Abstract
Chondral defects of the knee are prevalent and often encountered during arthroscopic procedures. Despite the limited healing potential of chondral defects, several treatment options have been proposed. However, microfracture, osteochondral autograft (or allograft) transfer, autologous chondrocyte implantation, and matrix-induced autologous chondrocyte implantation are all associated with their respective shortcomings. As such, the optimal treatment for chondral defects of the knee remains unclear. Recently, many authors have advocated treating chondral defects with biological therapies and scaffold-based treatments. Bone marrow aspirate concentrate, a cell-based injection, has gained particular attention because of its differentiation capacity and potential role in tissue regeneration. In addition, scaffold cartilage treatments have emerged and reached clinical practice. BioCartilage is one form of scaffold, which consists of extracellular matrix, and has been claimed to promote the regeneration of hyaline-like cartilage. This article presents our technique of arthroscopic chondral defect repair using BMAC and BioCartilage.
Chondral defects of the knee are extremely prevalent and often identified during arthroscopic procedures.1,2 Despite the limited healing potential of chondral defects,3,4 several treatment options have been proposed. Among the treatment options, microfracture (MF) has been considered the treatment of choice for lesions that measure than 2 cm2.5 On the other hand, osteochondral autograft (or allograft) transfer, autologous chondrocyte implantation, and matrix-induced autologous chondrocyte implantation (MACI) are typically reserved for larger defects.6,7 However, MF has been associated with poor long-term outcomes, osteochondral autograft transfer may lead to donor-site morbidity, and MACI requires multiple operations and is costly.8, 9, 10, 11, 12 As such, the optimal treatment for chondral defects of the knee remains unclear.13
Recently, many authors have advocated treating chondral defects with biological therapies and scaffold-based treatments. Bone marrow aspirate concentrate (BMAC), a cell-based injection, has gained particular attention because of its differentiation capacity and potential role in tissue regeneration.14 Early preclinical studies have found that defects treated with BMAC generate an increased fill with well-integrated repair tissue composed of greater type II collagen content.15,16 In addition, scaffold cartilage treatments have emerged and reached clinical practice.17 Although cell-based scaffolds have been more readily studied, cell-free constructs have shown early promising outcomes.18, 19, 20 BioCartilage (Arthrex, Naples, FL) is one form of scaffold, which consists of extracellular matrix, and has been claimed to promote the regeneration of hyaline-like cartilage.21 This article presents our technique of arthroscopic chondral defect repair using BMAC and BioCartilage.
Technique
Indications
Patients who present with knee pain, swelling, mechanical symptoms, and limited motion and functional capabilities and receive a diagnosis of a chondral defect may be candidates for this procedure. Patients with chondral lesions in whom conservative measures have failed, including physical therapy, nonsteroidal inflammatory drug use, and intra-articular injections, may be indicated for this procedure.22,23
Surgery
Bone marrow aspiration is the first step in this procedure. The patient is placed in the supine position, and the anterior iliac crest is aseptically prepared. The bone marrow aspirate is obtained from the iliac crest in a standard fashion. An 11- or 13-gauge-diameter fenestrated blunt trocar (ISTO Biologics, Hopkinton, MA) is inserted percutaneously down to bone and into the medullary cavity 3 fingerbreadths posterior to the anterior superior iliac spine24 (Fig 1, Video 1). This posterior access point protects the lateral femoral cutaneous nerve from injury. The syringe is attached to the trocar, and a total of 60 mL of bone marrow is aspirated by rotating the syringe 90° approximately every 5 mL. The bone marrow aspirate is then centrifuged (Magellan; ISTO Biologics). During this process, the red blood cells are separated and extracted, and the buffy coat is concentrated. Approximately 7 mL of BMAC is yielded in which the nucleated cells, platelets, and growth factors are ready for immediate use.
Fig 1.
(A) Prior to bone marrow aspiration, the patient is positioned supine and the harvest site of the contralateral iliac crest of the affected knee is prepared in a sterile fashion. Bone marrow aspirate is obtained from the right iliac crest using a trocar (black arrow) inserted down to bone 3 fingerbreadths (black H) posterior to the anterior superior iliac spine. (B) Aspiration of 60 mL of bone marrow is performed by rotating the trocar (white arrows) 90° every 5 mL of removal.
