Abstract
Circular RNAs (circRNAs) are reported to be aberrantly expressed and perform different functions in numerous types of tumor; however, their expression levels in cutaneous squamous cell carcinoma (CSCC) remain largely unclear. Thus, the purpose of the present study was to investigate the function of circRNA_001937 in CSCC. Differential circRNA expression profiles of CSCC were analyzed using the Arraystar Human circRNAs chip and reverse transcription-quantitative PCR (RT-qPCR); and the effects of circRNA_001937 on cell behavior, in particular its regulation over the microRNA (miRNA)-597-3p/Fos-related antigen 2 (FOSL2) pathway, was investigated using a dual-luciferase reporter assay, and verified using RT-qPCR and western blotting. circRNA_001937 expression levels were significantly increased in CSCC tissues and cell lines compared with the corresponding adjacent tissues and control cells (P<0.05). The genetic silencing of circRNA_001937 with small interfering RNA significantly inhibited cell proliferation, and induced cell apoptosis (P<0.05). circRNA_001937 was observed to directly bind to miRNA-597-3p and serve as a sponge, which indirectly increased the expression levels of FOSL2, a miRNA-597-3p target gene. In conclusion, circRNA_001937 expression was increased in CSCC and silencing circRNA_001937 gene expression may inhibit CSCC progression by sponging the miRNA-597-3p/FOSL2 pathway.
Keywords: cutaneous squamous cell carcinoma, circular RNA_001937, microRNA-597-3p, Fos-related antigen 2, proliferation
Introduction
Cutaneous squamous cell carcinoma (CSCC) is one of the most common skin malignancies, accounting for 25-35% of all skin cancer types (1). The extent of tumor infiltration and metastasis greatly influences the clinical stage and prognosis of the disease; and although numerous treatment options are available for CSCC, including surgical excision, radiotherapy, photodynamic therapy and topical drug treatment, the prognosis of invasive and metastatic CSCC remains relatively poor and is associated with substantial levels of mortality (2). Furthermore, the incidence of CSCC continues to increase, which is largely due to the increased prevalence of risk factors including older populations, immunosuppression, chronic sun exposure and sensitivity to sunlight or ultraviolet radiation (1,3).
Circular RNAs (circRNAs) are a newly identified group of non-coding RNAs that lack 5'-caps and 3'-tails (4), which leaves them resistant to exonuclease- or ribonuclease-medi-ated degradation and permits their stable expression in numerous types of organisms. A number of studies have reported that circRNA expression levels are significantly increased in various types of tumor, where they serve as important molecules for tumor metastasis and recur-rence (5). circRNA may competitively bind to microRNA (miRNA) response elements to inhibit miRNA expression or function, which ultimately affects target genes (6,7). Fos-related antigen 2 (FOSL2) is a member of the activator protein 1 (AP-1) transcription factor family, which includes the various isoforms of Fos and Jun (8-10). Previous studies have demonstrated that FOSL2 is abnormally expressed in numerous different types of tumor, where it serves important functions in cell adhesion, migration, invasion, metastasis and proliferation (11-12).
The aim of the present study was to investigate the function of circRNA_001937 in CSCC. In the present study, differ-ential circRNA expression profiles of CSCC were analyzed using the Arraystar Human circRNAs chip and verified by RT-qPCR. In addition, the effects of circRNA on cell behavior, in particular its regulation of the miRNA-mRNA axis, were also investigated.
Materials and methods
Patient samples
The present study was approved by the Ethics Committee of the First Hospital of Jilin University (Jilin, China) and written informed consent was obtained from all patients. Three pairs of CSCC tissues and corresponding adjacent tissues were collected from the Department of Plastic Surgery at the First Hospital of Jilin University between September 2015 and November 2018. The clinicopathological features are shown in detail in Table SI. All specimens were confirmed by clinical and pathological diagnosis.
Cell lines
The human CSCC cell lines A431 and SCL-1, and the human immortal keratinocyte cell line HaCaT, were purchased from Guangzhou Genio Biotechnology Co., Ltd.
Reagents
circRNA_001937, miRNA-597-3p and FOSL2 mRNA primer sequences were purchased from Kangcheng Co., Ltd. Anti-FOSL2 primary antibody (1:500; cat. no. H00116173-B01P) and anti-GAPDH primary antibody (1:1,000; cat. no. R2655) were purchased from Sigma-Aldrich (Merck KGaA). RPMI-1640 medium, fetal bovine serum (FBS), crystal violet, Annexin V fluorescein isothiocyanate (FITC)/propidium iodide (PI) Cell Apoptosis Detection kit (cat. no. sc-4252 AK), MTT assays, Transwell plates, Matrigel and dimethyl sulfoxide were purchased from Santa Cruz Biotechnology, Inc.
Profiling of circRNA expression
The Arraystar Human circRNAs chip (Arraystar Inc.) was used to analyze the expres-sion of circRNAs in the CSCC tissues and corresponding adjacent tissues. Total RNA was extracted from the samples using an RNeasy Mini kit (cat. no. 74104; Qiagen GmbH), and the RNA was analyzed on the circRNAs chips. The expression levels were analyzed and quantified by Kangcheng Co., Ltd.
