Abstract
Paraquat is a highly toxic pesticide, which often causes pulmonary interstitial fibrosis after poisoning, and there is no specific antidote. At present, limited studies have reported that tacrolimus, as an immunosuppressant, can inhibit pulmonary fibrosis, but the specific mechanism remains unknown. The aim of the present study was to demonstrate the effect of tacrolimus on the TGF-β1 pathway associated with pulmonary fibrosis in paraquat exposed alveolar type II epithelial cells, and to identify the antipulmonary fibrosis mechanism of tacrolimus The rat alveolar epithelial type II RLE-6TN cell line was exposed to paraquat and treated with or without tacrolimus for 24 h, or with a TGF-β1 receptor type I/II inhibitor (LY2109761) for 1, 4, 8 or 16 h. MTT assays were used to detect the viability of rat alveolar type II epithelial cells under these different treatment conditions, while the concentrations of TGF-β1, SMAD3, SMAD7 and connective tissue growth factor (CTGF) in the cell culture supernatant were determined using ELISAs. Additionally, reverse transcription-quantitative PCR and immunofluorescence were used to analyze the mRNA and protein expression levels of TGF-β1, SMAD3, CTGF and SMAD7. The results demonstrated that the inhibition of the proliferation of RLE-6TN cells exposed to 200 nmol/l paraquat was 26.05±2.99%. The inhibition rate of 10 ng/ml tacrolimus on paraquat-exposed alveolar type II epithelial cells was 18.40±3.49%. The inhibition rate caused by 5 µmol/l LY2109761 was 26.56±4.49%. The expression levels of TGF-β1, SMAD3 and CTGF, as well as their concentrations in the culture supernatant, were significantly downregulated in the tacrolimus group compared with the paraquat group. However, both the concentration and expression levels of SMAD7 were significantly upregulated in the tacrolimus group compared with the paraquat group. In conclusion, tacrolimus can reduce the levels of TGF-β1, SMAD3 and CTGF, increase the level of SMAD7 in TGF-β1 signaling pathway and protect the development of pulmonary fibrosis in paraquat exposed alveolar epithelial cells.
Keywords: tacrolimus, paraquat, alveolar type II epithelial cell, transforming growth factor-β1, pulmonary fibrosis
Introduction
Although paraquat is one of the most widely used herbicides, it is highly toxic and lethal (1,2). Paraquat poisoning has been identified to cause severe damage to the lungs, digestive tract, liver, kidney, heart and brain (3). Nonetheless, lung damage is the most apparent, which often culminates in death due to respiratory failure (3). In fact, the survival of the patients with paraquat poisoning is notably affected by the development of pulmonary interstitial fibrosis (4,5). Current treatments for paraquat poisoning include hormones, antioxidants, immunosuppressants and hemoperfusion (6,7). Cyclophosphamide is also commonly used as an immunosuppressive therapy for paraquat poisoning (8,9); however, it can cause severe adverse effects (10).
Tacrolimus (also known as FK506) is a macrolide immunosuppressive drug, which inhibits the transcription of several cytokine genes (NF-AT, IL-2) by binding with calcineurin (11). Tacrolimus was discovered to inhibit T cell activation and T helper cell-dependent B cell proliferation and it has been widely used in organ transplantation and for the treatment of numerous types of autoimmune disease, such as myasthenia gravis (11–14). However, to the best of our knowledge, only a few studies have documented the use of tacrolimus for the treatment of paraquat-induced lung injury and its underlying mechanism of action remains unclear. A previous study by Nagano et al (15) indicated that tacrolimus inhibited collagen synthesis in murine pulmonary fibroblasts treated with bleomycin. In addition, Staab-Weijnitz et al (16) demonstrated that the intracellular chaperone FK506 binding protein 10 (FKBP10) was upregulated in idiopathic pulmonary fibrosis stromal fibroblasts. Furthermore, FKBP10 inhibition in primary idiopathic pulmonary fibrosis fibroblasts reduced the expression of fibrinogen and prevented collagen synthesis.
Fibrosis of various organs is a highly complex process. Previous studies have suggested that the transforming growth factor-β1/SMAD signaling pathway served an important role in the process of organ fibrosis (17,18). It has been shown that the upregulation of TGF-β1 is associated with the pathological changes of pulmonary fibrosis and the severity of the disease. Notably, TGF-β1 increased the formation of connective tissue and prevented its degradation through the SMAD signaling, thereby initiating pulmonary fibrosis (17,18).
After paraquat enters the human body, alveolar type I and II epithelial cells will actively ingest paraquat, causing serious lung injury (1). The present study aimed to determine the role of tacrolimus as a putative therapeutic agent for pulmonary injury in rat alveolar epithelial type II cells (RLE-6TN cells) exposed to paraquat. The potential effects of tacrolimus were evaluated in vitro by analyzing the expression levels of inflammatory factors involved in the TGF-β1/SMAD signaling pathway and known to serve a role in pulmonary fibrosis. The present findings may provide supporting evidence for the use of tacrolimus in the treatment of pulmonary interstitial fibrosis induced by paraquat.
