Abstract
Diabetes-induced oxidative stress is one of the major contributors to dysfunction of endothelial progenitor cells (EPCs) and impaired endothelial regeneration. Thus, we tested whether increasing antioxidant protein metallothionein (MT) in EPCs promotes angiogenesis in a hind limb ischemia (HLI) model in endothelial MT transgenic (JTMT) mice with high-fat diet– and streptozocin-induced diabetes. Compared with littermate wild-type (WT) diabetic mice, JTMT diabetic mice had improved blood flow recovery and angiogenesis after HLI. Similarly, transplantation of JTMT bone marrow–derived mononuclear cells (BM-MNCs) stimulated greater blood flow recovery in db/db mice with HLI than did WT BM-MNCs. The improved recovery was associated with augmented EPC mobilization and angiogenic function. Further, cultured EPCs from patients with diabetes exhibited decreased MT expression, increased cell apoptosis, and impaired tube formation, while cultured JTMT EPCs had enhanced cell survival, migration, and tube formation in hypoxic/hyperglycemic conditions compared with WT EPCs. Mechanistically, MT overexpression enhanced hypoxia-inducible factor 1α (HIF-1α), stromal cell–derived factor (SDF-1), and vascular endothelial growth factor (VEGF) expression and reduced oxidative stress in ischemic tissues. MT’s pro-EPC effects were abrogated by siRNA knockdown of HIF-1α without affecting its antioxidant action. These results indicate that endothelial MT overexpression is sufficient to protect against diabetes-induced impairment of angiogenesis by promoting EPC function, most likely through upregulation of HIF-1α/SDF-1/VEGF signaling and reducing oxidative stress.
Introduction
Diabetes has attained global pandemic status with >415 million patients and with an estimated 193 million people living with undiagnosed diabetes worldwide (1). Type 2 diabetes (T2D) accounts for >90% of patients with diabetes and leads to microvascular and macrovascular complications that cause profound psychological and physical distress (1). For example, critical limb ischemia is the underlying cause of tissue loss in patients with peripheral artery disease (PAD), and PAD accounts for the high rate of limb amputation in patients with T2D. Thus, a better understanding of diabetic PAD may spur more specific and tailored management of patients with T2D. Defective endothelial regeneration impairs neovascularization and likely contributes to the pathophysiological changes that lead to vascular complications of PAD (2). Yet, specific therapeutic interventions aimed at accelerating repair of dysfunctional endothelium and restoring limb blood flow in patients with T2D remain to be established.
Endothelial progenitor cells (EPCs) participate in angiogenesis in vivo and have the ability to differentiate into endothelial cells in vitro as first described by Asahara et al. (3) in 1997. Accumulated evidence shows that EPCs play a vital role in vascular repair through increased mobilization, migration, homing, differentiation, and enhanced neovascularization of ischemic/hypoxic areas (4). Furthermore, EPC mobilization by low-energy shock waves or transplantation of functional EPCs appears to be a potential therapeutic strategy developed in murine models of diabetic ischemia (5,6). Nonetheless, the underlying mechanisms of dysfunction of resident EPCs as impaired in diabetes remains unresolved, and thus, there is a critical need to better understand EPCs in diabetic conditions to improve EPC therapeutic application in PAD of patients with T2D.
Metallothionein (MT) is a cysteine-rich protein that binds metals such as zinc (Zn). As a potent antioxidant, MT is highly efficient in scavenging or quenching various reactive oxygen species (ROS) that play critical roles in the onset and development of diabetes and diabetic complications (7). The importance of MT in diabetic complications has been demonstrated in previous studies in MT transgenic mice. For example, mice with cardiac-specific MT overexpression are resistant to diabetes-induced cardiac oxidative stress, insulin resistance, metabolic disorder, cardiomyocyte cell death, pathological remodeling, and the development of diabetic cardiomyopathy (8–11). Likewise, mice with podocyte-specific overexpression of MT are resistant to the development of diabetic nephropathy by decreasing oxidative damage in podocytes of OVE26 diabetic mice (12). Recently, we demonstrate that mice with endothelium MT overexpression (JTMT) are significantly protected against diabetic nephropathy in OVE26 diabetic mice (13). However, the role of MT in the regulation of EPC function and of endothelial repair in diabetes is unclear. Thus, the objectives of the current study were 1) to test whether endothelium MT overexpression in mice is sufficient to prevent diabetes-induced impairment in ischemia angiogenesis in vivo and 2) to delineate the molecular basis whereby MT promotes endothelial repair capacity (e.g., oxidative stress, EPC survival, migration, tube formation) in mice under diabetic conditions.
Research Design and Methods
Human Subjects and Peripheral Blood EPC Isolation and Culture
Patients with T2D (n = 6) were recruited from the First Affiliated Hospital of Chengdu Medical College. Healthy individuals (n = 6) admitted to the same hospital for preventive examinations were enrolled as control subjects. Written informed consent was obtained from all study participants. The baseline characteristics are shown in Supplementary Table 1. The study protocol was approved by the research ethics committee of the First Affiliated Hospital of Chengdu Medical College.
Human EPCs were isolated and cultured as detailed previously (14). Blood samples (20 mL) were mixed with 1 mL heparin (Sigma-Aldrich, St. Louis, MO), and mononuclear cells (MNCs) were isolated by density gradient centrifugation using Histopaque-1077 (Sigma-Aldrich) within 2 h of sampling. MNCs were suspended in endothelial growth factor–supplemented media (EGM-2 Bullet Kit; Lonza, Basel, Switzerland) with 10% FBS (Gibco, Carlsbad, CA) and seeded into six-well plates precoated with 50 μg/mL fibronectin (Sigma-Aldrich). Cells were maintained at 37°C with 5% CO2 in a humidified incubator. After 3 days of incubation, dead cells were washed away with PBS, and new medium was added. Medium was changed daily for 7 days and then every 3 days. After 2 weeks of culture, late EPCs were harvested for the following experiments.
Animal Models, Hind Limb Ischemia, and Cell Therapy
All the protocols described below were approved by the institutional animal care and use committee of the University of Louisville. JTMT mice were designed to overexpress human MTII gene specifically and ubiquitously in endothelial cells driven by murine Tie-2 promoter (13). JTMT mice were obtained by breeding JTMT heterozygotes with FVB wild-type (WT) mice. Age- and sex-matched littermate WT mice were used as controls. The db/db mice with FVB background [FVB.BKS(D)-Leprdb/+/ChuaJ] were purchased from The Jackson Laboratory (Bar Harbor, ME) and maintained under specific pathogen–free conditions at the University of Louisville animal facility. The db/db mice were generated by breeding heterozygous male and female (db/+ × db/+) mice following The Jackson Laboratory’s instructions.
The T2D mouse model was created by high-fat-diet (HFD) feeding and streptozotocin (STZ) induction in JTMT and WT control mice as described in our previous report (15). In this model, STZ damages pancreatic β-cells, which prevents the usual hyperinsulinemia response to HFD-induced insulin resistance to mimic late-stage T2D (16). As illustrated in Supplementary Fig. 1, male mice at age 8–10 weeks were fed an HFD (12492, 60% kcal from fat; Research Diets) or a normal diet (ND) (12450B, 10% kcal from fat; Research Diets) for 23 weeks. Body weight was monitored every week (Supplementary Fig. 1B), and blood glucose levels were monitored every other week (Supplementary Fig. 1C). After 8 weeks on HFD, intraperitoneal glucose tolerance test was performed to confirm glucose intolerance status (Supplementary Fig. 1D and E). After 15 weeks on HFD, mice with glucose intolerance were injected with a single dose of STZ (100 mg/kg) (Sigma) in 0.1 mol/L sodium citrate buffer (pH 4.5) to induce insulin deficiency and hyperglycemia. Seven days after the injection of STZ, mice with blood glucose levels ≥250 mg/dL were considered diabetic (Supplementary Fig. 1C). Mice fed an ND were injected with a single dose of 0.1 mol/L sodium citrate buffer (pH 4.5) as controls.
JTMT and WT mice at 4 weeks after T2D induction and db/db T2D mice at the age of 8–10 weeks were used in hind limb ischemia (HLI) experiments as reported in our previous studies (6,17). Briefly, under isoflurane anesthesia (1–3% isoflurane in 100% oxygen at a flow rate of 1 L/min), the right hind limbs were shaved and the entire right superficial femoral artery and vein (from just below the deep femoral arteries to the popliteal artery and vein) were ligated with 6-0 silk sutures, cut, and excised with an electric coagulator, and the skin was closed with 4-0 silk sutures. Real-time microcirculation imaging analysis was performed using a PeriCam perfusion speckle imager (PSI) (Perimed) to evaluate the foot pad blood perfusion ratio (ischemic limb [right]/normal limb [left]) at days 0, 3, 7, 14, 21, and 28 postsurgery. Mice then were euthanized, and HLI gastrocnemius and soleus muscles were collected for analysis of capillary density by staining with anti-CD31 antibody (Thermo Fisher Scientific). Some WT and JTMT mice were euthanized at day 3 and db/db mice at day 7 after HLI surgery according to our previous study (17) to document early changes in angiogenic signaling and EPC mobilization by staining peripheral blood with phycoerythrin-conjugated anti-mouse CD34 and allophycocyanin-conjugated anti-mouse vascular endothelial growth factor (VEGF) receptor (VEGFR) 2 (BioLegend) antibodies.
For cell therapy in db/db mice, 2.5 × 106 mouse bone marrow (BM)–derived MNCs were isolated from WT or JTMT mice (male, 8–10 weeks old). Then 2.5 × 106 BM-MNCs in 200 μL PBS were infused into db/db mice through the tail vein within 1 h after HLI. The same volume of suspension medium PBS was used as a vehicle control.
