Abstract
SARS-CoV-2 is responsible for the 2019 coronavirus disease (COVID-19), a global pandemic that began in March 2020 and is currently in progress. To date, COVID-19 has caused about 935,000 deaths in more than 200 countries. The respiratory system is most affected by injuries caused by COVID-19, but other organs may be involved, including the cardiovascular system. SARS-CoV-2 penetrates host cells through the angiotensin 2 conversion enzyme (ACE-2). ACE-2 is expressed not only in the lungs, but also in other organs, including the cardiovascular system. Several studies have found that a good percentage of patients with severe COVID-19 have cardiac lesions, including myocardial fibrosis, edema and pericarditis. Pathological remodeling of the extracellular matrix caused by viral infection leads to myocardial fibrotic lesions. These fibrotic scars can cause cardiac dysfunction, reducing the ejection fraction caused by the presence of stiffened myocardial matrix, or cardiac arrhythmias that cause an alteration in the electrical conduction system of the heart. These cardiac dysfunctions can cause death. It is therefore essential to identify cardiac involvement early in order to act with appropriate therapeutic treatments. In this review, we describe what is known about cardiac injury from COVID-19, highlighting effective pharmacological therapeutic solutions to combat cardiac injury, particularly cardiac fibrosis, caused by COVID-19.
Keywords: SARS-CoV-2, ACE-2, Cardiovascular, Angiotensin, Cardiac injury, COVID-19
Graphical abstract
The massive and sudden release of pro-inflammatory cytokines can be responsible for contractile dysfunction and myocytic cardiac apoptosis with consequent Cardiac lesion. The TNF-a is a cytokine with pro-inflammatory properties that has an inotropic negative effect on the heart.
1. Introduction
1.1. SARS-CoV-2 (COVID-19)
On March 11, 2020 the World Health Organization (WHO) declared the state of global pandemic caused by the SARS-CoV-2, virus responsible for the coronavirus disease (COVID-19). To date, the data provided by the Coronavirus Resource Center of Johns Hopkins University (JHU) (https://coronavirus.jhu.edu/) recorded 29.6 million infected people and 935,000 deaths worldwide [1]. The global COVID-19 pandemic is causing serious and unexpected medical and socio-economic consequences and represents one of the greatest health challenges in human history. To date, 7 vaccines have entered phase 3 trials, representing an important hope for the final victory against SARS-CoV-2 (https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines [2]. Therefore, pending effective vaccines, the understanding of the pathophysiology and clinical features of COVID-19 infection and the use of valid therapeutic strategies to reduce serious complications are of paramount importance [3,4]. SARS-CoV-2 infection (COVID-19) usually occurs asymptomatically or with mild flu-like symptoms such as fever, cough, fatigue, less common gastrointestinal symptoms such as diarrhea and vomiting. In a small number of infected patients, however, especially in elderly patients or patients with existing diseases, the infection can seriously damage the patient's health by causing respiratory distress syndrome, which causes serious lung injury, which can even lead to death. SARS-CoV-2 penetrates host cells through the angiotensin 2 conversion enzyme (ACE-2). ACE-2 is expressed not only in the lungs, but also in other organs, including the cardiovascular system. Several studies have found that a good percentage of patients with severe COVID-19 have cardiac lesions, including myocardial fibrosis, edema and pericarditis [5,6,7].
1.2. Cardiovascular risk factors associated with COVID-19
Patients in old age and with pre-existing pathologies may have an increased risk of severe clinical outcome COVID-19 [8]. In particular, physiological conditions of the cardiovascular system (CV) appear to be of particular importance for COVID-19 outcome and severity. The COVID-19 itself can also induce severe myocardial lesions, cardiac arrhythmia, acute coronary syndrome and venous thromboembolism [9].
Epidemiological data indicate that mortality from COVID-19 is higher in patients with diseases such as diabetes, heart disease, chronic respiratory diseases (e.g. COPD, asthma) [10]. Some data show that COVID-19 severity is more influenced by CV pathologies present, compared to respiratory pathologies [11], although further epidemiological data are needed to define it, however, if the effects that comorbidities of the cardiovascular system may have a higher susceptibility and risk of infection remain to be clarified.
