Abstract
At the end of last century a prominent biochemist once opened the discussion of a controversial issue in the field of Bioenergetics with the following statement: “This is a long story, that shouldn’t be long, but it will take a long time to make it short”. As it happens, such a statement would apply perfectly well to the story of chloroquine (CQ) and hydroxychloroquine (HCQ) in the COVID-19 infection: it has become a veritable saga, with conflicting views that have often gone beyond the normal scientific dialectic, and with conclusions that have frequently been polluted by non scientific opinions: thus, for instance, when National Agencies have taken positions against CQ and HCQ, the move has been seen as a pro-vaccine attempt to block low cost therapy means. And it is difficult to avoid the feeling that the opposition to CQ and HCQ has in large measure been shaped not by scientific arguments, but by the fact that their use has been strongly endorsed by National leaders whose popularity among Western intellectuals is extremely low. The role of the two drugs in the COVID-19 infection thus deserves an objective analysis solely based on scientific facts. This contribution will attempt to produce it.
1. Chemistry and mechanism of action of chloroquine and hydroxychloroquine
CQ and HCQ are derivatives of 4-aminoquinoline: they have an aromatic core and a side chain that makes them weak bases (Fig. 1 ). For decades CQ has been the most important drug in the treatment and prophylaxis of malaria, particularly in the African continent however, its efficacy has gradually declined due to the emergence of chloroquine-resistant strains of P. falciparum, which is responsible for about 75% of the cases of malaria in Africa. The Plasmodium degrades the hemoglobing of the host [1], liberating free heme that is toxic: in the host, heme oxygenase detoxifies the heme into biliverdin, which is then converted by biliverdin reductase to water soluble bilirubin, which is excreted [2]. The plasmodium lacks heme oxygenase, and detoxifies instead the heme by polymerazing it into inert hemozoin [3]. The polymerization of the toxic heme is the target of chloroquine: its inhibition leads to the accumulation of toxic heme which eventually kills the parasite [4,5]. Other effects of the Plasmodium infection have also been described: in general, they are related to the overstimulation of the immune system and of the cytokine network, e.g., tumor necrosis factor alpha and interleukin-6 [6]. Chloroquine had positive effects on these accessory aspects of the malarial infection as well, as was first recognized from the improvements of the cutaneous lupus rashes and inflammatory arthritis in soldiers of the Second World War treated with antimalarials. Its use was thus successfully extended to the treatment of other autoimmune disease like systemic lupus erythematosus, Siogren’s syndrome, rheumatoid arthritis, and sarcoidosis [7]. Outside malaria, the spectrum of possible activities of CQ, and of its derivative HCQ, has been further enlarged by the finding that their biochemical effects were associated with the inhibition of the inflammatory response commonly observed in bacterial and viral infections: CQ inhibits the production/release of tumor necrosis factor alpha (TNF alpha) and of interleukin-6, which are responsible for the pathological symptomatology of viral diseases. Thus, at the turn of last century, CQ was repurposed against HIV and other viruses [[8], [9], [10]].
Fig. 1.
Chloroquine and hydroxychloroquine.
In their immunomodulatory/antiinflammatory effects, CQ and HCQ have multiple modes of action. Outside cells, in vitro experiments have shown that they inhibit the glycosylation of the SARS coronavirus cell surface target, the ACE2 receptor [11]. And a recent structural and molecular modelling study has identified a ganglioside binding domain in the N-terminal portion of the S-protein of COVID-19 that interacts with surface gangliosides of the host cells, facilitating contact with the ACE receptor: CQ and HCQ inhibit the interaction, and thus the penetration of the virus into the target cell [12].
