Abstract
Introduction
Angiotensin-converting enzyme 2 (ACE2) provides an adhesion site for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Patients with COPD could have severe outcomes after SARS-CoV-2 infection. The objective of this study was to investigate ACE2 regulation by air pollution during the development of COPD.
Methods
Sprague Dawley rats were exposed to unconcentrated traffic-related air pollution for 3 and 6 months. We examined lung injury markers, oxidative stress, inflammation, emphysema, ACE2 and angiotensin II receptor type 1 (AT1) and 2 (AT2) in the lungs after exposure.
Results
Lung injury occurred due to an increase in permeability and lactate dehydrogenase cytotoxicity was observed after 6 months of exposure to fine particulate matter of <1 μm in aerodynamic diameter (PM1). An α1-antitrypsin deficiency and neutrophil elastase production with emphysema development were observed after 6 months of PM1 exposure. 8-isoprostane and interleukin-6 were increased after 3 and 6 months of PM1 exposure. Caspase-3 was increased after exposure to PM1 for 6 months. Upregulation of ACE2 was found after 3 months of PM1 exposure; however, ACE2 had decreased by 6 months of PM1 exposure. AT1 and AT2 had significantly decreased after exposure to PM1 for 6 months. Furthermore, smooth muscle hypertrophy had occurred after 6 months of PM1 exposure.
Conclusions
In conclusion, short-term exposure to PM1 increased the ACE2 overexpression in lungs. Long-term exposure to PM1 decreased the ACE2 overexpression in emphysema. Air pollution may be a risk for SARS-CoV-2 adhesion during the development of COPD.
Short abstract
Short-term exposure to PM1 increases ACE2 overexpression in lungs. Long-term exposure to PM1 decreases the ACE2 overexpression in emphysema. Air pollution may be a risk for #SARSCoV2 adhesion during the development of COPD. https://bit.ly/2Vfykur
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) or coronavirus disease 2019 (COVID-19) has been declared a public health emergency of international concern by the World Health Organization. The SARS-CoV-2 epidemic has resulted in 334 981 confirmed cases with 14 652 deaths reported (as of 24 March 2020). SARS-CoV-2 was identified as a highly infectious novel coronavirus [1], which is highly infectious in adults and older adults. A recent report indicated a lower incidence rate of SARS-CoV-2 infection in patients with chronic lung diseases [2]; however, patients with a chronic lung disease could have severe outcomes once they are infected.
A report indicated that SARS-CoV-2 uses angiotensin-converting enzyme 2 (ACE2) for entry and the serine protease TMPRSS2 for S protein priming [3]. The S1 domain contains the ACE2 receptor-binding domain, which is responsible for host cell entry in the first stage [4]. The S2 domain facilitates fusion between the cell and virus membrane, which is required for cellular infiltration [5]. Recent studies demonstrated that the modified S protein of SARS-CoV-2 has a higher affinity for ACE2 [6]. SARS-CoV-2 is likely to bind to ACE2 in human cells [6]. ACE2 is involved in the synthesis of bioactive components of the renin–angiotensin system (RAS). The RAS is involved in regulating proinflammatory pathways as well as reactive stress in inflammatory-related diseases [7]. ACE2 acts as an anti-inflammatory protein by counteracting the actions of angiotensin II (AngII) [8]. Therefore, alteration in ACE2 may not only result in regulation of SARS-CoV-2 infection ability, but also associate with inflammatory responses.
COPD is known to cause deficits in ACE2 in the lungs [9, 10]. Air pollution is a risk factor for the development of COPD, which is also suspected to be associated with the SARS-CoV-2 infection ability [11, 12]. The objective of this study was to investigate the expressions of ACE2 by air pollution during the development of COPD. The data can explain the role of air pollution in SARS-CoV-2 infections.
Materials and methods
Animals
Male 6-month-old Sprague Dawley rats were obtained from the National Laboratory Animal Center (Taipei, Taiwan), and were housed at a constant temperature of 22±2°C and a relative humidity of 55±10% with a 12-h light–dark cycle. Animal experiments were conducted in compliance with the Animal and Ethics Review Committee of the Laboratory Animal Center at Taipei Medical University (Taipei, Taiwan).