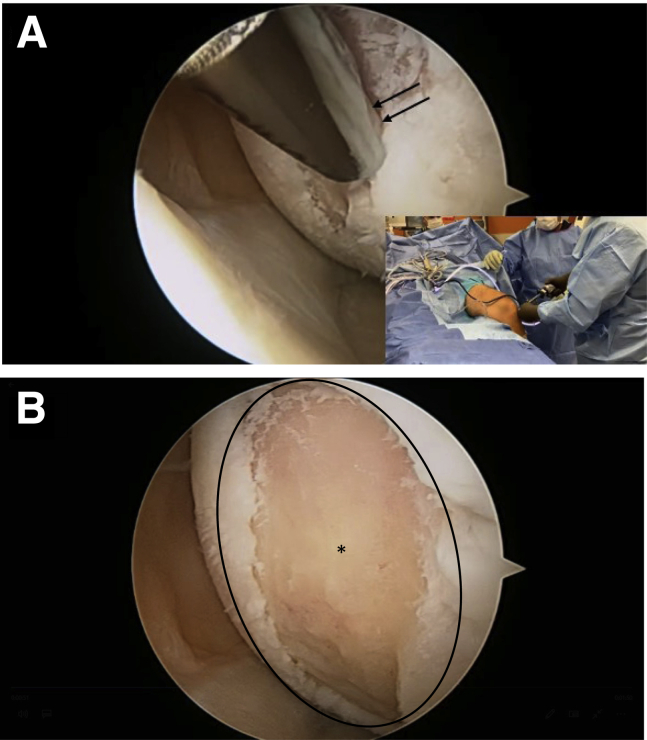
Standard diagnostic arthroscopy is then performed by anteromedial and anterolateral portals to assess damage to the cartilage and underlying bone of the afflicted region. The area is debrided with a shaver in forward-down mode to a bed of healthy bleeding bone (Fig 2). A stable rim is also achieved.
Fig 2.
Standard diagnostic arthroscopy of the left knee is performed via anteromedial and anterolateral portals. (A) Via the anterolateral viewing portal, the area of the chondral defect is debrided with a shaver in forward mode (black arrows). (B) Debridement is performed until a healthy bed of bleeding bone is reached (asterisk) and a stable rim (black oval) is achieved.
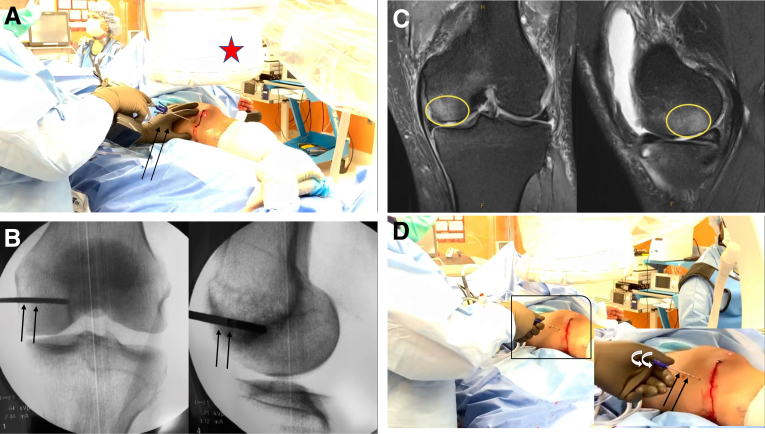
Next, subchondroplasty (Zimmer Biomet, Warsaw, IN) is performed with calcium phosphate cement behind the chondral defect, under fluoroscopic guidance, to address the associated subchondral insufficiency fracture (Fig 3). After a small incision is made, a subchondroplasty trocar is inserted behind the chondral defect, which is confirmed by correlating orthogonal fluoroscopic views with the subchondral edema on the preoperative magnetic resonance imaging scan. The trocar is inserted to an appropriate depth based on the location of the subchondral insufficiency fracture, and it is rotated such that the perforation is faced toward the joint line. In total, no more than 3 mL of calcium phosphate cement is injected through the trocar, rotating the trocar back and forth slowly while injecting to evenly fill the lesion in 3 dimensions. If performed adequately, calcium phosphate cement leakage should not occur and the substrate should be maintained within the location of the lesion. Currently, it is unknown whether interaction through improper technique between the calcium phosphate cement and the BMAC and BioCartilage scaffold would lead to the inhibition of the BMAC and scaffold mixture. However, there should be minimal contact between the cement injected into the subchondral insufficiency fracture and the biological materials applied arthroscopically to the chondral surface. Thus, proper technique including avoiding aggressive debridement on the lesion remains important during this and all steps. After this injection is completed, the inner sleeve is replaced and the trocar is left in place for 10 minutes to allow the cement time to harden.