BM-EPC Isolation, Culture, Hypoxia Plus High-Glucose Treatment, and siRNA Transfection
EPCs were isolated from BM of WT and JTMT mice using density gradient centrifugation according to our previous studies (6). Cells were maintained in basal endothelial cell growth medium (PromoCell) supplemented with 10% FBS and growth supplement from bovine neural tissue (Sigma-Aldrich) at 37°C with 5% CO2 in a humidified incubator for 7 days; these cells were defined as early EPCs (6).
For in vitro mechanistic study, EPCs were cultured under hypoxic with high-glucose (HG) conditions to mimic diabetic ischemia. Briefly, EPCs were treated with either HG (25 mmol/L) or equivalent mannitol (osmotic control) and then placed in a tightly sealed hypoxia chamber (Billups-Rothenberg, Inc.) containing low oxygen gas (1% O2-5% CO2-balance N2) as previously reported (18). The hypoxia chamber was refilled with 1% O2-5% CO2 gas every 3 h to ensure hypoxia conditions during culture, and EPCs were treated for 24 h before harvest.
To knock down hypoxia-inducible factor 1α (HIF-1α) expression in early EPCs from JTMT mice, siRNAs against mouse HIF-1α or a Silencer Select Negative Control (Sigma-Aldrich) were transfected into EPCs using Lipofectamine 2000 (Thermo Fisher Scientific). The siRNA efficiency against HIF-1α was optimized in a preliminary study using two different siRNA sequences (Supplementary Fig. 2). Forty-eight hours after transfection, the level of HIF-1α and signal of stromal cell–derived factor 1 (SDF-1), VEGF, protein kinase B (Akt), phospho-Akt (p-Akt), 3-nitrotyrosine (3-NT), and 4-hydroxynonenal (4-HNE) were detected by Western blot. EPC functions, including tube formation, migration, and apoptosis, were examined as described below.
Tube Formation, Transendothelial Migration, Apoptosis, and Oxidative Stress Assays
Tube formation assay was used to measure the angiogenic capability, and transwell assay was used to evaluate the transendothelial migration (TEM) ability of EPCs according to our previous studies (6). Flow cytometry (Accuri C6 Plus; BD Biosciences) was used for EPC apoptotic detection with allophycocyanin-conjugated annexin V and propidium iodide (PI) staining as described previously (6). Early apoptotic cells were defined as annexin V+/PI−. Dihydroethidium (DHE) (Molecular Probes) staining was used to detect ROS levels in frozen sections and in cultured EPCs according to previous description (6). Nuclear DHE-positive staining indicates superoxide generation in cells. The fluorescence intensity was captured by microscopy (DP74; Olympus) and analyzed using Image J software.
Western Blot and ELISA
As previous described (6), gastrocnemius muscle and/or soleus muscle tissues and harvested EPCs were homogenized or lysed in ice-cold radioimmunoprecipitation lysis buffer (Santa Cruz Biotechnology). Total proteins were extracted and separated by 10% SDS-PAGE and transferred to nitrocellulose membranes (Bio-Rad). The membranes were probed with the primary antibodies against 3-NT, 4-HNE, HIF-1α, β-actin, Akt, and p-Akt (serine 473), SDF-1, and VEGF overnight and then further probed with appropriate horseradish peroxidase–conjugated secondary antibodies. Blots were visualized with SuperSignal West Femto Maximum Sensitivity Substrate (Thermo Fisher Scientific) and quantified with Quantity 5.2 software (Bio-Rad). Plasma SDF-1 and VEGF concentration were tested using Quantikine ELISA Kit (R&D Systems) according to the manufacturer’s protocol.
Statistical Analysis
All data are presented as mean ± SD. Statistical analysis was performed using GraphPad Prism 5.0 software. One-way or two-way ANOVA was used where appropriate followed by post hoc Tukey multiple comparison test. Statistical significance was considered as P < 0.05.
Data and Resource Availability
The data sets generated or analyzed during this study are included here in the published article. The original data sets and JTMT mice analyzed during this study will be available to the research community upon reasonable request.
Results
JTMT Mice Exhibit Improved Blood Perfusion and Ischemic Angiogenesis in Diabetic HLI
The T2D model was established by HFD/STZ induction (Supplementary Fig. 1). Both WT and JTMT mice on HFD for 8 weeks were obese and glucose intolerant as reflected by significantly increased body weight (Supplementary Fig. 1B) and impaired intraperitoneal glucose tolerance test (Supplementary Fig. 1D and E) yet without significant changes in fed blood glucose levels (Supplementary Fig. 1C) compared with the mice on ND. After induction with a single dose of STZ, all mice on HFD were hyperglycemic (Supplementary Fig. 1C). No significant differences were observed in body weight, blood glucose levels, and glucose tolerance between WT and JTMT mice with or without diabetes (Supplementary Fig. 1B–E).
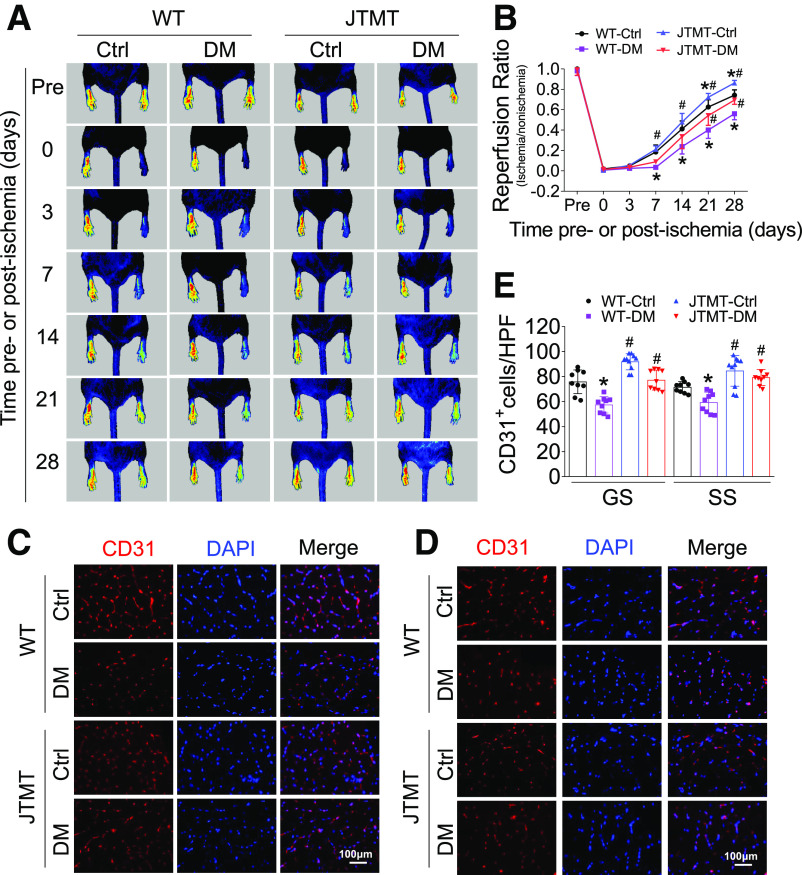
To determine whether endothelial MT overexpression was protective against diabetic ischemia, HLI was induced by femoral artery ligation (Supplementary Fig. 1). Blood perfusion of the hind limb footpad was evaluated by a PSI on days 3, 7, 14, 21, and 28 after surgery. The results showed that diabetes impaired hind limb blood perfusion recovery in both WT and JTMT mice in a time-dependent manner (Fig. 1A and B). However, MT endothelial overexpression in JTMT mice promoted greater flow recovery at days 7–28 after HLI surgery compared with diabetic WT mice, although statistical differences in blood flow levels were only observed at days 21 and 28 (Fig. 1A and B). JTMT mice with or without diabetes also had increased capillary density in both gastrocnemius and soleus muscles measured 28 days after HLI compared with diabetic WT mice (Fig. 1C–E). Thus, endothelial MT overexpression promoted blood flow recovery despite not altering systemic diabetes, indicating a selective augmentation of ischemic angiogenesis.
Figure 1.
JTMT mice exhibit improved blood perfusion and angiogenesis in diabetic HLI. T2D was induced by HFD plus STZ in JTMT and littermate WT mice. The blood perfusion was monitored before and at 0, 3, 7, 14, 21, and 28 days after HLI using a PeriCam PSI (A) and quantified by Image J (B). Ischemic hind limb muscle tissues were then collected at day 28 after HLI. Transverse sections of gastrocnemius (GS) muscle (C) and soleus (SS) muscle (D) were stained for CD31 to enumerate CD31+-stained cell number as a proxy of capillary density. Capillary density was expressed as CD31+ capillaries per high-power field (HPF) (E), and DAPI was used to recognize nuclei. Data are mean ± SD. n ≥ 9 mice/group. *P < 0.05 vs. WT control (Ctrl); #P < 0.05 vs. WT diabetes mellitus (DM).
