Abstract
Background
Extremely preterm infants are often exposed to endotracheal tube continuous positive airway pressure (ETT-CPAP) trials to assess extubation readiness. The effects of ETT-CPAP trial on their diaphragmatic activity (Edi) and breathing variability is unknown.
Methods
Prospective observational study enrolling infants with birth weight ≤1250 g undergoing their first extubation attempt. Diaphragmatic activity, expressed as the absolute minimum (Edi min) and maximum values (Edi max), area under the Edi signal, and breath-by-breath analyses for breath areas, amplitudes, widths, and neural inspiratory and expiratory times, were analyzed during mechanical ventilation (MV) and ETT-CPAP. Neural breathing variability of each of these parameters was also calculated and compared between MV and ETT-CPAP.
Results
Thirteen infants with median (interquartile range) birth weight of 800 g [610–920] and gestational age of 25.4 weeks [24.4–26.3] were included. Diaphragmatic activity significantly increased during ETT-CPAP when compared to MV:Edi max (44.2 vs. 38.1 μV), breath area (449 vs. 312 μV·s), and amplitude (10.12 vs. 7.46 μV). Neural breathing variability during ETT-CPAP was characterized by increased variability for amplitude and area under the breath, and decreased for breath time and width.
Conclusions
A 5-min ETT-CPAP in extremely preterm infants undergoing extubation imposed significant respiratory load with changes in respiratory variability.
Impact
ETT-CPAP trials are often used to assess extubation readiness in extremely preterm infants, but its effects upon their respiratory system are not well known.
Diaphragmatic activity analysis demonstrated that these infants are able to mount an important response to a short trial.
A 5-min trial imposed a significant respiratory load evidenced by increased diaphragmatic activity and changes in breathing variability.
Differences in breathing variability were observed between successful and failed extubations, which should be explored further in extubation readiness investigations.
This type of trial cannot be recommended for preterm infants in clinical practice until clear standards and accuracy are established.
Introduction
Extremely preterm infants are commonly exposed to extubation readiness trials as a means to assess whether they can be successfully disconnected from invasive mechanical ventilation (MV). During these trials, ventilator inflation pressures are typically removed, and the patient only receives continuous positive airway pressure (CPAP) via the endotracheal tube (ETT) for a short period of time. Although well described in adults and pediatric patients as a good tool to assess extubation readiness,1–3 there is little evidence supporting the use of these trials in extremely preterm infants.4–7 Indeed, a recent systematic review in neonates demonstrated that those trials perform well at identifying successful extubation, but lack accuracy at detecting failures.8 Nevertheless, one-third of neonatal intensive care units (NICUs) have reported the use of ETT-CPAP trials, conducted in various ways, as part of their extubation readiness assessment in extremely preterm infants.4,9,10
The objective of an ETT-CPAP trial is to challenge the patient as a way to test readiness, but how the trial affects breathing variability and what is the magnitude of any response has not yet been studied in fragile infants. The electrical activity of the diaphragm (Edi) is a measure of the central respiratory drive, taking into account all respiratory reflexes and responses, such as blood gases and respiratory loads. All of this information generates an output response from the respiratory centers, transmitting the signal through the phrenic nerve to the diaphragm (Edi). As an electromyographic signal, the measures of Edi represent the activation and contraction of the motor units of the diaphragm, reflecting respiratory efforts and responses.11 and used to evaluate respiratory drive and work of breathing.12–14
In adult and pediatric patients, Edi increases during ETT-CPAP as a result of the increased respiratory load imposed by the trial.15–18 However, extremely preterm infants have immature respiratory control, differences in respiratory mechanics and diaphragm apposition, and reduced diaphragm muscle mass.19–21 These differences limit the extrapolation of those results to the extremely preterm population. While the diaphragmatic activity of preterm infants during the peri-extubation period has been recently described,22 the study included more mature infants and an ETT-CPAP trial was not performed.
Breathing, under normal conditions, is a highly variable physiological process representing adaptation of the respiratory control systems; most importantly, alterations in respiratory variability have been demonstrated in response to increases in respiratory mechanical loading,23–25 and in relation to respiratory morbidities.26,27 Differences in respiratory variability have also been found between infants who succeed or fail extubation,28 but it was calculated from analysis of respiratory inductive plethysmography signals or ventilator measurements, which are subject to artifacts. Therefore, the primary objective of this study was to describe diaphragmatic activity and neural breathing variability during a 5-min ETT-CPAP period in extremely preterm infants undergoing their first extubation attempt. The second objective aimed to investigate the association between those parameters and extubation outcomes.
Methods
Population
This is a secondary analysis on a subset of patients enrolled in two studies concurrently performed at the Montreal Children’s Hospital (ClinicalTrials.gov NCT01909947 and NCT02723123). The studies enrolled extremely preterm infants with birth weight ≤ 1250 g receiving invasive MV and undergoing their first planned extubation. The first study was a multicenter collaboration to investigate automated analysis of cardiorespiratory signals to predict extubation outcomes29 and the second was a randomized crossover trial comparing physiological differences between nasal CPAP, noninvasive positive pressure ventilation and noninvasive neurally adjusted ventilatory assist (NAVA) applied immediately after extubation. Therefore, in some infants at our site, cardiorespiratory signal recordings included the use of a special catheter to capture the electrical activity of the diaphragm (Edi) during MV and ETT-CPAP: the NAVA catheter. The research ethics board of the institution approved both studies and informed consent was obtained from parents or legal guardians. All infants that enrolled in both studies were consecutively included in the secondary analysis until the planned sample size was reached.
All clinical decisions concerning intubation, adjustments and/or weaning from MV, and timing of extubation were made exclusively by the attending physician. All premature infants were on assisted control mode using a permissive hypercapnia strategy (PaCO2 between 45 and 55 mmHg for the first 3 days and between 55 and 65 mmHg afterwards, if pH > 7.20) and oxygen saturation (SpO2) target between 91 and 95%. Both research protocols suggested infants to be considered “ready” for extubation in the presence of all the following criteria: for infants <1000 g − mean airway pressure (MAP) ≤ 7 cmH2O and fraction of inspired oxygen (FiO2) ≤ 0.3 and for infants ≥1000 g − MAP ≤ 8 cmH2O and FiO2 ≤ 0.3.