Reverse transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from A431 and SCL-1 cells using TRIzol® reagent (Thermo Fisher Scientific, Inc.). Total RNA was reverse transcribed into cDNA at room temperature using TaqMan™ reverse transcription reagents (cat. no. 4304134; Thermo Fisher Scientific, Inc.). qPCR was subsequently performed using the iScript™ cDNA Synthesis kit (cat. no. 1708890; Bio-Rad Laboratories, Inc.). The following thermocycling conditions were used for the qPCR: 40 cycles at 94°C for 15 sec; 20 cycles at 55°C for 30 sec; and 20 cycles at 70°C for 30 sec. The following primers were used: circRNA_001937 forward, 5'-TGA AGA ACA GCT CTC TGG CTG-3' and reverse, 5'-GCC CAC TTA ATC AGG GTC AGG T-3'; miRNA-597-3p forward, 5'-CGG AAT TCA TCT CAA GCC AAC-3' and reverse, 5'-CGG GAT CCC TTC ATT CAA GGT CAA TG-3'; FOSL2 forward, 5'-GAG AGG AAC AAG CTG GCT GC-3' and reverse, 5'-GCT TCT CCT TCT CCT TCT GC-3'; U6 (control for miRNA) forward, 5'-TTT AGG GCT TCG ATA CT-3' and reverse, 5'-TCT GCT GCA GCA CA-3'; and GAPDH (control for circRNA and mRNA) forward, 5'-GGT CCT GTT GTT TA-3' and reverse, 5'-TGC TCA TTC CCT C-3'. Expression levels were quantified using the 2-Δ∆Cq method (13) and the relative expression levels of target RNAs were normalized to the loading control U6.
Cell transfection
Small interfering RNA (siRNA/si) targeting circRNA_001937 (si-circRNA_001937) and the nega-tive control (si-NC) labeled with green fluorescent protein, miRNA-597-3p mimic, miRNA-597-3p inhibitor and the NC were synthesized by Kangcheng Co., Ltd. The sequence of si-circRNA_001937 was 5'-GGC AGC ACA TGT CAG GC-3' and the sequence of si-NC was 5'-TCT TTA GGG GTG TGC GTA GG-3'. The quantity of siRNA transfected was 50 nM. A431 and SCL-1 cells (1x105/well) were transfected using Lipofectamine® 2000 reagent (Thermo Fisher Scientific, Inc.) according to the manufacturer's protocol. Cells were trans-fected for 48 h prior to subsequent experimentation.
MTT assay
Following transfection, A431 and SCL-1 cells in the logarithmic growth phase were seeded at a density of 1x105/well into 96-well culture plates. Following 0, 12, 24 or 48 h of culture, 20 µl MTT solution was added/well for 6 h to determine cell proliferation. Following the MTT incubation, cells were washed with phosphate buffered saline (PBS) and the purple formazan crystals were dissolved using 100 µl dimethyl sulfoxide/well. Proliferation was subsequently analyzed at a wavelength of 490 nm using a microtiter plate reader.
Colony formation assay
Following transfection, A431 and SCL-1 cells (1x104/well) in the logarithmic growth phase were plated into 6-well culture plates and cultured for 2 weeks at 37°C. Following incubation, the cells were washed twice with PBS, fixed with 2% paraformaldehyde at 37°C for 30 min and subsequently stained with 0.5% crystal violet at 37°C for 15 min. Colonies were visualized using a Nikon electron microscope (magnification, x100; Nikon Corporation) and analyzed using ImageJ version 1.47 software (National Institutes of Health).
Matrigel invasion assay
Following transfection, A431 and SCL-1 cells (1x105/well) in the logarithmic growth phase were plated in the upper chambers of Transwell plates with Matrigel and fibronectin was also added to the upper chamber. RPMI-1640 medium supplemented with 20% FBS was plated in the lower chambers. Following incubation at 37°C for 24 h, the lower chamber cells were fixed with 2% parafor-maldehyde at 37°C for 30 min and stained with 0.5% crystal violet at 37°C for 15 min. Stained A431 and SCL-1 cells were visualized using a confocal microscope (magnification, x100) and ImageJ version 1.47 software was used to quantify the number of invasive cells.
Flow cytometric analysis of apoptosis
Cell apoptosis was performed using the Annexin V-FITC/PI apoptosis detection kit, according to the manufacturer's protocol. Following trans-fection, A431 and SCL-1 cells in the logarithmic growth phase were seeded into 96-well culture plates and were subsequently incubated with Annexin V-FITC and PI solution at 37°C for 20 min in the dark. Apoptotic cells were visualized using a BD FACSCalibur flow cytometer (BD Biosciences).