Fig 3.
Subchondroplasty of the left knee is performed through a small incision in the subchondral bone marrow insufficiency. (A) By use of fluoroscopic guidance (red star), a subchondroplasty trocar (black arrows) is inserted through a small incision behind the chondral defect of the left knee. Correct placement is confirmed by correlating anteroposterior and lateral orthogonal fluoroscopic views of the trocar (black arrows) (B) with preoperatively obtained anteroposterior and lateral magnetic resonance imaging views showing subchondral edema (yellow ovals) (C). (D) The trocar (black arrows) is inserted at the appropriate depth (with inset showing zoomed-in view of insertion) and rotated so that the perforation is faced toward the joint line (white arrows). (E) Injection of 3 mL of calcium phosphate cement (yellow arrows) is performed through the trocar (red arrows), rotating the trocar 180° back and forth during injection (white arrows) to ensure even filling of the lesion. (F) After injection, the inner sleeve of the trocar (red arrows) is replaced and the cement is allowed 10 minutes to harden.
With subchondroplasty complete, attention is turned to the application of BMAC and BioCartilage to the chondral defect (Fig 4). An 18-gauge spinal needle is placed percutaneously such that the tip rests just below the lesion; this will be used to keep the area dry during the procedure. The joint is thoroughly dried with the use of suction, the fluid inflow tubing is removed, and the stopcock on the arthroscope is left open to avoid closure of soft-tissue space in the joint due to lack of airflow while suctioning. A second 18-gauge spinal needle is placed percutaneously above the lesion; this will be used for injection of fibrin glue (TISSEEL; Baxter Healthcare, Franklin Lakes, NJ). By use of suction through the BioCartilage application suction paddle (Arthrex), the joint is fully evacuated of fluid, confirming that the anterior fat pad has been resected adequately and will not interfere with visualization of the lesion directly or as a reservoir for fluid accumulation during the procedure.
Fig 4.
Preparation for BioCartilage application to the chondral defect of the left knee is performed. (A) Via the anterolateral viewing portal, one 18-gauge spinal needle (white arrows) is placed percutaneously such that the tip rests just below the lesion (white star) and will be used to keep the area dry during the procedure. (B) Another 18-gauge spinal needle (white arrows) is placed percutaneously above the lesion (white star) and will be used for injection of fibrin glue.
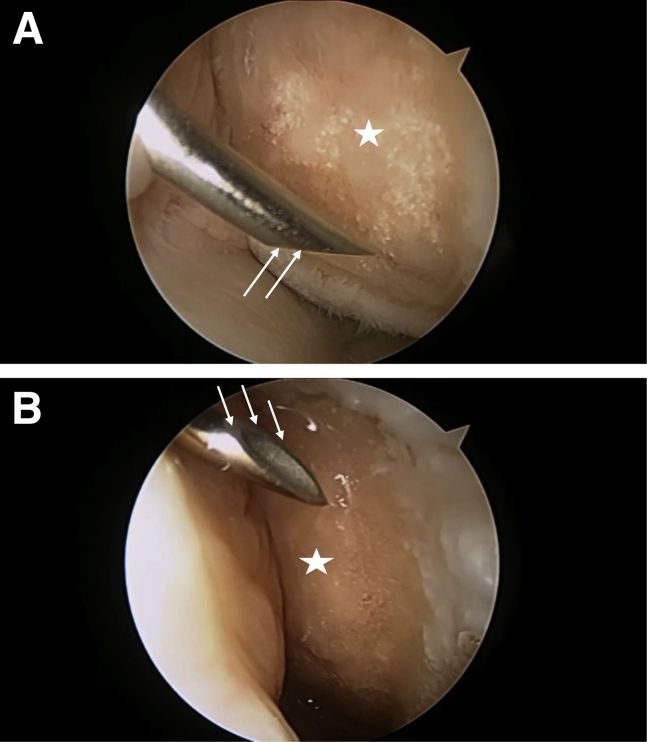
The BioCartilage mixed with 1 mL of BMAC is injected through the trocar and smoothed down to fill the lesion with the paddle (Fig 5). Throughout this process, an assistant intermediately applies suction to the inferior spinal needle via a Frazier-tip suction device, keeping the working area free of fluid. Once the BioCartilage has been applied, fibrin glue is injected over it via the superior spinal needle, sealing it in place. The fibrin glue is given time to harden prior to removal of the scope and closure. The remaining 6 mL of BMAC is injected into the joint after closure of the portals. Standard perioperative care includes 1 preoperative dose of intravenous antibiotics and postoperative thromboembolic prophylaxis with 325 mg of aspirin twice a day.
