Abstract
The world faces a severe and acute public health emergency due to the ongoing coronavirus disease 2019 (COVID-19) global pandemic caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Healthcare workers are in the front line of the COVID-19 outbreak response and are exposed to the risk of SARS-CoV-2 infection daily. Personal protective equipment (PPE) is their main defense against viral contamination; gloves, visors, face masks, and gown materials are designed to eliminate viral transfer from infected patients. Here, we review research investigating the stability of SARS-CoV-2 and similar viruses on surfaces and highlight opportunities for materials that can actively reduce SARS-CoV-2 surface contamination and associated transmission and improve PPE.
Keywords: surface, polymer, virus, SARS-CoV-2, personal protective equipment, contact transfer
Graphical Abstract
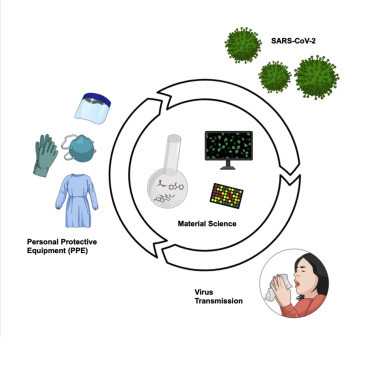
Progress and Potential
It remains to be seen if any of the existing materials commonly used to fabricate PPE may offer improvements in viral binding and inactivation rate at their surface; for example, comparative viral lifetime data cannot be found for the three types of polymer gloves commonly used clinically. The theoretical basis for describing viral particle interactions at synthetic surfaces is not well developed. One means of identifying materials that does not require such a theoretical framework, and could help to build it, is to use high-throughput polymer microarray screening.
There also appears to be an opportunity to develop new polymers for use, i.e., those that bind virus particles strongly and speed the inactivation of adsorbed virus under conditions modeling ambient transmission. In the medium term, there are a number of lines of enquiry for the development of anti-SARS-CoV-2 PPE materials that could reduce contact transfer of infectious virus in clinical and public settings.
The world faces a severe and acute public health emergency caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Healthcare workers are on the front line and exposed to the risk of infection daily. Personal protective equipment (PPE) is their main defense against viral contamination. We review research investigating the binding and stability of SARS-CoV-2 and other viruses on surfaces, and highlight opportunities for materials that can actively reduce infectious SARS-CoV-2 surface contamination and associated transmission to improve PPE.
Introduction
Respiratory diseases caused by viruses have become a serious global public health concern, in particular, as new viruses emerge. The most well-known pandemic in recent history is the Spanish flu pandemic, caused by a strain of the influenza A virus known as H1N1, which led to at least 50 million killed worldwide in 1918.1 Swine flu, caused by H1N1, was responsible for the pandemic in 2009–2010, which spread rapidly from country to country.2 In addition, recent coronavirus outbreaks that caused the respiratory diseases called severe acute respiratory syndrome (SARS, the virus known as SARS-CoV-1), which originated in China in 2002, and Middle East respiratory syndrome (MERS, the virus known as MERS-CoV), first identified in the Middle East in 2012, both caused major disruption and threatened people's lives and livelihoods.3 , 4
The ongoing coronavirus disease 2019 (COVID-19) pandemic, the latest example, has spread rapidly since December 2019. Although the rate of new infections in some countries has reduced significantly recently, other countries are still facing exponential growth. Like other human respiratory viruses, SARS-CoV-2 is reported to spread primarily in small droplets released when an infected person sneezes or coughs, although conversation and breathing have recently been highlighted as potential routes for virus transmission.5 Whereas larger droplets are expected to land on surrounding surfaces within a relatively short time frame, some smaller ones (<5 μm in diameter) may remain airborne and potentially travel larger distances, for example, up to 7–8 m if a turbulent cloud is created.6 , 7 Consequently, the virus is envisaged as spreading through direct inhalation of virus-laden droplets or, more rarely, aerosols, or by hand contact with contaminated surfaces and subsequent transfer to mucus membranes. The infectious dose of SARS-CoV-2, namely the average number of viral particles required to establish an infection for COVID-19, is unknown, but data from other respiratory viruses, such as influenza, indicate that the initial viral dose is directly correlated with the severity of disease symptoms.8
The best approach to prevent viral infections is vaccination, and antiviral drugs are the only treatment option once infected. However, at the moment, in the absence of an effective vaccine or drug, and with the majority of the population unexposed or without immune protection after recovering from COVID-19,9 there is considerable risk of large-scale future outbreaks across the world. Health care workers are on the front line of the COVID-19 outbreak response and are exposed to the risk of SARS-CoV-2 virus infection daily. Personal protective equipment (PPE) is their main defense against viral contamination. In addition to the effort made on vaccines and drugs, new and effective prevention strategies, for example, the development of effective antiviral protective materials for PPE or environmental infection control, potentially reducing the contact transmission of virus pandemics in the future, need to be developed urgently.
