Abstract
Roles of environmental factors in transmission of COVID-19 have been highlighted. In this study, we sampled the high-touch environmental surfaces in the quarantine room, aiming to detect the distribution of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) on the environmental surfaces during the incubation period of coronavirus disease 2019 (COVID-19) patients. Fifteen sites were sampled from the quarantine room, distributing in the functional areas such as bedroom, bathroom and living room. All environmental surface samples were collected with sterile polyester-tipped applicator pre-moistened in viral transport medium and tested for SARS-CoV-2. Overall, 34.1% of samples were detected positively for SARS-CoV-2. The positive rates of Patient A, B and C, were 46.2%, 0% and 61.5%, respectively. SARS-CoV-2 was detected positively in bedroom and bathroom, with the positive rate of 50.0% and 46.7%, respectively. In contrast, living room had no positive sample detected. Environmental contamination of SARS-CoV-2 distributes widely during the incubation period of COVID-19, and the positive rates of SARS-CoV-2 on environmental surfaces are relatively high in bathroom and bedroom.
Keywords: Coronavirus disease 2019 (COVID-19), Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), Environmental contamination, Incubation period
Highlights
-
•
This study reports detection of SARS-CoV-2 on the high-touch surfaces of dwelling environment.
-
•
Environmental contamination of SARS-CoV-2 distributes widely during the incubation period.
-
•
The positive rates of SARS-CoV-2 on environmental surfaces are relatively high in bathroom and bedroom.
1. Introduction
Since a novel human coronavirus, named as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was first detected in Wuhan, China, in late 2019, the coronavirus disease 2019 (COVID-19) caused by this virus has been reported drastically all over the world. As of May 25, 2020, about 5,408,301 confirmed COVID-19 cases including 345,064 deaths have been reported in 188 countries (Johns Hopkins University of Medicine (JHU), 2020b). So far, compared with severe acute respiratory syndrome coronavirus 1 (SARS-CoV-1) and Middle East respiratory syndrome coronavirus (MERS-CoV), a relatively low fatality (about 7%) has been summarized from SARS-CoV-2 infected cases, however, it is obvious that SARS-CoV-2 is much more contagious as evidenced by its spread to 188 countries across the globe within a very short time.
Roles of environmental factors in transmission of COVID-19 have been highlighted before. Several impressive published works on environmental roles revealed the potential risk of environmental factors on the transmission and prevalence of COVID-19 pandemics, such as climate change, fomites, water transfer, air and food (Eslami and Jalili, 2020, Qu et al., 2020, Wigginton and Boehm, 2020). Kampf et al. emphasized that inanimate surface contact is an important way of transmitting the SARS-CoV-2 (Kampf et al., 2020). SARS-CoV-2 can survive on surfaces for hours to days, which can live on inanimate surfaces such as metals, glass and plastic at room temperature increasing the opportunity for transmission via touch (Kampf et al., 2020, van Doremalen et al., 2020). Eslami and Jalili concluded that reducing the frequency of touching surfaces by hands and disinfecting surfaces can reduce the amount of coronavirus load on surfaces and the rate of transmission (Eslami and Jalili, 2020). Although transmission from contaminated surfaces and fomites are emphasized as an indirect transmission route of SARS-CoV-2, detection of the virus on the environmental surfaces is largely unknown.
From March 18–24, 2020, three Chinese oversea students returned to Qingdao, China. After tested negative nucleic acid without any symptoms at the entry quarantine, they were then transferred to the hotel for 14 days quarantine. During quarantine period in hotel, they presented initial symptoms or were tested positive SARS-CoV-2 as a presymptomatic person. Meanwhile, they were transferred to the local hospital for further diagnosis and treatment. In addition, we synchronously sampled the high-touch environmental surfaces in the quarantine room, aiming to detect the SARS-CoV-2 distribution on the environmental surfaces during the incubation period of COVID-19 patients. Our findings would extremely address the importance of touching the contaminated environment transmission, and help extend an effective protocol to interrupt the indirect environmental transmission of SARS-CoV-2, limit its spread, and mitigate its risks.
