Abstract
The severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) pandemic has provoked major stresses on the health‐care systems of several countries, and caused the death of more than a quarter of a million people globally, mainly in the elderly population with preexisting pathologies. Previous studies with coronavirus (SARS‐CoV) point to gender differences in infection and disease progression with increased susceptibility in male patients, indicating that estrogens may be associated with physiological protection against the coronavirus. Therefore, the objectives of this work are threefold. First, we aim to summarize the SARS‐CoV‐2 infection pathway and the roles both the virus and patient play in COVID‐19 (Coronavirus disease 2019) progression, clinical symptomatology, and mortality. Second, we detail the effect estrogen has on viral infection and host infection response, including its role in both the regulation of key viral receptor expression and the mediation of inflammatory activity. Finally, we describe how ERs (estrogen receptors) and RAGE (receptor for advanced glycation end‐products) play a critical role in metabolic pathways, which we envisage could maintain a close interplay with SARS‐CoV and COVID‐19 mortality rates, despite a current lack of research directly determining how. Taken together, we present the current state of the field regarding SARS‐CoV‐2 research and illuminate where research is needed to better define the role both estrogen and metabolic comorbidities have in the COVID‐19 disease state, which can be key in screening potential therapeutic options as the search for effective treatments continue.
Keywords: ACE2, COVID‐19, estrogen, RAGE
Abbreviations
- AC
adenylyl cyclase 1
- ACE1
angiotensin‐converting enzyme 1
- ACE2
angiotensin‐converting enzyme 2
- AECC
American‐European Consensus Conference
- AGE
advanced glycation end‐products
- AGT
angiotensin
- AngA
angiotensin A
- AP1
activator protein I
- ARDS
acute respiratory distress syndrome
- AT2
alveolar type 2 pneumocytes
- ATR1
angiotensin II receptor type 1
- CFR
case fatality rate
- COPD
chronic obstructive pulmonary disease
- COVID‐19
coronavirus disease 2019
- eNOS
endothelial nitric oxide synthase
- ERE
estrogennonbreakingspaceresponse element
- ERK 1/2
extracellular signal‐regulated kinase 1/2
- ERs
estrogen receptors
- ERα
estrogen receptors α
- ESR1
estrogen receptor 1
- ESR2
estrogen receptor 1
- G‐CSF
granulocyte colony‐stimulating factor)
- GPER1
G protein‐coupled estrogen receptor 1
- HCV
hepatitis C virus
- HMGB1
high mobility group box 1 protein
- IL‐10
interleukin‐10
- IL‐2
interleukin‐2
- IL‐7
interleukin‐7
- IP10
inducible protein 10
- JNK
c‐Jun N‐terminalnonbreakingspacekinase
- KEGG
Kyoto Encyclopedia of Genes and Genomes
- MAPK
mitogen‐activated proteinnonbreakingspacekinase
- MCP1
monocyte chemoattractant protein‐1
- MERS‐CoV
Middle East respiratory Syndrome Coronavirus
- MIP1a
macrophage inflammatory protein 1 alpha
- NADPH
nicotinamide adenine dinucleotide phosphate
- NF‐κB
nuclear factor Kappa B
- NO
nitric oxide
- PAMP
pathogen‐associated molecular patterns
- PI3K‐AKT
phosphatidylinositol 3‐kinase‐ proteinnonbreakingspacekinasenonbreakingspaceB
- PKC
protein kinase C
- PLC
phospholipase C
- PMN
polymorphonuclear cells
- RAGE
receptor for advanced glycation end‐products
- RNS
reactive nitrogen species
- ROS
reactive oxygen species
- Rp3 NP
Rp3nonbreakingspacenucleocapsid protein
- S100A4/Mts1
S100 calcium‐binding protein A4
- SARS‐CoV
Severe Acute Respiratory Syndrome Coronavirus
- SARS‐CoV‐2
Severe Acute Respiratory Syndrome Coronavirus 2
- SERMs
selective estrogen receptor modulators
- SRC
proto‐oncogene non‐receptor tyrosine kinase
- TMPRSS2
type II serine protease
- TNFα
tumor necrosis factor‐α
- WHO
World Health Organization
1. INTRODUCTION
1.1. SARS‐CoV‐2 and COVID‐19
Coronavirus disease 2019 (COVID‐19) is a disease caused by the new coronavirus called SARS‐CoV‐2 (SARS‐coronavirus 2). In December 2019, the first case of COVID‐19 was diagnosed in the city of Wuhan, China. 1 The virus disseminated rapidly and the World Health Organization (WHO) declared SARS‐CoV‐2 as a pandemic on March 11th 2020, given that it had already spread to more than 188 countries on five continents. Updated epidemiological data from the Johns Hopkins University indicator shows that there are more than 20 million cases and over 730 thousand deaths due to COVID‐19 by the beginning of August 2020 (https://coronavirus.jhu.edu/map.html—accessed on August 11, 2020 14:00 GMT). The Case Fatality Rate (CFR) for COVID‐19 increases exponentially with age. 2 , 3 For example, for patients aged between 65 and 74 years old, the CFR is 3%‐5%, 4%‐11% CFR for 75 and 84 years old and 10%‐27% CFR for the patients above 85 years old. 2
Obesity, diabetes, and hypertension are comorbidities associated with increased risk for developing the severe form of COVID‐19. Data attributed to the Centers for Disease Control and Prevention (https://www.cdc.gov/coronavirus/2019‐ncov/need‐extraprecautions/groups‐at‐higher‐risk.html) shows that diabetes is one of the major risk factors for fatal outcomes from COVID‐19. Considering that diabetic patients usually have hyperglycemia, impaired immune function, and several comorbidities such as hypertension, dyslipidemia, and cardiovascular disease, this group is more susceptible to be severely infected by SARS‐CoV‐2. 4 Another important risk for fatal COVID‐19 is obesity, particularly in males. 5 , 6 Moreover, COVID‐19 also affects more severely individuals with metabolic syndrome, probably because these patients have a pro‐inflammatory condition that may contribute to enhance the COVID‐19‐mediated host immune dysregulation. 7 In addition, data from Italy show the CFR is over a third higher for men as compared to women. 3 Interestingly, the authors pointed that although the bias is observed at all ages apparently there is a reduction in the relative risk in older man. 3 To date, many recent publications are arising to reinforce the role of sex bias in COVID‐19, 8 , 9 which suggests that it might be potentially considered in future public health policies.
Coronaviruses are single‐stranded and enveloped RNA viruses that belong to the Coronaviridae family. 10 Several members of this family circulate in the human population and usually lead to mild respiratory diseases. However, two coronavirus subtypes, Severe Acute Respiratory Syndrome Coronavirus (SARS‐CoV) and Middle East Respiratory Syndrome Coronavirus (MERS‐CoV), cause the severe respiratory diseases SARS and MERS, respectively. 11 , 12 Coronaviruses have four structural proteins––spike proteins (S, spike), envelope (E), membrane (M), and nucleocapsid (N). 13 Protein S is important for virus binding, fusion and entry into host cell, and therefore, is a potential target for drugs or vaccines. Once in the respiratory system, coronavirus entry into the host cells appears to depend on the interaction between S protein on the surface of the virus and angiotensin‐converting enzyme 2 (ACE2) molecules on the outer side of lung epithelial cells, 14 and uses the type II serine protease (TMPRSS2) for spike protein priming. 15 When viral S protein binds to ACE2, it is cleaved by TMPRSS2 into S1 and S2 subunits. S1 is bound to ACE2, and S2 is sequentially cleaved into S2' which has the role of promoting the fusion of the viral envelope with the cell membrane. 16 , 17 Besides lung localization, ACE2 and TMPRSS2 are also expressed in intestine, kidney epithelial cells, and endothelial cells. 18 The respiratory and intestinal epithelial cells are the target cells for the replication of SARS‐CoV‐2, which leads to cytological changes and clinical symptoms. 19 Therefore, the ACE2 enzyme acts as the main receptor, which mediates SARS‐CoV and SARS‐CoV‐2 entry into human gut and lung cells, and both ACE2 and TMPRSS2 are possible therapeutic targets for COVID‐19. An important factor is that SARS‐CoV‐2 appears to have a higher affinity for ACE2 than SARS‐CoV, 20 which could explain the significantly larger number of both infected patients. However, the presence of ACE2 alone does not seem to be sufficient to make cells susceptible to infection. SARS‐CoV has failed to infect some cell types, such as endothelial and intestinal cells, that have high ACE2 expression. 21 In contrast, some cells that have a low amount of these receptors, such as hepatocytes, could be infected, 22 which reinforces the importance of looking for other mechanisms.
1.2. ACE2 in lung and differences in age and gender
ACE2 is a surface membrane protein that is implicated in heart function, diabetes, hypertension, and several viral infections in the pulmonary system, such as the SARS‐CoV and SARS‐CoV‐2. 23 , 24 , 25 ACE2 is prominent in pulmonary tissue––including alveolar and bronchial epithelium, the lung parenchyma, and pulmonary vascular structures. 17 ACE2 appears to be increased in the lungs of patients with lung diseases such as chronic obstructive pulmonary disease (COPD), 26 and when this protein is overexpressed it confers a protective effect in pulmonary hypertension, and subsequent cardiac remodeling. 27 , 28 , 29
Jia et al determined several features regarding ACE2 distribution in pulmonary cells and the impact it has on coronavirus infection susceptibility. Localization of ACE2 on the surface of pulmonary cells is highly polarized in human airway epithelia. As a result, coronavirus had significantly higher transduction efficiency in the highly ACE2 populated, apical cell surface in cultured primary human pulmonary epithelial cells, compared to basolateral exposed groups. 25 Pulmonary cell differentiation state is also highly associated with cell susceptibility to coronaviruses. These authors also demonstrated that while fully differentiated pulmonary epithelial cells highly express ACE2 mRNA and have a high density of ACE2 protein in their membranes, undifferentiated epithelial cell membranes have little to no ACE2 expression on both the mRNA and protein level and, as expected, show strong resistance to transduction in comparison to their fully differentiated counterparts. 25
Besides the cellular factors of membrane polarization and differentiation state that play important roles in ACE2 localization and expression in pulmonary epithelial cells, organism‐scale factors such as age and sex also impact ACE2 presence in the pulmonary system. ACE2 maps to the X sex chromosome and has been considered to potentially have a sex‐dependent expression profile in both gene and protein form. 30 On a fetal and neonatal level, both mRNA and protein expression of renal ACE2 increased from birth through the first year of life with no sex difference in ACE2 expression present at this early stage of development in a sheep model. 31 This absence of sex differences in ACE expression in early life stages was observed in other species as well. For example, the ACE2 expression in lung epithelium of young adult and middle‐aged rats demonstrated no sex differences, although both male and female groups showed decline in ACE2 protein levels with age. 30 It is only in the old rat group that a significant sex difference in ACE2 expression is seen in this study––with old male rats demonstrating a significantly lower level of ACE2 when compared to the corresponding female group. 30 Taken together, these studies suggest that though ACE2 is located on the X sex chromosome, that ACE2 expression is stable and similar between the sexes from fetal development through middle age, and that only once late‐stage life is reached will males experience significantly lower levels of ACE2 than female counterparts. 30 , 31 Furthermore a recently published review paper that shed light on the ACE2‐estrogen pathway in COVID‐19, which relates the loss of ACE2 to the development of venous thromboembolism in patients with SARS‐CoV‐2––adding evidence to the current trend that suggests the lower levels of ACE2 in SARS‐CoV‐2 patients may be associated to higher rates of negative patient outcomes. 32 Dalpiaz and colleagues showed that ACE2 expression is elevated in spontaneously hypertensive male mice compared to female mice, and that it correlated with cardiac hypertrophy. This was reversed by orchiectomy, followed by improvement of cardiac performance, and females ovariectomized had more ACE2 expression and a higher incidence cardiac hypertrophy. 33
Epidemiological studies have shown that aging can be an important risk factor of viral infection. A study on SARS‐CoV showed that individuals under 25 years old had mild to moderate symptoms, whereas elderly people over 60 years old have a mortality rate greater than 50%. 34 , 35 The same profile was observed in the laboratory with animal models, where young B6 mice (6 to 10 weeks old) were resistant to SARS‐CoV infection, and mice over 5 months of age were highly susceptible to infection. 36 , 37 An epidemiological study by Karlberg et al showed that incidence and mortality rates were lowest in young women (0‐44 years old) and were increased in women between the ages of 45 and 74. Notably, the protective effect is completely lost in patients over 75 years old, where similar mortality rates are seen in both sexes. 38 These results suggest that there is a time‐dependent loss of protection to viral infection in women. Likewise, data from MERS outbreaks showed that the age groups most affected were 45 to 59 years old and ≥60 years old. It is also important to highlight that the number of deaths in women is higher with age from 45 to 59 years old, 39 which typically corresponds to the beginning of the menopause.
