Highlights
-
•
SARS infection transcription profiling is hypothesised as a rout to drug repurposing for the COVID-19 pandemic.
•Potential drugs are selected based on their recapitulating the lung cell SARS infection defence response.
-
•
Of the resulting repurposing candidates half have reported antiviral activities and nine shown to block SARS2.
-
•
This approach may serve to broaden the spectrum of approved drugs that should be further assessed as potential COVID-19 therapeutics.
Keywords: COVID-19, Gene expression, Repurposing
Abstract
We have utilised the transcriptional response of lung epithelial cells following infection by the original Severe Acute Respiratory Syndrome coronavirus (SARS) to identify repurposable drugs for COVID-19. Drugs best able to recapitulate the infection profile are highly enriched for antiviral activity. Nine of these have been tested against SARS-2 and found to potently antagonise SARS-2 infection/replication, with a number now being considered for clinical trials. It is hoped that this approach may serve to broaden the spectrum of approved drugs that should be further assessed as potential anti−COVID-19 agents and may help elucidate how this seemingly disparate collection of drugs are able to inhibit SARS-2 infection/replication.
1. Introduction
TheCOVID-19 pandemic has spawned a global drug development effort. With a vaccine still many months away and novel drugs requiring years to reach the clinic, drug repurposing has become an attractive alternative. To date the most successful intervention in the pandemic has been the redeployment of dexamethasone for patients in intensive care (Horby et al., 2020). Still further effort is needed to identify other approved drugs that can be repositioned against SARS-2. One approach is based on the observation that the gene expression changes seen in disease states can serve as effective disease descriptors or quantitative phenotypes (Golub et al., 1999; Lee and Young, 2013) and drugs can then be repurposed based on an ability to drive expression in the opposite direction (Marton et al., 1998; Hughes et al., 2000; Wei et al., 2006; Zhang et al., 2012; Williams, 2012; Walf-Vorderwulbecke et al., 2018). However, in the case of SARS, transcriptional data are largely limited to the cellular response to infection established in in vitro assays, which is characterised by the up regulation of a cellular viral defence mechanism. The hypothesis behind the present work is that drugs driving these defensive gene expression changes may bolster the cellular response to infection and thus present candidate therapeutics to fight the SARS-2 coronavirus.
2. Methods
In total, 17 infection SARS-associated transcriptional profiles were generated from NCBI GEO deposited expression series, see Table S1 for details. CMAP (Lamb et al., 2006) profiles were defined as previously described (Williams et al., 2019). LINCS profiles were generated based on the deposited dataset series GSE92742 and GSE70138 (Subramanian et al., 2017). The LINCS portal of SPIED (www.spied.org.uk) hosts profiles in the form of categorical calls on the up/down status of genes generated based on combining expression data for drug/cell replicates. In the present work the LINCS profiles for different cell types are combined for each drug based on a given gene being assigned an up or down regulated status based on a majority vote across the cell types. See Supplementary file for further Methods details.
3. Results
A high degree of correlation between the profiles was found, Table S1, facilitating the definition of a composite signature for SARS infection comprising 192 up and 136 down regulated genes, Table S2. Pathway and gene ontology enrichment analyses of the up regulated gene set highlight the involvement of a viral defence mechanism and immune response genes, Tables S3, S4. The signature also returns high ranking virus infection related transcription studies in a search of publicly available data, Table S5.
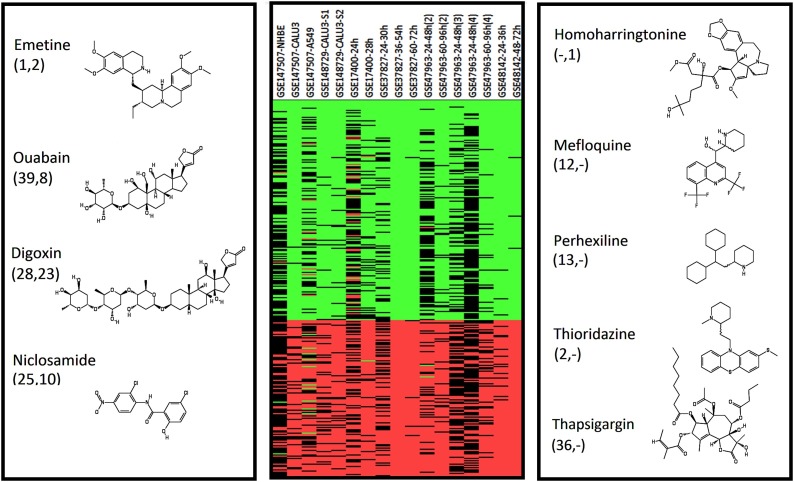
The CMAP and LINCS repositories of the transcriptomic effects of approved drugs were queried for positive correlates of the SARS signature with the striking result that half of the high scoring drugs have reported anti-viral activities (23/45 in CMAP and 22/45 in LINCS) of which 9 have recently shown to be potent inhibitors of SARS-2 (Jeon et al., 2020; Choy et al., 2020), see Fig. 1 and Table S6.