2. Methods
2.1. Patients
Three COVID-19 patients, as Chinese oversea students in America and England, returned to Qingdao from March 18–24, 2020. They had no fever and other symptoms at the entry quarantine, and transferred to a hotel for 14 days quarantine. During the quarantine, their nasopharyngeal swabs were collected every 1 to 3 days. If the swab was tested negatively for SARS-CoV-2, the student would be still in quarantine in hotel, whereas if the swab was tested positively for SARS-CoV-2, or presenting initial symptoms of COVID-19, such as fever, cough, myalgia and fatigue, etc., they would be immediately transferred to the hospital for further diagnosis and treatment. Personal, clinical, and radiological characteristics onset of illness were obtained with standardized data collection forms from the interview, field reports as well as electronic medical records. Additionally, their frequency of face washing, hands washing, tooth brushing, bathing and excrement during the quarantine were interviewed by telephone after discharged from hospital.
This study was approved by the Ethics Commission of Municipal Centre of Disease Control and Prevention of Qingdao and written informed consent was waived considering the emergency of infectious disease.
2.2. Approach to environmental surface sampling
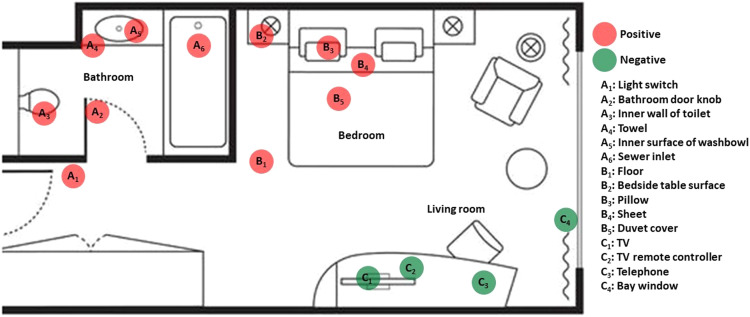
Environmental surface samples were collected with sterile polyester-tipped applicator pre-moistened in viral transport medium. Fifteen sites were sampled from the quarantine room, including light switch, bathroom door knob, inner wall of toilet, towel, inner surface of washbowl, sewer inlet, floor (0.5–1.5 m from the bed), bedside table surface, pillow, sheet, duvet cover, television (TV), TV remote controller, telephone, and bay window. All sampling sites were marked in Fig. 1. The surface of the entire item was swabbed except for pillow, sheet, and duvet cover by swabbing 10 times vigorously from two directions (horizontally and vertically) in an about 100 cm2 area that they contacted. All sites were sampled within 4 h after the positive nucleic acid test of the patients.
Fig. 1.
Distribution of environmental sampling sites in the quarantine room.
2.3. SARS-CoV-2 detection
All samples after sampling were transferred to the Qingdao Municipal Center for Disease Control and Prevention for SARS-CoV-2 detection. Tests were carried out in biosafety level 2 facilities, using a commercial Novel Coronavirus Nucleic Acid Detection Kit (Shanghai BioGerm Medical Technology Company) in a total reaction volume of 25 μL, targeting SARS-CoV-2 virus frame1ab (ORFab1). Viral RNA was extracted from sample material and collected in elution buffer, and then underwent real-time reverse-transcription-polymerase-chain-reaction (RT-PCR) with SARS-CoV-2-specific primers and probes. Reverse transcription was performed at 50 °C for 10 min, 95 °C for 5 min, followed by 40 cycles of RT-PCR analysis at 95 °C for 10 s, and annealing/elongation/fluorescence detection at 55 °C for 40 s. A cycle threshold (Ct) value were used to approximately reflect the viral loads (inversely related to Ct-value) in the respiratory tract, and lower Ct values indicated higher viral loads and vice versa (Xu et al., 2020, Zhu et al., 2020). According to the Technical Guidelines for Laboratory Testing of COVID-19 issued by National Health Commission the People's Republic of China (Chinese Center for Disease Control and Prevention (China CDC), 2020a), a Ct value less than 37 was defined as a positive test, and a Ct value of 40 or more was considered as a negative test. An equivocal result, defined as a Ct value between 37 and 40, required confirmation by retesting. If the repeated Ct value was less than 40 and an obvious peak was observed, or if the repeated Ct value was less than 37, the result was deemed positive.