Chronic air pollution exposure
The experimental design was previously reported [13]. Rats were randomly divided into three groups: 1) a clean-air control group housed in the Laboratory Animal Center of Taipei Medical University (Taipei, Taiwan); 2) a high-efficiency particulate air (HEPA) group (exposed to gaseous pollution only; New Taipei City, Taiwan); and 3) a PM1 (particulate matter <1 μm in aerodynamic diameter) exposure group (exposed to particulate and gaseous pollution; New Taipei City, Taiwan). There were 16 rats in each group in this study. Rats were whole-body exposed to PM1 using a whole-body exposure system for 3 and 6 months. The whole-body exposure system for rodents was described previously [13, 14] and was located at a traffic-dominated area near a highway and an expressway. PM1 mass (by a tapered element oscillating microbalance) and number concentrations (by a scanning mobility particle sizer and average particle size), black carbon (by an aethalometer) and lung deposition surface area concentrations (by a nanoparticle surface area monitor) were continuously monitored during the exposure period. Gaseous pollution was referenced from the nearby Taiwan Environmental Protection Administration Yonghe air quality monitoring station (https://taqm.epa.gov.tw/taqm/en/default.aspx). The left lung was used for bronchoalveolar lavage fluid (BALF) and tissue collection, whereas the right lung was used for histology. An animal necropsy was performed and tissues were collected as described previously [15].
Biochemistry
Total protein (Thermo Fisher Scientific, Waltham, MA, USA) and lactate dehydrogenase (LDH) cytotoxicity (Thermo Fisher Scientific) were determined in BALF. α1-antitrypsin (Cusabio, Wuhan, Hubei, China), neutrophil elastase (Cusabio), 8-isoprostane (Cayman, Ann Arbor, MI, USA), interleukin (IL)-6 (R&D Systems, Minneapolis, MN, USA), caspase-3 (Elabscience, Houston, TX, USA), ACE2 (Elabscience), AngII receptor type 1 (AT1) (Elabscience) and AngII receptor type 2 (AT2) (Elabscience) were determined in lung lysates using ELISA. All analyses were in accordance with the manufacturers’ instructions. Data obtained from lung lysates are presented after adjusting for the β-actin level (Cell Signaling, Danvers, MA, USA).
