Abstract
A critical step in the life cycle of a virus is spread to a new target cell, which generally involves the release of new viral particles from the infected cell which can then initiate infection in the next target cell. While cell-free viral particles released into the extracellular environment are necessary for long distance spread, there are disadvantages to this mechanism. These include the presence of immune system components, the low success rate of infection by single particles, and the relative fragility of viral particles in the environment. Several mechanisms of direct cell-to-cell spread have been reported for animal viruses which would avoid the issues associated with cell-free particles. A number of viruses can utilize several different mechanisms of direct cell-to-cell spread, but our understanding of the differential usage by these pathogens is modest. Although the mechanisms of cell-to-cell spread differ among viruses, there is a common exploitation of key pathways and components of the cellular cytoskeleton. Remarkably, some of the viral mechanisms of cell-to-cell spread are surprisingly similar to those used by bacteria. Here we summarize the current knowledge of the conventional and non-conventional mechanisms of viral spread, the common methods used to detect viral spread, and the impact that these mechanisms can have on viral pathogenesis.
Keywords: Virus, Cell-to-cell spread, Syncytia, Filopodia, Nanotubes
1. Introduction
Viruses are intracellular parasites that have coexisted with cells from ancient times. Viruses hijack living cells and redirect many cellular processes to allow for efficient viral protein synthesis and replication. The success of the virus depends on the spread of some of these copies to new target cells to initiate a de novo infection. Many animal viruses have evolved more than one mechanism of viral spread, revealing a previously unexpected heterogeneity. Remarkably, some of these mechanisms allow the transfer of viral particles or components directly from cell-to-cell, without the need for virus release. Cell-to-cell spread has important advantages over the well-studied mechanism of spread by diffusion of cell-free viral particles. To achieve direct cell-to-cell spread, some viruses induce a dramatic remodeling of cell architecture to reach uninfected cells and transfer viral particles or components. It is critical for our understanding of viral propagation to comprehend what circumstances promote the use of one mechanism of spread over others, and whether different mechanisms can operate simultaneously at different stages of the infectious cycle. In this review we have focused on the current knowledge of different mechanisms of cell-to-cell spread in animal viruses.
2. Modes of cell-to-cell spread
2.1. Cell-free viral particles
Release of viral particles into the extracellular space and the subsequent re-entry of these particles into new target cells is one of the best described and understood mechanisms of virus spread. The release of cell-free particles is critical for viral spread between distant cells, or for spread between hosts. Virions released from a cell must interact with molecules at the cell surface of a new target cell as the first step to enter and initiate an infectious cycle. These molecules include receptors, co-receptors, and attachment factors. Cell-free viral particles can be released into the extracellular space through different mechanisms, such as: (a) cell lysis induced by viral proteins, as is the case for many non-enveloped viruses such as reoviruses, rotaviruses, adenoviruses and picornaviruses (Giorda and Hebert, 2013; Hu et al., 2012; Nieva et al., 2012); (b) by budding directly from the plasma membrane, where virions acquire their envelope, as is the case of human immunodeficiency virus (HIV-1), influenza, paramyxoviruses, and pneumoviruses (Lorizate and Krausslich, 2011; Votteler and Sundquist, 2013; Weissenhorn et al., 2013); (c) by exocytosis of intracellularly assembled viral particles, as is the case for bunyaviruses, flaviviruses and coronaviruses (Cifuentes-Munoz et al., 2014; Lorizate and Krausslich, 2011). Once released, different environmental and host factors affect the stability of viral particles and therefore their ability to initiate a new infection. Many of the viruses that can be released as cell-free virions can use additional mechanisms of spread that are summarized below.
2.2. Formation of actin tails
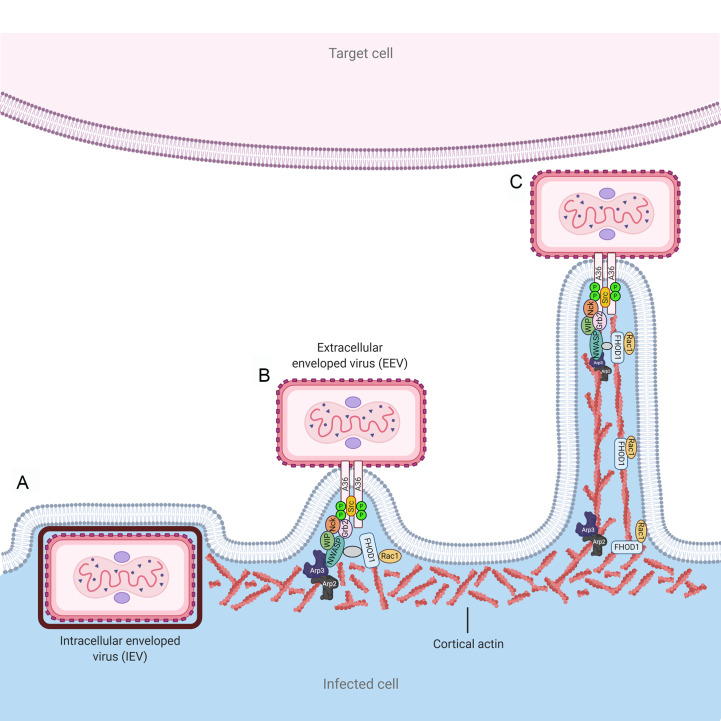
Vaccinia virus (VACV) replication occurs in cytosolic factories, and involves the formation of two forms of infectious virus, the intracellular mature virus (IMV) and extracellular enveloped virus (EEV) (Fig. 1 ). These two forms differ not only antigenically and structurally, but also in the way they exit the cell (Smith and Law, 2004). The majority of IMV particles are released from cells after lysis. Late in infection, IMV particles can also exit the cell through budding, but the significance of this mechanism is not completely understood. Budding from the plasma membrane, together with exocytosis, are the proposed ways that EEV exits cells. However, an important role of the actin cytoskeleton in VACV assembly was identified after early observations using high-voltage electron microscopy (Stokes, 1976). In infected cells, microvilli containing enveloped VACV particles at their tips were observed at the cell periphery (Stokes, 1976). The formation of the microvilli was shown to be sensitive to cytochalasin B but not to nocodazole, suggesting actin polymerization could play a role in their stabilization. The term actin tails was proposed for VACV-induced microvilli, due to their resemblance to actin tails formed in bacterial infections with Listeria, Shigella or Rickettsia (Cudmore et al., 1995; Welch and Way, 2013). Actin tails are initially observed at the interior of the cell, but as the infection progresses they project from the cell surface up to 20 μm. Enveloped virions located at the tip of actin tails were shown to project toward uninfected cells for direct cell-to-cell spread (Fig. 1) (Cudmore et al., 1995). Soon after the discovery of actin tails, it was demonstrated that phosphorylation of the viral protein A36R by Src and Abl family kinases was essential for the actin-based motility of vaccinia (Frischknecht et al., 1999a, Frischknecht et al., 1999b; Newsome et al., 2006). Phosphorylated A36R recruits the adaptor proteins Nck and Grb2 and the downstream effector N-WASP (Wiscott-Aldrich syndrome protein) together with the WASP Interacting Protein (WIP) (Donnelly et al., 2013; Frischknecht et al., 1999b; Scaplehorn et al., 2002). N-WASP can stimulate the actin-nucleating activity of the Arp2/3 complex (Taylor et al., 2011). In addition, activation of the formin FHOD1 by the small GTPase Rac1 was shown to stimulate vaccinia virus-induced actin tail initiation and elongation, a mechanism independent of the N-WASP-Arp2/3 pathway (Alvarez and Agaisse, 2013) (Fig. 1). Two isoforms of cytoplasmic actin, β and γ-actin, are present in actin tails, but only β-actin was found to be involved in actin nucleation induced by VACV (Marzook et al., 2017). Therefore, the signaling pathway initiated through phosphorylation of VACV A36R to induce actin tail formation for direct cell-to-cell spread was found to be an elegant mimic of cellular pathways. More recently, it was shown that two VACV proteins, A33 and A36, are necessary and sufficient to induce actin tail formation. The early expression of both proteins was shown to be crucial for rapid spread and repulsion of virions to neighboring uninfected cells (Doceul et al., 2010). Additional cellular factors such as clathrin and the clathrin adaptor protein AP-2 enhance actin-based motility of vaccinia. Clathrin and AP-2 are recruited by the extracellular virus during its egress to promote clustering of A36, thus potentiating actin-based motility and spread of the infection (Humphries et al., 2012). Casein kinase 2 (CK2) also enhances actin tail formation of vaccinia virus, potentially through direct phosphorylation of A36 and the recruitment of phosphorylated Src (Alvarez and Agaisse, 2012). Though actin tail formation has been explored in detail for vaccinia virus, the sequence homology of key viral proteins and additional evidence suggest that actin tail formation is a common strategy for orthopoxviruses (Duncan et al., 2018; Newsome and Marzook, 2015; Reeves et al., 2011; Welch and Way, 2013).
Fig. 1.
Formation of actin tails. Vaccinia virus (VACV) can spread from cell-to-cell through different mechanisms. In VACV infected cells, intracellular enveloped particles are transported to budding sites, where the outer viral membrane fuses with the plasma membrane (A). Extracellular enveloped viral particles remain attached to the plasma membrane and several cellular factors are recruited to the site through a cascade of events initiated by phosphorylation of the A36R protein cytosolic tail (B). Polymerization of F-actin underneath the plasma membrane occurs through activation of the cellular pathways N-WASP/Arp2/3 and FHOD1/Rac1, leading to elongation of actin tails (C). Viral particles can thus reach adjacent cells for rapid direct cell-to-cell spread.
2.3. Syncytia
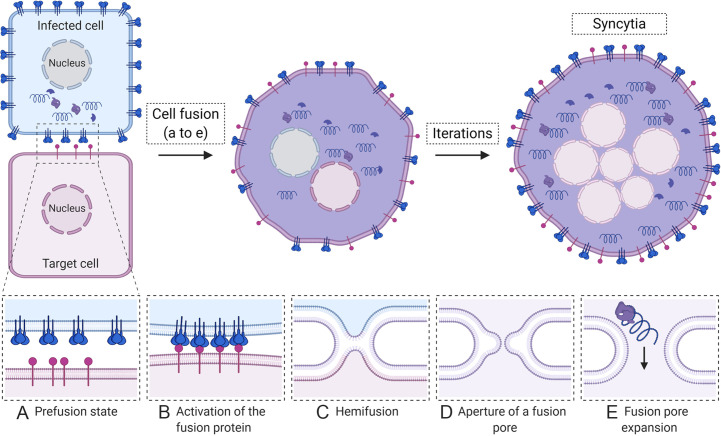
The fusion of membranes from adjacent cells results in multi-nucleated giant cells, also called syncytia. Infection with different viruses including paramyxoviruses (Takeuchi et al., 2003), pneumoviruses (Hamelin et al., 2004; Neilson and Yunis, 1990; Vargas et al., 2004), herpesviruses (Cole and Grose, 2003) and retroviruses (Nardacci et al., 2005) results in syncytia formation in vitro and in vivo. Cells infected with these viruses display a high concentration of viral fusion protein at the plasma membrane which can promote membrane fusion with neighboring cells. Specific fusion proteins have different requirements for triggering the conformational changes needed to drive fusion, such as exposure to low pH or the presence of receptors which bind the fusion protein or an associated receptor binding protein (Hernandez et al., 1996; Kielian, 2014). If the needed factors are present, the fusion protein can be activated to promote fusion between the membrane of the infected cell and the membrane of the target cell. The process of membrane fusion driven by viral fusion proteins involves several sequential steps, including: (a) activation of the fusion protein, which exposes a fusion peptide (FP); (b) insertion of the FP into the adjacent membrane; (c) clustering of fusion proteins at the site of fusion; (d) refolding of the fusion protein, which pushes together both adjacent membranes; (e) hemifusion, where only the outer leaflets of the membranes merge; (e) formation of a small fusion pore, and (f) enlargement of the fusion pore until complete merging of both membranes (Fig. 2 ) (Hernandez et al., 1996; Kielian, 2014; Lorizate and Krausslich, 2011). Iterations of this process generate massive syncytia containing numerous nuclei with the complete mixing of cytoplasmic material, which allows propagation of infection rapidly without the need for assembly of complete virions. Large rearrangements of the actin cytoskeleton are induced during cell fusion and syncytia formation. These rearrangements are regulated by the action of Rho GTPases Cdc42, Rac1 and RhoA (Eitzen, 2003). For example, it has been demonstrated that HIV-1 Env-coreceptor interactions activate the GTPase Rac1, which promotes actin rearrangements necessary for membrane fusion (Pontow et al., 2004). For the paramyxoviruses Hendra virus and parainfluenza virus type 5 (PIV5), constitutively active Rac1 and Cdc42 increased cell-to-cell fusion by their respective viral glycoproteins, as shown by quantitative reporter gene fusion assays. In contrast, constitutively active RhoA decreased Hendra virus Fusion protein (F)-mediated fusion and had minor effects on PIV5 cell-to-cell fusion (Schowalter et al., 2006). Cell-to-cell fusion mediated by the respiratory syncytial virus (RSV) fusion protein was shown to require RhoA signaling (Gower et al., 2005). Interestingly, a recent report correlated a RSV fusion protein with a hyperfusogenic phenotype with increased pathogenesis in mice (Hotard et al., 2015). Hypersyncytial viral strains (syn) have been extensively described for α-herpesviruses (Ambrosini and Enquist, 2015). Mutations associated with the syn phenotype have been mapped to viral genes encoding the glycoproteins, with a predominance in the gB and gK genes (Ambrosini and Enquist, 2015; Bond et al., 1982). Early and recent reports have associated the hyperfusogenic strains of α-herpesviruses with increased electrical activity in infected neurons (Mayer et al., 1985; McCarthy et al., 2009). Additionally, syncytia formed between neurons and satellite cells during varicella-zoster virus (VZV) infection have been suggested to contribute to neuropathogenesis (Reichelt et al., 2008). In contrast, syncytia formation by VZV mutant hyperfusogenic glycoproteins might be detrimental, rather than beneficial, for viral spread and pathogenesis in skin cells (Yang et al., 2014). Whether syncytia formation contributes to pathogenicity of other viruses such as HIV-1 is still a matter of controversy (Compton and Schwartz, 2017). Remarkably, while viral fusion proteins are well studied in enveloped viruses, another type of viral fusogen that promotes syncytia formation has been described for the non-enveloped orthoreoviruses (Duncan, 2019; Shmulevitz and Duncan, 2000). This family of non-structural viral fusion proteins, named fusion-associated small transmembrane (FAST) proteins, are unusual in that they do not mediate viral entry but rather fusion between cells using combinations of membrane effector motifs. Cell-to-cell fusion mediated by the reptilian orthoreovirus p14 fusogen is accomplished by recruitment of the Src kinase, the Grb adaptor protein and N-WASP, thus initiating branched actin assembly (Chan et al., 2020). The factors recruited by the reptilian orthoreovirus p14 for cell-to-cell fusion are thus remarkably similar to those recruited by vaccinia virus to induce actin tail formation, as described above. Despite their role in orthoreovirus cell-to-cell spread, syncytia formation is not essential for virus replication or progeny production of orthoreoviruses in vitro (Duncan et al., 1996). However, the maintenance of these proteins across reovirus species strongly suggests they provide a competitive advantage to the virus (Duncan, 2019).
Fig. 2.
Syncytia formation. Infected cells (blue) display a high concentration of the viral fusion protein (shown as blue spikes) on cell surfaces. When infected cells come into proximity with an uninfected cell (purple), several sequential steps lead to syncytia formation and spread of infection. (A) Initially, the fusion protein is in an inactive prefusion state; (B) upon the proper stimulus (such as binding a receptor shown in purple on the target cell), the fusion protein is triggered, which induces conformational changes, insertion of a fusion peptide into the adjacent membrane, and conformational changes that bring both membranes, viral and cellular into close proximity, followed by a partial merging known as hemifusion (C). Full merger of both membranes results in the opening of a fusion pore (D) that expands allowing the mixing of cytoplasmic material and spread of viral components between cells (E). Iterations of the fusion process result in formation of large multinucleated syncytia.
