Abstract
The microbiological quality of pre-packed sandwiches, prepared by a company which had implemented the Hazard Analysis Critical Control Points system, was assessed at retail level, in Crete, Greece. Totally, we analyzed 225 sandwiches (S1: ham, cheese; S2: ham, cheese, tomato; S3: tuna salad), for specific pathogens (Listeria monogytenes, Salmonella spp, Staphylococcus aureus) and hygiene indicators (Escherichia coli, Enterobacteriaceae, Aerobic Colony Count-ACC). Pathogens were not detected. The E. coli numbers enumerated in day 0 (factory level) were found within acceptable levels < 100 cfu/g; limited samples had unsatisfactory values at the 3rd day of retailing storage (7%, 7%, and 27% > 100 cfu/g for S1, S2, and S3, respectively), which were further increased at the 7th day (20%, 33% and 53% > 100 cfu/g for S1, S2, and S3, respectively). The Enterobacteriaceae numbers mean log CFU/g were in the satisfactory or acceptable category with an increase in the range of 19.5–49.5% at the 7th day, nevertheless never exceeded the borderline of 4 log CFU/g. All ACC values were satisfactory or acceptable as no value higher than 7 log CFU/g was recorded. Overall there was a difference between the three sandwiches types, with S2 and S3, exhibiting higher levels than S1, possibly due to the extra ingredients. A number of corrective actions can be applied, as i.e. revision of cooking-chilling times, sanitizing procedures, staff hygiene practices and training etc.
Keywords: Ready to eat (RTE) foods, Sandwiches, Microbiological quality, Retailing, HACCP
Introduction
Crete, the most heavily populated island of Greece, is the fifth largest island in the Mediterranean Sea, with more than 1000 km coastline. It has three major airports (two of them serve international routes) and six ports, while the main economic activities are tourism and agriculture. Crete’s airports received more than 2.5 million passengers during 2010, second after only Athens’s airport, while tourists have the opportunity to choose among 1600 hotels for accommodation purposes (Greek Tourism Confederation, SETE, 2019). Cretan nutrition and diet have been proven to promote human health and longevity (Hertog et al. 1995; Kromhout et al. 1995; Willett et al. 1995; Padilla 2001; Mentella et al. 2019) and visitors have the chance to try various food recipes (traditional or international) either in commercial establishments (such as hotels, restaurants, supermarkets etc.) or small retail shops and kiosks (ready-to-eat food, without further preparation before eating).
All composite foods, which before consumption do not require significant further processing other than unpacking, reheating or completion of cooking, are included in the ready-to-eat food (RTF) category. Characteristic examples of RTF can be substances derived from plants (i.e. spices, seasonings); plant food (fruits and vegetables) that does not require any further washing, cooking or processing; bakery items (bread, cakes, pies) for which further cooking is not required; dried products (meat, poultry); fermented or salt cured products (sausages, meat); thermally processed low acid foods packaged in hermetically sealed containers (canned corn, green beans, mushrooms, variety of vegetables, pickled foods, sauces and dressings, fruits or fruit mixtures), or combinations of the previous defined as ready-to-eat prepacked food, such as pre-packed sandwiches. Main requirement for all these RTF is the appropriate lethality treatment for pathogen microorganisms (Evans and Redmond 2016).
Ready-to-eat foods provide a high degree of readiness, completion and convenience, while there is a considerable variation in the composition of these foods. Ready-to-eat foods, if not consumed immediately, must be stored under refrigeration to ensure shelf-life (FSAI 2019). Several modern trends and changes in food consumption patterns (due to advertising, demographic and price factors), along with the lack of time to prepare cooked food, the long-distance daily transportation of people in order to work, the more demanding daily working-schedules etc.; lead to an increasing demand for convenience foods, hence enhancing food safety risks, due to consumption of ready-to-eat prepacked foods, including sandwiches (Ritson and Hutchins 1995; Hyebin et al. 2014; Okrent and Kumcu 2014).
Sandwiches microbiological quality is influenced by the quality of raw materials as well as by handling, processing, transportation, and storage (Angelidis et al. 2006). Sandwiches may be a source of foodborne illnesses because ingredients may be exposed to contaminants throughout the production process i.e., facilities, equipment, handlers. Often, sandwich fillings are shredded or chopped by food handlers. Furthermore, the lack of processing in high temperatures may lead to pathogen survival and proliferation in the final product, causing food poisoning (Gillespie et al. 2000; Fang et al. 2003). The hygiene risks from sandwiches consumption are higher when they are sold in small shops (Jang et al. 2013), and especially when the sandwich ingredients include salad, and the temperatures are high, as during in the summer period. On the other hand the introduction of food safety systems, such as Hazard Analysis Critical Control Points (HACCP), has a substantial positive effect in the quality of the final food product (Eves and Cervisi 2005; Walker et al. 2003; ISO (International 2018b).