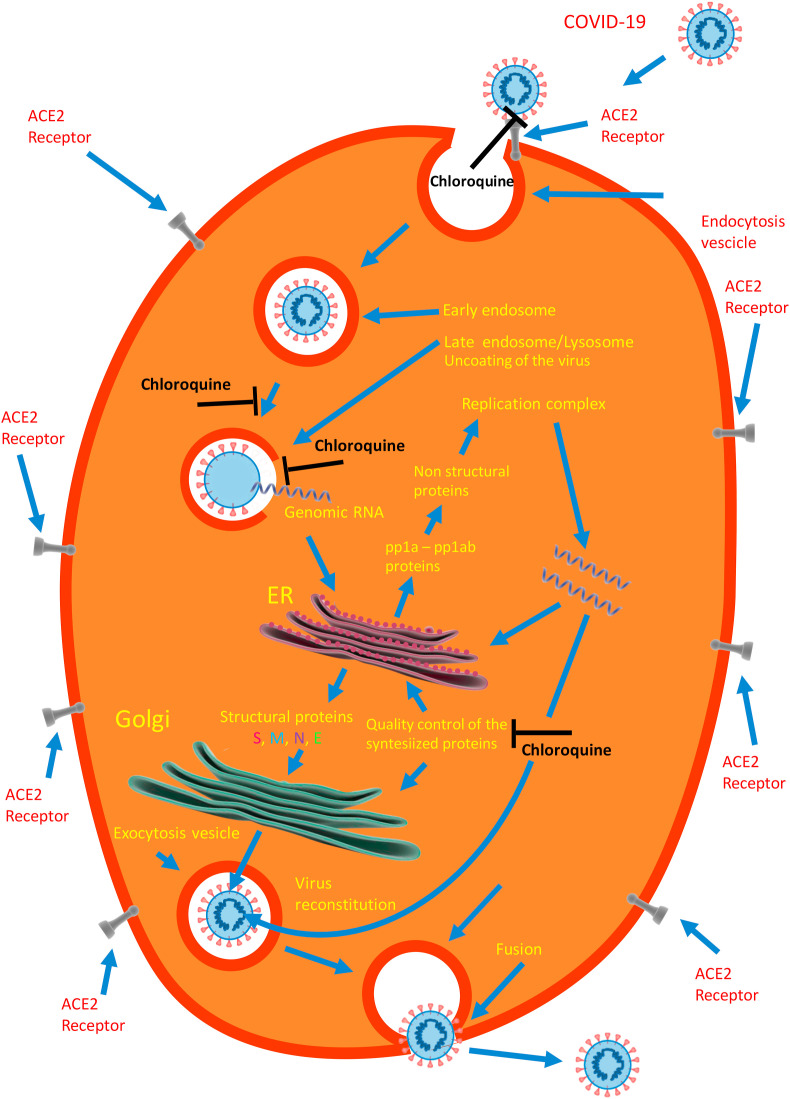
The non-protonated form of CQ and HCQ diffuses across the plasma membrane to the cytoplasm, where it is reprotonated and accumulated in the acidic intracellular organelles such as the endosomes, the trans-Golgi network, and the lysosomes, increasing their pH and thus decreasing the activity of their hydrolytic enzymes. Early in vitro experiments had indeed shown that the pH in endosomes of macrophages increased from 4.5 to 6.5 after a few minutes of exposure to 100 μM CQ [13]. In the absence of CQ, the low internal pH mediates the fusion of the endosomal and viral membranes and thus the internationalization of the virus [14]A quantitative analysis of COVID-19 entry in Vero E6 cells exposed to 50 μM CQ or HCQ [15] has shown that, in untreated cells, after 90 min, 16.2% of the internalized virions were in the early endosomes (EEs), and 34.3% had been transported to late endosomes (ELs). By contrast, in the treated cells, only 2.4% of the virions had remained in the EEs in the case of CQ and 0.03% in the case of HCQ, whereas 35.3% had been transported to the ELs in the case of CQ and 29.2% in the case of HCQ. CQ and HCQ had thus blocked the transport of the COVID-19 virions from the early endosomes to the late endosomes. Eventually,in the absence of CQ or HCQ, the virions ends up in the lysosomes, where the hydrolytic enzymes uncoat it, liberating into the cytoplasm the infectious genomic RNA which is translated into pp1a and pp1ab proteins. These two proteins are cleaved by a protease to make a total of 16 non structural proteins (nsp) [16], some of which form the replication/transcription complex (RNA-dependent RNA polymerase). Work on several viruses has shown that the CQ-induced increase of pH blocks the uncoating activity of the lysosomal hydrolytic enzymes, thus abrogating the entire replication cycle of the virus [17]. The cartoon of Fig. 2 shows the phases of the virus challenge to the target cell, from its penetration into it, to the liberation of the genomic RNA into the cytoplasm, to its process of replication and de novo assembly, to its final exit from the cell to propagate the infection. For reasons of completion, it could be mentioned that the cell penetration of COVID-19 could also occur via a pH independent pathway: whereas in most cell lines the penetration of the virus is promoted by the activation of the S-protein by pH dependent endosomal proteases, in other cell lines, e.g., airway epithaeial cells, a pH-independent protease (TMPRSS2) activates instead the S-protein.
Fig. 2.
A cartoon depicting the attack of a target cell by COVID-19, and the inhibitory actions of chloroquine on it. The endocytosed virus travels from early endosomes to late endosomes-lysosomes, where the acidic environment activates their hydrolytic enzymes the uncoat the virus, liberating its genomic RNA into the cytoplasm. In the endoplasmic reticulum of the host cell (ER) the open reading frames (ORFs) of the genome are translated into pp1a and pp1ab proteins, which are cleaved by proteases of the host cell to 16 non structural proteins (nsps). They form the replicase/transcriptase complex that will produce the new viral RNA genome, which is translated into the four structural proteins E, M, N, and S to reconstitute a new viral particle. After suitable modification and quality control of the structural proteins in the ER and Golgi complex, the new virus travels to the surface of the host cell to be escreted. The text contains additional details.
In a number of viruses, from HIV, to Dengue, to herpes simplex, to human coronaviruses like MERS, CQ has also been shown to interfere with the post-translational modification of viral proteins mediated by proteases and glycosyltransferases in the endoplasmic reticulum and trans-Golgi network of the host cell [[18], [19], [20]] Finally, CQ and HCQ have been found to reduce the production of pro-inflammatory cytokines like IL-1, IL-6, TNF, and IFNgamma by several cell types, e.g., mononuclear cells [21].