2.4. Spread at cell junctions
Cell junctions are structures composed by several different transmembrane proteins, whose main function is to form a seal between polarized epithelial cells. Adhesion between epithelial cells is achieved by three main types of seals: tight junctions (TJ), adherens junctions (AJ) and desmosomes. These barriers cannot be penetrated by viruses unless there is substantial damage to the epithelia (Mateo et al., 2015). However, epithelial cells can be infected from the apical or basolateral sides, with viral particles then transmitted to adjacent cells through specialized sites located in the vicinity of cellular junctions that are at least partially inaccessible from the extracellular environment. For example, in addition to cell-free spread, hepatitis C virus (HCV) can spread from cell-to-cell in a neutralizing antibody-independent manner (Brimacombe et al., 2011; Timpe et al., 2008). Claudin 1 (CLDN1) and occludin (OCLN), components of tight junctions, are critical for cell-to-cell spread of HCV (Brimacombe et al., 2011; Timpe et al., 2008). In addition, the cellular receptor CD81, scavenger receptor BI (SR-BI), apolipoprotein E, and low density lipoprotein receptor (LDLR) were shown to have a role in the cell-to-cell, but not cell-free, spread mechanism of HCV (Brimacombe et al., 2011; Fan et al., 2017; Timpe et al., 2008). Direct transmission of assembled HCV particles was proposed to occur in cell-cell contacts across partially sealed cell junctions (Brimacombe et al., 2011).
Robust evidence shows the use of adherens junctions by herpesviruses to spread from cell-to-cell (Johnson and Baines, 2011; Johnson and Huber, 2002). Herpes simplex virus type 1 (HSV-1) particles have been shown to be preferentially sorted towards cell junctions rather than to the apical surface of polarized epithelial cells (Johnson et al., 2001). The transport of particles towards these sites is primarily directed by the glycoprotein complex gI/gE, with the cytosolic domain of gE having a critical role (Dingwell et al., 1994; Dingwell and Johnson, 1998; Farnsworth and Johnson, 2006). Indeed, mutant viruses lacking gI/gE grow poorly in monolayers of human fibroblasts, and form plaques of small size (Dingwell et al., 1994). This phenotype was corroborated in vivo, as the △gI/gE mutant viruses produced small, punctate lesions in the corneal epithelium of rabbits and mice (Dingwell et al., 1994). The cell-to-cell spread mediated by gI/gE was found to be independent of neutralizing antibodies, suggesting that it occurs at sites of cellular junctions that are not accessible to these molecules (Dingwell et al., 1994). Immunofluorescent staining showed that gI/gE colocalized with the adherens junction protein β-catenin, but not with ZO-1, a component of tight junctions (Dingwell and Johnson, 1998). In addition, nectin-1, a component of adherens junctions, has been reported as a receptor for the herpes simplex virus glycoprotein D (Sakisaka et al., 2001; Yoon and Spear, 2002; Zhang et al., 2011). In this context, one accepted model is that gI/gE complexes accumulate at trans-Golgi network (TGN) subdomains, from where nascent virions are sorted to basolateral cell junctions for cell-to-cell spread (Farnsworth and Johnson, 2006; Johnson and Baines, 2011). At these sites, release of particles and their subsequent entry into the adjacent cell occur almost simultaneously. However, the critical function of gI/gE complexes extends beyond the spread between epithelial cells, and their role, together with pUS9, in the anterograde transport (from cell body to axons) of HSV-1 particles has been extensively studied (Howard et al., 2014; Kratchmarov et al., 2013; McGraw and Friedman, 2009; Snyder et al., 2008). Remarkably, the complex gI/gE together with pUS9 promote anterograde transport of enveloped particles, nucleocapsids, and glycoproteins through kinesin-mediated vesicular transport using microtubules (Diwaker et al., 2020; Johnson and Baines, 2011; Kratchmarov et al., 2013). The role of the gI/gE complex and US9 for cell-to-cell spread has additionally been reported for other herpesviruses including pseudorabies and VZV (Cohen and Nguyen, 1997; Diwaker et al., 2020; Johnson and Huber, 2002; Lyman et al., 2007; Zsak et al., 1992).
The use of desmosomes for cell-to-cell spread has been studied less than spread at the other cell junctions described above. However, some reports describe an important role of keratin 1 in infection by lymphocytic choriomeningitis virus (LCMV) (Labudova et al., 2009) (Labudova et al., 2019). Keratin filaments are an important component of desmosomes, and the LCMV nucleoprotein was shown to bind and stabilize keratin 1 (Labudova et al., 2009). This interaction resulted in increased desmosome formation and cell-cell adhesion capacity, facilitating cell-to-cell spread of LCMV (Labudova et al., 2009).
2.5. Virological synapses
Formation of virological synapses (VS) is a mechanism of cell-to-cell spread that has been shown to be used by retroviruses (Alvarez et al., 2014; Dupont and Sattentau, 2020; Jolly and Sattentau, 2004). In the VS, several cellular factors, including cytoskeletal proteins, receptors, and adhesion molecules, are recruited to sites where virus particles can be transmitted quickly from an infected cell to a target cell. In lymphocytes infected with human T-cell leukemia virus type 1 (HTLV-1), Env is distributed evenly at the cell surface and clusters of Gag are observed close to the plasma membrane (Igakura et al., 2003). Upon contact with uninfected T-cells, a rapid recruitment of a large number of Env, Gag and viral RNA molecules at cell-cell junctions is observed. Subsequently, transfer of Gag molecules and viral RNA is detected from the infected T cell to the uninfected T cell, likely utilizing a mechanism in which viral particles are rapidly released by the infected cell and taken up by the target cell in the junction region (Igakura et al., 2003). The cellular adhesion molecule talin was frequently found at sites of cell-cell contact, and an important reorientation of the microtubule organizing center (MTOC) to cell-cell contact adjacent sites was shown to be critical for transport of Gag molecules. The polarization of the MTOC towards sites of cell-cell contact by HTLV-1 is triggered by the transcriptional transactivator viral protein Tax (Nejmeddine et al., 2005). Polarization of the MTOC/Tax was shown to depend on intact microtubules, actin filaments, and the function of Rac and Cdc42 small GTPases (Nejmeddine et al., 2005). Further evidence indicates that the engagement of the adhesion molecule ICAM-1 on the surface of HTLV-1 infected cells is sufficient to trigger the intracellular polarization of the MTOC (Barnard et al., 2005). Analogous observations are seen in cells infected with HIV-1. Dendritic cells (DC) infected with HIV-1 rapidly recruit virus to cell junctions formed between DCs and T lymphocytes (McDonald et al., 2003). Remarkably, the un-infected T cells recruit receptor and co-receptor molecules including CD4, CCR5 and CXCR4 at the VS, which facilitates HIV transmission from cell-to-cell (McDonald et al., 2003). Similar to HTLV-1, in HIV-1-infected T cells (effector cells) Env and Gag are quickly recruited to the VS (Do et al., 2014; Jolly et al., 2004). Polarization of the MTOC and associated organelles towards the VS is induced in HIV-infected lymphocytes and DCs to hijack components of the secretory pathway (Bayliss et al., 2020; Jolly et al., 2011). In adjacent uninfected T CD4 + cells, an actin-dependent recruitment of CD4, CXCR4 and lymphocyte function-associated antigen 1 (LFA-1) was found to facilitate spread of Gag molecules between cells (Jolly et al., 2004). Quantitative 3D live microscopy revealed “buttons” of oligomerized HIV-1 Gag at sites of cell-cell contact, which were shown to be the preferential route of infection between CD4-T cells (Hubner et al., 2009). Thus, the proposed model of cell-to-cell infection involves budding of viral particles into the synaptic cleft and subsequent fusion of viral particles with the adjacent uninfected cell (Hubner et al., 2009; Miyauchi et al., 2009). Remarkably, it has been shown that HIV cell-to-cell spread can occur simultaneously from one infected lymphocyte to multiple adjacent recipient cells, a process named polysynapse formation (Rudnicka et al., 2009). Moreover, cell-to-cell transmission of retroviruses through the VS has been shown to play a critical role in viral dissemination in vivo (Murooka et al., 2012; Sewald et al., 2015). In this context, studies that examined the accessibility of the VS to entry inhibitors have concluded that synapse-mediated spread is less sensitive than cell-free particle spread to neutralizing antibodies in vitro and vivo (Abela et al., 2012; Gombos et al., 2015; Smith and Derdeyn, 2017; Zhong et al., 2013). Cell-to-cell contact sites have additionally been described as the main route of viral dissemination for the murine leukemia virus (MLV) by using live-cell imaging (Jin et al., 2009). Interestingly, a mechanism that resembles the VS was reported for spread of the non-lymphotropic HSV-1 (Aubert et al., 2009). Infection of CD4 + and CD8 + T lymphocytes by HSV-1 was shown to occur in vivo. Further, infection was more efficient by using cell-to-cell contacts than by cell-free virus, in a manner independent of the gI/gE glycoprotein complex (Aubert et al., 2009). Whether or not other non-lymphotropic viruses can establish VSs warrants future research.
2.6. Viral biofilms
HTLV-1 cell-to-cell transmission does not only rely on the formation of VSs. Lymphocytes infected with HTLV-1 have also been shown to store viral particles in an unconventional way, as clusters located on the surface of T CD4 + lymphocytes (Thoulouze and Alcover, 2010). These clusters correspond to viral assembly sites and are enriched in carbohydrates and extracellular matrix components including collagen and Agrin, a heparan sulfate proteoglycan (Pais-Correia et al., 2010; Thoulouze and Alcover, 2010). High-level expression of the host cell actin-bundling protein fascin is induced by HTLV-1 Tax in infected T-cells (Kress et al., 2011). Fascin contributes to cell-to-cell HTLV transmission, and has been suggested to be involved in transport and fitting gag into viral biofilms (Gross et al., 2016). Remarkably, the clusters are transferred from infected cells to recipient cells during cell contact, and it was estimated that 80% of the infectious capacity of HTLV-1 comes from these structures (Pais-Correia et al., 2010; Thoulouze and Alcover, 2010). The mechanism shares several similarities with bacterial biofilms, and both improved spread and escape from the immune response might be advantages of this mechanism of viral cell-to-cell spread.
2.7. Tunneling nanotubes
Tunneling nanotubes (TNTs) were first described in 2004 as a new mechanism of cell-to-cell communication (Rustom et al., 2004). They are thin, elongated membrane-bound structures, between 50 and 200 nm in diameter, that connect two distant cells and allow the transfer of material including organelles, ions, proteins and miRNAs. A very recent complete description of nanotube properties and their role in viral spread has been reported by Jansens et al. (Jansens et al., 2020). Based on their diameter and composition, two classes of TNTs are reported in macrophages: “thin TNTs” that contain filamentous actin (F-actin) and “thick TNTs” (greater than 0.7 μm) that contain F-actin and microtubules (Onfelt et al., 2006). Organelles such as mitochondria, lysosomes and peroxisomes were detected in thick, but not in thin, TNTs. Interestingly, bacteria could “surf” along the surface of thin nanotubes using a constitutive flow (Onfelt et al., 2006). Viruses can also exploit nanotubes for cell-to-cell spread, and this was first demonstrated for HIV (Sowinski et al., 2008). Tunneling nanotubes containing F-actin were shown to connect distant T cells, to have a close-ended nature and not to be tethered to the substratum, in contrast to what is seen for filopodia. Rapid spread of HIV particles through TNTs was found to be receptor-dependent (Sowinski et al., 2008). The induction of TNTs by HIV was additionally demonstrated in macrophages (Eugenin et al., 2009). Interestingly, the authors identified three different processes in uninfected and HIV-infected macrophages: long TNTs, short TNTs and filopodia, based on intercellular communication and length. The authors reporter that TNTs, but not filopodia, are open-ended and longer than filopodia, with lengths up to 150 μm (Eugenin et al., 2009). Another retrovirus, HTLV-1, was shown to form conduits in T cells that share characteristics of TNTs (Van Prooyen et al., 2010). Induction of these conduits was shown to depend on the viral p8 protein and to be facilitated by the cellular factors ICAM-1 and LFA-1 (Van Prooyen et al., 2010). Further, the induction of TNTs in infected cells was recently reported for influenza. Influenza virus nucleocapsids were observed in long intercellular structures, likely TNTs formed between lung epithelial cells (Kumar et al., 2017; Roberts et al., 2015). Direct transfer of influenza virus genomes and proteins was shown to require F-actin and actin dynamics. The process was also shown to be unaffected by the presence of neutralizing antibodies or oseltamivir, suggesting an open-ended nature of these TNTs (Kumar et al., 2017). Similar observations were made in cells infected with porcine reproductive and respiratory syndrome virus (PRRSV) (Guo et al., 2016). Infection by PRRSV induces the formation of TNTs that contain F-actin and myosin II-A, a motor-binding protein. Viral proteins and RNA were observed through the length of TNTs, and it was shown that viral proteins might interact directly or indirectly with myosin II-A. The presence of neutralizing antibodies does not prevent cell-to-cell transmission of a fluorescently labeled PRRSV and spread could occur in absence of cellular receptors, suggesting either a direct cytosolic connection between cells through TNTs or transfer through sites of close-contact provided by the TNTs that are inaccessible to antibodies (Guo et al., 2016). Exploitation of TNTs was additionally reported for pseudorabies virus (Jansens et al., 2017). Formation of TNTs capable of intercellular spread of molecules was achieved by the expression of the viral protein US3 in the absence of other viral proteins (Favoreel et al., 2005; Jansens et al., 2017). The TNTs were associated with enhanced viral intercellular spread and were very stable. This stability was suggested to be the result of the presence of stabilized microtubules and an enrichment of the adherens junctions components β-catenin and E-cadherin at the site of contact between the TNT and the target cell. In this scenario, enveloped virions were shown to be transported by vesicles through the TNT and released by exocytosis from this structure at sites of contact between the TNT and the acceptor cells (Favoreel et al., 2005; Jansens et al., 2017). Recently, the induction of long intercellular connections has been reported upon infection by DNA viruses, including VACV and the bovine herpesvirus 1 (BoHV-1) (Panasiuk et al., 2018; Xiao et al., 2017). The lack of adherence to substratum, their open-ended nature, length of up to 110 μm, and presence of F-actin and tubulin led to the proposal that the BoHV-1-induced structures correspond to TNTs. Viral proteins were detected along the length of the TNTs, and direct transfer of recombinant BoHV-1 in a neutralizing antibody independent manner was observed, similar to what was reported for RNA viruses (Panasiuk et al., 2018).
2.8. Filopodia
Filopodia are cellular structures involved in a wide array of functions including cell migration, wound healing, adhesion to the extracellular matrix and cell-cell interactions (Faix and Rottner, 2006; Mattila and Lappalainen, 2008). Their formation is induced through actin polymerization underneath the plasma membrane, which generates finger-like extensions that range between 0.1 and 0.3 μm in diameter. Filopodia are filled with parallel bundles of F-actin (Mattila and Lappalainen, 2008). Several signaling pathways drive the protrusion of filopodia, including members of the family of Rho GTPases like Cdc42 (Faix and Rottner, 2006; Mattila and Lappalainen, 2008; Nobes and Hall, 1995). Because of the heterogeneous nature of the previously described TNTs, it can be challenging to differentiate between TNTs and filopodia. Indeed, it was proposed that TNTs arise from filopodium-like protrusions that connect to neighboring cells (Rustom et al., 2004). A more recent report suggests that two filopodial connections can interact with each other through N-cadherin, forming a bridge (Chang et al., 2018). Rotation of the actin filaments inside these bridges by myosin proteins separates both filopodia, with one of them reaching a cell body and establishing a stable TNT-like connection via N-cadherin/β-catenin clustering (Chang et al., 2018). Moreover, it has been shown that the Cdc42/IRSp53/VASP network that is upregulated during filopodia elongation acts as a negative regulator of TNT formation and vesicle transfer function (Delage et al., 2016).
