Abstract
Background:
Sodium-glucose cotransporter-2 inhibitors (SGLT-2i’s) improve heart failure (HF) related outcomes. The mechanisms underlying these benefits are not well understood, but diuretic properties may contribute. Traditional diuretics, such as furosemide, induce substantial neurohormonal activation contributing to the limited improvement in intravascular volume often seen with these agents. However, the proximal tubular site of action of the SGLT-2i’s may help circumvent these limitations.
Methods:
20 patients with type-2 diabetes and chronic, stable HF completed a randomized placebo-controlled crossover study of empagliflozin 10mg daily vs. placebo. Patients underwent an intensive 6-hour biospecimen collection and cardio-renal phenotyping at baseline and again after 14 days of study drug. After a 2-week washout, patients crossed over to the alternate therapy with repeat of the above protocol.
Results:
Oral empagliflozin was rapidly absorbed as evidenced by a 27-fold increase in urinary glucose excretion by 3 hours (p<0.0001). Fractional excretion of sodium (FENa) increased significantly with empagliflozin monotherapy vs. placebo (FENa 1.2 ± 0.7% vs. 0.7 ± 0.4% p=0.001) and there was a synergistic effect in combination with bumetanide (FENa 5.8 ± 2.5% vs. 3.9 ± 1.9%, p=0.001). At 14 days, the natriuretic effect of empagliflozin persisted, resulting in a reduction in blood volume (−208mL, IQR −536 to 153 mL vs −14mL, IQR −282 to 335 mL, p=0.035), and plasma volume (−138mL, IQR −379 to 154mL ± 453 mL, p=0.04). This natriuresis was not, however, associated with evidence of neurohormonal activation as change in norepinephrine was superior (p = 0.02) and all other neurohormones similar (p<0.34) during the empagliflozin vs. placebo period. Furthermore, there was no evidence of potassium wasting (p=0.20), or renal dysfunction (p>0.11 for all biomarkers), whereas both serum magnesium (p<0.001) and uric acid levels (p=0.008) improved.
Conclusion:
Empagliflozin causes significant natriuresis, particularly when combined with loop diuretics, resulting in an improvement in blood volume. However, off-target electrolyte wasting, renal dysfunction, and neurohormonal activation were not observed. This favorable diuretic profile may offer significant advantage in the management of volume status in HF patients and may represent a mechanism contributing to the superior long-term HF outcomes observed with these agents.
ClinicalTrials.gov number, NCT03027960
Keywords: natriuresis, SGLT-2 inhibitor, blood volume
Introduction:
Central to the pathophysiology of heart failure (HF) is dysregulation of sodium and fluid homeostasis.1–3 Specifically, there is maladaptive activation of sodium conserving pathways despite appropriate or even excess intravascular volume. The resulting congestion is a primary driver of symptoms and hospitalizations and is strongly associated with worsened survival.4–7 Currently, loop diuretics are the mainstay of therapy to counteract this sodium avidity.8 However, loop diuretics augment sodium excretion at the expense of neurohormonal activation, which occurs in part due to direct antagonism of sodium chloride entry into the salt sensor of the kidney, the macula densa.9–11 In addition to the well-established contribution of neurohormonal activation to HF progression and mortality, these pathways evolved to defend intravascular volume by increasing renal tubular sodium reabsorption.12 As such, it is not surprising that loop diuretic resistance and persistent volume overload is common with these agents.13, 14
Sodium glucose cotransporter-2 inhibitors (SGLT-2is) are glucose-lowering drugs that have been shown to reduce heart failure hospitalizations in patients with type 2 diabetes and, more recently, to improve clinical HF outcomes in patients with HF with reduced ejection fraction, including those without diabetes.15–18 While the mechanisms behind these benefits are unknown, one candidate contributor is the diuretic effect of these agents. Several studies in presumably euvolemic individuals with diabetes have demonstrated reduction in measured blood volume with these agents and evidence of hemoconcentration has been reproducibly observed in large SGLT2i trials.15, 19, 20 This apparent decrease in intravascular volume occurs despite these agents being substantially weaker natriuretics than loop diuretics, drugs that do not reduce blood volume in euvolemic subjects.21 Another important difference as compared to loop diuretics is the proximal tubular location action of the SGLT2i’s, which leads to increased rather than decreased sodium chloride delivery to the macula densa. This physiology may explain the small or absent neurohormonal response to SGLT2i’s compared to what would normally be expected with the observed reduction in blood volume.22 The purpose of the current investigation was to study the acute and intermediate (14-day) effects of SGLT-2i’s on natriuresis, volume status, and neurohormonal activation in patients with HF.
Methods:
De-identified data that support the findings of this study are available from the corresponding author upon reasonable request. Due to the small sample size single center nature of the study, creating a higher potential for re-identification, data will be provided only to qualified researchers with training in human subject confidentiality protocols.
Study Oversight
The study was an investigator-initiated trial that was conceived of and designed by the investigators with funding support from Boehringer-Ingelheim. Approval was obtained by the Yale University Institutional Review Board and written informed consent was obtained from all patients. The study was registered on clinicaltrials.gov (NCT03027960).
Study Population
Inclusion criteria included: (a) Stable HF (diagnosed by an advanced HF cardiologist) as defined by: 1) No hospitalizations during the preceding 60 days, 2) Stable HF medications for at least two weeks and stable diuretics for four weeks, 3) Opinion of the HF cardiologist that the patient is at optimal volume status; (b) Diagnosis of type 2 diabetes mellitus; (c) Regular home monitoring of blood glucose; (d) eGFR ≥ 45 mL/min/1.73m2; (e) 18 years of age or older. Exclusion criteria included: (a) Active titration of chronic HF medications expected during the study period; (b) Use of a non-loop diuretic aside from an aldosterone antagonist (≤ 25 mg spironolactone or ≤ 50 mg eplerenone); (c) Critical stenotic valvular disease, complex congenital heart disease, or prior heart transplant; (d) History of diabetic ketoacidosis, brittle diabetes and/or frequent hypoglycemia, or severe hypoglycemic episodes requiring emergency intervention in the last 6 months; (e) History of bladder dysfunction, incontinence, pyelonephritis, urosepsis, or frequent urinary tract infections; (f) Anemia with hemoglobin < 8 g/dL; (g) Pregnant or breastfeeding; (h) History of serious hypersensitivity; (i) Participation in another trial with an investigational drug within 30 days prior to informed consent; (j) Use of another SGLT2i; (k) Anticipated inability to participate in the required study procedures; (l) Inability to give written informed consent. In an effort to improve enrollment rate, and the cumulative safety experience gained in the ongoing large SGLT-2i trials, the eGFR inclusion criteria was modified to ≥ 20 mL/min/1.73m2 in July 2018.
Study Design
The study was a randomized, double-blind, placebo-controlled, crossover study consisting of treatment with either 10 mg empagliflozin or matched placebo daily for 14 days followed by a two-week washout period and crossover to 14 days of treatment with the alternate therapy (Supplemental Figure I). Randomization was performed in permuted blocks by the Yale-New Haven Hospital Investigational Drug Service.
Study Procedures
On days 1 and 14 of each study arm, patients underwent body fluid space measurements followed by biospecimen collection. Upon arrival to the clinical research unit, an IV was placed and the participant then underwent a one-hour period of quiet recumbency. The angle of the bed was noted at visit 1 and the same bed angle set for visit 2. Blood was collected from the existing IV into chilled potassium EDTA tubes and immediately placed on ice. All biomarkers, including neurohormones, were measured at the start of each study visit prior to study drug administration.
Patients were asked to perform an overnight fast the night prior to the study visit. At the beginning of each study visit, baseline body weight, vital signs, and blood and urine samples were collected. Following a 60-minute recumbency period, blood volume was determined via indicator dilution using I-131 albumin (Daxor Inc, New York, NY, USA). Next, empagliflozin or matched placebo and 10g of 99.9% deuterium oxide were administered orally. All patients then received a 500 mL bolus of 5% dextrose in sterile water administered intravenously over 30 minutes followed by a continuous infusion of 100 mL/hour in order to optimize the fidelity of urine clearance periods throughout the early part of the study visit. In 12 patients, due to a national shortage of 5% dextrose in water, the 500 mL bolus was replaced with consumption of 500 mL of an oral sports beverage (Gatorade) over 30 minutes followed by 100 mL per hour. Each patient received the same hydration route for testing during their crossover treatment.