Histology and emphysema score
Haematoxylin and eosin staining of the lungs was performed according to our previous report [16]. The mean linear intercept (MLI), an indicator of the mean alveolar diameter, was assessed in 10 nonoverlapping fields according to a previous report [17]. Histological examinations were conducted under light microscopy.
Statistical analysis
Comparisons among multiple groups were performed using an ANOVA with Tukey's post hoc test. Statistical analyses were performed using GraphPad version 5 for Windows. The level of significance was set to p<0.05. Data are expressed as the mean±sd.
Results
Emphysema occurred after PM1 exposure
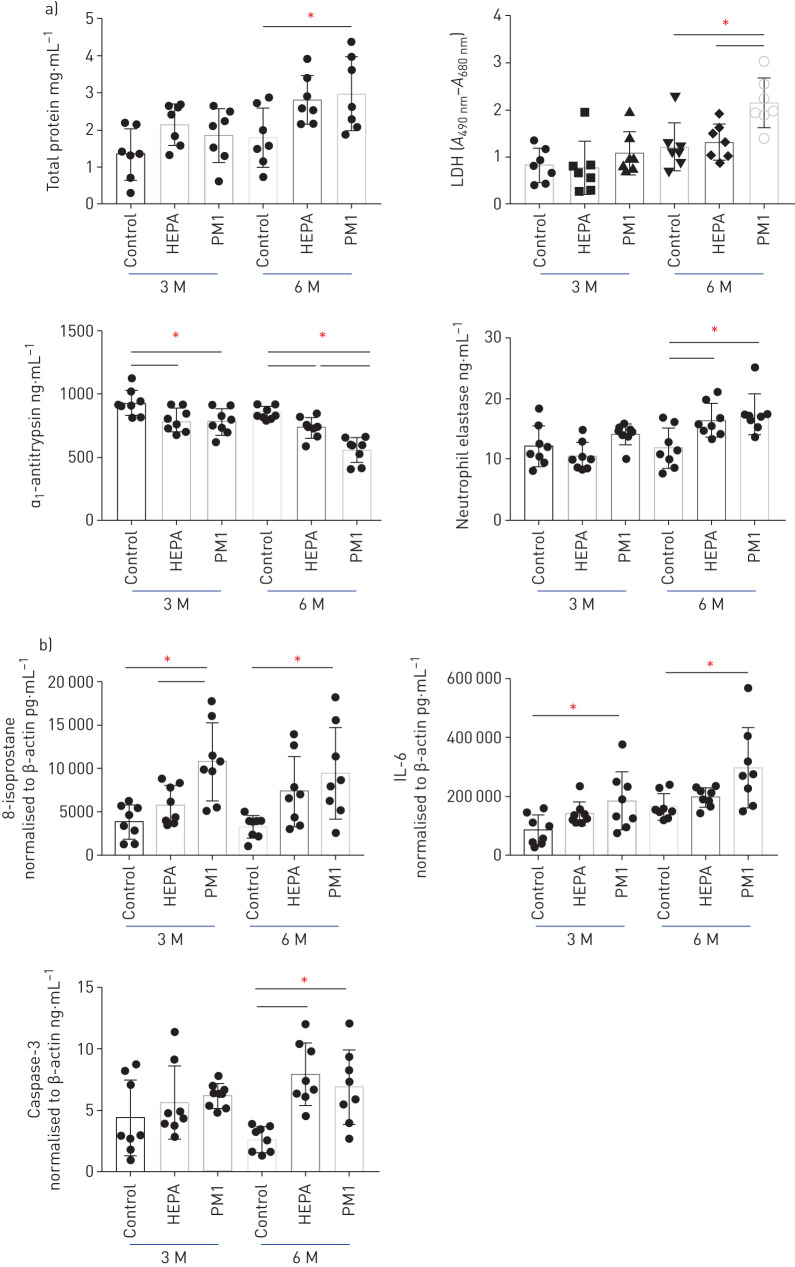
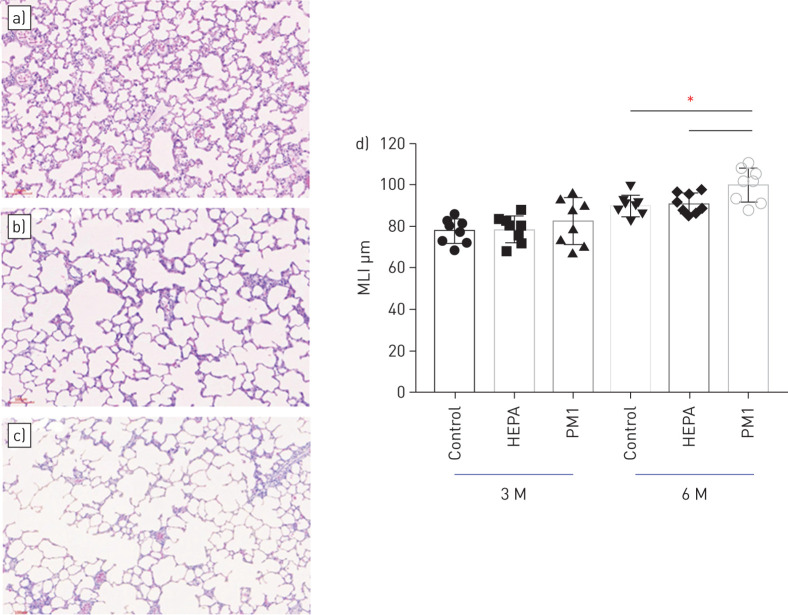
Rats were whole-bodily exposed to unconcentrated traffic-related air pollution for 3 and 6 months. After exposure, we observed that the total protein and LDH in BALF had significantly increased at 6 months compared to the control and the HEPA groups (p<0.05) (figure 1a). Levels of α1-antitrypsin had significantly decreased in both the HEPA and PM1 groups after exposure (p<0.05), especially at 6 months (figure 1a), whereas neutrophil elastase had significantly increased after 6 months of exposure (p<0.05). Levels of 8-isoprostane and IL-6 were significantly increased by 3 and 6 months of PM1 exposure (p<0.05) (figure 1b). We observed that caspase-3 had significantly increased in rats after PM1 exposure for 6 months (p<0.05) (figure 1b). Next, we examined the MLI score in lungs after 3 and 6 months of exposure (figure 2). The MLI score had significantly increased due to PM1 exposure compared to the control and HEPA groups at 6 months (p<0.05).