Fig. 1.
Transcription-based repurposing candidates. The SARS signature gene expression across the 17 component profiles is shown in the middle. The four common hits that are SARS-2 antagonists are shown at the left with their respective CMAP and LINCS ranks. The hits unique to the two datasets are shown to the right. The structures illustrate the power of transcription profiling to group chemically diverse compounds into a biological activity class.
Ouabain, the most potent of the SARS-2 antagonist, and Digoxin are plant derived toxins used at low concentrations to treat hypertension and cardiac arrhythmia and have been shown to inhibit SARS-2 and MERS replication in the nanomolar range (Jeon et al., 2020; Ko et al., 2020). Both inhibit the sodium/potassium ATPase, or sodium/potassium pump (Ogawa et al., 2009; Laursen et al., 2015), and CTS antiviral activity has been linked to the ionic changes they induce in the cell being less favourable for viral replication (Hartley et al., 2006; Grosso et al., 2017). Low doses of Ouabain also reduce inflammatory cytokine production in LPS treated rats by blocking the nuclear translocation of NF-kB (Kinoshita et al., 2014). At low picomolar concentrations Ouabain promotes an interaction between the pump and the Angiotensin Type 1 Receptor (AT1R) (Ketchem et al., 2016). It has also been demonstrated that AT1R directly interacts with ACE2 (Deshotels et al., 2014), the port of entry for the SARS and SARS-2 (Hoffmann et al., 2020) coronaviruses. It is plausible then that at very low concentrations Ouabain, and by extension Digoxin, cause the internalisation of the pump in complex with AT1R and ACE2, thereby reducing the cell surface expression levels of ACE2 and inhibiting the ability of SARS-2 to enter cells and replicate. The therapeutic range of Ouabain and Digoxin is limited, as at high doses they are extremely toxic. However, the effects outlined above appear to occur at very low concentration, well below toxic levels. Interestingly, in the LINCS data set there are two molecules with very close homology to the CTSs; witherferin-A, the active ingredient in Ashwagandha, and cucurbitacin, from the squirting cucumber. As these are not FDA approved drugs they have not been tested in in vitro assays but now warrant urgent testing for an ability to inhibit SARS-2 infecting cells.
Interestingly, in response to the recent data on anti-SARS-2 activity (Jeon et al., 2020; Ko et al., 2020) C2 pharma (www.c2pharma.com) is opening access to its stocks of Digoxin for trial purposes. There are 4 other CTS like drugs among our top hits, these too should now be assessed for their ability to inhibit SARS-2 infection as a matter of some urgency.
The closely related ipecac root extracts Emetine (our top CMAP and second LINCS hit) and Cephaeline (3rd and 35th respectively) are prescribed as expectorants and at high doses emetics. Of these Emetine has been shown to potently inhibit SARS-2 (Choy et al., 2020) and has been singled out as a possible COVID-19 therapeutic (Bleasel and Peterson, 2020). Emetine is currently being developed for COVID-19 by Acer Therapeutics (www.acertx.com).
The broad spectrum antiviral Niclosamide (Jurgeit et al., 2012; Xu et al., 2020) has been repurposed three times over its 60 year history and recently been shown to have sub-micromolar potency against SARS-2 (Jeon et al., 2020). Niclosamide is now in Phase 2 trials for COVID-19 sponsored by Tufts Medical Center (trial reference NCT04399356).
Of the hits unique to LINCS, Homoharringtonine, a protein translation inhibitor, also inhibits SARS-2 replication at low micromolar concentrations (Choy et al., 2020). The four unique CMAP hits Terfenadine, an antihistamine, the antimalarial Mefloquine, the antipsychotic Thioridazine and the antianginal Perhexiline have been shown to inhibit SARS-2 replication at low micromolar concentration (Jeon et al., 2020).