Study design and data acquisition
All infants were studied in resting supine position immediately prior to extubation while receiving MV. In the hour preceding extubation, a 6-French/49 cm specialized feeding tube with small, embedded electrodes was placed at the level of the diaphragm and the position was confirmed by the Servo-i ventilator as previously described before being secured in place.30 Research staff were by the bedside during the entire study period, and the catheter position was monitored throughout. These catheters were inserted prior to extubation to capture Edi signals during MV and the 5 min ETT-CPAP and used for the noninvasive neurally adjusted ventilatory assist (NAVA) module of the Servo-i ventilator as part of the post-extubation study (Maquet Critical Care, Solna, Sweden). Edi signal was recorded and stored for offline analysis using the Servo Tracker software (v4.2, Maquet Critical Care, Solna, Sweden). After 1 h of recordings during MV, infants were placed on a 5-min ETT-CPAP using the same positive end-expiratory pressure, and then extubated within the following hour.
Edi analysis
Edi segments selection
Edi signals were analyzed using 3-minute segments obtained during MV and ETT-CPAP recordings. During MV, the segment was selected immediately prior to the switch to ETT-CPAP, and during ETT-CPAP, the segment was selected as the middle 3-min of the whole 5-min period if possible. In these cases, the first minute was not analyzed to circumvent misinterpretations of any abrupt response related to switching from MV to ETT-CPAP, and the last minute for any possible tiredness after 4 min of ETT-CPAP. Previous studies of cardiorespiratory behavior from our collaborative group have found that the middle of the ETT-CPAP trial was most useful and predictive of extubation outcome.31–33 Infants with ETT-CPAP durations of <3 min were excluded from Edi analysis. The major reason for exclusion was the inability to analyze all parameters of interest in a very short segment and to maintain consistent lengths of time (segment length) for comparison of variability measurements between patients.
Edi signal analysis: diaphragmatic activity
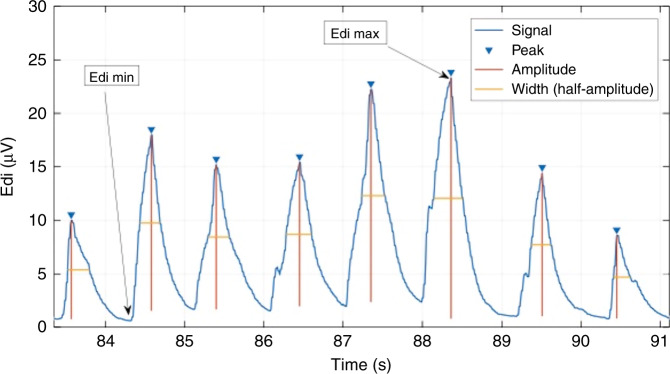
From the entire 3-min selected segments, the following parameters were obtained: (a) resting tonic state (Edi min) = minimum (or lowest) value of Edi, (b) maximal inspiratory effort (Edi max) = maximum value of Edi, and (c) work of breathing (Edi area) = area under the entire signal. These were single values obtained from each segment for each patient. Then, for each individual neural breath, the following parameters were calculated: (a) breath area = area under the curve; (b) breath amplitude = minimum vertical distance that the signal descends on either side of the peak; (c) breath width = distance between the points to the left and right of the peak at half the amplitude; (d) neural inspiratory time (NTi) = time from trough to peak; and (e) neural expiratory time (NTe) = time from peak to trough. After that, the median values of these individual neural breath parameters were calculated over the entire 3-min segment to obtain a single value per patient. The latter values, in combination with Edi min, Edi max, and Edi area, were described across groups and used for comparisons (as described in the “Statistical analysis” below). All parameters were calculated using a semi-automated algorithm developed on MATLAB (R2017a, MathWorks, Natick, USA), as peak detection functions needed manual adjustment depending on signal amplitude and breathing frequency. An example of Edi parameter calculation is provided in Fig. 1.
Fig. 1. Electrical activity of the diaphragm signal analysis.
Example of electrical activity of the diaphragm (Edi) signal analysis to obtain the maximum Edi (Edi max), minimum Edi (Edi min), breath amplitudes, and widths.
Neural breathing variability analysis
From the five parameters obtained from individual breaths analysis (breath area, breath amplitude, breath width, NTi, NTe), as well as the breath-to-breath (BB) time intervals, the following measures of variability were calculated: standard deviation (SD), coefficient of variation (CV), standard deviation of the successive differences (SDSD), and the triangular index (TI) of each of them. Variability of any biological signal can be calculated in several different ways. While SD and CV are common methods, the SDSD and TI are less generally used, but have been applied for the assessment of heart rate and respiratory variability.34,35 SDSD measures the degree of variability with respect to rapid, high-frequency changes, or successive differences in the data. TI is a geometric method of assessing variability based on the construction of a histogram with specified bin widths. It is calculated as the total number of values divided by the number of values at the highest peak of the histogram. A larger TI value indicates a flatter, wider histogram, thus a greater degree of dispersion of the data. In our study, the histogram bin widths were set empirically after visual inspection of the histograms: 0.2 s for BB intervals, 100 μV·s for breath area, 0.1 s for NTi, NTe, and breath width, and 5 μV for breath amplitude.
Clinical data
Baseline neonatal data (birth weight, gestational age, post-menstrual age at extubation, weight at extubation, and sex) and the most recent pre-extubation blood gas results within 36 h prior to extubation (pH, partial pressure of carbon dioxide, bicarbonate, base excess, and total hemoglobin) were collected. In addition, the ventilation settings during MV and ETT-CPAP were collected: ETT size, peak inflation pressure, positive end-expiratory pressure (PEEP) during MV and ETT-CPAP, rate, tidal volume (VT), and FiO2. Extubation failure was defined as reintubation within 7 days.
Sample size and statistical analysis
As this study aimed to analyze and describe the electrical activity of the diaphragm of each patient, a convenience sample size of 12 patients with adequate recordings was planned. Given our experience with biological signal acquisition and analysis, we expected up to 20% loss of patients for poor quality of signals or technical issues.36,37 As such, we aimed to include 16 patients in total. All values are presented as median (interquartile range [IQR]) or numbers (%). For the primary objective, the nonparametric Wilcoxon’s signed-rank test was used for paired comparisons of diaphragmatic activity and respiratory variability parameters between MV and ETT-CPAP. For the secondary objective, values during MV and ETT-CPAP, as well as the relative percent changes from MV to ETT-CPAP, were calculated and compared between success and failure infants using the Wilcoxon’s rank-sum test. The most significant parameters were then evaluated further for their ability to identify extubation success by calculating the area under the receiver-operating characteristics curves (AUCROC), sensitivity, and specificity.