Dual-luciferase reporter assay
A431 and SCL-1 cells in the logarithmic growth phase were digested and seeded into 24-well culture plates. Subsequently, the A431 and SCL-1 cells were co-transfected with miRNA-597-3p mimics or the miRNA-597-3p NC and the wild-type (wt) or mutated (mut) 3'-untranslated region (UTR) of circRNA_001937 and FOSL2. Following 36 h of transfection, the luciferase activity was performed by a Dual-Luciferase Reporter Assay system (cat. no. E1910; Promega Corporation). Relative luciferase activity was normalized to the Renilla luciferase internal control.
Western blotting
A431 and SCL-1 cells of each group were lysed with RIPA buffer (Sigma-Aldrich; Merck KGaA). Protein quantification was carried out using a BCA protein assay kit (Promega Corporation). A total of 50 µg protein/lane was separated by 10% SDS-PAGE and subsequently transferred to polyvinylidene difluoride membranes. The membranes were then blocked for 1 h at room temperature with non-fat dry milk in TBST (Bio-Rad Laboratories, Inc.). The membranes were incubated with the anti-FOSL2 primary antibody (1:500; cat. no. H00116173-B01P; Gibco; Thermo Fisher Scientific, Inc.) and anti-β-actin (1:1,000; cat. no. R2655; Sigma-Aldrich; Merck KGaA) at 37°C overnight. Then, membranes were washed with PBS-Tween-20 buffer and subsequently incu-bated with a horseradish peroxidase-conjugated anti-rabbit antibody (1:1,000; cat. no. G-21234; Invitrogen; Thermo Fisher Scientific, Inc.) for 2 h at 4°C. Protein bands were visualized using the Pierce ECL Western blotting substrate (Pierce; Thermo Fisher Scientific, Inc.).
Statistical analysis
Statistical analysis was performed using SPSS 17.0 software (SPSS, Inc.) and the data were presented as the mean ± standard deviation. Statistical differences between two groups were determined using an unpaired Student's t-test or an χ2 test, whereas statistical differences between >2 groups were analyzed using a one-way analysis of variance, followed by Tukey's multiple comparison test. P<0.05 was considered to indicate a statistically significant difference.
Results
circRNA_001937 expression levels are significantly increased in CSCC
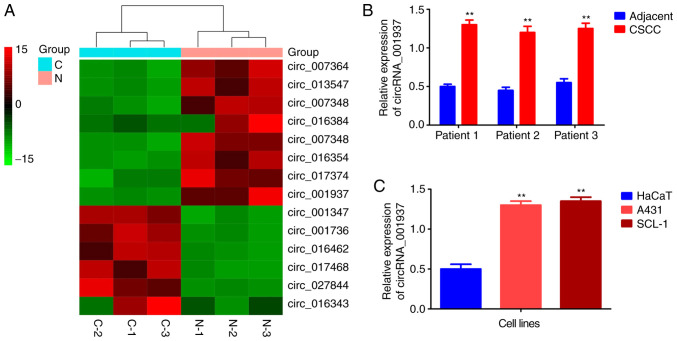
The cluster heat map demonstrated that circRNA_001937 expression levels were increased by 14.58-fold in CSCC tissues (Fig. 1A). Similarly, results from the RT-qPCR also reported that circRNA_001937 levels were significantly increased in CSCC tissues (P<0.01; Fig. 1B), and A431 and SCL-1 cells (P<0.01; Fig. 1C) compared with the corresponding adjacent tissues and HaCaT cells.
Figure 1.
circRNA_001937 was upregulated in CSCC (n=3). (A) Cluster heat map of circRNA chip. (B) RT-qPCR assay revealed that circRNA_ 001937 was significantly increased in three pairs of CSCC tissues compared with corresponding adjacent tissues. **P<0.01 vs. adjacent tissues. (C) RT-qPCR assay revealed that circRNA_001937 was significantly increased in A431 and SCL-1 cells compared with HaCaT cell lines. **P<0.01 vs. HaCaT cell lines. N, adjacent normal tissues; C, CSCC tissues; CSCC, cutaneous squamous cell carcinoma; circRNA, circular RNA; RT-qPCR, reverse transcription-quantitative PCR.
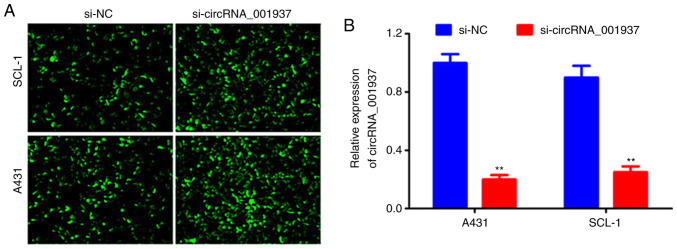
si-circRNA_001937 transfection
Following cell transfection with si-circRNA_001937 or si-NC, bright green fluorescence was observed in the si-NC and si-circRNA_001937 groups (Fig. 2A). The expression levels of circRNA_001937 in the si-circRNA_001937 group were significantly decreased compared with the si-NC group (P<0.01; Fig. 2B).