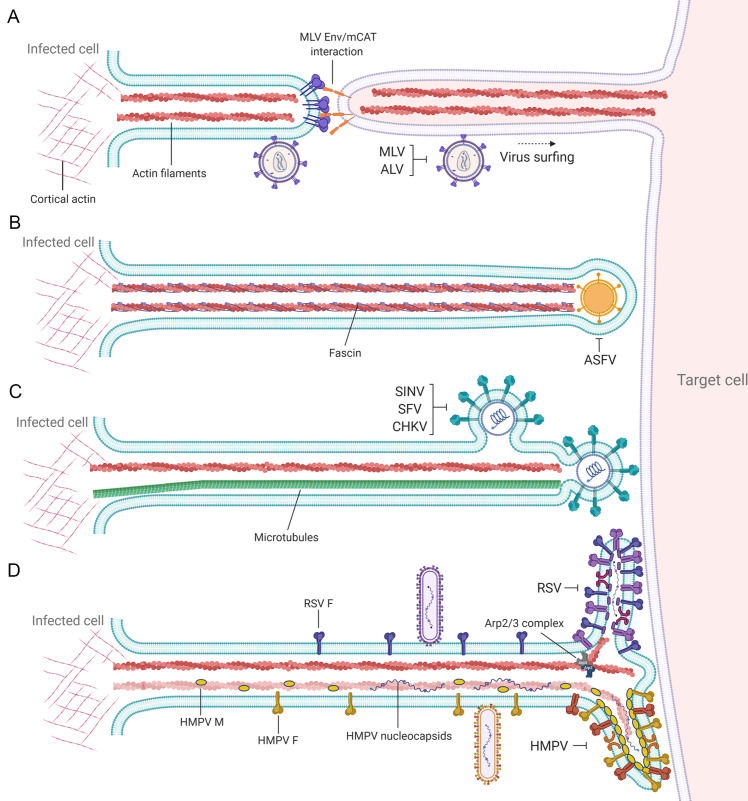
An interesting case of exploitation of filopodia during viral infection is reported for murine leukemia virus. After binding to receptors, MLV particles move along filopodia to reach the cell body and permit cell entry, a process dependent on actin and myosin that has been termed virus surfing (Fig. 3A) (Lehmann et al., 2005). Later in infection, it was shown that MLV particles move directly from infected to uninfected cells using filopodial bridges, named viral cytonemes, of about 5.8 μm in length (Sherer et al., 2007). Movement of fluorescently labeled MLV particles was observed on the outer surface of cytonemes. The formation of cytonemes was shown to be dependent on Env-receptor interactions, and antibodies targeting the extracellular domain of Env could disrupt viral cytonemes. Viral surfing through retrograde transport on the outer surface of filopodial structures has been also reported for DNA viruses including HSV-1 and human papillomavirus types 16 and 31 (Dixit et al., 2008; Schelhaas et al., 2008; Smith et al., 2008).
Fig. 3.
Different strategies of cell-to-cell spread by filopodia. Filopodial bridges have been described as a mechanism for direct cell-to-cell transmission by different viruses. (A) Cells infected with some retroviruses including MLV and avian leukemia virus (ALV) can establish filopodial bridges with uninfected cells, with viral particles surfing along the surface of filopodia toward the cell body to initiate infection. Single as farvirus particles have been observed projected at the tip of filopodial structures (B), contrasting with alphaviruses (C), where multiple particles budding from filopodial extensions facilitate direct cell-to-cell spread. (D) Filamentous pneumovirus particles, including respiratory syncytial virus (RSV) and human metapneumovirus (HMPV), bud from filopodia reaching the surface of adjacent cells for direct cell-to-cell spread. Viral proteins and genome have been detected in filopodia induced by pneumoviruses.
Further evidence of animal virus infection inducing filopodia formation has been reported for asfarviruses (Jouvenet et al., 2006). It was shown that African swine fever virus (ASFV) induces filopodia formation at the plasma membrane with an average length of 10.5 μm. Each projection contains the cellular proteins actin and fascin and single viral particles at their tips (Fig. 3B) (Jouvenet et al., 2006). Similar observations were made for alphaviruses, as infection induces a dramatic remodeling of the cell architecture including formation of long intercellular projections (˃10 μm) (Martinez et al., 2014; Martinez and Kielian, 2016). As observed for other viruses, projections induced by alphaviruses contain actin and microtubules. However, they are different from those seen with ASFV, where single virions are present at the tip of filopodia, as multiple alphavirus virions budding from the projections are observed (Fig. 3C). An interesting observation made for alphaviruses is that the filopodia-like extensions are not able to transfer cytosolic or plasma membrane components, suggesting they are not open-ended connections like TNTs. Instead, viral particles are hypothesized to bud into a protected space at the filopodial tip and then rapidly enter the target cell, preventing access of neutralizing antibodies. Recent work has demonstrated that filopodia-like extensions are also a mechanism that respiratory viruses use for direct cell-to-cell spread. Infection of human bronchial epithelial cells by human metapneumovirus (HMPV) resulted in a dramatic reorganization of the cell cytoskeleton (El Najjar et al., 2016). Two major forms of projections were observed: long intercellular extensions of up to 18 μm, which resemble filopodia, and shorter abundant branching filaments (up to 6 μm) budding from the cell body and from the intercellular extensions (Fig. 3D). We speculate that the branching filaments correspond to filamentous forms of budding virus. As was seen for other viruses, F-actin and the Rho GTPases Cdc42 and Rac1 were important for the formation of HMPV intercellular extensions. Cdc42 is one of the main small GTPases involved in the formation of filopodia (Nobes and Hall, 1995). Up to 50% of cell-to-cell spread facilitated by HMPV intercellular extensions was shown to occur in the presence of neutralizing antibodies and in the absence of appropriate attachment factors, leading to the suggestion that direct intercellular transfer of viral components could occur. Like HMPV, RSV induces formation of filopodial structures in lung epithelial cells which facilitate viral spread (Mehedi et al., 2016). Clusters of filamentous structures were observed at the surface of RSV-induced filopodia, similar to branching filaments induced by HMPV (Fig. 3D). Additionally, formation of filopodia was shown to confer increased motility to infected cells, thus facilitating cell-to-cell spread. Filopodia induced by RSV were shown to be dependent on the RSV F protein and the cellular actin-related protein 2 (Arp2), an important actin-nucleation factor (Mehedi et al., 2016). Recently, a significant induction of filopodia containing budding particles was reported for the severe acute respiratory syndrome virus 2 (SARS-CoV-2) (Bouhaddou et al., 2020). Filopodia induction was associated with an increase in signaling by casein kinase 2. Indeed, CK2 was found in filopodia co-localizing with the SARS-CoV-2 nucleoprotein (Bouhaddou et al., 2020).
2.9. Membrane pores
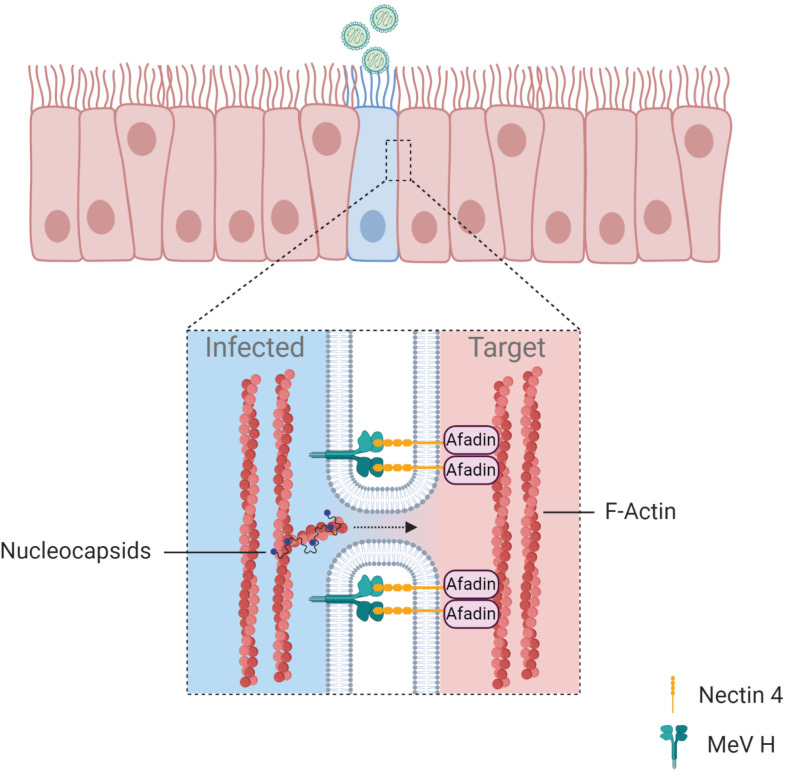
Analysis of measles virus (MeV) infection in well-differentiated primary cultures of human airway epithelial (HAE) cells revealed a previously unidentified mechanism of direct cell-to-cell spread that involves the opening of intercellular pores (Singh et al., 2015). A recombinant GFP-MeV rapidly spreads in HAE cell cultures to form infectious centers, but no syncytia were observed. These infectious centers are larger than those formed by other viruses such as RSV, Sendai or PIV5. Cytoplasmic material from the infected cell was shown to suddenly flow into the adjacent cell through lateral pores of an estimated size of 250 nm (Singh et al., 2015). The formation of infectious centers by MeV was dependent on an interaction between the actin filament binding protein afadin and nectin-4, the cellular receptor for MeV entry (Fig. 4 ). Recent work from the same group demonstrated that MeV ribonucleoprotein complexes can spread to neighbor human airway epithelial cells by moving along the circumapical F-actin ring (Fig. 4) (Singh et al., 2019). The circumapical F-actin ring encircles the uppermost portion of cells in HAE cultures, close to and at the level of adherens junctions. Further studies demonstrated that MeV uses cell-to-cell spread promoted by cell-cell contacts to move from infected immune cells to epithelial cells, rather than spread by cell-free virus (Singh et al., 2016). Very recently, another mechanism of cell-to-cell spread exploited by MeV has been reported. This mechanism involves the capture of membranes containing nectin-4 by cells expressing nectin-1, a process known as trans-endocytosis (Generous et al., 2019). Cargo cytosolic material, including MeV ribonucleocapsids, was shown to be transferred by trans-endocytosis and to remain functional in the recipient cell. This mechanism of spread was shown to occur between epithelial cells but also from infected epithelial cells to primary neurons. This type of spread does not require the assembly of complete virions, and demonstrates that transfer of ribonucleocapsids is sufficient to initiate infection in a target cell (Generous et al., 2019).
Fig. 4.
Formation of intercellular membrane pores. Infection of polarized epithelial cells by measles virus results in the opening of membrane pores between adjacent cells. Intercellular pores are stabilized by interactions between the viral glycoprotein H and the cellular receptor Nectin 4, which is anchored to the cell cytoskeleton through the Afadin protein. It has been suggested that physical constraints due to the presence of the circumapical F-actin ring thwart expansion of the intercellular pores, in contrast to what is observed during syncytia formation. However, the pore has a size that permits direct spread of ribonucleoprotein complexes that can initiate infection in the adjacent cell. Blue dots represent polymerase complexes bound to viral RNA.
2.10. Extracellular vesicles
A novel mechanism for transfer of multiple virus particles has been recently reported for enteroviruses. This virus family has historically been described as non-enveloped viruses that exit the cell by lysis. However, Coxsackie virus B3 (CVB3) particles were shown to alternatively exit the cell enclosed within vesicles (Robinson et al., 2014). These extracellular microvesicles (EMVs) were shown to contain autophagosomal membranes and to be infectious once added to recipient cells (Robinson et al., 2014). Further evidence supporting extracellular vesicles as a mechanism for viral spread came from the demonstration of non-lytic release of poliovirus (PV) capsids (Bird et al., 2014; Chen et al., 2015). Large extracellular vesicles enriched with phosphatidyl serine (PS) of up to 500 nm in size were shown to contain multiple mature PV particles. The same observations were made for other enteroviruses including CVB3 and rhinovirus (Chen et al., 2015). The question of how these large PV-containing vesicles can enter a target cell was addressed, and the process was shown to be dependent on both the cellular receptor CD155 and the presence of PS on the membranes of vesicles. Interestingly, it was shown that the PV particles contained within vesicles infect more efficiently than cell-free viral particles on a per-particle basis, potentially due to the synergistic effect of multiple particles entering the cell at the same time (Chen et al., 2015). Non-lytic release of vesicles containing rotavirus from cultured cells was recently reported (Santiana et al., 2018). The biological relevance of this mechanism was strengthened by the observation of vesicles containing clusters of rotavirus in animal stool samples. On average, more than 15 rotavirus active particles were shown to be present in each vesicle, and these virus-containing vesicles were shed into stool. Similar to what was found for PV-containing vesicles, rotavirus within vesicles was found to be more efficient in infection than cell-free viral particles (Santiana et al., 2018). Norovirus particles were also shown to be released non-lytically within vesicles, but these vesicles were slightly smaller in size and had cellular markers of exosomes (Santiana et al., 2018). The first demonstrations of viral particles released in exosomal membranes came from studies with HCV and the non-enveloped hepatitis A (HAV) virus (Feng et al., 2013; Ramakrishnaiah et al., 2013). Exosomes containing viral particles were shown to transmit HCV infection to recipient cells, a mechanism partly resistant to neutralizing antibodies (Ramakrishnaiah et al., 2013). Similarly, enveloped vesicles with markers of exosomes were found to contain multiple HAV particles and to mediate cell-to-cell transmission within the liver (Feng et al., 2013). These quasi-enveloped HAV particles (eHAV), are resistant to the action of neutralizing antibodies and can enter target cells through clathrin and dynamin-dependent endocytosis (Feng et al., 2013; Rivera-Serrano et al., 2019). Endocytosed eHAV are recognized by endolysosomal gangliosides that act as receptors, and the eHAV is then trafficked to lysosomes, where the exosomal membrane is degraded and virus uncoated (Das et al., 2020; Rivera-Serrano et al., 2019). These novel mechanisms of spread represent an advantage for the virus, as neutralizing antibodies should not affect infectivity of vesicles the same way they neutralize cell-free particles since the particles are protected by the vesicular membrane. This was demonstrated for the human JC polyomavirus that also associates with extracellular vesicles (Morris-Love et al., 2019). Furthermore, vesicles containing JC polyomavirus were shown to enter target cells in a manner that is independent of cellular receptors (Morris-Love et al., 2019). Thus, production of extracellular vesicles containing infectious virus seems to be a mechanism shared by a number of RNA and DNA viruses, whether they are enveloped or not (Altan-Bonnet et al., 2019).
3. Advantages of direct cell-to-cell spread
While budding and release of cell-free virus particles enables dissemination from host-to-host and spread across long distances to infect distant tissues within a host, direct cell-to-cell spread and spread at specialized cell-cell contact sites have several potential advantages for viral spread and infection, and thus has implications for viral disease and pathogenesis.