4. Discussion
Our data indicate that a seemingly disparate group of nine drugs each able to inhibit SARS-2 infection/replication, a number of which are being considered for trials, are linked though an ability to drive the expression of the SARS infection profile genes. Thus transcriptional data points to a common mechanism of action and is likely to reveal the therapeutic potential of drugs with as yet no reported antiviral activity but with well-known safety profiles.
Our data suggest that the other plant based cardiotonic steroids we have identified, for which there are no data concerning their ability to block SARS-2, should be tested as soon as possible, and if effective added to the list of candidate therapeutics.
In conclusion, these findings validate transcription-based repurposing as a rapid and effective means to identify potential treatments for the current COVID-19 pandemic and potentially for other zoonotic coronavirus pandemics that will doubtless inflict mankind in the future.
Author statement
Autor contributions to ‘Transcription-based drug repurposing for COVID-19′. RK and GW conceived the study and wrote the paper. GW did the bioinformatics analysis. PD and CB contributed to the discussion on pharmacology of candidate drugs.
Footnotes
Supplementary data associated with this article can be found, in the online version, at https://doi.org/10.1016/j.virusres.2020.198176.
Appendix A. Supplementary data
The following are Supplementary data to this article:
References
- Bleasel M.D., Peterson G.M. Emetine, ipecac, ipecac alkaloids and Analogues as potential antiviral agents for coronaviruses. Pharmaceuticals (Basel) 2020;13 doi: 10.3390/ph13030051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choy K.T., Wong A.Y., Kaewpreedee P., Sia S.F., Chen D., Hui K.P.Y. Remdesivir, lopinavir, emetine, and homoharringtonine inhibit SARS-CoV-2 replication in vitro. Antiviral Res. 2020;178 doi: 10.1016/j.antiviral.2020.104786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deshotels M.R., Xia H., Sriramula S., Lazartigues E., Filipeanu C.M. Angiotensin II mediates angiotensin converting enzyme type 2 internalization and degradation through an angiotensin II type I receptor-dependent mechanism. Hypertension. 2014;64:1368–1375. doi: 10.1161/HYPERTENSIONAHA.114.03743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golub T.R., Slonim D.K., Tamayo P., Huard C., Gaasenbeek M., Mesirov J.P. Molecular classification of cancer: class discovery and class prediction by gene expression monitoring. Science. 1999;286:531–537. doi: 10.1126/science.286.5439.531. [DOI] [PubMed] [Google Scholar]
- Grosso F., Stoilov P., Lingwood C., Brown M., Cochrane A. Suppression of adenovirus replication by cardiotonic steroids. J. Virol. 2017;91 doi: 10.1128/JVI.01623-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartley C., Hartley M., Pardoe I., Knight A. Ionic Contra-Viral Therapy (ICVT); a new approach to the treatment of DNA virus infections. Arch. Virol. 2006;151:2495–2501. doi: 10.1007/s00705-006-0824-x. [DOI] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horby P., Lim W.S., Emberson J., Mafham M., Bell J., Linsell L. Effect of dexamethasone in hospitalized patients with COVID-19: preliminary report. medRxiv. 2020;2020 06.22.20137273. [Google Scholar]
- Hughes T.R., Marton M.J., Jones A.R., Roberts C.J., Stoughton R., Armour C.D. Functional discovery via a compendium of expression profiles. Cell. 2000;102:109–126. doi: 10.1016/s0092-8674(00)00015-5. [DOI] [PubMed] [Google Scholar]