Vital signs, blood and urine samples, blood glucose levels, and sonographic post-void residual volumes were obtained at 1.5, 3, 4.5 and 6 hours after empagliflozin or matched placebo administration. During this time, all urine produced was collected in 1.5-hour cumulative collections, ending with each specified time point. Three hours after taking empagliflozin or matched placebo, intravenous (IV) bumetanide was administered in a dose equivalent to the patient’s home loop diuretic dose, up to 4mg. Patients who were not on a chronic loop diuretic (n=1) received 0.5mg IV bumetanide. At 6 hours, body weight was measured. Patients also had a safety visit on days 3 and 7 of each study arm to ensure stable renal function, electrolytes, and blood pressure. After a two-week washout, participants were crossed over to the opposite therapy, and the above protocol was repeated in an identical fashion.
Trial Endpoints
The study was designed to evaluate both the acute (day 1) and 14-day diuretic and cardiorenal effects of empagliflozin. The primary acute endpoint was the natriuretic effect of empagliflozin both as monotherapy and in combination with loop diuretics. The primary 14-day endpoint was to understand if these acute natriuretic effects would translate into improved volume status after 14 days of therapy, as assessed by change in blood volume. Secondary endpoints were the change in neurohormones within 14 days of therapy. We also sought to extensively characterize the cardio-renal effects of empagliflozin in HF patients, thus a number of exploratory endpoints and biomarkers were also included.
Assays and Calculations
Throughout the manuscript, fractional excretion of sodium (FENa) is the primary metric used to describe sodium handling. FENa was chosen because it offers the best instantaneous assessment of sodium excretion, which is required given the various natriuretic experimental conditions during each study visit. When referring to other metrics such as absolute sodium excretion (mmol of sodium excretion during a timed collection), we explicitly specify the metric used. We defined HF with reduced ejection fraction as those with a left ventricular ejection fraction ≤40%. eGFR was calculated with the cystatin-based and creatinine-based Chronic Kidney Disease Epidemiology formulas.23
Blood volume, plasma volume, and red cell mass were determined on the BVA 100 semi-automated blood volume machine (Daxor Inc; New York, NY, USA) by I-131 albumin indicator dilution and spun hematocrit values. Plasma renin activity (PRA), norepinephrine, and aldosterone were measured using the commercially available ELISA kit from ALPCO as per manufacturer’s instructions (ALPCO, Salem, NH, USA). Total renin measured using ELISA kits from R&D Systems (Minneapolis, USA). The total renin immunoassay kit from R&D systems recognizes both active as well as prorenin. Further assay description can be found in the supplemental methods section. All neurohormonal and inflammatory biomarkers were log-transformed prior to analysis.
Statistical Analysis
Descriptive analysis and statistical tests were performed using SPSS, version 26 (IBM; Armonk, NY, USA), SAS software, version 9.4 (SAS Institute Inc; Cary, NC, USA), and Stata version 13.1 (StataCorp LP; College Station, TX, USA). Data with a normal distribution are presented as mean ± standard deviations. Categorical values are presented as frequencies and percentages and data with a skewed distribution are shown as median with interquartile ranges (IQR). We examined the difference between two interventions over time using linear mixed model (LMM) with random intercepts which incorporate correlated outcomes within-subject. The interactions between time (categorized as either within visit timepoint or between visit time point) and intervention (empagliflozin vs placebo) were tested for the intervention effects at a 5% significance level. To account for crossover design effect, the LMM was adjusted for the interaction between time and the order of the interventions
Results
A consort diagram can be found in the supplemental appendix (Supplemental Figure II). The cohort was predominately male (75%) and obese (BMI of 37±7 kg/m2), with an average age of 60 ± 12 years. The median hemoglobin A1c was 7.1% (6.2–10.5%) and 40% (8/20) were administering insulin at home. Patients with HF with reduced ejection fraction comprised 45% of the cohort (9/20). Participants were largely diuretic resistant, as the mean daily home loop diuretic dose was 244 ± 306 mg furosemide equivalents and the peak FENa after receiving 1.9 ± 1.4 mg bumetanide during the placebo period was 3.9 ± 1.9% (normal response would be an increase in FENa of >20%).24 Full baseline characteristics can be found in Table 1. To check for carryover effect, we examined the differences in serum and urinary electrolytes, in addition to the primary endpoints. We found no difference with respect to those who received empagliflozin or placebo first (p > 0.18 for all).
Table 1.
Characteristics of the Patients at Baseline.
| Characteristic | Cohort N=20 |
|---|---|
| Age – years | 60 ± 12 |
| Female sex – no. (%) | 5 (25) |
| Body mass index (kg/m2) | 37 ± 7 |
| Race – no. (%) | |
| Black | 8 (40) |
| NYHA functional classification – no. (%) | |
| 3 | 6 (30) |
| 4 | 0 (0) |
| Heart rate – beats/min | 74 ± 12 |
| Systolic blood pressure – mmHg | 126 ± 18 |
| Left ventricular ejection fraction (LVEF) – % | 42.9 ± 15.0 |
| HFrEF – no. (%)* | 9 (45) |
| NT-proBNP (IQR), pg/mL | 399 (139–2000) |
| Principal cause of heart failure – no. (%) | |
| Ischemic | 5 (25) |
| Non-ischemic | 15 (75) |
| Medical History – no. (%) | |
| Hypertension | 19 (95) |
| Coronary artery disease | 12 (60) |
| Arrhythmia | 11 (55) |
| Hyperlipidemia | 16 (80) |
| Hemoglobin A1C (IQR), % | 7.1 (6.2–10.5) |
| Renal Function | |
| eGFR by CKD-EPI, mL/min/1.73 m2 | 69.1 ± 19.0 |
| <60 mL/min/1.73 m2 – no. (%) | 7 (35) |
| Device therapy – no. (%) | |
| Implantable cardioverter-defibrillator | 10 (50) |
| Cardiac resynchronization therapy | 2 (10) |
| Heart failure medication – no. (%) | |
| Loop diuretic | 19 (95) |
| Oral furosemide equivalents – median (IQR), mg | 80 (40–300) |
| ACEi, ARB, or ARNi | 17 (85) |
| Beta-blocker | 19 (95) |
| Mineralocorticoid receptor antagonist | 10 (50) |
| Digoxin | 2 (10) |
| Glucose lowering medications – no. (%) | |
| Any form of insulin** | 8 (40) |
| Metformin | 12 (60) |
| Sulfonylurea | 3 (15) |
| DPP-4 inhibitor | 2 (10) |
HFrEF was defined as ejection fraction ≤ 40%
Includes short, intermediate, and long-acting forms of insulin.
NYHA = New York Heart Association; HFrEF = heart failure with reduced ejection fraction; NT-proBNP = n-terminal pro-hormone brain natriuretic peptide; eGFR CKDEPI = estimated glomerular filtration rate by chronic kidney disease epidemiology collaboration; ACEi = angiotensin converting enzyme inhibitor; ARB = Angiotensin II receptor blocker; ARNi = angiotensin receptor-neprilysin inhibitor; DPP-4 = dipeptidyl peptidase 4 inhibitor
Effect of Empagliflozin on Renal Glucose Handling
Oral empagliflozin was rapidly absorbed and delivered to the renal tubular site of action, as evidenced by a 27-fold increase in urinary glucose excretion which peaked at 3 hours and remained stable at that level for the remainder of the 6-hour study visit (Figure 1). After 14 days of empagliflozin therapy, there was no evidence of either tachyphylaxis to or intensification of the glucosuric effect, as repeated dosing yielded similar peak levels of glucose excretion at both the day 1 and day 14 visit (Figure 1, p=0.43). There was also no evidence of a carryover effect as pre-empagliflozin glucose excretion was similar between empagliflozin and placebo, regardless of randomization order (p=0.82). As previously reported, patients with a higher eGFR had substantially greater glucose excretion than patients with a lower eGFR (p interaction=0.001, Supplemental Figure III).