Results
A total of 16 patients were enrolled and studied, but three were excluded due to the catheter position being dislodged to such a degree that the Edi signal quality was very low (n = 1) or premature interruption of the ETT-CPAP (duration of only 1–2 min; n = 2). Of the two infants who were excluded for short ETT-CPAP trials, one was successfully extubated and the other failed. The 13 included infants were mostly male (69%), with median [IQR] birth weight of 800 g [610–920] and gestational age of 25.4 weeks [24.4–26.3]. Infants were extubated at median [IQR] day of life 8 [4–25], weight of 870 g [775–990], and post-menstrual age of 27.4 weeks [26.6–28.7] (Table 1). All infants received caffeine and had blood gas analysis prior to extubation (Table 1). Most infants were intubated with a 2.5 mm ETT (n = 11). The two infants with a 3.0 mm ETTs were successfully extubated (Supplementary Table S1 online). Individual demographics and extubation data are provided in Supplementary Table S1 (online).
Table 1.
Patient demographics.
| Variables | All patients (n = 13) | Success (n = 7) | Failure (n = 6) |
|---|---|---|---|
| Neonatal data | |||
| Birth weight (g) | 800 [610–920] | 920 [860–993] | 605 [600–618]** |
| Gestational age (week) | 25.4 [24.4–26.3] | 26.3 [25.5–27.9] | 24.5 [23.7–25.3]* |
| Weight at extubation (g) | 870 [775–990] | 990 [805–1110] | 825 [776–900] |
| PMA at extubation (week) | 27.4 [26.6–28.7] | 28.1 [26.7–29.1] | 27.3 [26.7–27.5] |
| Day of life at extubation (days) | 8 [4–25] | 7 [3–8] | 24 [20–27] |
| Male sex | 9/13 (69) | 6/7 (88) | 3/6 (50) |
| Pre-extubation blood gas | |||
| pH | 7.35 [7.30–7.39] | 7.34 [7.27–7.35] | 7.38 [7.36–7.39] |
| PCO2 (mmHg) | 47 [39–53] | 47 [39–51] | 46 [42–52] |
| Base excess (mmol/L) | −0.8 [−4.4 to 1.9] | −3.9 [−5.2 to −1.9] | 1.9 [0.4–3.4] |
| Total hemoglobin (g/L) | 133 [116–141] | 139 [119–150] | 129 [115–138] |
| Pre-extubation settings | |||
| Mechanical ventilation | |||
| PEEP (cmH2O) | 5 [5, 6] | 5 [5, 6] | 5 [5, 6] |
| Mean airway pressure (cmH2O) | 7.2 [6.2–7.9] | 7.2 [6.5–8.0] | 6.9 [6.3–7.7] |
| Maximum FiO2 | 0.25 [0.21–0.29] | 0.24 [0.21–0.25] | 0.28 [0.25–0.31] |
| Tidal volume (mL/kg)a | 4.8 [4.5–4.9] | 4.8 [4.8–5.3] | 4.6 [4.5–4.7] |
| Ratea | 20 [10, 45] | 20 [20, 45] | 20 [10, 20] |
| ETT-CPAP | |||
| PEEP (cmH2O) | 5 [5, 6] | 5 [5, 6] | 5 [5, 6] |
| Maximum FiO2 | 0.30 [0.24–0.32] | 0.24 [0.23–0.28] | 0.32 [0.30–0.38]* |
| Extubation failure | |||
| Time to reintubation | |||
| Reintubation within 24 h | – | – | 4/6 (67) |
| Reintubation within 72 h | – | – | 4/6 (67) |
| Reintubation within 7 days | – | – | 6/6 (100) |
| Reason for reintubation | |||
| Apneas and bradycardias | – | – | 5/6 (83) |
| Respiratory acidosis | – | – | 1/6 (17) |
PCA post-conceptional age, PCO2 partial pressure of carbon dioxide, PEEP positive end-expiratory pressure, FiO2 fraction of inspired oxygen, NCPAP nasal continuous positive airway pressure, NIPPV nasal intermittent positive airway pressure, NIV-NAVA noninvasive neurally adjusted ventilatory assist.
Values are presented as median [IQR], median [min, max], or n/N (%). All patients were on assisted control mode before extubation.
*P value <0.05 and **p value <0.01 for comparisons between extubation success vs. failure (Wilcoxon’s rank-sum test).
aBackup rate available for 10 patients.
Primary objective
Differences in diaphragmatic activity were noted in the analysis of the entire 3-min selected segments and individual breaths, with higher values during the ETT-CPAP period when compared to MV for the following parameters: Edi max (44.2 μV [31.2–53.9] vs. 38.1 μV [25.7–52.5]; p = 0.027), breath area (449 μV·s [316–523] vs. 312 μV·s [231–416]; p = 0.002), and amplitude (10.12 μV [7.99–14.09] vs. 7.46 μV [5.50–10.03]; p = 0.008) (Table 2).
Table 2.
Diaphragmatic activity during mechanical ventilation weaning.
| MV (n = 13) | ETT-CPAP (n = 13) | P value | |
|---|---|---|---|
| Edi analysis | |||
| Edi min (μV) | 0.24 [0.05–0.29] | 0.18 [0.12–0.27] | 0.931 |
| Edi max (μV) | 38.1 [25.7–52.5] | 44.2 [31.2–53.9] | 0.027 |
| Edi area (μV·s) | 1005 [666–1303] | 1077 [810–1648] | 0.068 |
| Individual breath analysis | |||
| Neural breaths (n) | 151 [129–184] | 154 [145–195] | 0.718 |
| RR (breaths per min) | 50 [44–60] | 51 [49–64] | 0.751 |
| Breath area (μV·s) | 312 [231–416] | 449 [316–523] | 0.002 |
| NTi (s) | 0.39 [0.37–0.42] | 0.43 [0.36–0.45] | 0.191 |
| NTe (s) | 0.57 [0.48–0.66] | 0.63 [0.49–0.69] | 0.505 |
| Breath amplitude (μV) | 7.46 [5.50–10.03] | 10.12 [7.99–14.09] | 0.008 |
| Breath width (s) | 0.35 [0.33–0.38] | 0.35 [0.31–0.40] | 0.839 |
MV mechanical ventilation, ETT-CPAP endotracheal continuous positive airway pressure, Edi electrical activity of the diaphragm, RR respiratory rate, NTi neural inspiratory time, NTe neural expiratory time.
