Abstract
Background
The provision of facemasks must be prioritised when supplies are interrupted. These include supplies to operating rooms. The aim of this review is to evaluate the available evidence to determine the relative priority for the provision of facemasks in operating rooms to prevent surgical site infection.
Methods
A systematic search of OVID Medline, Embase & Cochrane Central was completed. Candidate full-text articles were identified and analysed by two reviewers who also assessed risk of bias.
Findings
Six studies were identified that described infections with and without facemask usage. The pooled effect of not wearing facemasks was a risk ratio for infection of 0.77 (0.62–0.97) in favour of not wearing masks. Only one case-controlled study evaluated facemask usage in implant surgery and demonstrated an odds ratio for developing infection of 3.34 (95% CI 1.94–5.74) if facemasks were not worn by the operating surgeon.
Four studies collected microbiological cultures during periods in surgery with or without facemasks. Two demonstrated an increase in colony forming units in surgery where the wound was directly below the surgeon. One study showed equivocal results when masks were worn, and one was terminated early limiting interpretation.
Conclusion
The use of facemasks by scrubbed staff during implant surgery should be mandatory to prevent infection. We recommend the use of facemasks by all scrubbed staff during other forms of surgery to protect the patient and staff, but the supporting evidence is weak. There is insufficient evidence to show that non-scrubbed staff must wear masks during surgery.
Keywords: Facemask, Surgical infection, Elective surgery, Emergency surgery, Bacterial infection
Introduction
An estimated 187·2–281·2 million major surgical procedures are undertaken worldwide each year.1 Healthcare related infection is a significant cause of patient morbidity and cost around the world, with surgical site infection being the top cause of healthcare related infection in low and middle income countries and the second most frequent type of healthcare related infection in high income countries.2
Many international agencies have developed guidelines for surgical attire to minimise the risk of patients developing surgical site infections.3, 4, 5, 6, 7, 8, 9, 10 The surgical facemask is recommended to prevent the transmission of nasal and oral pathogens from the surgical team to the surgical wound and has been used in routine surgical practice for more than 100 years. However, many of these guidelines also acknowledge that the evidence supporting the use of these facemasks by the scrubbed and non-scrubbed members of the surgical team is uncertain.2 , 3 , 6
The recent coronavirus pandemic has resulted in a global shortage of surgical facemasks.11, 12, 13 This echoes the shortage in Hong Kong experienced during the 2003 SARS pandemic14 and may be a feature of future respiratory viral infection outbreaks. The FDA has recommended that in a crisis of supply of surgical facemasks, the use must be prioritised for procedures that expose patients and healthcare professionals to blood and body fluids (though these recommendations do not cover protection against surgical aerosols).15 It is unclear if this is expected to include all staff in the operating room.
Facemasks and respirators have now formed part of the routine personal protective equipment being used to protect healthcare workers from coronavirus infection from aerosol generating procedures in the operating theatre such as airway management, laparoscopy, orthopaedic procedures and other forms of surgery.
The aim of this systematic review is to evaluate the evidence surrounding the use of surgical facemasks in the operating theatre by scrubbed and non-scrubbed staff to prevent surgical site infection. This evidence may be used to prioritise the distribution of facemasks when operating on patients without coronavirus infection if the facemask supply becomes interrupted in a potential second wave or future pandemics.
Methods
A systematic review of all English language titles was performed to identify relevant comparative studies for inclusion. The protocol was prospectively registered on the PROSPERO database (PROSPERO CRD42020172714).16
OVID Medline, EMBASE and the Cochrane CENTRAL databases were searched on the 7th March 2020. Titles were screened by two independent reviewers against the inclusion criteria which were studies that compared surgical facemask usage in the operating theatre with no mask usage during surgery. All surgical disciplines were included, with no date restriction. Additional candidate studies were identified through bibliographic review of included studies, previous systematic reviews and relevant guidelines.
The primary outcome of interest was the number of patients who developed an infection following surgery. The secondary outcome was the number of colony forming units that were demonstrated through microbiological cultures or air sampling as indirect evidence of facemask effect.
A prespecified subgroup analysis of surgical type was planned. Where possible, reported surgery was categorised and analysed by the Centre of Disease Control (CDC) classification of dirty or infected, contaminated, clean-contaminated and clean surgery.9 An additional category of implant surgery was included for studies that reported surgery where permanent biomaterials were being implanted.