A possible explanation for this age‐related increase in the number of cases and mortality rate may be associated with the decline in immune response in older populations. 40 The immune system declines with age, a phenomenon also known as immunosenescence. 41 Age leads to reduced adaptive immune function and increased pro‐inflammatory activity. 42 In addition, preexisting diseases common in older adults including hypertension, coronary heart disease, and diabetes can increase the risk of COVID‐19. 43 However, these factors do not explain the potentially increased susceptibility of the male gender to these infections.
1.3. Lung inflammation induced by COVID‐19
Inflammation is a natural defense mechanism of the body to remove harmful stimuli such as pathogens and initiate the recovery process. SARS‐CoV‐2 induces COVID‐19 that, in its extreme form, can induce severe pneumonia with intense lung inflammation and the release of high levels of cytokines. Acute lung injury present in COVID‐19 is associated with coagulation alterations and pulmonary embolism that can impair gas exchange and quickly lead the patient to death.
The epithelial tissue of the respiratory system acts as a barrier that actively regulates local immunity with the ability to signal and produce cytokines when activated, and is essential for maintaining the respiratory system tissue homeostasis. 44 , 45 Lung tissue biopsy of SARS‐CoV‐2 patients revealed diffuse alveolar damage, epithelial cells peeling with AT2 (alveolar type 2) pneumocytes reactive hyperplasia, fibrinous exudate associated with interstitial fibrosis and chronic inflammation. 46 Immunohistochemistry stains revealed the presence of Rp3 NP SARS‐CoV‐2 protein mainly in alveolar epithelium cells, including those that were peeled and injured in the alveolar space. 46 The radiological findings of COVID‐19 are variable, but there is a consensus that most of the patients have bilateral lung involvement with the presence of ground‐glass opacity in computed tomography. 46 , 47 , 48 Radiological changes that occur in SARS‐CoV‐2‐induced pneumonia are compatible with radiological findings of severe respiratory infection, in many cases similar to what is observed in acute respiratory distress syndrome (ARDS). Severe pneumonia, ARDS, sepsis, and septic shock 19 are the most common consequences of COVID‐19 pulmonary infection.
Among patients with SARS‐CoV‐2 infection admitted in intensive care units (ICU), approximately 67%‐85% developed ARDS 1 making it the leading cause of mortality. 49 ARDS is defined as an acute and diffuse inflammatory lung injury, which triggers pulmonary vascular permeability, an increase in lung mass due to alveolar edema, and loss of pulmonary tissue due to tissue destruction. The pathophysiology of ARDS is marked by the recruitment and the activation of PMN (polymorphonuclear cells), especially neutrophils, and the consequent release of pro‐inflammatory mediators, such as cytokines and chemokines, as well as ROS (reactive oxygen species) and RNS (reactive nitrogen species). Importantly, alveolar edema and the cytokine storm have a major impact on respiratory failure observed in ARDS. According to the American‐European Consensus Conference (AECC), ARDS is classified by oxygenation level as mild (PaO2/FiO2 200 ≤ 300 mm Hg), moderate (PaO2/FiO2 100 ≤ 200 mm Hg), and severe (PaO2/FiO2 ≤ 100 mm Hg). 50 Similar symptoms have been observed in patients with SARS‐CoV‐2‐induced respiratory failure with impairment of both lungs and severe acute respiratory failure with oxygen‐refractory hypoxemia. The incidence of ARDS in the US population among adolescents is relatively low when compared with elderly populations, 51 , 52 which may contribute to the higher mortality rate due to SARS‐CoV‐2 the elderly and immunosuppressed individuals.
It has been recognized that epithelial dysfunction is an important contributor to pulmonary injury in patients with ARDS. 53 Airway epithelial cells are involved in the secretion of several molecules as surfactant components and pro‐inflammatory mediators. 54 , 55 Interleukin 6 (IL‐6) and Interleukin 8 (IL‐8) are strongly involved in respiratory acute lung injury. Pires‐Neto et al showed that airway epithelial cells from patients with ARDS have increased expression of IL‐8 and IL‐6 compared with controls. 56 In ARDS patients, these cytokines are found in high levels in both serum and bronchoalveolar lavage. 57 , 58 In sepsis, IL‐6 is one of the cytokines that is initially released in acute phase 59 and the chemotactic cytokine IL‐8 is correlated to neutrophil recruitment and severity of lung injury. 60 Is important to consider that epithelial cells can also interact with immune cells such as neutrophils, influencing the signaling pathways. Once in the lung, SARS‐CoV‐2 also can activate immune cells and cytokines.
Several studies have analyzed the bronchoalveolar lavage of patients with ARDS and also observed an increase in Tumor Necrosis Factor‐α (TNFα) levels. 61 , 62 Interestingly, in patients who developed severe SARS‐CoV‐2‐induced infection, high levels of several cytokines such as IL‐2 (Interleukin‐2), IL‐7 (Interleukin‐7), IL‐10 (Interleukin‐10), G‐CSF (Granulocyte colony‐stimulating factor), IP10 (Inducible protein 10), MCP1 (monocyte chemoattractant protein‐1), MIP1a (Macrophage Inflammatory Protein 1 alpha), and TNF‐α were observed. 1 Some evidence described the occurrence of cytokine storm syndrome in patients who developed severe forms of COVID‐19. 63 Lagunas‐Rangel and Chávez‐Valencia suggested that high ratio of IL‐6/IFN‐γ can be associated with the severe form of COVID‐19. 64
It was proposed that T lymphocytes are involved in the pathogenesis of COVID‐19 and provide defense against SARS‐CoV‐2. Wan et al showed that the CD4 + T and CD8 + T were more reduced in the group that developed the most severe form of the disease compared with those that developed the mild form of COVID‐19. 65 This has been previously reported in SARS‐CoV by Chen et al. 66
It should be noted that ARDS as well as SARS‐CoV‐2‐induced severe pneumonia and acute lung inflammation and ARDS do not have a specific treatment, which reinforces the importance of several research groups seeking therapeutic alternatives to reduce mortality and impact on national health systems.
1.4. Estrogen role in lung inflammation
The progress of COVID‐19 plays a major role in the lungs––therefore, targeted modulation of cytokine secretion and hyperinflammation can be an important therapeutic strategy. Evidence from both clinical and experimental studies strongly suggests that estrogen modulates innate and adaptive immune responses. Estrogens can act in either pro or anti‐inflammatory roles depending on the cell type or dose, but it is recognized that this hormone interferes with the prevalence and severity of lung diseases. 67 17β‐estradiol, the predominant circulating estrogen, can modulate both immune cells as well as cytokine release. ERs (estrogen receptors) were detected in immune cells such as neutrophils and macrophages 68 and ERα was detected in both resident lung and inflammatory cells. 69 Low doses of 17β‐estradiol can enhance pro‐inflammatory cytokines production (IL‐1, IL‐6, and TNF‐α), whereas high or sustained concentrations were able to reduce pro‐inflammatory cytokines release. 70 The activation of the ERs can modulate pro‐inflammatory cytokines due to inhibition of NF‐κB, an important nuclear factor for cellular signaling, limiting the severity of the inflammation. 71 , 72
In ARDS, one of the severe complications of COVID‐19, experimental studies have suggested a protective role of estrogen. Rats submitted to acute lung injury induced by seawater aspiration have pulmonary edema reductions by downregulation of aquaporins after the administration of 17β‐ estradiol. 73 Doucet et al showed a reduction in lung injury in ovariectomized rats treated with 17β‐estradiol or agonist receptors, 74 and Vieira et al demonstrated that lung injury is attenuated by 17β‐estradiol in brain‐dead rats and this effect is related to the regulation of NO (nitric oxide) synthases by estrogen. 75 In addition, Fantozzi et al showed that treatment with 17β‐estradiol administered before the induction of acute lung injury induced by intestinal ischemia and reperfusion prevented the systemic and pulmonary release of pro‐inflammatory cytokines. 76
In virus‐induced lung inflammation, exogenous estrogen treatment in female mice infected with H1N1 reduced pulmonary inflammation and the levels of pro‐inflammatory genes, protecting females from a severe form of influenza. 77 High estrogen concentrations in females with SARS‐CoV was able to reduce cytokine storm and eliminated the inflammatory cells. 78
Neutrophils are the central cells to host defense against viral infection. Infected female mice that received the administration of 17β‐estradiol showed elevated chemo attractants recruiting neutrophils into the lungs and adaptive T cell responses. 79 In COVID‐19 patients, T cells seem to be downregulated and are correlated to hospital death and severity of lung damage. 80 Estrogen‐regulated T cell‐mediated autoimmune inflammatory diseases reduced genes associated with pro‐inflammatory cytokines in the lungs and reduced antibody titers during Influenza infection. 77
Together, these data suggest that estrogen can be a tool to be considered in the treatment of COVID‐19 since estrogen not only acts on virus receptors, but also affects acute lung inflammation by the modulation of immune responses.