3.1. Rapid spread
Transmission of virus particles at cell-cell contact sites allows rapid viral dissemination within a tissue. In cell-to-cell spread, virus particles or virus genetic material are delivered right to target cells, making infection of target cells faster than with extracellular release and re-infection. Cell-to-cell spread also overcomes the rate limiting fluid-phase virus diffusion step encountered by released particles. Fitting with this, several studies have shown that the rate of HIV-1 spread through cell-to-cell transmission is faster than spread with cell-free virus (Boulle et al., 2016; Chen et al., 2007; Dimitrov et al., 1993; Sourisseau et al., 2007; Spouge et al., 1996). Transfer of virus particles across cell-cell contacts can also facilitate receptor binding and entry. Cell-cell contacts can specifically recruit receptors to the appropriate site, making the receptor attachment and entry steps more efficient (Chen et al., 2007; Sherer et al., 2007; Vasiliver-Shamis et al., 2009). In some cases, like with measles virus, complete assembly of virus particles does not occur and direct spread is mediated by transfer of virus genetic material in the form of ribonucleoprotein (RNP) complexes to target cells (Lawrence et al., 2000; Singh et al., 2015, Singh et al., 2019). Intercellular spread is mediated by cytoskeletal forces and protein trafficking pathways, thus allowing faster transmission than that seen with particle diffusion. The actin comet tails induced by vaccinia virus act to propel virus particles quickly across long distances, allowing for faster spread (Cudmore et al., 1995; Doceul et al., 2010). Comparative analysis revealed that cell-to-cell spread can be significantly more efficient than cell-free spread for a number of viruses, as determined by higher infection rates that occur under conditions of cell-to-cell compared to lower infection by cell-free virus infection. HIV cell-to-cell transmission is 10-18,000 × more efficient than extracellular spread (Chen et al., 2007; Kolodkin-Gal et al., 2013; Martin et al., 2010; Sourisseau et al., 2007). Cell-to-cell spread of HSV and PRV through intercellular extensions was shown to be essential for efficient spread in vitro (Favoreel et al., 2005).
3.2. Multiple genome transfer
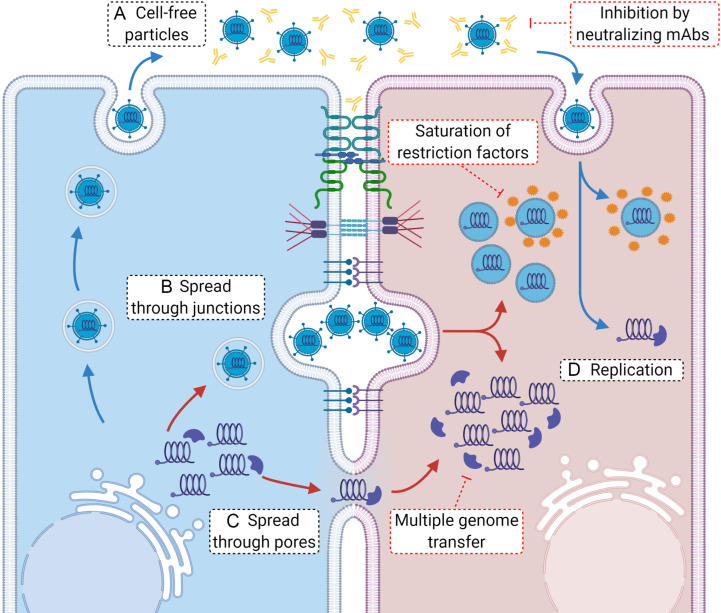
Cell-to-cell transmission was initially identified for viruses that have low infectivity to particle ratio but show efficient transmission in cell culture (Bangham, 2003; Carr et al., 1999; Dimitrov et al., 1993). Several studies have shown that spread of viruses at cell-cell contacts is associated with transfer of many virus particles or genomes from the donor to the target cell (Del Portillo et al., 2011; Hubner et al., 2009; Jin et al., 2009; Jung et al., 2002; Rager et al., 2002; Russell et al., 2013; Sherer et al., 2007; Shirogane et al., 2012; Sigal et al., 2011; Zhong et al., 2013). Enhanced budding at cell-cell contact sites and transfer of multiple genomes provide high local multiplicity of infection (MOI) (Fig. 5 ). This high MOI makes cell-to-cell spread more efficient than cell-free transmission by increasing the probability of successful infection through the introduction of multiple genomes into a single target cell, thus allowing spread even with low viral gene expression and poor particle release. It has been suggested that the transfer of multiple genomes during cell-to-cell spread has implications to virus fitness and evolution (Sanjuan, 2017). Other advantages of high MOI include trans-complementation of genetic defects, evasion of innate immune responses in the target cell, avoidance of the loss of viral components necessary for replication, alleviation of transmission bottlenecks and maintainance of genetic diversity (Sanjuan, 2017; Sanjuan and Thoulouze, 2019). Although many virus particles or genomes can be transferred, only a limited number of genomes likely initiate infection in the target cell (Miyashita et al., 2015). It has been estimated that as many as 102 or 103 HIV-1 particles can be transmitted through a virological synapse (Chen et al., 2007; Hubner et al., 2009); however, HIV-1 infects with an average of 3.62 proviruses after cell-to-cell spread (Del Portillo et al., 2011) which is relevant in vivo where 3–4 genomes are detected in infected cells (Jung et al., 2002; Law et al., 2016). The alpha-herpesviruses HSV and PRV infect with an average of 1.4 and 1.6 genomes, respectively, following cell-to-cell transmission although multiple virions were seen transported on distal axon sites (Taylor et al., 2012a). This suggests that although many virions or genomes can be transmitted at sites of cell-cell contacts, only a limited number establish infection which may be advantageous for the virus. Further studies are needed to determine if this is true for other viruses that use cell-to-cell spread for transmission.
Fig. 5.
Advantages of cell-to-cell spread. (A) Cell-free viral particles released by budding can be affected by the presence of neutralizing antibodies in the extracellular environment. In contrast, viral particles that are disseminated from cell-to-cell at cell junction-specialized membrane contacts (as one example of cell-to-cell spread) are not accessible to neutralizing antibodies (B). Once they enter a cell, individual viral particles can be targeted by intracellular restriction factors during uncoating. In contrast, multiple viral particles entering a target cell can saturate restriction factors available in the cell and proceed with infection. After successful uncoating, individual viral particles will initiate genome replication starting from single genomes (D), a highly inefficient process. In contrast, several viral genomes coming from particles will be more efficient in replication, leading to a rapid infection. Alternatively, genomes can be transmitted from cell-to-cell directly through intercellular pores (as an example), which can bypass the uncoating step and inhibition by restriction factors, proceeding more efficiently with replication (C).
3.3. Evasion of anti-viral and immune barriers
Spread of viruses by cell-to-cell transmission allows evasion of multiple barriers that exist for cell-free infection. One of the hallmarks of cell-to-cell spread is that it allows transmission in the presence of neutralizing antibodies (Fig. 5). Spread at tight cell-cell contacts provides limited exposure time to neutralizing antibodies in the extracellular environment and can physically restrict access of neutralizing antibodies to the virus. Neutralizing-antibody resistant spread has been shown for viruses that use different modes of cell-to-cell spread. For example, transmission of viruses such as HCV, HIV-1 and HSV-1 across tight cell-cell contacts has been shown to shield the virus from neutralizing antibodies (Brimacombe et al., 2011; Favoreel et al., 2006). Viruses that use nanotubes, filopodia, or intercellular extensions, whether open- or close-ended, show a neutralizing antibody- resistant mode of spread. This includes the alphaherpesviruses PRV and HSV-1 (Favoreel et al., 2005; Jansens et al., 2017; Sarfo et al., 2017), the alphavirus Sindbis virus (Martinez and Kielian, 2016), the respiratory viruses HMPV, influenza A virus, and PIV5 (El Najjar et al., 2016; Kumar et al., 2017; Roberts et al., 2015), PRRSV (Guo et al., 2016, Guo et al., 2018), and Coxsackievirus B3 (Paloheimo et al., 2011). Antibody resistant cell-to-cell spread of enveloped VACV particles occurs in an actin-dependent or -independent manner likely mediated by protein A33 (Krupovic et al., 2010; Law et al., 2002). The formation of viral biofilms by HTLV-1 may also provide a way of physically protecting particles from circulating antibodies which recognize the Env protein (Pais-Correia et al., 2010; Thoulouze and Alcover, 2010). For HIV-1, several reports show conflicting results on the role of neutralizing antibodies in inhibiting cell-to-cell transmission, with some studies showing decreased neutralization during cell-to-cell spread and others showing no inhibition of neutralizing antibodies on direct virus transfer. Variations in the effects of the neutralizing antibodies depend on the epitope on Env protein targeted by the antibody, the donor and target cell type, and virus strain used (Abela et al., 2012; Chen et al., 2007; Ganesh et al., 2004; Gupta et al., 1989; Martin et al., 2010; Massanella et al., 2009; Pantaleo et al., 1995; van Montfort et al., 2007; Zhong et al., 2013). In addition to evasion from neutralizing antibodies, cell-to-cell spread can play a role in overcoming intrinsic antiviral restriction factors. For retroviruses, restriction factors such as TRIM5α and tetherin can effectively inhibit cell-free virus spread, but are less effective, or fail completely, in preventing cell-to-cell transmission (Celestino et al., 2012; Jolly et al., 2010; Richardson et al., 2008; Zhong et al., 2013). This may occur due to saturation of the restriction factors by accumulation of virus particles at sites of cell-cell contacts or because cell-associated virus is inaccessible to a restriction factor. It has been suggested that tetherin can promote accumulation of HIV-1 virus particles at the VS and even help to stabilize the synapse during cell-to-cell spread (Jolly et al., 2010). Cell-to-cell transmission may also act on overcoming the innate antiviral response. It has been suggested that transfer of influenza A virus NS protein through tunneling nanotubes may overcome the innate immune response in the target cell (Kumar et al., 2017).
3.4. Role in pathogenesis
Cell-to-cell spread has implications for the pathogenesis of viral infection as it contributes to viral persistence and latency, therapy failure/resistance, and immune evasion. The cell-to-cell mode of transmission is inherent to the pathogenesis of HSV-1; it is the main mode of spread in the epithelium and from epithelial cells to neurons during primary infection where it establishes latency and moves back through axons to epithelial cells during reactivation (Dingwell et al., 1994; Wang et al., 2010). Cell-to-cell transmission has also been implicated in persistence of HCV infection and establishment of chronic infection in the liver, in addition to resistance to direct-acting antiviral drugs and therapy failure (Timpe et al., 2008; Witteveldt et al., 2009; Xiao et al., 2014; Zeisel et al., 2013). Resistance to antivirals due to direct cell-to-cell spread has also been shown for influenza virus with resistance to oseltamivir treatment (Kumar et al., 2017; Mori et al., 2011; Roberts et al., 2015). The different modes of cell-to-cell transmission that HIV-1 utilizes contribute to the persistence of viral reservoirs (Costiniuk and Jenabian, 2014) and resistance to antiretroviral therapy (Duncan et al., 2013; Sigal et al., 2011). Understanding mechanisms of cell-to-cell spread is thus important for development of a deeper understanding of viral pathogenesis and for the development of potent antiviral therapies.
4. Experimental approaches for studying cell-to-cell spread
4.1. Cell culture experiments
Multiple approaches have been used in in-vitro cell culture models to study the different modes of virus transmission. Experimental conditions were developed for blocking cell-free or cell-to-cell spread to determine the contribution of either in infection dynamics. Addition of a viscous media such as methylcellulose or agarose to an infected cell culture suppresses diffusion of released virus particles and thus decreases spread by cell-free virus and not by direct cell-to-cell spread (El Najjar et al., 2016; Jin et al., 2009; Martin et al., 2010; Timpe et al., 2008). Another approach for blocking cell-free virus infection that provides more convincing experimental support is assaying spread in the presence of virus-neutralizing antibodies. While neutralizing antibodies are successful in completely, or almost completely, inhibiting cell-free infection, cell-to-cell transmission is generally resistant to neutralizing antibodies. Spread in the presence of neutralizing antibodies has been used to demonstrate cell-to-cell transmission for several viruses including HCV-1, herpesviruses, alphaviruses, coxsackievirus B3, IAV, HMPV and PRRSV (Brimacombe et al., 2011; Favoreel et al., 2005, Favoreel et al., 2006; Guo et al., 2016; Jansens et al., 2017; Martinez and Kielian, 2016; Roberts et al., 2015; Sarfo et al., 2017). In some cases however, such as HIV-1 and CMV, the effect of neutralizing antibodies can be inconsistent, as discussed above, and further validation of transmission modes requires additional investigation (Dufloo et al., 2018; Jacob et al., 2013). In addition, some neutralizing antibodies can prevent cell-to-cell transmission by inhibiting establishment or stability of cell-cell contacts between an infected donor cell and an uninfected target cell. For instance, cell-to-cell transmission of HIV-1 depends on cell-cell contacts that form by the interaction of Env protein in the donor cell with the CD4 receptor on the target cell; thus neutralizing antibodies against Env protein can block cell-cell contact and cell-to-cell spread (Chen et al., 2007; Jolly et al., 2004; Rudnicka et al., 2009). It is therefore important to use neutralizing antibodies that do not interfere with the formation of cell-cell contacts in order to differentiate the susceptibility of cell-free and cell-to-cell transmission to neutralization. Experimental assays to assess the role of direct contact between infected cells and neighboring cells in spreading infection include using a trans-well to physically separate donor and target cells and prevent cell migration, cell shaking to abolish the formation of cell-cell contacts or lowering cell density to prevent close cell contact. Coculturing of donor infected cells with target cells is another widely used assay to identify cell-to-cell transmission. In this assay, infected target cells are harvested and cocultured with uninfected target cells under conditions that prevent infection by cell-free virus such as addition of neutralizing antibodies or blocking receptor binding (Branscome et al., 2019; El Najjar et al., 2016; Martinez and Kielian, 2016; Xiao et al., 2014; Zhong et al., 2013). A fluorescent dye is usually used to differentiate target cells from donor cells, and quantification of cell spread is determined microscopically or by flow cytometry of a reporter virus or a labeled viral antigen. The cell coculture system has been used to identify viral and host factors that contribute to direct cell spread of several viruses. Cell-to-cell transmission of HCV depends on viral envelope proteins and utilizes the same receptors used for cell-free infection including scavenger receptor BI, CD81, epidermal growth factor receptor (EGFR) and the tight junction proteins claudin-1 and occludin (Catanese et al., 2013; Fofana et al., 2013; Lupberger et al., 2011; Witteveldt et al., 2009; Zahid et al., 2013). Cell-to-cell spread for other viruses such as HMPV and SINV occurs in a manner that is independent, or partially independent, of the receptor binding which is needed for cell-free entry (El Najjar et al., 2016; Martinez and Kielian, 2016). For studying spread of herpesviruses, two cell culture systems are primarily used: a dissociated model where rat superior cervical ganglia (SCG) are plated and form a network of axons; and a compartmentalized system that provides isolation of the neuron cell body from the distal axon termini (Curanovic et al., 2009; Ch'ng et al., 2005). The latter system has been used to study the number and diversity of viral genomes after anterograde direct spread of HSV-1 and PRV particles, with results showing limited virus transmission from neurons to epithelial cells (Taylor et al., 2012a). Experiments conducted in 2-dimensional cell culture models provide valuable information on the mechanisms of cell-to-cell spread of viruses; however, they do not take into account how the complexity and environment of the tissue impacts virus spread in vivo. To overcome some of these limitations, different 3-dimensional cell culture models have been utilized for studying spread for a number of viruses. A 3-D cell culture system using HIV-1-infected primary human CD4 T lymphocytes in a 3-D scaffold showed the importance of cell motility and density to HIV-1 spread (Imle et al., 2019). Studying measles virus in a 3-D model of primary human differentiated airway epithelial cells revealed a novel mechanism of RNP horizontal spread along apical F-actin rings (Singh et al., 2019). A neutralizing antibody-resistant mode of spread was recently described for HMPV in a similar 3-D model of differentiated human bronchial epithelial cells (Kinder et al., 2020). Further studies in 3-D tissue models are necessary to elucidate how viruses actually spread within the host and identify antivirals that affect one mode of virus transmission but not the other.