One last aspects of the molecular/cellular effects of CQ and HCQ must be mentioned at the end of this chapter on the mechanisms of action of the two drugs: their possible negative side effects. CQ and HCQ have a well studied toxicity profile, and the safety of CQ use has been demonstrated by the decades of its administration to humans in malaria-endemic countries. They are now widely used to treat autoimmune diseases, and, at the dosages that are routinely used, their safety profile is considered very good. Occasionally, however, adverse side effects have been described [22], and it is important to mention them because they have been greatly emphasized in the descriptions and discussions of the results of therapy attempts, including international trials, of COVID-19 using CQ and, especially, HCQ. The most important adverse side effects that have been described are cardiac rhythm disorders, i.e., a prolongation of the electrocardiographic Q-T interval [23], and the development of retinopathy [24]: which is more commonly associated with CQ than with HCQ, and depends on the cumulative dose more than on the daily dose. The retinal damage is attributed to the disruption of the lysosomal degradation process in photoreceptor outer segments.
2. Chloroquine and hydroxychloroquine in the treatment of the COVID-19 infection
As mentioned above, the arrival of CQ (and HCQ) on the scene of antiretroviralviral therapy was sparked by their beneficial effects on the inflammatory response commonly observed in bacterial, fungal, and even viral diseases, but, especially, by the need for treatments less expensive than those under study for a number of diseases: at the end of last century the most important, and thus the prime target, was the HIV infection [25,26]. However, in the following years the antiviral efficiency of CQ was also tested on a number of other viruses, including the SARS coronavirus. Eventually, a study by Wang et al. [27] tested CQ on Vero E6 cells infected with the COVID-19, and compared its effects with those of 4 FDA-approved antiviral drugs, and of the antiviral drugs favipiravid and remdesivir. The study revealed that CQ and remdesivir were highly effective against the COVID-19 infection of the cells. At that time, remdesivir had already shown efficacy against the COVID-19 infection on a patient in the USA, but the results on CQ were novel. The study by Wang et al. was published in March 2020, when the COVID-19 infection was reaching epidemic dimensions in Wuhan in China, and was immediately followed by about 20 Chinese studies that had the atypical pneumonias of the COVID-19 disease as the main target. They were not long range randomized trials, but their apparently beneficial results nevertheless induced the Chinese sanitary authorities to officially recommend CQ and HCQ for the treatment of the infection. Beneficial results were also found in a small French study [28] (at that time COVID-19 had already spread to Europe): HCQ had reduced or eliminated the viral load in the patients, the effect being reinforced by azithromycin.
The French study had a number of serious shortcomings that later commentaries have convincingly discussed in detail [29,30], but it nevertheless immediately attracted worldwide attention. Its findings were rapidly disseminated by the press and amplified by the social media, initiating a period of great popularity of HCQ. Government and institutional leaders endorsed it, to the point that it was proclaimed to be “game changer” in the fight against COVID-19. These overtones, and the enthusiastic endorsements by political Leaders whose popularity indexes were abysmally low among Western opinion makers, were clearly damaging: they were instrumental in generating a feeeling of annoyance against CQ and HCQ, and the ideological backlash that soon hit them. However, it wasn’t only Governments and leaders who had so promptly and generally accepted CQ and HCQ: Hospitals and doctors in general, and also National Agencies, started administering and recommending HCQ. As an example, it may be interesting to quote here a document of the Italian Federation of Family Doctors, that included HCQ in the treatment of certified, and even only suspected, cases of COVID-19 infection. Adverse feelings were nevertheless beginning to surface: for instance, the document of the Italian Family Doctors had an appendix that contained a long list of conditions, that in effect embraced most human pathologies, that were incompatible with the HCQ therapy. In hindsight, it was an indirect indication of the mounting wave of alarming warnings on the dangers of the HCQ therapy. And so, in a short while CQ and HCQ became the subjects of a dispute that was gradually corrupted by non-scientific arguments that divided the scientific community in a way similar to that of supporters of competing sports teams. From the scientific angle, the unpleasant aspect of the dispute was the rush to publish as rapidly as possible results that would either prove, or diprove, the efficacy of CQ or HCQ, and/or their safety or their dangers. The standard in the generation and interpretation of the results rapidly became increasingly relaxed, with the result that a lot of bad science inundated the literature from both sides. The most striking example of this sort of corrupted scientific behaviour undoubtedly was the first large scale international trial on HCQ, that had at long last been sponsored by the WHO to provide the final answer to the question of the efficacy of CQ and HCQ in the fight against COVID-19. The trial had involved 96.000 COVID-19 patients in 671 hospitals around the world, and the article that described the results had been submitted at the end of May 2020 to the leading Journal Lancet. The article had been promptly accepted: It had found that the mortality and the cardiac complications were higher in the patients treated with the two drugs that in the untreated controls [31].: as a result, the WHO immediately interrupted the trial, proclaming the inefficacy, and even the dangers, of CQ and HCQ therapy. National Agencies that had previously recommended the use of CQ and HCQ immediately followed suit, and cancelled the recommendation. Was it the end of the story ? Not at all, because the findings in the Lancet paper, and the decision of WHO, started a veritable immediate revolt in the scientific community: 182 specialists, many of them actually “opponents” of HCQ, literally cut the Lancet paper to pieces, showing that it was based on unsupported and fraudulent data provided by a small, relatively unknown private firm, that had somehow been involved in the trial. The reviewers of the journal had inexplicably