1.5. ACE2 and estrogen interaction
SARS‐CoV‐2 has been associated with higher mortality rate in male patients than in female patients. 81 According to New York City Health officials, updated data from August 11th 2020 the death rate per 100 000 people was over 283 men vs 173 women (https://github.com/nychealth/coronavirus‐data/blob/master/by‐sex.csv, accessed on 08/11/2020 14:00 GMT). Some hypotheses have been raised, such as a greater tendency among men to become smokers or adopt detrimental health habits though this is contradicted by a study conducted in 2017 by Channappanavar et al with SARS‐CoV, which showed that in experiments with mice this proportion is repeated. 78 In this study, experiments performed on young and elderly mice demonstrated that the susceptibility to infection is age‐dependent. This group also tested the hypothesis that the infection may be sex‐dependent, which showed that mice ovariectomized or treated with ERs antagonist (fulvestrant (ICI)) presented increased susceptibility to infection, evolution to severe cases, and even lethality similar to male mice. This susceptibility may be related to a more aggressive and less specialized immune response and a greater sensibility to infectious agents in men, as well as a stronger adaptive innate response and a greater resistance to viral infections in women. These characteristics are attributed to steroidal hormones and to different numbers of copies of immune response genes linked to the X chromosome. Estrogen in low concentrations has an immune‐stimulation function and its signaling pathway blocks viral replication by modulating genes that regulate metabolic functions. 78 Moreover, as discussed above estrogens also modulate pulmonary inflammation and lung damage in several models of acute inflammation including lung inflammation induced by virus exposure. 76 , 77 Thus, these findings suggest that estrogen can have a protective effect against SARS‐CoV‐2‐induced pneumonia.
As already described, the presence of ACE2 is necessary for virus entry inside host cells. Higher expression of the ACE2 may cause a more efficient viral infection, which could make diabetics and people who use antihypertensive drugs more susceptible. 82 Recent studies demonstrate that G1 ((±)‐1‐[(3aR*,4S*,9bS*)‐4‐(6‐Bromo‐1,3‐benzodioxol‐5‐yl)‐3a,4,5,9b‐tetrahydro‐3H‐cyclopenta[c]quinolin‐8‐yl]‐ ethanone), a selective GPER1 (G protein‐coupled estrogen receptor 1) agonist, reduced ACE2 activity, expression of angiotensin II receptor type 1 (ATR1), and immunoreactivity to angiotensin II. 83 Lu et al showed that ACE2 silencing was able to reduce the replication of SARS‐CoV in vitro, suggesting a possible combined therapy between RNAi and non‐RNAi strategies could reduce viral infection. 84 A network map showing the interactions among ESR1, ESR2, RAGE (receptor for advanced glycation end‐products, see below), ACE2, and TMPRSS2 are shown in Figure 1.
FIGURE 1.
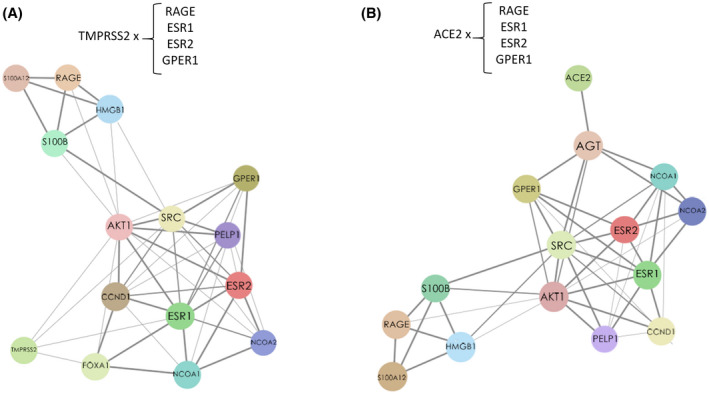
Network map of correlative expression of ERs (estrogen receptors) and related signaling genes (GPER1, ESR1, and ESR2) with RAGE, also associated to TMPRSS2 (A) and ACE2 (B) expression. Adapted from https://string‐db.org/ 144 , 145
Angiotensin I and II is cleaved by ACE2 in angiotensin 1‐9 and angiotensin 1‐7 (with vasodilator actions), respectively. For this reason, ACE2 has been pointed to as a potential participator in the regulation of heart function and target in cardiovascular diseases, hypertension. 85 Although ACE2 has a protein structure that is similar to ACE, it is not inhibited by ACE inhibitors. Instead, ACE inhibitors and angiotensin receptors blockers, usually prescribed to treat hypertension, may increase the amount of ACE2 formation. 86 Therefore, an issue of intense debate has been raised in the medical community, suggesting that hypertensive patients may be more vulnerable to SARS‐CoV‐2 infection, potentially by the use of those drugs. 82 It is important to highlight that this is still controversial and there is currently no consensus in the field regarding these medications. 87 Carey et al showed that the use of angiotensin II receptor blockers and ACE inhibitors in hypertensive patients should not be discontinued during the pandemic induced by COVID‐19. 88 There is evidence that shows that estrogen agonists are capable to reduce ACE2 activity. 83 Considering these findings and given the long time and expensive costs associated with licensing new therapies, the study and use of drugs that modulate the estrogenic route as a possible therapeutic target for COVID‐19 is a promising strategy.
Experimental and clinical studies have shown that the renin‐angiotensin system undergoes sex‐related changes. 89 , 90 17β‐estradiol may be involved in this effect since it regulates the expression of ACE2. 91 , 92 , 93 In a clinical study, it was observed that treatment with estrogen increased the gene expression of ACE2 and reduced ACE in the atrial tissue of male donors, while treatment with the ERα antagonist (MPP—1,3‐Bis(4‐hydroxyphenyl)‐4‐methyl‐5‐[4‐(2‐piperidinyl ethoxy)phenol]‐1H‐pyrazole) reversed this effect. 94 Similarly, a discussion was conducted in a paper by da Silva et al, which showed that in ovariectomized female rats there was a reduction in the expression and activity of ACE2. 95 The direct effect of estrogen to increase ACE2 expression can occur through the interaction of ERα and ERE on the ACE2 promoter. 96 However, these effects may change depending on the tissue involved, since in human endothelial cells there was an estrogen‐mediated increase in the protein expression of ACE1, but not ACE2. 97 The lung tissue of young and middle‐aged male and female rats (3 and 12 months) have a similar expression of ACE2. However, in old animals (24 months) there was a more significant decline in ACE2 expression in male rats. 30
Of note, the exact role of estrogen in ACE2 expression is not fully elucidated. Recently, Stelzig et al reported that 17β‐estradiol is able to reduce the ACE2 mRNA expression in normal human bronchial epithelial cells. 98 In addition, according to Kuba et al, ACE2 knocked out mice showed less SARS‐CoV infection rate induced and demonstrated an attenuation in lung pathological alterations. 99 However, these authors also showed that the infection with SARS‐CoV in wild‐type mice reduced the ACE2 expression in the lung. It is known that ACE can be involved in the protection of acute lung injury, 100 including the potential association of angiotensin II to lung injury and viral load found in COVID‐19 infected patients. 101 Although ACE2 is relevant to the coronavirus cell entry, the reduction in ACE2 levels after the infection can be associated with severe lung injury. 99
Thus, it is not clear yet why women are less susceptible to COVID‐19 infections if the expression of ACE2 is greater in the lung tissue. For example, estrogen and androgen compounds reduce TMPRSS2 levels in MCF‐7 cells, 102 suggesting its potential role of steroid hormones in COVID‐19 therapeutic strategy.
1.6. The role of RAGE related to estrogen signaling: possible connection to risk factors in COVID‐19
Many factors are associated with enhanced susceptibility to cardiovascular diseases and diabetes, which has recently been linked to declining clinical conditions in COVID‐19 patients. One receptor involved in those actions is the receptor for advanced glycation end‐products (RAGE), which is mostly expressed in the lungs (Figure 2). RAGE activation produces a pro‐inflammatory response via NF‐κB maintenance response by enhancing NF‐kappaBp65 expression and degradation of IkappaB. 103 There are many endogenous ligands described, such as S100A, 104 and AGE (advanced glycation end‐products), 105 produced by nonenzymatic glycation of proteins, which increases with age 106 and correlates to disease comorbidities. 107 , 108 These receptors also recognize pathogens and promote the activation of immune responses to infection. Other endogenous ligands of RAGE, such as HMGB1 could be potentially associated with viral infection such as SARS‐CoV 109 (Figure 1). Other molecules, such Pathogen‐associated molecular patterns (PAMP) also activate RAGE, 110 and SARS‐CoV was previously suggested to be an antigen for RAGE activation. 111
FIGURE 2.
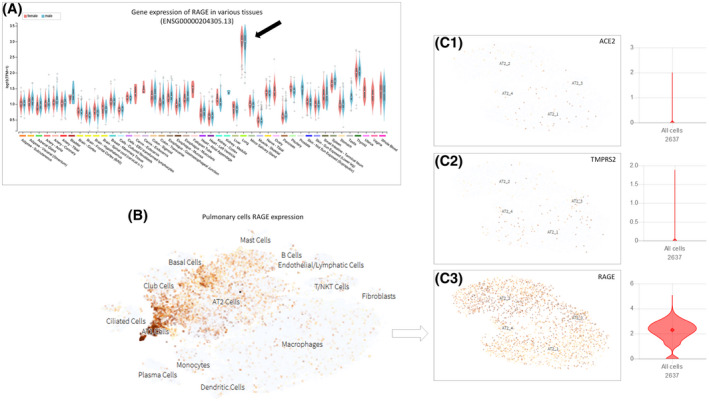
Expression of RAGE in different tissues, mostly expressed in AT2 (alveolar type 2 pneumocyte) cells in lung. A, Values and correlations of RAGE gene expression in many organs, mostly in the lung (dark arrow). Adapted from https://gtexportal.org/. 146 , 147 B, Data set Genes collected from integrated single‐cell RNA‐Seq analysis of patients with pulmonary fibrosis. RAGE (C3) detectable expression is far more pronounced in AT2 cells compared to other important proteins in SARS‐CoV‐2 infection process, such as ACE2 (C1) and TMPRSS2 (C2). Adapted from https://www.nupulmonary.org/ 148
Some of SARS‐CoV proteins, particularly M and 3a of coronavirus can be glycosylated by N‐linked or by O‐linked oligosaccharides, 112 and this glycosylation pattern could be potentially investigated in SARS‐CoV‐2 (Figure 3). Kumar et al analyzed the amino acid sequence of SARS‐CoV and SARS‐CoV‐2 and found 23.6% variation in S glycoprotein receptor‐binding domain, which suggests a significant difference in binding and infectivity of the new coronavirus. 113 Moreover, glycosylation is important in several viral infections such as influenza, Zika, and coronavirus, due to virus strategy to evade immune system and life cycle. 114
FIGURE 3.