4.2. Imaging techniques
Imaging fixed cells by fluorescence and electron microcopy has been widely used for showing virus transfer between cells. Detection of viral proteins in intercellular connections or at sites of cell-cell contact by immunofluorescence (IF) is usually added to the line of evidence to demonstrate cell-to-cell spread. A number of studies showed localization of viral proteins in TNTs, filopodia, and other intercellular projections for viruses of different families that spread by direct cell-to-cell transmission, including the herpesvirus BoHV-1, alphavirus SINV, PRRSV and HMPV. Localization of viral genomes within these cellular structures, as detected by fluorescence in situ hybridization (FISH), indicated that not only viral antigens are localizing to intercellular connections but also genetic material, thus suggesting passage of virus particles or viral genomes across these structures (El Najjar et al., 2016; Favoreel et al., 2005; Guo et al., 2016; Martinez and Kielian, 2016). For HIV-1, a VS was initially described by IF showing clustering of receptor on the target cell and Env protein on the effector cell (Jolly et al., 2004; McDonald et al., 2003), and transmission electron microscopy provided cross sectional views of the synapse (Wang et al., 2007). Advances in microscopic imaging facilitated by the generation of fluorescently labeled recombinant viruses helped to further deliver mechanistic insights underlying cell-to-cell spread of viruses. Live cell imaging provides a powerful tool for demonstrating the dynamics of virus transport between cells. Combining IF and time lapse microscopy with correlative fluorescence and scanning microscopy revealed dynamics of movement of single MLV particles on the surface of cytonemes that form by interaction of Env protein with the receptor mCAT1 (Jin et al., 2009; Sherer et al., 2007). Live cell 3-D microscopy and expansion microscopy of influenza virus infected cells, in addition to scanning electron microscopy (SEM), showed that the ends of TNT are continuous with the cytoplasm of cells and that there are bundled fibers inside the TNTs (Kumar et al., 2017). Generation of fluorescently labeled alphaherpesviruses with fluorophore tagged capsid, tegument, or glycoproteins allowed examination of egress and spread mechanisms. Live imaging of neurons infected with different labeled viruses indicated that both capsids and enveloped particles move across axons and that the US9 membrane protein is needed for anterograde transport of progeny virus particles (Taylor et al., 2012b; Wisner et al., 2011). Imaging alphaherpesviruses HSV-1 and PRV, tagged with different fluorophores, allowed determination of the diversity and number of viral genomes following anterograde-directed spread from isolated neurites to epithelial cells in a compartmentalized cell culture model (Taylor et al., 2012a). Imaging techniques have also advanced our understanding of measles virus spread. Time lapse microscopy with 3-D z-stack imaging of GFP-tagged measles virus, or more recently of a recombinant virus containing GFP/P fusion protein, in a model of airway epithelial tissue was used to reveal rapid spread. Infectious centers and RNP were shown along the apical F actin network stained with the LifeAct marker as mentioned in more detail in Section 2.9 (Singh et al., 2015, Singh et al., 2019). More information has been gathered in recent years on the formation of the VS mediating HIV-1 spread. Analysis using ion abrasion SEM and electron tomography provided 3-D visualization of the VSs with invaginations forming in the donor cell and a projection extending from the target cell (Felts et al., 2010). Kinetics of virus clustering and transfer were determined by quantitative 3-D live imaging (Hubner et al., 2009; Real et al., 2018). Recently, live imaging using a GFP-tagged Env mutant virus and cells stably expressing mCherry-Gag revealed that the Env protein is first transferred across the VS followed by redistribution of Gag to cell-cell contact sites (Wang et al., 2019). Super resolution microscopy revealed an organized localization of N and M proteins of HMPV in budding filaments with N protein on the inside surrounded by the M protein (El Najjar et al., 2016).
Combining functional and imaging approaches is thus essential for elucidating the mechanisms underlying cell-to-cell spread of different viruses. In addition, mathematical models have been employed in recent years to provide better quantitation of the contribution of the different modes of spread in infection dynamics. Such models account for the role of space and effects of the immune response on viral spread and utilize experimental and clinical data to assess modes of virus spread in vivo (Goyal and Murray, 2016; Graw et al., 2015; Graw and Perelson, 2016; Kumberger et al., 2018).
5. Concluding remarks
Research in the last several decades has highlighted both the variety of mechanisms that viruses utilize for spreading to new target cells and the surprising commonality of many mechanisms. Recent work suggests that these mechanisms may be important for pathogenesis, providing a more efficient means of propagating infection under many circumstances. Future work dissecting the role of cell-to-cell spread in animal models will be critical to understand the importance of these mechanisms in overall pathogenesis. In addition, dissection of these mechanisms may provide important new targets for antiviral treatment.
Acknowledgments
This work was supported in part by NIH grants AI051517 and AI140758 to R.E.D. and Fondecyt Inicio grant 11180269 to N.C.M. Images were created using BioRender.com.
References
- Abela I.A., Berlinger L., Schanz M., Reynell L., Gunthard H.F., Rusert P., Trkola A. Cell-cell transmission enables HIV-1 to evade inhibition by potent CD4bs directed antibodies. PLoS Pathog. 2012;8:e1002634. doi: 10.1371/journal.ppat.1002634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altan-Bonnet N., Perales C., Domingo E. Extracellular vesicles: vehicles of en bloc viral transmission. Virus Res. 2019;265:143–149. doi: 10.1016/j.virusres.2019.03.023. [DOI] [PubMed] [Google Scholar]
- Alvarez D.E., Agaisse H. Casein kinase 2 regulates vaccinia virus actin tail formation. Virology. 2012;423:143–151. doi: 10.1016/j.virol.2011.12.003. [DOI] [PubMed] [Google Scholar]
- Alvarez D.E., Agaisse H. The formin FHOD1 and the small GTPase Rac1 promote vaccinia virus actin-based motility. J. Cell Biol. 2013;202:1075–1090. doi: 10.1083/jcb.201303055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alvarez R.A., Barria M.I., Chen B.K. Unique features of HIV-1 spread through T cell virological synapses. PLoS Pathog. 2014;10:e1004513. doi: 10.1371/journal.ppat.1004513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ambrosini A.E., Enquist L.W. Cell-fusion events induced by α-herpesviruses. Futur. Virol. 2015;10:185–200. [Google Scholar]
- Aubert M., Yoon M., Sloan D.D., Spear P.G., Jerome K.R. The virological synapse facilitates herpes simplex virus entry into T cells. J. Virol. 2009;83:6171–6183. doi: 10.1128/JVI.02163-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bangham C.R.M. The immune control and cell-to-cell spread of human T-lymphotropic virus type 1. J. Gen. Virol. 2003;84:3177–3189. doi: 10.1099/vir.0.19334-0. [DOI] [PubMed] [Google Scholar]
- Barnard A.L., Igakura T., Tanaka Y., Taylor G.P., Bangham C.R. Engagement of specific T-cell surface molecules regulates cytoskeletal polarization in HTLV-1-infected lymphocytes. Blood. 2005;106:988–995. doi: 10.1182/blood-2004-07-2850. [DOI] [PubMed] [Google Scholar]
- Bayliss R., Wheeldon J., Caucheteux S.M., Niessen C.M., Piguet V. Identification of host trafficking genes required for HIV-1 virological synapse formation in dendritic cells. J. Virol. 2020;94:JVI.01597–19. doi: 10.1128/JVI.01597-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bird S.W., Maynard N.D., Covert M.W., Kirkegaard K. Nonlytic viral spread enhanced by autophagy components. Proc. Natl. Acad. Sci. U. S. A. 2014;111:13081–13086. doi: 10.1073/pnas.1401437111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bond V.C., Person S., Warner S.C. The isolation and characterization of mutants of herpes simplex virus type 1 that induce cell fusion. J. Gen. Virol. 1982;61(Pt. 2):245–254. doi: 10.1099/0022-1317-61-2-245. [DOI] [PubMed] [Google Scholar]
- Bouhaddou M., Memon D., Meyer B., White K.M., Rezelj V.V., Marrero M.C., Polacco B.J., Melnyk J.E., Ulferts S., Kaake R.M. The global phosphorylation landscape of SARS-CoV-2 infection. Cell. 2020;182:685–712. doi: 10.1016/j.cell.2020.06.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boulle M., Muller T.G., Dahling S., Ganga Y., Jackson L., Mahamed D., Oom L., Lustig G., Neher R.A., Sigal A. HIV cell-to-cell spread results in earlier onset of viral gene expression by multiple infections per cell. PLoS Pathog. 2016;12:e1005964. doi: 10.1371/journal.ppat.1005964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Branscome H., Paul S., Khatkar P., Kim Y., Barclay R.A., Pinto D.O., Yin D., Zhou W., Liotta L.A., El-Hage N. Stem cell extracellular vesicles and their potential to contribute to the repair of damaged CNS cells. J. Neuroimmune Pharmacol. 2019;15:520–537. doi: 10.1007/s11481-019-09865-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brimacombe C.L., Grove J., Meredith L.W., Hu K., Syder A.J., Flores M.V., Timpe J.M., Krieger S.E., Baumert T.F., Tellinghuisen T.L. Neutralizing antibody-resistant hepatitis C virus cell-to-cell transmission. J. Virol. 2011;85:596–605. doi: 10.1128/JVI.01592-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr J.M., Hocking H., Li P., Burrell C.J. Rapid and efficient cell-to-cell transmission of human immunodeficiency virus infection from monocyte-derived macrophages to peripheral blood lymphocytes. Virology. 1999;265:319–329. doi: 10.1006/viro.1999.0047. [DOI] [PubMed] [Google Scholar]
- Catanese M.T., Loureiro J., Jones C.T., Dorner M., von Hahn T., Rice C.M. Different requirements for scavenger receptor class B type I in hepatitis C virus cell-free versus cell-to-cell transmission. J. Virol. 2013;87:8282–8293. doi: 10.1128/JVI.01102-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Celestino M., Calistri A., Del Vecchio C., Salata C., Chiuppesi F., Pistello M., Borsetti A., Palu G., Parolin C. Feline tetherin is characterized by a short N-terminal region and is counteracted by the feline immunodeficiency virus envelope glycoprotein. J. Virol. 2012;86:6688–6700. doi: 10.1128/JVI.07037-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan K.M.C., Son S., Schmid E.M., Fletcher D.A. A viral fusogen hijacks the actin cytoskeleton to drive cell-cell fusion. Elife. 2020;9:e51358. doi: 10.7554/eLife.51358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang M., Oh J., Doh J., Lee J.-B. F-actin dynamics transform filopodial bridges into intercellular nanotubes capable of distant cell communication. BioRxiv. 2018:405340. [Google Scholar]
- Chen P., Hubner W., Spinelli M.A., Chen B.K. Predominant mode of human immunodeficiency virus transfer between T cells is mediated by sustained Env-dependent neutralization-resistant virological synapses. J. Virol. 2007;81:12582–12595. doi: 10.1128/JVI.00381-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y.H., Du W., Hagemeijer M.C., Takvorian P.M., Pau C., Cali A., Brantner C.A., Stempinski E.S., Connelly P.S., Ma H.C. Phosphatidylserine vesicles enable efficient en bloc transmission of enteroviruses. Cell. 2015;160:619–630. doi: 10.1016/j.cell.2015.01.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ch'ng T.H., Flood E.A., Enquist L.W. Culturing primary and transformed neuronal cells for studying pseudorabies virus infection. Methods Mol. Biol. 2005;292:299–316. doi: 10.1385/1-59259-848-x:299. [DOI] [PubMed] [Google Scholar]
- Cifuentes-Munoz N., Salazar-Quiroz N., Tischler N.D. Hantavirus Gn and Gc envelope glycoproteins: key structural units for virus cell entry and virus assembly. Viruses. 2014;6:1801–1822. doi: 10.3390/v6041801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen J.I., Nguyen H. Varicella-zoster virus glycoprotein I is essential for growth of virus in Vero cells. J. Virol. 1997;71:6913–6920. doi: 10.1128/jvi.71.9.6913-6920.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole N.L., Grose C. Membrane fusion mediated by herpesvirus glycoproteins: the paradigm of varicella-zoster virus. Rev. Med. Virol. 2003;13:207–222. doi: 10.1002/rmv.377. [DOI] [PubMed] [Google Scholar]
- Compton A.A., Schwartz O. They might be giants: does syncytium formation sink or spread HIV infection? PLoS Pathog. 2017;13:e1006099. doi: 10.1371/journal.ppat.1006099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costiniuk C.T., Jenabian M.A. Cell-to-cell transfer of HIV infection: implications for HIV viral persistence. J. Gen. Virol. 2014;95:2346–2355. doi: 10.1099/vir.0.069641-0. [DOI] [PubMed] [Google Scholar]
- Cudmore S., Cossart P., Griffiths G., Way M. Actin-based motility of vaccinia virus. Nature. 1995;378:636–638. doi: 10.1038/378636a0. [DOI] [PubMed] [Google Scholar]
- Curanovic D., Ch'ng T.H., Szpara M., Enquist L. Compartmented neuron cultures for directional infection by alpha herpesviruses. Curr. Protoc. Cell Biol. 2009;Chapter 26:unit 26.24. doi: 10.1002/0471143030.cb2604s43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das A., Barrientos R., Shiota T., Madigan V., Misumi I., McKnight K.L., Sun L., Li Z., Meganck R.M., Li Y. Gangliosides are essential endosomal receptors for quasi-enveloped and naked hepatitis A virus. Nat. Microbiol. 2020;5:1069–1078. doi: 10.1038/s41564-020-0727-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Del Portillo A., Tripodi J., Najfeld V., Wodarz D., Levy D.N., Chen B.K. Multiploid inheritance of HIV-1 during cell-to-cell infection. J. Virol. 2011;85:7169–7176. doi: 10.1128/JVI.00231-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delage E., Cervantes D.C., Penard E., Schmitt C., Syan S., Disanza A., Scita G., Zurzolo C. Differential identity of Filopodia and Tunneling Nanotubes revealed by the opposite functions of actin regulatory complexes. Sci. Rep. 2016;6:39632. doi: 10.1038/srep39632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dimitrov D.S., Willey R.L., Sato H., Chang L.J., Blumenthal R., Martin M.A. Quantitation of human immunodeficiency virus type 1 infection kinetics. J. Virol. 1993;67:2182–2190. doi: 10.1128/jvi.67.4.2182-2190.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dingwell K.S., Johnson D.C. The herpes simplex virus gE-gI complex facilitates cell-to-cell spread and binds to components of cell junctions. J. Virol. 1998;72:8933–8942. doi: 10.1128/jvi.72.11.8933-8942.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dingwell K.S., Brunetti C.R., Hendricks R.L., Tang Q., Tang M., Rainbow A.J., Johnson D.C. Herpes simplex virus glycoproteins E and I facilitate cell-to-cell spread in vivo and across junctions of cultured cells. J. Virol. 1994;68:834–845. doi: 10.1128/jvi.68.2.834-845.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diwaker D., Murray J.W., Barnes J., Wolkoff A.W., Wilson D.W. Deletion of the Pseudorabies Virus gE/gI-US9p complex disrupts kinesin KIF1A and KIF5C recruitment during egress, and alters the properties of microtubule-dependent transport in vitro. PLoS Pathog. 2020;16:e1008597. doi: 10.1371/journal.ppat.1008597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dixit R., Tiwari V., Shukla D. Herpes simplex virus type 1 induces filopodia in differentiated P19 neural cells to facilitate viral spread. Neurosci. Lett. 2008;440:113–118. doi: 10.1016/j.neulet.2008.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Do T., Murphy G., Earl L.A., Del Prete G.Q., Grandinetti G., Li G.H., Estes J.D., Rao P., Trubey C.M., Thomas J. Three-dimensional imaging of HIV-1 virological synapses reveals membrane architectures involved in virus transmission. J. Virol. 2014;88:10327–10339. doi: 10.1128/JVI.00788-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doceul V., Hollinshead M., van der Linden L., Smith G.L. Repulsion of superinfecting virions: a mechanism for rapid virus spread. Science. 2010;327:873–876. doi: 10.1126/science.1183173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donnelly S.K., Weisswange I., Zettl M., Way M. WIP provides an essential link between Nck and N-WASP during Arp2/3-dependent actin polymerization. Curr. Biol. 2013;23:999–1006. doi: 10.1016/j.cub.2013.04.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dufloo J., Bruel T., Schwartz O. HIV-1 cell-to-cell transmission and broadly neutralizing antibodies. Retrovirology. 2018;15:51. doi: 10.1186/s12977-018-0434-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan R. Fusogenic reoviruses and their fusion-associated small transmembrane (FAST) proteins. Annu. Rev. Virol. 2019;6:341–363. doi: 10.1146/annurev-virology-092818-015523. [DOI] [PubMed] [Google Scholar]
