CASE HISTORY
A 66-year-old male, being evaluated as a potential stem cell donor, was found to be leukopenic. There was no evidence of lymphadenopathy or organomegaly by clinical examination as well as ultrasonographic (USG) evaluation of neck and abdomen. The peripheral white blood cell count was 2.59 × 109/L with 52.4% lymphocytes (absolute lymphocyte count of 1.36 × 109/L) and 1.0% monocytes. The hemoglobin was 129 g/L and platelets were 287 × 109/L. Flow cytometric immunophenotyping (FCI) and molecular studies using polymerase chain reaction (PCR) were performed on a peripheral blood (PB) sample. Subsequently, FCI as well as morphological evaluation including immunohistochemistry (IHC) was performed on the bone marrow (BM) sample.
FLOW CYTOMETRIC STUDIES
Flow cytometry (FC; BD Biosciences, San Jose, CA; FACS Calibur) was performed first on PB sample and subsequently on the BM specimen. Erythrocytes were lysed by incubating with lysing solution (150 mM NH4Cl, 10 mM KHCO3, and 0.1 mM EDTA) for 10 min at room temperature at a ratio of 1:9 (volume of sample:volume of lysing solution). After incubation, cells were pelleted by centrifugation (500g for 5 min at room temperature), the medium was aspirated, and the cells were washed twice in a phosphate-buffered saline solution containing 0.1% NaN3. A comprehensive panel of antibodies is used as shown in Table 1. The antibody cocktail of CD14, CD16+56, CD19, and CD3 was used to enumerate T, B, and NK cells. The absolute B-cell count was calculated using dual platform (FC and an automated blood cell counter).
Table 1.
Antibody and Fluorochrome Combinations Per Tube Used in the Flow Cytometric Analysis
| Cocktails | FITC | PE | PerCP or PerCP-Cy5.5 | APC |
|---|---|---|---|---|
| 1 | CD14 | CD16+56 | CD19 | CD3a |
| 2 | CD4 | CD8 | CD45 | CD3b |
| 3 | CD103b | CD25 | CD20 | CD11c |
| 4 | Kappa | Lambda | CD19 | CD11c |
| 5 | Kappa | CD22 | CD20 | CD11c |
| 6 | Lambda | CD22 | CD20 | CD11c |
| 7 | CD5 | CD22 | CD20 | CD11c |
| 8 | CD5 | Lambda | CD19 | CD11c |
| 9 | – | CD123 | CD20 | CD11c |
Antibodies were purchased from Beckman Coulter.
Antibodies were purchased from DAKO.
Remaining antibodies were purchased from BD biosciences.
DISCUSSION
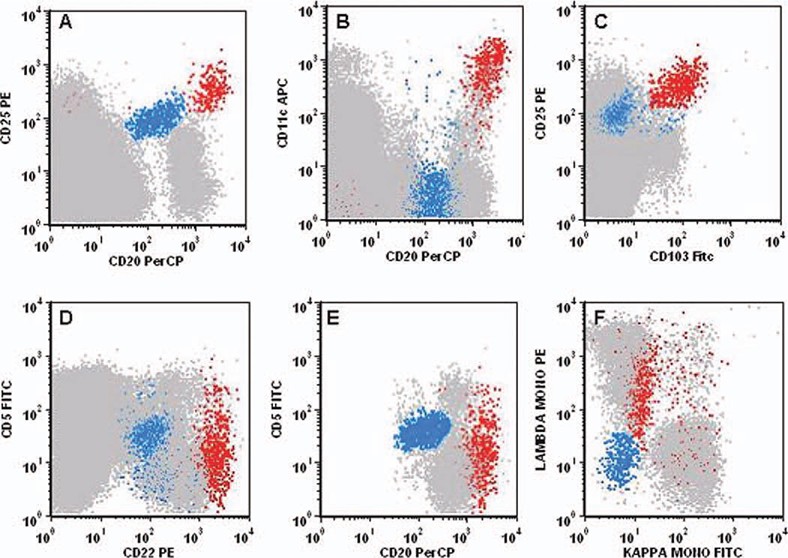
FC analysis of PB revealed two subsets of clonal B-cells (shown in Fig. 1). One subset (0.6% of lymphoid cells) expressed CD5, CD19, weak CD20, weak CD22, weak CD25, and weak lambda surface light chain but was negative for kappa surface light chain, CD11c, CD103, and CD123 (Fig. 1, blue population); and the other subset (0.3% of lymphoid cells) expressed bright CD20, bright CD22, bright CD11c, bright CD25, CD103, CD123, and lambda surface light chain but was negative for kappa surface light chain and CD5 (Fig. 1, red population). The immunophenotypic data of the first subset is consistent with monoclonal B-cells with chronic lymphocytic leukemia (CLL)-like immunophenotype; however, the absolute CLL-like clonal B-cell count was calculated to be 6.4 cells/μL. As the CLL-like clonal B-cell count was less than 5,000/μL (i.e., less than the cut-off criteria for the diagnosis of CLL) (1) and there was no evidence of lymphadenopathy, this subset was diagnosed as CLL-like monoclonal B-cell lymphocytosis (MBL). The immunophenotype of other subset was consistent with the diagnosis of classical hairy cell leukemia (HCL). PCR clonality studies demonstrated the presence of two discrete peaks, consistent with either the presence of two independent clonal populations or biallelic rearrangements occurring in one B-cell clone. BM was sampled and submitted for FCI and morphological review. FCI demonstrated significant involvement with HCL. Histopathological and IHC results revealed diffuse interstitial involvement by atypical lymphoid cells which were positive for CD20, CD79a, and TRAP confirming the diagnosis of HCL. The patient was entered onto an NCI clinical protocol for treatment of his HCL.