The purpose of this study, carried out during the summer season, was to monitor and evaluate the effects imposed by storage conditions (i.e. chilling conditions 1–4 °C) on the microbiological quality of sandwiches that were produced by a factory, which had implemented the HACCP system and were offered for sale in small retail shops throughout Crete, Greece. The presence of three specific pathogens and three indicator bacterial populations was recorded for a period of seven days after the production of sandwiches. Based on the results we present a number of suggestions to the industry and retailing of ready-to-eat prepacked sandwiches.
Materials and methods
Sampling
We selected the biggest factory in Crete which had implemented and certified the HACCP system, producing sandwiches transported by factory’s own vehicles and sold in super/mini-markets, food stalls or kiosks, in Crete, as well as at other tourist islands and Greece mainland. Our research included initial sampling (Fig. 1) in the factory, where samples were taken and production codes recorded to secure traceability, and then in 6 different retailing points (at 6 different major tourist areas of the island). Three different sandwiches types were assessed (S1: ham, cheese; S2: ham, cheese, tomato; S3: tuna salad). The product distribution schedule of the factory was followed, in order to track the initial product codes and perform product sampling at the end-points of sales (traceability process). Arrangements were made with each owner at sale points, to store the individual sandwiches, in order to evaluate storage conditions effect in microbial quality of product. The second sampling was carried out during the third 3rd day, and the seventh 7th day of the products life. Three batches of each type (S1, S2, and S3), five sandwiches per batch, were sampled in the factory; totally 45 freshly produced sandwiches. In addition, five samples from each type were collected from each of the six retailing points during the following 3rd and 7th day; that is 180 sandwiches, in total 225 food samples. In general, sandwiches which had passed their expiry date or had damaged packaging, were excluded from sampling. The samples taken were immediately transported to the laboratory in an icebox (2–6 °C) and stored in cooling conditions (1–4 °C) until microbiological analysis. The food samples analysis was performed the following day and the samples were analyzed with classical (ISO) protocols for the detection and enumeration of pathogens and commercial microbiological analytical kits for indicator microorganisms: we analyzed samples for detection and enumeration of Listeria monocytogenes (LMO), Salmonella species (SLM), Staphylococcus aureus (SAU), Escherichia coli (E. coli), Enterobacteriaceae, and Aerobic Colony Count (ACC).
Fig. 1.
Sandwiches sampling scheme
Microbial analysis
Standard microbiological guidelines and criteria for sampling and evaluation of ready-to-eat foods were followed ( FSA (Food Standards Agency) 2000; Gilbert et al. 2000; ICMSF (International 1986; ICMSF (International 2002). The L. monocytogenes detection followed horizontal methods (ISO 11,290–1:2017) ( ISO (International 2017a); a two-stage enrichment method was used as described ( Harrigan 1998). Food samples (25 g) were added into “half Fraser” broth (225 ml), (Biokar Diagnostics), homogenized and incubated for 24 h at 30 °C. A loopful of the primary enrichment broth was used to inoculate a plate of Oxford agar (Biokar Diagnostics) that was incubated for 48 h at 35 °C. For the secondary enrichment, 0.1 ml of the primary enriched broth was used to inoculate 10 ml full-strength Fraser-broth that was incubated for 48 h at 35 °C, and used to inoculate Oxford agar plates that were further incubated for 48 h at 35 °C. All “suspicious” colonies, from the enrichments, were picked, subcultured onto tryptone soy yeast extract agar (TSYEA) (Difco), and incubated for 48 h at 35 °C. Further identification tests were carried out with the API Listeria kit (bioMerieux).
Salmonella spp detection followed a horizontal method (ISO 6579–1:2017) ( Harrigan 1998; ISO (International 2017b). Food samples (25 g) were homogenized into 225 ml buffered peptone water (BPW) (Biokar Diagnostics) and incubated for 24 h at 37 °C. After this pre-enrichment, 10 ml were added to both, 100 ml of selenite-cystine broth, and 100 ml of Rappaport-Vassiliadis (RV) (Biokar Diagnostics) medium, and incubated for 24 h, at 37 °C, and 42 °C, respectively. These enriched media were used to inoculate brilliant green phenol red agar (BGA) (Oxoid), and xylose lactose deoxycholate (XLD) (Biokar Diagnostics) plates, that were incubated for at 37 °C, for 24 h, and 48 h, respectively. Further tests were carried out with the API 20E kit (bioMerieux).