Materials and methods
Main reagents
Paraquat and tacrolimus were purchased from Shanghai Aladdin Bio-Chem Technology Co., Ltd. High glucose DMEM and FBS were obtained from HyClone; Cytiva. MTT reagent was purchased from Amresco, LLC. Rat TGF-β1 (cat. no. CSB-E04727r), SMAD3 (cat. no. CSB-EL021788RA), SMAD7 (cat. no. CSB-E09225r) and connective tissue growth factor (CTGF; cat. no. CSB-E07876r) levels were analyzed using ELISA kits purchased from Cusabio Technology LLC. BSA (cat. no. A8020), the TGF-β1 receptor type I/II dual inhibitor (LY2109761; cat. no. ab29286) and anti-SMAD3 antibody (cat. no. ab40854) were obtained from Abcam, and the anti-SMAD7 antibody (cat. no. 42-0400) was purchased from Thermo Fisher Scientific, Inc. The Cy3 conjugated Goat Anti-Rabbit IgG secondary antibody (cat. no. GB21303) was obtained from Wuhan ServiceBio Technology Co., Ltd. RNA extracting fluid (cat. no. G3013) was obtained from Wuhan ServiceBio Technology Co., Ltd. HyPure™ Molecular Biology Grade Water (cat. no. SH30538.02) was obtained from HyClone. RevertAid First Strand cDNA Synthesis kit was obtained from Thermo Fisher Scientific, Inc. FastStart Universal SYBR Green Master (Rox) was obtained from Roche Diagnostics. Primers were obtained from Wuhan ServiceBio Technology Co., Ltd.
Cell culture
The rat alveolar epithelial type II RLE-6TN cell line was obtained from the Beijing Beina Chuanglian Biotechnology Research Institute (http://www.bnbio.com/default.html). RLE-6TN cells were cultured in high glucose DMEM, supplemented with 10% FBS, and maintained at 37°C with 5% CO2. Upon reaching 70–80% confluence, the cells were incubated with 0.25% trypsin solution and passaged at a ratio of 1:3. The culture medium was changed every 48 h. Cells in the exponential growth phase were used for further experiments.
MTT assay
RLE-6TN cells in logarithmic growth phase were digested with trypsin to prepare cell suspension. A total of 1×104 cells were inoculated into 96-well plate with 200 µl cells in each well. The cells were cultured in CO2 (5%) incubator overnight at 37°C and adhered to the wall. The marginal pores were filled with sterile PBS. Following 24 h of incubation, the culture medium was discarded and new culture medium containing different concentrations of paraquat (10, 50, 100, 200, 400, 600, 800, 1,000 or 2,000 nmol/l) was added for an additional 24 h at 37°C. Following the treatment, 20 µl MTT (5 g/l) was added to each well and incubated at 37°C for 4 h. Culture medium containing MTT reagent was removed, 150 µl DMSO was added into each well and spun at low speed 120 × g for 10 min to fully dissolve the crystal. The absorbance was measured at 490 nm (A490nm; RT-2100c; Rayto Life and Analytical Sciences Co., Ltd.) in order to assess the paraquat concentration required for 50% inhibition of cellular viability (IC50). GraphPad Prism 8 was used to calculate the IC50 (GraphPad Software, Inc.). In addition, an inhibitory rate (%) was calculated using the following formula: [(A490nm of control group-A490nm of experimental group)/A490nm of control group] ×100.
In further experiments, RLE-6TN cells (1×104 cells/well) were treated with a range of concentrations of tacrolimus (0, 0.1, 1, 10 or 20 ng/ml) for 4 h at 37°C and subsequently incubated in the presence of tacrolimus and 200 nmol/l paraquat for 24 h at 37°C to determine the effects of different concentrations of tacrolimus on the survival rate of RLE-6TN cells exposed to paraquat. LY2109761 was also incubated with RLE-6TN cells (1×104 cells/well) at a range of concentrations (0, 1, 5, 10, 20 or 30 µmol/l) for 16 h at 37°C and subsequently incubated in the presence of LY2109761 and 200 nmol/l paraquat for 16 h at 37°C. Following these treatments, the inhibitory rate (%) was performed as described above.
Experimental groupings
RLE-6TN cells were divided into the following seven groups: i) control group, which contained untreated cells incubated in culture medium for 24 h at 37°C; ii) paraquat group, which contained cells cultured in DMEM containing 200 nmol/l paraquat for 24 h at 37°C; iii) tacrolimus group, which was comprised of cells cultured in DMEM containing 10 ng/ml tacrolimus for 4 h and subsequently in DMEM containing 10 ng/ml tacrolimus and 200 nmol/l paraquat for 24 h at 37°C; and iv-vii) LY2109761 groups, which was comprised of cells cultured in DMEM containing 5 µmol/l LY2109761 for 16 h and subsequently in DMEM containing 5 µmol/l LY2109761 and 200 nmol/l paraquat for 1, 4, 8 or 16 h at 37°C, which was used to assess the action of LY2109761 at different time points. Each group had three experimental wells.