An additional subgroup analysis was planned for the different theatre team members who were not wearing masks (scrubbed vs non-scrubbed), recognising that theatre team members who are non-scrubbed staff may be also be a source of contamination where they are circulating near to open sterile trays.
Following title screening, full texts were analysed by two reviewers and data extracted onto a standard form. Any disagreements were resolved through discussion, and through moderation with the senior author if necessary.
Heterogeneity of study outcomes was evaluated through visual inspection of forest plots and calculation of the I2 statistic which was interpreted in line with guidance from the Cochrane collaboration.17 Where the studies were sufficiently homogeneous, meta-analysis was performed using a random effects model using Review Manager 5.1.18 Risk of bias was assessed by two reviewers using the risk of bias 2.0 (RoB 2.0) tool for randomised trials19 or the Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool for non-randomised studies.20 Publication bias was evaluated through the calculation of a funnel plot.
Results
The PRISMA study flow diagram is shown in Fig. 1 . Our initial searches of the electronic databases yielded 653 candidate studies. Following de-duplication, 451 titles and abstracts were screened to identify 124 studies. Full texts were analysed with 10 studies eligible for inclusion in the qualitative synthesis and six for quantitative analysis.
Fig. 1.
PRISMA flow diagram.
The characteristics of the included studies included with direct evidence of infection following surgery with or without facemasks are shown in Table 1 . Risk of bias in randomised and non-randomised studies is shown in Table 2 and Table 3 .
Table 1.
Characteristics of included studies. qRCT-quasi randomised controlled trial, cRCT – cluster randomised trial.
| Authors | Study design | Study participants | Comparison | Outcomes |
|---|---|---|---|---|
| Orr 198121 | Before and after | 2066 general surgical patients | 6 months with no facemasks in theatre compared to 4 years with facemask | Wound infection (“wounds with a positive culture”) |
| Chamberlain & Houang 198422 | qRCT | 41 gynaecological patients | Alternate lists by masked and unmasked staff | Wound infections requiring antibiotics |
| Webster et al 201023 | cRCT | 811 gynaecological, obstetric, general, urology or breast surgery patients | Non-scrubbed staff wearing or not wearing facemasks | Surgical site infection |
| Tunevall 199124 | qRCT | 3088 surgical patients (orthopaedics and urology excluded) | Alternate weeks of masked and unmasked surgery | Pus or cellulitis requiring surgical debridement/drainage and antibiotics |
| Singh et al* 200025 | Unclear | 921 urology patients | Cases with masks then cases without masks | Infection |
| Alwitry et al 200227 | RCT | 221 patients with cataracts for phacoemulsification | Randomised to surgeon wearing or not wearing mask | Settle plates and infective complications |
*Indicates the article was only available in abstract form.
Table 2.
Risk of bias for randomised trials identified for surgical site infection following use and non-use of surgical masks.
| Chamberlain & Houang 198422 | Webster et al. 201023 | Tunevall 199124 | Singh et al. 200025 | Alwitry et al. 200227 | Berger et al. 199330 | |
|---|---|---|---|---|---|---|
| Risk of bias arising from the randomization process | Some concerns | Low | High | Some concerns | Low | Some concerns |
| Risk of bias due to deviations from the intended interventions (effect of assignment to intervention) | Some concerns | Low | Some concerns | High | Low | Some concerns |
| Risk of bias due to deviations from the intended interventions (effect of adhering to intervention) | Low | Low | Low | High | Low | Some concerns |
| Risk of bias due to missing outcome data | Low | Some concerns | Low | High | Low | Low |
| Risk of bias in measurement of the outcome | Some concerns | Low | Some concerns | High | Low | Low |
| Risk of bias in selection of the reported result | Low | Low | Low | High | Low | Low |
| Overall risk of bias | Some concerns | Some concerns | High | High | Low | Some concerns |
Table 3.
Risk of bias for non-randomised trials identified for surgical site infection following use and non-use of surgical masks.
| Orr 198121 | Kamalarajah et al. 200726 | Wright et al. 196828 | Tunevall & Jorbeck 199231 | |
|---|---|---|---|---|
| Bias due to confounding | Serious | Moderate | Serious | Low |
| Bias in selection of participants into the study | Low | Moderate | Serious | Low |
| Bias in classification of interventions | Low | Low | Serious | Low |
| Bias due to deviations from intended interventions | Low | Low | Moderate | Low |
| Bias due to missing data | Low | Moderate | Serious | Low |
| Bias in selection of the reported result | Moderate | Moderate | Serious | Moderate |
| Overall risk of bias | Serious | Moderate | Serious | Low |
The use of facemasks in routine practice was evaluated by Orr in 1981 through a before and after study involving 2066 general surgical patients.21 Infection surveillance from 4 years of general surgical operating where facemasks were worn as standard was compared to 6 months where facemasks were not worn. A non-significant reduction in infection for patients when masks were not worn was demonstrated with a risk ratio of 0.57 (95% CI 0.27–1.19).