Baird-Parker agar (BPA), (Biokar Diagnostics) with egg-yolk tellurite emulsion (Oxoid) was used for the detection and enumeration of Staphylococcus aureus (ISO 6888–1:1999/AMD2:2018) (ISO 2018a). Food samples (25 g) were added into 225 ml peptone water (PW) (Biokar Diagnostics), homogenized, and used to inoculate BPA plates. After incubation for 48 h at 37 °C, the plates were examined for the typical black colonies of Staphylococcus aureus, while further identification tests were carried out with the API Staph kit (bioMerieux).
Enumeration of Enterobacteriaceae was carried out using the 3 M™ Petrifilm™ Enterobacteriaceae Count Plate method (Silbernagel and Lindberg 2003). The 3 M™ Petrifilm® Ecoli/Coliform Count (3 M™ Healthcare, Loughborough, UK) method based on AOAC 991.14 (AOAC 2000), and the 3 M™ Petrifilm® Aerobic Count (AC) methodology were used for enumeration of E. coli, and ACC, respectively (Gracias and McKillip 2004). Sample preparation was performed as before, according to ISO 6887–1:2017 ( ISO (International 2017c), food samples (25 g) were homogenized into 225 ml buffered peptone water (BPW), 1 ml sample was placed on each petrifilm and incubated at 35 °C for 24 h.
Statistical analysis
Experimental data were analyzed by descriptive statistics (mean and standard deviation) and ANNOVA (two-way), in order to determine the correlation of microbial population levels between the three different types of sandwiches (at a significance level p < 0.05). Data shown in Table 1 and Table 2 are described by means and standard deviation, while two-way ANOVA using food item and days of analysis as factors were applied in raw data.
Table. 1.
E. coli, identified in the 3 types (S1, S2, and S3) of sandwiches tested
| Microbe | Factory sandwiches (n = 15/type = 3 × 5 = 45) | Retail sandwiches (n = 5/type, from 6 retailing points = 5 × 3x6 = 180) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Day 0 (n = 45) | 3rd day (n = 90) | 7th day (n = 90) | |||||||
| Type S1: sandwiches with ham and cheese | |||||||||
| < 20 | 20–100 | > 100 | < 20 | 20–100 | > 100 | < 20 | 20–100 | > 100 | |
| E. coli(%) | 73 | 27 | 0 | 73 | 20 | 7 | 47 | 33 | 20 |
| Type S2: sandwiches with ham, cheese, and tomato | |||||||||
| < 20 | 20–100 | > 100 | < 20 | 20–100 | > 100 | < 20 | 20–100 | > 100 | |
| E. coli(%) | 67 | 33 | 0 | 53 | 40 | 7 | 20 | 47 | 33 |
| Type S3: sandwiches with tuna salad | |||||||||
| < 20 | 20–100 | > 100 | < 20 | 20–100 | > 100 | < 20 | 20–100 | > 100 | |
| E. coli(%) | 53 | 47 | 0 | 53 | 20 | 27 | 14 | 33 | 53 |
n number of samples
Table. 2.
Enterobacteriaceae, and aerobic colony count levels in the 3 types (S1, S2, and S3) of sandwiches tested
| Microbe | Factory sandwiches (n = 15/type = 3 × 5 = 45) | Retail sandwiches (n = 5/type, from 6 retailing points = 5 × 3x6 = 180) | ||||
|---|---|---|---|---|---|---|
| Day 0 (n = 45) | 3rd day (n = 90) | 7th day (n = 90) | ||||
| Mean (logCFU/g) | Range (logCFU/g) | Mean (logCFU/g) | Range (logCFU/g) | Mean (logCFU/g) | Range (logCFU/g) | |
| Type S1: sandwiches with ham and cheese | ||||||
| Enterobacteriaceae | 1.86 | 1.74–1.98 | 1.94 | 1.82–2.05 | 2.72 | 2.65–2.79 |
| ACC | 4.94 | 4.75–5.12 | 5.02 | 4.83–5.20 | 5.89 | 5.81–5.97 |
| Type S2: sandwiches with ham, cheese, and tomato | ||||||
| Enteroba-cteriaceae | 1.94 | 1.80–2.07 | 2.02 | 1.91–2.12 | 2.90 | 2.82–2.97 |
| ACC | 5.23 | 5.19–5.27 | 5.27 | 5.21–5.33 | 6.02 | 5.95–6.08 |
| Type S3: sandwiches with tuna salad | ||||||
| Enteroba-cteriaceae | 2.71 | 2.55–2.87 | 2.76 | 2.60–2.91 | 3.24 | 3.01–3.47 |
| ACC | 5.48 | 5.21–5.75 | 5.82 | 5.78–5.86 | 6.33 | 6.11–6.55 |
n number of samples
Results and discussion
Pathogens presence/absence in the sandwiches
According to the EU Commission Regulation No 2073/2005 on “Microbiological Criteria for Foodstuffs” concerning foods ready to eat that can be consumed by infants, and adults, no pathogens: Listeria monocytogenes, Salmonella spp, and Staphylococcus aureus, must be present within 25 g of these foods (EU 2005). In this research no such pathogens were detected at any of the three different types of sandwiches tested; hence, the HACCP system (ISO 22,000:2018) was successfully implemented as reported in other studies also (Kokkinakis et al. 2008). According to HACCP methodology and Good Hygiene Practices (GHP) when vegetables are used raw in sandwiches or prepared as salads for the final products, they were sanitized in order to reduce microbes (Kokkinakis and Fragkiadakis 2007).