1.3. Cardiac injury and COVID-19
Cardiovascular diseases have been associated and observed already in epidemics caused by SARS and MERS [12,13]. Epidemiological evidence shows that cardiac disease is also associated with COVID-19 [14]. The cardiovascular system (CV) seems to have complex interactions with COVID19.
As already mentioned, in a good percentage of patients hospitalized with COVID19 the presence of myocardial lesions and high levels of cardiac troponin I and NT-pro-BNP has been observed.
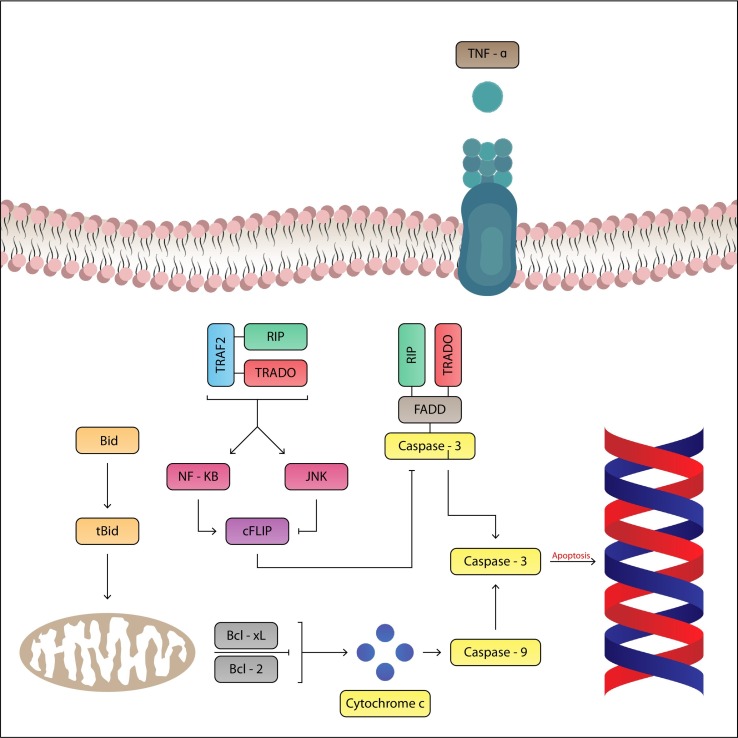
In particular, severe cases of COVID-19 are associated with a rapid progression of systemic inflammation, caused by a sudden release of pro-inflammatory mediators, called “cytokine storm”, which causes multi-organ lesions particularly in the lungs and heart. In particular, the excessive and sudden release of pro-inflammatory mediators can cause direct cardiac lesions (Fig. 1 ).
Fig. 1.
The massive and sudden release of pro-inflammatory cytokines can be responsible for contractile dysfunction and myocytic cardiac apoptosis with consequent Cardiac lesion. The TNF-a is a cytokine with pro-inflammatory properties that has an inotropic negative effect on the heart.
TNFa acts by binding to two specific receptors: TNF-R1 and TNF-R2. TNF-R1 has cytotoxic activity and induces the proliferation of fibroblasts and mediates apoptosis with negative inotropic effect on the heart. The activity mediated by TNF-R1 may play an important role in cardiac lesions COVID-19 induced.
In a situation of systemic inflammation, cardiac lesions caused, such as myocarditis, cardiac fibrosis, edema, and pericarditis [15] are responsible for a poor prognosis in COVID-19 patients. Cardiac injury caused by COVID-19 may have a direct cause mediated by virus penetration into the organ or an indirect cause mediated by the abnormal inflammatory/immune response that may be generated by the infection.