A quasi-randomised trial was conducted by Chamberlain and Houang in gynaecological surgery.22 This study had to be halted early after three infections in the unmasked group compared to none in the masked group leaving a small sample size that potentially biases the results in favour of the masked group.
Webster et al. conducted a RCT regarding the use of surgical masks for the non-scrubbed staff including anaesthetists.23 While the methods suggest that all the hospital's 17 operating rooms participated, no vascular or orthopaedic operations were included. Lists were randomised daily with 811 included patients. A small reduction in risk of infection was demonstrated for patients where facemasks were not worn with a risk ratio of 0.79 (95% CI 0.52–1.19) but this again did not achieve statistical significance.
Tunevall reported a quasi-randomised trial where patients were operated during masked or unmasked weeks.24 A power calculation required recruitment of 3000 patients, and the sample size was 3088. Patients undergoing orthopaedic, urology or surgery involving a synthetic graft were excluded. A small but statistically insignificant difference was shown in infection rates when masks were not worn with a risk ratio of 0.75 (95% CI 0.55–1.05).
The use of masks in 921 urological surgery was reported by Singh et al., in 2000.25 Unfortunately, study data is only available in abstract form with the full text unavailable despite attempted contact with study authors and the British Library. The precise study design is unavailable and so the study is at high risk of bias. A reduction in infections with a risk ratio of 0.88 (95% CI 0.51–1.52) was present when facemasks were not worn.
A national case control study was completed of infective endophthalmitis following phacoemulsification.26 In a multivariate regression, the odds ratio of developing endophthalmitis when a cataract procedure was performed without facemasks was 3.34 (95% CI 1.94–5.74). However, the denominator number of cases was not reported so it was not possible to pool data from this study. A randomised trial by Alwitry et al. comparing mask wearing or non-wearing during cataract surgery using phacoemulsification included 221 patients but found no infective complications in either group.27
Wright et al. reported no change the infection rates in their hospital of 30% for emergency surgery and 15% for elective surgery when gauze and cellophane surgical facemasks were abandoned.28 Cases were not broken down into surgical type, and precise rates were not reported. These results have not been included in any further analysis as the gauze facemask is not commonly used in modern practice and the study is at significant risk of bias as the baseline infection rate was not stable during this time period.
Pooled results from all surgeries
The heterogeneity between these studies for all infections in all patient groups was unlikely to be important with an I2 statistic of 2%. In a pooled analysis using the Mantel-Haenszel random effects model the overall effect of removing facemasks was a risk ratio of 0.77 (0.62–0.97) in favour of not wearing masks (Fig. 2 ).
Fig. 2.
Forest plot of included studies for the use of facemasks during all surgery.
Facemask usage during implant surgery
In a case-controlled study, Kamalarajah et al. found that the odds ratio of developing infective endophthalmitis following cataract replacement was 3.34 (95% CI 1.94–5.74) if facemasks were not worn by the operating surgeon.26 The only randomised trial where implants were implanted was that of Alwitry et al. where the use of a mask or non-wearing during cataract surgery using phacoemulsification with 221 patients but found no infective complications in either group.27
Facemask usage during clean surgery
Two studies presented results from the use of facemasks by all staff (scrubbed and un-scrubbed) during clean surgery.22 , 24 The number of patients who experienced infections in the Webster et al. study was obtained by personal communication by Vincent & Edwards and reported in a previous review relating to the use of facemasks by non-scrubbed staff only.23 , 29
In clean surgery, no difference in risk of infection was demonstrated between groups wearing and not wearing facemasks with a pooled risk ratio of 0.89 (95%CI 0.47–1.68) as shown in Fig. 3 . Moderate heterogeneity was demonstrated between the studies.
Fig. 3.
Forest plot of included studies for the use of facemasks during clean surgery.