Figure 1. Fractional excretion of glucose (FEGlucose) on day 1 (dotted line) and day 14 (solid line) of treatment.
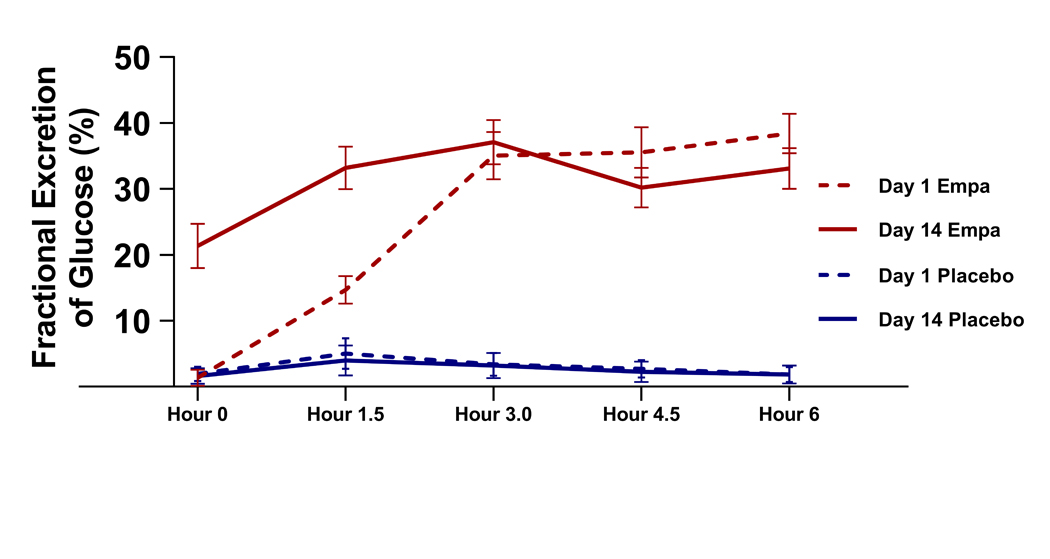
Empa = Empagliflozin.
Effect of Empagliflozin on Renal Sodium Handling
A modest natriuretic effect was observed with empagliflozin monotherapy (p<0.001), with a greater natriuresis over the 3-hour period after empagliflozin both during the day 1 (p<0.001) and day 14 (p=0.02) visits (Figure 2). The natriuresis demonstrated a similar temporal pattern to glucose excretion with the 3-hour FENa highest during the day 1 visit but a more sustained increase was observed at the day 14 visit. However, there was no significant attenuation in FENa with monotherapy between the day 1 visit and after 14 days of therapy (p=0.44). Additionally, natriuresis was greater with empagliflozin at the day 3 and day 7 safety visits (FENa= 2.4± 2.6% vs. 1.6± 1.6%, p=0.041). Following the administration of bumetanide, a significant synergistic effect on natriuresis was observed in patients receiving empagliflozin both during the day 1 (p<0.001) and day 14 (p=0.008) visits (Figures 2 & 3). Similarly, there was no detectable attenuation of the synergistic natriuretic effect following 14 days of empagliflozin therapy (p=0.15). With respect to FENa, there was no difference between patients who received empagliflozin first or second (p = 0.74). Over the 6-hour study visits urine output (1735mL, IQR 1365 to 2225 mL vs. 1405mL, IQR 1150 – 2065 mL, p=0.001), net fluid balance (−732mL IQR −382 to −1086mL vs. −377mL, IQR −75 to −933mL, p=0.001), and total sodium output (137mmol, IQR 87 to 180mmol vs. 111mmol, IQR 68 to 174 mmol, p=0.03) were greater with empagliflozin than placebo with no difference in this effect between the acute and day 14 visits (p=NS for all comparisons).
Figure 2. Empaglaflolzin increased natiuresis as monotherapy (panel A) and in combination with a loop diuretic (panel B) both with the first dose (top) and after 14 days of therapy (bottom).
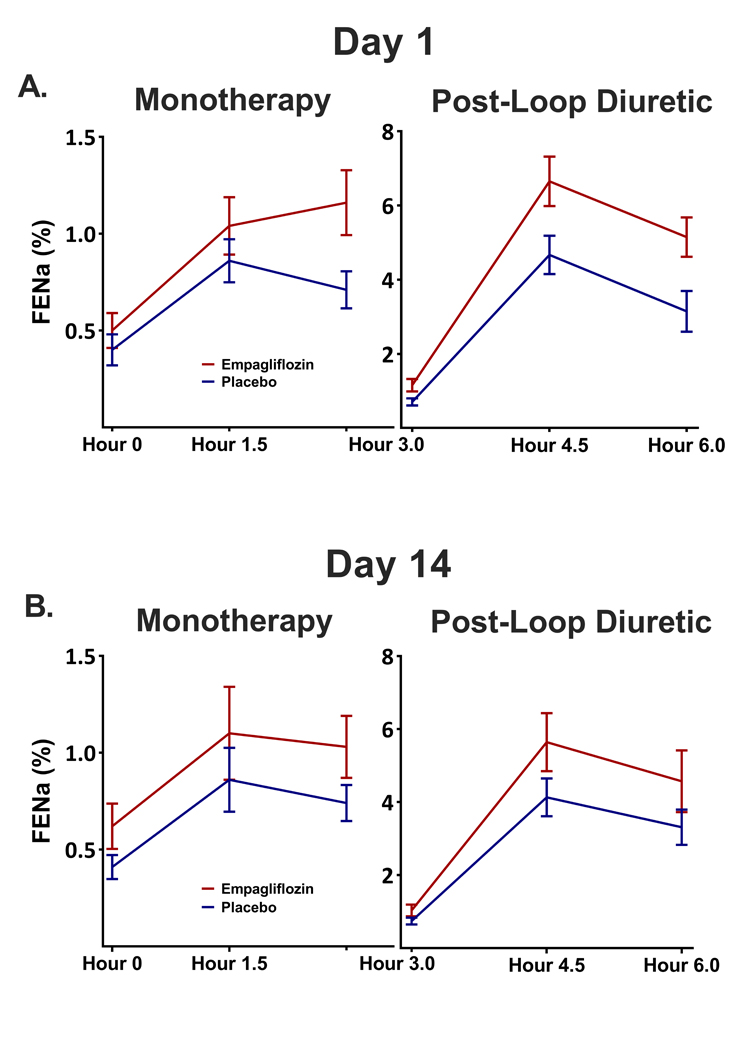
FENa: Fractional Excretion of Sodium
Figure 3. Natriuresis at the Day 1 Visit.