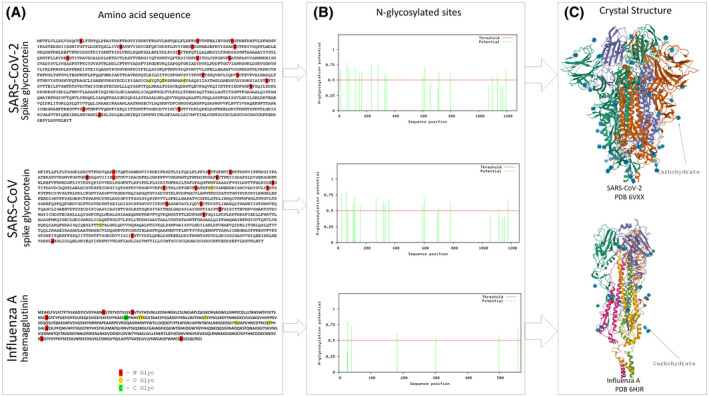
Comparison of potential glycosylation sites in amino acid sequence (A) of the spike protein of SARS‐CoV‐2 (GenBank QIC53213.1) and SARS‐CoV (GenBank AAU04646.1) vs hemagglutinin Influenza A (GenBank BAA01280.1), shows similar numbers of N, O, C glycosylation in coronavirus and fewer glycosylation points in H1N1, as illustrated in histograms of N‐glycosylation sites (B). Adapted from www.cbs.dtu.dk/services/NetNGlyc (Gupta, Jung and Brunak. In preparation, 2004) www.cbs.dtu.dk/services/NetCGlyc 149 ; www.cbs.dtu.dk/services/NetOGlyc. 150 C, The crystallography structure of spike protein SARS‐CoV‐2 and hemagglutinin of Influenza A extracted from Protein Data Bank (https://www.rcsb.org; PDB ID: 6VXX, 151 PDB ID: 6HJR1 152 ) shows dark arrows pointing carbohydrate sites, which are more frequent in SARS‐CoV‐2
The inhibition of RAGE‐signaling pathway has been suggested to be in close association with estrogenic‐mediated protection in cardiovascular diseases. 115 In addition to the effects on AGEs and TNF‐α, direct evidence of the regulation of RAGE by ERs, which are mediated by Sp‐1 protein complex, suggests that it might be a contributing factor for diabetic microvasculopathy. 116 To exemplify this possible association, pyridoxamine, an inhibitor of AGE production, was shown to affect the expression of kidney ERα in vivo, 117 one of the most affected systems by SARS‐CoV‐2 infection. Interestingly, lifestyle changes can affect AGE production which would benefit the patient, as breast cancer survivors who practiced physical activity had reduced circulating AGE levels, 118 and Vlassara et al showed that AGE dietary restrictions positively correlated to insulin resistance amelioration. 119 To our knowledge, the comparison of RAGE expression in male and female has not been fully addressed yet. However, males showed higher levels of HMGB1‐mediated renal injury induced by ischemia and reperfusion compared to females, 120 and HMGB1 is a RAGE ligand.
The impact of estrogen in inflammation was reviewed by Chakrabarti et al, considering several aspects of estrogen therapy in vascular inflammation related to increased tyrosine nitration of proteins and the production of ROS and NO, including RAGE pro‐inflammatory actions. 121 As an example, postmenopausal women treated with conjugated estrogens combined with progestin had elevated levels of NO in serum. 122 In a model of acute lung inflammation induced by intestinal ischemia and reperfusion, the anti‐inflammatory effects of 17β‐estradiol are dependent on the effects of NO produced by eNOS (endothelial nitric oxide synthase). 123 Therefore, considering the regulation of RAGE and its association to diabetes and hypertension, it is noteworthy that one possible cause of the gender disparity in COVID‐19 cases could be linked to RAGE and steroid hormone signaling. The possible effects of estrogen on RAGE signaling are detailed in Table 1.
TABLE 1.
Summary of possible effects of estrogen on RAGE and RAGE ligands
| Effects on RAGE ligands | Possible effects on inflammation | Reference |
|---|---|---|
| Estrogen inhibits RAGE expression and oxidative stress and for this reason, estrogen therapy did not have effect in diabetic woman | Estrogen has a protective effect on inflammation | 115 |
| Estrogen inhibits the synthesis of AGE, the substrate of RAGE in vaginal epithelial tissues of postmenopausal women | These findings indicate a potential anti‐inflammatory and protective role for estrogen | 138 |
| Direct evidence of the regulation of RAGE by ERs (in addition to AGE and TNF‐α) has been demonstrated in endothelial cells | Estrogen has a protector effect on inflammation | 116 |
| The inhibition of intracellular AGE accumulation with pyridoxamine may protect glomeruli against age‐related oxidant stress by preventing an increase of TGFβ production and by regulation of the estrogen receptor | Inhibition of RAGE has a protector effect on inflammation | 117 |
| Lifestyle changes can affect AGE production which would benefit the patient, as breast cancer survivors who practiced physical activity had reduced circulating AGE levels | Reduced AGE production is beneficial in estrogen‐positive breast cancer | 118 |
| Several aspects of estrogen therapy in vascular inflammation related to increased tyrosine nitration of proteins and the production of ROS and NO, including RAGE pro‐inflammatory actions | Estrogen has a pro‐inflammatory action by increasing RAGE | 121 |
| Postmenopausal women treated with conjugated estrogens combined with progestin had elevated levels of NO in serum | High NO levels may produce benefits in cardiovascular system | 122 |
| Female mice that overexpress S100A4/Mts1 (ligand for RAGE) presented greater expression of this protein in pulmonary arterial compared to male, which correlated to elevated pulmonary vascular remodeling and risk to develop pulmonary arterial hypertension in female mice, despite the similar levels of RAGE in both sexes | Treatment with ligands for RAGE is deleterious particularly in female animals | 139 |
RAGE: receptor for advanced glycation end‐products; AGE: advanced glycation end‐products.
Abbreviations: ER, estrogen receptors; NO, nitric oxide.
Specifically in the lung, treatment with either anti‐RAGE mAb (monoclonal antibody) or sRAGE (soluble RAGE) is suggested to increase arterial oxygenation, reduce alveolar inflammation, and improve lung damage in acute lung inflammation 124 , 125 suggesting that the inhibition of RAGE can have a potential therapeutic effect in ARDS. Although there is evidence that RAGE signaling is involved in lung inflammation, 126 there is a lack of studies focused on inhibitors of this pathway in models of lung diseases.
Drucker et al have raised a possible association between diabetes and coronavirus infection by considering the impact of glucose‐lowering therapies on the levels of ACE2 in urine samples, which are found to be increased in diabetes type 1 and 2. 127 , 128 , 129 Therefore, this disease are present in many cases of severe COVID‐19, and it has been suggested that particularly uncontrolled status of hyperglycemia may induce changes in glycosylation of the ACE2, together with glycosylation of the viral spike protein. 130 In addition, it has been discussed by Rojas et al the potential participation of RAGE in COVID‐19’s inflammatory response in lungs, exploring the role of AT1R‐mediated signaling in RAGE transactivation, then, associating angiotensin to inflammation. 131 In this context, the role of RAGE and estrogens should be investigated to draw a comprehensive connection between metabolic activity and susceptibility to COVID‐19. In pulmonary fibrosis, RAGE is located in alveoli, but not in fibrotic tissue, 132 suggesting that the inflammatory process is reduced over time.
1.7. Theoretical effects of estrogen on COVID‐19
A recent paper potentially correlates sex hormones to disease severity by relating preexisting chronic diseases and insulin resistance to defective ER signaling in humans. 133 Importantly, immune response can be also correlated to gender‐associated viral infection. 133 In animal models, ERα knockout mice of both sexes present insulin resistance, glucose tolerance, and obesity, 134 all comorbidities associated with COVID‐19 aggravation.
Selective estrogen receptor modulators (SERMs) that have been developed and approved as anticancer therapies, such as tamoxifen and toremifene, have demonstrated activity against SARS‐CoV and MERS‐CoV, HCV (hepatitis C virus) and Ebola virus and show low toxicity in cell lines. 135 , 136 , 137 The activity of SERMs can occur through gene transcription activation/inhibition after its interaction with classic ERs (ERα/ESR1; and ERβ/ESR2) or modulation of the GPER1 signaling. Watashi et al (2007) showed that tamoxifen inhibits HCV replication by classic estrogenic pathway blockage. It was observed that the ERα located in the endoplasmic reticulum promotes the interaction between HCV replication complex and NS5B polymerase and favors the replication of this virus, an effect that was revoked by tamoxifen treatment. 135 However, in Zika virus infections, ERα overexpression reduces the replication of this virus by 2000 times, suggesting a broader role for this receptor in viral replication. 138 Very importantly, female mice that overexpress S100A4/Mts1 (ligand for RAGE) presented greater expression of this protein in pulmonary arterial compared to male, which correlated to elevated pulmonary vascular remodeling and risk to develop pulmonary arterial hypertension in female mice, despite the similar levels of RAGE in both sexes. 139 Mukhopadhyay and Mukherjee (2005) reviewed several literature data and contrasted the conflicting aspects of RAGE‐estrogen signaling association. 115 For instance, in vaginal epithelium estrogen promotes inhibition of the AGEs’ synthesis, 140 but in endothelial cells cultures it upregulates RAGE expression. 116 Considering that estrogen exerts several anti‐inflammatory effects in lungs, the role of RAGE is still elusive, keeping this issue under debate in literature, and could be further explored in COVID‐19.
SERMs can also play important roles in viral replication by ER‐independent pathways. 136 A study performed in HEK293T cells infected with Ebola virus showed that treatment with toremifene inhibited viral infection by membrane glycoprotein conversion blockage into its functional subunits, thus, preventing fusion between the cell and viral membrane. This result was not reproduced in treatments with tamoxifen, 4‐hydroxy‐tamoxifen and clomiphene for Ebola virus. 141
How to correlate the production of oxygen‐related species, such as ROS and NO, in RAGE/ER signaling is challenging. 115 As estrogen elevates NO levels, and considering that AGE leads to excessive generation of ROS, 142 this may result in peroxynitrite generation, 143 promoting deleterious effects on many organs and promoting a pro‐inflammatory response.
RAGE evokes an inflammatory response in many types of cells. This pathway is also modulated by estrogen signaling, through the rapid activation of MAPK by estrogen membrane receptors. Figure 4 represents a proposed signaling pathway that could be further explored in SARS‐CoV‐2 infection. In this context, we are keen on investigating estrogen‐related interactions with susceptibility to SARS‐CoV‐2 clinical condition aggravation, considering the role in modulating ACE2 levels and RAGE intracellular signaling response, which eventually may result in reduced inflammation and cell protection.
FIGURE 4.
