-contaminated site in aluminium removal and recovery from wastewater. Environ. Technol. Innov. 2019;15:100422. [Google Scholar]
- Purwanti I.F., Kurniawan S.B., Ismail N. ‘Izzati, Imron M.F., Abdullah S.R.S. Aluminium removal and recovery from wastewater and soil using isolated indigenous bacteria. J. Environ. Manag. 2019;249:109412. doi: 10.1016/j.jenvman.2019.109412. [DOI] [PubMed] [Google Scholar]
- Purwanti I.F., Kurniawan S.B., Simanjuntak D.Y. Removal of aluminium in contaminated soil using locally isolated vibrio alginolyticus. J. Ecol. Eng. 2019;20:135–140. [Google Scholar]
- Rasmussen P.E., Subramanian K.S., Jessiman B.J. A multi-element profile of house dust in relation to exterior dust and soils in the city of Ottawa, Canada. Sci. Total Environ. 2001 doi: 10.1016/s0048-9697(00)00775-0. [DOI] [PubMed] [Google Scholar]
- Rezaee M., Kargar Ghomeshe P., Mohammad Hosseini A. Electrokinetic remediation of zinc and copper contaminated soil: a simulation-based study. Civ. Eng. J. 2017;3:690–700. [Google Scholar]
- Rezaee M., Nasrollahi Gisel M., Saffari S. Mathematical modeling and sensitivity analysis on cadmium transport in Kaolinite under direct current electric field. Civ. Eng. J. 2017;3:1097. [Google Scholar]
- Said N.S.M., Abdullah S.R.S., Ismail N. ‘Izzati, Hasan H.A., Othman A.R. Phytoremediation of real coffee industry effluent through a continuous two-stage constructed wetland system. Environ. Technol. Innov. 2020 [Google Scholar]
- Sampanpanish P., Tippayasak K., Chairat-utai P. Chromium accumulation by phytoremediation with monocot weed plant species and a hydroponic sand culture system. J. Envin. Res. Dev. 2010;4:654–666. [Google Scholar]
- Santiago Mahimairaja, Shenbagavalli S., Naidu R. Remediation of chromium-contaminated soil due to tannery waste Disposal : potential for phyto- and bioremediation. Jpn Soc. Pedol. 2011 [Google Scholar]
- Sarwoko M., Samudro G. 2006. Fitoteknologi Terapan, Computers & Education. [Google Scholar]
- Setiadi Y., Anira Early detection of aluminum toxicity on bridelia monoica merr. To post coal mined land in PT. Jorong barutama greston, South Kalimantan. Silvikultur Trop. - J. Trop. Silvic. Sci. Technol. 2015 [Google Scholar]
- Shahandeh H., Hossner L.R. Plant screening for chromium phytoremediation. Int. J. Phytoremediation. 2000 [Google Scholar]
- Supriyanto . Jana Tek; 2009. Analisis Hasil Pengecoran Aluminium Dengan Variasi Media Pendinginan. [Google Scholar]
- Tahira S.A. University of The Punjab; 2006. Phytoremediation of Tannery Effluents and Associated Contaminated Soil of Kasur District. [Google Scholar]
- Talebi M., Tabatabaei B.E.S., Akbarzadeh H. Hyperaccumulation of Cu, Zn, Ni, and Cd in Azolla species inducing expression of methallothionein and phytochelatin synthase genes. Chemosphere. 2019;230:488–497. doi: 10.1016/j.chemosphere.2019.05.098. [DOI] [PubMed] [Google Scholar]
- Tangahu B.V. Growth rate measurement of scirpus grossus plant as preliminary step to apply the plant in wastewater treatment using reedbed system. J. Civ. Environ. Eng. 2016 [Google Scholar]
- Tangahu B.V., Abdullah S.R.S., Basri H., Idris M., Anuar N., Mukhlisin M. Regional Engineering Postgraduate Confrence (EPC) 2010. Range finding test of lead (Pb) on scirpus grossus and measurement of plant wet dry weight as preliminary study of pythotoxicity. [Google Scholar]
- Tangahu B.V., Sheikh Abdullah S.R., Basri H., Idris M., Anuar N., Mukhlisin M. A review on heavy metals (As, Pb, and Hg) uptake by plants through phytoremediation. Int. J. Chem. Eng. 2011;2011:1–31. [Google Scholar]
- Titah H.S., Rozaimah S., Abdullah S.R.S., Idris M., Anuar N., Basri H., Mukhlisin M., Tangahu B.V., Purwanti I.F., Kurniawan S.B. Arsenic resistance and biosorption by isolated Rhizobacteria from the roots of Ludwigia octovalvis. Internet J. Microbiol. 2018;2018:1–10. doi: 10.1155/2018/3101498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Titah H.S., Purwanti I.F., Tangahu B.V., Kurniawan S.B., Imron M.F., Abdullah S.R.S., Ismail N. ‘Izzati. Kinetics of aluminium removal by locally isolated Brochothrix thermosphacta and Vibrio alginolyticus. J. Environ. Manag. 2019;238:194–200. doi: 10.1016/j.jenvman.2019.03.011. [DOI] [PubMed] [Google Scholar]
- Tong Y.P., Kneer R., Zhu Y.G. Vacuolar compartmentalization: a second-generation approach to engineering plants for phytoremediation. Trends Plant Sci. 2004;9:7–9. doi: 10.1016/j.tplants.2003.11.009. [DOI] [PubMed] [Google Scholar]
- Tsakiridis P.E. Aluminium salt slag characterization and utilization – a review. J. Hazard Mater. 2012;217–218:1–10. doi: 10.1016/j.jhazmat.2012.03.052. [DOI] [PubMed] [Google Scholar]
- Watanabe T., Osaki M., Tadano T. Al uptake kinetics in roots of Melastoma malabathricum L. - an al accumulator plant. Plant Soil. 2001 [Google Scholar]
- Zhang K., Zhou Q. Toxic effects of Al-based coagulants on Brassica chinensis and Raphanus sativus growing in acid and neutral conditions. Environ. Toxicol. 2005;20:179–187. doi: 10.1002/tox.20093. [DOI] [PubMed] [Google Scholar]
- Zołotajkin M., Ciba J., Kluczka J., Skwira M., Smoliński A. Exchangeable and bioavailable aluminium in the mountain forest soil of Barania Góra Range (Silesian Beskids, Poland). Water. Air. Soil Pollut. 2011;216:571–580. doi: 10.1007/s11270-010-0554-2. [DOI] [PMC free article] [PubMed] [Google Scholar]