FIGURE 1.
Levels of a) total protein, lactate dehydrogenase (LDH), α1-antitrypsin and neutrophil elastase in bronchoalveolar lavage fluid (BALF) and b) 8-isoprostane, interleukin (IL)-6 and caspase-3 in lungs after 3 and 6 months (M) of exposure to traffic-related ambient air. Rats were exposed to particulate matter with an aerodynamic diameter <1 µm (PM1; PM1 group) or high-efficiency particulate air (HEPA)-filtered air (HEPA group, gaseous pollution). Rats in the control group were housed in the animal centre. n=8 per group. *: p<0.05.
FIGURE 2.
Haematoxylin and eosin staining of lungs after 6 months (M), from a) control rats, and from rats exposed to b) high-efficiency particulate air (HEPA)-filtered air or c) particulate matter with an aerodynamic diameter <1 μm (PM1). d) Emphysema mean linear intercept (MLI) score after air pollution exposure. Significant emphysema occurred after 6 months of PM1 exposure in rats. n=8 per group. *: p<0.05.
ACE2 regulation by PM1 exposure
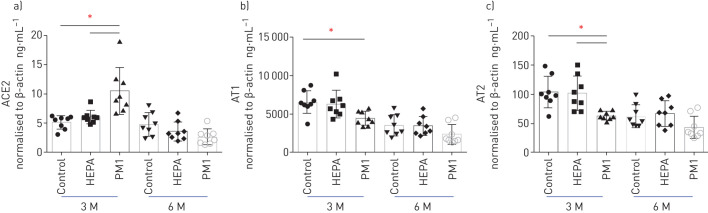
Expression of ACE2 was examined in rat lungs after air pollution exposure (figure 3). Levels of ACE2 had increased at 3 months of PM1 exposure (p<0.05). However, ACE2 was significantly decreased at 6 months of PM1 exposure (p<0.05).
FIGURE 3.
Levels of a) angiotensin-converting enzyme 2 (ACE2), b) angiotensin II receptor type 1 (AT1) and c) angiotensin II receptor type 2 (AT2) in rat lungs after 3 and 6 months (M) of exposure to traffic-related ambient air. Rats were exposed to high-efficiency particulate air (HEPA)-filtered air or particulate matter with an aerodynamic diameter <1 μm (PM1). Rats in the control group were housed in the animal centre. n=8 per group. *: p<0.05.
Smooth muscle hypertrophy occurred after PM1 exposure
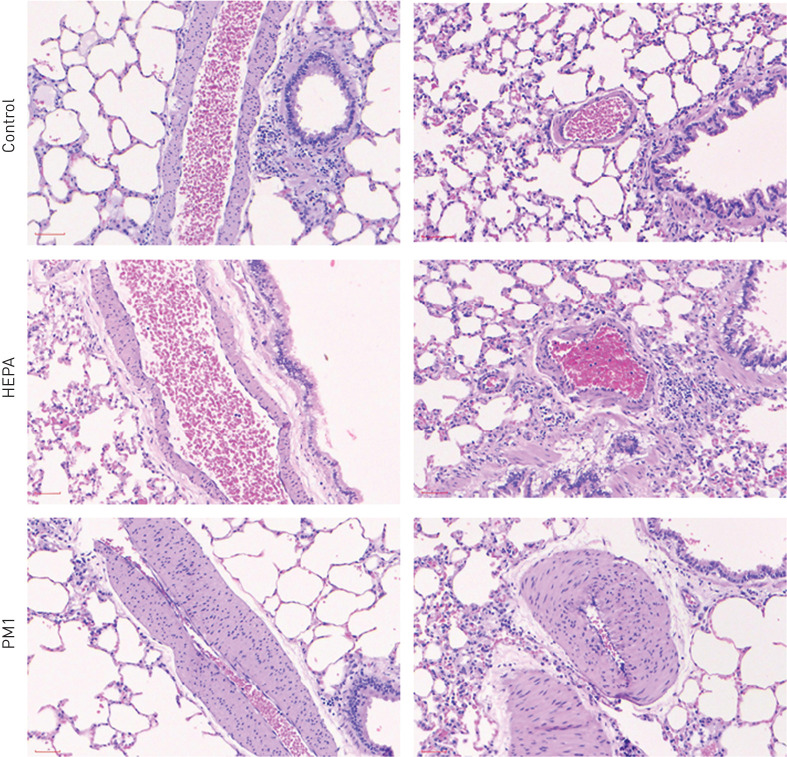
Both AT1 and AT2 had significantly decreased by PM1 at 3 months (p<0.05) (figure 3). Furthermore, we observed that smooth muscle hypertrophy occurred after 6 months of PM1 exposure in rat lungs (figure 4).