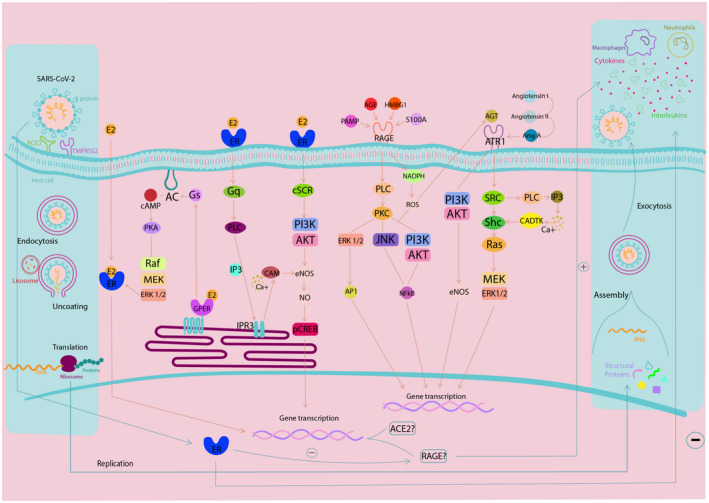
Integrated signaling pathway and summary of events promoted by SARS‐CoV‐2 infection in human cells. This scheme describes the cellular response to SARS‐CoV‐2 entry into the cell mediated by ACE2, which can be modulated by estrogen signaling via transcriptional pathway. Under a pro‐inflammatory response, RAGE can be stimulated by AGE, PAMP, S100A, or HMGB1, which activates NADPH and PLC (phospholipase C). NADPH elevates intracellular ROS levels, which may act in the PI3K‐AKT or AGT (angiotensin) pathway. The PLC activates the PKC, which activates the signaling pathways of ERK 1/2, JNK, and PI3K‐AKT. This results in activation of NF‐κB and ERK 1/2 that ends in AP1 and gene transcription. Regarding renin‐angiotensin signaling, AGT and AngA (angiotensin A) stimulate ATR1, leading to the activation of PI3K‐AKT signaling pathway, promoting eNOS release. In turn, ATR1 also activates the SRC (proto‐oncogene non‐receptor tyrosine kinase), which activates Shc‐Ras and finally MEK‐ERK 1/2, heading to gene transcription. The estrogen pathway can also interfere with the internalization of SARS‐CoV‐2, due to its modulation of ACE2 gene transcription via Raf‐MEK‐ERK 1/2 pathway, which can be mediated by nuclear membrane receptors or associated with G protein. Adapted from KEGG (Kyoto Encyclopedia of Genes and Genomes) 153 and Xiang et al 154 Therefore, according to literature data, here, we propose that estrogen receptors activation may participate in modulation of ACE2 levels and modulation of RAGE cell response, which in turn could reduce inflammation in COVID‐19
2. CONCLUSIONS AND PERSPECTIVES
Overall, regarding COVID‐19 risk factors, metabolic syndromes are exacerbated in the aging process, and therefore, may be closely associated with deteriorating clinical conditions of patients caused by SARS‐CoV‐2 disease progression. To date, no evidence exists to illuminate the role of RAGE or AGE to COVID‐19, however, given that metabolic diseases are rampant in vulnerable aging populations, they should be the focus of further investigations regarding the complex SARS infection pathway given that both diabetes and hypertension are comorbidities associated with high fatality rates. Remarkably, the clinical condition aggravation in men compared to women opens the possibility to explore the role of estrogens in disease management, considering that experimental data correlates ERs stimulation to ACE2 levels. In addition, the well‐known connection of estrogens to inflammation could be critical in attempting to balance immune responses to SARS infection. Furthermore, there is a long list of literature and clinical data that supports ER‐targeting drugs in the treatment of different diseases, which opens the opportunity for drug‐repurposing, with the main advantage of using drugs with human safety already determined. Therefore, future pharmacological directions should address the modulation of the estrogen pathway as a therapeutic strategy for COVID‐19.
CONFLICT OF INTEREST
The authors have stated explicitly that there are no conflicts of interest in connection with this article.
AUTHOR CONTRIBUTIONS
Designed research (R.P. Ureshino, R.S. Stilhano, C.M. Prado); Performed research (R.P. Ureshino, R.S. Stilhano, C.M. Prado, M.S. Nishino, S. Shams, A.J. Costa, C.S. Bartolomeo, A.L. Ramirez, A.C. Breithaupt‐Faloppa, E.A. Silva); Contributed new reagents or analytic tools (R.P. Ureshino, R.S. Stilhano, C.M. Prado, M.S. Nishino, S. Shams, A.J. Costa, C.S. Bartolomeo, A.L. Ramirez, A.C. Breithaupt‐Faloppa, E.A. Silva); Analyzed data (R.P. Ureshino, R.S. Stilhano, C.M. Prado, M.S. Nishino, S. Shams, A.J. Costa, C.S. Bartolomeo, A.L. Ramirez, A.C. Breithaupt‐Faloppa, E.A. SilvaE.A. Silva); Wrote the paper (R.P. Ureshino, R.S. Stilhano, C.M. Prado, M.S. Nishino, S. Shams, A.J. Costa, C.S. Bartolomeo, A.L. Ramirez, A.C. Breithaupt‐Faloppa, E.A. Silva).
ACKNOWLEDGMENTS
The authors thank to the Fundação de Amparo à Pesquisa do Estado de São Paulo—FAPESP: 2016/20796‐2 (RPU), 2020/04709‐8 (RPU), 2018/16719‐8 (MSN), 2018/06088‐0 (CMP); Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES: code 001 (AJC, CSB), Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq: 303035/2018‐8 (CMP); FAP‐FCMSCSP 2019/2021; NIGMS‐funded Pharmacology Training Program: T32GM099608 (SS); American Heart Association grant number 19IPLOI34760654 (EAS).
Stilhano RS, Costa AJ, Nishino MS, et al. SARS‐CoV‐2 and the possible connection to ERs, ACE2, and RAGE: Focus on susceptibility factors. The FASEB Journal. 2020;34:14103–14119. 10.1096/fj.202001394RR
REFERENCES
- 1. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497‐506. 10.1016/S0140-6736(20)30183-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Le Couteur DG, Anderson RM, Newman AB. COVID‐19 through the lens of gerontology. J Gerontol A Biol Sci Med Sci: Series A. 2020:glaa077. 10.1093/gerona/glaa077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Promislow DEL. A geroscience perspective on COVID‐19 mortality. J Gerontol A Biol Sci Med Sci: Series A. 2020:glaa094. 10.1093/gerona/glaa094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Huang I, Lim MA, Pranata R. Diabetes mellitus is associated with increased mortality and severity of disease in COVID‐19 pneumonia—a systematic review, meta‐analysis, and meta‐regression. Diabetes Metab Syndr. 2020;14(4):395‐403. 10.1016/j.dsx.2020.04.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Uzzan M, Corcos O, Martin JC, Treton X, Bouhnik Y. Why is SARS‐CoV‐2 infection more severe in obese men? The gut lymphatics—lung axis hypothesis. Med Hypotheses. 2020;144:110023. 10.1016/j.mehy.2020.110023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Deng M, Qi Y, Deng L, et al. Obesity as a potential predictor of disease severity in young COVID‐19 patients: a retrospective study. Obesity (Silver Spring). 2020:1–11. 10.1002/oby.22943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Bansal R, Gubbi S, Muniyappa R. Metabolic syndrome and COVID 19: endocrine‐immune‐vascular interactions shapes clinical course. Endocrinology. 2020;161:bqaa112. 10.1210/endocr/bqaa112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Maleki Dana P, Sadoughi F, Hallajzadeh J, et al. An insight into the sex differences in covid‐19 patients: what are the possible causes? Prehosp Disaster Med. 2020;35(4):438‐441. 10.1017/S1049023X20000837 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Li Y, Jerkic M, Slutsky AS, Zhang H. Molecular mechanisms of sex bias differences in COVID‐19 mortality. Crit Care. 2020;24(1):405. 10.1186/s13054-020-03118-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Chan JF, Kok KH, Zhu Z, et al. Genomic characterization of the 2019 novel human‐pathogenic coronavirus isolated from a patient with atypical pneumonia after visiting Wuhan. Emerg Microbes Infect. 2020;9(1):221‐236. 10.1080/22221751.2020.1719902 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Fehr AR, Channappanavar R, Perlman S. Middle east respiratory syndrome: emergence of a pathogenic human coronavirus. Annu Rev Med. 2017;68:387‐399. 10.1146/annurev-med-051215-031152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Channappanavar R, Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin Immunopathol. 2017;39(5):529‐539. 10.1007/s00281-017-0629-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Tai W, He L, Zhang X, et al. Characterization of the receptor‐binding domain (RBD) of 2019 novel coronavirus: implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cell Mol Immunol. 2020;17(6):613‐620. 10.1038/s41423-020-0400-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Xu X, Chen P, Wang J, et al. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci China Life Sci. 2020;63(3):457‐460. 10.1007/s11427-020-1637-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Hoffmann M, Kleine‐Weber H, Schroeder S, et al. SARS‐CoV‐2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271‐280.e8. 10.1016/j.cell.2020.02.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Glowacka I, Bertram S, Muller MA, et al. Evidence that TMPRSS2 activates the severe acute respiratory syndrome coronavirus spike protein for membrane fusion and reduces viral control by the humoral immune response. J Virol. 2011;85(9):4122‐4134. 10.1128/JVI.02232-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203(2):631‐637. 10.1002/path.1570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. D'Amico F, Baumgart DC, Danese S, Peyrin‐Biroulet L. Diarrhea during COVID‐19 infection: pathogenesis, epidemiology, prevention and management. Clin Gastroenterol Hepatol. 2020;18(8):1663‐1672. 10.1016/j.cgh.2020.04.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Wujtewicz M, Dylczyk‐Sommer A, Aszkielowicz A, Zdanowski S, Piwowarczyk S, Owczuk R. COVID‐19—what should anaethesiologists and intensivists know about it? Anaesthesiol Intensive Ther. 2020;52(1):34‐41. 10.5114/ait.2020.93756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Wrapp D, Wang N, Corbett KS, et al. Cryo‐EM structure of the 2019‐nCoV spike in the prefusion conformation. Science. 2020;367(6483):1260‐1263. 10.1126/science.abb2507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Chan PK, To KF, Lo AW, et al. Persistent infection of SARS coronavirus in colonic cells in vitro. J Med Virol. 2004;74(1):1‐7. 10.1002/jmv.20138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. To KF, Lo AW. Exploring the pathogenesis of severe acute respiratory syndrome (SARS): the tissue distribution of the coronavirus (SARS‐CoV) and its putative receptor, angiotensin‐converting enzyme 2 (ACE2). J Pathol. 2004;203(3):740‐743. 10.1002/path.1597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Crackower MA, Sarao R, Oudit GY, et al. Angiotensin‐converting enzyme 2 is an essential regulator of heart function. Nature. 2002;417(6891):822‐828. 10.1038/nature00786 [DOI] [PubMed] [Google Scholar]
- 24. Tikellis C, Johnston CI, Forbes JM, et al. Characterization of renal angiotensin‐converting enzyme 2 in diabetic nephropathy. Hypertension. 2003;41(3):392‐397. 10.1161/01.HYP.0000060689.38912.CB [DOI] [PubMed] [Google Scholar]