Figure 2.
si-circRNA_001937 transfection. (A) Bright green fluorescence was observed under a confocal laser scanning microscope. (B) Reverse transcription-quantitative PCR results revealed that the expression of circRNA_001937 in the si-circRNA_001937 group were significantly lower compared with the si-NC group. **P<0.01 vs. si-NC group. siRNA/si, small interfering RNA; circRNA, circular RNA; NC, negative control.
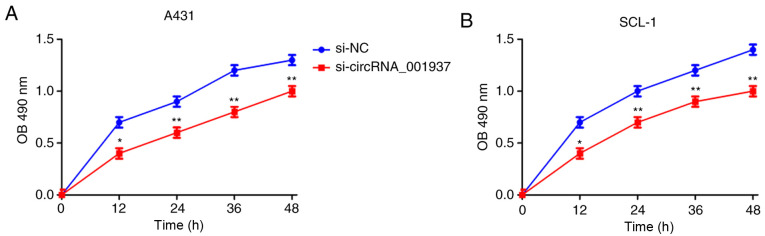
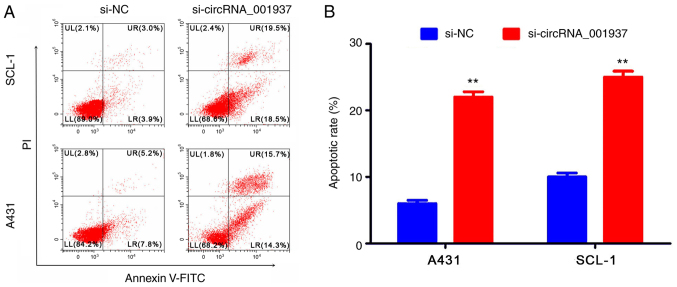
Silencing circRNA_001937 expression inhibits CSCC prolif- eration, and induces apoptosis
Subsequently, the effect of circRNA_001937 on cell behavior was investigated. The proliferative rate (P<0.05) were significantly reduced in the si-circRNA_001937 group compared with the si-NC group (Fig. 3). In addition, flow cytometric analysis identified that the apoptotic rate was significantly increased in the si-circRNA_001937 group compared with the si-NC group (P<0.01; Fig. 4).
Figure 3.
Silencing circRNA_001937 inhibits cell proliferation (n=3). (A) A431 cells. (B) SCL-1 cells. *P<0.05 and **P<0.01 vs. si-NC group. si, small inter-fering RNA; circRNA, circular RNA; OD, optical density; NC, negative control.
Figure 4.
Silencing circRNA_001937 inhibits cell invasion and induces cell apoptosis (n=3). (A) Cell apoptosis rate determined using flow cytometry. (B) Analysis of the apoptotic rate. **P<0.01 vs. si-NC group. si, small interfering RNA; circRNA, circular RNA; NC, negative control; PI, propidium iodide.
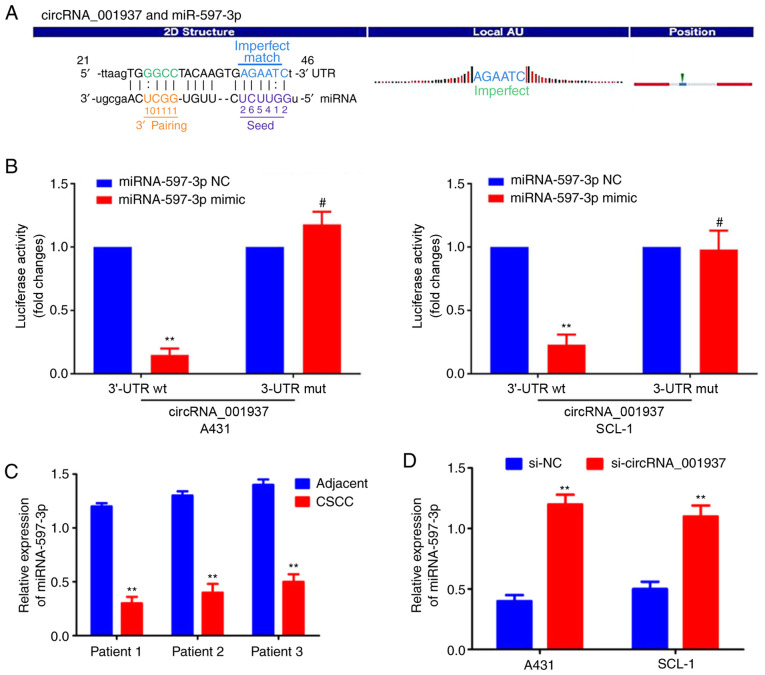
circRNA_001937 functions as an miRNA sponge for miRNA-597-3p
The specific binding sequences between circRNA_001937 and miRNA-597-3p are presented in Fig. 5A. Dual-luciferase reporter assays were used to demonstrate that the relative luciferase activity in the circRNA_001937-3'-UTR-Wt and miRNA-597-3p mimic co-transfection group was significantly decreased compared with the groups co-transfected with miRNA-597-3p NC or circRNA_001937-3'-UTR-Mut (P<0.01; Fig. 5B). miRNA-597-3p expression levels were also significantly decreased in CSCC tissues compared with corresponding adjacent tissues (P<0.01; Fig. 5C); however, these miRNA-597-3p expression levels were significantly increased following transfection with si-circRNA_001937 compared with the si-NC group (P<0.01; Fig. 5D).