FIGURE 4.
Haematoxylin and eosin staining of blood vessels after 6 months of air pollution exposure. Rats were exposed to high-efficiency particulate air (HEPA)-filtered air or particulate matter with an aerodynamic diameter <1 μm (PM1). Rats in the control group were housed in the animal centre. Two different sections of blood vessels are shown for each group. A significant increase in smooth muscle hypertrophy occurred after 6 months of PM1 exposure in rats.
Discussion
Air pollution has been reported to increase the risk of SARS-CoV-2 infection [11, 12]. ACE2 is considered to play an important role in regulation of SARS-CoV-2 infection ability [18]. In the present study, we observed that 3 months of exposure to fine particles (PM1) upregulated ACE2 levels of the lungs. The 6 months of exposure to air pollution, particularly PM1, caused an ACE2 deficiency in the lungs with the occurrence of emphysema-combined smooth muscle hypertrophy in rats. Our results suggest that: 1) short-term exposure to PM1 increased the ACE2 overexpression in lungs, and 2) long-term exposure to PM1 decreased the ACE2 overexpression in emphysema. The alteration in ACE2 expression of lungs may associate with SARS-CoV-2 infection during emphysema development.
Air pollution has been identified as a risk factor for COPD; however, the scientific evidence is mainly based on epidemiological evidence. Currently, there is still no natural route for air pollution exposure in a rodent model. To our best knowledge, this is the first report to study ACE2 expression in a rodent model with 6 months’ inhalation of unconcentrated traffic-related ambient air using a whole-body exposure system. Our previous study indicated that the whole-body exposure system had the higher penetration rate of PM (from outdoor to exposure system) with a particle size <1 μm; therefore PM1 was the main size fraction for the whole-body exposure in this system [13, 14]. Rats were exposed to an average of 16.3 μg·m−3 PM1, a particle number concentration of 11 257 particles·m−3, 1800 ng·m−3 of black carbon and 55.1 μm2·cm−3 of lung deposition surface area in the alveolar region during the exposure period [13]. The mean geometric mean diameter was 55.8 nm during the study period. The average ambient PM2.5 was 19.7±9.8 (obtained from the Yonghe air quality monitoring station). The PM1 to PM2.5 ratio 0.827, which is commonly observed from traffic-dominated urban area. The correlation (r2) between PM1 and PM2.5 was 0.829 (p<0.001) (supplementary figure S1), suggested that the rats were mainly exposed to the outdoor air pollution during the experimental periods. Gaseous pollutants averaged 32.9 ppb for oxides of nitrogen, 2.5 ppb for SO2 and 29.7 ppb for O3. After 6 months of exposure to PM1, the permeability (total protein) and cytotoxicity (LDH) of the lungs had increased. For the pathogenesis of COPD, we observed an α1-antitrypsin deficiency and neutrophil elastase elevation in the lungs due to PM1 exposure. The data suggest that subchronic and chronic PM1 exposure may cause extensive tissue damage and malfunctioning of the airways. We next investigated the development of emphysema, which is considered a phenotype of air pollution-induced COPD [19], after exposure in rats. First, we observed elevated levels of caspase-3 by PM1 and gaseous pollutants. The data showed activation of apoptosis in lung cells after exposure. Consistent with caspase-3 activation, emphysema was induced by 6 months of PM1 exposure. Our data indicated that emphysema occurred after 6 months of exposure to PM1.
