Abstract
Several studies have described unusually high incidence of vascular thrombosis in coronavirus disease-2019 (COVID-19) patients. Pathogenesis of the vascular thrombosis in COVID-19 is least understood for now and presents a challenge to the treating physicians. Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), the causative pathogen for COVID-19, has been shown to bind to angiotensin converting enzyme 2 (ACE2) protein in human epithelial cells which facilitates its entry in the organ and mediate tissue specific pathogenesis. For ACE2 mediated cell entry of the SARS-CoV-2, co-expression of one more protein—Transmembrane protease serine 2 (TMPRSS2) is essential. Existing studies suggested significant expression of ACE2 and TMPRSS2 in human vascular endothelium. Vascular endothelial dysfunction can potentially activate coagulation cascade eventually resulting in thrombosis. ACE2 has proven role in the maintenance of endothelial integrity inside the vessels. Existing in situ evidence for SARS-CoV-1 (the causative agent for SARS pandemic of 2002, which shared ACE2 as cell entry receptor) suggested that virus binding can downregulate ACE2, thus can induce endothelial dysfunction. Recently, in situ evidence has been presented that SARS-CoV-2 can infect cells in engineered human vascular endothelium, which can be effectively blocked by using clinical-grade recombinant human ACE2. Based on the circumstantial evidence present in the literature, we propose a SARS-CoV-2 cell entry receptor ACE2 based mechanism for vascular thrombosis in COVID-19 patients.
Keywords: SARS-CoV-2, COVID-19, ACE2, TMPRSS2, Thrombosis, Vascular endothelial dysfunction
Introduction
Several studies have described high incidence of vascular thrombosis in coronavirus disease-2019 (COVID-19) patients [1], [2], [3]. Elevated levels of prothrombotic markers including D-dimer and fibrin degradation products (FDPs), thrombocytopenia, abnormalities in prothrombin time and partial thromboplastin time are among the frequent findings in COVID-19 patients [2] (Table 1 ), especially those with severe symptoms and in ICU admission [1], [2], [3], [4]. Though, probability of vascular thrombosis remains relatively high in ICU admitted patients, in COVID-19 it has been found to be unusually high [1], [3]. Pathogenesis of the vascular thrombosis in COVID-19 is rather obscure and hence management of such cases presents a considerable challenge to the treating physicians [1].
Table 1.
A literature study of blood coagulation markers status in COVID-19 patients*.
| Blood coagulation markers | Fan et al1 (n = 67) | Wang et al2 (n = 26) | Chen et al3 (n = 99) | Huang et al4 (n = 40) | Peng et al5 (n = 58) | Chan et al6 (n = 150) | Ming et al7 (n = 23) | Yan et al8 (n = 9) | Dan et al9 (n = 30) | Qiu et al10 (n = 153) | Shi et al11 (n = 109) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Platelets (×109/L; normal range 100.0–300.0) | 185 (148–259) | 191.0 (156.8 – 250.3) | 213·5 | 164·5 (131·5–263·0) | – | – | 176 (91–292) | 187 (137.56–236.44) | 251.90 (170.1–333.6) | – | – |
| Increased in | 49 (73.1%) | 4 (15.4%) | 4 (4%) | 2 (5%) | – | – | 22 (95.7%) | – | – | – | – |
| Decreased in | – | – | 12 (12%) | 38 (95%) | – | – | 1 (4.3%) | – | – | – | – |
| Prothrombin time (s; normal range 11.0–14.0) | – | 12.6 (12.2–13.1) | 11·3 (9.4–13.2) | 11·1 (10·1–12·4) | – | 11.5 (10.1–12.9) | 18.6 (12.7–20.8) | – | 11.06 (10.12–12) | – | – |
| Increased in | – | 2 (7.7%) | 5 (5%) | – | – | 2 (1%) | – | – | – | – | – |
| Decreased in | – | 1 (3.9%) | 30 (30%) | – | – | – | – | – | – | – | – |
| Activated partial thromboplastin time (s; normal range 22.0–38.0) | – | 28.7 (25.5–33.5) | 27·3 (10·2) | 27·0 (24·2–34·1) | – | – | 36.5 (23.3–58.0) | – | 25.39 (19.21–31.57) | – | – |
| Increased in | – | 2 (7.7%) | 6 (6%) | – | – | – | – | – | – | – | – |
| Decreased in | – | 2 (7.7%) | 16 (16%) | – | – | – | – | – | – | – | – |
| D-dimer (μg/mL; normal range 0.0–1.0) | – | 0.3 (0.2–0.4) | 0·9 (0·5–2·8) | 0·5 (0·3–1·3) | 0·45 (0·3–1·3) | – | 0.50 (0.30–1.00) | – | 0.68 (0.74) | 0.22 (0.16–0.37) | 1.89 |
| Increased in | – | 1 (3.9%) | 36 (36%) | – | 45 (77.5%) | – | – | – | – | – | – |
| Decreased in | – | – | – | – | – | – | – | – | – | – | – |
*References provided as supplementary information. Parameters primarily indicating prothrombosis have been marked in red.
Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), the causative pathogen for COVID-19 has been shown to bind to angiotensin converting enzyme 2 (ACE2) protein in human epithelial cells, which facilitates its entry in the organ and mediate tissue specific pathogenesis [5], [6]. ACE2 is also known as a receptor for SARS-CoV-1 (causative agent for SARS epidemic of 2002) and human coronavirus NL63 (HCoV-NL63) [7]. For ACE2 mediated cell entry of the SARS viruses, co-expression of one more protein—Transmembrane protease serine 2 (TMPRSS2) is essential [5]. In this article, we propose a SARS-CoV-2 cell entry receptor ACE2 based mechanism for vascular thrombosis in COVID-19 patients.
Hypothesis
SARS-CoV-2 binding to the cell entry receptor ACE2 downregulates receptor expression that in turn induces vascular endothelial dysfunction, which activates prothrombotic cascade and eventually leads to vascular thrombosis observed in COVID-19 patients.
Evaluation of the hypothesis
SARS-CoV-2 cell entry receptor ACE2 and entry associated receptor TMPRSS2 are co-expressed across various tissue and cell types in humans [5]. ACE2 expression is known in endothelial cells of human blood vessels and microvasculature [8], [9]. Though, studies are limited, TMPRSS2 expression has been noted in human vascular endothelium [10], [11]. ACE2 is an analogue of angiotensin converting enzyme (ACE)—a key regulator of renin angiotensin system regulating hemodynamic homeostasis in body [12]. An ACE/ACE2 balance appears to be crucial in maintaining the RAS and a dysregulation of this ratio is associated with vascular thrombosis [13], [14]. An ACE/ACE2 balance is also critical for maintenance of endothelial integrity in vessels, a breach of which can potentially induce thrombosis [15], [16]. Although, any SARS-CoV-2 specific evidence is lacking for now, existing in vitro studies for SARS-CoV1 and HCoV-NL63 binding on ACE2 in lung tissue indicate that virus binding downregulates ACE2 (but not ACE) creating ACE/ACE2 imbalance and thereby promotes tissue injury which potentially can activate prothrombotic cascades inside the vessels [7], [17]. A lower ACE/ACE2 ratio in the vascular endothelium prevents prothrombotic cascade from activation by catalyzing degradation of angiotensin II to angiotensin 1–7. Angiotensin 1–7 also induces antithrombotic effects through binding to G-protein coupled Mas receptors [18]. Conversely, a higher ACE/ACE2 ratio will allow binding of angiotensin II to AT1 receptors which will induce vasoconstriction, inflammation, and eventually thrombosis [15], [18]. SARS-CoV-2 mediated ACE2 downregulation in vascular endothelium also can activate the kallikrein–bradykinin pathway inducing platelet aggregation and leaking of the vessels which can further contribute to thrombosis [15], [19].
