Abstract
Islet transplantation is regarded as the most promising treatment for type 1 diabetes (T1D). However, the function of grafted islet could be damaged on account of transplant rejection and/or hypoxia several years later after transplantation. We proposed a hypothetical functionalized hydrogel model, which encapsulates sufficient A20 high-expressing islets and supporting cells, and performs as a drug release system releasing immunosuppressants and growth factors, to improve the outcome of pancreatic islet transplantation. Once injected in vivo, the hydrogel can gel and offer a robust mechanical structure for the A20 high-expressing islets and supporting cells. The natural biomaterials (eg, heparin) added into the hydrogel provide adhesive sites for islets to promote islets’ survival. Furthermore, the hydrogel encapsulates various supporting cells, which can facilitate the vascularization and/or prevent the immune system attacking the islet graft. Based on the previous studies that generally applied one or two combined strategies to protect the function of islet graft, we designed this hypothetical multifunctional encapsulation hydrogel model with various functions. We hypothesized that the islet graft could survive and maintain its function for a longer time in vivo compared with naked islets. This hypothetical model has a limitation in terms of clinical application. Future development work will focus on verifying the function and safety of this hypothetical islet transplantation hydrogel model in vitro and in vivo.
Keywords: islet transplantation, hydrogel, T1D, encapsulation, biomaterials
Introduction
Type 1 diabetes (T1D) is characterized by the destruction of insulin-secreting β cells by autoimmunity, which increases blood sugar (glucose) levels and results in a high incidence of life-long diabetes-related complications.1,2 T1D mostly affects young people and is an increasing health issue with an estimated prevalence ranged between 5% and 15% of the total cases of diabetes mellitus worldwide.3 Currently, life-long administration of exogenous insulin on daily is the primary treatment for T1D to maintain blood glucose close to the normal range and to reduce complications associated with the disease.4 However, insulin replacement therapy for patients with T1D cannot restore normal glucose homeostasis and leads to a high risk of secondary complications. It has been confirmed that the transplantation of pancreas or islets could reach the best therapeutic outcome.5 The first case of clinical pancreas transplantation was performed by Kelly et al at the University of Minnesota in the late 1960s.6 Since the “Edmonton protocol” with glucocorticoid-free immunosuppressive regimen was made, islet transplantation has become available for clinical treatment strategy.7 However, less than 20% of the transplant recipients remain the independence of insulin 3–5 years later after this clinical procedure.8 The main reason for loss of β cells after transplantation is the immune-mediated inflammatory reaction and the scarcity of oxygen. After islet transplantation, the rejection and recurrence of autoimmunity impair islet’s function and eventually result in graft loss.9 To overcome the limitation of clinical islet transplantation, various strategies have been studied recently. It was confirmed that the hydrogel made from various biomaterials for encapsulating islets could facilitate the functionality and stability of islets.10
The hydrogel used for encapsulating islets consists of natural biomaterials and synthetic biomaterials. The natural hydrogels are biocompatible. Polymers occurring crosslinked naturally include fibrin, collagen, fibronectin, alginate, heparan sulfate, chitosan, or hyaluronic acid. Among the natural materials, alginate is widely used in islet encapsulation; it shows a degree of biocompatibility and can be crosslinked in divalent cations solution.11,12 Typical synthetic hydrogels include PEG (polyethylene glycol), PEG-PLA (polyethylene glycol-polylactic acid), PEGDA (polyethylene glycol diacrylate) and PVA (polyvinyl alcohol), PHEMA-MMA (polyhydroxyethylmethacrylate-methyl methacrylate) and so on.13 In tissue engineering applications, synthetic hydrogels have advantages over natural polymer hydrogels due to their adjustable properties (eg, porosity, stability, mechanical strength, and biocompatibility).14 Among the synthetic hydrogels, PEG and its derivatives are the most widely used in islet encapsulation.15,16 The matrix pore size of PEG nanofilm on the surface of islet is tunable. The concentration and molecular weight of precursor PEG diacrylate can adjust the optimal pore size before gelation.17 The pore size can be adjusted to exclude large immune antibodies, while permitting glucose and insulin to pass through. Herein, PEG was introduced into the hypothetical islet encapsulating model.