Endothelial MT Overexpression Preserves EPC Mobilization and Homing and Alleviates Oxidative Stress in Diabetic HLI
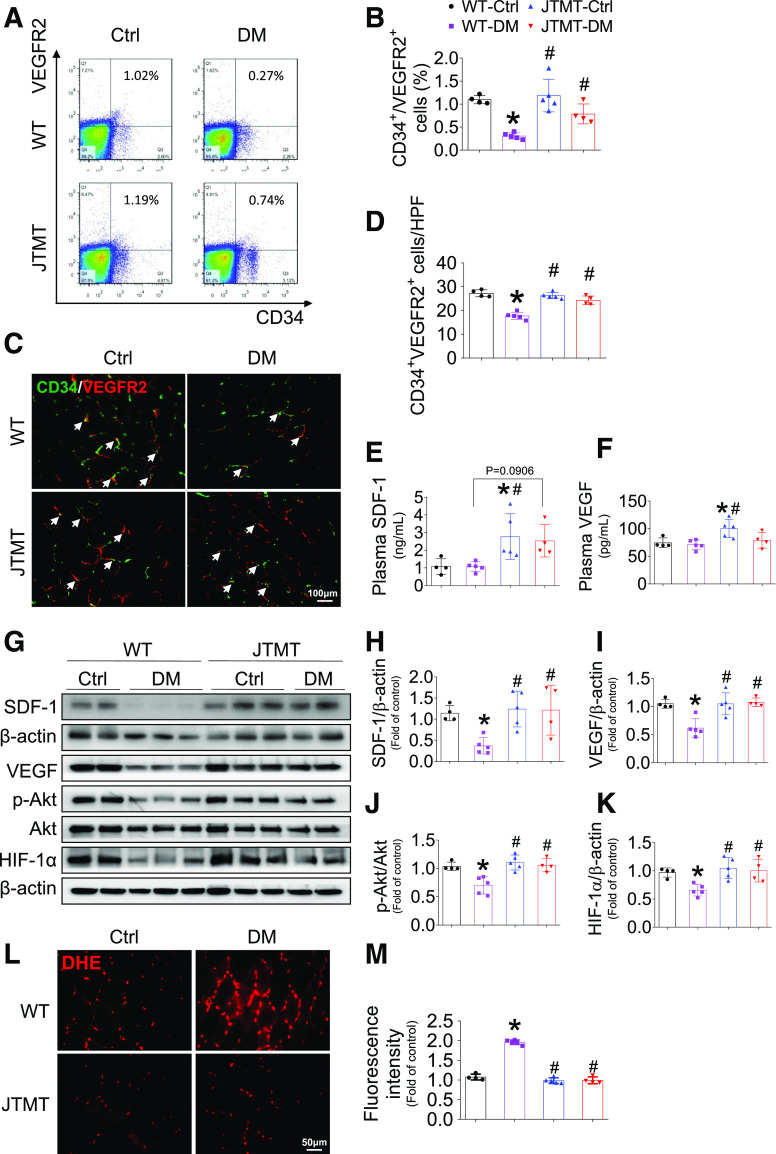
Because EPCs are important contributors in ischemic angiogenesis (6), we quantified the circulating number of EPCs by flow cytometry using two cell surface markers, CD34+/VEGFR2+, at day 3 after HLI surgery. The results demonstrate that following HLI surgery, diabetes in WT mice reduced EPC mobilization compared with healthy WT controls and that EPC mobilization in diabetes was better preserved in diabetic JTMT mice compared with diabetic WT mice; however, JTMT did not significantly alter EPC mobilization in mice without diabetes (Fig. 2A and B). Mobilization and homing of EPCs to ischemic sites are key steps in ischemic angiogenesis (6). Homing EPCs (CD34+/VEGFR2+) in ischemic skeletal muscle were significantly reduced in diabetic WT mice yet preserved in healthy and diabetic JTMT transgenic mice (Fig. 2C and D).
Figure 2.
Endothelial MT overexpression preserves EPC mobilization and homing and alleviates oxidative stress in diabetic HLI. T2D was induced as described in Fig. 1. At day 3 after HLI surgery, peripheral blood was collected to evaluate the percentage of EPCs (CD34+/VEGFR2+) in circulation by a flow cytometry assay (A and B); transverse sections of gastrocnemius muscle were stained by CD34 and VEGFR2 (C) to evaluate EPC infiltration (arrow); the infiltration incidence was expressed as CD34+/VEGFR2+ cells per high-power field (HPF) (D). The plasma SDF-1 (E) and VEGF (F) levels were detected by ELISA. The expression of SDF-1 (G and H), VEGF (G and I), and HIF-1α (G and K) and phosphorylation of Akt (G and J) in gastrocnemius muscle were detected by Western blot, with β-actin used as loading control. The transverse sections of gastrocnemius muscle were also used to probe superoxide production by DHE staining (L), and the fluorescence intensity was quantified by Image J (M). Data are mean ± SD. n ≥ 4 mice/group. *P < 0.05 vs. WT control (Ctrl); #P < 0.05 vs. WT diabetes mellitus (DM).
SDF-1 and VEGF are major proangiogenic factors that promote endothelial repair by mobilizing and recruiting EPCs from BM to injured tissues (6,17). Thus, we tested the hypothesis that enhanced EPC mobilization and infiltration in JTMT transgenic mice resulted from induction of SDF-1 and VEGF. In partial confirmation, blood levels of SDF-1 (Fig. 2E) and VEGF (Fig. 2F) after HLI were elevated in control JTMT transgenic mice compared with WT controls. However, no differences were observed in blood levels of SDF-1 and VEGF as a result of diabetes in either WT or JTMT transgenic mice. In contrast, diabetes led to a significant reduction of SDF-1 (Fig. 2G and H) and VEGF (Fig. 2G and I) expression in ischemic muscle of WT mice. However, SDF-1 (Fig. 2G and H) and VEGF (Fig. 2G and I) expression in ischemic muscle of JTMT transgenic mice were preserved in diabetes and significantly higher than levels in WT diabetic mice. SDF-1– and VEGF-mediated activation of Akt plays a critical role in ischemic angiogenesis in vivo and EPC function in vitro (6,19). Consistent with the changes of SDF-1 and VEGF expression in ischemic muscle, phosphorylation status (activation) of Akt was reduced in WT diabetic mice, but preserved in diabetic JTMT transgenic mice (Fig. 2G and J). HIF-1α is considered a central oxygen sensor in eukaryotic cells (20) that orchestrates ischemic angiogenesis through transcription regulation of several proangiogenic factors, including SDF-1 and VEGF (21,22). Thus, we tested whether augmented levels of SDF-1 and VEGF and Akt signaling was paralleled by preservation of HIF-1α expression. Supporting this idea, diabetes markedly reduced HIF-1α expression in WT mice, whereas HIF-1α expression was preserved in diabetic JTMT transgenic mice (Fig. 2G and K). However, these changes were not observed in the contralateral (nonischemic) gastrocnemius muscles between WT and JTMT mice with or without diabetes (Supplementary Fig. 3). Together, these findings indicate that the improved angiogenesis and blood perfusion in JTMT transgenic mice was a likely consequence of MT preserving HIF-1α, SDF-1, VEGF, and Akt signaling and EPC mobilization and homing in diabetic HLI.
Oxidative stress plays a causal role in diabetes-induced impairment of EPC mobilization, homing, and ischemic angiogenesis (6,23), so we assessed whether endothelial MT overexpression prevented diabetes-induced oxidative stress in ischemic muscle. The results show that diabetes increased superoxide production in ischemic tissue in WT mice, as measured by DHE staining, and this effect of diabetes was completely prevented by endothelial MT overexpression in diabetic JTMT mice (Fig. 2L and M). Furthermore, using 3-NT (Supplementary Fig. 4A) and 4-HNE (Supplementary Fig. 4B) as oxidative damage markers, we obtained similar results: MT overexpression completely prevented diabetes-induced elevation of 3-NT and 4-HNE levels compared with WT mice with diabetes. These findings imply that endothelial cell MT overexpression preserves EPC mobilization and homing in part attributable to MT alleviation of diabetes-induced ischemic oxidative stress in diabetic HLI.
MT-Overexpressing BM-MNC Transplantation Improves Blood Perfusion and Ischemic Angiogenesis in Diabetic HLI
To test whether MT endothelium overexpression in EPCs enhances ischemic angiogenesis and blood perfusion in diabetes, we performed transplantation of BM-MNCs into db/db mice with HLI. BM-MNCs were used because they contain all the EPCs, and fresh BM-MNCs (albeit a mix of leukocytes and stem cells) avoid potential issues with culturing EPCs (24,25).
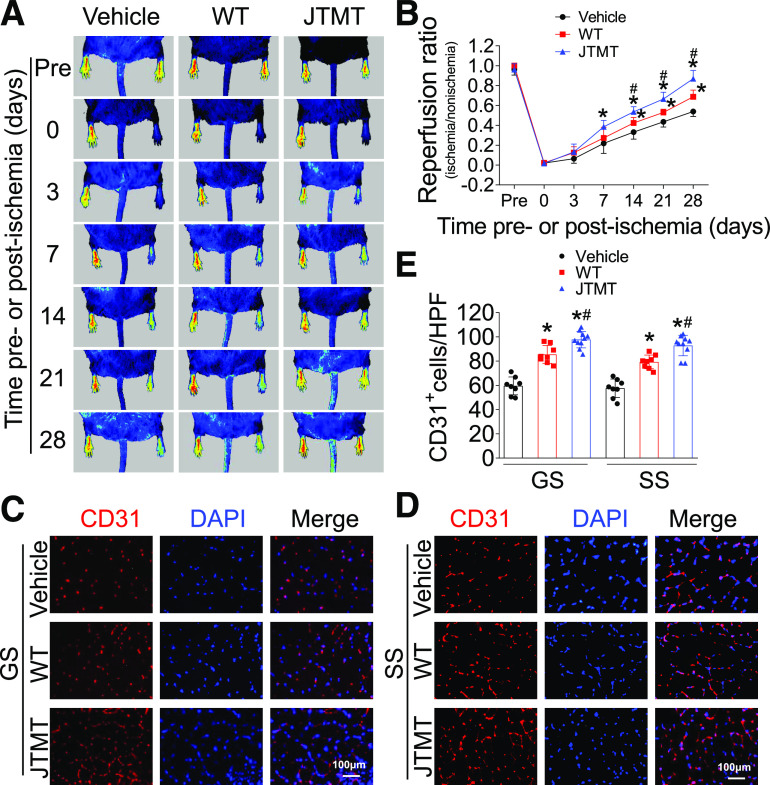
We compared MT expression in BM-MNCs with early BM-EPCs before transplantation to ensure their level of MT overexpression. MT expression in both BM-MNCs and BM-EPCs of JTMT mice was significantly higher than in WT controls, similar to what was observed in aorta; no obvious difference in MT expression levels between MNCs and EPCs was observed (Supplementary Fig. 5). Transplantation with WT or JTMT MNCs had no obvious effects on hyperglycemia or body weight in db/db mice compared with vehicle-infused db/db mice (data not shown). Notably, transplantation of WT or JTMT BM-MNCs time dependently improved hind limb blood perfusion at all time points from days 7 to 28 after HLI compared with db/db mice infused with vehicle (Fig. 3A and B). In support of increased blood flow, mice with JTMT BM-MNC transplanted had increased capillary density in both gastrocnemius and soleus muscles compared with mice receiving WT BM-MNC infusion (Fig. 3C–E).