SARS-CoV-2 and Surfaces
Viruses cannot reproduce outside the host, relying instead on the host's cellular machinery to produce RNA and to build proteins for their own use. A completely assembled virus, ready to infect a host, is known as a virion. The basic structure of a virion usually composes a nucleic acid (RNA or DNA) core and a protein capsid to protect its nucleic acid. Some viruses, such as coronaviruses, have an additional lipid envelope. The first step of host infection/cell entry is virus attachment to host tissue by recognizing and binding to cell surface receptors with their externally displayed proteins. It is anticipated that the viral surface proteins also play a role when viruses interact with human-made substrates outside a host.
Viral attachment to human-made surfaces is expected to be a function of the physicochemical properties of the material. The binding of viruses on synthetic surfaces has typically been described using nonspecific electrostatic10, 11, 12, 13 and hydrophobic11, 12, 13, 14 interactions. Early studies have shown that the virus’s survival and ability to infect mammalian cells increase with the amount of adsorption to contact transfer surfaces.15 However, more recent studies have demonstrated that strong irreversible adsorption on hydrophobic and polycationic surfaces damaged and inactivated viruses.11, 12, 13 To date, there is still limited information regarding the correlation of viruses’ attachment to materials and their stability outside a host. Virus stability and infectivity under ambient conditions are affected by a combination of the biology (type) of the virus, the environment (temperature, light, relative humidity [RH]), and the physicochemical characteristics of the surface, along with local environmental factors, including carrier liquid identity (saliva/mucus), toxic agents, pH, and salts.16 It is therefore difficult to compare among published studies, as experimental conditions, methods, and types of viruses vary in the conditions chosen to model this complex situation, in the absence of information on the conditions dominant in transmission. The classification of surfaces as either “soft” or “hard” has gained attention. “Soft surfaces,” i.e., cardboard, paper, fabric (which might better be classified as porous surfaces), have been grouped, since they have been observed to support infectious virus for shorter periods, whereas “hard surfaces,” plastic and steel, have been observed to support active virus for longer periods.17 , 18 Here we look in more detail at the importance of polymer surfaces and outline what research is required to choose optimal materials to reduce contact transmission. We also consider what material aspects influence adsorption and virus survival and, in the context of PPE, suggest what improvements can be achieved.
It has been reported that the transmission of SARS-CoV-1 (the most closely related human coronavirus to SARS-CoV-2) starts only after symptoms develop and is associated with detectable viral loads in the respiratory tract that reach a peak after about 10 days.19 In contrast, SARS-CoV-2 virus has been widely detected in the upper respiratory tract in the absence of symptoms,20 highlighting the possibility of viral spread through microdroplets (bioaerosol) generated during breathing or conversation.5 These asymptomatic cases have been shown to have a similar viral load compared with symptomatic patients.21 Like other coronaviruses, SARS-CoV-2 particles are spherical and have proteins protruding from their surface, with the spike protein being the most characteristic. Binding of spike protein to the angiotensin-converting enzyme 2 (ACE2) receptors on human cell surfaces drives structural changes to the protein, which facilitates membrane fusion and entry of the nucleocapsid into the cell cytoplasm.22 , 23 It has been found that the SARS-CoV-2 spike protein has between 10- and 20-fold increased affinity to the ACE2 receptor, compared with SARS-CoV-1, which might facilitate efficient replication in the upper respiratory tract and enable more efficient human transmission.24 Although the major routes of transmission are not fully understood, it is likely that transfer of virus from contaminated surfaces plays a major role. Importantly, virus-laden aerosols may also be generated by the doffing (removing) of PPE, through cleaning, or via the movement of staff, so there could be a benefit to a surface that encourages strong binding.25 , 26
Critically, it is unclear how long the SARS-CoV-2 virus can remain viable outside the host. Viral degradation and inactivation on surfaces are influenced by environmental factors such as humidity, temperature, and light, but critically also the identity of the surface.15 A review article27 published early this year summarized research on the persistence of coronavirus on a range of potential contact transfer surfaces: SARS-CoV-1 and MERS-CoV were found to remain infectious on metal (steel),28 , 29 glass,28 and plastic (type not specified)28, 29, 30 for up to 9 days, under ambient conditions.31 Higher temperature and humidity appear to hasten viral degradation on surfaces, for example, SARS-CoV-1 was reported to remain infectious on plastic (polystyrene) at room temperature and RH of 40%–50% with only 1 log10 loss of titer after 5 days, while a 0.25–2 log10 loss of titer was observed at 38°C and 80%–90% RH within 24 h. Although the surface state of the 24-well plastic plate was unknown, for tissue culture applications oxygen modification of the polystyrene surface is often used.32 More recently, human coronavirus 229E (HCoV-229E) was inoculated onto a variety of surfaces, including steel, polytetrafluoroethylene (Teflon), poly(vinyl chloride) (PVC), ceramic tiles, glass, and silicone rubber. The virus remained infectious for at least 5 days on all surfaces, as shown by a plaque assay that measures the infection of hosts cells in vitro,33 but was reduced to 3 days for silicon rubber.34 In addition, influenza A viruses, the respiratory viruses that cause avian flu, were found to remain detectable up to 2 weeks on stainless steel and up to 1 week on cotton and microfiber under ambient conditions.35 Instead of using qPCR to measure the amount of viral nucleic acid that could arise from inactivated viruses, in most of these studies, the 50% tissue culture infective dose (TCID50) endpoint titration and viral plaque assays were used. These measure the viral infectivity in tissue culture cells and therefore represent a more relevant functional quantification of infectious virus on surfaces.