- 25. Jia HP, Look DC, Shi L, et al. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J Virol. 2005;79(23):14614‐14621. 10.1128/JVI.79.23.14614-14621.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Leung JM, Yang CX, Tam A, et al. ACE‐2 expression in the small airway epithelia of smokers and COPD Patients: implications for COVID‐19. Eur Respir J. 2020;55(5):2000688. 10.1183/13993003.00688-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Shenoy V, Qi Y, Katovich MJ, Raizada MK. ACE2, a promising therapeutic target for pulmonary hypertension. Curr Opin Pharmacol. 2011;11(2):150‐155. 10.1016/j.coph.2010.12.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Li G, Liu Y, Zhu Y, et al. ACE2 activation confers endothelial protection and attenuates neointimal lesions in prevention of severe pulmonary arterial hypertension in rats. Lung. 2013;191(4):327‐336. 10.1007/s00408-013-9470-8 [DOI] [PubMed] [Google Scholar]
- 29. Liao W, Wu J. The ACE2/Ang (1–7)/MasR axis as an emerging target for antihypertensive peptides. Crit Rev Food Sci Nutr. 2020;1‐15. 10.1080/10408398.2020.1781049 [DOI] [PubMed] [Google Scholar]
- 30. Xie X, Chen J, Wang X, Zhang F, Liu Y. Age‐ and gender‐related difference of ACE2 expression in rat lung. Life Sci. 2006;78(19):2166‐2171. 10.1016/j.lfs.2005.09.038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Chen K, Bi J, Su Y, Chappell MC, Rose JC. Sex‐specific changes in renal angiotensin‐converting enzyme and angiotensin‐converting enzyme 2 gene expression and enzyme activity at birth and over the first year of life. Reprod Sci. 2016;23(2):200‐210. 10.1177/1933719115597760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. La Vignera S, Cannarella R, Condorelli RA, Torre F, Aversa A, Calogero AE. Sex‐specific SARS‐CoV‐2 mortality: among hormone‐modulated ACE2 expression, risk of venous thromboembolism and hypovitaminosis D. Int J Mol Sci. 2020;21(8):2948. 10.3390/ijms21082948 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Dalpiaz PL, Lamas AZ, Caliman IF, et al. Sex hormones promote opposite effects on ACE and ACE2 Activity, Hypertrophy And Cardiac Contractility In Spontaneously Hypertensive Rats. PLoS One. 2015;10(5):e0127515. 10.1371/journal.pone.0127515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Nicholls JM, Poon LL, Lee KC, et al. Lung pathology of fatal severe acute respiratory syndrome. Lancet. 2003;361(9371):1773‐1778. 10.1016/s0140-6736(03)13413-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Chen J, Subbarao K. The Immunobiology of SARS. Annu Rev Immunol. 2007;25:443‐472. 10.1146/annurev.immunol.25.022106.141706 [DOI] [PubMed] [Google Scholar]
- 36. Zhao J, Zhao J, Legge K, Perlman S. Age‐related increases in PGD(2) expression impair respiratory DC migration, resulting in diminished T cell responses upon respiratory virus infection in mice. J Clin Invest. 2011;121(12):4921‐4930. 10.1172/JCI59777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Roberts A, Paddock C, Vogel L, Butler E, Zaki S, Subbarao K. Aged BALB/c mice as a model for increased severity of severe acute respiratory syndrome in elderly humans. J Virol. 2005;79(9):5833‐5838. 10.1128/JVI.79.9.5833-5838.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Karlberg J, Chong DS, Lai WY. Do men have a higher case fatality rate of severe acute respiratory syndrome than women do? Am J Epidemiol. 2004;159(3):229‐231. 10.1093/aje/kwh056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Alghamdi IG, Hussain II, Almalki SS, Alghamdi MS, Alghamdi MM, El‐Sheemy MA. The pattern of Middle East respiratory syndrome coronavirus in Saudi Arabia: a descriptive epidemiological analysis of data from the Saudi Ministry of Health. Int J Gen Med. 2014;7:417‐423. 10.2147/IJGM.S67061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;180(7):934. 10.1001/jamainternmed.2020.0994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Siegrist CA, Aspinall R. B‐cell responses to vaccination at the extremes of age. Nat Rev Immunol. 2009;9(3):185‐194. 10.1038/nri2508 [DOI] [PubMed] [Google Scholar]
- 42. Marquez EJ, Chung CH, Marches R, et al. Sexual‐dimorphism in human immune system aging. Nat Commun. 2020;11(1):751. 10.1038/s41467-020-14396-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID‐19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054‐1062. 10.1016/S0140-6736(20)30566-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Hallstrand TS, Hackett TL, Altemeier WA, Matute‐Bello G, Hansbro PM, Knight DA. Airway epithelial regulation of pulmonary immune homeostasis and inflammation. Clin Immunol. 2014;151(1):1‐15. 10.1016/j.clim.2013.12.003 [DOI] [PubMed] [Google Scholar]
- 45. Munkholm M, Mortensen J. Mucociliary clearance: pathophysiological aspects. Clin Physiol Funct Imaging. 2014;34(3):171‐177. 10.1111/cpf.12085 [DOI] [PubMed] [Google Scholar]
- 46. Zhang H, Zhou P, Wei Y, et al. Histopathologic changes and SARS‐CoV‐2 immunostaining in the lung of a patient with COVID‐19. Ann Intern Med. 2020;172(9):629‐632. 10.7326/M20-0533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Lai CC, Shih TP, Ko WC, Tang HJ, Hsueh PR. Severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) and coronavirus disease‐2019 (COVID‐19): The epidemic and the challenges. Int J Antimicrob Agents. 2020;55(3):105924. 10.1016/j.ijantimicag.2020.105924 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Xu X, Yu C, Qu J, et al. Imaging and clinical features of patients with 2019 novel coronavirus SARS‐CoV‐2. Eur J Nucl Med Mol Imaging. 2020;47(5):1275‐1280. 10.1007/s00259-020-04735-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID‐19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46(5):846‐848. 10.1007/s00134-020-05991-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Force ADT, Ranieri VM, Rubenfeld GD, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307(23):2526‐2533. 10.1001/jama.2012.5669 [DOI] [PubMed] [Google Scholar]
- 51. Rubenfeld GD, Caldwell E, Peabody E, et al. Incidence and outcomes of acute lung injury. N Engl J Med. 2005;353(16):1685‐1693. 10.1056/NEJMoa050333 [DOI] [PubMed] [Google Scholar]
- 52. Hecker L. Mechanisms and consequences of oxidative stress in lung disease: therapeutic implications for an aging populace. Am J Physiol Lung Cell Mol Physiol. 2018;314(4):L642‐L653. 10.1152/ajplung.00275.2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Jain M, Sznajder JI. Peripheral airways injury in acute lung injury/acute respiratory distress syndrome. Curr Opin Crit Care. 2008;14(1):37‐43. 10.1097/MCC.0b013e3282f37976 [DOI] [PubMed] [Google Scholar]
- 54. Hohlfeld JM. The role of surfactant in asthma. Respir Res. 2002;3:4. 10.1186/rr176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Shi L, Dong N, Ji D, et al. Lipopolysaccharide‐induced CCN1 production enhances interleukin‐6 secretion in bronchial epithelial cells. Cell Biol Toxicol. 2018;34(1):39‐49. 10.1007/s10565-017-9401-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Pires‐Neto RC, Morales MM, Lancas T, et al. Expression of acute‐phase cytokines, surfactant proteins, and epithelial apoptosis in small airways of human acute respiratory distress syndrome. J Crit Care. 2013;28(1):111.e9‐111.e15. 10.1016/j.jcrc.2012.05.013 [DOI] [PubMed] [Google Scholar]
- 57. Parsons PE, Eisner MD, Thompson BT, et al. Lower tidal volume ventilation and plasma cytokine markers of inflammation in patients with acute lung injury. Crit Care Med. 2005;33(1):1‐6; discussion 230–2. 10.1097/01.ccm.0000149854.61192.dc [DOI] [PubMed] [Google Scholar]
- 58. Schutte H, Lohmeyer J, Rosseau S, et al. Bronchoalveolar and systemic cytokine profiles in patients with ARDS, severe pneumonia and cardiogenic pulmonary oedema. Eur Respir J. 1996;9(9):1858‐1867. 10.1183/09031936.96.09091858 [DOI] [PubMed] [Google Scholar]
- 59. Voisin L, Breuille D, Ruot B, et al. Cytokine modulation by PX differently affects specific acute phase proteins during sepsis in rats. Am J Physiol. 1998;275(5):R1412‐R1419. 10.1152/ajpregu.1998.275.5.R1412 [DOI] [PubMed] [Google Scholar]
- 60. Aggarwal A, Baker CS, Evans TW, Haslam PL. G‐CSF and IL‐8 but not GM‐CSF correlate with severity of pulmonary neutrophilia in acute respiratory distress syndrome. Eur Respir J. 2000;15(5):895‐901. 10.1034/j.1399-3003.2000.15e14.x [DOI] [PubMed] [Google Scholar]
- 61. Li T, Luo N, Du L, Liu J, Gong L, Zhou J. Early and marked up‐regulation of TNF‐alpha in acute respiratory distress syndrome after cardiopulmonary bypass. Front Med. 2012;6(3):296‐301. 10.1007/s11684-012-0219-1 [DOI] [PubMed] [Google Scholar]
- 62. Reper P, Heijmans W. High‐frequency percussive ventilation and initial biomarker levels of lung injury in patients with minor burns after smoke inhalation injury. Burns. 2015;41(1):65‐70. 10.1016/j.burns.2014.05.007 [DOI] [PubMed] [Google Scholar]
- 63. Zhou G, Chen S, Chen Z. Advances in COVID‐19: the virus, the pathogenesis, and evidence‐based control and therapeutic strategies. Front Med. 2020;14(2):117‐125. 10.1007/s11684-020-0773-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Lagunas‐Rangel FA, Chavez‐Valencia V. High IL‐6/IFN‐gamma ratio could be associated with severe disease in COVID‐19 patients. J Med Virol. 2020;92(2):1789‐1790. 10.1002/jmv.25900 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Wan Y, Shang J, Graham R, Baric RS, Li F. Receptor recognition by the novel coronavirus from wuhan: an analysis based on decade‐long structural studies of SARS coronavirus. J Virol. 2020;94(7). 10.1128/JVI.00127-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Chen H, Hou J, Jiang X, et al. Response of memory CD8+ T cells to severe acute respiratory syndrome (SARS) coronavirus in recovered SARS patients and healthy individuals. J Immunol. 2005;175(1):591‐598. 10.4049/jimmunol.175.1.591 [DOI] [PubMed] [Google Scholar]
- 67. Fuentes N, Silveyra P. Endocrine regulation of lung disease and inflammation. Exp Biol Med (Maywood). 2018;243(17–18):1313‐1322. 10.1177/1535370218816653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Cheskis BJ, Greger JG, Nagpal S, Freedman LP. Signaling by estrogens. J Cell Physiol. 2007;213(3):610‐617. 10.1002/jcp.21253 [DOI] [PubMed] [Google Scholar]
- 69. Vegeto E, Cuzzocrea S, Crisafulli C, et al. Estrogen receptor‐alpha as a drug target candidate for preventing lung inflammation. Endocrinology. 2010;151(1):174‐184. 10.1210/en.2009-0876 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Straub RH. The complex role of estrogens in inflammation. Endocr Rev. 2007;28(5):521‐574. 10.1210/er.2007-0001 [DOI] [PubMed] [Google Scholar]
- 71. Garcia‐Alvarez J, Ramirez R, Sampieri CL, et al. Membrane type‐matrix metalloproteinases in idiopathic pulmonary fibrosis. Sarcoidosis Vasc Diffuse Lung Dis. 2006;23(1):13‐21. [PubMed] [Google Scholar]