Values are presented as median [IQR] and p value was calculated by using the Wilcoxon’s signed-rank test.
Significant changes in neural breathing variability were observed during ETT-CPAP, with increased variability for the amplitude and area under the breath and decreased variability for time and width, characterizing an increased work of breathing during ETT-CPAP. Values of each significantly different parameters are provided in Table 3; results of all variability measures are provided in Supplementary Table S2 (online).
Table 3.
Variability of neural breathing pattern during mechanical ventilation weaning.
| MV (n = 13) | ETT-CPAP (n = 13) | P value | |
|---|---|---|---|
| Breath area (μV·s) | |||
| SDArea | 312 [209–415] | 326 [230–458] | 0.040 |
| SDSDArea | 402 [221–468] | 424 [287–549] | 0.021 |
| TIArea | 4.08 [3.39–5.49] | 5.00 [3.58–7.38] | 0.033 |
| Neural Ti (s) | |||
| CVNTi | 0.74 [0.52–0.87] | 0.48 [0.42–0.55] | 0.046 |
| Breath amplitude (μV) | |||
| SDSDAmp | 7.57 [5.52–9.43] | 9.50 [6.45–10.74] | 0.013 |
| Breath width (s) | |||
| SDWidth | 0.15 [0.12–0.23] | 0.11 [0.11–0.15] | 0.033 |
| CVWidth | 0.37 [0.32–0.50] | 0.34 [0.28–0.37] | 0.046 |
| SDSDWidth | 0.19 [0.15–0.31] | 0.16 [0.14–0.21] | 0.040 |
MV mechanical ventilation, ETT-CPAP endotracheal continuous positive airway pressure, Ti inspiratory time, Te expiratory time.
Results are expressed as median [IQR]; statistical analysis was performed by using the Wilcoxon’s signed-rank test.
Secondary objective
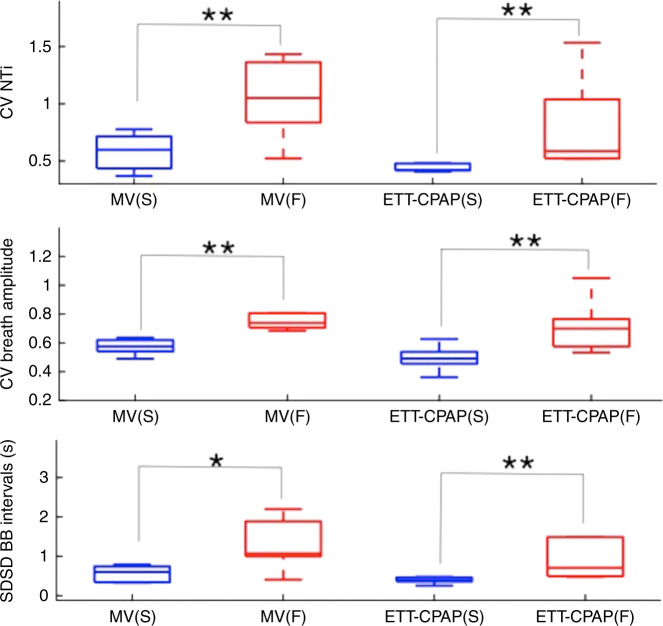
Of the 13 patients studied, seven were successfully extubated and six failed. A sample of Edi signals for an extubation success and a failure patient are provided in Supplementary Fig. S1 (online). For the diaphragmatic activity, no significant differences were observed between the two groups. Significant changes in variability measures, however, were found between the groups during both MV and ETT-CPAP. Comparisons of success and failure infants for all variability parameters are reported in Table 4; significant variability measures during both MV and ETT-CPAP included SDBB, SDNTi, CVArea, CVNTi, CVNTe, CVAmp, SDSDBB, and SDSDNTi, with SDNTe found to only be significant during MV. The most significant changes were found for CVNTi, CVAmp, and SDSDBB parameters as demonstrated in Fig. 2. The ability of these parameters to identify extubation success was strong: AUCROC values ranged between 0.91 and 1.00, sensitivity between 85.7 and 100%, and specificity between 83.3 and 100% (Supplementary Table S3). For the relative percent changes from MV to ETT-CPAP, neither diaphragmatic activity nor respiratory variability was found to be different between success and failure.
Table 4.
Respiratory variability and extubation outcomes.