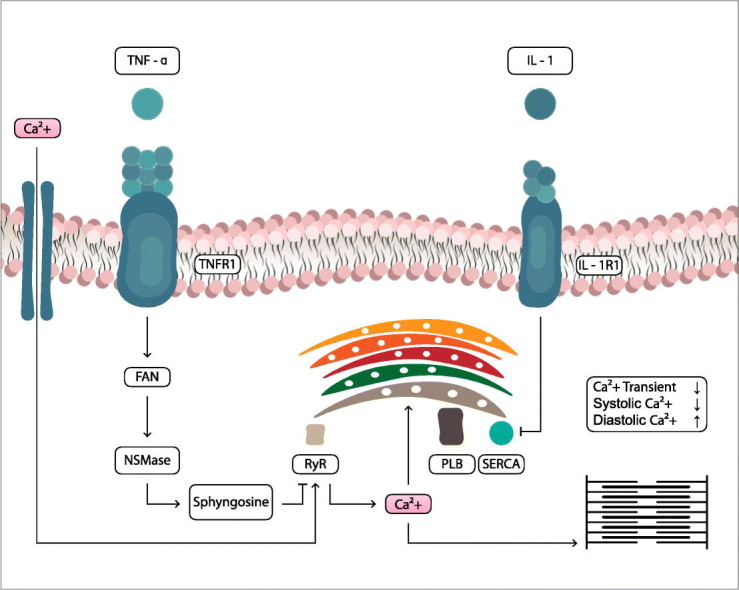
SARS-CoV-2 penetrates through the transmembrane protein ACE-2 into host cells, particularly type 2 pneumocytes, macrophages, endothelial cells and cardiac myocytes, causing inflammation and multi-organ damage. In particular, virus penetration into endothelial cells can be responsible for microvascular and macrovascular dysfunctions, as well as inflammatory/immune hyperreactivity, can destabilize atherosclerotic plaques and cause acute coronary syndromes. In addition, an increase in D-Dimer and a procoagulative effect have been observed in patients with severe COVID-19. Epidemiological data indicate that high levels of serum creatine kinase (CK) and lactate dehydrogenase (LDH) were found in patients with COVID-19 [16]. Similarly, viral penetration into cardiac cells [17] can cause damage and cardiac electrophysiology dysfunction. In addition, systemic inflammation and high levels of circulating cytokines can indirectly cause serious cardiac lesions and fulminating myocarditis [18]. A recent study provides important clinical and molecular information on cardiac involvement during COVID-19, advocating an association between (hyper)inflammation and myocardial damage and demonstrating that inflammatory/immune dysregulation SARS-CoV-2-induced and high cytokine concentrations may contribute to myocardial lesions and a worse prognosis [19]. Another interesting study analyzing autopsy cases COVID-19 showed that the SARS-CoV-2 genome was present in myocardial tissue, confirming that SARS-CoV-2 receptors are expressed within myocardial cells, and demonstrating that cytokine-induced organ dysfunction contributes to the disease process [20]. Finally, recent evidence shows the presence of pericardial effusion in COVID-19 patients with no history of cardiovascoular disease [21]. These studies show that direct or indirect cardiac damage induced by SARS-CoV-2 can result in short-term and acute effects leading to cardiac dysfunction, or in long-term effects. Another serious condition caused by the unregulated inflammatory/immune state typical of the more severe stages of SARS-CoV-2 infection is the formation of cardiac fibrotic tissue [22,23,24].
The presence of cardiac fibrotic tissue is probably a consequence of the cytokine storm and the sudden and elevated release of proinflammatory mediators, such as tumor necrosis factor alpha (TNF-α) and interleukin-1-beta (IL-1β).
Cardiac fibrosis is a process of pathological remodeling of the extracellular matrix (ECM), which leads to structural abnormalities and impaired cardiac function [25]. Excessive and continuous deposition of ECM leads to impairment of cardiac tissue function. These described aspects underline the importance of the need for multidisciplinary evaluation and treatment of the COVID-19 patient, including assessment of CV system conditions and the most appropriate therapy, to reduce mortality but also serious complications after the post-positive SARS-CoV-2 infection, in the convalescence period.
A combination therapy that acts with anti-inflammatory and antifibrotic properties in a synergistic way, and with agents effective in preventing or reducing cardiac damage is inevitable to reduce mortality from COVID-19.