A more recent study36 compared the stability of SARS-CoV-2 and SARS-CoV-1 in aerosols and on various surfaces at 21°C to 23°C and 40% RH (65% RH for aerosols). A virus dose of 50 μL of 105 TCID50/mL, which has been shown by qPCR to be equivalent to viral loads present in the upper respiratory tract of infected individuals,21 was placed on a variety of surfaces. Due to the nature of liquid absorption on cardboard, the inoculum was recovered from this by first swabbing the surface and then extracting with medium from the swab at predetermined time points, while those on the other materials were recovered directly from the surfaces. All samples were then quantified by TCID50 endpoint titration on Vero E6 cells. In particular, the researchers estimated the decay rates of viable viruses on surfaces using a Bayesian regression model, which allowed them to account for differences in the sources of experimental noise, including the initial inoculum level. Under the experimental conditions, SARS-CoV-2 remained detectable up to 4 h on reddish copper (99.9%, Metal Remnants), up to 24 h on cardboard, and up to 2–3 days on plastic (polypropylene) and AISI 304 alloy stainless steel. SARS-CoV-1 showed similar stability on these tested surfaces compared with SARS-CoV-2, with shorter viability times reported for cardboard. More comprehensive follow-up studies, for example, experiments performed at varying levels of temperature and RH, are being planned by the authors.
Polymers and SARS-CoV-2
The majority of PPE (gloves, gowns, visors, and face masks), and indeed many work surfaces where contact transfer may occur, is polymeric. Disposable polymer gloves are ubiquitous in clinical settings to help prevent cross-contamination between patients and health care workers and to protect against hazardous chemicals and bacteria/viruses. For instance, health care workers wear medical gloves as a barrier to contact with blood, other body fluids, wounds or mucus membranes, and the skin of patients, and higher risk surfaces, to reduce the chance of bacteria/virus transfer. Currently, there are three main types of commercially available medical gloves, including latex, nitrile, and vinyl gloves. Specifically, latex gloves, made of natural rubbers with polyisoprene as their primary chemical constituent, offer a great deal of flexibility, comfort, and fit for health care givers who perform sensitive work. However, as the number of people suffering from allergies to latex is increasing,37 latex-free gloves are now chosen by many professional users. Vinyl gloves, made of PVC, are the most cost-efficient latex-free gloves and are often preferred for low-risk and shorter tasks that still require some level of protection. Polyethylene gloves are not recommended for medical use as they are loose fitting, with limited tensile strength, but are often used in food preparation and serving. Nitrile gloves, made of a synthetic copolymer of acrylonitrile and butadiene monomers, are preferred in clinical areas, as they are stretchable, highly durable, and puncture resistant. Importantly, the protection levels offered by each type of material also vary. Whereas latex gloves were reported to provide the best protection against bacteria and viruses after puncture,38 synthetic gloves offer a higher degree of chemical protection. Medical visors, gowns, and face masks vary in terms of the polymers from which they are made and for what purpose they are worn. Face visors are often made from polyethylene terephthalate (PET), such as a recent COVID-19-specific EU CE-approved design.39 Disposable aprons, usually made from polyethylene to protect against fluids, designed with over-the-head neck and long sleeves, are recommended for use in high-risk areas or aerosol-generating procedures by the UK government's Guidance on Infection Prevention and Control for COVID-19.40 Face masks to reduce the risk of exhaling and/or inhaling viruses, usually made of an outer hydrophobic nonwoven layer (e.g., polypropylene, polyester, and polyaramid), a middle melt-blown/filter layer (e.g., synthetic fibers such as nonwoven polypropylene), and an inner soft absorbent nonwoven layer (e.g., terry cloth towel, quilting cotton, and flannel), vary in their quality and levels of protection.41, 42, 43, 44
An early study compared the maintenance of influenza A virus infectivity on PPE surfaces, including rubber glove (type not specified), N95 particulate respirator, surgical mask (nonwoven fabric), gown made of Dupont Tyvek (polyethylene fiber), coated (coating material not specified) wooden desk, and stainless steel.45 The influenza A viruses were found to remain infectious (≥102.8 TCID50/mL) for at least 8 h on all the surface materials at 25.2°C and 55% RH, but this was increased to 24 h for the rubber glove surface. A recent preprint article studied the SARS-CoV-2 stability on some of the PPE currently used by health care workers.46 A virus dose of 10 μL of 107.88 TCID50/mL in organic components to mimic the typical virus-containing fluids was loaded on each sample surface and dried before the study was started. SARS-CoV-2 was found to be detectable, determined by endpoint titration in Vero E6 cells, up to 7 days on nitrile gloves (copolymer of acrylonitrile and butadiene), 4 days on chemical-resistant gloves (type not specified, but usually made of nitrile rubber), 21 days on plastic face shield (type not specified, but usually made of PET) and N95/N100 particulate respirators, and 14 days on Tyvek (polyethylene fiber) and stainless steel under ambient conditions (20°C and 35%–40% RH). The infectivity of SARS-CoV-2 on cotton was reduced within 4 h of drying, and not detectable by 24 h in the same study. To date, we have been unable to find a systematic study of the stability of SARS-CoV-2, nor of its similar viruses, on different polymers.