- Duncan R., Chen Z., Walsh S., Wu S. Avian reovirus-induced syncytium formation is independent of infectious progeny virus production and enhances the rate, but is not essential, for virus-induced cytopathology and virus egress. Virology. 1996;224:453–464. doi: 10.1006/viro.1996.0552. [DOI] [PubMed] [Google Scholar]
- Duncan C.J., Russell R.A., Sattentau Q.J. High multiplicity HIV-1 cell-to-cell transmission from macrophages to CD4 + T cells limits antiretroviral efficacy. AIDS. 2013;27:2201–2206. doi: 10.1097/QAD.0b013e3283632ec4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan M.L., Horsington J., Eldi P., Al Rumaih Z., Karupiah G., Newsome T.P. Loss of actin-based motility impairs ectromelia virus release in vitro but is not critical to spread in vivo. Viruses. 2018;10:111. doi: 10.3390/v10030111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dupont M., Sattentau Q.J. Macrophage cell-cell interactions promoting HIV-1 infection. Viruses. 2020;12:492. doi: 10.3390/v12050492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eitzen G. Actin remodeling to facilitate membrane fusion. Biochim. Biophys. Acta. 2003;1641:175–181. doi: 10.1016/s0167-4889(03)00087-9. [DOI] [PubMed] [Google Scholar]
- El Najjar F., Cifuentes-Munoz N., Chen J., Zhu H., Buchholz U.J., Moncman C.L., Dutch R.E. Human metapneumovirus induces reorganization of the actin cytoskeleton for direct cell-to-cell spread. PLoS Pathog. 2016;12:e1005922. doi: 10.1371/journal.ppat.1005922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eugenin E.A., Gaskill P.J., Berman J.W. Tunneling nanotubes (TNT) are induced by HIV-infection of macrophages: a potential mechanism for intercellular HIV trafficking. Cell. Immunol. 2009;254:142–148. doi: 10.1016/j.cellimm.2008.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faix J., Rottner K. The making of filopodia. Curr. Opin. Cell Biol. 2006;18:18–25. doi: 10.1016/j.ceb.2005.11.002. [DOI] [PubMed] [Google Scholar]
- Fan H., Qiao L., Kang K.D., Fan J., Wei W., Luo G. Attachment and postattachment receptors important for hepatitis C virus infection and cell-to-cell transmission. J. Virol. 2017;91:e00280–17. doi: 10.1128/JVI.00280-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farnsworth A., Johnson D.C. Herpes simplex virus gE/gI must accumulate in the trans-Golgi network at early times and then redistribute to cell junctions to promote cell-cell spread. J. Virol. 2006;80:3167–3179. doi: 10.1128/JVI.80.7.3167-3179.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Favoreel H.W., Van Minnebruggen G., Adriaensen D., Nauwynck H.J. Cytoskeletal rearrangements and cell extensions induced by the US3 kinase of an alphaherpesvirus are associated with enhanced spread. Proc. Natl. Acad. Sci. U. S. A. 2005;102:8990–8995. doi: 10.1073/pnas.0409099102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Favoreel H.W., Van Minnebruggen G., Van de Walle G.R., Ficinska J., Nauwynck H.J. Herpesvirus interference with virus-specific antibodies: bridging antibodies, internalizing antibodies, and hiding from antibodies. Vet. Microbiol. 2006;113:257–263. doi: 10.1016/j.vetmic.2005.11.003. [DOI] [PubMed] [Google Scholar]
- Felts R.L., Narayan K., Estes J.D., Shi D., Trubey C.M., Fu J., Hartnell L.M., Ruthel G.T., Schneider D.K., Nagashima K. 3D visualization of HIV transfer at the virological synapse between dendritic cells and T cells. Proc. Natl. Acad. Sci. U. S. A. 2010;107:13336–13341. doi: 10.1073/pnas.1003040107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng Z., Hensley L., McKnight K.L., Hu F., Madden V., Ping L., Jeong S.H., Walker C., Lanford R.E., Lemon S.M. A pathogenic picornavirus acquires an envelope by hijacking cellular membranes. Nature. 2013;496:367–371. doi: 10.1038/nature12029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fofana I., Xiao F., Thumann C., Turek M., Zona L., Tawar R.G., Grunert F., Thompson J., Zeisel M.B., Baumert T.F. A novel monoclonal anti-CD81 antibody produced by genetic immunization efficiently inhibits Hepatitis C virus cell-cell transmission. PLoS One. 2013;8:e64221. doi: 10.1371/journal.pone.0064221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frischknecht F., Cudmore S., Moreau V., Reckmann I., Rottger S., Way M. Tyrosine phosphorylation is required for actin-based motility of vaccinia but not listeria or Shigella. Curr. Biol. 1999;9:89–92. doi: 10.1016/s0960-9822(99)80020-7. [DOI] [PubMed] [Google Scholar]
- Frischknecht F., Moreau V., Rottger S., Gonfloni S., Reckmann I., Superti-Furga G., Way M. Actin-based motility of vaccinia virus mimics receptor tyrosine kinase signalling. Nature. 1999;401:926–929. doi: 10.1038/44860. [DOI] [PubMed] [Google Scholar]
- Ganesh L., Leung K., Lore K., Levin R., Panet A., Schwartz O., Koup R.A., Nabel G.J. Infection of specific dendritic cells by CCR5-tropic human immunodeficiency virus type 1 promotes cell-mediated transmission of virus resistant to broadly neutralizing antibodies. J. Virol. 2004;78:11980–11987. doi: 10.1128/JVI.78.21.11980-11987.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Generous A.R., Harrison O.J., Troyanovsky R.B., Mateo M., Navaratnarajah C.K., Donohue R.C., Pfaller C.K., Alekhina O., Sergeeva A.P., Indra I. Trans-endocytosis elicited by nectins transfers cytoplasmic cargo, including infectious material, between cells. J. Cell Sci. 2019;132:jcs235507. doi: 10.1242/jcs.235507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giorda K.M., Hebert D.N. Viroporins customize host cells for efficient viral propagation. DNA Cell Biol. 2013;32:557–564. doi: 10.1089/dna.2013.2159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gombos R.B., Kolodkin-Gal D., Eslamizar L., Owuor J.O., Mazzola E., Gonzalez A.M., Korioth-Schmitz B., Gelman R.S., Montefiori D.C., Haynes B.F. Inhibitory effect of individual or combinations of broadly neutralizing antibodies and antiviral reagents against cell-free and cell-to-cell HIV-1 transmission. J. Virol. 2015;89:7813–7828. doi: 10.1128/JVI.00783-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gower T.L., Pastey M.K., Peeples M.E., Collins P.L., McCurdy L.H., Hart T.K., Guth A., Johnson T.R., Graham B.S. RhoA signaling is required for respiratory syncytial virus-induced syncytium formation and filamentous virion morphology. J. Virol. 2005;79:5326–5336. doi: 10.1128/JVI.79.9.5326-5336.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goyal A., Murray J.M. Modelling the impact of cell-to-cell transmission in hepatitis B virus. PLoS One. 2016;11:e0161978. doi: 10.1371/journal.pone.0161978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graw F., Perelson A.S. Modeling viral spread. Annu. Rev. Virol. 2016;3:555–572. doi: 10.1146/annurev-virology-110615-042249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graw F., Martin D.N., Perelson A.S., Uprichard S.L., Dahari H. Quantification of hepatitis C virus cell-to-cell spread using a stochastic modeling approach. J. Virol. 2015;89:6551–6561. doi: 10.1128/JVI.00016-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross C., Wiesmann V., Millen S., Kalmer M., Wittenberg T., Gettemans J., Thoma-Kress A.K. The tax-inducible actin-bundling protein fascin is crucial for release and cell-to-cell transmission of human T-cell leukemia virus type 1 (HTLV-1) PLoS Pathog. 2016;12:e1005916. doi: 10.1371/journal.ppat.1005916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo R., Katz B.B., Tomich J.M., Gallagher T., Fang Y. Porcine reproductive and respiratory syndrome virus utilizes nanotubes for intercellular spread. J. Virol. 2016;90:5163–5175. doi: 10.1128/JVI.00036-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo R., Davis D., Fang Y. Intercellular transfer of mitochondria rescues virus-induced cell death but facilitates cell-to-cell spreading of porcine reproductive and respiratory syndrome virus. Virology. 2018;517:122–134. doi: 10.1016/j.virol.2017.12.018. [DOI] [PubMed] [Google Scholar]
- Gupta P., Balachandran R., Ho M., Enrico A., Rinaldo C. Cell-to-cell transmission of human immunodeficiency virus type 1 in the presence of azidothymidine and neutralizing antibody. J. Virol. 1989;63:2361–2365. doi: 10.1128/jvi.63.5.2361-2365.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamelin M.E., Abed Y., Boivin G. Human metapneumovirus: a new player among respiratory viruses. Clin. Infect. Dis. 2004;38:983–990. doi: 10.1086/382536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernandez L.D., Hoffman L.R., Wolfsberg T.G., White J.M. Virus-cell and cell-cell fusion. Annu. Rev. Cell Dev. Biol. 1996;12:627–661. doi: 10.1146/annurev.cellbio.12.1.627. [DOI] [PubMed] [Google Scholar]
- Hotard A.L., Lee S., Currier M.G., Crowe J.E., Jr., Sakamoto K., Newcomb D.C., Peebles R.S., Jr., Plemper R.K., Moore M.L. Identification of residues in the human respiratory syncytial virus fusion protein that modulate fusion activity and pathogenesis. J. Virol. 2015;89:512–522. doi: 10.1128/JVI.02472-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard P.W., Wright C.C., Howard T., Johnson D.C. Herpes simplex virus gE/gI extracellular domains promote axonal transport and spread from neurons to epithelial cells. J. Virol. 2014;88:11178–11186. doi: 10.1128/JVI.01627-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu L., Crawford S.E., Hyser J.M., Estes M.K., Prasad B.V. Rotavirus non-structural proteins: structure and function. Curr. Opin. Virol. 2012;2:380–388. doi: 10.1016/j.coviro.2012.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hubner W., McNerney G.P., Chen P., Dale B.M., Gordon R.E., Chuang F.Y., Li X.D., Asmuth D.M., Huser T., Chen B.K. Quantitative 3D video microscopy of HIV transfer across T cell virological synapses. Science. 2009;323:1743–1747. doi: 10.1126/science.1167525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humphries A.C., Dodding M.P., Barry D.J., Collinson L.M., Durkin C.H., Way M. Clathrin potentiates vaccinia-induced actin polymerization to facilitate viral spread. Cell Host Microbe. 2012;12:346–359. doi: 10.1016/j.chom.2012.08.002. [DOI] [PubMed] [Google Scholar]
- Igakura T., Stinchcombe J.C., Goon P.K., Taylor G.P., Weber J.N., Griffiths G.M., Tanaka Y., Osame M., Bangham C.R. Spread of HTLV-I between lymphocytes by virus-induced polarization of the cytoskeleton. Science. 2003;299:1713–1716. doi: 10.1126/science.1080115. [DOI] [PubMed] [Google Scholar]
- Imle A., Kumberger P., Schnellbacher N.D., Fehr J., Carrillo-Bustamante P., Ales J., Schmidt P., Ritter C., Godinez W.J., Muller B. Experimental and computational analyses reveal that environmental restrictions shape HIV-1 spread in 3D cultures. Nat. Commun. 2019;10:2144. doi: 10.1038/s41467-019-09879-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacob C.L., Lamorte L., Sepulveda E., Lorenz I.C., Gauthier A., Franti M. Neutralizing antibodies are unable to inhibit direct viral cell-to-cell spread of human cytomegalovirus. Virology. 2013;444:140–147. doi: 10.1016/j.virol.2013.06.002. [DOI] [PubMed] [Google Scholar]
- Jansens R.J.J., Van den Broeck W., De Pelsmaeker S., Lamote J.A.S., Van Waesberghe C., Couck L., Favoreel H.W. Pseudorabies virus US3-induced tunneling nanotubes contain stabilized microtubules, interact with neighbouring cells via cadherins and allow intercellular molecular communication. J. Virol. 2017;91 doi: 10.1128/JVI.00749-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jansens R.J.J., Tishchenko A., Favoreel H.W. Bridging the gap: virus long-distance spread via tunneling nanotubes. J. Virol. 2020;94:e02120–19. doi: 10.1128/JVI.02120-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin J., Sherer N.M., Heidecker G., Derse D., Mothes W. Assembly of the murine leukemia virus is directed towards sites of cell-cell contact. PLoS Biol. 2009;7:e1000163. doi: 10.1371/journal.pbio.1000163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D.C., Baines J.D. Herpesviruses remodel host membranes for virus egress. Nat. Rev. Microbiol. 2011;9:382–394. doi: 10.1038/nrmicro2559. [DOI] [PubMed] [Google Scholar]
- Johnson D.C., Huber M.T. Directed egress of animal viruses promotes cell-to-cell spread. J. Virol. 2002;76:1–8. doi: 10.1128/JVI.76.1.1-8.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D.C., Webb M., Wisner T.W., Brunetti C. Herpes simplex virus gE/gI sorts nascent virions to epithelial cell junctions, promoting virus spread. J. Virol. 2001;75:821–833. doi: 10.1128/JVI.75.2.821-833.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jolly C., Sattentau Q.J. Retroviral spread by induction of virological synapses. Traffic. 2004;5:643–650. doi: 10.1111/j.1600-0854.2004.00209.x. [DOI] [PubMed] [Google Scholar]
- Jolly C., Kashefi K., Hollinshead M., Sattentau Q.J. HIV-1 cell to cell transfer across an Env-induced, actin-dependent synapse. J. Exp. Med. 2004;199:283–293. doi: 10.1084/jem.20030648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jolly C., Booth N.J., Neil S.J. Cell-cell spread of human immunodeficiency virus type 1 overcomes tetherin/BST-2-mediated restriction in T cells. J. Virol. 2010;84:12185–12199. doi: 10.1128/JVI.01447-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jolly C., Welsch S., Michor S., Sattentau Q.J. The regulated secretory pathway in CD4(+) T cells contributes to human immunodeficiency virus type-1 cell-to-cell spread at the virological synapse. PLoS Pathog. 2011;7:e1002226. doi: 10.1371/journal.ppat.1002226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jouvenet N., Windsor M., Rietdorf J., Hawes P., Monaghan P., Way M., Wileman T. African swine fever virus induces filopodia-like projections at the plasma membrane. Cell. Microbiol. 2006;8:1803–1811. doi: 10.1111/j.1462-5822.2006.00750.x. [DOI] [PubMed] [Google Scholar]