Fig 5.
(A) Via the anterolateral viewing portal, BioCartilage mixed with 1 mL of bone marrow aspirate concentrate (white star) is injected arthroscopically through the trocar and smoothed with the paddle portion of the trocar (white arrows). (B) Throughout this process, an assistant (black star) intermediately applies suction through the inferior spinal needle via a Frazier-tip suction device (white arrows), keeping the working area free of fluid. (C) Once the BioCartilage has been applied, fibrin glue (white star) is injected over the filled lesion via the superior spinal needle (white oval), sealing it in place. (D) Fibrin glue (white star) is given time to harden prior to removal of the scope and closure, and the remaining bone marrow aspirate is injected into the joint after closure of the portals.
Rehabilitation
Postoperatively, patients were allowed 50% partial weight bearing for 4 to 6 weeks without a brace. In patients with patellofemoral lesions, weight bearing was allowed as tolerated with a hinged knee brace locked in extension for 4 to 6 weeks. All patients were allowed passive range of motion as tolerated with isometric strengthening for 6 weeks, followed by a progressive strengthening program with return-to-sport protocols initiated at 3 to 4 months provided that full range of motion and strength were achieved.
Discussion
The healing potential of chondral defects has been shown to be considerably limited. Despite the current treatment options, the ideal treatment for chondral defects of the knee remains unclear. Recently, many authors have advocated treating chondral defects with biological therapies such as BMAC and scaffold-based treatments including BioCartilage. Whereas BMAC has gained particular attention because of its differentiation capacity and potential role in tissue regeneration, BioCartilage is an extracellular matrix scaffold that has been claimed to promote the regeneration of hyaline-like cartilage. Thus, we have presented our technique of arthroscopic chondral defect repair using BMAC and BioCartilage. Table 1 shows the pearls and pitfalls associated with this technique.
Table 1.
Pearls and Pitfalls of Arthroscopic BMAC and BioCartilage Treatment for Chondral Defects
| Step | Pearls | Pitfalls |
|---|---|---|
|
The surgeon can position the patient supine when BMAC is harvested from the contralateral ASIS to allow for simultaneous draping of the affected knee. | The surgeon should ensure that the needle position is at least 3 fingerbreadths posterior to the ASIS to avoid lateral femoral cutaneous nerve irritation. |
|
The needle trajectory should be parallel to the iliac crest, and the surgeon should grip the inner and outer table to ensure a safe trajectory. No culture expansion is required, and there is no risk of allogeneic disease transmission. Harvesting is technically easy to achieve and may be performed with concomitant procedures. |
The surgeon should ensure that the trocar and syringes are properly treated with heparin per the specific system’s protocol. |
|
The surgeon should use as many portals as necessary to allow for proper visualization and trocar trajectory. | The surgeon should ensure visualization of the chondral defect before proceeding. |
|
A shaver in forward mode should be used to adequately prepare a healthy bleeding bed of subchondral bone. | Compromised healing may occur if an adequate base of bleeding bone is not achieved. We recommend avoiding microfracture. |
|
Subchondroplasty is performed through a small incision in the subchondral bone marrow insufficiency correlating with orthogonal fluoroscopic views and preoperative MRI scans showing subchondral edema. | Overfilling of the lesion with calcium phosphate cement should be avoided. The appropriate aperture position should be ensured to avoid extravasation into the joint. Overfilling of calcium phosphate cement or improper technique can result in leakage into the joint cavity. |
|
The joint is thoroughly dried with the use of suction, the fluid inflow tubing is removed, and the stopcock on the arthroscope is left open to avoid closure of the soft-tissue space in the joint due to lack of airflow while suctioning. One spinal needle is placed above the lesion for application of fibrin glue, and another spinal needle is placed below to keep the area dry. | Improper placement of spinal needles can result in fluid gathering around the lesion and visualization obstruction from the anterior fat pad. |
|
The BioCartilage and BMAC mixture should be applied smoothly with a paddle to ensure even filling. | If the lesion is not cleared of fluid during filling, it can interfere with the healing process and mixture application. |
|
The fibrin glue should be applied on top of the mixture; use of a dual-lumen applicator is recommended to prevent clogging of fibrin in the needle. | A sufficient amount should be used to cover the mixture but over-application must be avoided because it can cause the construct to sit proud in the joint. |
ASIS, anterior superior iliac spine; BMAC, bone marrow aspirate concentrate; MRI, magnetic resonance imaging.