Figure 3.
MT-overexpressing BM-MNC transplantation improves blood perfusion and ischemic angiogenesis in db/db mice with HLI. BM-MNCs from WT and JTMT mice were transplanted in db/db mice immediately after HLI; MNC suspension buffer was used as vehicle control. The blood perfusion was monitored before and at 0, 3, 7, 14, 21, and 28 days after HLI using a PeriCam PSI (A) and quantified by Image J (B). Ischemic hind limb muscle tissues were then collected at day 28 after HLI. Transverse sections of gastrocnemius (GS) muscle (C) and soleus (SS) muscle (D) were stained by CD31 to evaluate angiogenesis. Capillary density was expressed as CD31+ capillaries per high-power field (HPF) (E), and DAPI was used to recognize nuclei. Data are mean ± SD. n ≥ 8 mice/group. *P < 0.05 vs. vehicle; #P < 0.05 vs. WT.
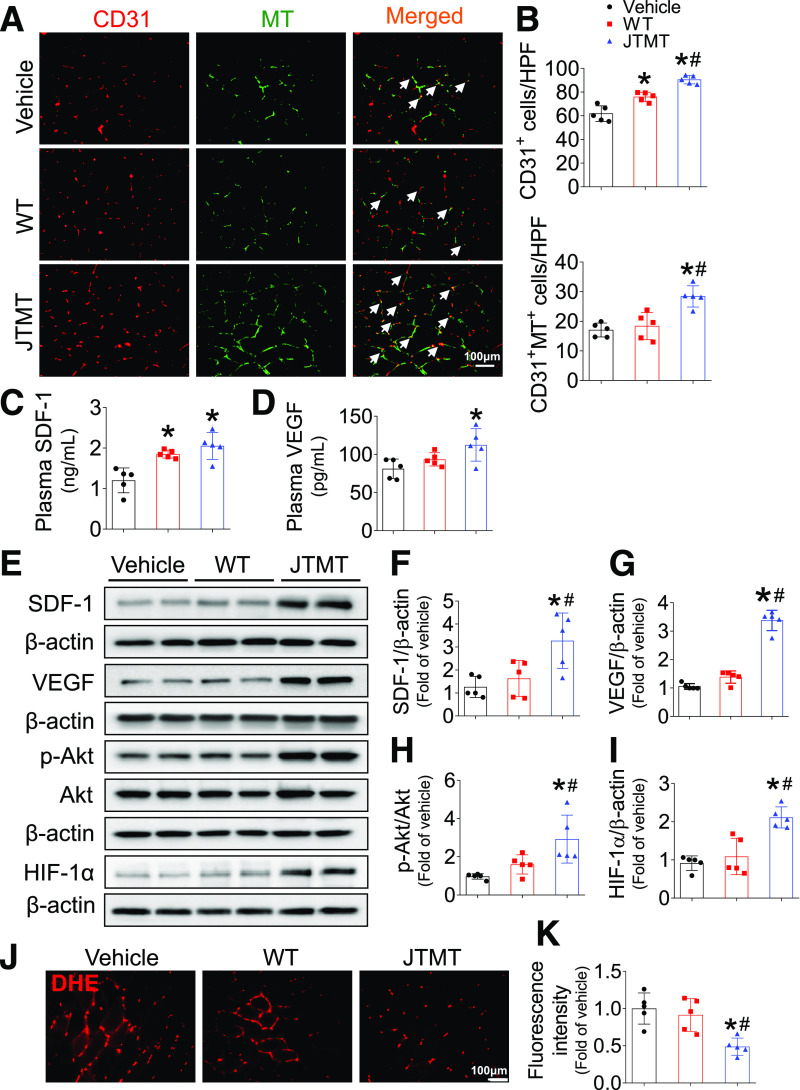
To test whether the transplanted MT-overexpressing MNCs were directly involved in angiogenesis in diabetic ischemia, we monitored the distribution of MT-overexpressing BM-MNCs in ischemic gastrocnemius muscle at day 7 after HLI surgery. The db/db mice infused with either WT BM-MNCs or JTMT BM-MNCs had a higher number of CD31+ cells compared with vehicle-infused db/db mice. Notably, a further increase in CD31+ cells was observed in db/db mice infused with JTMT BM-MNCs when compared with db/db mice infused with WT BM-MNCs (Fig. 4B). Only the JTMT BM-MNCs, identified as MT and CD31 double-positive stained cells, were clearly involved in capillary formation in ischemic gastrocnemius muscle in transplanted db/db mice (Fig. 4A and B), whereas a similar and lower level of double-positive stained cells were observed in vehicle- and WT BM-MNC–infused db/db mice (Fig. 4A and B). Plasma levels of proangiogenic proteins showed that blood SDF-1 levels were elevated with infusion of either BM-MNCs (Fig. 4C), yet only transplantation of JTMT BM-MNCs elevated both SDF-1 and VEGF blood levels (Fig. 4C and D) compared with controls. In agreement with these findings, transplantation of JTMT BM-MNCs elevated SDF-1, VEGF, Akt, and HIF-1α signaling in ischemic gastrocnemius muscle (Fig. 4E–I), which were not observed in the contralateral nonischemic gastrocnemius muscles between db/db mice infused with WT MNCs and JTMT MNCs (Supplementary Fig. 6). Moreover, transplantation of JTMT BM-MNCs was associated with lower levels of oxidative damage in ischemic gastrocnemius muscle as demonstrated by a reduction in superoxide production (Fig. 4J and K), 3-NT staining (Supplementary Fig. 7A), and 4-HNE protein staining (Supplementary Fig. 7B) compared with controls. Taken together, these findings indicate that the improved therapeutic efficacy of JTMT BM-MNC transplantation may be attributed to MT endothelial overexpression that preserves EPC angiogenic signaling and function as a consequence of MT antioxidative capacity.
Figure 4.
MT-overexpressing BM-MNCs involved in ischemic angiogenesis in db/db diabetic mice with HLI. BM-MNCs transplantation was performed as described in Fig. 3. At day 7 after HLI surgery, transverse sections of gastrocnemius muscle were stained by CD31 and MT (A) to evaluate the incorporation of MT-overexpressing BM-MNCs in capillaries (arrows), the capillary density was expressed as CD31+ cells per high-power field (HPF), and the incorporation incidence was expressed as CD31+/MT+ cells per HPF (B). The plasma SDF-1 (C) and VEGF (D) levels were detected by ELISA. The expression of SDF-1 (E and F), VEGF (E and G), and HIF-1α (E and I) and phosphorylation of Akt (E and H) in gastrocnemius muscle were detected by Western blot, with β-actin used as loading control. The transverse sections of gastrocnemius muscle were also used to probe superoxide production by DHE staining (J), and the fluorescence intensity was quantified by Image J (K). Data are mean ± SD. n = 5 mice/group. *P < 0.05 vs. vehicle; #P < 0.05 vs. WT.
Endothelial MT Overexpression Protects EPCs From Effects of HG and Hypoxia
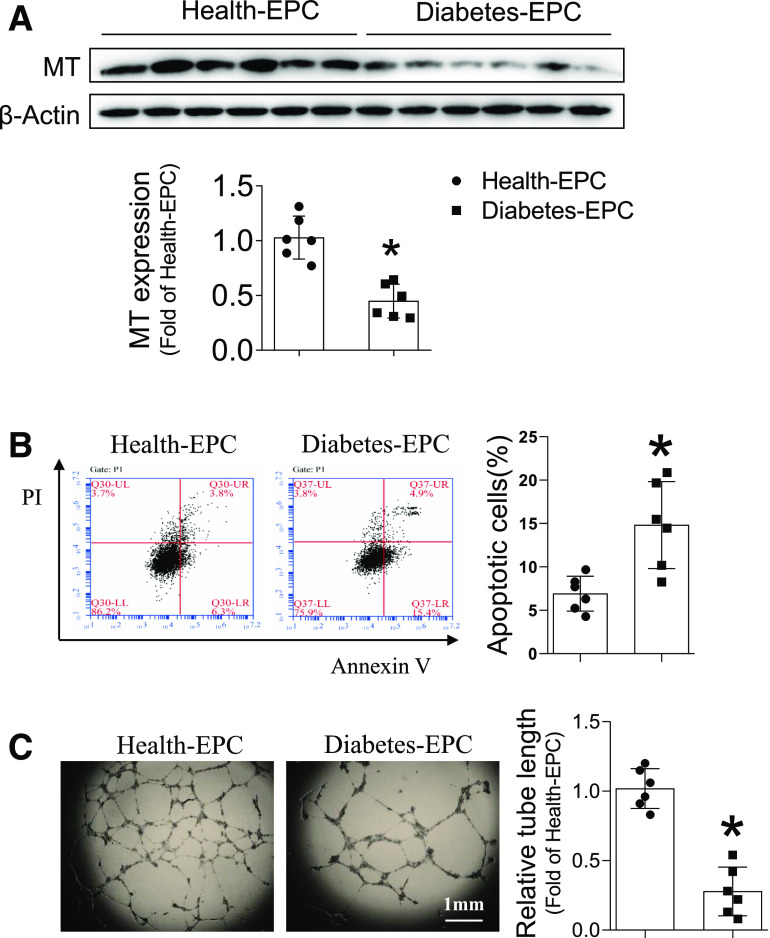
To test the role of MT in EPC function in diabetes, human EPCs were isolated from healthy subjects and patients with diabetes. Strikingly, EPCs from patients with diabetes exhibited significantly decreased MT expression (Fig. 5A), which was accompanied by significantly increased cell apoptosis (Fig. 5B) and impaired tube formation (Fig. 5C). These findings indicate that reduced MT expression plays an important role in EPC dysfunction in patients with diabetes.