Indicator organisms in the sandwiches
According to the Public Health Laboratory Services (PHLS) guidelines the E. coli values concerning sandwiches indicate a satisfactory condition if they are < 20 cfu/g, acceptable if < 100 cfu/g, and unsatisfactory if > 100 cfu/g (Gilbert et al. 2000). As presented in Table 1 the E coli numbers enumerated in day 0 (factory level) were all found within acceptable levels (with 73%, 67% and 53% within satisfactory levels < 20 cfu/g for S1, S2 and S3 respectively). However, a small number of samples were found with unsatisfactory values at the 3rd day of storage at the retailing points (7%, 7% and 27% > 100 cfu/g for S1, S2 and S3 respectively), which were further increased at the 7th day of storage, with a range of 20–53% of the samples at the unsatisfactory level (20%, 33% and 53% > 100 cfu/g for S1, S2 and S3 respectively). Concerning sampling at the factory level, 16 sandwiches out of 45 sampled were found with a high degree of hygiene status (< 20 cfu/g) and all the remaining were found with E. coli values within the acceptable level, ie below 100 cfu/g. In case the borderline of 100 cfu/g E coli was exceeded at factory level, then a number of investigations and corrective actions (according to HACCP methodology) could be performed in order to ensure subsequent batches of food are satisfactory (eg revision of cooking-chilling times, selection and origin of raw materials, cleaning and sanitizing procedures, staff hygiene practices and training, review and update of GHP procedures and HACCP in total).
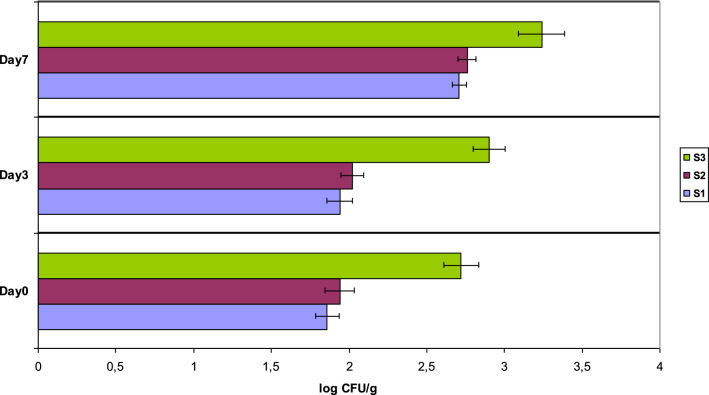
Enterobacteriaceae measurements are useful indicators of hygiene and post-processing contamination of heat processed foods. Enterobacteriaceae give a good indication of the likelihood of the presence of pathogens, and can provide information about the handling and storage of the foodstuff (FSAI 2019). According to the PHLS guidelines, the values of Enterobacteriaceae in sandwiches are: satisfactory for < 2 log CFU/g, acceptable for 2–4 log CFU/g, and unsatisfactory for > 4 log CFU/g (Gilbert et al. 2000). The Enterobacteriaceae numbers mean log CFU/g in this study were in the satisfactory or acceptable category with an increase of the mean log CFU/g in the range of 19.5–49.5% at the 7th day (Table 2), nevertheless never exceeded the borderline of 4 log CFU/g, as shown in Fig. 2. In case Enterobacteriaceae at factory level were found above acceptable levels, the same investigations and corrective measures should be performed, as described earlier in the case of E. coli, in order to ensure quality during food production.
Fig. 2.