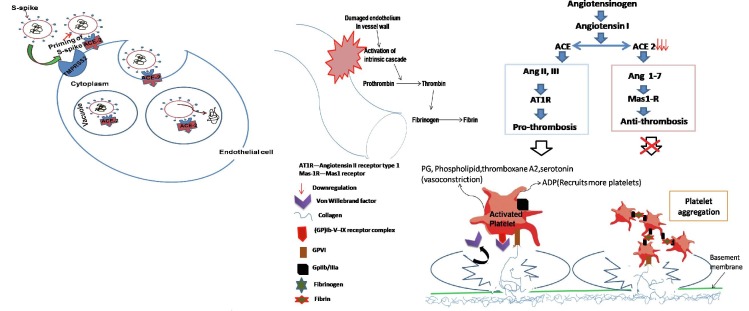
Fig. 1 gives a schematic description of proposed mechanism for ACE2 mediated dysfunction of vascular endothelium leading to thrombotic incidences in COVID-19 patients.)
Fig. 1.
Schema of ACE2 mediated mechanisms of vascular thrombosis in COVID-19 patients. (Binding of SARS-CoV-2 to ACE2 receptor present at the vascular endothelial cell surface leads internalization and replication of the virus inside the cell, and consequently cellular disintegration which activates prothrombotic cascade. Additionally, SARS-CoV-2 binding induces downregulation of ACE2 inturn imbalances ACE/ACE2 ratio favoring prothrombosis. Both of these stated mechanisms in consequence also induce activation and aggregation of the platelets, altogether culminating in intravascular thrombosis.)
Empirical data
Studies describing the effect of SARS-CoV-2 binding on ACE2 signaling in vessels are lacking. Knock-out of ACE2 gene in mice was found to induce vascular endothelial dysfunction leading to release of chemokines and pro-inflammatory markers [20]. ACE2 knock-out mediated release of chemokine like CCL2 and other pro-inflammatory cytokines and interleukins are known to induce platelet aggregation and adhesion to the vascular endothelium, which consequently leads to increased propensity for thrombosis [20], [21]. Conversely, activated ACE2 has been found to protect against vascular thrombosis [14]. SARS-CoV-2 infection of vascular endothelium is mediated by ACE2 was proven by a recent study. Monteil et al provided in situ evidence that SARS-CoV-2 can infect engineered human vessels which can be prevented by application of clinical grade recombinant human ACE2 [8].
Recent studies noted remarkable similarities in clinical presentations in SARS and COVID-19 (apart from sharing of the virus entry receptor). Cases of vascular thrombosis were also reported in SARS patients [22]. SARS-CoV-1:ACE2 binding analogy to COVID-19 can explain the pathogenesis of vascular thrombosis with articulate coherence.
ACE2 is secreted in blood and expresses in the endothelium of the small vessels [9], [23], [24]. The histopathological examination of autopsy tissues in SARS, and now in COVID-19, showed presence of the virus and cellular damage in ACE2 expressing tissues [22], [25], [26]. Damaged tissue also displays presence of pro-inflammatory markers and cytokines, (e.g. C-reactive protein, D-dimer, ferritin and interleukin-6), which can induce thrombosis [1], [22], [27]. Viral inclusion structures, an accumulation of inflammatory cells associated with endothelium, as well as apoptotic bodies were evidenced in vascular endothelial cells of multiple organs in autopsied patients [28], indicating for the widespread endothelial dysfunction as the etiology for the vascular thrombosis in COVID-19 [29].
Consequences of the hypothesis and discussion
Widespread thrombosis, more specifically in microvasculature, was found present in majority of the autopsied patients [30]. In some cases, thrombosis was observed despite full anticoagulation and regardless of timing of the disease course of the duration of onset of the disease, which indicated that it could be a key pathology in COVID-19 [30]. Thrombus rich in megacaryocytes and platelets was detected in lung, and many other viscera like liver, heart, kidney, and brain, and also in skeletal tissue like bone [30]. Pulmonary vessels were found most severely affected where distinctive vascular features, consisting of severe endothelial injury associated with the presence of intracellular virus and disrupted cell membranes, and perivascular T-cell infiltration were observed [31]. Presence of vascular thrombosis was associated with multi organ injury and higher fatality. Formations of clots were unusually high in patients who were critical and admitted in ICU. Clinically, early appearances of thrombotic symptoms predicted a more severe course of disease [30]. Uniquely, purple or red lesions in toes and skin rashes have been reported in some patients [27], which are supposedly caused by inflammation of the dermal micro-vessels and are complement system mediated [25].