To render polymers antimicrobial, the addition of toxic substances to kill surface-located cells has been widely employed, and silver has been proposed as a strategy to inactivate SARS-CoV-1.47 PPE made of PVC with silver coating is commercially available, but is normally much more expensive than the noncoated PPE. Antiviral textiles made of polymer fibers (e.g., nylon) embedded with nanocopper are potentially functional and cost-effective materials for PPE manufacture.48 The incorporation of metals has been proposed as a fast and efficient way to improve the function of existing PPE or coating materials. However, when the metals are employed as nanoparticles, for example, silver, this approach does have the disadvantage that it is likely to release these into the environment, which can potentially contaminate the food chain and be toxic to humans at the cellular and organismic levels.49 A recent review has summarized the advances in antiviral materials and their mechanisms of activity, and it highlighted that nanomaterials, including metal-based, carbon-based, silicon-based, organic-based, and intrinsic antiviral materials, should be studied to improve the antiviral capability of PPE (e.g., filtering materials for face masks).50 In addition, an emerging strategy to treat microbial infection is to create cell-membrane mimics as decoys to trap and detain the pathogens,51 which gives the chance to inactivate the viruses/bacteria while retaining immunogenicity. However, the active-loaded and protein-coated products currently available have limitations for long-term use, as they deplete or degrade over time.
An alternative approach taking advantage of the polymer surface chemistry to modulate cellular attachment has been employed to induce significant effects ranging from bacterial and mammalian cell death to modified viability and phenotype.52 , 53 There is now theoretical and experimental evidence to suggest that polymer surface chemistry may also be used to control virion adsorption strength and therefore reduce the viable lifetime of viruses attached to surfaces.16 , 54 , 55 For example, cationic polymers have shown high affinity for virus binding,10 and hydrophobic polycationic surfaces have been reported to inactivate influenza viruses owing to irreversible attachment on the surface and related viral structure damage and inactivation, which in turn altered the release of their genomic materials.12 , 13 This proposed mechanism was supported by the loss of infectivity and viral proteins of the exposed solution, indicating virion attachment on the surface, and the significant amount of viral RNA quantified in the disinfected solution due to the damage of viral structure.13 Similarly, a recent study reported that surfaces of polypropylene and other PPE materials engineered with both positive charge and hydrophobic features achieved strong binding with the surface spike protein of SARS-CoV-2 viruses.11 The strong binding between the hydrophobic spike protein and surfaces through electrostatic and hydrophobic interactions was proposed to cause protein conformational change and consequently inactivate the viruses. Noncharged oleophilic surfaces have also been demonstrated to destroy the viral lipid envelope upon contact.14 In another study, anionic copolymers with both hydrophobic and hydrophilic blocks were shown to be inherently self-sterilizing and to have broad-spectrum antimicrobial efficacy to successfully inactivate a broad range of bacteria and viruses, including both enveloped vesicular stomatitis virus and influenza A virus and nonenveloped human adenovirus-5, after only 5 min of contact.56 The authors proposed that the high antimicrobial efficacy of these polymers is due to the ability of hydrated sulfonic acid groups to dramatically reduce the pH of the medium, leading to enzyme damage, protein denaturation, and microbe death.
Progress
Looking forward, a systematic study on the lifetime of infectious viruses on a range of existing polymers under ambient conditions would be useful for those choosing which PPE to use; e.g., are viral particle lifetimes similar on vinyl and latex gloves? There also appears to be an opportunity to develop new polymers for use, i.e., those that bind virus particles strongly and speed the inactivation of adsorbed virus under conditions modeling ambient transmission (Figure 1 B). The theoretical basis for describing viral particle interactions at synthetic surfaces is not well developed. One means of identifying materials that does not require such a theoretical framework, and could help to build it, is to use high-throughput polymer microarray screening. Polymer microarray screening has mainly been explored in identifying simple polymers to control bacterial biofilm formation,57 stem cell pluripotency,58 and phenotype control.59 Recent work in screening polymer libraries using microarrays for rubella and Lassa fever virus-like particles illustrates its potential in identifying materials for differential virus binding (Figure 1C),60 but this needs to be progressed to include SARS-CoV-2 viral particle inactivation as a selection criterion in order to allow new polymers to be identified with desirable attachment and inactivation profiles.
Figure 1.
Opportunities for Materials to Actively Reduce SARS-CoV-2 Surface Contamination and Associated Transmission
(A) An image showing fluorescent powder, representing viruses, transferred from door handle to hand after contact.
(B) The to-be-developed new polymer surface binding virus particles strongly and speeding the inactivation of adsorbed virus under ambient conditions (blue, infectious virus particles; orange, inactivated virus particles).