Quality controls in SARS-CoV-2 detection was conducted as follows: environmental surface samples were at the room temperature for no more than 2 h before laboratory detection; viral RNA was isolated from sample material by automatic nucleic acid isolation instruments (MagNA Pure 96, Roche Diagnostics GmbH, Germany) to minimize the possibility of laboratory contamination; Novel Coronavirus Nucleic Acid Detection Kit recommended by National Health Commission of China was adopted, and laboratory steps were performed according to the instruction of the manufacturer. In addition, SARS-CoV-2 Medium Quality Control and Negative Quality Control (TMNQC, RANDOX, UK) were used for RNA isolation to conduct quality controls. After comparing with stability and sensitivity of products from six manufacturers, we purchased the most precise primers from Shanghai BioGerm Medical Technology Company. According to the stability of weak positive quality control results (Synthetic SARS-CoV-2-RNA, Twist Bioscience, #102024), the effectiveness of primers was monitored in real time.
3. Results
3.1. Characteristics of patients
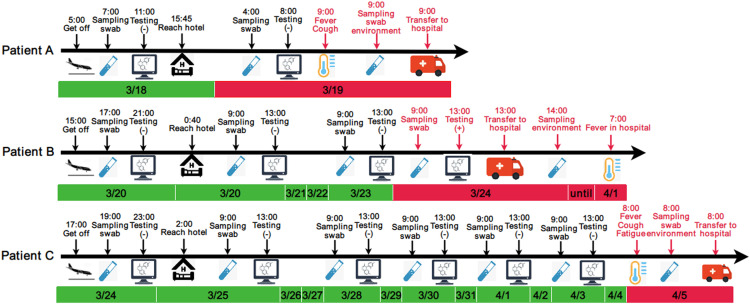
Three patients returned to Qingdao, China, during March 18 and 24, 2020, and their characteristics were shown in Table 1. Patient A and C were onset of illness (fever, cough or fatigue) during the quarantine time. When presenting initial symptoms, they immediately reported to professional medical staffs stayed in hotel. Then, medical staffs stayed in hotel immediately sampled their nasopharyngeal swabs and environmental surfaces, and transferred them to the hospital for further diagnosis and treatment. All samples were sent to Qingdao CDC within 2 h, and were conducted laboratory tests taking 2 h. However, Patient B was tested positive RT-PCR during the routine monitoring in the room. Once the positive result tested in laboratory (within 4 h after sampling nasopharyngeal swab), medical staffs stayed in the hotel immediately went the room to sample environmental surfaces and transferred her to hospital. On the seventh day of hospitalization, he presented fever, as the initial symptom. The timeline from getting off the flight to leaving quarantine room for each patient was shown in Fig. 2.
Table 1.
Characteristics of patients.
| Characteristics | Patient A | Patient B | Patient C |
|---|---|---|---|
| Date returned to China | March 18, 2020 | March 20, 2020 | March 24, 2020 |
| Check-in time for quarantine | March 18, 2020 | March 21, 2020 | March 25, 2020 |
| Duration of quarantine in hotel | 18 h | 3 days | 11 days |
| Date of SARS-CoV-2 positive detection and admission | March 19, 2020 | March 24, 2020 | April 5, 2020 |
| Initial symptoms | Fever, cough | Fever | Fever, cough, fatigue |
| Ct values of samples on admission | |||
| Nasopharyngeal swab | 24 | 27 | 20 |
| Sputum | 28 | 39 | 24 |
| Stool swab | 33 | > 40 | 34 |
| CT imaging | Normal | Normal | Normal |
| Disease severity | Moderate | Moderate | Moderate |
Fig. 2.
The timeline from getting off the flight to leaving quarantine room for each patient .
The nasopharyngeal swab, sputum and stool swab were tested by RT-PCR for all cases on admission, and they were all positive for SARS-CoV-2 except for sputum in patient B. The Ct values of nasopharyngeal swab and sputum were lowest in Patient C, and the lowest value of stool swab was in patient A. All cases were identified as moderate COVID-19, with normal computed tomography (CT) imaging.
Additionally, the frequency of washing behaviors of patients at the quarantine room, including face washing, hands washing, tooth brushing, bathing and excrement, were shown in Table 2.
Table 2.