Enterobacteriaceae in 3 different types of sandwiches (S1, S2, S3). Error bars are included, to indicate the uncertainty around the estimate of the mean measurement
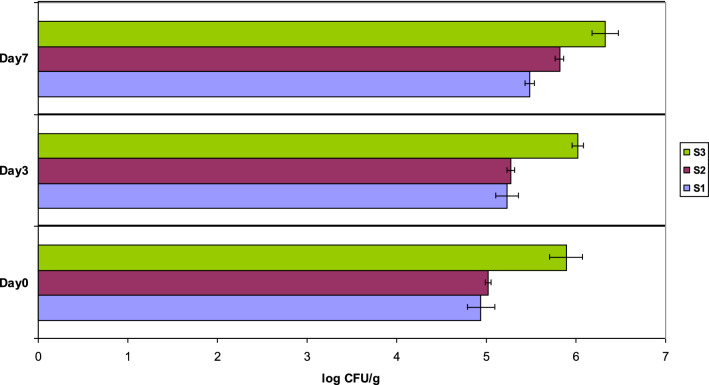
The Aerobic Colony Count (ACC) test is a generic test that counts various organisms which grow under aerobic conditions at mesophilic temperatures on a specific growth medium. This test provides useful information in order to assess food quality or food remaining shelf-life. Criteria concerning the Aerobic Colony Count (ACC) levels of sandwiches are reported mainly for those containing no salad and no cheese; since the additive effect of these ingredients on ACC cannot be predicted safely. Nevertheless, we used the guidelines for assessing the microbiological quality of ready-to-eat foods placed on the market, as suggested by Food Safety Authority of Ireland’s ( FSAI (Food 2019) and adapted by UK’s Health Protection Agency Guidelines (HPA (UK’s Health Protection Agency) 2009). According to these guidelines a combination of food categories was chosen (bakery and confectionery products, food mixed with dressings and dips, and ready to eat meat and fish) and the more strict guidelines were used to evaluate the ACC measurements; satisfactory were considered the levels for < 6 log CFU/g, acceptable for 6–7 log CFU/g, and unsatisfactory for > 7 log CFU/g (Gilbert et al. 2000). Additionally, for sandwiches containing salad and/or cheese, according to Gilbert et al (2000): “food examiners and microbiologists should use their own judgment to assess where a product would fit—based on the type of product, the processing it has received, and the potential for microbial growth during storage”. During our assessment all the ACC values were satisfactory or acceptable as no value higher than 7 log CFU/g was recorded, as shown in Fig. 3. All food items sampled at factory were found within satisfactory levels (< 6 log CFU/g), while the deteriorations of the mean ACC values in the 7th day of retailing were in the range of 15–19%, compared to day 0 of the product at the factory.
Fig. 3.
Aerobic colony count in 3 different types of sandwiches (S1, S2, S3). Error bars are included, to indicate the uncertainty around the estimate of the mean measurement
In total, statistical analysis (two-way ANOVA) using food item and days of analysis, revealed statistically significant results (p < 0.05). Variable 1 (S1, S2, S3), Variable 2 (D0, D3, D7) and Interaction, as calculated by the two-way ANOVA statistical test, were all found with p < 0.05. Hence, there was a difference between the three sandwiches types S1, S2, and S3 and days of analysis, in terms of E. coli, Enterobacteriaceae, and ACC levels, with S2 and S3 generally exhibiting higher levels than S1 (Table 1, and Table 2). These findings can be explained by each food item’s composition. The first product (S1) consisted of ham and cheese, the second (S2) had vegetables as extra ingredient, while the third (S3) had salad made from vegetables and mayonnaise in addition to tuna. Most possibly, the extra ingredients introduced higher number of microbes, yet without exceeding the acceptable limits in the fresh products, an observation reported in other studies also (Eleftheriadou et al. 2002).
Conclusion
The microbiological quality of the sandwiches tested in this study was generally good, with samples taken from the factory all within satisfactory and acceptable microbial levels, while no pathogen microbes were identified. This can be attributed to correct manufacturing procedures (Smith et al. 2001) and proper HACCP implementation. The same was observed during food storage at the retailer’s level (shopping points) as far as Enterobacteriaceae and Aerobic colony Counts (ACC) are concerned. Even at the 7th day of storage microbial levels were never above 4 log CFU/g and 7 log CFU/g, for Enterobacteriaceae and ACC respectively. However, there was a change of microbial levels of hygiene indicator microorganisms, in terms of E. coli, during the 7th day of storage at the retailer’s level indicating possible product deterioration (e.g. E. coli > > 100 CFU/g) and limiting the self-life of food product.