The optimum conditions leading to thrombosis like presence of proinflammatory markers, reduced clotting time, activated platelets and fibrin degradation products, and complement cascades were observed in the blood samples of COVID-19 patients (Table 1). Existing studies for SARS and MERS, and now emerging evidence from autopsy studies in COVID-19 provide clear indications that increased thrombosis is propagated by virus mediated tissue injury [28], [31]. An early presence of vascular thrombosis at systemic scale can be appropriately explained by ACE2 mediated dysfunction of vascular endothelium and consequent activation of prothrombosis cascade [15], [18], [29]. SARS-CoV-2 binding mediated downregulation of ACE2 signaling in vascular endothelium and consequent imbalance of ACE/ACE2 ratio and dysregulation of RAS, can potentially activate prothrombotic cascade at a scale matched to that observed in COVID-19 [15], [18], [29] (Fig. 1).
Other than SARS-CoV-2 induced vascular endothelial dysfunction ACE2 mediated injury of liver tissue can also contribute to vascular thrombosis. Liver cell injury and functional impairment are usual features in COVID-19 patients [32]. Owing to low expression of ACE2 in hepatocytes, it is not clear if liver cell injury is mediated through this molecule; howbeit, high expression of ACE2 (and TMPRSS2) is known in common bile duct and gallbladder epithelium [5]. Literature suggests that acute liver injury should be considered as an independent risk factor for developing vascular thrombosis [33].
Clinical outcomes in COVID-19 were found depending on multiple factors specific to the virus and host [34], that may apply to thrombotic incidences as well. Apart from direct SARS-CoV-2 inflicted tissue injury, vascular thrombosis can also be a resultant of cell toxicity by viral proteins, pro-inflammatory markers released after viral tissue injury, sepsis, or cellular stress caused by critical illness [35]. Additionally, host specific factors like old age and associated comorbid conditions which induce endothelial dysfunction like diabetes, hypertension, obesity, coronary artery diseases, atherosclerosis, chronic liver, renal or lung diseases, cancers, pregnancy, disorders related to clotting factors, and genetic predisposition (which are known risk factors for increase in vascular thrombosis) [15], [16], [36], or auto-immune reactions against viral protein component or viral receptor, virus-receptor complex [37], may have contributory roles in thrombotic incidences in COVID-19 patients.
Currently, the therapeutic approaches which are being suggested to prevent vascular thrombosis in COVID-19 patients are primarily directed at inhibition of coagulation cascade at certain steps or preventing thrombus formation [38]. Chief anticoagulants which are being used are low molecular weight heparin (LMWH) as prophylaxis and treatment, unfractionated heparin (UH) where disseminated intravascular coagulation (DIC) is threatened, or fibrinolytics like tissue plasminogen activation (tPA) inhibitors (as a salvage therapy). Anti-thrombotic drugs primarily being used are that inhibiting platelet aggregation and glycoprotein IIb/IIIa receptor [38]. Existing evidence for the antiviral properties of heparin against coronaviruses like SARS-CoV-1 and human coronavirus NL63 (HCoV-NL63) suggested comparative benefits of using this (however any direct proof for the antiviral property of heparin against SARS-CoV-2 is lacking for now) [39], [40].