ELISAs
A total of 106 RLE-6TN cells/ml were diluted in PBS (pH 7.2–7.4) and lysed using repeated freeze-thaw cycles. The cells were frozen below −20°C and thawed at room temperature ten times; due to the formation of ice particles in the cells, and the increase in salt concentration in the cell solution, the cells swelled and the cell structure was broken. The samples were then centrifuged at 3,000 × g for 20 min at room temperature. Following centrifugation, the supernatant was collected and the concentrations of TGF-β1, SMAD3, SMAD7 and CTGF were detected using their respective ELISA kits, according to the manufacturers' protocols.
Immunofluorescence staining
RLE-6TN cells (1×104 cells/well) were fixed with 4% paraformaldehyde for 30 min at room temperature and washed with PBS three times for 5 min each time. A total of 50–100 µl 0.5% Triton X-100 was added, and the samples were incubated at room temperature for 10 min. The samples were subsequently washed with PBS three times for 5 min and incubated with 5% BSA for 30 min at room temperature. The primary antibodies (1:200, LY2109761 inhibitor; 1:500, anti-SMAD3 antibody; 1:100, anti-SMAD7 antibody) were diluted in PBS and added dropwise to the samples, then incubated overnight at 4°C. Following the primary antibody incubation, the samples were washed three times with PBS for 5 min each time and incubated with a Cy3 conjugated Goat Anti-Rabbit IgG secondary antibody (1:300) at room temperature for 50 min. The samples were then washed three times with PBS for 5 min each time and subsequently stained with 0.5 µg/ml DAPI dye at room temperature for 10 min. The samples were washed three times with PBS for 5 min each time, dried, then the surface containing the cells was sealed with anti-fluorescence quenching, which involved adding a sealing tablet on a clean slide, holding the cells on the slide onto the sealing agent. The stained slides were visualized under fluorescence microscope (magnification, ×200 and ×400; Eclipse Ci-L; Nikon Corporation) and the images were analyzed using ImageJ 1.51K (National Institutes of Health).
Reverse transcription-quantitative PCR (RT-qPCR)
The mRNA expression levels of TGF-β1, SMAD3, SMAD7 and CTGF were analyzed using RT-qPCR. Briefly, total RNA was extracted from RLE-6TN cells using TRIzol® reagent (Invitrogen; Thermo Fisher Scientific, Inc.). Total RNA was then reverse transcribed into cDNA using RevertAid First Strand cDNA Synthesis kit, according to the manufacturer's instructions. Target genes were amplified by qPCR using FastStart Universal SYBR Green Master on a StepOne Real-Time PCR system (Applied Biosystems; Thermo Fisher Scientific, Inc.) and the expression levels were normalized to the internal reference gene, β-actin. The following thermocycling conditions were used for the qPCR: Initial denaturation at 95°C for 10 min; followed by 40 cycles of denaturation at 95°C for 15 sec and annealing/elongation at 60°C for 60 sec. The melting curve was analyzed via heating at 0.3°C every 15 sec from 60–90°C. The primer sequences used for the qPCR are presented in Table I. qPCR was performed and the data were analyzed using ABI 7500 Real-Time PCR (Thermo Fisher Scientific, Inc.). The relative gene expression levels were calculated using the 2−ΔΔCq method (19).
Table I.
Primer sequences used for the reverse transcription-quantitative PCR.
| Gene | Primer sequence (5′→3′) | Product length, bp |
|---|---|---|
| β-actin | F: TGCTATGTTGCCCTAGACTTCG | 240 |
| R: GTTGGCATAGAGGTCTTTACGG | ||
| Transforming growth factor-β1 | F: CTTTAGGAAGGACCTGGGTTG | 140 |
| R: GGTTGTGTTGGTTGTAGAGGG | ||
| SMAD3 | F: CGAGAACACTAACTTCCCCGCT | 112 |
| R: GTGGTTCATCTGGTGGTCGCTA | ||
| SMAD7 | F: TCGGAAGTCAAGAGGCTGTGTT | 148 |
| R: GTTTGAGAAAATCCATCGGGTA | ||
| Connective tissue growth factor | F: CCAACTATGATGCGAGCCAACT | 272 |
| R: TTAGCCCGGTAGGTCTTCACACT |
F, forward; R, reverse.
Statistical analysis
SPSS 18.0 (SPSS, Inc.) statistical software was used to analyze the data. Each experiment was repeated three times. All data are presented as the mean ± SD. A Shapiro-Wilk test was used to analyze the distribution of the data and multi-group comparisons were performed using a Tukey's test. P<0.05 was considered to indicate a statistically significant difference.
Results
Effects of paraquat, tacrolimus and LY2109761 on the viability of RLE-6TN cells
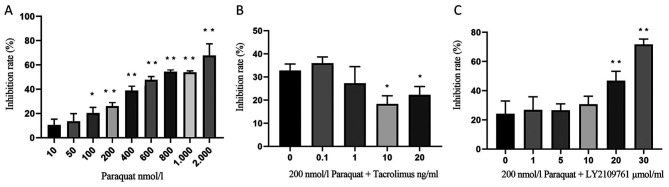
RLE-6TN cells were treated with 10, 50, 100, 200, 400, 600, 800, 1,000 or 2,000 nmol/l paraquat for 24 h and the cell inhibitory rate was determined using an MTT assay; the results revealed that the cell inhibitory rate increased with increasing concentrations of paraquat (Fig. 1A). Notably, the IC50 of paraquat was estimated to be 700 nmol/l; however, this induced high levels of cell death. Therefore, in subsequent experiments, 200 nmol/l paraquat was used, corresponding to an inhibitory rate of 26.05±2.99% (Fig. 1A).