Observations made during sampling from the retailing points confirmed that the majority of sandwiches were consumed within 1–3 days, which along with company’s policy of product redrawn every week and replacement with new ones, provided an excellent attitude towards food quality and safety. However, a number of observations indicated a possible lack of Good Hygiene Practices (GHP) at shops level: some personnel were found with not adequate knowledge about product safety and hygiene; a number of small refrigerators used to keep sandwiches were found to operate above safety temperatures (i.e. > > 4–8 °C); cleaning of refrigerators was not always satisfactory, while one refrigerator was found with no protection from the sun, causing the internal temperature to increase further. Concerning retailing conditions, special emphasis should be made on temperature control, since it’s a control measure (ByrneP (2002) 3rd Quarter National Microbiology Survey2002(NS3): Microbiological safety of pre-packed sandwiches.The Food Safety Authority of Ireland.(www.fsai.ie/uploadedFiles/3rdQuarter_prepacked_sandwiches.pdf), 2002) acting as a critical control point, ensuring that the microbiological quality/safety does not deteriorates during storage. To our knowledge, sandwiches from mini-markets, small corner shops, kiosks etc. are not sampled very often by the Greek hygiene authorities. However, the risk for a basic hygiene breakdown is considered to increase in such premises, especially when the sandwich ingredients include salad, and ambient temperatures are high during summer months (Williamson et al. 2000).
Storage conditions of pre-packed and ready-to-eat sandwiches at the point of retail are critical to product safety and can be greatly affected by training and hygiene of the personnel involved. We suggest that food factories implementing HACCP should provide even more specific guidelines and training to retail customers, in terms of product quality and safety. In addition, the retailing conditions, as well as the microbial quality of products at points of sale, must be checked more often by factories themselves, and independently of the local authorities’ inspections, as a requirement of the implemented HACCP system. This would further enhance factory’s brand name status. There is justifiable expectation that in perspective, research and development of antimicrobial materials for food applications such as packaging and other food contact surfaces will grow in the next decades, with the advent of new polymer materials and antimicrobials (Appendini and Hotchkiss 2002). These materials will be an asset for the sandwiches preparation technology/industry also. Finally, the shelf-life of fresh or minimally processed foods can be further secured and extended by using both innovative packing materials and techniques as modified atmosphere packaging (MAP), where the oxygen content within the pack is lowered or minimized by introducing another gas, as i.e. nitrogen (Caleb et al. 2013).
Acknowledgements
Authors declare they have received no financial support for the preparation of this manuscript
Full compliance with all ethics.
Compliance with ethical standards
Conflict of interest
All authors declared that they have no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Manolis N. Kokkinakis, Email: mkokkinakis@hmu.gr
Georgios A. Fragkiadakis, Email: fragkiadakis@hmu.gr
Nikolaos E. Lapidakis, Email: lapidakis@hmu.gr
Aikaterini N. Kokkinaki, Email: katrinakok@yahoo.gr
References
- Angelidis AS, Chronis EN, Papageorgiou DK, Kazakis II, Arsenoglou KC, Stathopoulos GA. Non-lactic acid, contaminating microbial flora in ready-to-eat foods: a potential food-quality index. Food Microbiol. 2006;23:95–100. doi: 10.1016/j.fm.2005.01.015. [DOI] [PubMed] [Google Scholar]
- AOAC (Association of Official Analytical Chemists) (2000) Official Methods of Analysis, 17th edition. Association of Official Analytical Chemists International Gaithersburg, 481 N. Frederick Ave, Suite 500, MD 20877, USA
- Appendini P, Hotchkiss J. Review of antimicrobial food packaging. Innov Food Sci Emerg Technol. 2002;3:113–126. doi: 10.1016/S1466-8564(02)00012-7. [DOI] [Google Scholar]
- Byrne P (2002) 3rd Quarter National Microbiology Survey 2002 (NS3): Microbiological safety of pre-packed sandwiches. The Food Safety Authority of Ireland. (www.fsai.ie/uploadedFiles/3rdQuarter_prepacked_sandwiches.pdf)