SARS-CoV-2 uses ACE2 receptors to invade cells, particularly in the lungs and heart [20]. SARS-CoV-2 shares similar mechanisms with other coronaviruses, and the outer membrane spike glycoprotein is the prime host-interacting protein with host cell targets [21]. ACE2, for example, plays important roles in cell adhesion and virulence [22]. In the present study, we first observed that 3 months of exposure to PM1 increased ACE2 expression in rat lungs. This observation is similar to that with smoking in a previous report [20]. Brake et al. [20] indicated that smoking upregulated ACE2 expression, which provides an adhesion site for SARS-CoV-2, leading to an increased risk of SARS-CoV-2 infection. In contrast, we observed ACE2 deficiencies after 6 months of exposure to PM1 in the development of emphysema. A report indicated that ACE2 mRNA expression is significantly reduced in COPD rat lung [23]. ACE2 overexpression mitigated reduction of lung function and pathological manifestations of COPD [10]. The reduction of ACE2 in emphysema could decrease the risk of SARS-CoV-2 infection. However, increasing reports indicated that COPD patients had higher severity after SARS-CoV-2 infection [24, 25].
The particle effects on ACE2 were also found in a previous study, which indicated that ACE2-knockdown mice showed increased pulmonary phosphorylated signal transducer and activator of transcription 3 (STAT3) and phosphorylated extracellular signal-regulated kinase (ERK) 1/2 levels after fine particle-induced acute lung injury [26]. It is worth noting that emphysema occurred after 6 months of exposure to PM1. The ACE2 deficiency implies that RAS dysregulation may play a pivotal role in the development of COPD. Inflammation is also an adverse effect resulting from ACE2 reduction in the lungs. The ACE2 deficiency may cause uncontrolled oxidative stress and inflammatory responses in COPD. Indeed, we observed that 8-isoprostane and IL-6 persisted at higher levels in lungs even after 6 months of PM1 exposure. Such adverse effects by ACE2 deficiency may induce more severe outcomes in COPD patients once they have been infected by SARS-CoV-2. Therefore, although there may be low levels of ACE2 adhesion sites for SARS-CoV-2, there may be higher levels of inflammatory responses in COPD after 6 months of PM1 exposure.
ACE2 is elevated during episodes of lung inflammation, which is a consequence of an increase in the vascular permeability. The main active peptides of the RAS include AngII, AngIII, AngIV and Ang-1–7. AngII and Ang-1–7 play more important roles in terms of health and disease [7]. Recently, ACE2 and their peptides were reported to play key roles in inflammatory processes in conditions such as pulmonary hypertension [27]. Therefore, in the present study, we further investigated AT1 and AT2 expression. We observed reductions in AT1 and AT2 after 3 months of PM1 exposure. Our observations were consistent with a previous study of placental structures [28], which suggested that PM1 altered the RAS pathway. Downregulation of AT1 and AT2 by PM1 could result in vasodilation and vasoconstriction dysfunction. Importantly, we observed smooth muscle hypertrophy of the pulmonary artery after 6 months of PM1 exposure. An association of air pollution with hypertrophy was also evident in cardiovascular diseases [29, 30]; however, this is the first study to identify smooth muscle hypertrophy of lungs by PM1 exposure. Together, patients with pulmonary hypertension could have low risk for SARS-CoV-2 infection.
The strength of this study is that the ACE2 expression during air pollution-induced emphysema was investigated. ACE2 has different expression between short-term and long-term PM1 exposure. We further showed that smooth muscle hypertrophy occurred due to PM1. The limitations of this study included that the contributions of physicochemical characteristics of PM1 to COPD remain unclear. Expression of other proteins such as TMPRSS2 and cathepsin L was not determined in the present study. A confirmatory experiment may need to be conducted on SARS-CoV-2 adhesion sites in the future. SARS-CoV-2 might replicate more efficiently in cells in COPD; therefore, leading to more severe outcomes.
Conclusions
In conclusion, we provide biological evidence to explain how short-term exposure to PM1 upregulated ACE2 for SARS-CoV-2 adhesion sites. Moreover, long-term exposure to PM1 downregulated ACE2 in emphysema. Air pollution may be a risk factor for SARS-CoV-2 infection during the development of COPD.
Supplementary material
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
FIGURE S1 Correlation between PM1 exposure to rats and ambient PM2.5. 00174-2020.figureS1 (115.3KB, pdf)
Acknowledgements
The authors heartily thank Yi-Syuan Lin (School of Respiratory Therapy, Taipei Medical University) and Zhe-Wei Lin (Graduate Institute of Environmental Engineering, National Taiwan University) for technical assistance during this project.