Based on the size of pores in the hydrogel, the islet encapsulating models can be classified into cell penetration restrictive devices and cell-permissive devices.10 Cell penetration restrictive devices limit the migration of immune cells to pass through the pores of immune-isolating nanofilm. Through that way, the doses of immunosuppressive can be reduced or avoided when islet graft is encapsulated in cell penetration restrictive devices. Lou et al18 developed an ultrathin nanofilm of star-PEG with incorporated heparin; it protected implanted cells from host immune rejections and modified the cellular landscape for better post-transplantation graft function and survival. Haque et al19 designed a novel method camouflaging porcine islets with PEG and its derivatives; this method could prevent islets dissociation and the invasion of various immune cells after transplantation. The camouflaged xenograft survived and sustained its function in mice for average 13.0 days. Cell-permissive devices are not equipped with the function of immune-isolating but are open to the bloodstream. Compared with cell penetration restrictive devices, the cell-permissive device can provide more oxygen and nutrition to the transplant. Different strategies for improving in situ vascularization can be applied to the cell-permissive devices, such as embedding vascular endothelial growth factor (VEGF) into the hydrogel to promote the rebuild of blood vessels.16
The hydrogel of encapsulating islet graft has emerged as an encouraging platform to eliminate the need for immunosuppressive and improve oxygen supplement, thereby allowing the wilder application of islet transplant. Although encapsulated islets with various polymeric hydrogels have shown tremendous promise in many studies, some deficiencies of this technology remain unsolved.
The Deficiencies of Current Islet Encapsulation
The main obstacles of islet encapsulation are poverty of vascularization around islet graft and host immune attack activated by the transplant. The two main obstacles cause the loss of a large percentage of islets after transplantation. Islets of Langerhans are vulnerable to hypoxia. Physically, the blood supply for islets is quite abundant. Islets are around 2% of the weight of the pancreas. However, they are rendered over 5 fold higher blood flow than the exocrine pancreas.20 Apart from providing nutrients and oxygen to islet, abundant blood supply enhances insulin sensitivity.21 The islets used for transplantation have lost its extracellular matrix (ECM) during the procedure of isolation; new blood vessels need to be established timely to maintain the survival of islets after transplanted into recipients. Cell-permissive devices are designed to maximize vascularization for islet graft encapsulated, but cannot prevent islet graft from contacting with immune cells which incurs antigen recognition by the immune system of the host.
Early islet loss after transplantation mainly results from immune rejection. The mechanism of immune rejection includes instant blood-mediated inflammatory reaction (IBMIR).22 It is reported that 50% of islet xenografts are rejected within 9 days after transplantation without immunosuppressants in animal models.23 Cell penetration restrictive devices can prevent infiltration and recognition of immune cells by selective permeability established by nano-sized pores on the surface of the devices. However, Brauker et al24 demonstrated that 5 μm pores, compared to 20 nm pores, were 80 folds more effective in promoting the growth of vessels.