| MV | ETT-CPAP | |||||
|---|---|---|---|---|---|---|
| Success | Failure | P values | Success | Failure | P values | |
| BB intervals (s) | ||||||
| SDBB | 0.55 [0.27–0.56] | 1.02 [0.90–1.42] | 0.014 | 0.36 [0.27–0.39] | 0.61 [0.42–1.31] | 0.014 |
| CVBB | 0.48 [0.36–0.67] | 0.50 [0.38–0.77] | 0.945 | 0.37 [0.33–0.39] | 0.40 [0.31–0.61] | 0.945 |
| SDSDBB | 0.60 [0.34–0.73] | 1.07 [1.00–1.70] | 0.014 | 0.40 [0.37–0.45] | 0.70 [0.52–1.32] | 0.002 |
| TIBB | 2.92 [2.46–3.36] | 3.65 [2.67–4.15] | 0.628 | 3.06 [2.69–3.76] | 3.04 [2.64–3.45] | 0.945 |
| Breath area (μV·s) | ||||||
| SDArea | 248 [169–346] | 347 [315–419] | 0.294 | 269 [220–392] | 384 [304–473] | 0.366 |
| CVArea | 0.67 [0.62–0.78] | 0.91 [0.86–0.98] | 0.014 | 0.58 [0.53–0.65] | 0.83 [0.64–0.99] | 0.013 |
| SDSDArea | 2789 [179–426] | 436 [404–467] | 0.294 | 334 [272–486] | 480 [379–631] | 0.366 |
| TIArea | 4.33 [3.08–5.67] | 3.86 [3.45–4.69] | 0.534 | 5.43 [3.76–7.45] | 4.60 [3.44–6.79] | 0.534 |
| Neural Ti (s) | ||||||
| SDNTi | 0.29 [0.18–0.33] | 0.64 [0.55–0.81] | 0.014 | 0.20 [0.18–0.21] | 0.40 [0.27–0.57] | 0.035 |
| CVNTi | 0.60 [0.44–0.69] | 1.05 [0.84–1.33] | 0.008 | 0.42 [0.42–0.48] | 0.59 [0.53–0.93] | 0.001 |
| SDSDNTi | 0.41 [0.24–0.45] | 0.81 [0.68–1.16] | 0.022 | 0.25 [0.23–0.26] | 0.49 [0.37–0.74] | 0.014 |
| TINTi | 2.85 [2.36–3.39] | 3.52 [2.60–4.15] | 0.534 | 2.65 [2.49–3.35] | 3.46 [2.80–4.37] | 0.347 |
| Neural Te (s) | ||||||
| SDNTe | 0.35 [0.21–0.43] | 0.62 [0.48–0.70] | 0.014 | 0.27 [0.20–0.30] | 0.37 [0.26–1.04] | 0.234 |
| CVNTe | 0.53 [0.37–0.57] | 0.75 [0.73–0.79] | 0.021 | 0.42 [0.35–0.46] | 0.63 [0.49–0.96] | 0.021 |
| SDSDNTe | 0.37 [0.27–0.56] | 0.76 [0.54–0.89] | 0.073 | 0.32 [0.28–0.35] | 0.43 [0.33–1.44] | 0.138 |
| TINTe | 4.04 [3.09–4.81] | 5.01 [4.81–5.83] | 0.138 | 3.57 [3.38–4.49] | 3.76 [3.44–6.77] | 0.836 |
| Breath amplitude (μV) | ||||||
| SDAmp | 6.29 [3.76–7.13] | 6.40 [5.31–8.67] | 0.534 | 4.97 [4.64–9.05] | 7.66 [6.56–8.92] | 0.445 |
| CVAmp | 0.58 [0.55–0.62] | 0.74 [0.71–0.79] | 0.001 | 0.49 [0.46–0.53] | 0.70 [0.60–0.75] | 0.002 |
| SDSDAmp | 7.57 [4.17–8.69] | 8.26 [6.87–9.68] | 0.062 | 6.45 [6.16–11.31] | 9.87 [8.76–10.61] | 0.445 |
| TIAmp | 2.55 [1.90–3.14] | 2.53 [2.12–3.15] | 0.836 | 2.48 [2.14–3.63] | 2.42 [1.70–3.29] | 0.534 |
| Breath width (s) | ||||||
| SDWidth | 0.13 [0.12–0.17] | 0.19 [0.14–0.31] | 0.234 | 0.11 [0.10–0.13] | 0.16 [0.12–0.20] | 0.138 |
| CVWidth | 0.36 [0.32–0.43] | 0.46 [0.37–0.70] | 0.279 | 0.33 [0.28–0.34] | 0.38 [0.32–0.49] | 0.276 |
| SDSDWidth | 0.17 [0.15–0.24] | 0.26 [0.19–0.45] | 0.181 | 0.16 [0.14–0.18] | 0.23 [0.16–0.27] | 0.138 |
| TIWidth | 2.56 [2.45–2.74] | 2.75 [2.53–2.89] | 0.628 | 2.30 [2.20–2.78] | 2.73 [2.37–3.34] | 0.389 |
MV mechanical ventilation, ETT-CPAP endotracheal tube continuous positive airway pressure, BB breath to breath, SD standard deviation, CV coefficient of variation, SDSD standard deviation of successive differences, TI triangular index, NTi neural inspiratory time, Ti inspiratory time, Te expiratory time.
Values are presented as median [IQR].
Fig. 2. Neural breathing variability and extubation outcomes.
MV mechanical ventilation, ETT-CPAP endotracheal tube continuous positive airway pressure, CV coefficient of variation, NTi neural inspiratory time, SDSD standard deviation of successive differences, BB breath to breath. Respiratory variability parameters during MV and ETT-CPAP significantly differ between infants with extubation success (S) or failure (F); p value *<0.05 and **<0.01 using the Wilcoxon’s rank sum.
Discussion
In this prospective observational study, we demonstrated that a 5-min ETT-CPAP trial imposed a significant respiratory load in extremely preterm infants undergoing their first elective extubation attempt. This was clearly shown by an increased diaphragmatic activity as well as significant changes in neural breathing variability during the trial. Although, it is well known that the presence of an ETT and ventilator apparatus increase dead space and respiratory resistance,38,39 the response of extremely preterm infants to this challenge has not been well outlined. It was also interesting to note that the ability to mount such respiratory effort occurred in response to a quite short trial.
An increased respiratory effort during an ETT-CPAP period has been well demonstrated in the pediatric and adult population.15–18 However, preterm infants have anatomical and physiological differences that might alter respiratory regulation, resting lung volumes, and subsequently the response to the challenge.19–21 These infants may simply not tolerate even a short-duration trial and quickly develop clinical instability. Indeed, a recent study from our group demonstrated that clinical instability (apneas, bradycardias, and desaturations) occurred in nearly 60% of extremely preterm infants during a 5-min ETT-CPAP,40 but the magnitude of the instability (or degree of response to the trial) was not assessed. In our study, the ETT-CPAP challenge led to a 16% increase in Edi max, 44% increase in breath area, and a 36% increase in breath amplitude. These important changes during ETT-CPAP that occurred after removal of the peak inflations and decrease of MAP (from 7 to 5 cmH2O) likely lead to a significant increase on respiratory system resistance.41 Indeed, this may explain why standard ETT-CPAP trials in preterm infants have a high number of false positives, that is, infants that fail the trial on the premise of clinical instability during an ETT-CPAP, but are successfully extubated.40
Typically, extubation readiness trials are meant to create a respiratory load sufficient enough to mimic the patient’s post-extubation condition. In adult and pediatric patients, the presence of positive end-expiratory pressure and/or pressure support during an extubation readiness trial significantly underestimates the patient’s respiratory load after extubation.42,43 Such studies are unfortunately not available in extremely preterm infants. Although the addition of pressure support to an ETT-CPAP trial may be appealing in the smallest infants, their fast respiratory rates and short inspiratory times may actually not allow sufficient time for pressure transmission. Therefore, studies evaluating the respiratory load during ETT-CPAP, at different CPAP levels and with or without pressure support, when compared to current standards of post-extubation noninvasive respiratory support are needed for the extremely preterm population.