Figure 1.
Inhibitory rate of RLE-6TN cell viability. (A) Inhibitory rate of RLE-6TN cells treated with varying doses of paraquat for 24 h. *P<0.05, **P<0.01 vs. 10 nmol/l paraquat. (B) Inhibitory effect of increasing concentrations of tacrolimus (0–20 ng/ml) and 200 nmol/l paraquat combined treatment. RLE-6TN cells were pretreated with increasing concentrations of tacrolimus for 4 h, then treated with 200 nmol/l paraquat for an additional 24 h. *P<0.05 vs. 0 ng/ml tacrolimus and 200 nmol/l paraquat. (C) Inhibitory effect of paraquat and LY2109761 treatment. RLE-6TN cells were pretreated with LY2109761 for 16 h, then treated with 200 nmol/l paraquat for an additional 16 h. **P<0.01 vs. 0 µmol/l LY2109761 and 200 nmol/l paraquat.
In addition, RLE-6TN cells were pretreated with 0.1, 1, 10 or 20 ng/ml tacrolimus for 4 h, then incubated with 200 nmol/l paraquat for an additional 24 h. The inhibition rate of 10 ng/ml tacrolimus on paraquat-exposed alveolar type II epithelial cells was 18.40±3.49%. Therefore, this concentration was used for subsequent experiments (Fig. 1B).
Lastly, RLE-6TN cells were cultured for 16 h in medium containing 1, 5, 10, 20 or 30 µmol/l LY2109761 and 200 nmol/l paraquat (Fig. 1C). The inhibitory rate was significantly increased following the treatment with 20 and 30 µmol/l LY2109761. Furthermore, the IC50 of LY2109761 was discovered to be 20 µmol/l; however, this concentration resulted in increased cell death. The inhibitory rate caused by 5 µmol/l was 26.56±4.49%. (Fig. 1C), and thus this concentration was used in further experiments.
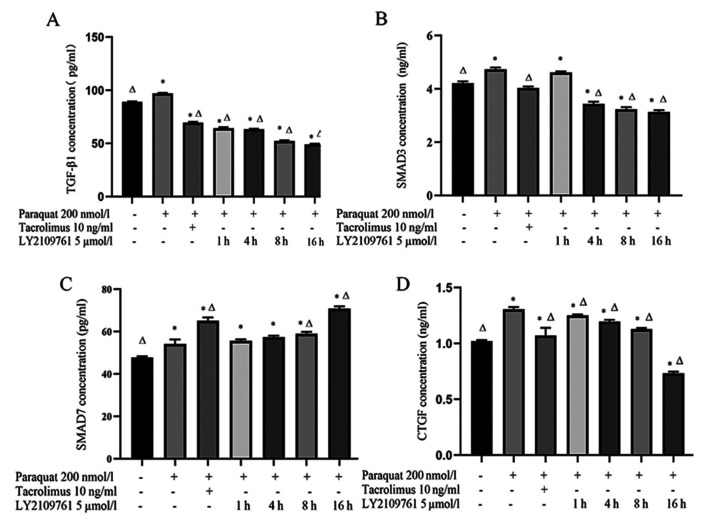
Tacrolimus decreases the concentration of TGF-β1 and SMAD3 in the supernatant of paraquat treated cells, and increases the concentration of SMAD7
The concentration of TGF-β1 in the supernatant of RLE-6TN cells treated with 200 nmol/l paraquat was significantly increased compared with the control group (Fig. 2A). However, the concentration of TGF-β1 in the tacrolimus group was significantly decreased compared with the control and paraquat groups. Notably, the levels of TGF-β1 gradually decreased following a 1, 4, 8 and 16 h incubation with 5 µmol/l LY2109761 compared with the paraquat group (Fig. 2A).
Figure 2.
TGF-β1, SMAD3, SMAD7 and CTGF concentrations in RLE-6TN cell culture supernatants. Concentrations of (A) TGF-β1, (B) SMAD3, (C) SMAD7 and (D) CTGF were determined using ELISAs following 200 nmol/l paraquat, 10 ng/ml tacrolimus and 5 µmol/l LY2109761 treatment. *P<0.05 vs. untreated cells; ΔP<0.05 vs. paraquat only. TGF-β1, transforming growth factor-β1; CTGF, connective tissue growth factor.
In addition, the concentration of SMAD3 in the paraquat group was significantly increased compared with the control cells (Fig. 2B). Following the treatment with tacrolimus, SMAD3 levels were significantly decreased compared with the paraquat groups. Moreover, the concentration of SMAD3 in RLE-6TN cells was decreased following the treatment with LY2109761 for 4, 8 and 16 h compared with the paraquat and control groups (Fig. 2B).