1.4. The role of ACE-2 in cardiac damage caused by COVID-19
To date, the pathophysiological mechanisms underlying the development of cardiac lesions caused by COVID-19 are not completely clear. However, the role that ACE-2 plays in specific tissue infection is the subject of scientific debate. As mentioned above, SARS-CoV-2 infection is triggered by the binding of the viral S protein to human ACE-2. ACE-2 is known to play a key role in cardiovascular function [26]. Therefore ACE-2 may play a dual role in cardiac damage caused by SARS-CoV-2, in particular its cardiac tissue expression may mediate the entry into cardiac cells, causing direct damage, moreover the decrease in ACE-2 concentrations highlighted in the most severe stages of viral infection may further cause lung and cardiac damage, considering the role of ACE-2 on cardiovascular and respiratory homeostasis.
1.5. Pharmacological agents and therapeutic strategies
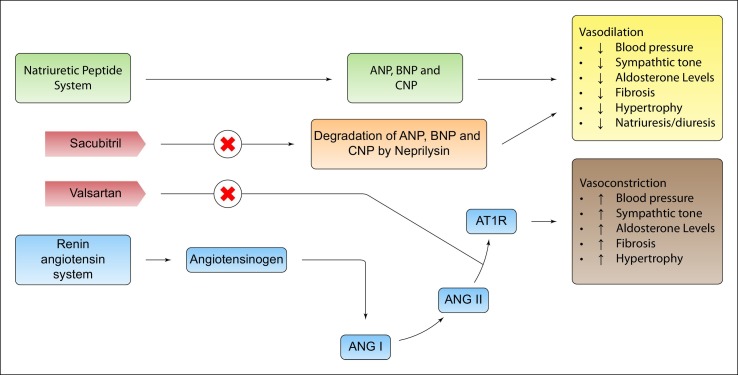
The management of the COVID-19 patient's therapeutic strategies is complex. To date there are no authorized vaccines or antiviral directed against SARS-CoV-2, there are ongoing clinical trials in this direction, about 300 studies are testing various therapeutic interventions against COVID-19 (ClinicalTrials.gov), 7 vaccines have entered phase 3 of clinical trials. In the meantime, the available and effective therapeutic treatments are pharmacological agents that can be considered to support the viral infection, among them there are immunomodulants, antiretrovirals, anticoagulants. In light of the above, it is also essential to adopt a therapeutic strategy aimed at preserving and protecting cardiovascular homeostasis and avoiding cardiac damage, especially in patients with more severe COVID-19. In view of the above, cardiac complications such as cardiac ischemia, cardiac arrhythmia, heart failure or venous thromboembolism caused by COVID-19 can be treated with common pharmacological therapeutic solutions. Finding added values to current therapeutic solutions may be important to preserve and eventually treat cardiac damage from COVID-19. Specifically, an interesting therapeutic strategy could be represented hypothetically by acting on the renin agiotensin system (RAS), in fact a decrease in ACE-2 concentrations could decrease the probability of SARS-CoV-2 penetration into cardiac cells. In this direction one could act with antibodies directed against ACE-2 [27], or alternatively by decreasing the expression of ACE-2 with renin angiotensin system modifiers (RAS), such as direct renin inhibitors (DRi). However, it should be considered that in the most severe stages of infection ACE-2 could play a protective role for both the respiratory and cardiac systems, In this case the use of ACE inhibitors or AT1-r receptor blockers (ARB), which have been shown to have in vitro modifying effects on ACE-2 concentrations, may have an added value. These scenarios are the subject of scientific discussion and at the moment there is no epidemiological evidence. In addition, with the use of RAS modifying agents such as AT1-r receptor blockers (ARB), it may be increased stimulation of AT2-r receptor with antifibrotic, antiflammatory and vasodilatory properties, in addition the vasoconstrictive, profibrotic, hyperproliferative and proinflammatory effects mediated by AT1r stimulation are also blocked. Finally, after ARB administration the response to hypertrophic and inflammatory growth induced by TNF-a is significantly attenuated [28]. Other drugs that can be used to influence the fibrotic response to cardiac lesions are beta-blockers, endothelin antagonists [29] and eplerenone (FDA approved since 2002) which was introduced as a drug that suppresses the formation of fibroses by blocking the aldosterone pathway [30]. Another pharmacological hypothesis to avoid cardiac lesions is the use of neprilisin inhibitors (NEPi) such as Sacubitril [31]. The beneficial effects of neprilisin inhibitor (NEPi) are attributable to the decrease in degradation of natriuretic peptides. Natriuretic peptides cause vasodilation by stimulating the guanylate cyclase receptor to produce cGMP. In addition, sacubitrile administration is known to decrease NT-proBNP, which in severe cases COVID-19 is increased. In addition, natriuretic peptides act to suppress the renin-angiotensin (RAS) and sympathetic systems and decrease endothelin secretion. In addition, natriuretic peptides also exert anti-inflammatory and antifibrotic effects. In particular, some evidence shows direct mediated anti-inflammatory effects (Fig. 2 ).