Variability in breathing has been examined in infants receiving different types of respiratory support.23,26,27,44 In this study, changes in neural breathing variability were observed from MV to ETT-CPAP and were characterized by an increased variability in breath depth and decreased variability in breath times. In other words, infants responded to the 5-min ETT-CPAP by breathing deeper and more constantly, and with increased variability of breathing efforts. Indeed, a strategy of slower respiratory rates and variability and increased VT to maintain minute ventilation has been demonstrated to be cost-effective under certain conditions such as increased respiratory system resistance.45 It is possible that adequate minute ventilation would not have been maintained if a longer trial duration was used.
Extubation success and failure
Differences in respiratory variability between extubation outcomes28 and respiratory morbidities26,27 have been previously described during MV weaning, and abnormal variability (too high or too low) has been associated with worse outcomes. In this study, infants that went on to fail their extubation showed increased variability of some parameters during ETT-CPAP. Unstable respiratory patterns have been described in preterm infants with sepsis, characterized by increased variability in the breathing cycle due to increased apneas.46 Furthermore, acute increases in periodic breathing, characterized by short repeated cycles of pauses and breathing, was found in preterm infants with septicemia and necrotizing enterocolitis.47 Thus, the increased respiratory variability in extubation failure infants are likely a reflection of these erratic and unstable breathing patterns of sicker infants. Interestingly, despite differences in respiratory variability, infants that failed extubation were able to mount a response to the 5-min ETT-CPAP trial similar to infants successfully extubated. In other words, diaphragmatic activity was not a good predictor of extubation readiness. Truly, current definitions of ETT-CPAP trials do not provide much additional benefit over clinical judgment to identify failures prior to extubation, but combinations of physiological parameters with clinical data showed better performance.8 The strong performances of respiratory variability parameters (up to 100% accuracy) to identify successes should be interpreted with caution given the small sample size. Nevertheless, analysis of respiratory variability parameters have potential and should be investigated in future research studies, without necessarily requiring an ETT-CPAP trial. Further research in this area is underway29 and may consider incorporation of measurements of diaphragm activity.
Study limitations and strengths
This study has some limitations. We used a small sample size as in most physiological studies involving extensive data analysis and paired comparisons, but additional infants could have provided more robust information. The analysis of Edi, although it is a good measure of the central respiratory drive, does not necessarily translate into changes in VT, therefore limiting interpretations about changes in ventilation. Infants in the failure group were smaller but based on previous research, diaphragmatic activity, as measured using Edi, does not appear to significantly change across weights and post-menstrual age.30,48 Moreover, the duration of the ETT-CPAP was empirically determined as 5 min with a 3-min segment selected within, and it remains unclear if a longer duration of ETT-CPAP or segment length would have altered the results. We also lost two patients due to short (<3 min) ETT-CPAP trials. It is possible that inclusion of these two infants could have provided useful information, but it would have rendered the analysis methodology more heterogeneous. Importantly, although the diaphragmatic activity has been previously described in preterm infants receiving noninvasive respiratory support,44,49,50 MV,22,51 and no respiratory support,30 the diaphragmatic response of these immature infants to a period of ETT-CPAP was never evaluated. The analysis was exclusively done in extremely preterm infants included in two large and well-designed studies, and investigators had experience with all recordings and physiological data analysis.
Conclusion
The use of a 5-min ETT-CPAP in extremely preterm infants undergoing their first planned extubation attempt imposed a respiratory load evidenced by significant changes in diaphragmatic activity and breathing variability. Therefore, until clear standards, definitions, and good accuracy to identify failures are established, this type of trial cannot be recommended in preterm infants in clinical practice. Neural breathing variability in combination with clinical judgment and other physiological parameters should be studied for the prediction of extubation outcome.
Supplementary information
Acknowledgements
We are indebted to all infants, their families, and the NICU staff for their participation and help with the study. S.L. and W.S. were both supported from Fonds de la recherche en sante du Quebec (FRQS) doctoral research awards in partnership with the Fondation Des Étoiles. G.S. received research funds from the Montreal Children’s Foundation.
Author contributions
S.L. assisted in patient screening and enrollment, was responsible for data acquisition, analyzed the data, and drafted the manuscript. M.B. and S.R. assisted in patient screening and enrollment, and data acquisition. W.S. was responsible for patient screening and enrollment, provided critical input into study design, data analyses, and writing of the manuscript. G.S. supervised the design and execution of the study, and provided critical input into the final data analyses and writing of the manuscript.
Competing interests
The authors declare no competing interests.
Patient consent
Informed consent was obtained from parents or legal guardians.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version of this article (10.1038/s41390-020-01159-x) contains supplementary material, which is available to authorized users.