Compared with the control group and paraquat group, the concentration of SMAD7 increased significantly after tacrolimus treatment. Compared with paraquat group, the levels of SMAD7 in LY2109761 8 and 16 h groups were significantly increased (Fig. 2C).
Finally, the concentration of CTGF in the paraquat group was significantly increased compared with the control group. However, CTGF levels in cells pretreated with tacrolimus were significantly decreased compared with the paraquat group. Consistent with the treatment with tacrolimus, CTGF levels in the LY2109761 groups were gradually decreased with time compared with the paraquat group (Fig. 2D).
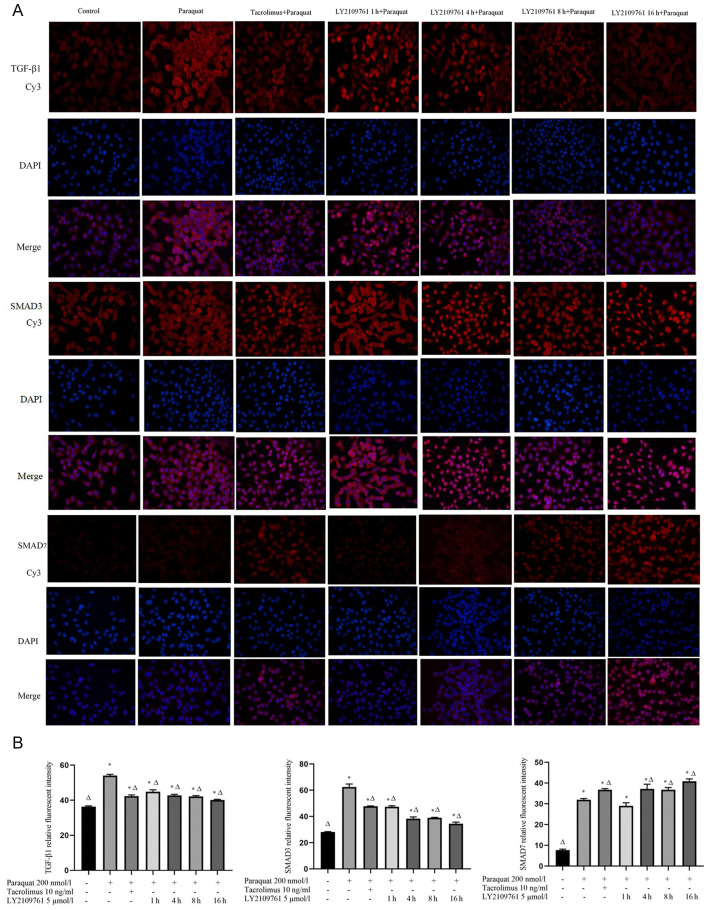
Tacrolimus downregulates the expression levels of TGF-β1 and SMAD3 in paraquat treated cells and upregulates the expression of SMAD7
The TGF-β1, SMAD3 and SMAD7 proteins were discovered to be localized in both the nucleus and cytoplasm (Fig. 3A). The expression levels of TGF-β1 and SMAD 3 in the paraquat group were upregulated compared with the control group. The pretreatment with tacrolimus downregulated the expression levels of TGF-β1 and SMAD3 compared with the cells exposed to paraquat alone, while the expression levels of SMAD7 were upregulated in the presence of tacrolimus compared with the paraquat group. The treatment with LY2109761 downregulated the expression levels of TGF-β1 and SMAD3. The expression levels of SMAD7 were upregulated following a 4 h incubation with LY2109761 compared with paraquat treatment alone (Fig. 3A and B).
Figure 3.
Expression levels of TGF-β1, SMAD3 and SMAD7 in RLE-6TN cells. Expression levels of TGF-β1, SMAD3 and SMAD7 in cells treated with 200 nmol/l paraquat without or without 10 ng/ml tacrolimus or 5 µmol/l LY2109761 (1, 4, 8 or 16 h) treatment. (A) The proteins of interest were stained with Cy3-conjugated secondary antibodies (red), while the cell nuclei were stained with DAPI (blue). Magnification, ×400. (B) The expression levels of TGF-β1, SMAD3 and SMAD7 were compared among the experimental groups. *P<0.05 vs. untreated cells; ΔP<0.05 vs. paraquat only. TGF-β1, transforming growth factor-β1.