Figure 5.
Diabetes impairs MT expression and function of EPC in patients. EPCs were collected from peripheral blood of healthy control subjects and patients with diabetes. The expression of MT was detected by Western blot, with β-actin used as loading control (A). The apoptosis was analyzed by flow cytometry using annexin V/PI staining (B). The angiogenic function was evaluated by tube formation assay (C). Data are mean ± SD. n = 6/group. *P < 0.05 vs. healthy subject EPCs (Health-EPC).
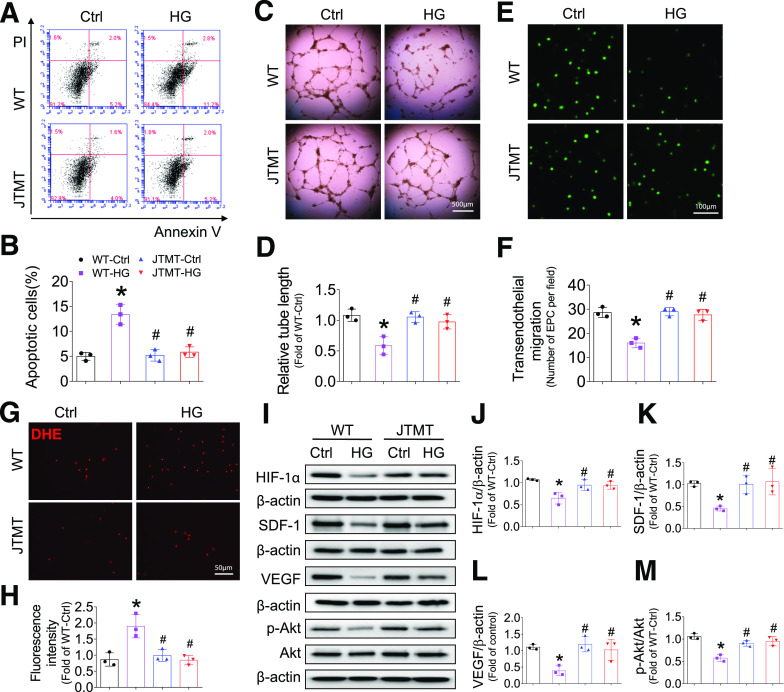
To test whether MT is proangiogenic in EPCs, MT expression in early mouse EPCs was confirmed (Supplementary Fig. 5), and the angiogenic functions of mouse EPCs were assayed under HG and hypoxia-mimicked hyperglycemic and ischemic conditions in vivo. Strikingly, EPCs from JTMT mice were almost completely resistant to HG/hypoxia-induced apoptotic cell death (Fig. 6A and B). HG and hypoxia significantly impaired tube formation in WT EPCs (Fig. 6C and D); however, these effects were eliminated in JTMT EPCs (Fig. 6C and D). Because TEM is essential for EPC homing to sites of blood vessel repair and formation, TEM in WT EPCs was impaired by HG/hypoxia treatment, yet SDF-1–induced migration was completely preserved in JTMT EPCs (Fig. 6E and F). As a major factor responsible for the diabetes-associated dysfunction of EPCs (6,26), ROS levels and oxidative damage induced by HG/hypoxia were markedly reduced by MT overexpression in JTMT EPCs compared with WT EPCs with HG/hypoxia treatment, as measured by DHE staining (Fig. 6G and H) and by oxidative markers 3-NT (Supplementary Fig. 8A) and 4-HNE (Supplementary Fig. 8B). More importantly, MT protection of EPC function was accompanied by protection from HG/hypoxia-induced impairment of HIF-1α/SDF-1/VEGF/Akt signaling (Fig. 6I–M), which is fully consistent with MT-associated protection of this signaling pathway in vivo (see Figs. 2 and 4).
Figure 6.
Endothelial MT overexpression protects EPCs from HG and hypoxia-induced apoptosis and angiogenic dysfunction and impaired HIF-1α/SDF-1/Akt signaling. BM-EPCs from WT and JTMT mice were exposed to HG (25 mmol/L) and hypoxia for 24 h; the equivalent concentration of mannitol was used as osmotic control (Ctrl). The apoptosis was analyzed by flow cytometry using annexin V/PI staining (A and B). The angiogenic function was evaluated by tube formation assay (C and D). The migration capability was evaluated by TEM assay (E and F). The oxidative damage was evaluated by DHE stain of superoxide production (G and H). The expression of HIF-1α (I and J), SDF-1 (I and K), VEGF (I and L), and phosphorylation of Akt (I and M) were tested by Western blot, with β-actin used as loading control. Three independent experiments were performed. Data are mean ± SD. *P < 0.05 vs. WT-Ctrl; #P < 0.05 vs. WT-HG.
Knockdown of HIF-1α Abolishes MT Overexpression Protecting EPC Function
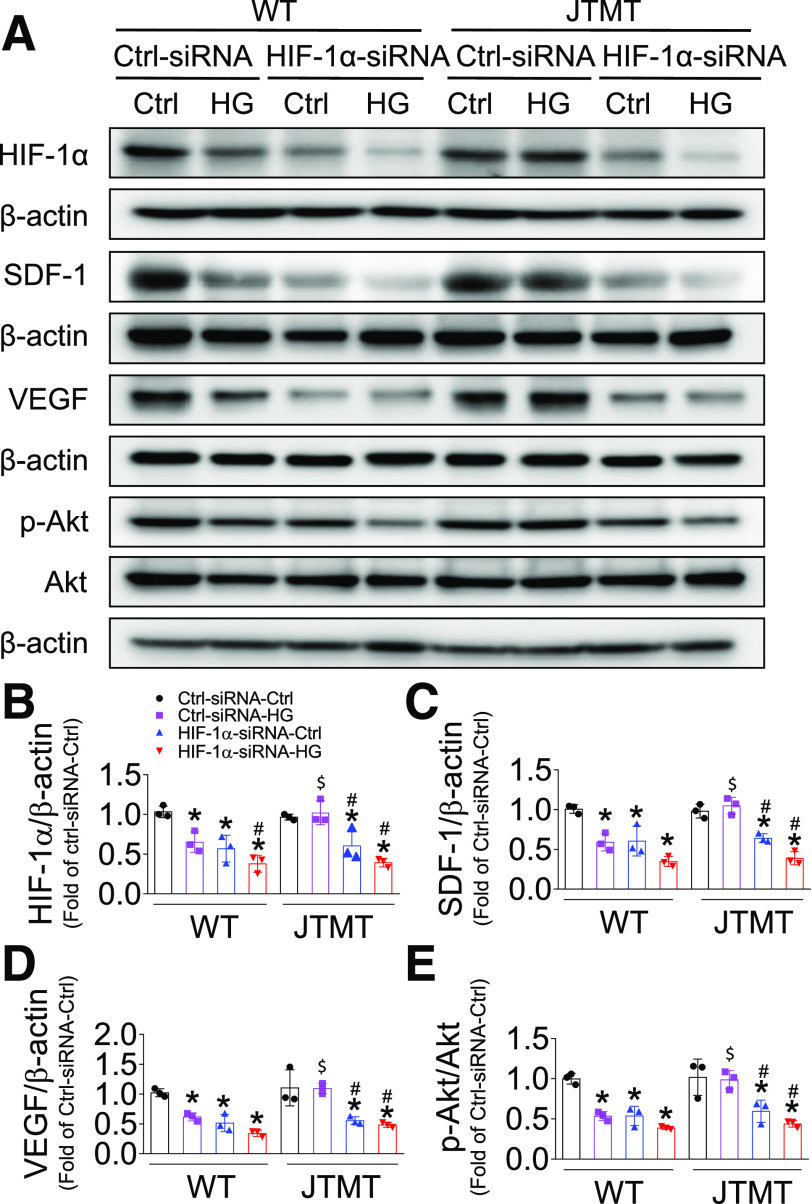
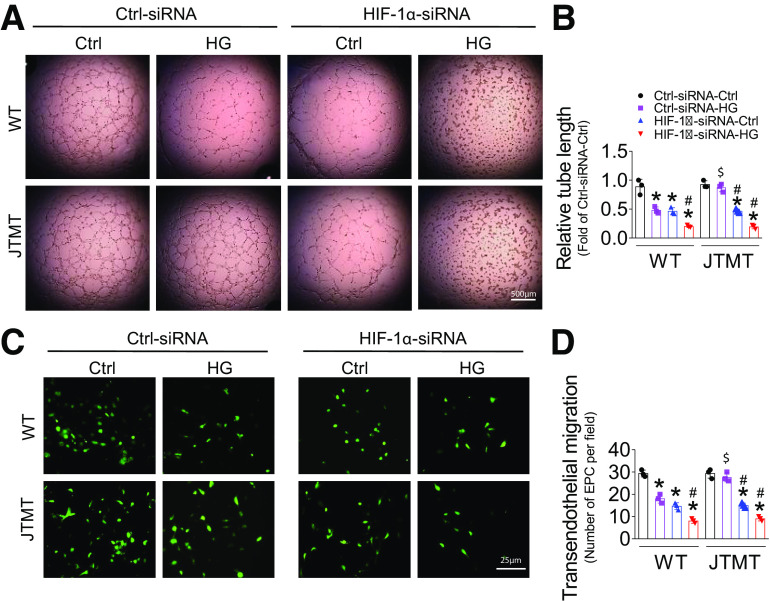
To test whether HIF-1α plays a pivotal role in MT protection of EPC function, we performed knockdown of HIF-1α gene expression with HIF-1α siRNA in both WT and JTMT EPCs. Consistent with the findings shown in Fig. 6, the nonsense control siRNA had no obvious effects on MT protection from HG/hypoxia-induced impairment of HIF-1α expression in JTMT EPCs compared with WT EPCs (Fig. 7A and B). However, the specific siRNA against HIF-1α significantly reduced HIF-1α expression compared with nonsense control siRNA with either mannitol/hypoxia or HG/hypoxia treatment, which was more apparent under HG/hypoxia conditions in both WT and JTMT EPCs (Fig. 7A and B). Similarly, siRNA knockdown of HIF-1α markedly decreased SDF-1 (Fig. 7A and C) and VEGF (Fig. 7A and D) protein levels as well as Akt phosphorylation (Fig. 7A and E); the magnitude of reduction of SDF-1 and VEGF levels and Akt phosphorylation was more apparent under HG/hypoxia conditions in both WT EPCs and JTMT EPCs (Fig. 7A–E). Furthermore, knockdown of HIF-1α not only aggravated HG/hypoxia-induced impairment in EPC tube formation (Fig. 8A and B) and TEM (Fig. 8C and D) in WT EPCs but also blocked the protective effects of MT overexpression against the effects of HG/hypoxia treatment on EPC tube formation (Fig. 8A and B) and TEM (Fig. 8C and D) in JTMT EPCs. More importantly, the essential role of HIF-1α in MT overexpression preserving EPC SDF-1/VEGF/Akt signaling and angiogenic function was validated with a second specific siRNA sequence knockdown of HIF-1α (Supplementary Figs. 9 and 10). These findings indicate that HIF-1α is equally important for both WT and JTMT EPC preservation of SDF-1/VEGF/Akt signaling and angiogenic function, and MT endothelial overexpression augments the HIF-1α/SDF-1/VEGF/Akt signaling pathway in promoting EPC survival and function. However, HIF-1α knockdown did not affect the antioxidative capacity of JTMT EPCs (Supplementary Fig. 11), demonstrating that MT antioxidant function is upstream of HIF-1α.