Few studies have evaluated the treatment of BMAC in conjunction with scaffold-based treatments for the treatment of cartilage defects. Gobbi and Whyte25 performed a prospective cohort study to assess patients with knee cartilage defects classified as grade IV by the International Cartilage Repair Society. One group of patients underwent treatment with MF (n = 50), whereas the other group received BMAC and a hyaluronic acid (HA)–based scaffold (n = 27). Higher functional outcome scores, as categorized by the Tegner score, International Knee Documentation Committee score, and Knee Injury and Osteoarthritis Outcomes Score, were found within the BMAC-HA group after 5 years of treatment. In another nonrandomized prospective trial, Gobbi et al.26 evaluated outcomes among 37 patients with patellofemoral chondral lesions. One group was treated with MACI (n = 19), whereas the other group was treated with BMAC and an HA-based scaffold (n = 18). After 3 years of follow-up, higher International Knee Documentation Committee subjective scores were found among the patients treated with BMAC and the HA-based scaffold. In contrast to our study, which used an extracellular matrix–based scaffold, these studies selected an HA-based scaffold. Another distinction from our study is that these studies performed a mini-arthrotomy as the approach for implantation. Our arthroscopic approach is intended to provide the advantages of a minimally invasive approach. Despite these differences, the aforementioned authors concluded that the concomitant treatment of BMAC with a scaffold is an appropriate option for the treatment of a chondral defect of the knee.
Our technique has its potential advantages and disadvantages24,27, 28, 29 (Table 2). For instance, it is performed arthroscopically, in contrast to an open procedure, which can be associated with more pain, greater blood loss, greater dissection, and a longer postoperative course. The less invasive nature of arthroscopy can potentially cause less pain, less blood loss, less dissection, and a potentially quicker recovery. Moreover, the use of BMAC and BioCartilage can potentially augment the healing process with growth factors and the provision of a tissue network to aid in cellular interactions. This theoretically would allow for better quality and durability of the restored cartilage. However, because this process takes time, several weeks to months may be required for patients begin to feel significant pain relief. The limitations of this technique with respect to defect size and depth are unclear and will require further investigation. Long-term and comparative studies using this technique are currently unavailable. Another disadvantage of this technique is that access to the lesion and maintenance of a dry environment for graft placement may be a technical challenge to perform arthroscopically.
Table 2.
Advantages and Disadvantages of Arthroscopic BMAC and BioCartilage Treatment for Chondral Defects
| Advantages | Disadvantages | |
|---|---|---|
| BMAC | Postoperative rehabilitation can begin immediately. The technique can provide growth factors and mesenchymal stem cells to begin the healing process in areas that lack blood supply and/or healing properties. Harvesting is easy, and no culture expansion is required. There is no risk of allogeneic disease transmission. |
Pain improvement can take 6-8 wk. Theoretically, NSAIDs may interfere with healing, which may require opioids; however, we allow for regular doses of NSAIDs and acetaminophen to avoid opioids postoperatively. Stem cell and growth factor quantity and quality are variable, depending on patient factors and harvest quality. |
| BioCartilage | The small particle size improves the ability to be injected and increases the surface area, providing attachment sites for bone marrow cells. A tissue network is provided that can signal autologous cellular interactions. The method is an inexpensive and has a shelf life of 5 yr. |
The use of this material is potentially questionable with larger and deeper lesions owing to poor integration of the graft. Abnormal bone growth, repair tissue growth, or detachment of the subchondral plate can result. |
BMAC, bone marrow aspirate concentrate; NSAIDs, nonsteroidal anti-inflammatory drugs.
In conclusion, we propose an arthroscopic technique with BMAC and BioCartilage implantation to treat patients with chondral defects of the knee. This technique is minimally invasive and is reproducible. With the goal of establishing a definitive treatment for chondral lesions of the knee, future studies should observe patients who undergo treatment with this technique and assess how other conventional techniques compare with it.