Design of the Hypothetical Hydrogel Model
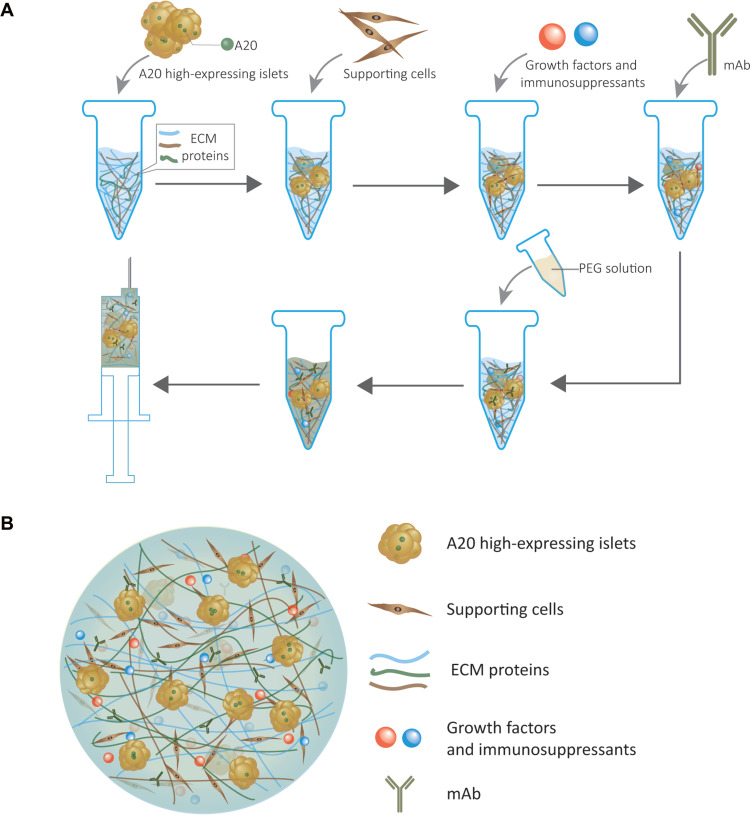
We proposed a hypothetical islet transplantation hydrogel model, which serves as not only a supporting culture matrix for islets but also a drug delivery system for releasing immunosuppressants and growth factors (Figure 1). The islets encapsulated in the model are genetically modified to increase the expression of A20, which can induce immune tolerance to survive longer.
Figure 1.
(A) Illustration of the hypothetical islet transplantation hydrogel model fabrication protocol. (B) Schematic illustrating the components of the hypothetical islet transplantation hydrogel model.
Ubiquitin editing protein A20 encoded by TNFAIP3 is a negative regulator of immunostimulatory factors.25 A20 is an ideal cytoprotective gene therapy candidate for islet transplantation. Grey et al found that genetic engineering of a suboptimal islet graft with A20 preserves β cell mass and function.26 Reducing expression of A20 in immune cells (eg, B cells) results in hyperactivation of the immune system.27 Zammit et al28 developed an islet cell line that can overexpress A20 via an adenoviral vector encoding human A20, and transplanted it beneath the kidney capsule in diabetic C57BL/6 mice. Their findings indicated that the forced expression of A20 reduced inflammation and allowed permanent islet allograft survival without the necessity of immunosuppression. A20 protects cells from apoptosis by stabilizing the linear (M1) ubiquitin network, which is associated with TNFR1 signaling complex when TNF binds to TNFR1.29 Hypoxia and inflammatory cause β cell destruction in the early post-transplantation period. Genetic engineering of islets with A20 will relieve the severe poverty of islet for clinical transplantation and improve their function in the early post-transplantation period.26 Benefited with the protection of A20,26 a relatively reduced islet transplant mass could be available in the hypothetical model. In this model, we proposed to transfer A20 gene (TNFAIP3) into islets by a novel safer and tissue-specific recombinant adenovirus vector (rAd.).28 However, the toxicity of adenoviruses may impede them to be used widely.