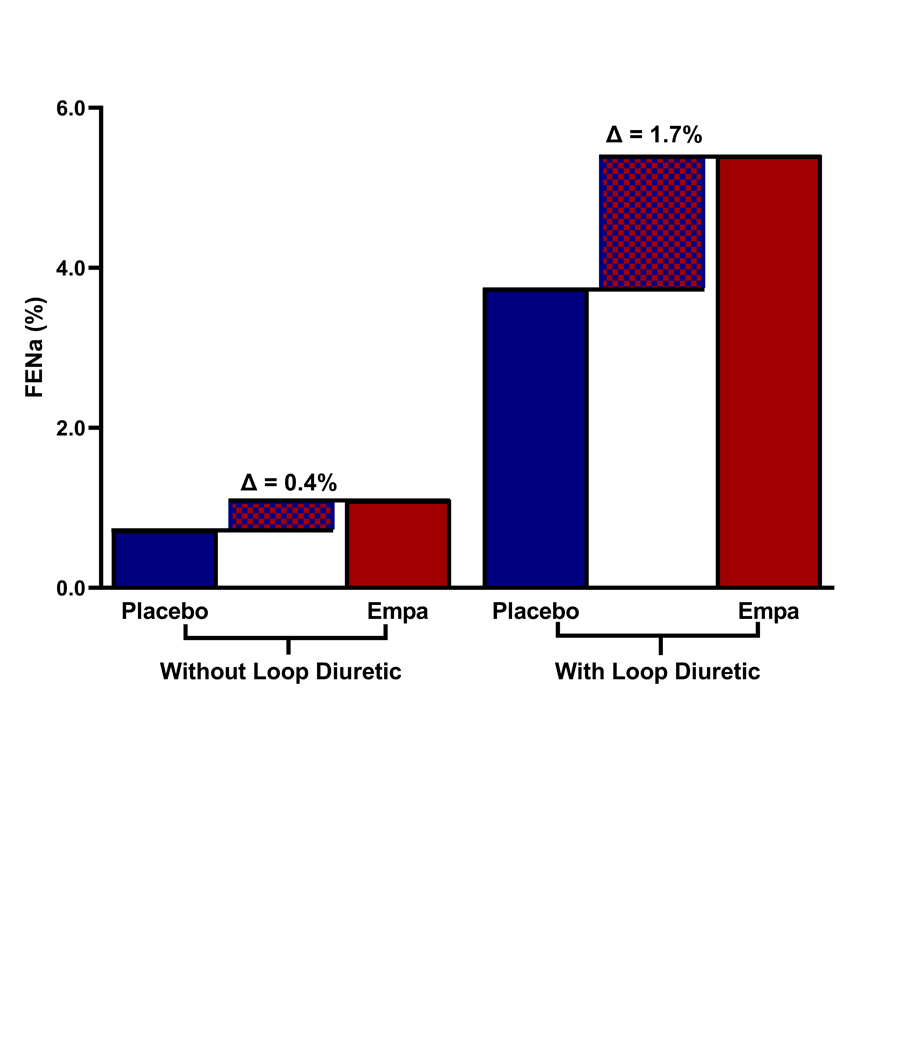
The solid bars represent measured sodium excretion under the 4 different experimental conditions. The thatched area represents the difference in natriuresis between the empagliflozin period and the placebo period. Notably the enhancement during the loop diuretic period is more than 4-fold greater than what is observed during monotherapy, illustrating the synergistic effect between loop diuretics and empagliflozin. Empa: Empagliflozin, Loop: Loop diuretic, FENa: Fractional Excretion of Sodium
Unlike the strong effect of eGFR on glucose excretion noted above, eGFR had a much smaller effect on the natriuretic effect of empagliflozin. For example, among patients receiving empagliflozin, the cumulative glucose excreted during the 6-hour study visits was linearly related to eGFR (r=0.61, p<0.001) but total sodium output was not (r=0.20, p=0.24), findings true both in the pre- and post-loop diuretic periods. Similarly, in patients with an eGFRs <60 mL/min/1.73m2, total sodium output was not significantly different than patients with an eGFRs ≥60 mL/min/1.73m2 (p=0.36) in contrast to the large differences noted in glucose excretion (Supplemental Figure III).
Mechanism of sodium excretion:
To better understand the mechanism of increased sodium excretion (i.e., direct effect vs. passive osmotic diuresis from glucosuria), we evaluated the relationship between the glucose osmotic load and metrics of sodium output and natriuresis. During the monotherapy period (when osmotic effects should be most prominent), urine osmolarity was not significantly increased by empagliflozin (401 ± 158 mOsm/L vs. 399 ± 179 mOsm/L, p=0.92), and there was only a small increase in total urine mOsms (221 ± 121 mOsm vs. 180 ± 119 mOsm, p=0.04). To further explore any possible contribution of urine osmolarity to sodium excretion, we examined the associations between several metrics of glycosuria (i.e. fractional excretion of glucose, total glucose excretion, urinary glucose concentration) and natriuresis, and found no significant positive correlation (Figure 4).
Figure 4. Relationship between natriuresis and metrics of glucosuria.

During the empagliflozin period, there was no association seen between FENa and FE Glucose (Panel A). There was an inverse correlation between both FENa and urinary glucose concentration (Panel B) and glucose excretion (Panel C) over the 6-hour study visits. FE= Fractional Excretion.
Changes in volume status and red cell indices:
14 days of empagliflozin therapy resulted in a greater reduction in total blood volume (p=0.035) and plasma volume (p=0.04) compared to placebo (Figure 5, Supplemental Figure IV). With respect to change in blood volume, there was no difference between patients who received empagliflozin first or second (p = 0.79). Baseline blood volume and the change in blood volume with empagliflozin did not differ between patients with HF with reduced ejection fraction and all others (p > 0.65 for both), and there was no difference in the primary endpoints when stratifying patients above or below the median baseline blood volume (Supplemental Table I). Total body water (p = 0.001) and body weight (p= 0.005) decreased during empagliflozin compared to placebo (Figure 6, Supplemental Figure IV). There was a moderately strong correlation between both weight loss and plasma volume loss (r = 0.50, p <0.001), as well as weight loss and total body water loss (r = 0.61, p <0.001).
Figure 5. Effects of Empagliflozin on total blood volume and plasma volume.
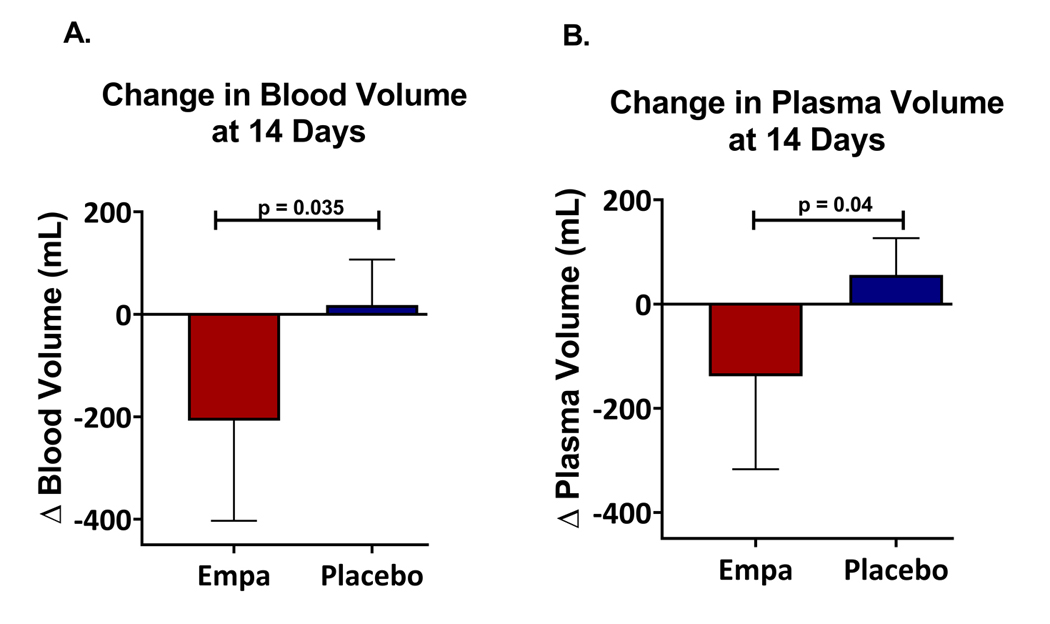
During the empagliflozin period, patients lost significantly more blood volume (Panel A) and plasma volume (Panel B) than during the placebo period. Empa = Empagliflozin.
Figure 6. Effects of Empagliflozin on weight and total body water.

During the empagliflozin period, patients lost significantly more weight (Panel A) and Total Body Water (Panel B). Empa = Empagliflozin.