Footnotes
This article has supplementary material available from openres.ersjournals.com
Author contributions: H-C. Chuang contributed to the interpretation of the data and the manuscript. H-C. Chuang, K-Y. Lee, H-P. Kuo and P-H. Feng contributed substantially to the concept, design, interpretation of the data, and completion of the study and manuscript. Y-Y. Chen and J-K. Chen contributed substantially to the completion of the study. T-C. Hsiao contributed to the air quality monitoring. H-C. Chuang contributed to the histological scoring. S-C. Ho and K-J. Chuang contributed to the biochemical analysis. All authors contributed to critically revising the manuscript for important intellectual content. All authors have read and approved the final manuscript.
Conflict of interest: H-C. Chuang has nothing to disclose.
Conflict of interest: Y-Y. Chen has nothing to disclose.
Conflict of interest: T-C. Hsiao has nothing to disclose.
Conflict of interest: H-C. Chou has nothing to disclose.
Conflict of interest: H-P. Kuo has nothing to disclose.
Conflict of interest: P-H. Feng has nothing to disclose.
Conflict of interest: S-C. Ho has nothing to disclose.
Conflict of interest: J-K. Chen has nothing to disclose.
Conflict of interest: K-J. Chuang has nothing to disclose.
Conflict of interest: K-Y. Lee has nothing to disclose.
Support statement: This study was funded by the Ministry of Science and Technology of Taiwan (108-2314-B-038-093) and Taipei Medical University (DP2-107-21121-T-04). Funding information for this article has been deposited with the Crossref Funder Registry.
References
- 1.Solomon R, Katzir O, Egozi Y, et al. Identification of two distinct protein carboxyl methyltransferases in eucaryotic cells. FEBS Lett 1988; 241: 131–135. doi: 10.1016/0014-5793(88)81045-7 [DOI] [PubMed] [Google Scholar]
- 2.Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382: 1708–1720. doi: 10.1056/NEJMoa2002032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020; 181: 271–280. doi: 10.1016/j.cell.2020.02.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li F, Li W, Farzan M, et al. Structure of SARS coronavirus spike receptor-binding domain complexed with receptor. Science 2005; 309: 1864–1868. doi: 10.1126/science.1116480 [DOI] [PubMed] [Google Scholar]
- 5.Coutard B, Valle C, de Lamballerie X, et al. The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade. Antiviral Res 2020; 176: 104742. doi: 10.1016/j.antiviral.2020.104742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wrapp D, Wang N, Corbett KS, et al. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020; 367: 1260–1263. doi: 10.1126/science.abb2507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gaddam RR, Chambers S, Bhatia M. ACE and ACE2 in inflammation: a tale of two enzymes. Inflamm Allergy Drug Targets 2014; 13: 224–234. doi: 10.2174/1871528113666140713164506 [DOI] [PubMed] [Google Scholar]
- 8.Boehm M, Nabel EG. Angiotensin-converting enzyme 2 – a new cardiac regulator. N Engl J Med 2002; 347: 1795–1797. doi: 10.1056/NEJMcibr022472 [DOI] [PubMed] [Google Scholar]
- 9.O'Reilly PJ, Ding Q, Akthar S, et al. Angiotensin-converting enzyme defines matrikine-regulated inflammation and fibrosis. JCI Insight 2017; 2: e91923. doi: 10.1172/jci.insight.91923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xue T, Wei N, Xin Z, et al. Angiotensin-converting enzyme-2 overexpression attenuates inflammation in rat model of chronic obstructive pulmonary disease. Inhal Toxicol 2014; 26: 14–22. doi: 10.3109/08958378.2013.850563 [DOI] [PubMed] [Google Scholar]
- 11.Zhu Y, Xie J, Huang F, et al. Association between short-term exposure to air pollution and COVID-19 infection: evidence from China. Sci Total Environ 2020; 727: 138704. doi: 10.1016/j.scitotenv.2020.138704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Domingo JL, Rovira J. Effects of air pollutants on the transmission and severity of respiratory viral infections. Environ Res 2020; 187: 109650. doi: 10.1016/j.envres.2020.109650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shih C-H, Chen J-K, Kuo L-W, et al. Chronic pulmonary exposure to traffic-related fine particulate matter causes brain impairment in adult rats. Part Fibre Toxicol 2018; 245: 226–234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hsiao TC, Chang J, Wang JY, et al. Serum neurofilament light polypeptide is a biomarker for inflammation in cerebrospinal fluid caused by fine particulate matter. Aerosol Air Qual Res 2020; 20: 1665–1674. [Google Scholar]