Facemask usage during clean-contaminated, contaminated and dirty surgery
Three studies presented results from non-clean surgery, though none differentiated cases into clean-contaminated, contaminated or dirty. Chamberlain and Houang reported no infections in non-clean surgery with or without masks but with group sizes too small to be included in the pooled analysis.22
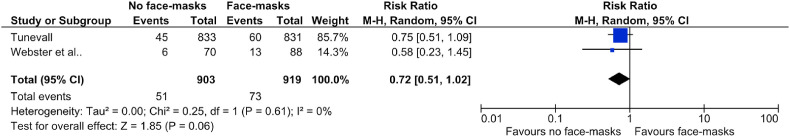
The pooled results from Tunevall and Webster et al. show a small reduction in risk of infection that is not statistically different with a risk ratio of 0.72 (95%CI 0.51–1.02) as shown in Fig. 4 .23 , 24
Fig. 4.
Forest plot of included studies for the use of facemasks during non-clean surgery.
Indirect evidence for efficacy of facemasks
Four studies were identified that provide indirect evidence for the use of facemasks during surgery. Alwitry et al. conducted a randomised trial of mask wearing or non-wearing during cataract surgery using phacoemulsification.27 221 patients were included with sequential blood agar settle plates collected from next to the patient's head. The rate of colony forming unit deposition was higher when facemasks were not used with a risk ratio for a colony forming unit rate of > 1CFUmin-1 of 4.28 (95%CI 1.68–10.48) in favour of wearing a mask.
Berger et al. conducted a study where mask position was changed and removed for 10 min periods during cardiac catherisation.30 The mask position was determined by a written, random schedule. When the masks were not worn there was a risk ratio of developing a positive blood agar culture of 1.12 (95% CI 0.95–1.3) in favour of wearing a mask. Wearing a mask reduced the mean number of colony forming units during the 10-min by 3.2 (95% CI 1.27–5.13).
As part of the clinical study, Chamberlain and Houang collected microbiological data for the use of facemasks in gynaecological surgery.22 Settle plates placed on the upper abdomen, lower abdomen and trolley for all cases with additional air sampling during 11 cases. Lower mean numbers of colony forming units was shown at all sites when masks were worn but standard deviations not presented and statistical significance not reported. Theatre air sampling showed higher counts of colony forming units when masks were worn, but again no tests for significance were performed.
Tunevall and Jorbeck followed up their comparative study with a microbiological study during thyroid surgery.31 In 14 operations no difference in numbers of colony forming units or bacterial species was identified on culture plates placed next to the wound during 30-min periods sampled while the surgeon was wearing and not wearing a facemask.
Discussion
During times of shortage of medical supplies there is a real need to be able to justify resource prioritisation based on the best available evidence. This review has identified and analysed the available comparative studies to evaluate if the provision of surgical facemasks for all surgical procedures should be a priority to prevent surgical site infections.
Overall, there is relatively little direct clinical evidence to support or refute the usage of masks in the operating room. The quality of the available studies is low, with all the studies supplying direct clinical evidence having significant risk of bias.
The efficacy of facemasks in preventing contamination of wounds has been explored experimentally through sham operations using tracer particles and bacterial cultures through settle plates. Several studies have demonstrated a reduction in contamination when masks are worn by scrubbed staff, but the clinical significance is unclear.32, 33, 34, 35 This is reinforced by two of the studies identified in this review that demonstrated higher contamination of settle plates when the wound was directly below the surgeon (in cardiac catherisation and cataract surgery).27 , 30 This confirmed the previous study that demonstrated that volunteers stood above a settle plate would generate 12–29 colony forming units while talking which reduces to 0 (across three plates) when the volunteers are stood 1 m away from the plate in a ventilated operating theatre.36
Equally, other studies exploring the impact of non-scrubbed staff wearing masks is less convincing. Ritter found no change in bacterial contamination plates placed in theatres when fibreglass facemasks were and were not worn by simulated non-scrubbed staff and Mitchell et al. conducted an simulated surgery study and found that four unmasked volunteers caused no colony forming units to develop on settle plates at the operating site if they were at least 1 m away.36 , 37 Zhiqing et al. demonstrated that the surface of the mask becomes progressively colonised with bacteria over time, concluding that masks should be replaced between cases.38
In a 2016 Cochrane review, Vincent and Edwards found no conclusive evidence that facemasks should be worn in the operating room at all to prevent surgical wound infection in clean surgery.29 However, these findings were not generalisable as the authors limited studies to those reporting clean surgery. None of the included studies included implant surgery where the development of infection is catastrophic.39, 40, 41, 42
Aside from the benefits in preventing infection, surgical facemasks also protect the staff members from contamination from the fluid splashes and debris during surgery.43, 44, 45 An additional benefit is the potential impact on surgical discipline. This is a sequence of routines that may not have an individual benefit on infection rates, but as a package contribute to improved theatre behaviours known to impact infection such as number of door openings and numbers of persons in a theatre. Several quality improvement studies have shown a benefit in infection rates when mask wearing has been included as part of such a package, but it is impossible to attribute this to the mask wearing directly.46, 47, 48 We would suggest that in times of plenty, surgical masks should be worn to prevent infection and protect staff, but it should still be possible to maintain high levels of surgical discipline if masks are not worn when supplies are limited.