Targeting SARS-CoV-2 binding to ACE2 receptor may potentially prevent endothelial dysfunction and consequent initiation of thrombotic cascade [41], however, application of this approach in COVID-19 is scarce in literature. In vitro/in vivo testing of the molecules which can counter SARS-CoV-2 mediated downregulation of ACE2 and consequently endothelial dysfunction, e.g., clinical grade recombinant human ACE2 [8] or an ACE2 activator [42] may signify therapeutic implications of our hypothesis. (Diminazene aceturate (DIZE), an ACE2 activator [42], is currently being used as an anti-protozoan veterinary drug (an active ingredient of ‘Berenil’), however, in clinically effective doses it has serious toxicity risk for vital organs such as liver and kidney, and more particularly for the central nervous system (CNS) and heart. Adequate safety testing, and determination of safe dose, in human subjects, will be necessary, before it can be repurposed in COVID-19 patients.)
Based on the circumstantial evidence present in the literature, we propose that thrombosis in organ vasculature and dermal micro-vessels in COVID-19 are primarily caused by virus binding mediated dysregulation of ACE2 signaling in vascular endothelium and consequent cellular injury and activation of hemostatic factors. However, contributory role of other prothrombotic factors (related to both virus and host) cannot be denied.
Further in vivo/in vitro studies investigating SARS-CoV-2 binding influence on ACE2 signaling and consequently on molecular cascade involved in clotting in vascular endothelium will be necessary to establish ACE2 based mechanisms in thrombotic incidences in COVID-19.
Authors contribution
A.K. designed research. A.K., R.K.N., and C.K. analyzed data. A.K., M.A.F., M.K., K.K., and V.P. wrote the paper.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.mehy.2020.110320.
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.Bikdeli B., Madhavan M.V., Jimenez D., Chuich T., Dreyfus I., Driggin E. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(23):2950–2973. doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ranucci M, Ballotta A, Di Dedda U. The procoagulant pattern of patients with COVID-19 acute respiratory distress syndrome [published online ahead of print 17 April 2020]. J Thromb Haemost. [DOI] [PMC free article] [PubMed]
- 3.Klok F.A., Kruip M.J., Meer N.J. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020 doi: 10.1016/j.thromres.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tang N., Li D., Wang X., Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18(4):844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sungnak W, Huang N, Bécavin C, Berg M, Queen R, Litvinukova M, Talavera-López C, Maatz H, Reichart D, Sampaziotis F, Worlock KB. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nature medicine. 2020 May;26(5):681-7.https://doi.org/10.1038/s41591-020-0868-6. [DOI] [PMC free article] [PubMed]
- 6.Song W, Gui M, Wang X, Xiang Y. Cryo-EM structure of the SARS coronavirus spike glycoprotein in complex with its host cell receptor ACE2. PLoS pathogens. 2018 Aug 13;14(8):e1007236. https://doi.org/10.1371/journal.ppat.1007236. [DOI] [PMC free article] [PubMed]
- 7.Glowacka I., Bertram S., Herzog P., Pfefferle S., Steffen I., Muench M.O. Differential downregulation of ACE2 by the spike proteins of severe acute respiratory syndrome coronavirus and human coronavirus NL63. J Virol. 2010;84(2):1198–1205. doi: 10.1128/jvi.01248-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Monteil V., Kwon H., Prado P., Hagelkrüys A., Wimmer R.A., Stahl M. Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell. 2020 doi: 10.1016/j.cell.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zulli A., Burrell L.M., Buxton B.F., Hare D.L. ACE2 and AT4R are present in diseased human blood vessels. Eur J Histochem. 2008;39–44 doi: 10.4081/1184. [DOI] [PubMed] [Google Scholar]