Figure 5.
circRNA_001937 functions as an miRNA sponge for miRNA-597-3p. (A) Specific binding sequences of circRNA_001937 and miRNA-597-3p. (B) Dual-luciferase reporter assay results demonstrated the relative luciferase activity in each groups. **P<0.01 vs. circRNA_001937 wt + miRNA-597-3p NC; #P<0.01 vs. circRNA_001937 mut + miRNA-597-3p NC. (C) RT-qPCR results demonstrated that miRNA-597-3p was significantly downregulated in CSCC tissues compared with adjacent normal tissues. **P<0.01 vs. corresponding adjacent tissues. (D) RT-qPCR revealed that miRNA-597-3p was significantly increased following si-circRNA_001937 transfection. **P<0.01 vs. si-NC group. si-, small interfering RNA; circRNA, circular RNA; miRNA, microRNA; RT-qPCR, reverse transcription-quantitative PCR; CSCC, cutaneous squamous cell carcinoma; NC, negative control; wt, wild type; mut, mutated type; UTR, untranslated region.
FOSL2 is a direct target of miRNA-597-3p
The specific binding sequences between FOSL2 3'-UTR and miRNA-597-3p were predicted using TargetScan 7.0 software (Fig. 6A). Dual-luciferase reporter assay results demonstrated that the relative luciferase activity in cells co-transfected with FOSL2-3'-UTR-Wt and miRNA-597-3p mimic was significantly decreased compared with the miRNA-597-3p NC or FOSL2-3'-UTR-Mut groups (P<0.01; Fig. 6B). In addition, FOSL2 mRNA expression levels were significantly increased in CSCC tissues compared with the adjacent noncancerous tissues (P<0.01; Fig. 6C), whereas FOSL2 expression levels were significantly decreased in the miRNA-597-3p mimic group, and significantly increased in the miRNA-597-3p inhibitor group, compared with the miRNA-597-3p NC group (P<0.01; Fig. 6D).
Figure 6.
FOSL2 was identified as a direct target of miRNA-597-3p. (A) Specific binding sequences of FOSL2 3'-UTR and miRNA-597-3p were predicted using TargetScan 7.0. (B) Dual-luciferase reporter assay results demonstrated the relative luciferase activity in each group. **P<0.01 vs. FOSL2 wt + miRNA-597-3p NC; #P<0.01 vs. FOSL2 mut + miRNA-597-3p NC. (C) Reverse transcription-quantitative PCR results demonstrated that FOSL2 mRNA was significantly upregulated in CSCC tissues. **P<0.01 vs. corresponding adjacent tissues. (D) FOSL2 mRNA were significantly decreased in a miRNA-597-3p mimic group, and substantially increased in miRNA-597-3p inhibitor group. **P<0.01 vs. miRNA-597-3p NC group. miRNA, microRNA; CSCC, cutaneous squamous cell carcinoma; NC, negative control; wt, wild type; mut, mutated type; UTR, untranslated region; FOSL2, Fos-related antigen 2.
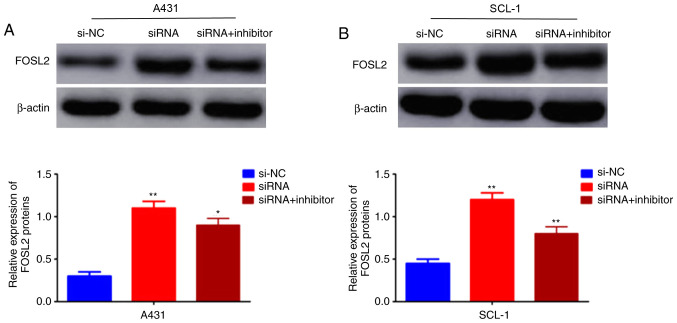
FOSL2 is activated by the circRNA_001937/miRNA-597-3p axis
The effects of the circRNA_001937/miRNA-597-3p axis on FOSL2 mRNA and protein expression levels was further investigated using RT-qPCR and western blotting, respectively. The expression levels of FOSL2 protein (P<0.01; Fig. 7A and B) and mRNA (P<0.01; Fig. 8) were significantly increased in the si-circRNA_001937 group compared with the si-NC group; and were substantially decreased in the si-circRNA_001937 and miRNA-597-3p inhibitor co-transfection group compared with the si-circRNA_001937 group.
Figure 7.
Effects of circRNA_001937/miRNA-597-3p axis on FOSL2. FOSL2 protein levels were examined in (A) A431 and (B) SCL-1 cells and were revealed to be significantly increased in the si-circRNA_001937 group, compared with the si-NC group, but substantially decreased in si-circRNA_001937 and miRNA-597-3p inhibitor co-transfection group, compared with the si-circRNA_001937 group. *P<0.05 and **P<0.01 vs. control group. circRNA, circular RNA; siRNA/si-, small interfering RNA; miRNA, microRNA; FOSL2, Fos-related antigen 2; NC, negative control.