- Jung A., Maier R., Vartanian J.P., Bocharov G., Jung V., Fischer U., Meese E., Wain-Hobson S., Meyerhans A. Recombination: multiply infected spleen cells in HIV patients. Nature. 2002;418:144. doi: 10.1038/418144a. [DOI] [PubMed] [Google Scholar]
- Kielian M. Mechanisms of virus membrane fusion proteins. Annu. Rev. Virol. 2014;1:171–189. doi: 10.1146/annurev-virology-031413-085521. [DOI] [PubMed] [Google Scholar]
- Kinder J.T., Moncman C.L., Barrett C., Jin H., Kallewaard N., Dutch R.E. Respiratory syncytial virus (RSV) and human metapneumovirus (HMPV) infections in 3-D human airway tissues expose an interesting dichotomy in viral replication, spread, and inhibition by neutralizing antibodies. J. Virol. 2020:JVI.01068–20. doi: 10.1128/JVI.01068-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolodkin-Gal D., Hulot S.L., Korioth-Schmitz B., Gombos R.B., Zheng Y., Owuor J., Lifton M.A., Ayeni C., Najarian R.M., Yeh W.W. Efficiency of cell-free and cell-associated virus in mucosal transmission of human immunodeficiency virus type 1 and simian immunodeficiency virus. J. Virol. 2013;87:13589–13597. doi: 10.1128/JVI.03108-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kratchmarov R., Kramer T., Greco T.M., Taylor M.P., Ch'ng T.H., Cristea I.M., Enquist L.W. Glycoproteins gE and gI are required for efficient KIF1A-dependent anterograde axonal transport of alphaherpesvirus particles in neurons. J. Virol. 2013;87:9431–9440. doi: 10.1128/JVI.01317-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kress A.K., Kalmer M., Rowan A.G., Grassmann R., Fleckenstein B. The tumor marker Fascin is strongly induced by the Tax oncoprotein of HTLV-1 through NF-kappaB signals. Blood. 2011;117:3609–3612. doi: 10.1182/blood-2010-09-305805. [DOI] [PubMed] [Google Scholar]
- Krupovic M., Cvirkaite-Krupovic V., Bamford D.H. Protein A33 responsible for antibody-resistant spread of Vaccinia virus is homologous to C-type lectin-like proteins. Virus Res. 2010;151:97–101. doi: 10.1016/j.virusres.2010.03.004. [DOI] [PubMed] [Google Scholar]
- Kumar A., Kim J.H., Ranjan P., Metcalfe M.G., Cao W., Mishina M., Gangappa S., Guo Z., Boyden E.S., Zaki S. Influenza virus exploits tunneling nanotubes for cell-to-cell spread. Sci. Rep. 2017;7:40360. doi: 10.1038/srep40360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumberger P., Durso-Cain K., Uprichard S.L., Dahari H., Graw F. Accounting for space-quantification of cell-to-cell transmission kinetics using virus dynamics models. Viruses. 2018;10:200. doi: 10.3390/v10040200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Labudova M., Tomaskova J., Skultety L., Pastorek J., Pastorekova S. The nucleoprotein of lymphocytic choriomeningitis virus facilitates spread of persistent infection through stabilization of the keratin network. J. Virol. 2009;83:7842–7849. doi: 10.1128/JVI.00309-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Labudova M., Zduriencikova M., Fabianova A., Baratova M., Ditte P., Radvak P., Nemcovicova I., Pastorekova S., Pastorek J. Absence of keratin 1 restricts the course of infection with lymphocytic choriomeningitis virus strain MX. Acta Virol. 2019;63:169–185. doi: 10.4149/av_2019_209. [DOI] [PubMed] [Google Scholar]
- Law M., Hollinshead R., Smith G.L. Antibody-sensitive and antibody-resistant cell-to-cell spread by vaccinia virus: role of the A33R protein in antibody-resistant spread. J. Gen. Virol. 2002;83:209–222. doi: 10.1099/0022-1317-83-1-209. [DOI] [PubMed] [Google Scholar]
- Law K.M., Komarova N.L., Yewdall A.W., Lee R.K., Herrera O.L., Wodarz D., Chen B.K. In vivo HIV-1 cell-to-cell transmission promotes multicopy micro-compartmentalized infection. Cell Rep. 2016;15:2771–2783. doi: 10.1016/j.celrep.2016.05.059. [DOI] [PubMed] [Google Scholar]
- Lawrence D.M., Patterson C.E., Gales T.L., D'Orazio J.L., Vaughn M.M., Rall G.F. Measles virus spread between neurons requires cell contact but not CD46 expression, syncytium formation, or extracellular virus production. J. Virol. 2000;74:1908–1918. doi: 10.1128/jvi.74.4.1908-1918.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehmann M.J., Sherer N.M., Marks C.B., Pypaert M., Mothes W. Actin- and myosin-driven movement of viruses along filopodia precedes their entry into cells. J. Cell Biol. 2005;170:317–325. doi: 10.1083/jcb.200503059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorizate M., Krausslich H.G. Role of lipids in virus replication. Cold Spring Harb. Perspect. Biol. 2011;3:a004820. doi: 10.1101/cshperspect.a004820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lupberger J., Zeisel M.B., Xiao F., Thumann C., Fofana I., Zona L., Davis C., Mee C.J., Turek M., Gorke S. EGFR and EphA2 are host factors for hepatitis C virus entry and possible targets for antiviral therapy. Nat. Med. 2011;17:589–595. doi: 10.1038/nm.2341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyman M.G., Feierbach B., Curanovic D., Bisher M., Enquist L.W. Pseudorabies virus Us9 directs axonal sorting of viral capsids. J. Virol. 2007;81:11363–11371. doi: 10.1128/JVI.01281-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin N., Welsch S., Jolly C., Briggs J.A., Vaux D., Sattentau Q.J. Virological synapse-mediated spread of human immunodeficiency virus type 1 between T cells is sensitive to entry inhibition. J. Virol. 2010;84:3516–3527. doi: 10.1128/JVI.02651-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez M.G., Kielian M. Intercellular extensions are induced by the alphavirus structural proteins and mediate virus transmission. PLoS Pathog. 2016;12:e1006061. doi: 10.1371/journal.ppat.1006061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez M.G., Snapp E.L., Perumal G.S., Macaluso F.P., Kielian M. Imaging the alphavirus exit pathway. J. Virol. 2014;88:6922–6933. doi: 10.1128/JVI.00592-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marzook N.B., Latham S.L., Lynn H., McKenzie C., Chaponnier C., Grau G.E., Newsome T.P. Divergent roles of beta- and gamma-actin isoforms during spread of vaccinia virus. Cytoskeleton. 2017;74:170–183. doi: 10.1002/cm.21356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massanella M., Puigdomenech I., Cabrera C., Fernandez-Figueras M.T., Aucher A., Gaibelet G., Hudrisier D., Garcia E., Bofill M., Clotet B. Antigp41 antibodies fail to block early events of virological synapses but inhibit HIV spread between T cells. AIDS. 2009;23:183–188. doi: 10.1097/QAD.0b013e32831ef1a3. [DOI] [PubMed] [Google Scholar]
- Mateo M., Generous A., Sinn P.L., Cattaneo R. Connections matter—how viruses use cell-cell adhesion components. J. Cell Sci. 2015;128:431–439. doi: 10.1242/jcs.159400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattila P.K., Lappalainen P. Filopodia: molecular architecture and cellular functions. Nat. Rev. Mol. Cell Biol. 2008;9:446–454. doi: 10.1038/nrm2406. [DOI] [PubMed] [Google Scholar]
- Mayer M.L., James M.H., Russell R.J., Kelly J.S., Wise J.C., Pasternak C.A. Spontaneous electrical activity induced by herpes virus infection in rat sensory neuron cultures. Brain Res. 1985;341:360–364. doi: 10.1016/0006-8993(85)91075-3. [DOI] [PubMed] [Google Scholar]
- McCarthy K.M., Tank D.W., Enquist L.W. Pseudorabies virus infection alters neuronal activity and connectivity in vitro. PLoS Pathog. 2009;5:e1000640. doi: 10.1371/journal.ppat.1000640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDonald D., Wu L., Bohks S.M., KewalRamani V.N., Unutmaz D., Hope T.J. Recruitment of HIV and its receptors to dendritic cell-T cell junctions. Science. 2003;300:1295–1297. doi: 10.1126/science.1084238. [DOI] [PubMed] [Google Scholar]
- McGraw H.M., Friedman H.M. Herpes simplex virus type 1 glycoprotein E mediates retrograde spread from epithelial cells to neurites. J. Virol. 2009;83:4791–4799. doi: 10.1128/JVI.02341-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehedi M., McCarty T., Martin S.E., Le Nouen C., Buehler E., Chen Y.C., Smelkinson M., Ganesan S., Fischer E.R., Brock L.G. Actin-related protein 2 (ARP2) and virus-induced filopodia facilitate human respiratory syncytial virus spread. PLoS Pathog. 2016;12:e1006062. doi: 10.1371/journal.ppat.1006062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyashita S., Ishibashi K., Kishino H., Ishikawa M. Viruses roll the dice: the stochastic behavior of viral genome molecules accelerates viral adaptation at the cell and tissue levels. PLoS Biol. 2015;13:e1002094. doi: 10.1371/journal.pbio.1002094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyauchi K., Kim Y., Latinovic O., Morozov V., Melikyan G.B. HIV enters cells via endocytosis and dynamin-dependent fusion with endosomes. Cell. 2009;137:433–444. doi: 10.1016/j.cell.2009.02.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori K., Haruyama T., Nagata K. Tamiflu-resistant but HA-mediated cell-to-cell transmission through apical membranes of cell-associated influenza viruses. PLoS One. 2011;6:e28178. doi: 10.1371/journal.pone.0028178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris-Love J., Gee G.V., O’Hara B.A., Assetta B., Atkinson A.L., Dugan A.S., Haley S.A., Atwood W.J. JC polyomavirus uses extracellular vesicles to infect target cells. MBio. 2019;10:e00379–19. doi: 10.1128/mBio.00379-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murooka T.T., Deruaz M., Marangoni F., Vrbanac V.D., Seung E., von Andrian U.H., Tager A.M., Luster A.D., Mempel T.R. HIV-infected T cells are migratory vehicles for viral dissemination. Nature. 2012;490:283–287. doi: 10.1038/nature11398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nardacci R., Antinori A., Larocca L.M., Arena V., Amendola A., Perfettini J.L., Kroemer G., Piacentini M. Characterization of cell death pathways in human immunodeficiency virus-associated encephalitis. Am. J. Pathol. 2005;167:695–704. doi: 10.1016/S0002-9440(10)62044-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neilson K.A., Yunis E.J. Demonstration of respiratory syncytial virus in an autopsy series. Pediatr. Pathol. 1990;10:491–502. doi: 10.3109/15513819009067138. [DOI] [PubMed] [Google Scholar]
- Nejmeddine M., Barnard A.L., Tanaka Y., Taylor G.P., Bangham C.R. Human T-lymphotropic virus, type 1, tax protein triggers microtubule reorientation in the virological synapse. J. Biol. Chem. 2005;280:29653–29660. doi: 10.1074/jbc.M502639200. [DOI] [PubMed] [Google Scholar]
- Newsome T.P., Marzook N.B. Viruses that ride on the coat-tails of actin nucleation. Semin. Cell Dev. Biol. 2015;46:155–163. doi: 10.1016/j.semcdb.2015.10.008. [DOI] [PubMed] [Google Scholar]
- Newsome T.P., Weisswange I., Frischknecht F., Way M. Abl collaborates with Src family kinases to stimulate actin-based motility of vaccinia virus. Cell. Microbiol. 2006;8:233–241. doi: 10.1111/j.1462-5822.2005.00613.x. [DOI] [PubMed] [Google Scholar]
- Nieva J.L., Madan V., Carrasco L. Viroporins: structure and biological functions. Nat. Rev. Microbiol. 2012;10:563–574. doi: 10.1038/nrmicro2820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nobes C.D., Hall A. Rho, rac, and cdc42 GTPases regulate the assembly of multimolecular focal complexes associated with actin stress fibers, lamellipodia, and filopodia. Cell. 1995;81:53–62. doi: 10.1016/0092-8674(95)90370-4. [DOI] [PubMed] [Google Scholar]
- Onfelt B., Nedvetzki S., Benninger R.K., Purbhoo M.A., Sowinski S., Hume A.N., Seabra M.C., Neil M.A., French P.M., Davis D.M. Structurally distinct membrane nanotubes between human macrophages support long-distance vesicular traffic or surfing of bacteria. J. Immunol. 2006;177:8476–8483. doi: 10.4049/jimmunol.177.12.8476. [DOI] [PubMed] [Google Scholar]
- Pais-Correia A.M., Sachse M., Guadagnini S., Robbiati V., Lasserre R., Gessain A., Gout O., Alcover A., Thoulouze M.I. Biofilm-like extracellular viral assemblies mediate HTLV-1 cell-to-cell transmission at virological synapses. Nat. Med. 2010;16:83–89. doi: 10.1038/nm.2065. [DOI] [PubMed] [Google Scholar]
- Paloheimo O., Ihalainen T.O., Tauriainen S., Valilehto O., Kirjavainen S., Niskanen E.A., Laakkonen J.P., Hyoty H., Vihinen-Ranta M. Coxsackievirus B3-induced cellular protrusions: structural characteristics and functional competence. J. Virol. 2011;85:6714–6724. doi: 10.1128/JVI.00247-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panasiuk M., Rychlowski M., Derewonko N., Bienkowska-Szewczyk K. Tunneling nanotubes as a novel route of cell-to-cell spread of herpesviruses. J. Virol. 2018;92:e00090–18. doi: 10.1128/JVI.00090-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pantaleo G., Demarest J.F., Vaccarezza M., Graziosi C., Bansal G.P., Koenig S., Fauci A.S. Effect of anti-V3 antibodies on cell-free and cell-to-cell human immunodeficiency virus transmission. Eur. J. Immunol. 1995;25:226–231. doi: 10.1002/eji.1830250137. [DOI] [PubMed] [Google Scholar]
- Pontow S.E., Heyden N.V., Wei S., Ratner L. Actin cytoskeletal reorganizations and coreceptor-mediated activation of rac during human immunodeficiency virus-induced cell fusion. J. Virol. 2004;78:7138–7147. doi: 10.1128/JVI.78.13.7138-7147.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rager M., Vongpunsawad S., Duprex W.P., Cattaneo R. Polyploid measles virus with hexameric genome length. EMBO J. 2002;21:2364–2372. doi: 10.1093/emboj/21.10.2364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramakrishnaiah V., Thumann C., Fofana I., Habersetzer F., Pan Q., de Ruiter P.E., Willemsen R., Demmers J.A., Stalin Raj V., Jenster G. Exosome-mediated transmission of hepatitis C virus between human hepatoma Huh7.5 cells. Proc. Natl. Acad. Sci. U. S. A. 2013;110:13109–13113. doi: 10.1073/pnas.1221899110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Real F., Sennepin A., Ganor Y., Schmitt A., Bomsel M. Live imaging of HIV-1 transfer across T cell virological synapse to epithelial cells that promotes stromal macrophage infection. Cell Rep. 2018;23:1794–1805. doi: 10.1016/j.celrep.2018.04.028. [DOI] [PubMed] [Google Scholar]