This novel model is based on injectable hydrogel, which consists of natural biomaterials and synthetic biomaterials. As the synthetic biomaterial, PEG can provide a robust mechanical structure to the model by covalent crosslinking. Before being crosslinked, several natural biomaterials (eg, collagen, hyaluronate, and heparin) will be added into the solution of PEG to gain better biocompatibility. Once injected, hydrogel formation occurs through chemical cross-linking. Collagen, hyaluronate, and heparin are all ECM proteins, which can compensate for the loss of matrix around islets during the procedure of isolation, and mimic the physiological microenvironment of islets. Furthermore, islets can be anchored in the hydrogel through the adhesion sites on the ECM proteins, which makes the islets stable.30 Among various ECM proteins, heparin is an excellent choice for the hydrogel model. Heparin can help reduce the inflammatory mediated by anticoagulation, and the thiolated heparin can combine various growth factors (eg, VEGF, bFGF, PDGF, IGF-2, and KGF) by ionic bond; the growth factors can slowly release from the hydrogel model for several weeks.31,32
As a drug delivery system, the model can encapsulate various growth factors. There are tremendous advantages that growth factors can bring to islets. The functionality of islets can be improved by the bond of IGF-2 and IGF-2 membrane receptors on the surface of islets. IGF-2 is a growth factor favorable for islet survival; the combination of IGF-2 and its receptor prevents islet aggregation during islet transplantation.33 Additionally, using growth factors embedded in hydrogels can stimulate blood vessels ingrowth. Basic fibroblast growth factor (bFGF) is identified to promote angiogenesis, which is beneficial to reconstruct new capillaries around islets when transplanted into recipients. Adding bFGF into recombinant human collagen could accelerate revascularization, and reduce the hypoxia-induced damage to islets.34 Various functional cells can be co-encapsulated with islets into this hypothetical model, and be co-transplanted in vivo. Mesenchymal stem cells (MSCs) are widely used to be co-transplanted with islets.35–39 When co-transplanted with islets in recipients, MSCs can promote angiogenesis in situ to prevent hypoxia and to attenuate immune rejection. MSCs can suppress various immune cells such as NK cells, macrophages, neutrophils, and T cells.35,40–43 Moreover, it was identified that MSCs could accelerate the maturation of neonatal porcine islets, which benefits the development of islets xenograft.39 The establishment of immune tolerance to islet grafts, is a significant strategy for inducing acceptance of histocompatibility complex (MHC)-mismatched allografts without compromising the host’s immune system. T regulatory cells (Tregs) play a central role in maintaining immune homeostasis and peripheral tolerance to foreign antigens.44 Herein, Tregs can also be added into the model as an immuno-modulatory strategy to improve islet grafts’ function. Multipotent adult progenitor cells can also be a candidate for co-transplantation with islets due to its similarities to MSCs in immunosuppressive and angiogenic advantages.45 When transplanted with islets, endothelial progenitor cells were confirmed to promote neovascularization around the islets graft.46
The drugs used in the immunosuppressive regimen for islets transplantation, such as sirolimus (mTOR inhibitor), can also be encapsulated in this hydrogel. Monoclonal antibodies can also be involved. For example, the anti-CD154 blockade possesses the potency of achieving transplantation tolerance, which results in the withdraw of chemical immunosuppressants.47 IL-2R antibody can also be added to the drug candidate list.
With the above-proposed strategies, the islet transplantation model provides a delivery vehicle for co-transplantation of A20 high-expressing islets and other supporting cells and the controlled release of immunosuppressants and growth factors. The injectable PEG hydrogel for islet encapsulation has been characterized in vitro and in vivo, illustrated the feasibility of using injectable PEG hydrogels for islet encapsulation.48 Herein, this multifunctional hydrogel model is a covalently cross-linked injectable hydrogel. Moreover, it is flowable before gelation. Hydrogel gelation was induced by the PEG crosslinker. The PEG crosslinker must be added before injection, and gels were left for a certain time to allow for complete gelation.
Conclusion
Islet transplantation is regarded as the most promising treatment for type 1 diabetes. Unfortunately, the clinical application of islet transplantation is limited mainly due to hypoxia and the immune-mediated destruction by the host. To overcome the obstacles, we design an islet encapsulated hydrogel model with the function of the controlled release of immunosuppressants and growth factors. Sufficient A20 high-expressing islets are encapsulated in this model to reverse the condition of hyperglycemia. However, as a macro-transplantation model, it could lack oxygen in its core. Therefore, we combined several strategies to reconstruct the vascularization. Co-transplantation with supporting cells and incorporation of various growth factors are identified to be beneficial to the revascularization around transplanted islets. Also, supporting cells (MSCs) can serve as an anti-immune factor to protect islets from immune attack by host. Given to the digestion of the extracellular matrix proteins around islets during isolation, the naked islets tend to aggregate together, resulting in the increasing risk of hypoxia in the core of the islet mass and leading to the loss of islet function. To address this issue, we add various ECM proteins to the hydrogel mimicking the natural microenvironment around islets, and the islet graft will be immobilized. The hydrogel can also serve as a materials platform to immobilize growth factors. However, as a cell-permissive device, the hydrogel model permits the islet to contact blood to get sufficient oxygen, which results in IBMIR. When IBMIR happens, the platelet will aggregate, and coagulation will occur, which are detrimental to the viability of islets.49 The particular gene-editing islets encapsulated in the model can express a high level of A20, and the threshold for the activation of immune response will be upregulated by the activation of NF-κB.28 The application of A20 high-expressing islets can achieve long-term survival in vivo.