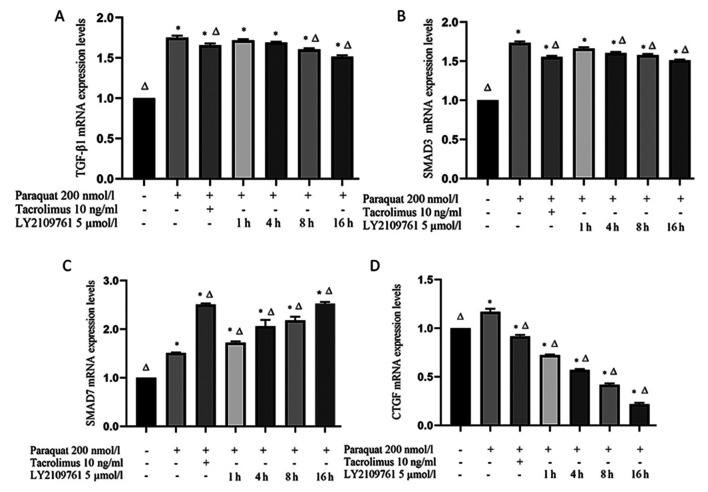
Tacrolimus downregulates the mRNA expression levels of TGF-β1, SMAD3 and CTGF in paraquat treated cells, and upregulates the expression of SMAD7 mRNA
The expression levels of TGF-β1 and SMAD3 in RLE-6TN cells exposed to paraquat were significantly upregulated compared with the control group (Fig. 4A and B). However, both TGF-β1 and SMAD3 mRNA expression levels were significantly downregulated following tacrolimus treatment compared with the paraquat group. Following the treatment with LY2109761, the expression levels of TGF-β1 gradually decreased over time; at the 8 and 16 h time points, TGF-β1 mRNA expression levels in the LY2109761 groups were significantly downregulated compared with the paraquat group. Similarly, SMAD3 mRNA expression levels were also downregulated following 4, 8 and 16 h of LY2109761 treatment compared with the paraquat group (Fig. 4A and B).
Figure 4.
TGF-β1, SMAD3, SMAD7 and CTGF mRNA expression levels. (A) TGF-β1, (B) SMAD3, (C) SMAD7 and (D) CTGF mRNA expression levels were analyzed in RLE-6TN cells treated with 200 nmol/l paraquat, 10 ng/ml tacrolimus and 5 µmol/lLY2109761 combinations using reverse transcription-quantitative PCR. *P<0.05 vs. β-actin; ΔP<0.05 vs. paraquat only. TGF-β1, transforming growth factor-β1; CTGF, connective tissue growth factor.
SMAD7 mRNA expression levels in RLE-6TN cells were significantly upregulated following paraquat exposure compared with the control group (Fig. 4C). Furthermore, following tacrolimus pretreatment, SMAD7 expression levels were significantly upregulated compared with paraquat treatment alone. Notably, SMAD7 mRNA expression levels were also significantly upregulated following the treatment with LY2109761 for 1, 4, 8 and 16 h compared with the paraquat and control groups (Fig. 4C).
Finally, CTGF expression levels were discovered to be significantly upregulated in paraquat-exposed cells compared with the control group (Fig. 4D); however, tacrolimus pretreatment significantly downregulated the mRNA expression levels of CTGF compared with the control and paraquat groups. Similarly, the expression levels of CTGF in the LY2109761 treatment for 1, 4, 8 and 16 h groups were significantly downregulated compared with those in the paraquat and control groups (Fig. 4D).
Discussion
The common poisoning method of paraquat poisoning is via oral administration into the digestive system. The toxicant is absorbed into the blood via the digestive tract, and the clinical manifestations associated with poisoning appear (20). Type I and II epithelial cells of the lung and Clara cells of the trachea include a polyamine transport system, which has been discovered to facilitate the transport of paraquat to the lungs (21). This process was reported to be characterized by inflammatory cell infiltration and pulmonary fibrosis in the lung tissue, and the transition of epithelial cells into interstitial cells was identified as an important pathological process of pulmonary fibrosis (22,23). As a result, alveolar epithelial cells were revealed to lose polarity, leading to cytoskeletal reconstruction and a spindle-shape type cellular morphology (22,23). The TGF-β1/SMAD signaling pathway was identified to be involved in pulmonary fibrosis. Briefly, TGF-β1 binding to TGF-β receptor leads to the phosphorylation of SMAD2 and SMAD3, and phosphorylated SMAD2 and SMAD3 subsequently form a complex with the phosphorylated form of SMAD4 (24). This complex then translocates to the nucleus and interacts with nuclear transcription factors, such as CTGF, where it regulates gene transcription (25). CTGF has been discovered to promote the transcription of genes involved in cell proliferation, cell adhesion and migration, as well as angiogenesis and other biological functions (25,26). Meanwhile, SMAD6 and SMAD 7 were reported to be strong inhibitors of SMAD2 and SMAD3 activation, resulting in the negative regulation of the TGF-β1/SMAD signaling pathway, and thus could prevent fibrosis (27,28). SMAD 6 and SMAD 7 were also identified to serve as intracellular antagonists of the TGF-β receptor (27,28).
In the present study, the levels of TGF-β1, SMAD 3 and CTGF were increased in RLE-6TN cells following paraquat exposure. However, tacrolimus treatment reversed this effect and led to TGF-β1, SMAD3 and CTGF downregulation, and SMAD7 upregulation. Moreover, the TGF-β receptor inhibitor LY2109761 produced similar effects to tacrolimus treatment. These findings suggested that tacrolimus may inhibit fibrosis through the TGF-β1/SMAD signaling pathway. Tacrolimus was found to inhibit the fibrosis of alveolar type II epithelial cells by inhibiting the expression of TGF-β1, SMAD 3 and CTGF, which are all known to promote fibrosis. Conversely, tacrolimus also prevented fibrosis through upregulating SMAD7, a protein known to inhibit fibrosis in alveolar type II epithelial cells. Therefore, due to its strong immunosuppressive effects, and its ability to inhibit the transformation of epithelial cells into mesenchymal cells, tacrolimus may represent a promising drug candidate for the treatment of acute paraquat poisoning.