The studies included in this review have focused on bacterial infections which are the predominant source of surgical site infections.49 , 50 An additional important consideration in the current pandemic is the transmission of viral pathogens where there is a reasonable chance that the patient may have an active coronavirus infection. While some of the studies reported occasions where the surgeon and the staff had a cough or other respiratory symptoms, none reported incidents where respiratory viruses were contracted by the surgical team from one another or the patient. In experimental situations the wearing of standard facemasks has been shown to reduce aerosol transmission by 97.14% (and 99.98% for N95 respirators) and reduce viral transmission.51 , 52 In a recent systematic review of observational studies, Chu et al. found a dramatic reduction in coronavirus infection when surgical facemasks were worn which improved further when N95 masks are used.53 Though there is a lack of definitive randomised trials, it seems prudent to use facemasks in the operating theatre to protect staff while the risk of coronavirus transmission remains high. When this risk of coronavirus transmission reduces, the future supplies of facemasks may be maintained by targeting the provision of facemasks for operating theatre staff based on the evidence identified in this review.
This review has some limitations. The searches were restricted to English language, though we are not aware of any randomised trials that were published in any other language. Three databases were queried, though only one additional study was identified from bibliographic search of included studies and systematic reviews, so we are confident that the relevant work has been identified and captured. There are relatively few studies published, though the number of participants in the pooled analyses is respectable for non-implant surgery. Analysis of funnel plot shows a reasonably symmetric distribution of studies. Review of clinicaltrials.gov does not yield any unpublished trials or ongoing studies that may lead to publication bias.
The pooled results in the meta-analyses suggest that the wearing of masks may increase the risk of infection. The randomised trials are all confounded by a lack of blinding, and this increase in infection may be related to changes in behaviour related to operating without masks.
Despite these limitations, the results of this review suggest that facemask wearing should be mandatory for scrubbed staff during implant surgery to prevent surgical site infection. There is insufficient evidence to recommend if facemasks should be worn by non-scrubbed staff to prevent surgical site infection. However, given the severity of surgical site infections, we suggest that non-masked staff should stand at least 1 m away from the wound or implant trays to reduce their bacterial dispersal in line with Mitchell and Hunt's experimental findings.36
For non-implant surgery there is no clear evidence that mask wearing by scrubbed or non-scrubbed staff is protective for surgical site infection. There is a suggestion that infection rates may be lower when masks are not worn, but this effect is modest, and the confidence intervals of the pooled estimates are close to the line of no effect. There may be a bias in all the studies as participants cannot be blinded, and this may impact in other behaviours such as quantity and volume of talking.
Conclusion
In the context of a global shortage of surgical masks and other healthcare equipment, evidence-based prioritisation is necessary. There is some evidence that mask wearing by scrubbed staff reduces implant related infections in ophthalmology, but it is unclear if this is generalisable to other implant surgery. Given the clinical consequences of an implant related infection, we suggest that mask wearing by scrubbed staff performing implant surgery should be mandatory to prevent surgical site infection. Equally, we recommend that non-scrubbed staff working within 1 m of surgical trays and wounds should wear a mask during implant surgery.
For non-implant surgery, the evidence does not show a reduction of surgical site infection following mask wearing during surgery. We would recommend that the scrubbed team are still encouraged to wear masks but have not found enough evidence to recommend that this is mandatory. Similarly, we have not identified enough evidence to recommend that non-scrubbed staff should or should not wear a mask during surgery to prevent surgical site infections. These conclusions are summarised in Table 4 .
Table 4.