In summary, based on the previous studies that generally applied one or two combined strategies to protect the function of islet graft, we proposed the multifunctional encapsulation hydrogel model with various functions to achieve a better outcome of islet transplantation. This hypothetical hydrogel model was designed with cocktail therapy; the combination of various factors might promote the function of the islet graft, compensate deficiency for each other. This hypothetical model might be injected into several suitable sites in vivo, such as the beneath of kidney capsule, the peritoneal cavity, and a subcutaneous site on the back.48,50 In the model, the hydrogel will be injected into body before gelation, then capillaries will creep into the hydrogel. With the release of immunosuppressants embedded in the model, the local immune isolation will be established. This hypothetical multifunctional hydrogel model has a limitation in terms of clinical application. Future development work will focus on verifying the function and safety of this hypothetical islet transplantation hydrogel model in vitro and in vivo.
Funding Statement
This study was partially supported by the Science and Technology Research Program of Chongqing Municipal Education Commission (KJQN201800418, KJQN201900418), the Project of Science and Technology of Chongqing Yuzhong District (20190114). The opinions, findings, and conclusions or recommendations expressed in this study are those of the authors and do not necessarily reflect the views of the funding agencies.
Disclosure
The authors declare no potential conflicts of interest for this work.
References
- 1.Ernst AU, Wang LH, Ma M. Interconnected toroidal hydrogels for islet encapsulation. Adv Healthc Mater. 2019;8(12):e1900423. doi: 10.1002/adhm.201900423 [DOI] [PubMed] [Google Scholar]
- 2.Nathan DM, Bayless M, Cleary P, et al. Diabetes control and complications trial/epidemiology of diabetes interventions and complications study at 30 years: advances and contributions. Diabetes. 2013;62(12):3976–3986. doi: 10.2337/db13-1093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Espona-Noguera A, Ciriza J, Canibano-Hernandez A, et al. Review of advanced hydrogel-based cell encapsulation systems for insulin delivery in type 1 diabetes mellitus. Pharmaceutics. 2019;11(11):597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vantyghem M-C, de Koning EJ, Pattou F, Rickels MR. Advances in β-cell replacement therapy for the treatment of type 1 diabetes. Lancet. 2019;394(10205):1274–1285. doi: 10.1016/S0140-6736(19)31334-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Farney AC, Sutherland DE, Opara EC. Evolution of islet transplantation for the last 30 years. Pancreas. 2016;45(1):8–20. doi: 10.1097/MPA.0000000000000391 [DOI] [PubMed] [Google Scholar]
- 6.Kelly WD, Lillehei RC, Merkel FK, Idezuki Y, Goetz FC. Allotransplantation of the pancreas and duodenum along with the kidney in diabetic nephropathy. Surgery. 1967;61(6):827–837. [PubMed] [Google Scholar]
- 7.Shapiro AM, Lakey JR, Ryan EA, et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N Engl J Med. 2000;343(4):230–238. doi: 10.1056/NEJM200007273430401 [DOI] [PubMed] [Google Scholar]