At 14 days, erythropoietin increased more in patients receiving empagliflozin compared to placebo (p=0.037). Empagliflozin appeared to be protective against decreasing hematocrit during the 14-day, phlebotomy-intensive study (total 193 mL of blood removed over the 6-week study), as subjects had a 0.7 ± 2.1% decrease in hematocrit during placebo whereas hematocrit was stable (0.0 ± 2.2% change) during empagliflozin (treatment by randomization order p=0.011). Change in measured red cell volume was not significantly different between empagliflozin vs. placebo periods (p=0.64). Change in NT-proBNP was not different between the empagliflozin (−20.8, IQR −230.3 to 369.4 pg/mL) and placebo periods (40.9, −316.4 to 382.9, p=0.67).
Effect of empagliflozin on potassium, magnesium, and uric acid handling:
There was no change in total 6-hour potassium excretion with empagliflozin vs. placebo (28.9± 11.2 mmol vs 26.6± 8.9 mmol, p=0.20). Serum potassium levels were not different between the empagliflozin and placebo periods (4.3± 0.5 mEq/L vs 4.3± 0.5 mEq/L, p=0.51). On day 1, there was a trend toward reduction in total urine magnesium excretion (3.8 ± 1.5 mmol vs. 4.6 ± 1.2 mmol, p=0.08) with empagliflozin, and the urine sodium to magnesium ratio substantially increased (95 ± 56 vs. 55 ± 23, p=0.002). At the 14-day visit the increase in sodium/magnesium ratio had significantly attenuated (64 ± 27, p=0.002 vs. 56 ± 24, p=0.037, time by treatment p=0.006). The increase in magnesium excretion after 14 days of empagliflozin therapy was likely related to re-establishment of magnesium balance as plasma magnesium level significantly increased with empagliflozin (14-day magnesium level 2.4 ± 0.3 mEq/L vs. 2.1 ± 0.3 mEq/L at baseline, p<0.001). Empagliflozin therapy increased the fractional excretion of uric acid (p<0.001), with the largest effect surprisingly seen during the combination therapy with loop diuretic (9.7 ± 3.1% vs. 7.7 ± 3.3%, p=0.001). This uricosuric effect resulted in a reduction in serum uric acid levels after 14 days of therapy with empagliflozin vs. placebo (7.0 ± 2.0 μmol/L vs. 7.6 ± 2.4 μmol/L, p=0.008). Lastly, we examined the effect of urine urea concentrations and found no difference between empagliflozin and placebo periods during either the day 1 (69± 29 mmol/L vs 99± 38 mmol/L, p = 0.13) or day 14 (87± 31 mmol/L vs 101± 35 mmol/L, p = 0.57) visits.
Neurohormonal Activation and Inflammatory Biomarkers
Despite a reduction in blood volume, there was no detectable activation of the renin-angiotensin-aldosterone system with stable levels of plasma renin activity, total renin, and aldosterone (Table 2). Change in plasma norepinephrine levels were significantly better with empagliflozin compared to the placebo period (p=0.023, Table 2). There were no differences in inflammatory biomarkers tested, including c-reactive protein, IL-6, IL-10, IL-18, TNF-R1, TNF-R2 or GDF-15 (Table 2). Fasting ketone levels tended to increase following 14 days of empagliflozin therapy vs. placebo (0.046 ± 0.131 mmol/L vs −0.012 ± 0.105 mmol/L), a finding of borderline statistical significance (p=0.05). Absolute levels of biomarkers are reported in Supplemental Table II.
Table 2.
Biomarkers
| Biomarker | Change from Beginning to End of Treatment Period | ||
|---|---|---|---|
| Empagliflozin | Placebo | P Value | |
| Neurohormones | |||
| Norepinephrine (nmol/L) | 0.09 (−1.39 – 0.71) | 0.7 (0.02 – 2.33) | 0.023* |
| Plasma Renin Activity (ng/mL/hr) | 0.84 (−4.90 – 13.81) | 0.56 (−2.07 – 10.69) | 0.63 |
| Total Renin (pg/mL) | 241.3 (−252.8 – 744.9) | 368.5 (−56.3 – 1062.1) | 0.50 |
| Aldosterone (pg/mL) | 18.3 (−3.0 – 41.4) | 1.7 (−16.4 – 23.9) | 0.35 |
| Copeptin (pg/mL) | −8.19 (−45.15 – 14.81) | −4.04 (−19.09 – 16.31) | 0.08 |
| Cardiac | |||
| NT-proBNP (pg/mL) | −20.8 (−230.3 – 369.4) | 40.9 (−316.4 – 382.9) | 0.67 |
| Endothelin (pg/mL) | −0.13 (−0.68 – 0.41) | −0.15 (−0.66 – 0.31) | 0.95 |
| Troponin I (ng/mL) | −5.6 (−71.5 – 226.0) | 41.7 (−71.6 – 127.6) | 0.17 |
| Renal | |||
| Urine albumin (mg/g) | 0.4 (−10.0 – 7.4) | 0.02 (−4.6 – 6.5) | 0.39 |
| Urine NGAL (pg/mg) | −67 (−948 – 1751) | 305 (−1246 – 1890) | 0.61 |
| Urine KIM-1 (ng/mg) | 90 (−514 – 1211) | 226 (−132 – 894) | 0.023* |
| Inflammatory | |||
| IL-6 (pg/mL) | 0.0 (−2.32 – 4.88) | −1.24 (−10.25 – 1.24) | 0.43 |
| IL-10 (pg/mL) | 0.0 (0.0 – 3.56) | 0.0 (−3.56 – 20.6) | 0.62 |
| IL-18 (pg/mL) | −4.5 (−15.8 – 13.9) | −0.3 (−17.6 – 9.8) | 0.99 |
| GDF-15 (pg/mL) | 370.1 (−479.1 – 1194.0) | 183.6 (−299.5 – 877.3) | 0.96 |
| CRP (mg/L) | 0.07 (−0.14 – 0.41) | −0.01 (−0.65 – 0.24) | 0.48 |
| TNF-R1 (pg/mL) | 78.5 (−141.7 – 205.6) | 45.4 (−137.5 – 224.3) | 0.52 |
| TNF-R2 (pg/mL) | 410.8 (−807.9 – 824.9) | 588.8 (−334.4 – 1319.4) | 0.22 |
| Other | |||
| Erythropoietin (mIU/mL) | 3.44 (−2.46 – 11.87) | 0.95 (−2.97 – 4.79) | 0.037* |
Values are reported as Median (IQR). Renal biomarkers were normalized to urine creatinine.
p < 0.05
NT-proBNP, N-terminal pro – B-type natriuretic peptide; NGAL, neutrophil gelatinase-associated lipocalin; KIM-1, kidney injury molecule-1; IL-6, interleukin 6; IL-10, interleukin 10; IL-18, interleukin 18; GDF-15, growth/differentiation factor 15, CRP, C-reactive protein; TNF-R1, tumor necrosis factor receptor 1; TNF-R2 tumor necrosis factor receptor 2
Effects on Glomerular Filtration, Tubular Injury, and Renal Secretory Capacity
Overall, empagliflozin was well tolerated from a renal perspective. Changes in creatinine-based eGFR (−5.2± 6.5 mL/min/1.73m2 vs −1.2± 7.6 mL/min/1.73m2, p=0.11), cystatin C eGFR (−4.1± 11.0 mL/min/1.73m2 vs −1.8± 8.6 mL/min/1.73m2, p=0.50), β2-microglobulin (0.81± 1.81 μg/mL vs −0.21 ± 1.77 μg/mL, p=0.13), six hour creatinine clearance (6.3 ± 49.5 mL/min/m2 vs. −2.8 ± 20.5 mL/min/m2, p=0.46), and albuminuria (0.4 mg/g of creatinine, IQR −10.0 to 7.4 vs 0.02 mg/g of creatinine, IQR −4.6 to 6.4, p= 0.20) did not differ significantly between 14 days of empagliflozin vs. placebo. The change in renal tubular injury biomarker urine KIM-1 was significantly better during empagliflozin therapy [90 ng/mg of creatinine (IQR −514 to 1211)] than the placebo period. [226 ng/mg of creatinine (IQR −132 to 894; p=0.023)]. Urine NGAL was not significantly different (p =0.61) during empagliflozin vs. placebo periods (Table 2).