The frequency of washing behaviors of patients at the quarantine room.
| Behaviors | Patient A | Patient B | Patient C |
|---|---|---|---|
| Brushing tooth | Morning and evening | Morning and evening | Morning and evening |
| Washing face | Morning and evening | Morning and evening | Morning and evening |
| Washing hands | Before and after eating | Before and after eating | Before and after eating |
| Bathing | None | None | Every 3 days |
| Excrement | Once a day | Once a day | Once a day |
| Hand disinfection | None | None | None |
3.2. SARS-CoV-2 in environmental surface samples
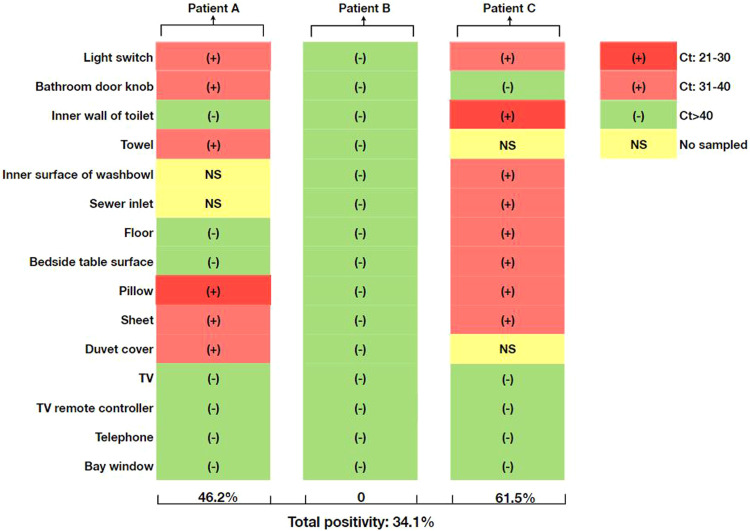
All sampling sites were marked in Fig. 1. In total, forty-one samples were collected from the quarantine rooms of the three COVID-19 patients. As shown in Fig. 3, overall, 14 of 41 (34.1%) samples were tested positively for SARS-CoV-2, and the Ct values ranged from 26 to 38, with the median of 35. For patient A, 6 of 13 (46.2%) samples were tested positively for SARS-CoV-2, and Ct values ranged from 28 and 37, with one sample Ct value below 30. For patient B, all samples were tested negatively for the virus. For patient C, 8 of 13 (61.5%) samples were tested positively for SARS-CoV-2, and the Ct values ranged from 26 to 38, with one sample Ct value below 30.
Fig. 3.
Distribution of Ct values of all samples in all cases.
As shown in Fig. 1 and Table 3, the quarantine room was divided into three functional areas. Bedroom and bathroom were tested positively for SARS-CoV-2, with the positivity rate of 50.0% (7/14) and 46.7% (7/15), respectively. In the bathroom, all the six sites were positive for RT-PCR tests, including light switch, bathroom door knob, inner wall of toilet, towel, inner surface of washbowl and sewer inlet, and the lowest Ct value was detected from inner wall of toilet. In the bedroom, all the five sites were positive for RT-PCR tests, including floor, beside table surface, pillow, sheet and duvet cover, and the lowest Ct value was detected from pillow. In contrast, living room was negative for virus, and all sites, such as TV, TV remote controller, telephone and bay window, were negative for RT-PCR test.
Table 3.
Distribution of Ct values of environmental surface samples among each functional area of the quarantine room and surface material.
| Functional area/surface material | Positive/total | Minimum | Median | Maximum |
|---|---|---|---|---|
| Bathroom | 7/15 (46.7%) | 26 | 33 | 37 |
| Bedroom | 7/14 (50.0%) | 29 | 36 | 38 |
| Living room | 0/4 (0) | > 40 | > 40 | > 40 |
| Cotton | 6/10 (60.0%) | 28 | 34 | 38 |
| Ceramic | 2/5 (40.0%) | 26 | – | 33 |
| Metal | 2/5 (40.0%) | 32 | – | 33 |
| Wood | 2/6 (33.3%) | 35 | – | 36 |
| Plastic | 2/12 (16.7%) | 32 | – | 37 |
In addition, all environmental sites came from five types of material. As shown in Table 3, the positive rate was highest in cotton sites (60.0%, 6/10), followed by ceramic sites (40.0%, 2/5), metal sites (40.0%, 2/5), wood sites (33.3%, 2/6) and plastic sites (16.7%, 2/12).