accepted a paper full of flaws, without noticing that the results had been fabricated from non-existing data. The 182 scientists had written a strong letter to Lancet: as a result, the fraudulent paper had to be retracted by the journal, and by the authorsas well [32], and WHO was forced to resume the trial. Understandably, the scandal had enormous worldwide resonance, which was reinforced by the fact that the results of a number of other less publicized trials on CQ and HCQ had appeared, or were about to appear, around the time of the WHO incident (10 at the end of July 2020) [[33], [34], [35], [36], [37], [38], [39], [40], [41], [42]]. One of these studies [33] had concentrated on the problem of the dosages of HCQ, and had concluded that those used for the prevention of malaria were insufficient to inhibit or suppress the acute respiratory COVID-19 syndrome. Another [34] was an extension -by the same authors-of the original study by Gautret and coworkers [28] that had had the phenomenal worldwide resonance. The study had combined HCQ and azithromycin in the treatment of 80 relatively mildly affected patients. They had detected clinical improvements in all but two of them, and had also detected the disappearance of nasopharyngeal viruses in about 90% of the patients in an 8-days follow up. The study had nevertheless concluded that larger scale studies were necessary, particularly on patients treated before irreversible respiratory symptomatology would become dominant. As for the remaining 8 studies, the conclusions of 4 were unfavourable to HCQ, those of 4 were instead favourable. They differed in a number of aspect, from the dosages of the administered drug, to the stage and the gravity of the disease, to the combination of therapeutic means. Anyway, in spite of these still mixed results, on May 12, 2020 a statement issued by the National Institute of Heath of the USA had concluded that the evidence available at that time was insufficient to suggest the use of HCQ in the treatment of the COVID-19 infection. And a few weeks earlier the Infectious Diseases Society of America had issued a similar statement. These very authoritative statements were widely considered to have put an end to the dispute on the role of chloroquine and hydroxychloroquine in the treatment opf the COVID-19 infection. However, it wasn’t so, since in the months that followed, happenings occurred that changed the picture, and resurrected the interest on CQ and HCQ. On August 19, 2020, in contrast to the statements of the North American Agencies, the New Official Chinese Guidelines for the treatment of COVID-19 infection(http://www.nhc.gov.cn/zygi/s7653p/202008/0a7bdf12bd4b46e5bd28ca7f9a7f5e5a.shtml), recommended the use of CQ. Surprisingly, it did not recommend that of HCQ, alone or in combination with azithromycin: the intriguing CQ-HCQ difference was probably due to the fact that in the preceeding Chinese Guidelines the dosage recommended for CQ were higher than that for HCQ. And then, the results of two studies appeared in August 2020 that offered support to the positive role of HCQ in the treatment of the COVID-19 infection [43,44]. The two studies were observational and retrospective, and had involved a total of more than 10,000 participants: one was an Italian study that had involved 3451 patients from 33 Hospitals, 75% of whom had been treated with low total HCQ doses, and 25% had not received HCQ. The other was a Belgian study that had had 8075 participants, 4542 of whom had received HCQ in low dosage monoterapy, and 3533 were in the no-HCQ group. Both studies had found a significant decrease in the mortality risk (about 30% in the Italian study) in the participants treated with HCQ. These were very positive results, and had been supported by a correct statistical analysis, but a word of caution on them is necessary, since they had not been randomized. However, the large number of patients involved certainly decreased the risk of errors.
One last recent finding, coming from a different research line, could also be mentioned, as it offers a possible additional reason for the controversial findings the CQ-HCQ story has experienced and is still experiencing [45]. The penetration of the COVID-19 into target cells depends on the pH of endosomes, and CQ and HCQ inhibits it by raising it. However, as mentioned above, in some cell types the virus could also use a pH independent pathway for entry into cells. The penetration of COVID-19 into Vero (kidney) cells was found to be dependent on pH dependent proteases, whereas its penetration into Calu-3 cells (lung) depended instead on the pH-independent TMPRSS2 protease.
All considered, then, it is fair to state that the problem of the role of CQ and HCQ in the COVID-19 infection is still open: in keeping with the opening statements of this contribution, chances are that it twill remain open for quite some time, even if hundreds of presumably reliable clinical research trials are currently ongoing. However, if one looks at the matter in a dispassionated way, it is clear that the development of the CQ-HCQ story has had peculiar aspects. The initial enthusiasm triggered by the original observations, which had evident flaws, had certainly been generated by the desperate need to find a cure for the devastating disease. But it had been premature and exaggerated. A couple of general indications could perhaps still be extracted with caution from the data that are presently available in the super-abundant literature. The first is that at late stages of the infection HCQ is probably ineffective; the second has to do with the frequency and seriousness of the described adverse side effects of HCQ, which appear to have been grossly exaggerated. Sooner or later, anyway, the matter of CQ and HCQ must be brought back to the clean realm of science, where the rules of the scientific method, uncorrupted by emotional or ideological deviations, are the only possible option. The request seems reasonable also on theorethical grounds, as it is hard to understand how a drug, which has all the documented mechanisms of action expected of an agent that would successfully fight the virus, could instead be ineffective and even damaging. And it is also necessary to deal with the fact that doctors who see patients at home, or in hospitals at the initial stages of the infection, are generally convinced of the efficacy of HCQ, and defend its use, sometimes even emphatically. Possibly, if one would look at the very early early stages of the disease, the outlook could be different. In this vein, a brief discussion on CQ and HCQ as chemoprophylactic agents rather than as therapeutic means is thus in order.