- 72. Smith LC, Moreno S, Robertson L, et al. Transforming growth factor beta1 targets estrogen receptor signaling in bronchial epithelial cells. Respir Res. 2018;19(1):160. 10.1186/s12931-018-0861-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Fan Q, Zhao P, Li J, et al. 17beta‐Estradiol administration attenuates seawater aspiration‐induced acute lung injury in rats. Pulm Pharmacol Ther. 2011;24(6):673‐681. 10.1016/j.pupt.2011.07.002 [DOI] [PubMed] [Google Scholar]
- 74. Doucet D, Badami C, Palange D, et al. Estrogen receptor hormone agonists limit trauma hemorrhage shock‐induced gut and lung injury in rats. PLoS One. 2010;5(2):e9421. 10.1371/journal.pone.0009421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Vieira RF, Breithaupt‐Faloppa AC, Matsubara BC, et al. 17beta‐Estradiol protects against lung injuries after brain death in male rats. J Heart Lung Transplant. 2018;37(11):1381‐1387. 10.1016/j.healun.2018.06.015 [DOI] [PubMed] [Google Scholar]
- 76. Fantozzi ET, Breithaupt‐Faloppa AC, Ricardo‐da‐Silva FY, et al. Estradiol mediates the long‐lasting lung inflammation induced by intestinal ischemia and reperfusion. J Surg Res. 2018;221:1‐7. 10.1016/j.jss.2017.07.038 [DOI] [PubMed] [Google Scholar]
- 77. Vermillion MS, Ursin RL, Attreed SE, Klein SL. Estriol reduces pulmonary immune cell recruitment and inflammation to protect female mice from severe influenza. Endocrinology. 2018;159(9):3306‐3320. 10.1210/en.2018-00486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Channappanavar R, Fett C, Mack M, Ten Eyck PP, Meyerholz DK, Perlman S. Sex‐based differences in susceptibility to severe acute respiratory syndrome coronavirus infection. J Immunol. 2017;198(10):4046‐4053. 10.4049/jimmunol.1601896 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Robinson DP, Hall OJ, Nilles TL, Bream JH, Klein SL. 17beta‐estradiol protects females against influenza by recruiting neutrophils and increasing virus‐specific CD8 T cell responses in the lungs. J Virol. 2014;88(9):4711‐4720. 10.1128/JVI.02081-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Xu B, Fan CY, Wang AL, et al. Suppressed T cell‐mediated immunity in patients with COVID‐19: A clinical retrospective study in Wuhan, China. J Infect. 2020;81(1):e51‐e60. 10.1016/j.jinf.2020.04.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Sun P, Lu X, Xu C, Sun W, Pan B. Understanding of COVID‐19 based on current evidence. J Med Virol. 2020;92(6):548‐551. 10.1002/jmv.25722 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID‐19 infection? The Lancet Respiratory Medicine. 2020;8(4):e21. 10.1016/S2213-2600(20)30116-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. da Silva JS, Sun X, Ahmad S, et al. G‐protein‐coupled estrogen receptor agonist G1 improves diastolic function and attenuates cardiac renin‐angiotensin system activation in estrogen‐deficient hypertensive rats. J Cardiovasc Pharmacol. 2019;74(5):443‐452. 10.1097/FJC.0000000000000721 [DOI] [PubMed] [Google Scholar]
- 84. Lu CY, Huang HY, Yang TH, Chang LY, Lee CY, Huang LM. siRNA silencing of angiotensin‐converting enzyme 2 reduced severe acute respiratory syndrome‐associated coronavirus replications in Vero E6 cells. Eur J Clin Microbiol Infect Dis. 2008;27(8):709‐715. 10.1007/s10096-008-0495-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Donoghue M, Hsieh F, Baronas E, et al. A novel angiotensin‐converting enzyme‐related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ Res. 2000;87(5):E1‐E9. 10.1161/01.res.87.5.e1 [DOI] [PubMed] [Google Scholar]
- 86. Vuille‐dit‐Bille RN, Camargo SM, Emmenegger L, et al. Human intestine luminal ACE2 and amino acid transporter expression increased by ACE‐inhibitors. Amino Acids. 2015;47(4):693‐705. 10.1007/s00726-014-1889-6 [DOI] [PubMed] [Google Scholar]
- 87. Yang G, Tan Z, Zhou L, et al. Effects of angiotensin II receptor blockers and ace (angiotensin‐converting enzyme) inhibitors on virus infection, inflammatory status, and clinical outcomes in patients with COVID‐19 and hypertension: a single‐center retrospective study. Hypertension. 2020;76(1):51‐58. 10.1161/HYPERTENSIONAHA.120.15143 [DOI] [PubMed] [Google Scholar]
- 88. Carey RM, Wang JG. Evidence that renin‐angiotensin system inhibitors should not be discontinued due to the COVID‐19 pandemic. Hypertension. 2020;76(1):42‐43. 10.1161/HYPERTENSIONAHA.120.15263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Fischer M, Baessler A, Schunkert H. Renin angiotensin system and gender differences in the cardiovascular system. Cardiovasc Res. 2002;53(3):672‐677. 10.1016/s0008-6363(01)00479-5 [DOI] [PubMed] [Google Scholar]
- 90. Bechtloff R, Goette A, Bukowska A, et al. Gender and age‐dependent differences in the bradykinin‐degradation within the pericardial fluid of patients with coronary artery disease. Int J Cardiol. 2011;146(2):164‐170. 10.1016/j.ijcard.2009.06.028 [DOI] [PubMed] [Google Scholar]
- 91. Gupte M, Thatcher SE, Boustany‐Kari CM, et al. Angiotensin converting enzyme 2 contributes to sex differences in the development of obesity hypertension in C57BL/6 mice. Arterioscler Thromb Vasc Biol. 2012;32(6):1392‐1399. 10.1161/ATVBAHA.112.248559 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Ji H, Menini S, Zheng W, Pesce C, Wu X, Sandberg K. Role of angiotensin‐converting enzyme 2 and angiotensin(1–7) in 17beta‐oestradiol regulation of renal pathology in renal wrap hypertension in rats. Exp Physiol. 2008;93(5):648‐657. 10.1113/expphysiol.2007.041392 [DOI] [PubMed] [Google Scholar]
- 93. Joyner J, Neves LA, Granger JP, et al. Temporal‐spatial expression of ANG‐(1–7) and angiotensin‐converting enzyme 2 in the kidney of normal and hypertensive pregnant rats. Am J Physiol Regul Integr Comp Physiol. 2007;293(1):R169‐R177. 10.1152/ajpregu.00387.2006 [DOI] [PubMed] [Google Scholar]
- 94. Bukowska A, Spiller L, Wolke C, et al. Protective regulation of the ACE2/ACE gene expression by estrogen in human atrial tissue from elderly men. Exp Biol Med (Maywood). 2017;242(14):1412‐1423. 10.1177/1535370217718808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. da Silva JS, Gabriel‐Costa D, Wang H, et al. Blunting of cardioprotective actions of estrogen in female rodent heart linked to altered expression of cardiac tissue chymase and ACE2. J Renin Angiotensin Aldosterone Syst. 2017;18(3):1470320317722270. 10.1177/1470320317722270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Wang Y, Shoemaker R, Thatcher SE, Batifoulier‐Yiannikouris F, English VL, Cassis LA. Administration of 17beta‐estradiol to ovariectomized obese female mice reverses obesity‐hypertension through an ACE2‐dependent mechanism. Am J Physiol Endocrinol Metab. 2015;308(12):E1066‐E1075. 10.1152/ajpendo.00030.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Mompeon A, Lazaro‐Franco M, Bueno‐Beti C, et al. Estradiol, acting through ERalpha, induces endothelial non‐classic renin‐angiotensin system increasing angiotensin 1–7 production. Mol Cell Endocrinol. 2016;422:1‐8. 10.1016/j.mce.2015.11.004 [DOI] [PubMed] [Google Scholar]
- 98. Stelzig KE, Canepa‐Escaro F, Schiliro M, Berdnikovs S, Prakash YS, Chiarella SE. Estrogen regulates the expression of SARS‐CoV‐2 receptor ACE2 in differentiated airway epithelial cells. Am J Physiol Lung Cell Mol Physiol. 2020;318(6):L1280‐L1281. 10.1152/ajplung.00153.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Kuba K, Imai Y, Rao S, et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus‐induced lung injury. Nat Med. 2005;11(8):875‐879. 10.1038/nm1267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Imai Y, Kuba K, Rao S, et al. Angiotensin‐converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436(7047):112‐116. 10.1038/nature03712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Liu Y, Yang Y, Zhang C, et al. Clinical and biochemical indexes from 2019‐nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63(3):364‐374. 10.1007/s11427-020-1643-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Baran‐Gale J, Purvis JE, Sethupathy P. An integrative transcriptomics approach identifies miR‐503 as a candidate master regulator of the estrogen response in MCF‐7 breast cancer cells. RNA. 2016;22(10):1592‐1603. 10.1261/rna.056895.116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Bierhaus A, Schiekofer S, Schwaninger M, et al. Diabetes‐associated sustained activation of the transcription factor nuclear factor‐kappaB. Diabetes. 2001;50(12):2792‐2808. 10.2337/diabetes.50.12.2792 [DOI] [PubMed] [Google Scholar]
- 104. Hofmann MA, Drury S, Fu C, et al. RAGE mediates a novel proinflammatory axis: a central cell surface receptor for S100/calgranulin polypeptides. Cell. 1999;97(7):889‐901. 10.1016/s0092-8674(00)80801-6 [DOI] [PubMed] [Google Scholar]
- 105. Neeper M, Schmidt AM, Brett J, et al. Cloning and expression of a cell surface receptor for advanced glycosylation end products of proteins. J Biol Chem. 1992;267(21):14998‐15004. [PubMed] [Google Scholar]
- 106. Chaudhuri J, Bains Y, Guha S, et al. The role of advanced glycation end products in aging and metabolic diseases: bridging association and causality. Cell Metab. 2018;28(3):337‐352. 10.1016/j.cmet.2018.08.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Glenn JV, Stitt AW. The role of advanced glycation end products in retinal ageing and disease. Biochim Biophys Acta. 2009;1790(10):1109‐1116. 10.1016/j.bbagen.2009.04.016 [DOI] [PubMed] [Google Scholar]
- 108. Stern DM, Yan SD, Yan SF, Schmidt AM. Receptor for advanced glycation endproducts (RAGE) and the complications of diabetes. Ageing Res Rev. 2002;1(1):1‐15. 10.1016/s0047-6374(01)00366-9 [DOI] [PubMed] [Google Scholar]
- 109. Wang H, Ward MF, Fan XG, Sama AE, Li W. Potential role of high mobility group box 1 in viral infectious diseases. Viral Immunol. 2006;19(1):3‐9. 10.1089/vim.2006.19.3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. van Zoelen MA, van der Sluijs KF, Achouiti A, et al. Receptor for advanced glycation end products is detrimental during influenza A virus pneumonia. Virology. 2009;391(2):265‐273. 10.1016/j.virol.2009.05.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Wang Y, Liu L. The membrane protein of severe acute respiratory syndrome coronavirus functions as a novel cytosolic pathogen‐associated molecular pattern to promote beta interferon induction via a toll‐like‐receptor‐related TRAF3‐independent mechanism. mBio. 2016;7(1):e01872‐15. 10.1128/mBio.01872-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Oostra M, de Haan CA, de Groot RJ, Rottier PJ. Glycosylation of the severe acute respiratory syndrome coronavirus triple‐spanning membrane proteins 3a and M. J Virol. 2006;80(5):2326‐2336. 10.1128/JVI.80.5.2326-2336.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Kumar S, Maurya VK, Prasad AK, Bhatt MLB, Saxena SK. Structural, glycosylation and antigenic variation between 2019 novel coronavirus (2019‐nCoV) and SARS coronavirus (SARS‐CoV). Virusdisease. 2020;31(1):13‐21. 10.1007/s13337-020-00571-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Watanabe Y, Bowden TA, Wilson IA, Crispin M. Exploitation of glycosylation in enveloped virus pathobiology. Biochim Biophys Acta Gen Subj. 2019;1863(10):1480‐1497. 10.1016/j.bbagen.2019.05.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Mukhopadhyay S, Mukherjee TK. Bridging advanced glycation end product, receptor for advanced glycation end product and nitric oxide with hormonal replacement/estrogen therapy in healthy versus diabetic postmenopausal women: a perspective. Biochim Biophys Acta. 2005;1745(2):145‐155. 10.1016/j.bbamcr.2005.03.010 [DOI] [PubMed] [Google Scholar]