- 8.Phelps EA, Headen DM, Taylor WR, Thule PM, Garcia AJ. Vasculogenic bio-synthetic hydrogel for enhancement of pancreatic islet engraftment and function in type 1 diabetes. Biomaterials. 2013;34(19):4602–4611. doi: 10.1016/j.biomaterials.2013.03.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Piemonti L, Everly MJ, Maffi P, et al. Alloantibody and autoantibody monitoring predicts islet transplantation outcome in human type 1 diabetes. Diabetes. 2013;62(5):1656–1664. doi: 10.2337/db12-1258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ernst AU, Bowers DT, Wang LH, et al. Nanotechnology in cell replacement therapies for type 1 diabetes. Adv Drug Deliv Rev. 2019;139:116–138. doi: 10.1016/j.addr.2019.01.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vegas AJ, Veiseh O, Gurtler M, et al. Long-term glycemic control using polymer-encapsulated human stem cell-derived beta cells in immune-competent mice. Nat Med. 2016;22(3):306–311. doi: 10.1038/nm.4030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Duvivier-Kali VF, Omer A, Parent RJ, O’Neil JJ, Weir GC. Complete protection of islets against allorejection and autoimmunity by a simple barium-alginate membrane. Diabetes. 2001;50(8):1698–1705. doi: 10.2337/diabetes.50.8.1698 [DOI] [PubMed] [Google Scholar]
- 13.Shrestha P, Regmi S, Jeong J-H. Injectable hydrogels for islet transplantation: a concise review. J Pharm Investig. 2019;50(1):29–45. doi: 10.1007/s40005-019-00433-3 [DOI] [Google Scholar]
- 14.Guan X, Avci‐Adali M, Alarçin E, et al. Development of hydrogels for regenerative engineering. Biotechnol J. 2017;12(5):1600394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Weaver JD, Headen DM, Hunckler MD, Coronel MM, Stabler CL, Garcia AJ. Design of a vascularized synthetic poly(ethylene glycol) macroencapsulation device for islet transplantation. Biomaterials. 2018;172:54–65. doi: 10.1016/j.biomaterials.2018.04.047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Marchioli G, Zellner L, Oliveira C, et al. Layered PEGDA hydrogel for islet of Langerhans encapsulation and improvement of vascularization. J Mater Sci Mater Med. 2017;28(12):195. doi: 10.1007/s10856-017-6004-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cruise GM, Scharp DS, Hubbell JAJB. Characterization of permeability and network structure of interfacially photopolymerized poly (ethylene glycol) diacrylate hydrogels. Biomaterials. 1998;19(14):1287–1294. doi: 10.1016/S0142-9612(98)00025-8 [DOI] [PubMed] [Google Scholar]
- 18.Lou S, Zhang X, Zhang J, Deng J, Kong D, Li C. Pancreatic islet surface bioengineering with a heparin-incorporated starPEG nanofilm. Mater Sci Eng C Mater Biol Appl. 2017;78:24–31. doi: 10.1016/j.msec.2017.03.295 [DOI] [PubMed] [Google Scholar]
- 19.Haque MR, Jeong JH, Byun Y. Combination strategy of multi-layered surface camouflage using hyperbranched polyethylene glycol and immunosuppressive drugs for the prevention of immune reactions against transplanted porcine islets. Biomaterials. 2016;84:144–156. doi: 10.1016/j.biomaterials.2016.01.039 [DOI] [PubMed] [Google Scholar]
- 20.Jansson L, Carlsson PO. Pancreatic blood flow with special emphasis on blood perfusion of the islets of Langerhans. Compr Physiol. 2019;9(2):799–837. [DOI] [PubMed] [Google Scholar]
- 21.Hogan MF, Hull RL. The islet endothelial cell: a novel contributor to beta cell secretory dysfunction in diabetes. Diabetologia. 2017;60(6):952–959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Özmen L, Ekdahl KN, Elgue G, Larsson R, Korsgren O, Nilsson B. Inhibition of thrombin abrogates the instant blood-mediated inflammatory reaction triggered by isolated human islets: possible application of the thrombin inhibitor melagatran in clinical islet transplantation. Diabetes. 2002;51(6):1779–1784. [DOI] [PubMed] [Google Scholar]