Nonetheless, the present study had several limitations. Firstly, the current findings were solely based on in vitro experimentation and thus, the effects of tacrolimus require further validation in vivo. Other aspects also require further validation, including the effect of tacrolimus on cell viability, apoptosis and the necrosis of RLE-6TN cells, for instance. Moreover, the relationship between tacrolimus and fibrosis markers, such as N-cadherin and E-cadherin, remains to be examined. Lastly, whether tacrolimus also affects the dynamics of SMAD3 and SMAD7 phosphorylation, the hallmark of SMAD signaling pathway activation, also requires further study.
In conclusion, the present study demonstrated that paraquat can increase the expression levels of TGF-β1, SMAD3 and CTGF in RLE-6TN cells, and tacrolimus can inhibit the increase of these pro-fibroinflammatory factors. Moreover, tacrolimus can serve an antifibrotic role by increasing the expression of the fibrosis inhibitor SMAD7, which reduces the lung injury caused by paraquat.
Acknowledgements
Not applicable.
Funding
The present study was supported by The Shandong Key Research and Development Plan Project (grant nos. 2015GSF118038 and 2016GSF201041).
Availability of data and materials
The datasets used and analyzed during the present study are available from the corresponding author on reasonable request.
Authors' contributions
XJ conceived the study; ZZ and QN contributed significantly to the data analysis and manuscript preparation; YR performed the data analysis and wrote the manuscript; and BK and LK interpreted the data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Authors' information
ORCID nos.: YR, 0000-0002-2136-0123; XJ, 0000-0002-2277- 6817; ZZ, 0000-0002-2469-1577; QN, 0000-0003-0039-9202; BK, 0000-0002-0312-686X; LK, 0000-0002-0988-1022.
References
- 1.Dinis-Oliveira RJ, Duarte JA, Sánchez-Navarro A, Remião F, Bastos ML, Carvalho F. Paraquat poisonings: Mechanisms of lung toxicity, clinical features, and treatment. Crit Rev Toxicol. 2008;38:13–71. doi: 10.1080/10408440701669959. [DOI] [PubMed] [Google Scholar]
- 2.Jeyaratnam J. Acute pesticide poisoning: A major global health problem. World Health Stat Q. 1990;43:139–144. [PubMed] [Google Scholar]
- 3.Gil HW, Hong JR, Jang SH, Hong SY. Diagnostic and therapeutic approach for acute paraquat intoxication. J Korean Med Sci. 2014;29:1441–1449. doi: 10.3346/jkms.2014.29.11.1441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yamashita M, Yamashita M, Ando Y. A long-term follow-up of lung function in survivors of paraquat poisoning. Hum Exp Toxicol. 2000;19:99–103. doi: 10.1191/096032700678815729. [DOI] [PubMed] [Google Scholar]
- 5.He X, Wang L, Szklarz G, Bi Y, Ma Q. Resveratrol inhibits paraquat-induced oxidative stress and fibrogenic response by activating the nuclear factor erythroid 2-related factor 2 pathway. J Pharmacol Exp Ther. 2012;342:81–90. doi: 10.1124/jpet.112.194142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eddleston M, Wilks MF, Buckley NA. Prospects for treatment of paraquat-induced lung fibrosis with immunosuppressive drugs and the need for better prediction of outcome: A systematic review. QJM. 2003;96:809–824. doi: 10.1093/qjmed/hcg137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Qian J, Ye Y, Lv L, Zhu C, Ye S. FTY720 attenuates paraquat-induced lung injury in mice. Int Immunopharmacol. 2014;21:426–431. doi: 10.1016/j.intimp.2014.05.025. [DOI] [PubMed] [Google Scholar]
- 8.Botella DMJ, Belenguer TJ. Paraquat poisoning. A study of 29 cases and evaluation of the effectiveness of the ‘Caribbean scheme’. Med Clin (Barc) 2000;115:530–533. doi: 10.1016/s0025-7753(00)71615-0. [DOI] [PubMed] [Google Scholar]
- 9.Gao J, Feng S, Li Y. Prolonged low-dose cyclophosphamide treatment after pulse therapy attenuates lung injury in rats with paraquat intoxication. Korean J Intern Med. 2018;33:1137–1142. doi: 10.3904/kjim.2017.334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Barnes H, Holland AE, Westall GP, Goh NSL, Glaspole IN. Cyclophosphamide for connective tissue disease-associated interstitial lung disease. Cochrane Database Sys Rev. 2018;1:CD010908. doi: 10.1002/14651858.CD010908.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Thomson AW, Starzl TE. New immunosuppressive drugs: Mechanistic insights and potential therapeutic advances. Immunol Rev. 1993;136:71–98. doi: 10.1111/j.1600-065X.1993.tb00655.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Artz MA, Boots JMM, Ligtenberg G, Roodnat JI, Christiaans MHL, Vos PF, Blom HJ, Sweep FCG, Demacker PNM, Hilbrands LB. Improved cardiovascular risk profile and renal function in renal transplant patients after randomized conversion from cyclosporine to tacrolimus. J Am Soc Nephrol. 2003;14:1880–1888. doi: 10.1097/01.ASN.0000071515.27754.67. [DOI] [PubMed] [Google Scholar]