Figure 8.
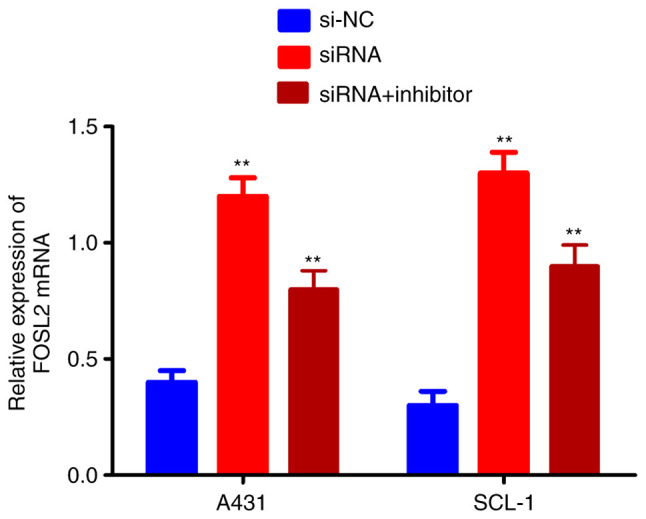
Effect of circRNA_001937/miRNA-597-3p axis on FOSL2. FOSL2 mRNA levels were significantly increased in the si-circRNA_001937 group compared with the si-NC group, but substantially decreased in the si-circRNA_001937 and miRNA-597-3p inhibitor co-transfection group compared with the si-circRNA_001937 group. **P<0.01 vs. control group. circRNA, circular RNA; siRNA/si-, small interfering RNA; miRNA, microRNA; FOSL2, Fos-related antigen 2; NC, negative control; CSCC, cutaneous squamous cell carcinoma.
Discussion
circRNAs serve as gene regulators in a variety of physiological functions and pathological processes, of which their functions in numerous types of cancer are currently being investigated. For example, circ_0003159 was identified as a potential biomarker for patients with gastric cancer. circ_001569 was identified to serve as a sponge for miR-145, which may prove beneficial as increased miR-145 expression promotes proliferation and invasion in colorectal cancer (14). Furthermore, circ_0043278 promoted non-small cell lung cancer (NSCLC) proliferation and migration through regulating miR-520 expression (15). Previous studies have also reported that circ_0016788 regu-lated hepatocellular carcinoma (HCC) tumorigenesis through the miR-486/cyclin-dependent kinase 4 pathway (16-18) and circRNA-mitochondrial tRNA translation optimization 1, whose expression was decreased in HCC, and was observed to sequester miRNA-9 and suppress HCC progression.
In the present study, the circRNAs chip and RT-qPCR results demonstrated that circRNA_001937 expression levels were increased in CSCC, and that silencing circRNA_001937 inhibited cell proliferation and invasion and induced cell apoptosis. circRNA_001937 is an exonic circRNA that is 2,850 nucleotides in length, and is located on chromosome 16 (19). Huang et al(20) reported that circRNA_001937 expression was significantly increased in the peripheral blood mononuclear cells of patients with tuberculosis, and that circRNA_001937 was correlated with tuberculosis severity, with expression levels successfully decreasing following treatment The results obtained in the present study suggest that circRNA_001937 may be used as a potential diagnostic biomarker for CSCC. However, the number of patients with CSCC used in the present study was small, and large-scale clinical samples and adequate follow-up studies are required for further verification. In addition, dual-luciferase reporter assays confirmed that circRNA_001937 served as a miRNA sponge towards miRNA-597-3p, which subsequently increased FOSL2 expres-sion. There are a limited number of studies examining the miRNA-597-3p/FOSL2 pathway. miRNA-597-3p is located on the 8p23.1 chromosome (21). Xie et al(22) reported that miR-597 targeting 14-3-3σ enhances cellular invasion and the epithelial-mesenchymal transition in nasopharyngeal carcinoma cells, whilst Zhang et al(23) revealed that a low expression of miR-597 is correlated with tumor stage and a poor outcome in breast cancer. FOSL2 is a member of the AP-1 transcription factor family (24). Previous studies have demonstrated that FOSL2 is abnormally expressed in numerous different types of tumor. Wang et al(25) identified that FOSL2 may positively regulate transforming growth factor-β1 signaling in NSCLC. Sun et al(26) confirmed that miR-143-3p inhibited the proliferation, migration and invasion of osteosarcoma through targeting FOSL2. Additionally, FOSL2 expression may be regulated through a number of different mechanisms. miRNA regulation is one method, including miRNA-30e (27) and miR-143-3p (26), which have been revealed to regulate the expression of FOSL2. These studies demonstrate that miRNA-597-3p and FOSL2 participate in the carcinogenesis and development of cancer. The present study demonstrated that miRNA-597-3p expression was significantly decreased, and FOSL2 expression was significantly increased, in CSCC tissues. The FOSL2 gene was additionally observed to be directly targeted by miRNA-597-3p, and FOSL2 expression levels were observed to be increased by circRNA_001937 serving as a sponge for miRNA-597-3p.