Vital Signs, Electrolytes, and Adverse Events
In general, empagliflozin was well-tolerated with few reported adverse events. Specifically, there were no instances of genitourinary infections, symptomatic hypoglycemia, or diabetic ketoacidosis. There were no significant differences in systolic blood pressure (121± 12 mmHg vs 127± 20 mmHg), diastolic blood pressure (74± 12 mmHg vs 74± 11 mmHg), or mean arterial pressure (90± 10 mmHg vs 92± 13 mmHg; p > 0.45 for all) or heart rate (76± 12 beats per minute vs 78± 12 beats per minute, p = 0.79) between the empagliflozin vs. placebo periods. Additionally, there was no significant effect on serum sodium (136± 7 mEq/L vs 136± 6 mEq/L), bicarbonate (22.0± 4.0 mEq/L vs 22.8± 5.7 mEq/L), chloride (101± 4 mEq/L vs 99± 4 mEq/L), calcium (9.4± 0.4 mg/dL vs 8.8± 2.0 mg/dL) or phosphorus levels (4.2± 1.1 mg/dL vs 3.7± 0.7 mg/dL; p= NS for all).
Discussion
The primary findings of this study of empagliflozin in patients with type 2 diabetes and HF are as follows: 1) Empagliflozin modestly enhanced natriuresis as monotherapy and when combined with loop diuretics exerted a meaningful synergistic natriuretic effect; 2) Unlike traditional diuretics, potassium wasting did not worsen and both magnesium and uric acid handling improved; 3) The natriuretic effect of empagliflozin was not driven by the urinary glucose load, indicating a direct natriuretic effect rather than an “osmotic” diuresis; 4) Unlike the effect on glucose excretion, the degree of renal dysfunction had limited importance in determining the natriuretic effect; 5) A natriuretic effect was sustained through 14 days of therapy with empagliflozin leading to a reduction in blood and plasma volume; 6) This intravascular volume contraction did not come at the expense of a significant RAAS or SNS activation, hypotension, or reflex tachycardia; 7) Empagliflozin therapy was not associated with a significant decline in GFR across multiple filtration markers and biomarkers of renal tubular injury were either unchanged or improved.
This study provides evidence that empagliflozin functions as a loop diuretic adjuvant with a clinically significant effect on natriuresis and what appears to be an excellent neurohormonal/renal/electrolyte safety profile. The most frequently employed loop diuretic adjuvant in patients with HF are the thiazide and thiazide-type diuretics.25 However, these agents are known to substantially increase potassium and magnesium wasting, worsen uric acid excretion, and commonly lead to a deterioration in renal function.26 Notably, the electrolyte abnormalities associated with combined loop-thiazide therapy are often clinically significant/severe, and the resultant hypokalemia may play a role in the signals for worsened survival associated with adjuvant thiazide use.27 In the current study, despite a significant adjuvant natriuretic effect, there was no change in potassium excretion and improvement in both magnesium and uric acid levels at 14 days. Furthermore, despite the significant reduction in plasma volume, we did not see any detectable worsening in renal function by several metrics, with evidence of some parameters actually improving. Overall these findings suggest empagliflozin may prove to be a preferred diuretic adjuvant over thiazides. Additional research to confirm these findings and understand the performance of empagliflozin in the setting of acute HF with active IV diuretics is needed.
It is well-described that despite days of aggressive intravenous dosing of loop diuretics, many patients hospitalized with decompensated HF do not experience an improvement in intravascular volume.13, 28–31 This inability of traditional diuretics to consistently improve intravascular volume is not unique to HF. For example, in otherwise healthy hypertensive volunteers, 30 days of 40mg twice daily furosemide did not reduce plasma volume.21 The lack of change in plasma volume is driven by the vigorous renal compensatory response to defend intravascular volume triggered by loop diuretics. Within minutes of loop diuretic administration, there is induction of neurohormonal systems and rapid deployment of mechanisms to conserve sodium, leading to development of diuretic resistance that is detectable within the first dose of diuretic.21, 32, 33 Some of this effect (e.g. renin secretion) is mediated by antagonism of sodium chloride entry into the kidney’s primary salt sensor, the macula densa.34 This effect theoretically could be observed with any proximally acting natriuretic agent and may not be specific to the SGLT2i class.
In contrast to the effect of loop diuretics, empagliflozin resulted in a persistent natriuresis at 14 days with a reduction in plasma volume. Counterintuitively, both occurred without triggering a detectable increase in neurohormonal activation. Several prior studies of SGLT2i in animal models or humans have identified a minimal increase or even suppression in neurohormonal levels.19, 35–39 One candidate mechanism that may explain the contrasting observation of SGLT-2is vs. loop diuretics is the resultant sodium chloride delivery to the macula densa with these drugs. While loop diuretics directly antagonize sodium chloride entry into the macula densa, increasing RAAS and SNS activity, SGLT2i’s inhibit sodium transport proximally thus increasing sodium chloride delivery.40 This increased sodium chloride delivery appears to be durable and of a physiologically relevant magnitude in humans, as evidenced by the decrease in GFR with initiation and increase in GFR with cessation of chronic SGLT2i therapy.41 These changes in GFR are thought to be secondary to tubuloglomerular feedback, which is also a response of the macula densa to increased salt delivery.42, 43 As a result, while a reduction in blood pressure and plasma volume would be expected to activate neurohormonal and sodium retaining systems, the increased sodium chloride delivery to the macula densa may counterbalance this effect.
Limitations:
Our study has several limitations. As this was a mechanistic study utilizing an intensive protocol, the sample size is small, and patients were highly selected from a single center. To facilitate the crossover design, only stable, euvolemic chronic HF patients were enrolled, so it remains unknown how these results may be applied to patients with acute decompensated HF and significant volume overload. Similarly, to ensure patients would have stable volume status and medical therapy for crossover, the treatment and washout periods were short by necessity. Given the lack of significant attenuation in the monotherapy natriuretic effect at 14 days, we can conclude these patients most likely had not re-established sodium balance. As a result, the ultimate chronic, fully adapted effects of empagliflozin on renal electrolyte handling and cardio-renal function cannot be determined from this study. While the natriuretic effects were unrelated to the glucosuric effects, the study only enrolled patients with diabetes and thus the results in patients without diabetes may differ. All patients in this study were fasting from the night prior to the study visit. It is known that sodium intake is a primary determinate of sodium output, both in the basal and diuretic induced states. As a result, it remains unknown how a dietary sodium load would influence empagliflozin induced natriuresis. Lastly, although this study focused natriuretic effects, multiple alternative hypothesis for the mechanism of benefit to SGLT2is have been proposed. Although our study highlighted the positive effects of empagliflozin on natriuresis and plasma volume, we cannot confirm or exclude these other mechanisms as important drivers of improved outcomes.
Conclusion
In this randomized, placebo-controlled crossover study of diabetic patients with chronic stable HF, empagliflozin resulted in a significant augmentation of natriuresis when combined with loop diuretics. This synergistic natriuresis persisted over the 14-day study period resulting in an improvement in plasma volume but did not occur at the expense of off target electrolyte wasting, renal dysfunction, or neurohormonal activation. This seemingly ideal diuretic profile may offer significant advantages in the management of volume status in HF patients and may represent an important mechanism underlying the superior long-term HF outcomes observed with these agents.
Supplementary Material
Clinical Perspective.
What is new?
Among patients with heart failure and diabetes, empagliflozin therapy resulted in increased natriuresis as monotherapy, and demonstrated a clinically meaningful synergistic effect when combined with a loop diuretic.
This enhanced natriuresis persisted over the 14-day study period, resulting in a reduction in plasma volume. However, it did not occur at the expense of off-target electrolyte wasting, renal dysfunction, or neurohormonal activation.
What are the clinical implications?
This favorable diuretic profile may offer significant advantage in the management of volume status in HF patients and may be a contributory mechanism to the superior long-term HF outcomes observed with these agents.
Additional study of the clinical utility of SGLT-2i’s as natriuretic agents is warranted.