Figure 7.
Knockdown of HIF-1α abolishes MT overexpression, protecting EPCs from HG and hypoxia-impaired HIF-1α/SDF-1/Akt signaling. BM-EPCs from WT and JTMT mice were transfected with the specific siRNA against HIF-1α for 48 h; the Silencer Select Negative Control was used as control siRNA (Ctrl-siRNA). The transfected EPCs were then exposed to HG (25 mmol/L) and hypoxia for an additional 24 h; the equivalent concentration of mannitol was used as osmotic control (Ctrl). The expression of HIF-1α (A and B), SDF-1 (A and C), VEGF (A and D), and phosphorylation of Akt (A and E) was tested by Western blot, with β-actin used as loading control. Three independent experiments were performed. Data are mean ± SD. *P < 0.05 vs. the respective Ctrl-siRNA-Ctrl; #P < 0.05 vs. the respective Ctrl-siRNA-HG; $P < 0.05 vs. WT-Ctrl-siRNA-HG.
Figure 8.
Knockdown of HIF-1α abolishes MT overexpression, protecting EPCs against HG and hypoxia-induced EPC dysfunction. BM-EPCs from WT and JTMT mice were transfected with the specific siRNA against HIF-1α for 48 h; the Silencer Select Negative Control was used as control siRNA (Ctrl-siRNA). Then the transfected EPCs were exposed to HG (25 mmol/L) and hypoxia for an additional 24 h, the equivalent concentration of mannitol was used as osmotic control (Ctrl). The angiogenic function was evaluated by tube formation assay (A and B). The migration capability was evaluated by TEM assay (C and D). Three independent experiments were performed. Data are mean ± SD. *P < 0.05 vs. Ctrl-siRNA-Ctrl; #P < 0.05 vs. Ctrl-siRNA-HG; $P < 0.05 vs. WT-Ctrl-siRNA-HG.
Discussion
In the current study, we found that diabetes or diabetic conditions reduce EPC numbers and impair EPC function, which is associated with aggravated oxidative stress. Endothelial overexpression of MT improves the mobilization and angiogenic function of EPCs in diabetic limb ischemia, accompanied by alleviated oxidative stress. Furthermore, transplantation of MT-overexpressing MNCs improves blood reperfusion and angiogenesis of diabetic limb ischemia, providing a novel potential therapeutic strategy to treat this pathology. Importantly, the benefits of MT on EPCs are predominantly mediated by the HIF-1α/SDF-1/VEGF/Akt pathway under diabetic ischemia conditions.
The most important finding of this study is that endothelium-selective scavenging oxidative stress by MT is sufficient to prevent diabetes-induced impairment in ischemia angiogenesis. Several previous studies have demonstrated the importance of MT in preventing diabetes-induced damage in cardiomyocytes (8–11) and in renal podocytes (12). Similarly, our previous study also showed renoprotection in JTMT mice (13) against several highly refractory complications of diabetic nephropathy. Consistent with our current study results, mice with global MT gene deficiency have impaired collateral flow recovery after the induction of acute HLI wherein MT deficiency is associated with endothelial cells, smooth muscle cells, and macrophage dysfunction in collateral remodeling (27). Because of ubiquitous MT expression, the specific contribution of endothelial MT in maintaining vascular homeostasis in diabetic limb ischemia was unclear. Thus, in this study, we provide direct evidence that endothelial MT overexpression provides significant promotion of blood flow recovery and angiogenesis in diabetic HLI (Fig. 1). Although transgenic approaches are challenging for clinical use, MT is a highly inducible protein with many inducers, such as Zn. Previously, we demonstrated that upregulation of cardiac MT by Zn supplementation provides significant protection against chronic cardiomyopathy in diabetic mice (9,28). Furthermore, Lim et al. (29) demonstrated that Tat sequence–fused MT recombinant protein effectively inhibits apoptosis, reduces fibrosis area, and enhances cardiac function in an ischemia/reperfusion rat myocardial infarction model. Therefore, induction of endogenous MT expression or supplementation of exogenous MT protein may be a practical approach to treat (or prevent) diabetic ischemic diseases.
Previous studies have shown EPCs to be an important contributor to neovascularization through mobilization, recruiting to injury location, and participating in angiogenesis and tissue repair in limb ischemia (30,31). Reduced number and function of EPCs are causally associated with diabetes-induced impairment in vasculogenesis (26,32). The current study demonstrates that the augmented angiogenesis in JTMT transgenic mice is attributable in part to an improved functionality of EPCs, as evidenced by increased mobilization of EPCs into the circulation and infiltration of EPCs into injured muscle tissue (Fig. 2). In addition, BM-MNCs are a major source of EPCs in vivo (24,33). Transplantation of BM-MNCs in clinical trials has provided solid evidence of feasibility and safety for treatment of myocardial infarction and peripheral limb ischemia (24,31,34). Thus, in the current study, transplantation of MT-overexpressed BM-MNCs showed greater therapeutic effects on ischemic blood reperfusion and angiogenesis than WT BM-MNCs in db/db mice with HLI (Fig. 3). We attribute the functional benefits of BM-MNCs to EPCs for several reasons: 1) BM-MNCs and BM-EPCs from JTMT mice have similar levels of MT, 2) there is more MT-positive staining in muscle of db/db mice transplanted with BM-MNCs of JTMT mice, and 3) cultured BM-EPCs of JTMT mice are protected from ex vivo hypoxia and HG-induced apoptosis and capillary tube formation and migration dysfunction (Fig. 6). These observations are consistent with previous reports showing that endothelium-specific AMPK overexpression protects against diabetes-induced aggravation of vascular injury by promoting mobilization and angiogenic function of EPCs (35), and studies showing that copper-Zn superoxide dismutase (CuZnSOD)–deficient mice demonstrate a reduction in the number of EPCs in the BM and an impairment in the functionality of isolated EPCs (36). Therefore, endothelium-selective overexpression of MT appears to exert its protective effects against diabetes-induced impairment in ischemic angiogenesis.
Accumulating evidence demonstrates that oxidative stress contributes to diabetic EPC dysfunction and impairment of vascular regeneration (6,26). Overexpression of the heme oxygenase-1 (HO-1) gene in hyperglycemic rats brings reduced superoxide production and decreases endothelial cell sloughing (37). Likewise, EPCs isolated from CuZnSOD-deficient mice have increased oxidative stress and a reduced ability to migrate and integrate into capillary-like networks (36). Importantly, the functional activities of CuZnSOD-deficient EPCs are rescued by treatment with the SOD mimetic Tempol (a membrane-permeable radical scavenger) (36). We also show herein that improved EPC function in diabetic JTMT transgenic mice coincides with a reduction in superoxide production and oxidative damage in ischemic muscle tissues (Fig. 2), JTMT BM-MNC transplanted into db/db mice (Fig. 4), and isolated EPCs exposed to diabetic conditions (Fig. 6). Thus, we support a central upstream role of oxidative stress as an important mediator of diabetic dysfunction, and MT-diminished oxidative stress accounts for the protective effects against diabetes-induced dysfunction of EPCs.