- 23.Triponez F, Oberholzer J, Morel P, et al. Xenogeneic islet re-transplantation in mice triggers an accelerated, species-specific rejection. Immunology. 2000;101(4):548–554. doi: 10.1046/j.1365-2567.2000.00130.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brauker JH, Carr-Brendel VE, Martinson LA, Crudele J, Johnston WD, Johnson RC. Neovascularization of synthetic membranes directed by membrane microarchitecture. J Biomed Mater Res. 1995;29(12):1517–1524. doi: 10.1002/jbm.820291208 [DOI] [PubMed] [Google Scholar]
- 25.Wertz IE, Newton K, Seshasayee D, et al. Phosphorylation and linear ubiquitin direct A20 inhibition of inflammation. Nature. 2015;528(7582):370–375. doi: 10.1038/nature16165 [DOI] [PubMed] [Google Scholar]
- 26.Grey ST, Longo C, Shukri T, et al. Genetic engineering of a suboptimal islet graft with A20 preserves β cell mass and function. J Immunol. 2003;170(12):6250–6256. doi: 10.4049/jimmunol.170.12.6250 [DOI] [PubMed] [Google Scholar]
- 27.Chu Y, Vahl JC, Kumar D, et al. B cells lacking the tumor suppressor TNFAIP3/A20 display impaired differentiation and hyperactivation and cause inflammation and autoimmunity in aged mice. Blood. 2011;117(7):2227–2236. doi: 10.1182/blood-2010-09-306019 [DOI] [PubMed] [Google Scholar]
- 28.Zammit NW, Walters SN, Seeberger KL, O’Connell PJ, Korbutt GS, Grey ST. A20 as an immune tolerance factor can determine islet transplant outcomes. JCI Insight. 2019;4(21):e131028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Priem D, Devos M, Druwe S, et al. A20 protects cells from TNF-induced apoptosis through linear ubiquitin-dependent and -independent mechanisms. Cell Death Dis. 2019;10(10):692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Krishnamurthy M, Li J, Fellows GF, Rosenberg L, Goodyer CG, Wang RJE. Integrin α3, but not β1, regulates islet cell survival and function via PI3K/Akt signaling pathways. Endocrinology. 2011;152(2):424–435. [DOI] [PubMed] [Google Scholar]
- 31.Pike DB, Cai S, Pomraning KR, et al. Heparin-regulated release of growth factors in vitro and angiogenic response in vivo to implanted hyaluronan hydrogels containing VEGF and bFGF. Biomaterials. 2006;27(30):5242–5251. doi: 10.1016/j.biomaterials.2006.05.018 [DOI] [PubMed] [Google Scholar]
- 32.Alkhoury H, Hautmann A, Fuhrmann B, et al. Studies on the mechanisms of anti-inflammatory activity of heparin- and hyaluronan-containing multilayer coatings-targeting NF-kappaB signalling pathway. Int J Mol Sci. 2020;21(10):3724. doi: 10.3390/ijms21103724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Forget A, Waibel M, Rojas-Canales DM, et al. IGF-2 coated porous collagen microwells for the culture of pancreatic islets. J Mater Chem B. 2017;5(2):220–225. doi: 10.1039/C6TB02748B [DOI] [PubMed] [Google Scholar]
- 34.Zhu Q, Lu C, Jiang X, et al. Using recombinant human collagen with basic fibroblast growth factor to provide a simulated extracellular matrix microenvironment for the revascularization and attachment of islets to the transplantation region. Front Pharmacol. 2020;10:1536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ishida N, Ishiyama K, Saeki Y, Tanaka Y, Ohdan H. Cotransplantation of preactivated mesenchymal stem cells improves intraportal engraftment of islets by inhibiting liver natural killer cells in mice. Am J Transplant. 2019;19(10):2732–2745. [DOI] [PubMed] [Google Scholar]
- 36.Bal T, Nazli C, Okcu A, Duruksu G, Karaoz E, Kizilel S. Mesenchymal stem cells and ligand incorporation in biomimetic poly(ethylene glycol) hydrogels significantly improve insulin secretion from pancreatic islets. J Tissue Eng Regen Med. 2017;11(3):694–703. doi: 10.1002/term.1965 [DOI] [PubMed] [Google Scholar]
- 37.Kuppan P, Seeberger K, Kelly S, et al. Co-transplantation of human adipose-derived mesenchymal stem cells with neonatal porcine islets within a prevascularized subcutaneous space augments the xenograft function. Xenotransplantation. 2020;e12581. [DOI] [PubMed] [Google Scholar]