(C) Schematic depiction of the process of polymer microarray screening, showing the printing of the monomers, the in situ UV polymerization of the monomers, and finally their incubation with differently fluorescently tagged virus-like particles.60
The many viral outbreaks in human history, and especially the ongoing COVID-19 pandemic, are urging us to look for effective strategies that might be used to deal with emergent viral diseases in the future. The best long-term population-wide approach to counter viral infections is prevention of infection by good public health measures, but if outbreaks do occur, vaccination is required. However, vaccines are not yet available against COVID-19, and despite the unprecedented speed in developing new candidates, which are showing promise in preclinical models,61 , 62 there is still a lag time between developing a promising lead formulation in the lab and bringing a safe and effective vaccine to the market. In the absence of effective therapeutics and vaccines, there is an urgent quest for broad-spectrum antiviral protective materials that can prevent the contact transmission of existing and emerging viruses. Generally, there are a range of considerations in the design of a material for antiviral surface transmission, for example, the interaction between virions (e.g., enveloped or nonenveloped) and surface materials (e.g., incorporation of antiviral agents or cell-membrane mimics, charge and hydrophobicity), the durability of the antiviral function of the surface, the cost effectiveness, the toxicity of the materials to the environment and mammalian cells, and the environmental conditions (e.g., temperature, RH, and light). However, most antiviral materials proposed so far have limitations for wide use as PPE or as surface coatings, including environmental issues, mammalian cell contact toxicity, loss of effectiveness over time, virus specificity, and cost. A route to identifying polymers using high-throughput polymer microarray screening and predictive quantitative structure-activity relationship has been exemplified for controlling bacterial-surface interactions.63 This approach may be adopted to the development of novel antiviral polymers with the potential to reduce contact transfer of a wide range of infectious viruses, including the emerging ones in the future.
Potential
Since December 2019 the COVID-19 pandemic has affected more than 200 countries and affected people's lives and livelihoods. With no vaccine available at this time and uncertainty over the duration of immunity following infection, the outlook is that we will be dealing with this virus for some time. To date, SARS-CoV-2 appears to spread easily in droplets in the air and via surfaces by infected individuals. Once outside the host cell, the virus cannot replicate, going on to either infect another host or degrade in the environment. Early work indicates that the virus survives longer on some surfaces compared with others. However, it is currently unclear what role the surface chemistry plays in viral survival, infectivity, and denaturation, and the role of the local environment (e.g., medium) is unclear. It remains to be seen if any of the existing materials commonly used to fabricate PPE may offer improvements in viral binding and inactivation rate at their surface; for example, comparative viral lifetime data cannot be found on the three types of polymer gloves commonly used clinically. These are experiments that can readily be undertaken in virology laboratories with access to the virus under biohazard containment conditions. In the medium term, there are a number of lines of enquiry for the development of anti-SARS-CoV-2 PPE materials that could reduce contact transfer of infectious virus in clinical and public settings.
Acknowledgments
M.R.A., C.A., and X.X. would like to thank the Engineering and Physical Sciences Research Council (grant EP/N006615/1) for funding. We also thank the Royal Society for a Wolfson Research Merit Award (WM150086) to C.A.
Author Contributions
M.R.A. and X.X. conceived the paper. X.X. wrote the original draft and revised and edited the manuscript. J.K.B., C.A., and M.R.A. commented on and revised the paper.
References
- 1.National Center for Immunization and Respiratory Diseases, Centers for Disease Control and Prevention . Centers for Disease Control and Prevention; 2018. History of 1918 Flu Pandemic. [Google Scholar]
- 2.Kelly H., Peck H.A., Laurie K.L., Wu P., Nishiura H., Cowling B.J. The age-specific cumulative incidence of infection with pandemic influenza H1N1 2009 was similar in various countries prior to vaccination. PLoS One. 2011;6:e21828. doi: 10.1371/journal.pone.0021828. 1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ksiazek T.G., Erdman D., Goldsmith C.S., Zaki S.R., Peret T., Emery S., Tong S., Urbani C., Comer J.A., Lim W. A novel coronavirus associated with severe acute respiratory syndrome. N. Engl. J. Med. 2003;348:1953–1966. doi: 10.1056/NEJMoa030781. [DOI] [PubMed] [Google Scholar]