In conclusion, to the best of our knowledge, the present study provides the first evidence that circRNA_001937 expression is significantly increased in CSCC, and that silencing circRNA_001937 inhibits CSCC proliferation and invasion and induces apoptosis. Silencing circRNA_001937 gene expression may inhibit CSCC progression by preventing the sponging of the miRNA-597-3p/FOSL2 pathway. These results suggest a novel, potential therapeutic target for the treatment of patients with CSCC. Large-scale, clinical and adequate follow-up studies are required for further verification of these results.
Supplementary Data
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
LG and QL designed the study and performed the experiments. HJJ and DZ analyzed the data. All authors have read and approved the final manuscript.
Ethics approval and consent to participate
The present study was approved by the Ethics Committee of the First Hospital of Jilin University (Jilin, China), and written informed consent was obtained from all patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Castelo B, Viñal D, Maseda R, Ostios L, Sánchez D, García-Salvatierra B, Escámez MJ, Martínez-Santamaría L, Del Río M, Mora-Rillo M, et al. Epidemiology and natural history of cutaneous squamous cell carcinoma in recessive dystrophic epidermolysis bullosa patients: 20 years' experience of a reference centre in Spain. Clin Transl Oncol. 2019;21:1573–1577. doi: 10.1007/s12094-019-02073-3. [DOI] [PubMed] [Google Scholar]
- 2.Xiong Y, Dresser K, Cornejo KM. Frequent TLE1 expression in cutaneous neoplasms. Am J Dermatopathol. 2019;41:1–6. doi: 10.1097/DAD.0000000000001186. [DOI] [PubMed] [Google Scholar]
- 3.Ogata D, Tsuchida T. Systemic immunotherapy for advanced cutaneous squamous cell carcinoma. Curr Treat Options Oncol. 2019;20:30. doi: 10.1007/s11864-019-0629-2. [DOI] [PubMed] [Google Scholar]
- 4.Ding L, Zhao Y, Dang S, Wang Y, Li X, Yu X, Li Z, Wei J, Liu M, Li G. Circular RNA circ-DONSON facilitates gastric cancer growth and invasion via NURF complex dependent activation of transcription factor SOX4. Mol Cancer. 2019;18:45. doi: 10.1186/s12943-019-1006-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li Q, Deng C, Zhu T, Ling J, Zhang H, Kong L, Zhang S, Wang J, Chen X. Dynamics of physiological and miRNA changes after long-term proliferation in somatic embryogenesis of Picea balfouriana. Trees. 2019;33:469–480. doi: 10.1007/s00468-018-1793-x. [DOI] [Google Scholar]
- 6.Niwa Y, Yamada S, Sonohara F, Kurimoto K, Hayashi M, Tashiro M, Iwata N, Kanda M, Tanaka C, Kobayashi D, et al. Identification of a serum-based miRNA signature for response of esophageal squamous cell carcinoma to neoadjuvant chemo-therapy. J Transl Med. 2019;17:1. doi: 10.1186/s12967-018-1762-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kogure A, Kosaka N, Ochiya T. Cross-talk between cancer cells and their neighbors via miRNA in extracellular vesicles: An emerging player in cancer metastasis. J Biomed Sci. 2019;26:7. doi: 10.1186/s12929-019-0500-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.He J, Mai J, Li Y, Chen L, Xu H, Zhu X, Pan Q. miR-597 inhibits breast cancer cell proliferation, migration and invasion through FOSL2. Oncol Rep. 2017;37:2672–2678. doi: 10.3892/or.2017.5558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wrann CD, Eguchi J, Bozec A, Xu Z, Mikkelsen T, Gimble J, Nave H, Wagner EF, Ong SE, Rosen ED. FOSL2 promotes leptin gene expression in human and mouse adipocytes. J Clin Invest. 2012;122:1010–1021. doi: 10.1172/JCI58431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jahangiri L, Sharpe M, Novikov N, González-Rosa JM, Borikova A, Nevis K, Paffett-Lugassy N, Zhao L, Adams M, Guner-Ataman B, et al. The AP-1 transcription factor component Fosl2 potentiates the rate of myocardial differentiation from the zebrafish second heart field. Development. 2016;143:113–122. doi: 10.1242/dev.126136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hu Y, Li J, Li S, Wang S, Gao G, Hou J. Expression level of FOSL2 mRNA in blood leukocytes of type 2 diabetes mellitus patients in Uygurs in Xinjiang and its clinical sinificance. J Jilin Univ. 2016;42:545–550. In Chinese. [Google Scholar]