- 10.Bertram S, Heurich A, Lavender H, Gierer S, Danisch S, Perin P, Lucas JM, Nelson PS, Pöhlmann S, Soilleux EJ. Influenza and SARS-coronavirus activating proteases TMPRSS2 and HAT are expressed at multiple sites in human respiratory and gastrointestinal tracts. PloS one. 2012 Apr 30;7(4):e35876.https://doi.org/10.1371/journal.pone.0035876. [DOI] [PMC free article] [PubMed]
- 11.Muus C., Luecken M.D., Eraslan G., Waghray A., Heimberg G., Sikkema L. Integrated analyses of single-cell atlases reveal age, gender, and smoking status associations with cell type-specific expression of mediators of SARS-CoV-2 viral entry and highlights inflammatory programs in putative target cells. BioRxiv. 2020 doi: 10.1101/2020.04.19.049254. [DOI] [Google Scholar]
- 12.Santos R.A., Ferreira A.J., Verano-Braga T., Bader M. Angiotensin-converting enzyme 2, angiotensin-(1–7) and Mas: new players of the renin-angiotensin system. J Endocrinol. 2013;216(2):R1–R7. doi: 10.1530/JOE-12-0341. [DOI] [PubMed] [Google Scholar]
- 13.Qaradakhi T., Gadanec L.K., McSweeney K.R., Tacey A., Apostolopoulos V., Levinger I. The potential actions of angiotensin-converting enzyme II (ACE2) activator diminazene aceturate (DIZE) in various diseases. Clin Exp Pharmacol Physiol. 2020;47(5):751–758. doi: 10.1111/1440-1681.13251. [DOI] [PubMed] [Google Scholar]
- 14.Fraga-Silva R.A., Sorg B.S., Wankhede M., deDeugd C., Jun J.Y., Baker M.B. ACE2 activation promotes antithrombotic activity. Mol Med. 2010;16(5):210–215. doi: 10.2119/molmed.2009.00160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Teuwen L.A., Geldhof V., Pasut A., Carmeliet P. COVID-19: the vasculature unleashed. Nat Rev Immunol. 2020;21:1–3. doi: 10.1038/s41577-020-0343-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pober J.S., Sessa W.C. Evolving functions of endothelial cells in inflammation. Nat Rev Immunol. 2007;7(10):803–815. doi: 10.1038/nri2171. [DOI] [PubMed] [Google Scholar]
- 17.Kuba K., Imai Y., Rao S. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med. 2005;11(8):875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Verdecchia P., Cavallini C., Spanevello A., Angeli F. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur J Int Med. 2020 doi: 10.1016/j.ejim.2020.04.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ottaiano T.F., Andrade S.S., de Oliveira C., Silva M.C., Buri M.V., Juliano M.A. Plasma kallikrein enhances platelet aggregation response by subthreshold doses of ADP. Biochimie. 2017;1(135):72–81. doi: 10.1016/j.biochi.2017.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Thomas M.C., Pickering R.J., Tsorotes D., Koitka A., Sheehy K., Bernardi S. Genetic Ace2 deficiency accentuates vascular inflammation and atherosclerosis in the ApoE knockout mouse. Circ Res. 2010;107(7):888–897. doi: 10.1161/CIRCRESAHA.110.219279. [DOI] [PubMed] [Google Scholar]
- 21.Chen I.Y., Chang S.C., Wu H.Y., Yu T.C., Wei W.C., Lin S. Upregulation of the chemokine (CC motif) ligand 2 via a severe acute respiratory syndrome coronavirus spike-ACE2 signaling pathway. J Virol. 2010;84(15):7703–7712. doi: 10.1128/jvi.02560-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ding Y., Wang H., Shen H., Li Z., Geng J., Han H. The clinical pathology of severe acute respiratory syndrome (SARS): a report from China. J Pathol. 2003;200(3):282–289. doi: 10.1002/path.1440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li J., Gao J., Xu Y.P., Zhou T.L., Jin Y.Y., Lou J.N. Expression of severe acute respiratory syndrome coronavirus receptors, ACE2 and CD209L in different organ derived microvascular endothelial cells. Zhonghua yi xue za zhi. 2007;87(12):833–837. [PubMed] [Google Scholar]
- 24.Blood atlas - ACE2 - The Human Protein Atlas. https://www.proteinatlas.org/ENSG00000130234-ACE2/blood (accessed April 29, 2020).