- 4.World Health Organization . World Health Organization; 2019. Middle East Respiratory Syndrome Coronavirus (MERS-CoV) [Google Scholar]
- 5.Fineberg H.V. The National Academies Press; 2020. Rapid Expert Consultation on the Possibility of Bioaerosol Spread of SARS-CoV-2 for the COVID-19 Pandemic (April 1, 2020) [Google Scholar]
- 6.Bourouiba L. Turbulent gas clouds and respiratory pathogen emissions: potential implications for reducing transmission of COVID-19. JAMA. 2020 doi: 10.1001/jama.2020.4756. [DOI] [PubMed] [Google Scholar]
- 7.World Health Organization . World Health Organization Guidelines; 2009. Natural Ventilation for Infection Control in Health-Care Settings. [PubMed] [Google Scholar]
- 8.Memoli M.J., Czajkowski L., Reed S., Athota R., Bristol T., Proudfoot K., Fargis S., Stein M., Dunfee R.L., Shaw P.A. Validation of the wild-type influenza A human challenge model H1N1pdMIST: an A(H1N1) pdm09 dose-finding investigational new drug study. Clin. Infect. Dis. 2015;60:693–702. doi: 10.1093/cid/ciu924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Seow J., Graham C., Merrick B., Acors S., Steel K.J.A., Hemmings O., O'Bryne A., Kouphou N., Pickering S., Galao R. Longitudinal evaluation and decline of antibody responses in SARS-CoV-2 infection. medRxiv. 2020 doi: 10.1101/2020.07.09.20148429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rosilo H., McKee J.R., Kontturi E., Koho T., Hytönen V.P., Ikkala O., Kostiainen M.A. Cationic polymer brush-modified cellulose nanocrystals for high-affinity virus binding. Nanoscale. 2014;6:11871–11881. doi: 10.1039/c4nr03584d. [DOI] [PubMed] [Google Scholar]
- 11.Pandey L.M. Surface engineering of personal protective equipments (PPEs) to prevent the contagious infections of SARS-CoV-2. Surf. Eng. 2020;36:901–907. [Google Scholar]
- 12.Haldar J., Chen J., Tumpey T.M., Gubareva L.V., Klibanov A.M. Hydrophobic polycationic coatings inactivate wild-type and zanamivir- and/or oseltamivir-resistant human and avian influenza viruses. Biotechnol. Lett. 2008;30:475–479. doi: 10.1007/s10529-007-9565-5. [DOI] [PubMed] [Google Scholar]
- 13.Hsu B.B., Wong S.Y., Hammond P.T., Chen J.Z., Klibanov A.M. Mechanism of inactivation of influenza viruses by immobilized hydrophobic polycations. Proc. Natl. Acad. Sci. U S A. 2011;108:61–66. doi: 10.1073/pnas.1017012108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mannelli I., Janner D., Sagues F., Reigada R. Assessing the optimal conditions for surface-mediated disinfection of Influenza A virus solutions. Environ. Chem. 2017;14:319–326. [Google Scholar]
- 15.Vasickova P., Pavlik I., Verani M., Carducci A. Issues concerning survival of viruses on surfaces. Food Environ. Virol. 2010;2:24–34. [Google Scholar]
- 16.Joonaki E., Hassanpouryouzband A., Heldt C.L., Areo O. Surface chemistry can unlock drivers of surface stability of SARS-CoV-2 in a variety of environmental conditions. Chem. 2020 doi: 10.1016/j.chempr.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gray R. Covid-19: how long does the coronavirus last on surfaces? BBC Future. 2020 https://www.bbc.com/future/article/20200317-covid-19-how-long-does-the-coronavirus-last-on-surfaces [Google Scholar]
- 18.Division of Viral Diseases, National Center for Immunization and Respiratory Diseases . Centers for Disease Control and Prevention; 2020. Cleaning and Disinfecting Your Home. [Google Scholar]
- 19.Lipsitch M., Cohen T., Cooper B., Robins J.M., Ma S., James L., Gopalakrishna G., Chew S.K., Tan C.C., Samore M.H. Transmission dynamics and control of severe acute respiratory syndrome. Science. 2003;300:1966–1970. doi: 10.1126/science.1086616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nathan W.F., John T.B., Jeremy S. Evidence supporting transmission of severe acute respiratory syndrome coronavirus 2 while presymptomatic or asymptomatic. Emerg. Infect. Dis. 2020;26:e201595. doi: 10.3201/eid2607.201595. 1-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zou L., Ruan F., Huang M., Liang L., Huang H., Hong Z., Yu J., Kang M., Song Y., Xia J. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N. Engl. J. Med. 2020;382:1177–1179. doi: 10.1056/NEJMc2001737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yan R.H., Zhang Y.Y., Li Y.N., Xia L., Guo Y.Y., Zhou Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science. 2020;367:1444–1448. doi: 10.1126/science.abb2762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shang J., Ye G., Shi K., Wan Y.S., Luo C.M., Aihara H., Geng Q.B., Auerbach A., Li F. Structural basis of receptor recognition by SARS-CoV-2. Nature. 2020 doi: 10.1038/s41586-020-2179-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Walls A.C., Park Y.J., Tortorici M.A., Wall A., McGuire A.T., Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181:281–292. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Casanova L., Alfano-Sobsey E., Rutala W.A., Weber D.J., Sobsey M. Virus transfer from personal protective equipment to healthcare employees' skin and clothing. Emerg. Infect. Dis. 2008;14:1291–1293. doi: 10.3201/eid1408.080085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu Y., Ning Z., Chen Y., Guo M., Liu Y., Gali N.K., Sun L., Duan Y., Cai J., Westerdahl D. Aerodynamic characteristics and RNA concentration of SARS-CoV-2 aerosol in Wuhan Hospitals during COVID-19 outbreak. bioRxiv. 2020 doi: 10.1101/2020.03.08.982637. [DOI] [Google Scholar]