- 38.Ren G, Rezaee M, Razavi M, Taysir A, Wang J, Thakor AS. Adipose tissue-derived mesenchymal stem cells rescue the function of islets transplanted in sub-therapeutic numbers via their angiogenic properties. Cell Tissue Res. 2019;376(3):353–364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.He S, Wang C, Du X, et al. MSCs promote the development and improve the function of neonatal porcine islet grafts. FASEB J. 2018;32(6):3242–3253. [DOI] [PubMed] [Google Scholar]
- 40.Madec AM, Mallone R, Afonso G, et al. Mesenchymal stem cells protect NOD mice from diabetes by inducing regulatory T cells. Diabetologia. 2009;52(7):1391–1399. doi: 10.1007/s00125-009-1374-z [DOI] [PubMed] [Google Scholar]
- 41.Yin Y, Hao H, Cheng Y, et al. Human umbilical cord-derived mesenchymal stem cells direct macrophage polarization to alleviate pancreatic islets dysfunction in type 2 diabetic mice. Cell Death Dis. 2018;9(7):760. doi: 10.1038/s41419-018-0801-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vasandan AB, Jahnavi S, Shashank C, Prasad P, Kumar A, Prasanna SJ. Human Mesenchymal stem cells program macrophage plasticity by altering their metabolic status via a PGE2-dependent mechanism. Sci Rep. 2016;6:38308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Magana-Guerrero FS, Dominguez-Lopez A, Martinez-Aboytes P, Buentello-Volante B, Garfias Y. Human amniotic membrane mesenchymal stem cells inhibit neutrophil extracellular traps through TSG-6. Sci Rep. 2017;7(1):12426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Krzystyniak A, Gołąb K, Witkowski P, Trzonkowski P. Islet cell transplant and the incorporation of Tregs. Curr Opin Organ Transplant. 2014;19(6):610–615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cunha JP, Leuckx G, Sterkendries P, et al. Human multipotent adult progenitor cells enhance islet function and revascularisation when co-transplanted as a composite pellet in a mouse model of diabetes. Diabetologia. 2017;60(1):134–142. doi: 10.1007/s00125-016-4120-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Grapensparr L, Christoffersson G, Carlsson PO. Bioengineering with endothelial progenitor cells improves the vascular engraftment of transplanted human islets. Cell Transplant. 2018;27(6):948–956. doi: 10.1177/0963689718759474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lee SJ, Kim HJ, Byun NR, Park CG. Donor-specific regulatory T cell-mediated immune tolerance in an intrahepatic murine allogeneic islet transplantation model with short-term anti-CD154 mAb single treatment. Cell Transplant. 2020;29:963689720913876. doi: 10.1177/0963689720913876 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Knobeloch T, Abadi SEM, Bruns J, Zustiak SP, Kwon G. Injectable polyethylene glycol hydrogel for islet encapsulation: an in vitro and in vivo characterization. Biomed Phys Eng Express. 2017;3:035022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Teramura Y, Oommen OP, Olerud J, Hilborn J, Nilsson B. Microencapsulation of cells, including islets, within stable ultra-thin membranes of maleimide-conjugated PEG-lipid with multifunctional crosslinkers. Biomaterials. 2013;34(11):2683–2693. doi: 10.1016/j.biomaterials.2013.01.015 [DOI] [PubMed] [Google Scholar]
- 50.Estil Les E, Tellez N, Nacher M, Montanya EA. Model for human islet transplantation to immunodeficient streptozotocin-induced diabetic mice. Cell Transplant. 2018;27(11):1684–1691. doi: 10.1177/0963689718801006 [DOI] [PMC free article] [PubMed] [Google Scholar]