Acknowledgments
Sources of Funding: This research was made possible by an independent grant from Boehringer-Ingelheim Pharmaceuticals, Inc. who provided both drug and financial support for the study. They had no role in study development, data interpretation, drafting of the manuscript, or decision to publish. The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE) and were fully responsible for all aspects of the trial and publication development. This work was also supported by NIH grant 5T32HL007950 (MG)
Dr Testani reports personal fees from Reprieve Medical, grants and personal fees from BMS, personal fees from AstraZeneca, personal fees from Novartis, grants and personal fees from 3ive Labs, personal fees from Cardionomic, personal fees from Bayer, grants and personal fees from Boehringer Ingelheim, personal fees from MagentaMed, grants from Otsuka, grants and personal fees from Sanofi, grants and personal fees from FIRE1, grants from Abbott, personal fees from W.L. Gore, personal fees from Windtree therapeutics, and grants from Merck outside the submitted work; in addition, Dr Testani has patents for treating diuretic resistance filed and issued.
Dr. Collins reports grants from NIH, PCORI, AHRQ and Astra Zeneca and personal fees from Ortho Clinical, Boehringer-Ingelheim, Roche and Relypsa Medical.
Dr. Riello reports consulting fees from Janssen, Johnson & Johnson, Pfizer, and Portola, and serves on advisory boards at AstraZeneca, Janssen, Johnson &Johnson, Medicure, and Portola.
Dr. Inzucchi serves on committees at Boehringer-Ingelheim, Lexicon/Sanofi, serves as a consultant for Novo Nordisk, AstraZeneca, VTV Therapeutics, Merck, Abott/Alere, Zafgen and has provided lectures for Merck and Boehringer-Ingelheim.
Dr. Butler has received research support from the National Institutes of Health, Patient Centered Outcomes Research and the European Union. He serves as a consultant for Abbott, Adrenomed, Amgen, Array, Astra Zeneca, Bayer, Boehringer-Ingelheim, Bristol Myers Squib, CVRx, G3 Pharmaceutical, Janssen, Lantheus, LivaNova, Luitpold, Medtronic, Merck, Novartis, NovoNordisk, Relypsa, Roche, Sanofi, Sequana, V-Wave Limited, Vifor.
The rest of the authors report no disclosures relevant to the content of this paper.
Nonstandard abbreviations:
- HF
Heart Failure
- SGLT-2i
Sodium Glucose Transporter 2 inhibitor
- nt-proBNP
n-terminal pro-brain natriuretic peptide
- GFR
glomerular filtration rate
References:
- 1.Sarraf M, Masoumi A and Schrier RW. Cardiorenal syndrome in acute decompensated heart failure. Clinical journal of the American Society of Nephrology : CJASN. 2009;4:2013–2026. [DOI] [PubMed] [Google Scholar]
- 2.Schrier RW. Use of diuretics in heart failure and cirrhosis. Semin Nephrol. 2011;31:503–12. [DOI] [PubMed] [Google Scholar]
- 3.Schrier RW and Ecder T. Gibbs memorial lecture. Unifying hypothesis of body fluid volume regulation: implications for cardiac failure and cirrhosis. Mt Sinai J Med. 2001;68:350–61. [PubMed] [Google Scholar]
- 4.Gheorghiade M, Filippatos G, De Luca L and Burnett J. Congestion in acute heart failure syndromes: an essential target of evaluation and treatment. The American Journal of Medicine. 2006;119:S3–S10. [DOI] [PubMed] [Google Scholar]
- 5.Gheorghiade M, Follath F, Ponikowski P, Barsuk JH, Blair JE, Cleland JG, Dickstein K, Drazner MH, Fonarow GC, Jaarsma T, Jondeau G, Sendon JL, Mebazaa A, Metra M, Nieminen M, Pang PS, Seferovic P, Stevenson LW, van Veldhuisen DJ, Zannad F, Anker SD, Rhodes A, McMurray JJ, Filippatos G, European Society of C and European Society of Intensive Care M. Assessing and grading congestion in acute heart failure: a scientific statement from the acute heart failure committee of the heart failure association of the European Society of Cardiology and endorsed by the European Society of Intensive Care Medicine. Eur J Heart Fail. 2010;12:423–33. [DOI] [PubMed] [Google Scholar]
- 6.Gheorghiade M and Pang PS. Acute heart failure syndromes. J Am Coll Cardiol. 2009;53:557–573. [DOI] [PubMed] [Google Scholar]
- 7.Gheorghiade M, Vaduganathan M, Fonarow GC and Bonow RO. Rehospitalization for heart failure: problems and perspectives. J Am Coll Cardiol. 2013;61:391–403. [DOI] [PubMed] [Google Scholar]
- 8.Felker GM, O’Connor CM and Braunwald E. Loop diuretics in acute decompensated heart failure: necessary? Evil? A necessary evil? Circulation Heart failure. 2009;2:56–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schnermann J Juxtaglomerular cell complex in the regulation of renal salt excretion. Am J Physiol. 1998;274:R263–79. [DOI] [PubMed] [Google Scholar]
- 10.Francis GS, Siegel RM, Goldsmith SR, Olivari MT, Levine TB and Cohn JN. Acute vasoconstrictor response to intravenous furosemide in patients with chronic congestive heart failure. Activation of the neurohumoral axis. Ann Intern Med. 1985;103:1–6. [DOI] [PubMed] [Google Scholar]
- 11.Francis GS, Benedict C, Johnstone DE, Kirlin PC, Nicklas J, Liang CS, Kubo SH, Rudin-Toretsky E and Yusuf S. Comparison of neuroendocrine activation in patients with left ventricular dysfunction with and without congestive heart failure. A substudy of the Studies of Left Ventricular Dysfunction (SOLVD). Circulation. 1990;82:1724–9. [DOI] [PubMed] [Google Scholar]
- 12.Brenner BM and Rector FC. Brenner & Rector’s the kidney. Philadelphia: Saunders Elsevier; 2008. [Google Scholar]
- 13.Miller WL and Mullan BP. Understanding the Heterogeneity in Volume Overload and Fluid Distribution in Decompensated Heart Failure Is Key to Optimal Volume Management Role for Blood Volume Quantitation. Jacc-Heart Fail. 2014;2:298–305. [DOI] [PubMed] [Google Scholar]
- 14.Testani JM, Brisco MA, Turner JM, Spatz ES, Bellumkonda L, Parikh CR and Tang WH. Loop diuretic efficiency: a metric of diuretic responsiveness with prognostic importance in acute decompensated heart failure. Circ Heart Fail. 2014;7:261–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zinman B, Lachin JM and Inzucchi SE. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med. 2016;374:1094. [DOI] [PubMed] [Google Scholar]
- 16.McMurray JJV, Solomon SD, Inzucchi SE, Kober L, Kosiborod MN, Martinez FA, Ponikowski P, Sabatine MS, Anand IS, Belohlavek J, Bohm M, Chiang CE, Chopra VK, de Boer RA, Desai AS, Diez M, Drozdz J, Dukat A, Ge J, Howlett JG, Katova T, Kitakaze M, Ljungman CEA, Merkely B, Nicolau JC, O’Meara E, Petrie MC, Vinh PN, Schou M, Tereshchenko S, Verma S, Held C, DeMets DL, Docherty KF, Jhund PS, Bengtsson O, Sjostrand M, Langkilde AM, Committees D-HT and Investigators. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N Engl J Med. 2019;381:1995–2008. [DOI] [PubMed] [Google Scholar]
- 17.Neal B, Perkovic V and Matthews DR. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N Engl J Med. 2017;377:644–657. [DOI] [PubMed] [Google Scholar]
- 18.Wiviott SD, Raz I and Sabatine MS. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. Reply. N Engl J Med. 2019;380:1881–1882. [DOI] [PubMed] [Google Scholar]