- Caleb OJ, Mahajan PV, Al-Said FA, Opara U. Modified atmosphere packaging technology of fresh and fresh-cut produce and the microbial consequences—a review. Food Bioprocess Technol. 2013;6:303–329. doi: 10.1007/s11947-012-0932-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eleftheriadou M, Varnava-Tello A, Metta-Loizidou NAS, Akkelidou D. The microbiological profile of foods in the Republic of Cyprus: 1991–2000. Food Microbiol. 2002;19:463–471. doi: 10.1006/fmic.2002.0508. [DOI] [Google Scholar]
- EU (European Union) (2005) Regulation No 2073/2005 of 15th Nov 2005 on Microbiological Criteria of Foodstuffs, Official Journal L338, 22.12.2005, pp. 1–26
- Evans EW, Redmond EC. Older adult consumer knowledge, attitudes, and self-reported storage practices of ready-to-eat food products and risks associated with listeriosis. J Food Prot. 2016;79:263–272. doi: 10.4315/0362-028X.JFP-15-312. [DOI] [PubMed] [Google Scholar]
- Eves A, Cervisi P. Experiences of the implementation and operation of hazard analysis critical control points in the food service sector. Hosp Manag. 2005;24:3–19. doi: 10.1016/j.ijhm.2004.04.005. [DOI] [Google Scholar]
- Fang TJ, Wei QT, Liao CW, Hung MJ, Wang TH. Microbiological quality of 18°C ready-to-eat food products sold in Taiwan. Int J Food Microbiol. 2003;80:241–250. doi: 10.1016/S0168-1605(02)00172-1. [DOI] [PubMed] [Google Scholar]
- FSA (Food Standards Agency) (2000) Code of Practice No.9: Food Hygiene Inspections: (Second Revision Oct 2000)
- FSAI (Food Safety Authority of Ireland) (2019) Guidance note no.3: guidelines for the interpretation of results of microbiological testing of ready-to-eat foods placed on the market (revision 3). Published by: Food Safety Authority of Ireland, Abbey Court, Lower Abbey Street, Dublin
- Gilbert RJ, De Louvois J, Donovan T, Little C, Nye K, Ribeiro CD, Richards J, Roberts D, Bolton FJ. Guidelines for the microbiological quality of some ready-to-eat food sampled at the point of sale. Commun Dis Public Health. 2000;3:163–167. [PubMed] [Google Scholar]
- Gillespie I, Little C, Mitchell R. Microbiological examination of cold ready-to-eat sliced meats from catering establishments in the United Kingdom. J Appl Microbiol. 2000;88:467–474. doi: 10.1046/j.1365-2672.2000.00981.x. [DOI] [PubMed] [Google Scholar]
- Gracias KS, McKillip JL. A review of conventional detection and enumeration methods for pathogenic bacteria in food. Can J Microbiol Rev. 2004;50:883–890. doi: 10.1139/w04-080. [DOI] [PubMed] [Google Scholar]
- Harrigan WF. Laboratory methods in food microbiology. 3. California: Academic Press; 1998. [Google Scholar]
- Hertog MG, Kromhout D, Aravanis C, Blackburn H, Buzina R, Fidanza F, Giampaoli S, Jansen A, Menotti A, Nedeljkovic S. Flavonoid intake and long-term risk of coronary heart disease and cancer in the seven countries study. Arch Intern Med. 1995;155:381–386. doi: 10.1001/archinte.1995.00430040053006. [DOI] [PubMed] [Google Scholar]
- HPA (UK’s Health Protection Agency) Guidelines for assessing the microbiological safety of ready-to-eat foods. London: Health Protection Agency; 2009. [Google Scholar]
- Hyebin J, Seoyoun L, Younchan CS (2014) Home Meal Replacement Market Segmentation: A Food-Related Life Style. Selected Paper prepared for presentation at the Agricultural and Applied Economics Association’s 2014 AAEA Annual Meeting, Minneapolis, MN, July 27–29, 2014
- ICMSF (International Commission on Microbiological Specifications for Foods) (1986) Microorganisms in Foods 2. Sampling for microbiological analysis: Principles and specific applications, 2nd Edition. Toronto: University of Toronto Press. Canada. ISBN: 0802056938
- ICMSF (International Commission on Microbiological Specifications for Foods) Microorganisms in Foods, 7 Microbiological testing in food safety management. New York: Kluwer Academic/Plenum Publishers; 2002. [Google Scholar]
- ISO (International Organization for Standardization) (ISO 11290–1:2017) (2017a) Microbiology of the food chain—Horizontal method for the detection and enumeration of Listeria monocytogenes and of Liesteria spp—Part 1: Detection method, Geneva: International Organization for Standardization
- ISO (International Organization for Standardization) (ISO 6579–1:2017) (2017b) Microbiology of the food chain—Horizontal method for the detection, enumeration and serotyping of Salmonella—Part 1: Detection of Salmonella spp. Geneva: International Organization for Standardization