- Reeves P.M., Smith S.K., Olson V.A., Thorne S.H., Bornmann W., Damon I.K., Kalman D. Variola and monkeypox viruses utilize conserved mechanisms of virion motility and release that depend on abl and SRC family tyrosine kinases. J. Virol. 2011;85:21–31. doi: 10.1128/JVI.01814-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reichelt M., Zerboni L., Arvin A.M. Mechanisms of varicella-zoster virus neuropathogenesis in human dorsal root ganglia. J. Virol. 2008;82:3971–3983. doi: 10.1128/JVI.02592-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson M.W., Carroll R.G., Stremlau M., Korokhov N., Humeau L.M., Silvestri G., Sodroski J., Riley J.L. Mode of transmission affects the sensitivity of human immunodeficiency virus type 1 to restriction by rhesus TRIM5alpha. J. Virol. 2008;82:11117–11128. doi: 10.1128/JVI.01046-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivera-Serrano E.E., Gonzalez-Lopez O., Das A., Lemon S.M. Cellular entry and uncoating of naked and quasi-enveloped human hepatoviruses. Elife. 2019;8:e43983. doi: 10.7554/eLife.43983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts K.L., Manicassamy B., Lamb R.A. Influenza A virus uses intercellular connections to spread to neighboring cells. J. Virol. 2015;89:1537–1549. doi: 10.1128/JVI.03306-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson S.M., Tsueng G., Sin J., Mangale V., Rahawi S., McIntyre L.L., Williams W., Kha N., Cruz C., Hancock B.M. Coxsackievirus B exits the host cell in shed microvesicles displaying autophagosomal markers. PLoS Pathog. 2014;10:e1004045. doi: 10.1371/journal.ppat.1004045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudnicka D., Feldmann J., Porrot F., Wietgrefe S., Guadagnini S., Prevost M.C., Estaquier J., Haase A.T., Sol-Foulon N., Schwartz O. Simultaneous cell-to-cell transmission of human immunodeficiency virus to multiple targets through polysynapses. J. Virol. 2009;83:6234–6246. doi: 10.1128/JVI.00282-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell R.A., Martin N., Mitar I., Jones E., Sattentau Q.J. Multiple proviral integration events after virological synapse-mediated HIV-1 spread. Virology. 2013;443:143–149. doi: 10.1016/j.virol.2013.05.005. [DOI] [PubMed] [Google Scholar]
- Rustom A., Saffrich R., Markovic I., Walther P., Gerdes H.H. Nanotubular highways for intercellular organelle transport. Science. 2004;303:1007–1010. doi: 10.1126/science.1093133. [DOI] [PubMed] [Google Scholar]
- Sakisaka T., Taniguchi T., Nakanishi H., Takahashi K., Miyahara M., Ikeda W., Yokoyama S., Peng Y.F., Yamanishi K., Takai Y. Requirement of interaction of nectin-1alpha/HveC with afadin for efficient cell-cell spread of herpes simplex virus type 1. J. Virol. 2001;75:4734–4743. doi: 10.1128/JVI.75.10.4734-4743.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanjuan R. Collective infectious units in viruses. Trends Microbiol. 2017;25:402–412. doi: 10.1016/j.tim.2017.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanjuan R., Thoulouze M.I. Why viruses sometimes disperse in groups?(dagger) Virus Evol. 2019;5:vez014. doi: 10.1093/ve/vez014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santiana M., Ghosh S., Ho B.A., Rajasekaran V., Du W.L., Mutsafi Y., De Jesus-Diaz D.A., Sosnovtsev S.V., Levenson E.A., Parra G.I. Vesicle-cloaked virus clusters are optimal units for inter-organismal viral transmission. Cell Host Microbe. 2018;24:208–220 e208. doi: 10.1016/j.chom.2018.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarfo A., Starkey J., Mellinger E., Zhang D., Chadha P., Carmichael J., Wills J.W. The UL21 tegument protein of herpes simplex virus 1 is differentially required for the syncytial phenotype. J. Virol. 2017;91:e01161–17. doi: 10.1128/JVI.01161-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scaplehorn N., Holmstrom A., Moreau V., Frischknecht F., Reckmann I., Way M. Grb2 and Nck act cooperatively to promote actin-based motility of vaccinia virus. Curr. Biol. 2002;12:740–745. doi: 10.1016/s0960-9822(02)00812-6. [DOI] [PubMed] [Google Scholar]
- Schelhaas M., Ewers H., Rajamaki M.L., Day P.M., Schiller J.T., Helenius A. Human papillomavirus type 16 entry: retrograde cell surface transport along actin-rich protrusions. PLoS Pathog. 2008;4:e1000148. doi: 10.1371/journal.ppat.1000148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schowalter R.M., Wurth M.A., Aguilar H.C., Lee B., Moncman C.L., McCann R.O., Dutch R.E. Rho GTPase activity modulates paramyxovirus fusion protein-mediated cell-cell fusion. Virology. 2006;350:323–334. doi: 10.1016/j.virol.2006.01.033. [DOI] [PubMed] [Google Scholar]
- Sewald X., Ladinsky M.S., Uchil P.D., Beloor J., Pi R., Herrmann C., Motamedi N., Murooka T.T., Brehm M.A., Greiner D.L. Retroviruses use CD169-mediated trans-infection of permissive lymphocytes to establish infection. Science. 2015;350:563–567. doi: 10.1126/science.aab2749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sherer N.M., Lehmann M.J., Jimenez-Soto L.F., Horensavitz C., Pypaert M., Mothes W. Retroviruses can establish filopodial bridges for efficient cell-to-cell transmission. Nat. Cell Biol. 2007;9:310–315. doi: 10.1038/ncb1544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirogane Y., Watanabe S., Yanagi Y. Cooperation between different RNA virus genomes produces a new phenotype. Nat. Commun. 2012;3:1235. doi: 10.1038/ncomms2252. [DOI] [PubMed] [Google Scholar]
- Shmulevitz M., Duncan R. A new class of fusion-associated small transmembrane (FAST) proteins encoded by the non-enveloped fusogenic reoviruses. EMBO J. 2000;19:902–912. doi: 10.1093/emboj/19.5.902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigal A., Kim J.T., Balazs A.B., Dekel E., Mayo A., Milo R., Baltimore D. Cell-to-cell spread of HIV permits ongoing replication despite antiretroviral therapy. Nature. 2011;477:95–98. doi: 10.1038/nature10347. [DOI] [PubMed] [Google Scholar]
- Singh B.K., Hornick A.L., Krishnamurthy S., Locke A.C., Mendoza C.A., Mateo M., Miller-Hunt C.L., Cattaneo R., Sinn P.L. The nectin-4/afadin protein complex and intercellular membrane pores contribute to rapid spread of measles virus in primary human airway epithelia. J. Virol. 2015;89:7089–7096. doi: 10.1128/JVI.00821-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh B.K., Li N., Mark A.C., Mateo M., Cattaneo R., Sinn P.L. Cell-to-cell contact and nectin-4 govern spread of measles virus from primary human myeloid cells to primary human airway epithelial cells. J. Virol. 2016;90:6808–6817. doi: 10.1128/JVI.00266-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh B.K., Pfaller C.K., Cattaneo R., Sinn P.L. Measles virus ribonucleoprotein complexes rapidly spread across well-differentiated primary human airway epithelial cells along F-actin rings. MBio. 2019;10:e02434–19. doi: 10.1128/mBio.02434-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith S.A., Derdeyn C.A. New connections: cell-to-cell HIV-1 transmission, resistance to broadly neutralizing antibodies, and an envelope sorting motif. J. Virol. 2017;91:e00149–17. doi: 10.1128/JVI.00149-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith G.L., Law M. The exit of vaccinia virus from infected cells. Virus Res. 2004;106:189–197. doi: 10.1016/j.virusres.2004.08.015. [DOI] [PubMed] [Google Scholar]
- Smith J.L., Lidke D.S., Ozbun M.A. Virus activated filopodia promote human papillomavirus type 31 uptake from the extracellular matrix. Virology. 2008;381:16–21. doi: 10.1016/j.virol.2008.08.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snyder A., Polcicova K., Johnson D.C. Herpes simplex virus gE/gI and US9 proteins promote transport of both capsids and virion glycoproteins in neuronal axons. J. Virol. 2008;82:10613–10624. doi: 10.1128/JVI.01241-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sourisseau M., Sol-Foulon N., Porrot F., Blanchet F., Schwartz O. Inefficient human immunodeficiency virus replication in mobile lymphocytes. J. Virol. 2007;81:1000–1012. doi: 10.1128/JVI.01629-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sowinski S., Jolly C., Berninghausen O., Purbhoo M.A., Chauveau A., Kohler K., Oddos S., Eissmann P., Brodsky F.M., Hopkins C. Membrane nanotubes physically connect T cells over long distances presenting a novel route for HIV-1 transmission. Nat. Cell Biol. 2008;10:211–219. doi: 10.1038/ncb1682. [DOI] [PubMed] [Google Scholar]
- Spouge J.I., Shrager R.I., Dimitrov D.S. HIV-1 infection kinetics in tissue cultures. Math. Biosci. 1996;138:1–22. doi: 10.1016/s0025-5564(96)00064-8. [DOI] [PubMed] [Google Scholar]
- Stokes G.V. High-voltage electron microscope study of the release of vaccinia virus from whole cells. J. Virol. 1976;18:636–643. doi: 10.1128/jvi.18.2.636-643.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeuchi K., Miyajima N., Nagata N., Takeda M., Tashiro M. Wild-type measles virus induces large syncytium formation in primary human small airway epithelial cells by a SLAM(CD150)-independent mechanism. Virus Res. 2003;94:11–16. doi: 10.1016/s0168-1702(03)00117-5. [DOI] [PubMed] [Google Scholar]
- Taylor M.P., Kobiler O., Enquist L.W. Alphaherpesvirus axon-to-cell spread involves limited virion transmission. Proc. Natl. Acad. Sci. U. S. A. 2012;109:17046–17051. doi: 10.1073/pnas.1212926109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor M.P., Kramer T., Lyman M.G., Kratchmarov R., Enquist L.W. Visualization of an alphaherpesvirus membrane protein that is essential for anterograde axonal spread of infection in neurons. MBio. 2012;3:e00063–12. doi: 10.1128/mBio.00063-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor M.P., Koyuncu O.O., Enquist L.W. Subversion of the actin cytoskeleton during viral infection. Nat. Rev. Microbiol. 2011;9:427–439. doi: 10.1038/nrmicro2574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thoulouze M.I., Alcover A. Viral extracellular biofilms: a novel spreading trick of viruses? Med. Sci. (Paris) 2010;26:571–573. doi: 10.1051/medsci/2010266-7571. [DOI] [PubMed] [Google Scholar]
- Timpe J.M., Stamataki Z., Jennings A., Hu K., Farquhar M.J., Harris H.J., Schwarz A., Desombere I., Roels G.L., Balfe P. Hepatitis C virus cell-cell transmission in hepatoma cells in the presence of neutralizing antibodies. Hepatology. 2008;47:17–24. doi: 10.1002/hep.21959. [DOI] [PubMed] [Google Scholar]
- van Montfort T., Nabatov A.A., Geijtenbeek T.B., Pollakis G., Paxton W.A. Efficient capture of antibody neutralized HIV-1 by cells expressing DC-SIGN and transfer to CD4 + T lymphocytes. J. Immunol. 2007;178:3177–3185. doi: 10.4049/jimmunol.178.5.3177. [DOI] [PubMed] [Google Scholar]
- Van Prooyen N., Gold H., Andresen V., Schwartz O., Jones K., Ruscetti F., Lockett S., Gudla P., Venzon D., Franchini G. Human T-cell leukemia virus type 1 p8 protein increases cellular conduits and virus transmission. Proc. Natl. Acad. Sci. U. S. A. 2010;107:20738–20743. doi: 10.1073/pnas.1009635107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vargas S.O., Kozakewich H.P., Perez-Atayde A.R., McAdam A.J. Pathology of human metapneumovirus infection: insights into the pathogenesis of a newly identified respiratory virus. Pediatr. Dev. Pathol. 2004;7:478–486. doi: 10.1007/s10024-004-1011-2. (discussion 421) [DOI] [PubMed] [Google Scholar]
- Vasiliver-Shamis G., Cho M.W., Hioe C.E., Dustin M.L. Human immunodeficiency virus type 1 envelope gp120-induced partial T-cell receptor signaling creates an F-actin-depleted zone in the virological synapse. J. Virol. 2009;83:11341–11355. doi: 10.1128/JVI.01440-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Votteler J., Sundquist W.I. Virus budding and the ESCRT pathway. Cell Host Microbe. 2013;14:232–241. doi: 10.1016/j.chom.2013.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J.H., Janas A.M., Olson W.J., Wu L. Functionally distinct transmission of human immunodeficiency virus type 1 mediated by immature and mature dendritic cells. J. Virol. 2007;81:8933–8943. doi: 10.1128/JVI.00878-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang F., Zumbrun E.E., Huang J., Si H., Makaroun L., Friedman H.M. Herpes simplex virus type 2 glycoprotein E is required for efficient virus spread from epithelial cells to neurons and for targeting viral proteins from the neuron cell body into axons. Virology. 2010;405:269–279. doi: 10.1016/j.virol.2010.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L., Izadmehr S., Kamau E., Kong X.P., Chen B.K. Sequential trafficking of Env and Gag to HIV-1 T cell virological synapses revealed by live imaging. Retrovirology. 2019;16:2. doi: 10.1186/s12977-019-0464-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weissenhorn W., Poudevigne E., Effantin G., Bassereau P. How to get out: ssRNA enveloped viruses and membrane fission. Curr. Opin. Virol. 2013;3:159–167. doi: 10.1016/j.coviro.2013.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welch M.D., Way M. Arp2/3-mediated actin-based motility: a tail of pathogen abuse. Cell Host Microbe. 2013;14:242–255. doi: 10.1016/j.chom.2013.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wisner T.W., Sugimoto K., Howard P.W., Kawaguchi Y., Johnson D.C. Anterograde transport of herpes simplex virus capsids in neurons by both separate and married mechanisms. J. Virol. 2011;85:5919–5928. doi: 10.1128/JVI.00116-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witteveldt J., Evans M.J., Bitzegeio J., Koutsoudakis G., Owsianka A.M., Angus A.G., Keck Z.Y., Foung S.K., Pietschmann T., Rice C.M. CD81 is dispensable for hepatitis C virus cell-to-cell transmission in hepatoma cells. J. Gen. Virol. 2009;90:48–58. doi: 10.1099/vir.0.006700-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao F., Fofana I., Heydmann L., Barth H., Soulier E., Habersetzer F., Doffoel M., Bukh J., Patel A.H., Zeisel M.B. Hepatitis C virus cell-cell transmission and resistance to direct-acting antiviral agents. PLoS Pathog. 2014;10:e1004128. doi: 10.1371/journal.ppat.1004128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao M., Xu N., Wang C., Pang D.W., Zhang Z.L. Dynamic monitoring of membrane nanotubes formation induced by vaccinia virus on a high throughput microfluidic chip. Sci. Rep. 2017;7:44835. doi: 10.1038/srep44835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang E., Arvin A.M., Oliver S.L. The cytoplasmic domain of varicella-zoster virus glycoprotein H regulates syncytia formation and skin pathogenesis. PLoS Pathog. 2014;10:e1004173. doi: 10.1371/journal.ppat.1004173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon M., Spear P.G. Disruption of adherens junctions liberates nectin-1 to serve as receptor for herpes simplex virus and pseudorabies virus entry. J. Virol. 2002;76:7203–7208. doi: 10.1128/JVI.76.14.7203-7208.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahid M.N., Turek M., Xiao F., Thi V.L., Guerin M., Fofana I., Bachellier P., Thompson J., Delang L., Neyts J. The postbinding activity of scavenger receptor class B type I mediates initiation of hepatitis C virus infection and viral dissemination. Hepatology. 2013;57:492–504. doi: 10.1002/hep.26097. [DOI] [PubMed] [Google Scholar]
- Zeisel M.B., Lupberger J., Fofana I., Baumert T.F. Host-targeting agents for prevention and treatment of chronic hepatitis C—perspectives and challenges. J. Hepatol. 2013;58:375–384. doi: 10.1016/j.jhep.2012.09.022. [DOI] [PubMed] [Google Scholar]
- Zhang N., Yan J., Lu G., Guo Z., Fan Z., Wang J., Shi Y., Qi J., Gao G.F. Binding of herpes simplex virus glycoprotein D to nectin-1 exploits host cell adhesion. Nat. Commun. 2011;2:577. doi: 10.1038/ncomms1571. [DOI] [PubMed] [Google Scholar]
- Zhong P., Agosto L.M., Ilinskaya A., Dorjbal B., Truong R., Derse D., Uchil P.D., Heidecker G., Mothes W. Cell-to-cell transmission can overcome multiple donor and target cell barriers imposed on cell-free HIV. PLoS One. 2013;8:e53138. doi: 10.1371/journal.pone.0053138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zsak L., Zuckermann F., Sugg N., Ben-Porat T. Glycoprotein gI of pseudorabies virus promotes cell fusion and virus spread via direct cell-to-cell transmission. J. Virol. 1992;66:2316–2325. doi: 10.1128/jvi.66.4.2316-2325.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]