- 25.Magro C., Mulvey J.J., Berlin D. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: A report of five cases. Transl Res. 2020;220:1–13. doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang H., Zhou P., Wei Y., Yue H., Wang Y., Hu M. Histopathologic Changes and SARS–CoV-2 Immunostaining in the Lung of a Patient With COVID-19. Ann Intern Med. 2020 doi: 10.7326/M20-0533. [DOI] [PubMed] [Google Scholar]
- 27.Zhang Y., Cao W., Xiao M. Clinical and coagulation characteristics of 7 patients with critical COVID-2019 pneumonia and acro-ischemia. Zhonghua Xue Ye Xue Za Zhi. 2020 doi: 10.3760/cma.j.issn.0253-2727.2020.0006. [DOI] [PubMed] [Google Scholar]
- 28.Varga Z., Flammer A.J., Steiger P. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395(10234):1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Huertas A., Montani D., Savale L., Pichon J., Tu L., Parent F. Endothelial cell dysfunction: a major player in SARS-CoV-2 infection (COVID-19)? Eur Respir J. 2020;2001634 doi: 10.1183/13993003.01634-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rapkiewicz A.V., Mai X., Carsons S.E., Pittaluga S., Kleiner D.E., Berger J.S. Megakaryocytes and platelet-fibrin thrombi characterize multi-organ thrombosis at autopsy in COVID-19: A case series. E Clin Med. 2020 doi: 10.1016/j.eclinm.2020.100434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ackermann M., Verleden S.E., Kuehnel M., Haverich A., Welte T., Laenger F. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. 2020 doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhang C., Shi L., Wang F.S. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5(5):428–430. doi: 10.1016/S2468-1253(20)30057-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kerr R., Newsome P., Germain L., Thomson E., Dawson P., Stirling D. Effects of acute liver injury on blood coagulation. J Thromb Haemost. 2003;1:754–759. doi: 10.1046/j.1538-7836.2003.00194.x. [DOI] [PubMed] [Google Scholar]
- 34.Zhang X., Tan Y., Ling Y. Viral and host factors related to the clinical outcome of COVID-19. Nature. 2020;583(7816):437–440. doi: 10.1038/s41586-020-2355-0. [DOI] [PubMed] [Google Scholar]
- 35.Goeijenbier M., van Wissen M., van de Weg C., Jong E., Gerdes V.E.A., Meijers J.C.M. Review: viral infections and mechanisms of thrombosis and bleeding. J Med Virol. 2012;84(10):1680–1696. doi: 10.1002/jmv.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Previtali E., Bucciarelli P., Passamonti S.M., Martinelli I. Risk factors for venous and arterial thrombosis. Blood Transfus. 2011;9(2):120–138. doi: 10.2450/2010.0066-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Amiral J., Vissac A.M., Seghatchian J. Covid-19, induced activation of hemostasis, and immune reactions: can an auto-immune reaction contribute to the delayed severe complications observed in some patients? Transfus Apher Sci. 2020;59(3) doi: 10.1016/j.transci.2020.102804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.McFadyen J.D., Stevens H., Peter K. The emerging threat of (Micro)thrombosis in COVID-19 and its therapeutic implications. Circ Res. 2020 doi: 10.1161/CIRCRESAHA.120.317447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lang J., Yang N., Deng J. Inhibition of SARS pseudovirus cell entry by lactoferrin binding to heparan sulfate proteoglycans. PLoS ONE. 2011;6(8) doi: 10.1371/journal.pone.0023710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Milewska A., Zarebski M., Nowak P., Stozek K., Potempa J., Pyrc K. Human coronavirus NL63 utilizes heparan sulfate proteoglycans for attachment to target cells. J Virol. 2014;88:13221–13230. doi: 10.1128/JVI.02078-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jung F., Krüger-Genge A., Franke R.P., Hufert F., Küpper J.H. COVID-19 and the endothelium. Clin Hemorheol Microcirc. 2020;75(1):7–11. doi: 10.3233/CH-209007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Castardeli C., Sartório C.L., Pimentel E.B., Forechi L., Mill J.G. The ACE 2 activator diminazene aceturate (DIZE) improves left ventricular diastolic dysfunction following myocardial infarction in rats. Biomed Pharmacother. 2018;107:212–218. doi: 10.1016/j.biopha.2018.07.170. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.