A novel finding of our present study is an uncovering of the critical relationship between MT and HIF-1α as a downstream target that mediates EPC mobilization and subsequent improved neovascularization. HIF-1 is a transcription factor that responds to decreased oxygen in the cellular environment and plays critical roles in angiogenesis (38). HIF-1 is a heterodimer composed of two subunits: the constitutively expressed HIF-1β and oxygen-regulated HIF-1α (39). HIF-1α is an important regulator of angiogenesis through activation of EPC function (40). HIF-1α deficiency impairs the expression of multiple angiogenic cytokines, mobilization of angiogenic cells, maintenance of tissue viability, and recovery of limb perfusion following HLI (41). Administration of an adenovirus encoding a constitutively active form of HIF-1α improves blood flow recovery in older mice to levels similar to those in young mice (41) and improves cardiac function after myocardial infarction in rats (42). The current study demonstrates that protein expression of HIF-1α decreases in ischemic muscle of diabetic WT mice, and it is similarly suppressed in WT EPCs treated with HG plus hypoxia in culture. In contrast, HIF-1α is markedly preserved in JTMT diabetic mice, in JTMT BM-MNCs transplanted into db/db mice, and in JTMT EPCs exposed to HG plus hypoxia (see Figs. 2, 4, and 6). Collectively, these results reveal an endothelial-specific (and EPC-specific) benefit of MT expression that preserves HIF-1α in conditions of ischemia and hyperglycemia as encountered in critical limb ischemia and PAD in patients with T2D.
It is clear that HIF-1α is an indispensable mediator of EPC-mediated vascular repair, including survival, mobilization, and differentiation of BM-EPCs, as well as migration, recruitment, and adhesion of EPCs to sites of injury by regulating multiple angiogenic factors (40–42). In particular, SDF-1 and VEGF, critical angiogenic factors downstream of HIF-1α (21,22,40), are known key regulators of ischemic angiogenesis in diabetes. SDF-1 activates angiogenesis through its cognate receptors CXCR4 and/or CXCR7, while VEGF regulates angiogenesis through its receptors VEGFR1 and/or VEGFR2 along with their downstream signaling mediators, such as Akt, in EPCs (6,22). In the current study, we found that JTMT transgenic mice and JTMT BM-MNC–infused db/db mice are resistant to diabetes-induced reduction in SDF-1 and VEGF expression as well as to reduced Akt phosphorylation in ischemic muscle (Figs. 2 and 4). A previous study reported elevated basal Akt phosphorylation in muscle tissues in hyperinsulinemic obese animals (43), whereas we observe reduced Akt phosphorylation in ischemic muscle tissues in WT diabetic mice compared with controls (Fig. 2). This difference could not be fully explained by the absence of hyperinsulinemia in the present HFD/STZ diabetic model because in the contralateral nonischemic muscle of the same diabetic animals, Akt phosphorylation levels were normal (Supplementary Fig. 3). More importantly, MT transgene prevented the decline in Akt phosphorylation levels in diabetic ischemic muscle tissues (Fig. 2). Therefore, preservation of ischemia-stimulated HIF-1α/SDF-1/VEGF signaling by MT could be an important factor in preventing Akt phosphorylation levels from declining in ischemic muscle tissues of diabetic animals. The protective effects of MT transgene could be attributable to both direct contribution of MT-expressing BM-MNCs infiltrated in ischemic muscle tissues and indirect mechanisms of these cells through a yet-to-be-defined paracrine action that preserves HIF-1α signaling in skeletal muscle cells and endothelial cells in the ischemic tissues. This is also demonstrated in EPCs treated with HG plus hypoxia (Fig. 6), providing a consistent mechanism across platforms. These protective signaling effects of MT overexpression as well as preservation of EPC functions are largely abrogated by siRNA knockdown of HIF-1α in EPCs (Figs. 7 and 8 and Supplementary Figs. 9 and 10), providing further mechanistic support. Because MT-augmented antioxidative capability is unaffected by HIF-1α knockdown (Supplementary Fig. 11), we conclude that HIF-1α lies downstream of MT and diabetic oxidative stimuli in EPCs. Several mechanisms underlying the potential deleterious effect of HG or hyperglycemia on HIF-1α activity under hypoxic conditions have been proposed (44,45). One major mechanism is the direct effects of HG- or hyperglycemia-induced overproduction of ROS that inhibits HIF-1α expression through repression of Ras-related C3 botulinum toxin substrate 1 expression or promotes HIF-1α degradation through activated proline hydroxylase in the presence of iron and increased ubiquitin-proteasome activity (44,45). Therefore, we hypothesize that MT protects diabetic EPC function predominantly through protection of HIF-1α transcriptional activity by scavenging ROS. However, the specific mechanism by which MT protects HIF-1α expression (in the presence of hypoxia and hyperglycemia) remains to be elucidated in future studies.
Although the current study focused on the effects of MT/HIF-1α in EPCs, MT and HIF-1α actions in endothelial cells are equally important in maintaining endothelial integrity and vascular homeostasis. Activation of MT and/or HIF-1α promotes migration, proliferation, survival, and angiogenic sprouting of endothelial cells (46,47). Therefore, the improved vascular function in JTMT transgenic mice may be attributable to the salutary effects of MT in both endothelial cells and EPCs, in part by suppression of oxidative stress and induction of HIF-1α/SDF-1/VEGF/Akt signaling. In addition to endothelial cells, Tie-2 is expressed in monocytes/macrophages, and Tie-2–expressing monocytes/macrophages play essential roles in tumor angiogenesis (48–50). Thus, it is possible that Tie-2 promoter–driven MT overexpression in monocytes/macrophages in JTMT mice may contribute to the promotion of diabetic HLI angiogenesis. Taken together, these findings suggest that therapeutic interventions to enhance endothelial MT expression and, thus, functionality may represent an attractive modality to prevent vascular complications in ischemia and diabetes.
Article Information
Acknowledgments. The authors acknowledge the reagent support and the insightful suggestions from Dr. Lu Cai (Pediatric Research Institute, Department of Pediatrics, University of Louisville School of Medicine). Most animal experiments were completed at the University of Louisville.
Funding. This study was supported in part by the American Diabetes Association Basic Research Award (1-11-BS-017, 1-15-BS-018) and Junior Faculty Award (1-13-JF-53), National Institute of General Medical Sciences (GM-103492), the National Natural Science Foundation of China project (81770305 and 81873466), a National Key R&D Program of China (2017YFA0506000), Zhejiang Provincial Natural Science Foundation of China grants (LY16H020010, LY14H070007), and an innovative team project of Chengdu Medical College (CYTD17-01). All personnel expenses and partial research-related expenses for K.W. were provided by Wenzhou Medical University through a collaborative research agreement between the University of Louisville and Wenzhou University.
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. K.W. and X.D. wrote, reviewed, and edited the manuscript. K.W., X.D., J.H., X.Ya., C.Y., S.S., J.F., X.Yu., and H.L. researched data. J.C., J.X., and Z.D. provided research material and techniques. X.F., E.C.C., F.S., K.A.W., D.J.C., P.N.E., and C.L. contributed to the data interpretation and discussion and/or edited the manuscript. Y.T. directed the project and wrote, reviewed, and edited the manuscript. Y.T. is the guarantor of this work and, as such, has full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This article contains supplementary material online at https://doi.org/10.2337/figshare.12280736.
K.W. and X.D. contributed equally to this study.