- 27.Kampf G., Todt D., Pfaender S., Steinmann E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J. Hosp. Infect. 2020;104:246–251. doi: 10.1016/j.jhin.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Duan S.M., Zhao X.S., Wen R.F., Huang J.J., Pi G.H., Zhang S.X., Han J., Bi S.L., Ruan L., Dong X.P. Stability of SARS coronavirus in human specimens and environment and its sensitivity to heating and UV irradiation. Biomed. Environ. Sci. 2003;16:246–255. [PubMed] [Google Scholar]
- 29.van Doremalen N., Bushmaker T., Munster V.J. Stability of Middle East respiratory syndrome coronavirus (MERS-CoV) under different environmental conditions. Euro. Surveill. 2013;18:7–11. doi: 10.2807/1560-7917.es2013.18.38.20590. [DOI] [PubMed] [Google Scholar]
- 30.Rabenau H.F., Cinatl J., Morgenstern B., Bauer G., Preiser W., Doerr H.W. Stability and inactivation of SARS coronavirus. Med. Microbiol. Immunol. 2005;194:1–6. doi: 10.1007/s00430-004-0219-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Reed L.J., Muench H. A simple method of estimating fifty per cent endpoints. Am. J. Epidemiol. 1938;27:493–497. [Google Scholar]
- 32.Chan K.H., Peiris J.S.M., Lam S.Y., Poon L.L.M., Yuen K.Y., Seto W.H. The effects of temperature and relative humidity on the viability of the SARS coronavirus. Adv. Virol. 2011;2011:734690–734697. doi: 10.1155/2011/734690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Smither S.J., Lear-Rooney C., Biggins J., Pettitt J., Lever M.S., Olinger G.G. Comparison of the plaque assay and 50% tissue culture infectious dose assay as methods for measuring filovirus infectivity. J. Virol. Methods. 2013;193:565–571. doi: 10.1016/j.jviromet.2013.05.015. [DOI] [PubMed] [Google Scholar]
- 34.Warnes S.L., Little Z.R., Keevil C.W. Human coronavirus 229E remains infectious on common touch surface materials. mBio. 2015;6:e01697. doi: 10.1128/mBio.01697-15. 1-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Thompson K.A., Bennett A.M. Persistence of influenza on surfaces. J. Hosp. Infect. 2017;95:194–199. doi: 10.1016/j.jhin.2016.12.003. [DOI] [PubMed] [Google Scholar]
- 36.van Doremalen N., Bushmaker T., Morris D.H., Holbrook M.G., Gamble A., Williamson B.N., Tamin A., Harcourt J.L., Thornburg N.J., Gerber S.I. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N. Engl. J. Med. 2020 doi: 10.1056/NEJMc2004973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cleenewerck M.B. Update on medical and surgical gloves. Eur. J. Dermatol. 2010;20:434–442. doi: 10.1684/ejd.2010.1000. [DOI] [PubMed] [Google Scholar]
- 38.Broyles J.M., O'Connell K.P., Korniewicz D.M. PCR-based method for detecting viral penetration of medical exam gloves. J. Clin. Microbiol. 2002;40:2725–2728. doi: 10.1128/JCM.40.8.2725-2728.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nottingham engineers produce certified 3D printed face shields for NHS. https://www.theengineer.co.uk/nottingham-3d-printed-face-shields-nhs/2020
- 40.A visual guide to safe PPE. Guidance COVID-19: infection prevention and control (IPC). 2020. https://www.cas.mhra.gov.uk/ViewandAcknowledgment/ViewAttachment.aspx?Attachment_id=103509
- 41.Lustig S.R., Biswakarma J.J.H., Rana D., Tilford S.H., Hu W., Su M., Rosenblatt M.S. Effectiveness of common fabrics to block Aqueous aerosols of virus-like nanoparticles. ACS Nano. 2020;14:7651–7658. doi: 10.1021/acsnano.0c03972. [DOI] [PubMed] [Google Scholar]
- 42.El-Atab N., Qaiser N., Badghaish H., Shaikh S.F., Hussain M.M. Flexible nanoporous template for the design and development of reusable anti-COVID-19 hydrophobic face masks. ACS Nano. 2020;14:7659–7665. doi: 10.1021/acsnano.0c03976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zangmeister C.D., Radney J.G., Vicenzi E.P., Weaver J.L. Filtration efficiencies of nanoscale Aerosol by cloth mask materials used to slow the spread of SARS-CoV-2. ACS Nano. 2020 doi: 10.1021/acsnano.0c05025. [DOI] [PubMed] [Google Scholar]