- 15.Li N, Wang M, Bramble LA, et al. The adjuvant effect of ambient particulate matter is closely reflected by the particulate oxidant potential. Environ Health Perspect 2009; 117: 1116–1123. doi: 10.1289/ehp.0800319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chuang HC, Juan HT, Chang CN, et al. Cardiopulmonary toxicity of pulmonary exposure to occupationally relevant zinc oxide nanoparticles. Nanotoxicology 2014; 8: 593–604. doi: 10.3109/17435390.2013.809809 [DOI] [PubMed] [Google Scholar]
- 17.Munoz-Barrutia A, Ceresa M, Artaechevarria X, et al. Quantification of lung damage in an elastase-induced mouse model of emphysema. Int J Biomed Imaging 2012; 2012: 734734. doi: 10.1155/2012/734734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Diaz JH. Hypothesis: angiotensin-converting enzyme inhibitors and angiotensin receptor blockers may increase the risk of severe COVID-19. J Travel Med 2020; 27: taaa041. doi: 10.1093/jtm/taaa041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang M, Aaron CP, Madrigano J, et al. Association between long-term exposure to ambient air pollution and change in quantitatively assessed emphysema and lung function. JAMA 2019; 322: 546–556. doi: 10.1001/jama.2019.10255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Brake SJ, Barnsley K, Lu W, et al. Smoking upregulates angiotensin-converting enzyme-2 receptor: a potential adhesion site for novel coronavirus SARS-CoV-2 (Covid-19). J Clin Med 2020; 9: 841. doi: 10.3390/jcm9030841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vankadari N, Wilce JA. Emerging WuHan (COVID-19) coronavirus: glycan shield and structure prediction of spike glycoprotein and its interaction with human CD26. Emerg Microbes Infect 2020; 9: 601–604. doi: 10.1080/22221751.2020.1739565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Song W, Gui M, Wang X, et al. Cryo-EM structure of the SARS coronavirus spike glycoprotein in complex with its host cell receptor ACE2. PLoS Pathog 2018; 14: e1007236. doi: 10.1371/journal.ppat.1007236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jia H. Pulmonary angiotensin-converting enzyme 2 (ACE2) and inflammatory lung disease. Shock 2016; 46: 239–248. doi: 10.1097/SHK.0000000000000633 [DOI] [PubMed] [Google Scholar]
- 24.Lippi G, Henry BM. Chronic obstructive pulmonary disease is associated with severe coronavirus disease 2019 (COVID-19). Respir Med 2020; 167: 105941–105941. doi: 10.1016/j.rmed.2020.105941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Halpin DMG, Faner R, Sibila O, et al. Do chronic respiratory diseases or their treatment affect the risk of SARS-CoV-2 infection? Lancet Respir Med 2020; 8: 436–438. doi: 10.1016/S2213-2600(20)30167-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lin CI, Tsai CH, Sun YL, et al. Instillation of particulate matter 2.5 induced acute lung injury and attenuated the injury recovery in ACE2 knockout mice. Int J Biol Sci 2018; 14: 253–265. doi: 10.7150/ijbs.23489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dai H, Jiang L, Xiao Z, et al. ACE2-angiotensin-(1–7)-Mas axis might be a promising therapeutic target for pulmonary arterial hypertension. Nat Rev Cardiol 2015; 12: 374. doi: 10.1038/nrcardio.2015.6-c1 [DOI] [PubMed] [Google Scholar]
- 28.Soto SF, Melo JO, Marchesi GD, et al. Exposure to fine particulate matter in the air alters placental structure and the renin-angiotensin system. PLoS One 2017; 12: e0183314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Leary PJ, Kaufman JD, Barr RG, et al. Traffic-related air pollution and the right ventricle. The multi-ethnic study of atherosclerosis. Am J Respir Crit Care Med 2014; 189: 1093–1100. doi: 10.1164/rccm.201312-2298OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu Y, Goodson JM, Zhang B, et al. Air pollution and adverse cardiac remodeling: clinical effects and basic mechanisms. Front Physiol 2015; 6: 162. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
FIGURE S1 Correlation between PM1 exposure to rats and ambient PM2.5. 00174-2020.figureS1 (115.3KB, pdf)