4. Discussion
Although researchers enhanced the importance of environmental contamination on the route of SARS-CoV-2 transmission (Faridi et al., 2020, Guo et al., 2020, Ong et al., 2020, Yung et al., 2020), there has been limit data revealing the contribution of direct surface contacts to the transmissibility of COVID-19. This case study explored the persistence of SARS-CoV-2 on the environmental surfaces, reporting detection of SARS-CoV-2 on the high-touch surfaces of dwelling environment in detail, especially for during the incubation period. All patients had no medication and disease symptoms, such as fever, before boarding the plane, and they all took personal protection by wearing mask and glove during the flight. When getting off the flight, they were immediately conducted nucleic acid test of SARS-CoV-2, and observed whether they had fever and other symptoms. Due to no disease symptoms and negative result of SARS-CoV-2 test, they were transferred to the hotel for 14 days quarantine. According to the prevention of COVID-19, going in and out of the quarantine room is strictly controlled, and anyone including care takers is not allowed to go in or out of the quarantine rooms during quarantine period except for professional medical staffs. During the quarantine, professional medical staffs with personal protective equipment went in the rooms to collect the nasopharyngeal swabs of quarantined students every one to three days, for early detection of COVID-19. The incubation period for COVID-19, is the time between exposure to the virus (becoming infected) and symptom onset (World Health Organization (WHO), 2020c). In our study, we concluded that three students were only possibly attacked by COVID-19 before or on the flight. Further, all environmental samples were collected when they were presenting initial symptom or tested positive RT-PCR during presymptomatic duration in quarantine room, which was during the incubation period.
Although we were unable to conduct virus culture, touching the environmental surfaces contaminated by SARS-CoV-2 in the quarantine room had great risk for viral infection. Several studies revealed a strong relationship between Ct values of RT-PCR and recovering infectious virus from positive respiratory tract samples in SARS-CoV-2 infected cases (La Scola et al., 2020, Perera et al., 2020, Singanayagam et al., 2020, Wölfel et al., 2020). Singanayagam et al. recently reported that the estimated odds ratio (OR) of recovering infectious virus decreased by 0.67 for each unit increase in Ct value (95%CI: 0.58–0.77). It also reported that 86.3% respiratory tract samples tested positive RT-PCR with Ct value below 30 were culture-positive, whereas 8.3% RT-PCR positive samples with Ct value over 35 were culture-positive. In addition, it identified that Ct values and the presence of infectious virus were similar in samples from asymptomatic and presymptomatic persons, compared with those who were symptomatic (Arons et al., 2020, Singanayagam et al., 2020). According to these findings, it is likely that the contaminated environmental surfaces during the incubation period of COVID-19 may have viral infectiousness, which decreases with the increase of Ct value.
Temperature, humidity and surface material types may be the important environmental factors for the survivability of SARS-CoV-2 on surfaces (Aboubakr et al., 2020, Biryukov et al., 2020, Ren et al., 2020). In our study, we found that environmental samples from bedroom and bathroom were detected positively for SARS-CoV-2 with close positive rates (50.0% and 46.7%, respectively), and the average Ct value in bathroom (32) was lower than that in bedroom (35). This may attribute to the indoor temperature and humidity in the different functional areas of the room. Due to moist space, bathroom had relatively low indoor temperature and high humidity compared with bedroom, which created the environmental condition delaying inactivation of SARS-CoV-2 on surfaces. Central heating was provided from 16 November, 2019–4 April, 2020 in Qingdao, and the quarantine period of three patients was within the central heating duration. A recent study investigating the central heating residential indoor thermal environment in Qingdao, suggested that the average temperature was 21.2 °C and the average humidity was 37.4% in the indoor environment during the central heating duration (Han et al., 2017), which provided an effective reference for our study. Although we did not measure other environmental factors during the quarantine, it was considered that temperature and humidity were consistently kept under control attributed to central heating and other similar accommodations. Additionally, we further explored the relationship between surface material types of sampled sites and viral detection. In our study, all 15 sampled sites came from five materials, including metal (door knob, sewer inlet), plastic (light switch, TV remote controller, telephone, TV), cotton (towel, pillow, sheet, duvet cover), ceramic (toilet, washbowl), wood (bedroom floor, bedside table surface) and stone (bay window). The positive rate was highest in cotton sites (60.0%, 6/10), followed by ceramic sites (40.0%, 2/5), metal sites (40.0%, 2/5), wood sites (33.3%, 2/6) and plastic sites (16.7%, 2/12). Although we were unable to control other potential factors, our findings provided implication for the survivability and infectivity of SARS-CoV-2 under temperature, humidity and different types of surface.