Fig. 2.
The use of AT1-r receptor blockers (ARB), can induce increased AT2-r receptor stimulation with antifibrotic, antifibrotic and vasodilatory properties, and the vasoconstrictive, profibrotic, hyperproliferative and proinflammatory effects mediated by AT1-r stimulation are also blocked. Finally, after ARB administration the response to hypertrophic and inflammatory growth induced by TNF-a is significantly attenuated. Neprilisin inhibitors decrease the degradation of natriuretic peptides. Natriuretic peptides cause vasodilation by stimulating the guanylate cyclase receptor to produce cGMP natriuretic peptides that also exert anti-inflammatory and antifibrotic effects.
On the basis of the evidence described and in relation to the hypotheses suggested, the use of sacubitril especially in severe cases, could be of therapeutic benefit, with cardioprotective, anti-inflammatory and antifibrotic effects [32,33,34]. The hyperactive and generalized inflammatory state caused by COVID 19 may be responsible for the above mentioned cardiac lesions. The use of immunomodulatory agents such as IL-6 Inhibitors [35] or steroids in this direction may be of clinical benefit. Pericarditis as described above may be another complication caused by COVID-19. In this direction the use of colchicine, a low cost drug used for many years for the treatment of diseases such as Bechet's disease, prevention and treatment of pericarditis and family Mediterranean fever, could be a valid therapeutic option. Colchicine's multiple effects with the reduction of IL-1, IL-6 and IL-18 that interfere with the NLRP3 inflammatory protein complex, a factor increasingly recognized for its role especially in recurrent idiopathic pericarditis and Mediterranean fever, could be of great benefit in patients with COVID-19. Finally, in patients with an altered profibrotic state, anticoagulant therapy is necessary to avoid further serious complications [36,37].
2. Conclusions
2.1. Perspectives and suggestions
In addition to lung damage, there may be significant cardiac involvement in patients with COVID-19, which is responsible for worsening the clinical condition of the host. The main cardiac manifestations can be edema, pericarditis, cardiac fibrosis, myocarditis and impairment of contractile function and cardiac electrophysiology. The cardiac status of patients with ongoing SARS-CoV-2 infection, or of surviving patients in convalescence period should be carefully monitored. There are many pharmacological agents therapeutic choices available to the clinician that, if used appropriately, and within the right timeframe, can be very useful to preserve cardiac homeostasis or reduce cardiac damage, lowering COVID-19 mortality. Finally, several pharmacological scientific hypotheses are to be demonstrated, well-structured clinical studies are necessary to generate further EBM in this direction.
Main statements
I, The undersigned, Francesco Ferrara and any other author, declare that:
-
•
We have no conflict of interest;
-
•
We have not received funding;
-
•
There are no sensitive data and no patients were recruited for this study;
-
•
The document does not conflict with ethical legislation.
-
•
All authors read and approved the final manuscript.
Founds
None.
Copyright
The authors certify that the manuscript is original, never submitted to other journal for publication before. All authors contributed equally to the manuscript and had the opportunity to revise and approve the final text.
Declaration of competing interest
None of the Authors have conflicts of interest to disclose.
Acknowledgements
None.