3. Chloroquine as a chemoprophylactic agent ?
As mentioned, CQ has been the most important therapeutic drug in the control of malaria, particularly of its resurgence after the international ban on dichloro-diphenyl-trichloroethane (DDT), which had practically eliminated the disease by exterminating the Anopheles mosquito. Unfortunately, the use of CQ had been compromised by the appearance of Plasmodium strains resistant to it because of a mutation in the pfcrt gene of the deadliest of them, P. Falciparum (46).Thus, at the end of last century, several countries of Africa, where the malaria is an absolute tragedy, followed a recommendation of WHO and officially discontinued the use of CQ, replacing it with artemisinin. In spite of the official ban, CQ has nevertheless remained the most commonly used antimalarial drug even in the African countries in which the resistance to the Plasmodia had been very substantial. This has been so essentially because the resistance of P. falciparum to CQ rapidly declined, and its sensitivity to CQ re-emerged [47]. An accurate analysis of the household use of chloroquine in sub-Saharan Africa [48] has indeed shown that the consumption of CQ does not reflect the national policies of the African Countries, as it continues to be used at high rates in most of them.
The arrival of the COVID-19 pandemic, and the initial results on the possible beneficial effects on it of CQ and HCQ have changed the scenario dramatically, increasing the demand for the two drugs in most countries. In India, for instance, the Indian Council of Medical Research, under the Ministry of Health and Family Welfare, has recommended prophylaxis with CQ for health care personnel who treat COVID-19 patients, and for asymptomatic household contacts of infected people, generating a rush to CQ and shortages in its market supply [49]. Such market shortages have also been reported in numerous other countries: and it could be added that some African countries have initiated large scale programs to produce CQ [[50], [51]].
The problem of a prophylactic role of CQ and HCQ against the COVID-19 infection targets a time frame that preceeds the necessity of therapy. In a sense, this would extend the observations, made by of most studies, that CQ and HCQ appear to be preferentially effective at the earliest stages of the infection. The idea would of course be compatible with the documented mechanism(s) of action of the two drugs. But could it also have something to do with a question that continues to challenge all COVID-19 specialists: why has COVID-19 so far it sub-Saharan Africa with far less force than in most other Countries ? At the moment the question has no satisfactory answer. Various factors have been considered, from the climate, tothe living patterns, to the nutrition habits, but no convincing evidence supports any of them. Genetics is ruled out, as it has become known that Afro- Americans are particularly susceptible to the COVID-19 infection. This uncertainty is the reason why, in a recent publication [46] we had added the documented widespread use of CQ and HCQ in countries where malaria is endemic, i.e. essentially sub-Saharn Africa, to the list of possible options, and had analyzed the pros and the cons of the hypothesis. We had not taken position, as we had only felt that it would have been useful to consider the problem in a more complete global perspective. A number of colleagues had indeed felt that our analysis had been fair and balanced. One of them, however -a scientist of some repute-felt otherwise, and his comment deserves to be quoted because it is perfectly in line with the statements I had chosen to open this contribution. Since I had written before on CQ and HCQ, he had singled me out from the list of the authors of that paper, and had written “he does not want to surrender to the evidence: he is worse than Trump.” Verbatim!
Declaration of competing interest
I declare no conflict of interest.
Acknowledgements
The author is very grateful to his coworker Andrea Nicoletto for his help in the preparation of Fig. 2.
References
- 1.Francis S.E., Sullivan D.J., Goldberg D.E. Hemoglobinmetabolism in the malaria parasite Plasmodium falciparum. Annu. Rev. Microbiol. 1997;51:97–123. doi: 10.1146/annurev.micro.51.1.97. [DOI] [PubMed] [Google Scholar]
- 2.Maines M.D. Heme oxygenase: function, multiplicity, regulatory mechanisms, and cklinical applications. Faseb. J. 1988;2:2257–2568. [PubMed] [Google Scholar]
- 3.Slater A.F. Malaria Pigment, Exp. Parasitol. 1992;74:362–365. doi: 10.1016/0014-4894(92)90162-4. [DOI] [PubMed] [Google Scholar]
- 4.Slater A.F. Chloroquine: mechanism of drug action and resistance in Plasodium falciparum. Pharmacol. Ther. 1993;56:203–235. doi: 10.1016/0163-7258(93)90056-j. [DOI] [PubMed] [Google Scholar]