References
- 1.MacIntyre NR, et al. Evidence-based guidelines for weaning and discontinuing ventilatory support: a collective task force facilitated by the American College of Chest Physicians; the American Association for Respiratory Care; and the American College of Critical Care Medicine. Chest. 2001;120:375s–395s. doi: 10.1378/chest.120.6_suppl.375s. [DOI] [PubMed] [Google Scholar]
- 2.Blackwood B, et al. Use of weaning protocols for reducing duration of mechanical ventilation in critically ill adult patients: Cochrane systematic review and meta-analysis. BMJ. 2011;342:c7237. doi: 10.1136/bmj.c7237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Newth CJ, et al. Weaning and extubation readiness in pediatric patients. Pediatr. Crit. Care Med. 2009;10:1–11. doi: 10.1097/PCC.0b013e318193724d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chawla S, Natarajan G, Gelmini M, Kazzi SNJ. Role of spontaneous breathing trial in predicting successful extubation in premature infants. Pediatr. Pulmonol. 2013;48:443–448. doi: 10.1002/ppul.22623. [DOI] [PubMed] [Google Scholar]
- 5.Kamlin COF, Davis PG, Argus B, Mills B, Morley CJ. A trial of spontaneous breathing to determine the readiness for extubation in very low birth weight infants: a prospective evaluation. Arch. Dis. Child. Fetal Neonatal Ed. 2008;93:F305–F306. doi: 10.1136/adc.2007.129890. [DOI] [PubMed] [Google Scholar]
- 6.Zhang Q, et al. Application of NT‐proBNP in ventilator weaning for preterm infants with RDS. Pediatr. Pulmonol. 2014;49:757–763. doi: 10.1002/ppul.22875. [DOI] [PubMed] [Google Scholar]
- 7.Andrade LB, et al. Spontaneous breathing trial evaluation in preterm newborns extubation. Rev. Bras. Ter. Intensiva. 2010;22:159–165. [PubMed] [Google Scholar]
- 8.Shalish W, Latremouille S, Papenburg J, Sant’Anna GM. Predictors of extubation readiness in preterm infants: a systematic review and meta-analysis. Arch. Dis. Child. Fetal Neonatal Ed. 2019;104:F89–F97. doi: 10.1136/archdischild-2017-313878. [DOI] [PubMed] [Google Scholar]
- 9.Al-Mandari H, et al. International survey on periextubation practices in extremely preterm infants. Arch. Dis. Child. Fetal Neonatal Ed. 2015;100:F428–F431. doi: 10.1136/archdischild-2015-308549. [DOI] [PubMed] [Google Scholar]
- 10.Al Mandhari H, Finelli M, Chen S, Tomlinson C, Nonoyama ML. Effects of an extubation readiness test protocol at a tertiary care fully outborn neonatal intensive care unit. Can. J. Respir. Ther. 2019;55:81–88. doi: 10.29390/cjrt-2019-011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stein H, Beck J, Dunn M. Non-invasive ventilation with neurally adjusted ventilatory assist in newborns. Semin. Fetal Neonatal Med. 2016;21:154–161. doi: 10.1016/j.siny.2016.01.006. [DOI] [PubMed] [Google Scholar]
- 12.Sinderby C, et al. Diaphragm activation during exercise in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2001;163:1637–1641. doi: 10.1164/ajrccm.163.7.2007033. [DOI] [PubMed] [Google Scholar]
- 13.Beck J, et al. Electrical activity of the diaphragm during pressure support ventilation in acute respiratory failure. Am. J. Respir. Crit. Care Med. 2001;164:419–424. doi: 10.1164/ajrccm.164.3.2009018. [DOI] [PubMed] [Google Scholar]
- 14.Sinderby C, et al. Neural control of mechanical ventilation in respiratory failure. Nat. Med. 1999;5:1433–1436. doi: 10.1038/71012. [DOI] [PubMed] [Google Scholar]
- 15.Dres M, et al. Diaphragm electromyographic activity as a predictor of weaning failure. Intens. Care Med. 2012;38:2017–2025. doi: 10.1007/s00134-012-2700-3. [DOI] [PubMed] [Google Scholar]
- 16.Barwing J, Pedroni C, Olgemöller U, Quintel M, Moerer O. Electrical activity of the diaphragm (EAdi) as a monitoring parameter in difficult weaning from respirator: a pilot study. Crit. Care. 2013;17:R182. doi: 10.1186/cc12865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wolf GK, Walsh BK, Green ML, Arnold JH. Electrical activity of the diaphragm during extubation readiness testing in critically ill children. Pediatr. Crit. Care Med. 2011;12:e220–e224. doi: 10.1097/PCC.0b013e3181fe28fc. [DOI] [PubMed] [Google Scholar]
- 18.Liu L, et al. Neuroventilatory efficiency and extubation readiness in critically ill patients. Crit. Care. 2012;16:R143. doi: 10.1186/cc11451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rehan VK, Laiprasert J, Wallach M, Rubin LP, McCool FD. Diaphragm dimensions of the healthy preterm infant. Pediatrics. 2001;108:e91. doi: 10.1542/peds.108.5.e91. [DOI] [PubMed] [Google Scholar]
- 20.Ashwell, K. W. S. & Waite, P. M. E. In The Human Nervous System 3rd edn (eds Jürgen, K. & Paxinos, G.) 14–30 (Academic Press, San Diego, 2012).