- 13.O'Grady JG, Hardy P, Burroughs AK, Elbourne D; UK, Ireland Liver Transplant Study Group Randomized controlled trial of tacrolimus versus microemulsified cyclosporin (TMC) in liver transplantation: Poststudy surveillance to 3 years. Am J Transplant. 2007;7:137–141. doi: 10.1111/j.1600-6143.2006.01576.x. [DOI] [PubMed] [Google Scholar]
- 14.Kim YH, Shin HY, Kim SM. Long-term safety and efficacy of tacrolimus in myasthenia gravis. Yonsei Med J. 2019;60:633–639. doi: 10.3349/ymj.2019.60.7.633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nagano J, Iyonaga K, Kawamura K, Yamashita A, Ichiyasu H, Okamoto T, Suga M, Sasaki Y, Kohrogi H. Use of tacrolimus, a potent antifibrotic agent, in bleomycin-induced lung fibrosis. Eur Respir J. 2006;27:460–469. doi: 10.1183/09031936.06.00070705. [DOI] [PubMed] [Google Scholar]
- 16.Staab-Weijnitz CA, Fernandez IE, Knüppel L, Maul J, Heinzelmann K, Juan-Guardela BM, Hennen E, Preissler G, Winter H, Neurohr C, et al. FK506-binding protein 10, a potential novel drug target for idiopathic pulmonary fibrosis. Am J Resp Crit Care. 2015;192:455–467. doi: 10.1164/rccm.201412-2233OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dhainaut JF, Charpentier J, Chiche JD. Transforming growth factor-beta: A mediator of cell regulation in acute respiratory distress syndrome. Crit Care Med. 2003;31(Suppl 4):S258–S264. doi: 10.1097/01.CCM.0000057901.92381.75. [DOI] [PubMed] [Google Scholar]
- 18.Bartram U, Speer CP. The role of transforming growth factor beta in lung development and disease. Chest. 2004;125:754–765. doi: 10.1378/chest.125.2.754. [DOI] [PubMed] [Google Scholar]
- 19.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 20.Sukumar CA, Shanbhag V, Shastry AB. Paraquat: The poison potion. Indian J Crit Care Med. 2019;23(Suppl 4):S263–S266. doi: 10.5005/jp-journals-10071-23306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hoet PH, Lewis CP, Demedts M, Nemery B. Putrescine and paraquat uptake in human lung slices and isolated type II pneumocytes. Biochem Pharmacol. 1994;48:517–524. doi: 10.1016/0006-2952(94)90281-X. [DOI] [PubMed] [Google Scholar]
- 22.Willis BC, Borok Z. TGF-beta-induced EMT: Mechanisms and implications for fibrotic lung disease. Am J Physiol Lung Cell Mol Physiol. 2007;293:L525–L534. doi: 10.1152/ajplung.00163.2007. [DOI] [PubMed] [Google Scholar]
- 23.Kim KK, Kugler MC, Wolters PJ, Robillard L, Galvez MG, Brumwell AN, Sheppard D, Chapman HA. Alveolar epithelial cell mesenchymal transition develops in vivo during pulmonary fibrosis and is regulated by the extracellular matrix. Proc Natl Acad Sci USA. 2006;103:13180–13185. doi: 10.1073/pnas.0605669103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Blank U, Karlsson S. The role of Smad signaling in hematopoiesis and translational hematology. Leukemia. 2011;25:1379–1388. doi: 10.1038/leu.2011.95. [DOI] [PubMed] [Google Scholar]
- 25.Chung AC, Zhang H, Kong YZ, Tan JJ, Huang XR, Kopp JB, Lan HY. Advanced glycation end-products induce tubular CTGF via TGF-beta-independent Smad3 signaling. J Am Soc Nephrol. 2010;21:249–260. doi: 10.1681/ASN.2009010018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen QK, Lee K, Radisky DC, Nelson CM. Extracellular matrix proteins regulate epithelial-mesenchymal transition in mammary epithelial cells. Differentiation. 2013;86:126–132. doi: 10.1016/j.diff.2013.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Krafft E, Lybaert P, Roels E, Laurila HP, Rajamaki MM, Farnir F, Myllarniemi M, Day MJ, Mc EK, Clercx C. Transforming growth factor beta 1 activation, storage, and signaling pathways in idiopathic pulmonary fibrosis in dogs. J Vet Intern Med. 2014;28:1666–1675. doi: 10.1111/jvim.12432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nie Y, Li S, Yi Y, Su W, Chai X, Jia D, Wang Q. Effects of astragalus injection on the TGFβ/Smad pathway in the kidney in type 2 diabetic mice. BMC Complement Altern Med. 2014;14:148. doi: 10.1186/1472-6882-14-148. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analyzed during the present study are available from the corresponding author on reasonable request.