References
- 1.John Hopkins University https://www.arcgis.com/apps/opsdashboard/index.html#/bda7594740fd40299423467b48e9ecf6
- 2.Worl Health of Organization (WHO) DRAFT landscape of COVID-19 candidate vaccines. https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines
- 3.Peng Z. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. (in) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jian S., Yushun W., Chuming L. Cell entry mechanisms of SARS-CoV-2. Proc. Natl. Acad. Sci. U. S. A. 2020;117(21):11727–11734. doi: 10.1073/pnas.2003138117. (in) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ruan Q., Yang K., Wang W., Jiang L., Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020 doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020;8:420–422.5. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shi S., Qin M., Shen B., Cai Y., Liu T., Yang F., et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020 doi: 10.1001/jamacardio.2020.0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Guan W.J., Ni Z.Y., Hu Y., Liang W.H., Ou C.Q., He J.X., et al. China medical treatment expert group for Covid-19. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nishiga M., Wang D.W., Han Y. COVID-19 and cardiovascular disease: from basic mechanisms to clinical perspectives. Nat. Rev. Cardiol. 2020;17:543–558. doi: 10.1038/s41569-020-0413-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ruan S. Likelihood of survival of coronavirus disease 2019. Lancet Infect. Dis. 2020 doi: 10.1016/S1473-3099(20)30257-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Epidemiology Working Group for NCIP Epidemic Response The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China. Chin J Epidemiol. 2020;41:145–151. [Google Scholar]
- 12.Madjid M., Safavi-Naeini P., Solomon S.D., Vardeny O. Potential effects of coronaviruses on the cardiovascular system: a review. JAMA Cardiol. 2020 doi: 10.1001/jamacardio.2020.1286. [DOI] [PubMed] [Google Scholar]
- 13.Peiris J.S. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361:1767–1772. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zheng Y.Y., Ma Y.T., Zhang J.Y., Xie X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020;17:259–260. doi: 10.1038/s41569-020-0360-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Knockaert D.C. Cardiac involvement in systemic inflammatory diseases. Eur. Heart J. 2007;28:1797–1804. doi: 10.1093/eurheartj/ehm193. [DOI] [PubMed] [Google Scholar]
- 16.Tang N., Li D., Wang X., Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020;18:844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vitiello A., Ferrara F. Correlation between renin-angiotensin system and severe acute respiratory syndrome coronavirus 2 infection: what do we know. Eur. J. Pharmacol. 2020;883:173373. doi: 10.1016/j.ejphar.2020.173373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Guzik T.J., Mohiddin S.A., Dimarco A. COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options. Cardiovasc. Res. 2020;116(10):1666–1687. doi: 10.1093/cvr/cvaa106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Duerr G.D., Heine A., Hamiko M. Parameters predicting COVID-19-induced myocardial injury and mortality. Life Sci. 2020;260:118400. doi: 10.1016/j.lfs.2020.118400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lindner D., Fitzek A., Bräuninger H. Association of cardiac infection with SARS-CoV-2 in confirmed COVID-19 autopsy cases. JAMA Cardiol. 2020 doi: 10.1001/jamacardio.2020.3551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Luetkens J.A., Isaak A., Zimmer S., Nattermann J., Sprinkart A.M., Boesecke C., et al. Diffuse myocardial inflammation in COVID-19 associated myocarditis detected by multiparametric cardiac magnetic resonance imaging. Circ Cardiovasc Imaging. 2020;13(5):e010897. doi: 10.1161/CIRCIMAGING.120.010897. [DOI] [PubMed] [Google Scholar]