Furthermore, Ct values in bathroom (median: 33; range: 26–37) were lower than those in bedroom (median: 36; range: 29–38), which indicated different severity and extent of viral contamination. However, the severity of the environmental contamination may be correlated with the viral shedding of the COVID-19 patient. Environmental surfaces, frequently contacted with positive excretion of infectious person, may have high risk on contamination with high viral load. Comparing with the Ct values of their nasopharyngeal swab, sputum specimen and stool specimen on admission and environmental samples, could provide possibility to find some clues. Among all the environmental samples, the Ct value of inner wall of toilet was the lowest in patient C, which was higher than those of his nasopharyngeal swab (Ct: 20) and sputum specimen (Ct: 24), whereas it was lower than that of stool specimen on admission (Ct: 34). It may be due to contacting mixed viral excretion from both defecating stool and spiting sputum into the toilet. Similarly, bathroom was the concentrated area of viral shedding from both upper respiratory tract and digestive tract excretion. This may explain why the contamination of bathroom was more severe than bedroom. Moreover, in Patient B, as the presymptomatic person without symptoms during the quarantine, the Ct values of nasopharyngeal swab, sputum specimen and stool specimen were the highest in all patients, which suggested a low viral load for shedding. It may explain why there was no positive sample detected on environmental surfaces. Additionally, the environmental contamination may be also correlated with the duration of quarantine in the hotel. The quarantine duration of Patient C was 11 days, which was much more than the durations of other patient, and this may correlate with more positive environmental sites.
SARS-CoV-2 in upper respiratory tract (Sputum and saliva) and on body surface (hands and face) could be brought into water by face washing, hands washing, tooth brushing and bathing, resulting in viral contamination of the inner surface of washbowl and sewer inlet. Although these two sites often have water flowing, virus in contaminated water may still remain on them under general open flowing time of clean water and the conventional washing behaviors. After patients were discharged from hospital several months, we conducted telephone interview to investigate their frequency of face washing, hands washing, tooth brushing, bathing and excrement during the quarantine, which could be helpful to discussing the reasons for some SARS-CoV-2 positive sites. All patients did not use disinfectant or other protective equipment during the hotel quarantine. Each of them brushed tooth and washed faces in the morning and evening. All patients had habit of washing hands before and after eating, and excrement once a day. Patient A had no bathing or excrement, and Patient B had no bathing during the quarantine. However, Patient C took bath every 3 days. Their washing behaviors may explain positive sites in bathroom, including inner wall of toilet, towel, inner surface of washbowl and sewer inlet. Similarly, light switch and bathroom door knob were also tested positively for virus, which may be attributed that they did not washing hands timely after contacting viral fomites. More information about individual behaviors is implicated for the reason for positive environmental surfaces. However, due to leaving the quarantine room for several months, they were unable to recall any other information. Thus, detailed questionnaire and individual body surface sampling during the quarantine should be conducted in future study, which could expand further understanding regarding mechanism of environmental contamination of SARS-CoV-2.
Several limitations should be known in our study. Firstly, we did not sample any indoor air, which should be strengthened in future study. Compared with other sampling sites in the room, the frequency of touching main door knob may be relatively low. However, main door knob should be still as necessary sampling site. Unfortunately, we did not sample it in all rooms, which should be paid for attention in our future study. Although we used central heating to describe the situation of room environment, other environmental factors, such as room temperature and humidity, were not detected accurately in the quarantine rooms. In addition, our study only analyzed the environmental contamination in the quarantine room based on a small size of COVID-19 patients, and further research on a larger size should be performed to evaluate the contribution of environmental contamination to the transmissibility of COVID-19.
5. Conclusion
Environmental contamination of SARS-CoV-2 distributes widely during the incubation period of CVOID-19, and the positive rates of SARS-CoV-2 on environmental surfaces are relatively high in bathroom and bedroom. Our findings would extremely address the importance of touching the contaminated environment transmission, and help extend an effective protocol to interrupt the indirect environmental transmission of SARS-CoV-2, limit its spread, and mitigate its risks.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
CRediT authorship contribution statement
Xiaowen Hu: Conceptualization, Writing - original draft. Wei Ni: Formal analysis, Writing - original draft. Zhaoguo Wang: Investigation, Writing - original draft. Guangren Ma: Investigation, Writing - original draft. Bei Pan: Investigation. Liyan Dong: Investigation. Ruqin Gao: Investigation, Writing - original draft. Fachun Jiang: Conceptualization, Writing - original draft.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
We are deeply thankful to all health-care workers involved in the diagnosis and treatment of patients in Qingdao.