- 12.Li Z, Liu Y, Yan J, Zeng Q, Hu Y, Wang H, Li H, Li J, Yu Z. Circular RNA hsa_circ_0056836 functions an onco-genic gene in hepatocellular carcinoma through modulating miR-766-3p/FOSL2 axis. Aging (Albany NY) 2020;12:2485–2497. doi: 10.18632/aging.102756. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 13.Livak KJ, Schmittgen TD. Analysis of relative gene expres-sion data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 14.Xie H, Ren X, Xin S, Lan X, Lu G, Lin Y, Yang S, Zeng Z, Liao W, Ding YQ, Liang L. Emerging roles of circRNA_001569 targeting miR-145 in the proliferation and invasion of colorectal cancer. Oncotarget. 2016;7:26680–26691. doi: 10.18632/oncotarget.8589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cui J, Li W, Liu G, Chen X, Gao X, Lu H, Lin D. A novel circular RNA, hsa_circ_0043278, acts as a potential biomarker and promotes non-small cell lung cancer cell proliferation and migration by regulating miR-520. Artif Cells Nanomed Biotechnol. 2019;47:810–821. doi: 10.1080/21691401.2019.1575847. [DOI] [PubMed] [Google Scholar]
- 16.Guan Z, Tan J, Gao W, Li X, Yang Y, Li X, Li Y, Wang Q. Circular RNA hsa_circ_0016788 regulates hepatocellular carci-noma tumorigenesis through miR-486/CDK4 pathway. J Cell Physiol. 2018;234:500–508. doi: 10.1002/jcp.26612. [DOI] [PubMed] [Google Scholar]
- 17.Han D, Li J, Wang H, Su X, Hou J, Gu Y, Qian C, Lin Y, Liu X, Huang M, et al. Circular RNA circMTO1 acts as the sponge of microRNA-9 to suppress hepatocellular carcinoma progression. Hepatology. 2017;66:1151–1164. doi: 10.1002/hep.29270. [DOI] [PubMed] [Google Scholar]
- 18.Zhou J, Zhang WW, Peng F, Sun JY, He ZY, Wu SG. Downregulation of hsa_circ_0011946 suppresses the migration and invasion of the breast cancer cell line MCF-7 by targeting RFC3. Cancer Manag Res. 2018;10:535–544. doi: 10.2147/CMAR.S155923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ojha R, Nandani R, Chatterjee N, Prajapati VK. Emerging role of circular RNAs as potential biomarkers for the diagnosis of human diseases. Adv Exp Med Biol. 2018;1087:141–157. doi: 10.1007/978-981-13-1426-1_12. [DOI] [PubMed] [Google Scholar]
- 20.Huang ZK, Yao FY, Xu JQ, Deng Z, Su RG, Peng YP, Luo Q, Li JM. Microarray expression profile of circular RNAs in peripheral blood mononuclear cells from active tuberculosis patients. Cell Physiol Biochem. 2018;45:1230–1240. doi: 10.1159/000487454. [DOI] [PubMed] [Google Scholar]
- 21.Bay A, Coskun E, Oztuzcu S, Ergun S, Yilmaz F, Aktekin E. Plasma microRNA profiling of pediatric patients with immune thrombocytopenic purpura. Blood Coagul Fibrinolysis. 2014;25:379–383. doi: 10.1097/MBC.0000000000000069. [DOI] [PubMed] [Google Scholar]
- 22.Xie L, Jiang T, Cheng A, Zhang T, Huang P, Li P, Wen G, Lei F, Huang Y, Tang X, et al. MiR-597 targeting 14-3-3σ enhances cellular invasion and EMT in nasopharyngeal carcinoma cells. Curr Mol Pharmacol. 2019;12:105–114. doi: 10.2174/1874467212666181218113930. [DOI] [PubMed] [Google Scholar]
- 23.Zhang XY, Liu DJ, Yuan RB, Zhang DH, Li SR, Zhang SH, Zhang LY. Low expression of miR-597 is correlated with tumor stage and poor outcome in breast cancer. Eur Rev Med Pharmacol Sci. 2018;22:456–460. doi: 10.26355/eurrev_201801_14195. [DOI] [PubMed] [Google Scholar]
- 24.Li S, Fang XD, Wang XY, Fei BY. Fos-like antigen 2 (FOSL2) promotes metastasis in colon cancer. Exp Cell Res. 2018;373:57–61. doi: 10.1016/j.yexcr.2018.08.016. [DOI] [PubMed] [Google Scholar]
- 25.Wang J, Sun D, Wang Y, Ren F, Pang S, Wang D, Xu S. FOSL2 positively regulates TGF-β1 signalling in non-small cell lung cancer. PLoS One. 2014;9:e112150. doi: 10.1371/journal.pone.0112150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sun X, Dai G, Yu L, Hu Q, Chen J, Guo W. miR-143-3p inhibits the proliferation, migration and invasion in osteosar-coma by targeting FOSL2. Sci Rep. 2018;8:606. doi: 10.1038/s41598-017-18739-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ling L, Zhang SH, Zhi LD, Li H, Wen QK, Li G, Zhang WJ. MicroRNA-30e promotes hepatocyte proliferation and inhibits apoptosis in cecal ligation and puncture-induced sepsis through the JAK/STAT signaling pathway by binding to FOSL2. Biomed Pharmacother. 2018;104:411–419. doi: 10.1016/j.biopha.2018.05.042. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.