Footnotes
The authors report the following potential conflicts of interest or sources of funding: V.K.M. is a board or committee member of the New Jersey Orthopaedic Society and MD Advantage Board of Trustees and receives other support from Topical Gear, outside the submitted work. A.J.S. receives research support from ISO-biologics; receives personal fees from Mitek; and is a board or committee member of the New Jersey Orthopaedic Society, outside the submitted work. Full ICMJE author disclosure forms are available for this article online, as supplementary material.
Supplementary Data
Arthroscopic chondral defect repair with bone marrow aspirate concentrate (BMAC) and BioCartilage. The bone marrow aspirate is obtained from the iliac crest in a standard fashion. The trocar is inserted percutaneously 3 fingerbreadths posterior to the anterior superior iliac spine. This posterior access point protects the lateral femoral cutaneous nerve from injury. A total of 60 mL of bone marrow is aspirated by rotating the syringe 90° approximately every 5 mL. The bone marrow aspirate is then centrifuged down to yield 7 mL of BMAC. Standard diagnostic arthroscopy is performed using anteromedial and anterolateral portals. The area is debrided with a shaver in forward-down mode to a bed of healthy bleeding bone. A stable rim is also achieved. Next, subchondroplasty is performed behind the chondral defect under fluoroscopic guidance. After a small incision is made, a subchondroplasty trocar is inserted behind the chondral defect, which is confirmed by correlating orthogonal fluoroscopic views with preoperatively obtained magnetic resonance imaging scans showing subchondral edema. The trocar is inserted to an appropriate depth, and it is rotated such that the perforation is faced toward the joint line. In total, 3 mL of calcium phosphate cement is injected through the trocar, rotating the trocar while injecting to evenly fill the lesion. After this is completed, the inner sleeve is replaced and the trocar is left in place for 10 minutes to allow the cement time to harden. With subchondroplasty complete, attention is turned to BioCartilage application to the chondral defect. A spinal needle is placed percutaneously such that the tip rests just below the lesion; this will be used to keep the area dry during the procedure. Another spinal needle is placed percutaneously above the lesion; this will be used for injection of fibrin glue. By use of suction through the BioCartilage application paddle, the joint is fully evacuated of fluid, confirming that the anterior fat pad has been resected adequately and will not interfere with visualization of the lesion during the procedure. The BioCartilage mixed with 1 mL of BMAC is injected through the trocar and smoothed down to fill the lesion with the paddle. Throughout this process, an assistant intermediately applies suction to the inferior spinal needle via a Frazier-tip suction device, keeping the working area free of fluid. Once the BioCartilage has been applied, fibrin glue is injected over it via the superior spinal needle, sealing it in place. The fibrin glue is given time to harden prior to removal of the scope and closure. The remaining 6 mL of BMAC is injected into the joint after closure of the portals.
References
- 1.Hjelle K., Solheim E., Strand T., Muri R., Brittberg M. Articular cartilage defects in 1,000 knee arthroscopies. Arthroscopy. 2002;18:730–734. doi: 10.1053/jars.2002.32839. [DOI] [PubMed] [Google Scholar]
- 2.Widuchowski W., Widuchowski J., Trzaska T. Articular cartilage defects: Study of 25,124 knee arthroscopies. Knee. 2007;14:177–182. doi: 10.1016/j.knee.2007.02.001. [DOI] [PubMed] [Google Scholar]
- 3.Buckwalter J.A. Articular cartilage: Injuries and potential for healing. J Orthop Sports Phys Ther. 1998;28:192–202. doi: 10.2519/jospt.1998.28.4.192. [DOI] [PubMed] [Google Scholar]
- 4.Scillia A.J., Aune K.T., Andrachuk J.S. Return to play after chondroplasty of the knee in national football league athletes. Am J Sports Med. 2015;43:663–668. doi: 10.1177/0363546514562752. [DOI] [PubMed] [Google Scholar]
- 5.Everhart J.S., Campbell A.B., Abouljoud M.M., Kirven J.C., Flanigan D.C. Cost-efficacy of knee cartilage defect treatments in the United States. Am J Sports Med. 2020;48:242–251. doi: 10.1177/0363546519834557. [DOI] [PubMed] [Google Scholar]