Edited by Dr. Yong Liang
References
- Aboubakr H.A., Sharafeldin T.A., Goyal S.M. Stability of SARS-CoV-2 and other coronaviruses in the environment and on common touch surfaces and the influence of climatic conditions: a review. Transbound. Emerg. Dis. 2020 doi: 10.1111/tbed.13707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arons M.M., Hatfield K.M., Reddy S.C., Kimball A., James A., Jacobs J.R., Taylor J., Spicer K., Bardossy A.C., Oakley L.P., Tanwar S., Dyal J.W., Harney J., Chisty Z., Bell J.M., Methner M., Paul P., Carlson C.M., McLaughlin H.P., Thornburg N., Tong S., Tamin A., Tao Y., Uehara A., Harcourt J., Clark S., Brostrom-Smith C., Page P.C., Kay M., Lewis S., Montgomery P., Stone N.D., Clark T.A., Honein M.A., Duchin J.S., Jernigan J.A., Public Health–Seattle and King County and CDC COVID-19 Investigation Team. Presymptomatic SARS-CoV-2 infections and transmission in a skilled nursing facility. N. Engl. J. Med. 2020;382(22):2081–2090. doi: 10.1056/NEJMoa2008457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biryukov J., Boydston J.A., Dunning R.A., Yeager J.J., Wood S., Reese A.L., Ferris A., Miller D., Weaver W., Zertouni N.E., Phillips A., Freeburger D., Hooper I., Ratnesar-Shumate S., Yolitz J., Krause M., Williams G., Dawson D.G., Herzog A., Dabisch P., Wahl V., Hevey M.C., Altamura L.A. Increasing temperature and relative humidity accelerates inactivation of SARS-CoV-2 on surfaces. mSphere. 2020;5(4) doi: 10.1128/mSphere.00441-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chinese Center for Disease Control and Prevention (China CDC), 2020a. Technical guidelines for laboratory testing of COVID-19. 〈http://med.china.com.cn/content/pid/158678/tid/1026〉. (Assessed 23 January 2020). [DOI] [PMC free article] [PubMed]
- van Doremalen N., Bushmaker T., Morris D.H., Holbrook M.G., Gamble A., Williamson B.N., Tamin A., Harcourt J.L., Thornburg N.J., Gerber S.I., Lloyd-Smith J.O., de Wit E., Munster V.J. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N. Engl. J. Med. 2020;382:1564–1567. doi: 10.1056/NEJMc2004973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eslami H., Jalili M. The role of environmental factors to transmission of SARS-CoV-2 (COVID-19) AMB Express. 2020;10:92. doi: 10.1186/s13568-020-01028-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faridi S., Niazi S., Sadeghi K., Naddafi K., Yavarian J., Shamsipour M., Jandaghi N.Z.S., Sadeghniiat K., Nabizadeh R., Yunesian M., Momeniha F., Mokamel A., Hassanvand M.S., MokhtariAzad T. A field indoor air measurement of SARS-CoV-2 in the patient rooms of the largest hospital in Iran. Sci. Total Environ. 2020;725 doi: 10.1016/j.scitotenv.2020.138401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Z.D., Wang Z.Y., Zhang S.F., Li X., Li L., Li C., Cui Y., Fu R.B., Dong Y.Z., Chi X.Y., Zhang M.Y., Liu K., Cao C., Liu B., Zhang K., Gao Y.W., Lu B., Chen W. Aerosol and surface distribution of severe acute respiratory syndrome coronavirus 2 in hospital wards, Wuhan, China, 2020. Emerg. Infect. Dis. 2020;26:26–1591. doi: 10.3201/eid2607.200885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han F., Hu S.T., Wang H.Y., Liu G.D., Wang L.Y. Test and analysis of the central heating residential indoor thermal environment in Qingdao city. J. Qingdao Univ. Technol. 2017;38(3):64–70. [Google Scholar]
- Johns Hopkins University of Medicine (JHU), 2020b. Coronavirus Resource Center. 〈https://coronavirus.jhu.edu/〉. (Accessed 25 May 2020).