- 44.Zhou S.S., Lukula S., Chiossone C., Nims R.W., Suchmann D.B., Ijaz M.K. Assessment of a respiratory face mask for capturing air pollutants and pathogens including human influenza and rhinoviruses. J. Thorac. Dis. 2018;10:2059–2069. doi: 10.21037/jtd.2018.03.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sakaguchi H., Wada K., Kajioka J., Watanabe M., Nakano R., Hirose T., Ohta H., Aizawa Y. Maintenance of influenza virus infectivity on the surfaces of personal protective equipment and clothing used in healthcare settings. Environ. Health Prev. Med. 2010;15:344–349. doi: 10.1007/s12199-010-0149-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kasloff S.B., Strong J.E., Funk D., Cutts T. Stability of SARS-CoV-2 on critical personal protective equipment. medRxiv. 2020 doi: 10.1101/2020.06.11.20128884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Han J., Chen L., Duan S.M., Yang Q.X., Yang M., Gao C., Zhang B.Y., He H., Dong X.P. Efficient and quick inactivation of SARS coronavirus and other microbes exposed to the surfaces of some metal catalysts. Biomed. Environ. Sci. 2005;18:176–180. [PubMed] [Google Scholar]
- 48.Copper nanoparticles being tested for anti-viral textiles. Innovation in Textiles. 2020. https://www.innovationintextiles.com/copper-nanoparticles-being-tested-for-antiviral-textiles/
- 49.Stensberg M.C., Wei Q., McLamore E.S., Porterfield D.M., Wei A., Sepúlveda M.S. Toxicological studies on silver nanoparticles: challenges and opportunities in assessment, monitoring and imaging. Nanomedicine. 2011;6:879–898. doi: 10.2217/nnm.11.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhou J., Hu Z., Zabihi F., Chen Z., Zhu M. Progress and perspective of antiviral protective material. Adv. Fiber Mater. 2020;2:123–139. doi: 10.1007/s42765-020-00047-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rao L., Tian R., Chen X. Cell-membrane-mimicking nanodecoys against infectious diseases. ACS Nano. 2020;14:2569–2574. doi: 10.1021/acsnano.0c01665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hasan J., Crawford R.J., Lvanova E.P. Antibacterial surfaces: the quest for a new generation of biomaterials. Trends Biotechnol. 2013;31:31–40. doi: 10.1016/j.tibtech.2013.01.017. [DOI] [PubMed] [Google Scholar]
- 53.Randazzo W., Fabra M.J., Falcó I., López-Rubio A., Sánchez G. Polymers and biopolymers with antiviral activity: potential applications for improving food safety. Compr. Rev. Food Sci. 2018;17:754–768. doi: 10.1111/1541-4337.12349. [DOI] [PubMed] [Google Scholar]
- 54.Kissmann J., Joshi S.B., Haynes J.R., Dokken L., Richardson C., Middaugh C.R. H1N1 influenza virus-like particles: physical degradation pathways and identification of stabilizers. J. Pharm. Sci. 2011;100:634–645. doi: 10.1002/jps.22304. [DOI] [PubMed] [Google Scholar]
- 55.Pereira H.G., Skehel J.J. Spontaneous and tryptic degradation of virus particles and structural components of adenoviruses. J. Gen. Virol. 1971;12:13–24. doi: 10.1099/0022-1317-12-1-13. [DOI] [PubMed] [Google Scholar]
- 56.Peddinti B.S.T., Scholle F., Vargas M.G., Smith S.D., Ghiladi R.A., Spontak R.J. Inherently self-sterilizing charged multiblock polymers that kill drug-resistant microbes in minutes. Mater. Horiz. 2019;6:2056–2062. [Google Scholar]
- 57.Hook A.L., Chang C.Y., Yang J., Luckett J., Cockayne A., Atkinson S., Mei Y., Bayston R., Irvine D.J., Langer R. Combinatorial discovery of polymers resistant to bacterial attachment. Nat. Biotechnol. 2012;30:868–878. doi: 10.1038/nbt.2316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Celiz A.D., Smith J.G.W., Langer R., Anderson D.G., Winkler D.A., Barrett D.A., Davies M.C., Young L.E., Denning C., Alexander M.R. Materials for stem cell factories of the future. Nat. Mater. 2014;13:570–579. doi: 10.1038/nmat3972. [DOI] [PubMed] [Google Scholar]
- 59.Patel A.K., Celiz A.D., Rajamohan D., Anderson D.G., Langer R., Davies M.C., Alexander M.R., Denning C. A defined synthetic substrate for serum-free culture of human stem cell derived cardiomyocytes with improved functional maturity identified using combinatorial materials microarrays. Biomaterials. 2015;61:257–265. doi: 10.1016/j.biomaterials.2015.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Blok A.J., Gurnani P., Xenopoulos A., Burroughs L., Duncan J., Urbanowicz R.A. Polymer microarrays rapidly identify competitive adsorbents of virus-like particles (VLPs) Biointerphases. 2020;15 doi: 10.1116/6.0000586. [DOI] [PubMed] [Google Scholar]
- 61.McKay P.F., Hu K., Blakney A.K., Samnuan K., Brown J.C., Penn R., Zhou J., Bouton C.R., Rogers P., Polra K. Self-amplifying RNA SARS-CoV-2 lipid nanoparticle vaccine candidate induces high neutralizing antibody titers in mice. Nat. Commun. 2020;11:3523–3530. doi: 10.1038/s41467-020-17409-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Folegatti P.M., Ewer K.J., Aley P.K., Angus B., Becker S., Belij-Rammerstorfer S., Bellamy D., Bibi S., Bittaye M., Clutterbuck E.A. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: a preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet. 2020 doi: 10.1016/S0140-6736(20)31604-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Dundas A.A., Sanni O., Dubern J.-F., Dimitrakis G., Hook A.L., Irvine D.J., Williams P., Alexander M.R. Validating a predictive structure–property relationship by discovery of novel polymers which reduce bacterial biofilm formation. Adv. Mater. 2019;31:1903513. doi: 10.1002/adma.201903513. 1-6. [DOI] [PMC free article] [PubMed] [Google Scholar]