- 6.Chui K., Jeys L., Snow M. Knee salvage procedures: The indications, techniques and outcomes of large chondral allografts. World J Orthop. 2015;6:340–350. doi: 10.5312/wjo.v6.i3.340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Richter D.L., Schenck R.C., Wascher D.C., Treme G. Knee articular cartilage repair and restoration techniques. Sports Health. 2016;8:153–160. doi: 10.1177/1941738115611350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Case J.M., Scopp J.M. Treatment of articular cartilage defects of the knee with microfracture and enhanced microfracture techniques. Sports Med Arthrosc. 2016;24:63–68. doi: 10.1097/JSA.0000000000000113. [DOI] [PubMed] [Google Scholar]
- 9.Goyal D., Keyhani S., Lee E.H., Hui J.H.P. Evidence-based status of microfracture technique: A systematic review of Level I and II studies. Arthroscopy. 2013;29:1579–1588. doi: 10.1016/j.arthro.2013.05.027. [DOI] [PubMed] [Google Scholar]
- 10.Andrade R., Vasta S., Pereira R. Knee donor-site morbidity after mosaicplasty—A systematic review. J Exp Orthop. 2016;3:31. doi: 10.1186/s40634-016-0066-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Derrett S., Stokes E.A., James M., Bartlett W., Bentley G. Cost and health status analysis after autologous chondrocyte implantation and mosaicplasty: A retrospective comparison. Int J Technol Assess Health Care. 2005;21:359–367. doi: 10.1017/s0266462305050476. [DOI] [PubMed] [Google Scholar]
- 12.de Windt T.S., Sorel J.C., Vonk L.A., Kip M.M.A., Ijzerman M.J., Saris D.B.F. Early health economic modelling of single-stage cartilage repair. Guiding implementation of technologies in regenerative medicine. J Tissue Eng Regen Med. 2017;11:2950–2959. doi: 10.1002/term.2197. [DOI] [PubMed] [Google Scholar]
- 13.Widuchowski W., Lukasik P., Kwiatkowski G. Isolated full thickness chondral injuries. Prevalance and outcome of treatment. A retrospective study of 5233 knee arthroscopies. Acta Chir Orthop Traumatol Cech. 2008;75:382–386. [PubMed] [Google Scholar]
- 14.Rizzello G., Longo U.G., Petrillo S. Growth factors and stem cells for the management of anterior cruciate ligament tears. Open Orthop J. 2012;6:525–530. doi: 10.2174/1874325001206010525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fortier L.A., Potter H.G., Rickey E.J. Concentrated bone marrow aspirate improves full-thickness cartilage repair compared with microfracture in the equine model. J Bone Joint Surg Am. 2010;92:1927–1937. doi: 10.2106/JBJS.I.01284. [DOI] [PubMed] [Google Scholar]
- 16.Ivkovic A., Pascher A., Hudetz D. Articular cartilage repair by genetically modified bone marrow aspirate in sheep. Gene Ther. 2010;17:779–789. doi: 10.1038/gt.2010.16. [DOI] [PubMed] [Google Scholar]
- 17.Kon E., Roffi A., Filardo G., Tesei G., Marcacci M. Scaffold-based cartilage treatments: With or without cells? A systematic review of preclinical and clinical evidence. Arthroscopy. 2015;31:767–775. doi: 10.1016/j.arthro.2014.11.017. [DOI] [PubMed] [Google Scholar]
- 18.Ferruzzi A., Buda R., Faldini C. Autologous chondrocyte implantation in the knee joint: Open compared with arthroscopic technique. Comparison at a minimum follow-up of five years. J Bone Joint Surg Am. 2008;90:90–101. doi: 10.2106/JBJS.H.00633. [DOI] [PubMed] [Google Scholar]
- 19.Manfredini M., Zerbinati F., Gildone A., Faccini R. Autologous chondrocyte implantation: A comparison between an open periosteal-covered and an arthroscopic matrix-guided technique. Acta Orthop Belg. 2007;73:207–218. [PubMed] [Google Scholar]
- 20.Zeifang F., Oberle D., Nierhoff C., Richter W., Moradi B., Schmitt H. Autologous chondrocyte implantation using the original periosteum-cover technique versus matrix-associated autologous chondrocyte implantation: A randomized clinical trial. Am J Sports Med. 2010;38:924–933. doi: 10.1177/0363546509351499. [DOI] [PubMed] [Google Scholar]