- Kampf G., Todt D., Pfaender S., Steinmann E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J. Hosp. Infect. 2020;104:246–251. doi: 10.1016/j.jhin.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ong S.W.X., Tan Y.K., Chia P.Y., Lee T.H., Ng O.T., Wong M.S.Y., Marimuthu K. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA. 2020;323:1610–1612. doi: 10.1001/jama.2020.3227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perera R.A.P.M., Tso E., Tsang O.T.Y., Tsang D.N.C., Fung K., Leung Y.W.Y., Chin A.W.H., Chu D.K.W., Cheng S.M.S., Poon L.L.M., Chuang V.W.M., Peiris M. SARS-CoV-2 virus culture and subgenomic RNA for respiratory specimens from patients with mild coronavirus disease. Emerg. Infect. Dis. 2020;26(11) doi: 10.3201/eid2611.203219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qu G.B., Li X.D., Hu L.G., Jiang G.B. An imperative need for research on the role of environmental factors in transmission of novel coronavirus (COVID-19) Environ. Sci. Technol. 2020;54:3730–3732. doi: 10.1021/acs.est.0c01102. [DOI] [PubMed] [Google Scholar]
- Ren S.Y., Wang W.B., Hao Y.G., Zhang H.R., Wang Z.C., Chen Y.L., Gao R.D. Stability and infectivity of coronaviruses in inanimate environments. World J. Clin. Cases. 2020;8(8):1391–1399. doi: 10.12998/wjcc.v8.i8.1391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- La Scola B., Le Bideau M., Andreani J., Hoang V.T., Grimaldier C., Colson P., Gautret P., Raoulr D. Viral RNA load as determined by cell culture as a management tool for discharge of SARS-CoV-2 patients from infectious disease wards. Eur. J. Clin. Microbiol. Infect. Dis. 2020;39(6):1059–1061. doi: 10.1007/s10096-020-03913-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singanayagam A., Patel M., Charlett A., Lopez B., Jamie S.V., Ellis J., Ladhani S., Zambon M., Gopal R. Duration of infectiousness and correlation with RT-PCR cycle threshold values in cases of COVID-19, England, January to May 2020. Euro Surveill. 2020;25(32) doi: 10.2807/1560-7917.ES.2020.25.32.2001483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wigginton K.R., Boehm A.B. Environmental engineers and scientists have important roles to play in stemming outbreaks and pandemics caused by enveloped viruses. Environ. Sci. Technol. 2020;54:3736–3739. doi: 10.1021/acs.est.0c01476. [DOI] [PubMed] [Google Scholar]
- Wölfel R., Corman V.M., Guggemos W., Seilmaier M., Zange S., Müller M.A., Niemeyer D., Jones T.C., Vollmar P., Rothe C., Hoelscher M., Bleicker T., Brunink S., Schneider J., Ehmann R., Zwirglamaier K., Drosten C., Wendtner C. Virological assessment of hospitalized patients with COVID-2019. Nature. 2020;581(7809):465–469. doi: 10.1038/s41586-020-2196-x. [DOI] [PubMed] [Google Scholar]
- World Health Organization (WHO), 2020c. Coronavirus disease 2019 (COVID-19)-WHO. 〈https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200402-sitrep-73-covid-19.pdf?sfvrsn=5ae25bc7_6〉. (Accessed 3 April 2020).
- Xu Y., Li X., Zhu B., Liang H., Fang C., Gong Y., Guo Q., Sun X., Zhao D., Shen J., Zhang H., Liu H., Xia H., Tang J., Zhang K., Gong S. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat. Med. 2020;26:502–505. doi: 10.1038/s41591-020-0817-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yung C.F., Kam K.Q., Wong M.S.Y., Maiwald M., Tan Y.K., Tan B.H., Thoon K.C. Environment and personal protective equipment tests for SARS-CoV-2 in the isolation room of an infant with infection. Ann. Intern. Med. 2020;173:240–242. doi: 10.7326/M20-0942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., Zhao X., Huang B., Shi W., Lu R., Niu P., Zhan F., Ma X., Wang D., Xu W., Wu G., Gao G.F., Tan W. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;2020(382):727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]