- 21.Butler, A. S. & Behrman, R. E. Preterm Birth: Causes, Consequences, and Prevention (National Academies Press, 2007). [PubMed]
- 22.Iyer, N. P. et al. Neural breathing pattern in newborn infants pre‐and postextubation. Acta Paediatr 106, 1928–1933 (2017). [DOI] [PubMed]
- 23.Baudin F, et al. Impact of ventilatory modes on the breathing variability in mechanically ventilated infants. Front. Pediatr. 2014;2:132. doi: 10.3389/fped.2014.00132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brack T, Jubran A, Tobin MJ. Effect of resistive loading on variational activity of breathing. Am. J. Respir. Crit. Care Med. 1998;157:1756–1763. doi: 10.1164/ajrccm.157.6.9704114. [DOI] [PubMed] [Google Scholar]
- 25.Schmidt M, et al. Neurally adjusted ventilatory assist increases respiratory variability and complexity in acute respiratory failure. Anesthesiology. 2010;112:670–681. doi: 10.1097/ALN.0b013e3181cea375. [DOI] [PubMed] [Google Scholar]
- 26.Hutten GJ, et al. Respiratory muscle activity related to flow and lung volume in preterm infants compared with term infants. Pediatr. Res. 2010;68:339–343. doi: 10.1203/PDR.0b013e3181eeeaf4. [DOI] [PubMed] [Google Scholar]
- 27.Usemann J, et al. Variability of tidal breathing parameters in preterm infants and associations with respiratory morbidity during infancy: a cohort study. J. Pediatr. 2019;205:61–69.e61. doi: 10.1016/j.jpeds.2018.10.002. [DOI] [PubMed] [Google Scholar]
- 28.Kaczmarek J, Kamlin CO, Morley CJ, Davis PG, Sant’anna GM. Variability of respiratory parameters and extubation readiness in ventilated neonates. Arch. Dis. Child. Fetal Neonatal Ed. 2013;98:F70–F73. doi: 10.1136/fetalneonatal-2011-301340. [DOI] [PubMed] [Google Scholar]
- 29.Shalish W, et al. Prediction of extubation readiness in extremely preterm infants by the automated analysis of cardiorespiratory behavior: study protocol. BMC Pediatr. 2017;17:167. doi: 10.1186/s12887-017-0911-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Stein H, Hall R, Davis K, White DB. Electrical activity of the diaphragm (Edi) values and Edi catheter placement in non-ventilated preterm neonates. J. Perinatol. 2013;33:707–711. doi: 10.1038/jp.2013.45. [DOI] [PubMed] [Google Scholar]
- 31.Kanbar LJ, et al. Undersampling and bagging of decision trees in the analysis of cardiorespiratory behavior for the prediction of extubation readiness in extremely preterm infants. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2018;2018:4940–4944. doi: 10.1109/EMBC.2018.8513194. [DOI] [PubMed] [Google Scholar]
- 32.Precup D, et al. Prediction of extubation readiness in extreme preterm infants based on measures of cardiorespiratory variability. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2012;2012:5630–5633. doi: 10.1109/EMBC.2012.6347271. [DOI] [PubMed] [Google Scholar]
- 33.Robles-Rubio CA, et al. Automated analysis of respiratory behavior in extremely preterm infants and extubation readiness. Pediatr. Pulmonol. 2015;50:479–486. doi: 10.1002/ppul.23151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cameron YL, Merazzi D, Mortola JP. Variability of the breathing pattern in newborn rats: effects of ambient temperature in normoxia or hypoxia. Pediatr. Res. 2000;47:813–818. doi: 10.1203/00006450-200006000-00022. [DOI] [PubMed] [Google Scholar]
- 35.Task Force of The European Society of Cardiology and The North American Society of Pacing and Electrophysiology. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Circulation. 1996;93:1043–1065. [PubMed] [Google Scholar]
- 36.Kaczmarek J, et al. Heart rate variability and extubation readiness in extremely preterm infants. Neonatology. 2013;104:42–48. doi: 10.1159/000347101. [DOI] [PubMed] [Google Scholar]
- 37.Latremouille S, et al. Heart rate variability in extremely preterm infants receiving nasal CPAP and non-synchronized noninvasive ventilation immediately after extubation. Respir. Care. 2018;63:62–69. doi: 10.4187/respcare.05672. [DOI] [PubMed] [Google Scholar]
- 38.Estay A, Claure N, D’Ugard C, Organero R, Bancalari E. Effects of instrumental dead space reduction during weaning from synchronized ventilation in preterm infants. J. Perinatol. 2010;30:479–483. doi: 10.1038/jp.2009.187. [DOI] [PubMed] [Google Scholar]
- 39.Nassabeh‐Montazami S, Abubakar KM, Keszler M. The impact of instrumental dead‐space in volume‐targeted ventilation of the extremely low birth weight (ELBW) infant. Pediatr. Pulmonol. 2009;44:128–133. doi: 10.1002/ppul.20954. [DOI] [PubMed] [Google Scholar]
- 40.Shalish W, et al. Assessment of extubation readiness using spontaneous breathing trials in extremely preterm neonates. JAMA Pediatr. 2020;174:178–185. doi: 10.1001/jamapediatrics.2019.4868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kenaley KM, et al. Impact of endotracheal tube biofilm and respiratory secretions on airway resistance and mechanics of breathing in a neonatal lung model. J. Appl. Physiol. 2018;125:1227–1231. doi: 10.1152/japplphysiol.00083.2018. [DOI] [PubMed] [Google Scholar]
- 42.Thille AW, Richard J-CM, Brochard L. The decision to extubate in the intensive care unit. Am. J. Respir. Crit. Care Med. 2013;187:1294–1302. doi: 10.1164/rccm.201208-1523CI. [DOI] [PubMed] [Google Scholar]
- 43.Khemani RG, et al. Pediatric extubation readiness tests should not use pressure support. Intens. Care Med. 2016;42:1214–1222. doi: 10.1007/s00134-016-4387-3. [DOI] [PubMed] [Google Scholar]
- 44.de Waal C, Hutten G, Kraaijenga J, de Jongh F, van Kaam A. Electrical activity of the diaphragm during nCPAP and high flow nasal cannula. Arch. Dis. Child. Fetal Neonatal Ed. 2017;102:F434–F438. doi: 10.1136/archdischild-2016-312300. [DOI] [PubMed] [Google Scholar]
- 45.Mortola JP. How to breathe? Respiratory mechanics and breathing pattern. Respir. Physiol. Neurobiol. 2019;261:48–54. doi: 10.1016/j.resp.2018.12.005. [DOI] [PubMed] [Google Scholar]
- 46.Navarro X, Porée F, Beuchée A, Carrault G. Artifact rejection and cycle detection in immature breathing: application to the early detection of neonatal sepsis. Biomed. Signal Process. Control. 2015;16:9–16. [Google Scholar]
- 47.Patel M, et al. Clinical associations with immature breathing in preterm infants: part 2—periodic breathing. Pediatr. Res. 2016;80:28–34. doi: 10.1038/pr.2016.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Iwasaki, E. et al. Postnatal physiological changes in electrical activity of the diaphragm in extremely preterm infants. Pediatr. Pulmonol. 55, 1969–1973 (2020). [DOI] [PubMed]
- 49.Nasef, N. et al. High‐flow nasal cannulae are associated with increased diaphragm activation compared with nasal continuous positive airway pressure in preterm infants. Acta Paediatr. 104, 337–343 (2015). [DOI] [PubMed]
- 50.Kraaijenga JV, de Waal CG, Hutten GJ, de Jongh FH, van Kaam AH. Diaphragmatic activity during weaning from respiratory support in preterm infants. Arch. Dis. Child. Fetal Neonatal Ed. 2016;102:F307–F311. doi: 10.1136/archdischild-2016-311440. [DOI] [PubMed] [Google Scholar]
- 51.Singh N, McNally MJ, Darnall RA. Does diaphragmatic electrical activity in preterm infants predict extubation success? Respir. Care. 2018;63:203–207. doi: 10.4187/respcare.05539. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.