- ISO (International Organization for Standardization) (ISO 6887–1:2017) (2017c)Microbiology of the food chain—Preparation of test samples, initial suspension and decimal dilutions for microbiological examination—Part 1: General rules for the preparation of the initial suspension and decimal dilutions. Geneva: International Organization for Standardization
- ISO (International Organization for Standardization) (ISO 6888–1:1999/AMD2:2018) (2018a) Microbiology of food and animal feeding stuffs—Horizontal method for the enumeration of coagulase-positive staphylococci (Staphylococcus aureus and other species)—Part 1: Technique using Baird-Parker agar medium—Amendment 2: Inclusion of an alternative confirmation test using RPFA stab method. Geneva: International Organization for Standardization
- ISO (International Organization for Standardization) (ISO 22000:2018) (2018b) Food safety management systems—Requirements for any organization in the food chain
- Jang H, Kim NH, Choi YM, Rhee M. Microbiological quality and risk factors related to sandwiches served in bakeries, cafes, and sandwich bars in South Korea. J Food Prot. 2013;76:231–238. doi: 10.4315/0362-028X.JFP-12-335. [DOI] [PubMed] [Google Scholar]
- Kokkinakis E, Fragkiadakis GA. HACCP effect on microbiological quality of minimally processed vegetables: a survey in six mass-catering establishments. Int J Food Sci Technol. 2007;42:18–23. doi: 10.1111/j.1365-2621.2006.01179.x. [DOI] [Google Scholar]
- Kokkinakis E, Boskou G, Fragkiadakis GA, Kokkinaki A. Changes in vegetable microbiological quality introduced by processing methods. Int J Food Sci Technol. 2008;43:603–609. doi: 10.1111/j.1365-2621.2006.01494.x. [DOI] [Google Scholar]
- Kromhout D, Menotti A, Bloemberg B, Aravanis C, Blackburn H, Buzina R, Dontas AS. Dietary saturated and trans fatty acids and cholesterol and 25 year mortality from coronary heart disease: the seven countries study. Prev Med. 1995;24:308–315. doi: 10.1006/pmed.1995.1049. [DOI] [PubMed] [Google Scholar]
- Mentella MC, Scaldaferri F, Ricci C, Miggiano GAD. Cancer and mediterranean diet: a review. Nutrients. 2019;11:2059. doi: 10.3390/nu11092059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okrent AM, Kumcu A (2014) What’s Cooking? Demand for Convenience Foods in the United States. Selected Paper prepared for presentation at the Agricultural and Applied Economics Association’s 2014 AAEA Annual Meeting, Minneapolis, MN, July 27–29, 2014
- Padilla M. Evolution of Mediterranean diets: facts, causes, effects. In: Amado R, Lairon D, Gerber M, Maiani G, Abt B, editors. Bioactive micronutrients in mediterranean diet and health. Luxembourg: European Communities; 2001. pp. 263–272. [Google Scholar]
- Ritson C, Hutchins R (1995) Food choice and the demand for food, in D. W. Marshall, Food choice and the consumer (pp. 43–77). Blackie Academic & Professional: An imprint of Chapman and Hall, ISBN 0–7514–0234–6
- SETE (Greek Tourism Confederation) (2019) https://sete.gr/en/ Accessed 9 Dec 2019
- Silbernagel KM, Lindberg KG. 3TM PetrifilmTM Enterobacteriaceae count plate method for enumeration of Enterobacteriaceae in selected foods: collaborative study. J AOAC Int. 2003;86:802–814. doi: 10.1093/jaoac/86.4.802. [DOI] [PubMed] [Google Scholar]
- Smith JL, Davies GJ, Bent AJ. Retail fast foods: overview of safe sandwich manufacture. J R Soc Promot Health. 2001;121:220–223. doi: 10.1177/146642400112100404. [DOI] [PubMed] [Google Scholar]
- Walker E, Pritchard C, Forsythe S. Hazard analysis critical control point and prerequisite programme implementation in small and medium size food businesses. Food Control. 2003;14:169–174. doi: 10.1016/S0956-7135(02)00061-0. [DOI] [Google Scholar]
- Willett WC, Sacks F, Trichopoulou A, Trichopoulos D. Mediterranean diet pyramid: a cultural model for healthy eating. Am J Clin Nutr. 1995;6:1402S–1406S. doi: 10.1093/ajcn/61.6.1402S. [DOI] [PubMed] [Google Scholar]
- Williamson K, Allen G, Bolton FJ (2000) Report of the greater Manchester/Lancashire/PHLS liaison group survey on the microbiological examination of meat/salad sandwiches from small retail premises, PHLS North West FESL—Preston PHL, Survey Code No. 904009, KW/KLM (182000), Date of report 6/3/00. (https://lancsscm.web-labs.co.uk/Documents/EnvHealth/Food/Sampling/904009%20Meat%20and%20Salad%20sandwiches%20from%20small%20retail%20premises.pdf)