- 5.Sullivan D.J., Gluzman I.Y., Russell D.G., Goldberg D.E. On the molecular mechanism of chloroquine’s antimalarial action. Proc. Natl. Acad. Sci. U.S.A. 1996;93:11865–11870. doi: 10.1073/pnas.93.21.11865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Picot S., Peyron F.S., Donaville A., Vuillez J.-P., Barbe G., Ambroise-Thimas P. Chloroquine-induced inhibition of the production of TNF, but not of IL-6, is affected by the disruption of iron metabolism. Immunnology. 1993;80:127–133. [PMC free article] [PubMed] [Google Scholar]
- 7.Ben-Zvi I., Kivity S., Langevitz P., Shoenfeld Y., Hydroxychloroquine From malaria to autoimmunity. Clin. Rev. Allergy Immunol. 2012;42:145–153. doi: 10.1007/s12016-010-8243-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Savarino A., Gennero L., Sperber K., Boelaert J.R. The anti-HIV 1 activity of chloroquine. J. Clin. Virol. 2001;20:131–135. doi: 10.1016/s1386-6532(00)00139-6. [DOI] [PubMed] [Google Scholar]
- 9.Boelaert J.R., Piette J., Sperber K. The potential place of chloroquine in the treatment of HIV-1-infected patients. J. Clin. Virol. 2001;20:137–140. doi: 10.1016/s1386-6532(00)00140-2. [DOI] [PubMed] [Google Scholar]
- 10.Yan Y., Zou Z., Sun Y., Li Y., Xu K._F., Wei Y., Jin N., Jiang C. Anti-malarial drug chloroquine is highly effective in treating avian influenza A H5N1 virus infection in an animal model. Cell Res. 2013;23:300–302. doi: 10.1038/cr.2012.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vincent M.J., Bergeron E., Benjeannet S., Erickson B.R., Rollin P.E., Ksiazek T.G., Seidah N.G., Nichol S.T. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol. J. 2005;2:69:1–10. doi: 10.1186/1743-422X-2-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fantini J., Di Scala C., Hahinian H., Yahi N. Structural and molecular modeling studies reveal a new mechanism of action of chloroquine and hudroxychloroquine against SARS-CoV-2. Int. J. Antimicrob. Agents. 2020;55:105960–105979. doi: 10.1016/j.ijantimicag.2020.105960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Okhuma S., Poole B. Fluorescence probe measurement of the intralysosomal pH in living cells and the perturbation of pH by various agents. Proc. Natl. Acad. Sci. U.S.A. 1978;75:3327–3331. doi: 10.1073/pnas.75.7.3327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yang Z.-Y., Huang Y., Ganesh L., Leung K., Kong W.-P., Schwartz O., Subbarao K., Nabel G.A. pH-dependent entry of severe acute respiratory syndrome coronavirus is mediated by the spike glycoprotein and enhanced by dendritic cell transfer through DC-SIGN. J. Virol. 2004;78:5642–5650. doi: 10.1128/JVI.78.11.5642-5650.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu J., Cao R., Xu M., Wang X., Zhang H., Hu H., Li Y., Hydroxychloroquine Z. Hu. A less toxicderivative of chloroquine , is effective in inhibiting SARS-Co-2 infection in vitro. Cell Discovery. 2020;6:1–4. doi: 10.1038/s41421-020-0156-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cassell S.E., Edwards J., Brown D.T. Effect of lysosomotropic weak bases on infection of BHF-21 cells by Sindbis virus. J. Virol. 1984;52:857–864. doi: 10.1128/jvi.52.3.857-864.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bishop N.E. Examination of potential inhibitors of Hepatitis A virus uncoating. Intervirol. 1998;41:261–271. doi: 10.1159/000024948. [DOI] [PubMed] [Google Scholar]
- 18.Savarino A., Gennero L., Chen H.C., Serrano D., Malavasi F., Boelaert J.R., Sperber K. Anti-HIV effects of chloroquine : mechanism of inhibition and spectrum of activity. AIDS. 2001;15:2221–2229. doi: 10.1097/00002030-200111230-00002. [DOI] [PubMed] [Google Scholar]
- 19.Harley C.A., Dasguota A., Wilson D.W. Characterization of herpes simplex virus-containing organelles by subcellular fractionation: role of organelle acidification in assembly of infectious particles. J. Virol. 2001;75:1236–1251. doi: 10.1128/JVI.75.3.1236-1251.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Perrier A., Bonnin A., Desmarets L., Danneels A., Goffard A., Roullié Y., Dubiuisson J., Belouzard S. The C-terminal domain of the MERS coronavirus Mprotein contains a trans-Golgi network localization signal. J. Biol. Chem. 2019;294:14406–14421. doi: 10.1074/jbc.RA119.008964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Van der Borne B.E., Dijkmans B.A., de Rooij H.H., le Cessie S., Verweij C.L. Chloroquine and hydroxychloroquine equally affect tumor necsis factor alpha, interleukin-6, and interferon-gamma production by peripheral blood mononuclear cells. J. Rheumatol. 1997;24:55–60. [PubMed] [Google Scholar]
- 22.Weniger H. World Health Organization; Geneva: 1979. Review of Side Effects and Toxicity of Chloroquine.https://www.apps.who.int/iris/handle/10665/65773 [Google Scholar]