- 19.Lambers Heerspink HJ, de Zeeuw D, Wie L, Leslie B and List J. Dapagliflozin a glucose-regulating drug with diuretic properties in subjects with type 2 diabetes. Diabetes Obes Metab. 2013;15:853–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sha S, Polidori D, Heise T, Natarajan J, Farrell K, Wang SS, Sica D, Rothenberg P and Plum-Morschel L. Effect of the sodium glucose co-transporter 2 inhibitor canagliflozin on plasma volume in patients with type 2 diabetes mellitus. Diabetes Obes Metab. 2014;16:1087–95. [DOI] [PubMed] [Google Scholar]
- 21.Loon NR, Wilcox CS and Unwin RJ. Mechanism of Impaired Natriuretic Response to Furosemide during Prolonged Therapy. Kidney Int. 1989;36:682–689. [DOI] [PubMed] [Google Scholar]
- 22.Skrtic M and Cherney DZ. Sodium-glucose cotransporter-2 inhibition and the potential for renal protection in diabetic nephropathy. Curr Opin Nephrol Hypertens. 2015;24:96–103. [DOI] [PubMed] [Google Scholar]
- 23.Inker LA, Schmid CH, Tighiouart H, Eckfeldt JH, Feldman HI, Greene T, Kusek JW, Manzi J, Van Lente F, Zhang YL, Coresh J, Levey AS and Investigators C-E. Estimating glomerular filtration rate from serum creatinine and cystatin C. N Engl J Med. 2012;367:20–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Beutler JJ, Boer WH, Koomans HA and Dorhout Mees EJ. Comparative study of the effects of furosemide, ethacrynic acid and bumetanide on the lithium clearance and diluting segment reabsorption in humans. J Pharmacol Exp Ther. 1992;260:768–72. [PubMed] [Google Scholar]
- 25.Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi JL, Johnson MR, Kasper EK, Levy WC, Masoudi FA, McBride PE, McMurray JJV, Mitchell JE, Peterson PN, Riegel B, Sam F, Stevenson LW, Tang WHW, Tsai EJ, Wilkoff BL, Anderson JL, Jacobs AK, Halperin JL, Albert NM, Bozkurt B, Brindis RG, Creager MA, Curtis LH, DeMets D, Guyton RA, Hochman JS, Kovacs RJ, Kushner FG, Ohman EM, Pressler SJ, Sellke FW, Shen WK, Stevenson WG, Commi W and Force AAT. 2013 ACCF/AHA Guideline for the Management of Heart Failure A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;62:E147–E239. [DOI] [PubMed] [Google Scholar]
- 26.Jentzer JC, DeWald TA and Hernandez AF. Combination of Loop Diuretics With Thiazide-Type Diuretics in Heart Failure. J Am Coll Cardiol. 2010;56:1527–1534. [DOI] [PubMed] [Google Scholar]
- 27.Brisco-Bacik MA, Ter Maaten JM, Houser SR, Vedage NA, Rao V, Ahmad T, Wilson FP and Testani JM. Outcomes Associated With a Strategy of Adjuvant Metolazone or High-Dose Loop Diuretics in Acute Decompensated Heart Failure: A Propensity Analysis. J Am Heart Assoc. 2018;7:e009149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Strobeck JE, Feldschuh J and Miller WL. Heart Failure Outcomes With Volume-Guided Management. Jacc-Heart Fail. 2018;6:940–948. [DOI] [PubMed] [Google Scholar]
- 29.Griffin M, Rao VS, Fleming J, Raghavendra P, Turner J, Mahoney D, Wettersten N, Maisel A, Ivey-Miranda JB, Inker L, Tang WHW, Wilson FP and Testani JM. Effect on Survival of Concurrent Hemoconcentration and Increase in Creatinine During Treatment of Acute Decompensated Heart Failure. Am J Cardiol. 2019;124:1707–1711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Testani JM, Chen J, McCauley BD, Kimmel SE and Shannon RP. Potential Effects of Aggressive Decongestion during the Treatment of Decompensated Heart Failure on Renal Function and Survival: Insights from the ESCAPE Trial Limited Dataset. J Card Fail. 2010;16:S104–S104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Testani JM, Brisco MA, Chen J, McCauley BD, Parikh CR and Tang WH. Timing of hemoconcentration during treatment of acute decompensated heart failure and subsequent survival: importance of sustained decongestion. J Am Coll Cardiol. 2013;62:516–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shah S, Pitt B, Brater DC, Feig PU, Shen W, Khwaja FS and Wilcox CS. Sodium and Fluid Excretion With Torsemide in Healthy Subjects is Limited by the Short Duration of Diuretic Action. J Am Heart Assoc. 2017;6:e006135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Felker GM and Mentz RJ. Diuretics and ultrafiltration in acute decompensated heart failure. J Am Coll Cardiol. 2012;59:2145–53. [DOI] [PubMed] [Google Scholar]
- 34.Martinezmaldonado M, Gely R, Tapia E and Benabe JE. Role of Macula Densa in Diuretics-Induced Renin Release. Hypertension. 1990;16:261–268. [DOI] [PubMed] [Google Scholar]
- 35.Herat LY, Magno AL, Rudnicka C, Hricova J, Carnagarin R, Ward NC, Arcambal A, Kiuchi MG, Head GA, Schlaich MP and Matthews VB. SGLT2 Inhibitor–Induced Sympathoinhibition. A Novel Mechanism for Cardiorenal Protection. 2020;5:169–179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang N, Feng B, Ma X, Sun K, Xu G and Zhou Y. Dapagliflozin improves left ventricular remodeling and aorta sympathetic tone in a pig model of heart failure with preserved ejection fraction. Cardiovasc Diabetol. 2019;18:107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wan N, Rahman A, Hitomi H and Nishiyama A. The Effects of Sodium-Glucose Cotransporter 2 Inhibitors on Sympathetic Nervous Activity. Front Endocrinol (Lausanne). 2018;9:421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yoshimoto T, Furuki T, Kobori H, Miyakawa M, Imachi H, Murao K and Nishiyama A. Effects of sodium-glucose cotransporter 2 inhibitors on urinary excretion of intact and total angiotensinogen in patients with type 2 diabetes. J Investig Med. 2017;65:1057–1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Matthews VB, Elliot RH, Rudnicka C, Hricova J, Herat L and Schlaich MP. Role of the sympathetic nervous system in regulation of the sodium glucose cotransporter 2. J Hypertens. 2017;35:2059–2068. [DOI] [PubMed] [Google Scholar]
- 40.Vallon V, Blantz RC and Thomson S. Glomerular hyperfiltration and the salt paradox in early [corrected] type 1 diabetes mellitus: a tubulo-centric view. J Am Soc Nephrol. 2003;14:530–7. [DOI] [PubMed] [Google Scholar]
- 41.Wanner C, Heerspink HJL, Zinman B, Inzucchi SE, Koitka-Weber A, Mattheus M, Hantel S, Woerle HJ, Broedl UC, von Eynatten M, Groop PH and Investigators E-RO. Empagliflozin and Kidney Function Decline in Patients with Type 2 Diabetes: A Slope Analysis from the EMPA-REG OUTCOME Trial. J Am Soc Nephrol. 2018;29:2755–2769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kidokoro K, Cherney DZI, Bozovic A, Nagasu H, Satoh M, Kanda E, Sasaki T and Kashihara N. Evaluation of Glomerular Hemodynamic Function by Empagliflozin in Diabetic Mice Using In Vivo Imaging. Circulation. 2019;140:303–315. [DOI] [PubMed] [Google Scholar]
- 43.Heerspink HJ, Perkins BA, Fitchett DH, Husain M and Cherney DZ. Sodium Glucose Cotransporter 2 Inhibitors in the Treatment of Diabetes Mellitus: Cardiovascular and Kidney Effects, Potential Mechanisms, and Clinical Applications. Circulation. 2016;134:752–72. [DOI] [PubMed] [Google Scholar]
- 44.Introduction to Body Composition Assessment Using the Deuterium Dilution Technique with Analysis of Urine Samples by Isotope Ratio Mass Spectrometry. Vienna: INTERNATIONAL ATOMIC ENERGY AGENCY; 2011. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