- 21.Carter A.H., Guttierez N., Subhawong T.K. MR imaging of BioCartilage augmented microfracture surgery utilizing 2D MOCART and KOOS scores. J Clin Orthop Trauma. 2018;9:146–152. doi: 10.1016/j.jcot.2017.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tyler T.F., Lung J.Y. Rehabilitation following chondral injury to the knee. Curr Rev Musculoskelet Med. 2012;5:72–81. doi: 10.1007/s12178-011-9108-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.da Cunha Cavalcanti F.M.M., Doca D., Cohen M., Ferretti M. Updating on diagnosis and treatment of chondral lesion of the knee. Rev Bras Ortop. 2012;47:12–20. doi: 10.1016/S2255-4971(15)30339-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chahla J., Mannava S., Cinque M.E., Geeslin A.G., Codina D., LaPrade R.F. Bone marrow aspirate concentrate harvesting and processing technique. Arthrosc Tech. 2017;6:e441–e445. doi: 10.1016/j.eats.2016.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gobbi A., Whyte G.P. One-stage cartilage repair using a hyaluronic acid-based scaffold with activated bone marrow-derived mesenchymal stem cells compared with microfracture: Five-year follow-up. Am J Sports Med. 2016;44:2846–2854. doi: 10.1177/0363546516656179. [DOI] [PubMed] [Google Scholar]
- 26.Gobbi A., Chaurasia S., Karnatzikos G., Nakamura N. Matrix-induced autologous chondrocyte implantation versus multipotent stem cells for the treatment of large patellofemoral chondral lesions: A nonrandomized prospective trial. Cartilage. 2015;6:82–97. doi: 10.1177/1947603514563597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Orthopaedic & Neurosurgery Specialists Bone marrow aspirate concentrate (BMAC) https://onsmd.com/bone-marrow-aspirate-concentrate-bmca/
- 28.Arthrex BioCartilage extracellular matrix. https://www.arthrex.com/orthobiologics/BioCartilage-extracellular-matrix
- 29.Schallmo M.S., Marquez-Lara A., Luo T.D., Rosas S., Stubbs A.J. Arthroscopic treatment of hip chondral defect with microfracture and platelet-rich plasma–infused micronized cartilage allograft augmentation. Arthrosc Tech. 2018;7:e361–e365. doi: 10.1016/j.eats.2017.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Arthroscopic chondral defect repair with bone marrow aspirate concentrate (BMAC) and BioCartilage. The bone marrow aspirate is obtained from the iliac crest in a standard fashion. The trocar is inserted percutaneously 3 fingerbreadths posterior to the anterior superior iliac spine. This posterior access point protects the lateral femoral cutaneous nerve from injury. A total of 60 mL of bone marrow is aspirated by rotating the syringe 90° approximately every 5 mL. The bone marrow aspirate is then centrifuged down to yield 7 mL of BMAC. Standard diagnostic arthroscopy is performed using anteromedial and anterolateral portals. The area is debrided with a shaver in forward-down mode to a bed of healthy bleeding bone. A stable rim is also achieved. Next, subchondroplasty is performed behind the chondral defect under fluoroscopic guidance. After a small incision is made, a subchondroplasty trocar is inserted behind the chondral defect, which is confirmed by correlating orthogonal fluoroscopic views with preoperatively obtained magnetic resonance imaging scans showing subchondral edema. The trocar is inserted to an appropriate depth, and it is rotated such that the perforation is faced toward the joint line. In total, 3 mL of calcium phosphate cement is injected through the trocar, rotating the trocar while injecting to evenly fill the lesion. After this is completed, the inner sleeve is replaced and the trocar is left in place for 10 minutes to allow the cement time to harden. With subchondroplasty complete, attention is turned to BioCartilage application to the chondral defect. A spinal needle is placed percutaneously such that the tip rests just below the lesion; this will be used to keep the area dry during the procedure. Another spinal needle is placed percutaneously above the lesion; this will be used for injection of fibrin glue. By use of suction through the BioCartilage application paddle, the joint is fully evacuated of fluid, confirming that the anterior fat pad has been resected adequately and will not interfere with visualization of the lesion during the procedure. The BioCartilage mixed with 1 mL of BMAC is injected through the trocar and smoothed down to fill the lesion with the paddle. Throughout this process, an assistant intermediately applies suction to the inferior spinal needle via a Frazier-tip suction device, keeping the working area free of fluid. Once the BioCartilage has been applied, fibrin glue is injected over it via the superior spinal needle, sealing it in place. The fibrin glue is given time to harden prior to removal of the scope and closure. The remaining 6 mL of BMAC is injected into the joint after closure of the portals.