Fig. 1.
Multiparameter (4-color) flow cytometry immunophenotyping of PB lymphoid cells. Hairy cells are highlighted in red and CLL-like cells highlighted in blue with background polyclonal B-cells and non-B-cells in gray. Hairy cells were CD20 bright(+), CD22 bright(+), CD11c bright(+), CD25 bright(+), CD103(+), and surface lambda(+), but CD5(−) and surface kappa(−). CLL-like monoclonal B-cells were CD5(+), CD20 dim(+), CD22 dim(+), CD25 dim(+), and surface lambda dim(+), but CD11c (−), CD103(−), and surface kappa(−). (FACS Caliber, BD Biosciences; Analysis software: FSC Express research version 3).
In 2008, The International Workshop on Chronic Lymphocytic Leukemia (iw-CLL) revised the diagnostic criteria for CLL to require 5,000 or more CLL-phenotype B-cells/μL (1). MBL is an asymptomatic hematologic condition defined as a flow cytometrically identified clonal MBL with less than 5 × 109 cells/L expressing a disease specific immunophenotype without any diagnostic features of B-cell lymphoproliferative disorders and persisting over the period greater than 3 months (2). In the majority of cases, the monoclonal B-cells express a CLL-like immunophenotype (2). The risk of progression varies according to the number of MBL cells and type of MBL (3) with approximately 1% per year risk of progression to CLL requiring treatment (4). The prevalence of MBL reportedly ranges from 3 to 5% (3,000–5,000 in 100,000) in the general population over the age of 50 years (5). A higher prevalence has been reported in individuals older than 60 years, and affects almost one in five individuals (6); however most patients never progress to CLL and remain stable over an extended period of time (3). It is therefore crucial that MBL not be mistaken for a more aggressive B-cell lymphoproliferative process.
The incidence rate per 100,000 persons for B-cell neoplasms is 26.13 and the incidence of lymphoma, particularly of B-cell origin, is increasing worldwide (7). The statistical data (prevalence rate of MBL and incidence rate of B-cell neoplasms) suggest the rising probability of cooccurrence of MBL with other B-cell neoplasms. This also raises concern about the original diagnostic criteria for MBL which demands the absence of other lymphoproliferative disorders. However, to our knowledge, no study has been reported focusing on MBL in the presence of other clonal B-cell disorders.
It has been shown that in more than half of the MBL cases, the percentage of monoclonal B-cells was very low (<0.01%) and hence there is a high probability that MBL populations will be missed in the presence of confounding normal B-cells or a more obvious other clonal B-cell process (8). For the detection of one CLL-like monoclonal cell per microliter, it requires analysis of at least 200,000 total events and for detection of CLL cells at even lower level more than 500,000 cells with six or more fluorescent channels is required (6). This highlights the requirement to analyze high numbers of lymphocytes in a given patient if co-incident MBL is to be detected. The use of a limited panel of antibodies may also contribute to under detection of co-existing MBL. To our knowledge this is the first reported case of MBL co-incident with HCL.