- 23.Chatre C., Roubille F., Vernhet H., Jorgensen C., Pers Y.-M. Cardiac complications attributed tomchloroquine and hydroxychloroquine: a systematic review of the literature. Drug Saf. 2018;41:919–931. doi: 10.1007/s40264-018-0689-4. [DOI] [PubMed] [Google Scholar]
- 24.Jorge A., Ung C., Young L.H., Melles R.B., K Choi H. Hydroxychloroquine retinopathy-implications of research advances for rheumatology care. Nat. Rev. Rheumatol. 2018;14:693–703. doi: 10.1038/s41584-018-0111-8. [DOI] [PubMed] [Google Scholar]
- 25.Boelaert J.R., Sperber K. Antiretroviral treatment. Lancet. 1998;352:1224–1225. doi: 10.1016/S0140-6736(05)60566-1. [DOI] [PubMed] [Google Scholar]
- 26.The Lancet AIDS, the unbridgable gap. 1998;351:1825. [PubMed] [Google Scholar]
- 27.Wang M., Cao R., Zhang L., Yang X., Liu J., Xu M., Shi Z., Hu Z., Zhong W., Xiao G. Cell Res. 2020;30:269–271. doi: 10.1038/s41422-020-0282-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gautret P., Lagier J.-C., Parola P., Huang V.T., Meddeb L., Mailhe M., Doudier B., Courjon J., Giordanengo V., Vieira V.E., Dupont H.T., Honoré S., Colson P., Chabriere E., La Scola B., Rolain J.-M., Broqui P., Raoult D. Hydroxychloroquineand azithromycin as a treatment of COVID-19: resultsof an open-label non-randomized trial. Int. J. Antimicrob. Agents. 2020;56:105949–105953. doi: 10.1016/j.ijantimicag.2020.105949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kim A.H.J., Sparks J.A., Liew J.W., Putnam M.S., Berenbaum F., Duarte-Garcia A., Graef E.R., Korsten P., Sattui S.E., Sirotich E., Ugarte-Gil M.F., Webb K., Grainger R. A rush to judgement ? Rapid reporting and dissemination of results and its consequences regarding thec use of hydroxychloroquine fr COVID-19. Ann. Intern. Med. 2020 doi: 10.7326/M20-1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.K.A.Pastick, E.C. Okafor, F. Wang, S.M. Lofgren, C.P. Skipper, M.R. Nicol, M.F. Pullen, R. Rajasingham, E.G. McDonald, T.C. Lee, I.S. Schwartz, L.E. Kelly, S.A. Lother, O. Mitià, E. Letang, M. Abassi, D.R. Boulware, Review: hydroxychloroquine and chloroquine for treatmentof SARS-CoV-2 (COVID-19), Open Forum Infectious Dis., doi:10.1093/ofid/ofaa130.eCollection2020Apr.PMID. 32363212M.R. [DOI] [PMC free article] [PubMed]
- 31.Mehra M.R., Desai S.S., Ruschitzka F., Patel A.N. Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis. Lancet. 2020 doi: 10.1016/S0140-6736(20)31180-6(Retracted. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 32.Mehra M.R., Ruschitzka F., Patel A.N. Retraction-hydroxychloroquine or chloroquiunecwith or without a macrolide for treatment of COVID-19: a multinational regiustry analysys. Lancet. 2020 doi: 10.1916/S0140-6736(20)31324-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Al Kofahi M., Jacobson P., Boulware D.R., Matas A., Kandarswamy R., Jaber M.M., Rajasingham R., Young J.-A.H., Nicol M.R. Finding the dose for Hydroxychloroquine prophylaxis for COVID-19: the desperate search for effectiveness. Clin. Pharmcol. Therapheutics. 2020:1–4. doi: 10.1002/cpt.1874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gautret P., Lagier J.-C., Parola P., Huang V.T., Meddeb L., Mailhe M., Doudier B., Aubry C., Amrane S., Seng P., Hocquart M., Eldin C., Finance J., Vieira V.E., Tissot H., Dupont H.T., Honoré S., Stein A., Million M., Colson P., La Scola B., Veit V., Jacquier A., Deharo J.-C., Drancourt M., Fournier P.E., Rolain J.-M., Broqui P., Raoult D. Clinical and microbiological effect of combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six –day follow up: a pilot ibservational study. Trav. Med. Infect. Dis. 2020;34:101663. doi: 10.1016/j.tmaid.2020.101663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Boulware D.R., Pullen M.F., Bangdiwala A.S., Pastick K.A., Lofgren S.M., Okafor E.C., Skipper C.P., Nascene A.A., Nicol M.R., Abassi M., Engen N.W., Cheng M.P., La Bar D., Lother S.A., MacKenzie L.J., Drobot G., Marten N., Zarychanski R., Kelly L.E., Schwartz I.S., McDonald E.G., Rajasingham R., Lee T.C., Hullsiek K.H. A randomized trialmof hydroxychloroquine aspostexposure prophylaxis for COVID-19. N. Engl. J. Med. 2020 doi: 10.1056/NEIMoa2016638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yu B., Li C., Cheng P., Zhou N., Wang L., Li J., Jiang H., Wang D.-W. Low Dose of Hydroxychloroquine Reducesfatality of Critically Ill Patientswith COVID-19. Sci China, Life Sci. 2020 doi: 10.1007/s11427-020-1732-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rosenberg E.S., Dufort E.M., Udo T., Wilbersfied L.A., Kumar J., Tesoriero J., Weinberg P., Kirkwood J., Muse A., DeHowitz J., Blog D.S., Hutton B., Holtgrave D.R., Zicker H. Association of treatment with hydroxychloroquine or azithromycin with in- hospital mortality in patients with COVID-19 in New York State. J. Am. Med. Assoc. 2020;323:2493–2502. doi: 10.1001/jama.2020.8630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Million M., Lagier J.