- 116. Tanaka N, Yonekura H, Yamagishi S, Fujimori H, Yamamoto Y, Yamamoto H. The receptor for advanced glycation end products is induced by the glycation products themselves and tumor necrosis factor‐alpha through nuclear factor‐kappa B, and by 17beta‐estradiol through Sp‐1 in human vascular endothelial cells. J Biol Chem. 2000;275(33):25781‐25790. 10.1074/jbc.M001235200 [DOI] [PubMed] [Google Scholar]
- 117. Pereira‐Simon S, Rubio GA, Xia X, et al. Inhibition of advanced glycation end products (AGEs) Accumulation By Pyridoxamine Modulates Glomerular And Mesangial Cell Estrogen Receptor Alpha Expression In Aged Female Mice. PLoS One. 2016;11(7):e0159666. 10.1371/journal.pone.0159666 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Walter KR, Ford ME, Gregoski MJ, et al. Advanced glycation end products are elevated in estrogen receptor‐positive breast cancer patients, alter response to therapy, and can be targeted by lifestyle intervention. Breast Cancer Res Treat. 2019;173(3):559‐571. 10.1007/s10549-018-4992-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Vlassara H, Cai W, Tripp E, et al. Oral AGE restriction ameliorates insulin resistance in obese individuals with the metabolic syndrome: a randomised controlled trial. Diabetologia. 2016;59(10):2181‐2192. 10.1007/s00125-016-4053-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Mohamed R, Rafikova O, O'Connor PM, Sullivan JC. Greater high‐mobility group box 1 in male compared with female spontaneously hypertensive rats worsens renal ischemia‐reperfusion injury. Clin Sci (Lond). 2020;134(13):1751‐1762. 10.1042/CS20200575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Chakrabarti S, Lekontseva O, Davidge ST. Estrogen is a modulator of vascular inflammation. IUBMB Life. 2008;60(6):376‐382. 10.1002/iub.48 [DOI] [PubMed] [Google Scholar]
- 122. Kesim MD, Aydin Y, Erdemir M, Atis A. Nitric oxide in postmenopausal women taking three different HRT regimens. Maturitas. 2005;50(1):52‐57. 10.1016/j.maturitas.2004.04.002 [DOI] [PubMed] [Google Scholar]
- 123. Breithaupt‐Faloppa AC, Fantozzi ET, de Assis Ramos MM, et al. Protective effect of estradiol on acute lung inflammation induced by an intestinal ischemic insult is dependent on nitric oxide. Shock. 2013;40(3):203‐209. 10.1097/SHK.0b013e3182a01e24 [DOI] [PubMed] [Google Scholar]
- 124. Blondonnet R, Audard J, Belville C, et al. RAGE inhibition reduces acute lung injury in mice. Sci Rep. 2017;7(1):7208. 10.1038/s41598-017-07638-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Oczypok EA, Perkins TN, Oury TD. All the “RAGE” in lung disease: The receptor for advanced glycation endproducts (RAGE) is a major mediator of pulmonary inflammatory responses. Paediatr Respir Rev. 2017;23:40‐49. 10.1016/j.prrv.2017.03.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Wang J, Li R, Peng Z, Hu B, Rao X, Li J. HMGB1 participates in LPSinduced acute lung injury by activating the AIM2 inflammasome in macrophages and inducing polarization of M1 macrophages via TLR2, TLR4, and RAGE/NFkappaB signaling pathways. Int J Mol Med. 2020;45(1):61‐80. 10.3892/ijmm.2019.4402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Drucker DJ. Coronavirus infections and type 2 diabetes—shared pathways with therapeutic implications. Endocr Rev. 2020;41(3):457‐470. 10.1210/endrev/bnaa011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Burns KD, Lytvyn Y, Mahmud FH, et al. The relationship between urinary renin‐angiotensin system markers, renal function, and blood pressure in adolescents with type 1 diabetes. Am J Physiol Renal Physiol. 2017;312(2):F335‐F342. 10.1152/ajprenal.00438.2016 [DOI] [PubMed] [Google Scholar]
- 129. Gutta S, Grobe N, Kumbaji M, et al. Increased urinary angiotensin converting enzyme 2 and neprilysin in patients with type 2 diabetes. Am J Physiol Renal Physiol. 2018;315(2):F263‐F274. 10.1152/ajprenal.00565.2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Brufsky A. Hyperglycemia, hydroxychloroquine, and the COVID‐19 pandemic. J Med Virol. 2020;92(7):770‐775. 10.1002/jmv.25887 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Rojas A, Gonzalez I, Morales MA. SARS‐CoV‐2‐mediated inflammatory response in lungs: should we look at RAGE? Inflamm Res. 2020;69(7):641‐643. 10.1007/s00011-020-01353-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Machahua C, Montes‐Worboys A, Llatjos R, et al. Increased AGE‐RAGE ratio in idiopathic pulmonary fibrosis. Respir Res. 2016;17(1):144. 10.1186/s12931-016-0460-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Suba Z. Prevention and therapy of COVID‐19 via exogenous estrogen treatment for both male and female patients. J Pharm Pharm Sci. 2020;23(1):75‐85. 10.18433/jpps31069 [DOI] [PubMed] [Google Scholar]
- 134. Heine PA, Taylor JA, Iwamoto GA, Lubahn DB, Cooke PS. Increased adipose tissue in male and female estrogen receptor‐alpha knockout mice. Proc Natl Acad Sci U S A. 2000;97(23):12729‐12734. 10.1073/pnas.97.23.12729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Watashi K, Inoue D, Hijikata M, Goto K, Aly HH, Shimotohno K. Anti‐hepatitis C virus activity of tamoxifen reveals the functional association of estrogen receptor with viral RNA polymerase NS5B. J Biol Chem. 2007;282(45):32765‐32772. 10.1074/jbc.M704418200 [DOI] [PubMed] [Google Scholar]
- 136. Johansen LM, Brannan JM, Delos SE, et al. FDA‐approved selective estrogen receptor modulators inhibit Ebola virus infection. Sci Transl Med. 2013;5(190):190ra79. 10.1126/scitranslmed.3005471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Dyall J, Coleman CM, Hart BJ, et al. Repurposing of clinically developed drugs for treatment of Middle East respiratory syndrome coronavirus infection. Antimicrob Agents Chemother. 2014;58(8):4885‐4893. 10.1128/AAC.03036-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Lasso G, Mayer SV, Winkelmann ER, et al. A structure‐informed atlas of human‐virus interactions. Cell. 2019;178(6):1526‐1541 e16. 10.1016/j.cell.2019.08.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Dempsie Y, Nilsen M, White K, et al. Development of pulmonary arterial hypertension in mice over‐expressing S100A4/Mts1 is specific to females. Respir Res. 2011;12:159. 10.1186/1465-9921-12-159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Jackson S, James M, Abrams P. The effect of oestradiol on vaginal collagen metabolism in postmenopausal women with genuine stress incontinence. BJOG. 2002;109(3):339‐344. 10.1111/j.1471-0528.2002.01052.x [DOI] [PubMed] [Google Scholar]
- 141. Zhao Y, Ren J, Harlos K, et al. Toremifene interacts with and destabilizes the Ebola virus glycoprotein. Nature. 2016;535(7610):169‐172. 10.1038/nature18615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Schmidt AM, Hori O, Brett J, Yan SD, Wautier JL, Stern D. Cellular receptors for advanced glycation end products. Implications for induction of oxidant stress and cellular dysfunction in the pathogenesis of vascular lesions. Arterioscler Thromb. 1994;14(10):1521‐1528. 10.1161/01.ATV.14.10.1521 [DOI] [PubMed] [Google Scholar]
- 143. Beckman JS, Koppenol WH. Nitric oxide, superoxide, and peroxynitrite: the good, the bad, and ugly. Am J Physiol. 1996;271(5 Pt 1):C1424‐C1437. 10.1152/ajpcell.1996.271.5.C1424 [DOI] [PubMed] [Google Scholar]
- 144. Szklarczyk D, Gable AL, Lyon D, et al. STRING v11: protein‐protein association networks with increased coverage, supporting functional discovery in genome‐wide experimental datasets. Nucleic Acids Res. 2019;47(D1):D607‐D613. 10.1093/nar/gky1131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Snel B, Lehmann G, Bork P, Huynen MA. STRING: a web‐server to retrieve and display the repeatedly occurring neighbourhood of a gene. Nucleic Acids Res. 2000;28(18):3442‐3444. 10.1093/nar/28.18.3442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Consortium GT . The genotype‐tissue expression (GTEx) project. Nat Genet. 2013;45(6):580‐585. 10.1038/ng.2653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Consortium GT . Human genomics. The genotype‐tissue expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science. 2015;348(6235):648‐660. 10.1126/science.1262110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148. Reyfman PA, Walter JM, Joshi N, et al. Single‐cell transcriptomic analysis of human lung provides insights into the pathobiology of pulmonary fibrosis. Am J Respir Crit Care Med. 2019;199(12):1517‐1536. 10.1164/rccm.201712-2410OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149. Julenius K. NetCGlyc 1.0: prediction of mammalian C‐mannosylation sites. Glycobiology. 2007;17(8):868‐876. 10.1093/glycob/cwm050 [DOI] [PubMed] [Google Scholar]
- 150. Steentoft C, Vakhrushev SY, Joshi HJ, et al. Precision mapping of the human O‐GalNAc glycoproteome through SimpleCell technology. EMBO J. 2013;32(10):1478‐1488. 10.1038/emboj.2013.79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Structure VD. Function, and antigenicity of the SARS‐CoV‐2 spike glycoprotein. Cell. 2020;181(2):281‐292 e6. 10.1016/j.cell.2020.02.058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. Benton DJ, Nans A, Calder LJ, et al. Influenza hemagglutinin membrane anchor. Proc Natl Acad Sci U S A. 2018;115(40):10112‐10117. 10.1073/pnas.1810927115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153. Kanehisa M, Goto S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000;28(1):27‐30. 10.1093/nar/28.1.27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Xiang J, Liu X, Ren J, et al. How does estrogen work on autophagy? Autophagy. 2019;15(2):197‐211. 10.1080/15548627.2018.1520549 [DOI] [PMC free article] [PubMed] [Google Scholar]