FCI is being extensively used in the diagnosis, classification, and follow-up of hemato-lymphoid neoplasms. Many FC laboratories practice the approach of primary screening antibody panel and if required, it is followed by secondary antibody panel. Due to low incidence of HCL, the antibodies specific for HCL, i.e., CD11c, CD25, CD103, and CD123 are usually not included in the primary screening antibody panel. If a limited panel is run in such a case, the MBL might be confused as CLL; and HCL might have been missed. In this case, patient presented with low WBC count and monocytopenia without lymphadenopathy or organomegaly. Although monocytopenia provides a clue toward HCL, an absence of splenomegaly does not favor it. Because of use of a comprehensive antibody panel (shown in Table1), we could diagnose this case of HCL (0.3% of lymphoid cells) with CLL-like MBL (0.6% of lymphoid cells). The leucopenia was caused by the HCL and the MBL was noncontributory to the clinical management of this patient.
Moreover, due to the routine use of limited antibody panel, MBL might be confused as a part of the coincident B-cell lymphoproliferative process and may lead to the miscalculation of the extent of disease involvement and in the case of co-incident B-cell NHL to false positive staging in such cases. Also, the persistence of these MBL cells during monitoring of response to therapy, especially for determination of minimal residual disease, may lead to inaccurate results. It was therefore important in following this patient to differentiate between the two different monoclonal B-cell lymphoproliferative disorders and to quantitate the response of just the HCL to therapy. The MBL cell count was very low and unlikely to progress to CLL (3).
In summary, the prevalence of the CLL-like MBL is relatively high in the general population over the age of 50 years and it may co-exist with other frank B-cell NHL or low grade B-cell leukemias. This case highlights the necessity of a multiparametric flow cytometric approach along with panels for differentiation of MBL from other B-cell lymphoproliferative processes. Furthermore, the existence of cases in which MBL clearly is an incidental finding in a patient with another lymphoma or leukemia supports a modification of current criteria for MBL.
CASE 1 DIAGNOSIS: Hairy Cell Leukemia with CLL-Like Monoclonal B Lymphocytosis
Supplementary Material
Acknowledgments
Grant sponsor: Intramural Research Program of the NIH, NCI.
Footnotes
Additional Supporting Information may be found in the online version of this article.
LITERATURE CITED
- 1.Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Dohner H, Hillmen P Keating MJ, Montserrat E, Rai KR, et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: A report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood 2008;111:5446–5456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Marti GE, Rawstron AC, Ghia P, Hillmen P Houlston RS, Kay N, Schleinitz TA, Caporaso N. Diagnostic criteria for monoclonal B-cell lymphocytosis. Br J Haematol 2005;130:325–332. [DOI] [PubMed] [Google Scholar]
- 3.Rawstron AC, Shanafelt T, Lanasa MC, Landgren O, Hanson C, Orfao A, Hillmen P, Ghia P. Different biology and clinical outcome according to the absolute numbers of clonal B-cells in monoclonal B-cell lymphocytosis (MBL). Cytometry Part B Clin Cytom 2010;78B(Suppl 1): S19–S23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Caporaso NE, Marti GE, Landgren O, Azzato E, Weinberg JB, Goldin L, Shanafelt T. Monoclonal B cell lymphocytosis: clinical and population perspectives. Cytometry Part B Clin Cytom 2010;78B(Suppl 1):S115–S119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Landgren O, Albitar M, Ma W Abbasi F, Hayes RB, Ghia P, Marti GE, Caporaso NE. B-cell clones as early markers for chronic lymphocytic leukemia. N Engl J Med 2009;360:659–667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shanafelt TD, Ghia P Lanasa MC, Landgren O, Rawstron AC. Monoclonal B-cell lymphocytosis (MBL): Biology, natural history and clinical management. Leukemia 2010;24:512–520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jaffe ES HN, Stein H, Campo E, Pileri SA, Swerdlow SH.Introduction and overview of classification of lymphoid neoplasms In: Swerdlow SH,Campo E,Harris NL,Jaffe ES,Pileri SA,Stein H,Thiele J,Vardiman JW, editors. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues.Lyon, France:IARC Press; 2008. pp 158–166. [Google Scholar]
- 8.Nieto WG, Almeida J, Romero A, Teodosio C, Lopez A, Henriques AF, Sanchez ML, Jara-Acevedo M, Rasillo A, Gonzalez M, et al. Increased frequency (12%) of circulating chronic lymphocytic leukemia-like B-cell clones in healthy subjects using a highly sensitive multicolor flow cytometry approach. Blood 2009;114:33–37. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.