- 22.Ferrara F., Granata G., Pelliccia C., La Porta R., Vitiello A. The added value of pirfenidone to fight inflammation and fibrotic state induced by SARS-CoV-2: anti-inflammatory and anti-fibrotic therapy could solve the lung complications of the infection? Eur. J. Clin. Pharmacol. 2020:1–4. doi: 10.1007/s00228-020-02947-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vitiello A., Pelliccia C., Ferrara F. COVID-19 patients with pulmonary fibrotic tissue: clinical pharmacological rational of antifibrotic therapy. SN Compr Clin Med. 2020:1–4. doi: 10.1007/s42399-020-00487-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lu H., Peijun Z., Dazhong T., Tong Z., Rui H., Chenao Z., et al. Cardiac involvement in patients recovered from COVID-2019 identified using magnetic resonance imaging. J. Am. Coll. Cardiol. Img. 2020 doi: 10.1016/j.jcmg.2020.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jiang F. Angiotensin-converting enzyme 2 and angiotensin 1–7: novel therapeutic targets. Nat. Rev. Cardiol. 2014;11:413–426. doi: 10.1038/nrcardio.2014.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jellis C., Martin J., Narula J., Marwick T.H. Assessment of nonischemic myocardial fi- brosis. J. Am. Col. Cardiol. 2010;56:89–97. doi: 10.1016/j.jacc.2010.02.047. [DOI] [PubMed] [Google Scholar]
- 27.Tan WSD, Liao W, Zhou S, Mei D, Wong WF. Targeting the renin-angiotensin system as novel therapeutic strategy for pulmonary diseases. CurrOpinPharmacol. 201; 40: 9–17. doi: 10.1016/j.coph.2017.12.002. (Epub 2017 Dec 27). [DOI] [PubMed]
- 28.Flesch M., Hoper A., Dell’Italia L., Evans K., Bond R., Peshock R., et al. Activation and functional significance of therenin-angiotensin system in mice with cardiac restricted overexpression of tumor necrosis factor. Circulation. 2003;108:598–604. doi: 10.1161/01.CIR.0000081768.13378.BF. [DOI] [PubMed] [Google Scholar]
- 29.Ertl G., Frantz S. Healing after myocardial infarction. Cardiovasc. Res. 2005;66:22–32. doi: 10.1016/j.cardiores.2005.01.011. [DOI] [PubMed] [Google Scholar]
- 30.Neefs J., Van den Berg N.W.E., Limpens J., Berger W.R., Boekholdt S.M., Sanders P., et al. Aldosterone pathway blockade to prevent atrial fibrilla- tion: a systematic review and meta-analysis. Int. J. Cardiol. 2017;231:155–161. doi: 10.1016/j.ijcard.2016.12.029. [DOI] [PubMed] [Google Scholar]
- 31.Vitiello A., La Porta R., Ferrara F. Sacubitril valsartan and SARS-CoV-2. BMJ Evidence-Based Medicine. 2020 doi: 10.1136/bmjebm-2020-111497. [DOI] [PubMed] [Google Scholar]
- 32.Obata H., Yanagawa B., Tanaka K., Ohnishi S., Kataoka M., Miyahara Y., et al. CNP infusion attenuates cardiac dysfunction and inflammation in myocarditis. Biochem. Biophys. Res. Commun. 2007;356:60–66. doi: 10.1016/j.bbrc.2007.02.085. [DOI] [PubMed] [Google Scholar]
- 33.Soeki T., Kishimoto I., Okumura H., Tokudome T., Horio T., Mori K., et al. C-type natriuretic peptide, a novel antifibrotic and antihypertrophic agent, prevents cardiac remodeling after myocardial infarction. J. Am. Coll. Cardiol. 2005;45:608–616. doi: 10.1016/j.jacc.2004.10.067. [DOI] [PubMed] [Google Scholar]
- 34.Bukulmez H., Khan F., Bartels C.F., Murakami S., Ortiz-Lopez A., Sattar A., et al. Protective effects of C-type natriuretic peptide on linear growth and articular cartilage integrity in a mouse model of inflammatory arthritis. Arthritis Rheumatol. 2014;66:78–89. doi: 10.1002/art.38199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Atal S., Fatima Z. IL-6 inhibitors in the treatment of serious COVID-19: a promising therapy? Pharm Med. 2020;34:223–231. doi: 10.1007/s40290-020-00342-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tardif J.C., Kouz S., Waters D.D. Efficacy and safety of low-dose colchicine after myocardial infarction. N. Engl. J. Med. 2019;381:2497–2505. doi: 10.1056/NEJMoa1912388. [DOI] [PubMed] [Google Scholar]
- 37.Vitiello A., Ferrara F., Pelliccia C., Granata G., La Porta R. Cytokine storm and colchicine potential role in fighting SARS-CoV-2 pneumonia. Italian Journal of Medicine. 2020;14(2):88–94. doi: 10.4081/itjm.2020.1284. [DOI] [Google Scholar]