-C., Gautret P., Colson P., Fournier P.E., Amrane S., Hocquart M., Mailhe M., Esteves-Vieira V., Doudier B., Aubry C., Correard F., Giraud-Gatineau A., Roussel Y., Berenger C., Cassir N., Seng P., Zandotti C., Dhiver C., Ravaux I., Tomei C., Eldin C., Tissot-Dupont H., Honoré S., Stein A., Jacquier A., Deharo J._C., Chabrière S., Levasseur A., Fenollar F., Rolain J.-M., Obadia Y., Broqui P., Drancourt M., La Scola B., Parola P., Raoult D. Early treatment of COVID-19 patients with hydroxychloroquine and azithromycin: a retrospective analysis of 1061cases in Marseille, France. Trvel Med. Infect. Disease. 2020;35:101738. doi: 10.1016/j.tmaid.2020.101738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.M.G. Silva-Borba, F.F. Almeida-Val, V. Souzasampaio, M. Almeida Arauio Alexandre, G. Cardoso Melo, M. Brito, M.P. Gomes-Mourao, J. D. Brito-Sousa, D. Baia-da-Silva, M.V. Farias-Guerra, L. Abracao Hajjar, R. Costa Pinto, A. Alcirley Silva Balieiro, A.G. Fonseca Pacheco, J. D. Oliveira Santos, F. Gomes Naveca, M. Simao Xavier, A. Machado Siqueira A. Schwartzbold, J. Croda, M. Lacerda Nogueira, G.A. Sierra Romero, Q. Bassat, C.J. Fontes. B. C. Albuquerque, C-T. Daniel-Ribeiro, W. M. Monteiro, M.V. Guimaraes Lacerda, J. Am. Med. Assoc..Doi 10.1001/jamanetworkopen2020.8857
- 40.J. Geleris, Y. Sun, J. Platt, J. Zucker, M. Baldwin, G. Hripcsak, A. Labella, D.K. Manson, C. Kubin, D.R. Graham Barr, M.E. Sobieszkzyk, N.W. Schluger, Observational study of hydroxychloroquine in hospitalizedpatients with COVID-19, NewEngl. J. Med. DOI: 10.1056/NEIMoa2012410 [DOI] [PMC free article] [PubMed]
- 41.Molina J.M., Delaguerre C., Le Goff J., Mela-Lima B., Ponscarme D., Goldwirt L., de Castro N. No evidence of rapid antiviral clearance or clinical benefit with the combinaation of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection. Médecine et maldies infectieuses. 2020;50:384. doi: 10.1016/j.medmal.2020.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Arshad S., Kilgore P., Chaudhry Z., Jacobsen G., Wang D.D., Huitsing K., Brar I., Alangaden G.J., Ramesh M.S., McKinnon J.E., O’Neillm W., Zervos M., Ford Henry. COVID-19 Task Force,Treatment with hydroxychloroquine, axithromycin, and combination inn patients hospitalized with COVID-19. Int. J. Infect. Dis. 2020;97:396–403. doi: 10.1016/j.ijid.2020.06.099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Iacoviello L. 101 collaboration authors of the COVID-19 Risk and Treatments (CORIST), Use of hydroxychloroquine in hospitalized patients is associated with reduced mortality:Findings from the observational multicentre Italian CORIST study. Eur. J. Intern. Med. 2020 doi: 10.1016/j.ejim.2020.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Catteau L., Dauby N., Montourcy M., Bottieau E., Hautekiet J., Goetghebeur E., Van Ierssel S., Duysburg E., Van Oyen H., Wyndham-Thomas C., Van Beckhoven D. Low-dose hydroxychloroquine therapy and mortalitynin hospitalized patients with COVID-19: a nationwide observational study on 8075 paricipants. J. Antimicrob. Agents. 2020 doi: 10.1016/j.ijantimicag.2020.106144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hoffmann M., Moesbauer K., Hofmann-Winkler K., Kaul A., Kleine-Weber H., Krueger N., Gassen N.C., Mueller M.A., Drosten C., Poehlmann S. Chloroquine does not inhibit infection ofv human lung cells with SARS-CoV-2. Nature. 2020 doi: 10.1038/s41586-020-2575-3. [DOI] [PubMed] [Google Scholar]
- 46.Djimde A., Doumbo O.K., Cortese J.F., Kayentao K., Doumbo S., Dicko Y. Diourte A., Su X.Z., Nomura T., Fidock D.A., Wellems T.E., Plowe C.V., Koulibaly D. A molecular marker for chloroquine-resistant falciparum malaria. N. Engl. J. Med. 2001;344:257–263. doi: 10.1056/NEJM200101253440403. [DOI] [PubMed] [Google Scholar]
- 47.Sharma A., Sharma S.K., Shi Y., Bucci E., Carafoli E., Bhattacherjee G. MelinoA., Das G. BCG vaccinatio policy and preventive chloroquine usage: do they have an impat on COVID-19 pandemic ? Cell Death Dis. 2020;11:516. doi: 10.1038/s41419-020-2721-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Frosch A.E.P., Venkatesan M., Laufer M.K. Pattern of chloroquine use and resistance in sub-Saharan Africa: a systematic review of household survey and moleclar data. Malar. J. 2011;10:116–126. doi: 10.1186/1475-2875-10-116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rathy S., Ish P., Kalantri P., Kalantri S. Hydroxychloroquinecprophylaxis for COVID-19 in India. Lancet Infect. Dis. 2020 doi: 10.1016/s1473-3099(20)30313-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.African rush for chloroquine as coronavirus tsunami looms. https://www.scmp.com/news/worl/africa/article/3078039/africans-rush-chloroquine-coronavirus-tsunami-looms (Accessed on)
- 51.Cameroon begins large-scale chloroquine production. https://www.woanews.com/science-health/coronavirus-outbreak/cameroon.begins.large-scale chloroquine production Accessed on.