References
- 1.Chatterjee S, Khunti K, Davies MJ. Type 2 diabetes. Lancet 2017;389:2239–2251 [DOI] [PubMed] [Google Scholar]
- 2.Avogaro A, Albiero M, Menegazzo L, de Kreutzenberg S, Fadini GP. Endothelial dysfunction in diabetes: the role of reparatory mechanisms. Diabetes Care 2011;34(Suppl. 2):S285–S290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Asahara T, Murohara T, Sullivan A, et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science 1997;275:964–967 [DOI] [PubMed] [Google Scholar]
- 4.Hristov M, Erl W, Weber PC. Endothelial progenitor cells: mobilization, differentiation, and homing. Arterioscler Thromb Vasc Biol 2003;23:1185–1189 [DOI] [PubMed] [Google Scholar]
- 5.Aicher A, Heeschen C, Sasaki K, Urbich C, Zeiher AM, Dimmeler S. Low-energy shock wave for enhancing recruitment of endothelial progenitor cells: a new modality to increase efficacy of cell therapy in chronic hind limb ischemia. Circulation 2006;114:2823–2830 [DOI] [PubMed] [Google Scholar]
- 6.Dai X, Yan X, Zeng J, et al. Elevating CXCR7 improves angiogenic function of EPCs via Akt/GSK-3β/Fyn-mediated Nrf2 activation in diabetic limb ischemia. Circ Res 2017;120:e7–e23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Park Y, Zhang J, Cai L. Reappraisal of metallothionein: clinical implications for patients with diabetes mellitus. J Diabetes 2018;10:213–231 [DOI] [PubMed] [Google Scholar]
- 8.Cai L, Wang J, Li Y, et al. Inhibition of superoxide generation and associated nitrosative damage is involved in metallothionein prevention of diabetic cardiomyopathy. Diabetes 2005;54:1829–1837 [DOI] [PubMed] [Google Scholar]
- 9.Gu J, Yan X, Dai X, et al. Metallothionein preserves Akt2 activity and cardiac function via inhibiting TRB3 in diabetic hearts. Diabetes 2018;67:507–517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tan Y, Ichikawa T, Li J, et al. Diabetic downregulation of Nrf2 activity via ERK contributes to oxidative stress-induced insulin resistance in cardiac cells in vitro and in vivo. Diabetes 2011;60:625–633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cai L, Wang Y, Zhou G, et al. Attenuation by metallothionein of early cardiac cell death via suppression of mitochondrial oxidative stress results in a prevention of diabetic cardiomyopathy. J Am Coll Cardiol 2006;48:1688–1697 [DOI] [PubMed] [Google Scholar]
- 12.Zheng S, Carlson EC, Yang L, Kralik PM, Huang Y, Epstein PN. Podocyte-specific overexpression of the antioxidant metallothionein reduces diabetic nephropathy. J Am Soc Nephrol 2008;19:2077–2085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Carlson EC, Chhoun JM, Grove BD, et al. Renoprotection from diabetic complications in OVE transgenic mice by endothelial cell specific overexpression of metallothionein: a TEM stereological analysis. Anat Rec (Hoboken) 2017;300:560–576 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yoon CH, Hur J, Park KW, et al. Synergistic neovascularization by mixed transplantation of early endothelial progenitor cells and late outgrowth endothelial cells: the role of angiogenic cytokines and matrix metalloproteinases. Circulation 2005;112:1618–1627 [DOI] [PubMed] [Google Scholar]
- 15.Gu J, Cheng Y, Wu H, et al. Metallothionein is downstream of Nrf2 and partially mediates sulforaphane prevention of diabetic cardiomyopathy. Diabetes 2017;66:529–542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang Z, Wang S, Zhou S, et al. Sulforaphane prevents the development of cardiomyopathy in type 2 diabetic mice probably by reversing oxidative stress-induced inhibition of LKB1/AMPK pathway. J Mol Cell Cardiol 2014;77:42–52 [DOI] [PubMed] [Google Scholar]
- 17.Dai X, Zeng J, Yan X, et al. Sitagliptin-mediated preservation of endothelial progenitor cell function via augmenting autophagy enhances ischaemic angiogenesis in diabetes. J Cell Mol Med 2018;22:89–100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Xue W, Liu Y, Zhao J, Cai L, Li X, Feng W. Activation of HIF-1 by metallothionein contributes to cardiac protection in the diabetic heart. Am J Physiol Heart Circ Physiol 2012;302:H2528–H2535 [DOI] [PubMed] [Google Scholar]
- 19.Tan Y, Li Y, Xiao J, et al. A novel CXCR4 antagonist derived from human SDF-1beta enhances angiogenesis in ischaemic mice. Cardiovasc Res 2009;82:513–521 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Simon MC. The hypoxia response pathways - hats off! N Engl J Med 2016;375:1687–1689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Youn SW, Lee SW, Lee J, et al. COMP-Ang1 stimulates HIF-1α-mediated SDF-1 overexpression and recovers ischemic injury through BM-derived progenitor cell recruitment. Blood 2011;117:4376–4386 [DOI] [PubMed] [Google Scholar]
- 22.Rey S, Semenza GL. Hypoxia-inducible factor-1-dependent mechanisms of vascularization and vascular remodelling. Cardiovasc Res 2010;86:236–242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kim YW, Byzova TV. Oxidative stress in angiogenesis and vascular disease. Blood 2014;123:625–631 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Idei N, Soga J, Hata T, et al. Autologous bone-marrow mononuclear cell implantation reduces long-term major amputation risk in patients with critical limb ischemia: a comparison of atherosclerotic peripheral arterial disease and Buerger disease. Circ Cardiovasc Interv 2011;4:15–25 [DOI] [PubMed] [Google Scholar]
- 25.Fadini GP, Losordo D, Dimmeler S. Critical reevaluation of endothelial progenitor cell phenotypes for therapeutic and diagnostic use. Circ Res 2012;110:624–637 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sorrentino SA, Bahlmann FH, Besler C, et al. Oxidant stress impairs in vivo reendothelialization capacity of endothelial progenitor cells from patients with type 2 diabetes mellitus: restoration by the peroxisome proliferator-activated receptor-gamma agonist rosiglitazone. Circulation 2007;116:163–173 [DOI] [PubMed] [Google Scholar]
- 27.Zbinden S, Wang J, Adenika R, et al. Metallothionein enhances angiogenesis and arteriogenesis by modulating smooth muscle cell and macrophage function. Arterioscler Thromb Vasc Biol 2010;30:477–482 [DOI] [PubMed] [Google Scholar]
- 28.Wang J, Song Y, Elsherif L, et al. Cardiac metallothionein induction plays the major role in the prevention of diabetic cardiomyopathy by zinc supplementation. Circulation 2006;113:544–554 [DOI] [PubMed] [Google Scholar]
- 29.Lim KS, Cha MJ, Kim JK, et al. Protective effects of protein transduction domain-metallothionein fusion proteins against hypoxia- and oxidative stress-induced apoptosis in an ischemia/reperfusion rat model. J Control Release 2013;169:306–312 [DOI] [PubMed] [Google Scholar]
- 30.Iwaguro H, Yamaguchi J, Kalka C, et al. Endothelial progenitor cell vascular endothelial growth factor gene transfer for vascular regeneration. Circulation 2002;105:732–738 [DOI] [PubMed] [Google Scholar]
- 31.Takahashi T, Kalka C, Masuda H, et al. Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nat Med 1999;5:434–438 [DOI] [PubMed] [Google Scholar]
- 32.Tepper OM, Galiano RD, Capla JM, et al. Human endothelial progenitor cells from type II diabetics exhibit impaired proliferation, adhesion, and incorporation into vascular structures. Circulation 2002;106:2781–2786 [DOI] [PubMed] [Google Scholar]
- 33.Kawamoto A, Tkebuchava T, Yamaguchi J, et al. Intramyocardial transplantation of autologous endothelial progenitor cells for therapeutic neovascularization of myocardial ischemia. Circulation 2003;107:461–468 [DOI] [PubMed] [Google Scholar]
- 34.Shantsila E, Watson T, Lip GY. Endothelial progenitor cells in cardiovascular disorders. J Am Coll Cardiol 2007;49:741–752 [DOI] [PubMed] [Google Scholar]
- 35.Li FY, Lam KS, Tse HF, et al. Endothelium-selective activation of AMP-activated protein kinase prevents diabetes mellitus-induced impairment in vascular function and reendothelialization via induction of heme oxygenase-1 in mice. Circulation 2012;126:1267–1277 [DOI] [PubMed] [Google Scholar]
- 36.Groleau J, Dussault S, Haddad P, et al. Essential role of copper-zinc superoxide dismutase for ischemia-induced neovascularization via modulation of bone marrow-derived endothelial progenitor cells. Arterioscler Thromb Vasc Biol 2010;30:2173–2181 [DOI] [PubMed] [Google Scholar]
- 37.Abraham NG, Rezzani R, Rodella L, et al. Overexpression of human heme oxygenase-1 attenuates endothelial cell sloughing in experimental diabetes. Am J Physiol Heart Circ Physiol 2004;287:H2468–H2477 [DOI] [PubMed] [Google Scholar]
- 38.Pugh CW, Ratcliffe PJ. Regulation of angiogenesis by hypoxia: role of the HIF system. Nat Med 2003;9:677–684 [DOI] [PubMed] [Google Scholar]
- 39.Wang GL, Jiang BH, Rue EA, Semenza GL. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci U S A 1995;92:5510–5514 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Krock BL, Skuli N, Simon MC. Hypoxia-induced angiogenesis: good and evil. Genes Cancer 2011;2:1117–1133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bosch-Marce M, Okuyama H, Wesley JB, et al. Effects of aging and hypoxia-inducible factor-1 activity on angiogenic cell mobilization and recovery of perfusion after limb ischemia. Circ Res 2007;101:1310–1318 [DOI] [PubMed] [Google Scholar]
- 42.Li M, Cui Y, He W, et al. Effects of triple-mutated hypoxia-inducible factor-1α on angiogenesis and cardiac function improvement in rats with myocardial infarction. Cell Physiol Biochem 2018;50:2329–2340 [DOI] [PubMed] [Google Scholar]
- 43.Liu HY, Hong T, Wen GB, et al. Increased basal level of Akt-dependent insulin signaling may be responsible for the development of insulin resistance. Am J Physiol Endocrinol Metab 2009;297:E898–E906 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xiao H, Gu Z, Wang G, Zhao T. The possible mechanisms underlying the impairment of HIF-1α pathway signaling in hyperglycemia and the beneficial effects of certain therapies. Int J Med Sci 2013;10:1412–1421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Catrina SB, Okamoto K, Pereira T, Brismar K, Poellinger L. Hyperglycemia regulates hypoxia-inducible factor-1alpha protein stability and function. Diabetes 2004;53:3226–3232 [DOI] [PubMed] [Google Scholar]
- 46.Schulkens IA, Castricum KC, Weijers EM, Koolwijk P, Griffioen AW, Thijssen VL. Expression, regulation and function of human metallothioneins in endothelial cells. J Vasc Res 2014;51:231–238 [DOI] [PubMed] [Google Scholar]
- 47.Schuermann A, Helker CS, Herzog W. Metallothionein 2 regulates endothelial cell migration through transcriptional regulation of vegfc expression. Angiogenesis 2015;18:463–475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Venneri MA, De Palma M, Ponzoni M, et al. Identification of proangiogenic TIE2-expressing monocytes (TEMs) in human peripheral blood and cancer. Blood 2007;109:5276–5285 [DOI] [PubMed] [Google Scholar]
- 49.Lewis CE, De Palma M, Naldini L. Tie2-expressing monocytes and tumor angiogenesis: regulation by hypoxia and angiopoietin-2. Cancer Res 2007;67:8429–8432 [DOI] [PubMed] [Google Scholar]
- 50.Chen L, Li J, Wang F, et al. Tie2 expression on macrophages is required for blood vessel reconstruction and tumor relapse after chemotherapy. Cancer Res 2016;76:6828–6838 [DOI] [PubMed] [Google Scholar]