- Jeon S., Ko M., Lee J., Choi I., Byun S.Y., Park S. Identification of antiviral drug candidates against SARS-CoV-2 from FDA-approved drugs. Antimicrob. Agents Chemother. 2020 doi: 10.1128/AAC.00819-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jurgeit A., McDowell R., Moese S., Meldrum E., Schwendener R., Greber U.F. Niclosamide is a proton carrier and targets acidic endosomes with broad antiviral effects. PLoS Pathog. 2012;8 doi: 10.1371/journal.ppat.1002976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ketchem C.J., Conner C.D., Murray R.D., DuPlessis M., Lederer E.D., Wilkey D. Low dose ouabain stimulates NaK ATPase alpha1 subunit association with angiotensin II type 1 receptor in renal proximal tubule cells. Biochim. Biophys. Acta. 2016;1863:2624–2636. doi: 10.1016/j.bbamcr.2016.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinoshita P.F., Yshii L.M., Vasconcelos A.R., Orellana A.M., Lima Lde S., Davel A.P. Signaling function of Na,K-ATPase induced by ouabain against LPS as an inflammation model in hippocampus. J. Neuroinflammation. 2014;11:218. doi: 10.1186/s12974-014-0218-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ko M., Chang S.Y., Byun S.Y., Choi I., d’Alexandry d’Orengiani A.-L.P.H., Shum D. Screening of FDA-approved drugs using a MERS-CoV clinical isolate from South Korea identifies potential therapeutic options for COVID-19. bioRxiv. 2020;(2020) doi: 10.3390/v13040651. 02.25.965582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamb J., Crawford E.D., Peck D., Modell J.W., Blat I.C., Wrobel M.J. The Connectivity Map: using gene-expression signatures to connect small molecules, genes, and disease. Science. 2006;313:1929–1935. doi: 10.1126/science.1132939. [DOI] [PubMed] [Google Scholar]
- Laursen M., Gregersen J.L., Yatime L., Nissen P., Fedosova N.U. Structures and characterization of digoxin- and bufalin-bound Na+,K+-ATPase compared with the ouabain-bound complex. Proc. Natl. Acad. Sci. U S A. 2015;112:1755–1760. doi: 10.1073/pnas.1422997112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee T.I., Young R.A. Transcriptional regulation and its misregulation in disease. Cell. 2013;152:1237–1251. doi: 10.1016/j.cell.2013.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marton M.J., DeRisi J.L., Bennett H.A., Iyer V.R., Meyer M.R., Roberts C.J. Drug target validation and identification of secondary drug target effects using DNA microarrays. Nat. Med. 1998;4:1293–1301. doi: 10.1038/3282. [DOI] [PubMed] [Google Scholar]
- Ogawa H., Shinoda T., Cornelius F., Toyoshima C. Crystal structure of the sodium-potassium pump (Na+,K+-ATPase) with bound potassium and ouabain. Proc. Natl. Acad. Sci. U S A. 2009;106:13742–13747. doi: 10.1073/pnas.0907054106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subramanian A., Narayan R., Corsello S.M., Peck D.D., Natoli T.E., Lu X. A next generation connectivity map: L1000 platform and the first 1,000,000 profiles. Cell. 2017;171:1437–1452. doi: 10.1016/j.cell.2017.10.049. e17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walf-Vorderwulbecke V., Pearce K., Brooks T., Hubank M., van den Heuvel-Eibrink M.M., Zwaan C.M. Targeting acute myeloid leukemia by drug-induced c-MYB degradation. Leukemia. 2018;32:882–889. doi: 10.1038/leu.2017.317. [DOI] [PubMed] [Google Scholar]
- Wei G., Twomey D., Lamb J., Schlis K., Agarwal J., Stam R.W. Gene expression-based chemical genomics identifies rapamycin as a modulator of MCL1 and glucocorticoid resistance. Cancer Cell. 2006;10:331–342. doi: 10.1016/j.ccr.2006.09.006. [DOI] [PubMed] [Google Scholar]
- Williams G. A searchable cross-platform gene expression database reveals connections between drug treatments and disease. BMC Genomics. 2012;13:12. doi: 10.1186/1471-2164-13-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams G., Gatt A., Clarke E., Corcoran J., Doherty P., Chambers D. Drug repurposing for Alzheimer’s disease based on transcriptional profiling of human iPSC-derived cortical neurons. Transl. Psychiatry. 2019;9:220. doi: 10.1038/s41398-019-0555-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J., Shi P.Y., Li H., Zhou J. Broad spectrum antiviral agent niclosamide and its therapeutic potential. ACS Infect. Dis. 2020;6:909–915. doi: 10.1021/acsinfecdis.0c00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang D., Ciciriello F., Anjos S.M., Carissimo A., Liao J., Carlile G.W. Ouabain mimics low temperature rescue of F508del-CFTR in cystic fibrosis epithelial cells. Front. Pharmacol. 2012;3:176. doi: 10.3389/fphar.2012.00176. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.