Recommendations for the use of surgical facemask usage to prevent surgical site infection during facemask shortages for different staff members during different types of surgery.
| Type of surgery | Scrubbed staff | Non scrubbed staff (Within 1 m of wound or open trays) | Non-scrubbed staff (More than 1 m away from wound or trays) |
|---|---|---|---|
| Implant | Mandatory | Recommended | Insufficient evidence |
| Clean | Recommended | Insufficient evidence | Insufficient evidence |
| Clean-contaminated | Recommended | Insufficient evidence | Insufficient evidence |
| Contaminated | Recommended | Insufficient evidence | Insufficient evidence |
| Dirty | Recommended | Insufficient evidence | Insufficient evidence |
Funding
Ben Marson is funded by a National Institute for Health Research (NIHR), Doctoral Fellowship NIHR300240 for this research project. This report presents independent research funded by the National Institute for Health Research (NIHR). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
Declaration of competing interest
None.
References
- 1.Weiser T.G., Regenbogen S.E., Thompson K.D., Haynes A.B., Lipsitz S.R., Berry W.R. An estimation of the global volume of surgery: a modelling strategy based on available data. Lancet. 2008 Jul;372(9633):139–144. doi: 10.1016/S0140-6736(08)60878-8. [DOI] [PubMed] [Google Scholar]
- 2.World Health Organization . 2016. Global guidelines for the prevention of surgical site infection. [PubMed] [Google Scholar]
- 3.AORN . 2015. Guidelines for perioperative practice. Denver. [Google Scholar]
- 4.Association of Surgical Technologists . Hand Hygiene and Hand Washing; Littleton: 2008. AST standards of practice for surgical attire, surgical scrub. [Google Scholar]
- 5.Australian Commission for Safety and Quality in Healthcare . vol. 39. National Health Medical Research Council; 2010. p. 80. (Australian guidelines for the prevention and control of infection in healthcare). [Google Scholar]
- 6.NICE . 2013. Surgical site infection.https://www.nice.org.uk/guidance/qs49 [Internet] [cited 2020 Mar 30]. Available from: [Google Scholar]
- 7.Roy M.-C. A guide to infection control in the hospital. Infect Contr Urol Care. 2014;7(1):16. [PubMed] [Google Scholar]
- 8.NHS Infection Control Team . 2012. Standard infection control precautions literature Review : personal protective equipment ( PPE ) gloves; pp. 1–29. April 2014. [Google Scholar]
- 9.Mangram A.J., Horan T.C., Pearson M.L., Silver L.C., Jarvis W.R. Guideline for prevention of surgical site infection, 1999. Am J Infect Contr. 1999 Apr;27(2):97–134. [PubMed] [Google Scholar]
- 10.Berríos-Torres S.I., Umscheid C.A., Bratzler D.W., Leas B., Stone E.C., Kelz R.R. Centers for disease control and prevention guideline for the prevention of surgical site infection. JAMA Surg. 2017;152(8):784. doi: 10.1001/jamasurg.2017.0904. 2017 Aug 1. [DOI] [PubMed] [Google Scholar]
- 11.Andrew S., Yeung J. price-gouging and serious fear for the future. 2020. Masks can't stop the coronavirus in the US, but hysteria has led to bulk-buying.https://edition.cnn.com/2020/02/29/health/coronavirus-mask-hysteria-us-trnd/index.html [Internet]. CNN. [cited 2020 Mar 30]. Available from: [Google Scholar]
- 12.Mahase E. Novel coronavirus: Australian GPs raise concerns about shortage of face masks. BMJ. 2020 Feb 5:m477. doi: 10.1136/bmj.m477. [DOI] [PubMed] [Google Scholar]
- 13.Wu H., Huang J., Zhang C.J.P., He Z., Ming W.-K. Facemask shortage and the novel coronavirus disease (COVID-19) outbreak: reflections on public health measures. EClinicalMedicine. 2020 Apr:100329. doi: 10.1016/j.eclinm.2020.100329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hung L.S. The SARS epidemic in Hong Kong: what lessons have we learned? JRSM. 2003 Aug 1;96(8):374–378. doi: 10.1258/jrsm.96.8.374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.FDA . 2020. Surgical mask and gown conservation strategies - letter to healthcare providers.https://www.fda.gov/medical-devices/letters-health-care-providers/surgical-mask-and-gown-conservation-strategies-letter-healthcare-providers [Internet] [cited 2020 Mar 30]. Available from: [Google Scholar]
- 16.Marson B.A. 2020. Surgical mask usage by scrubbed and non-scrubbed staff to reduce surgical site infections.https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42020172714 [Internet] [cited 2020 Mar 30]. Available from: [Google Scholar]
- 17.Higgins J., Green S. 2011. Cochrane handbook for systematic reviews of Interventions. 5.1.0. Oxford: the Cochrane collaboration. [Google Scholar]
- 18.The Nordic Cochrane Centre . 2014. Review manager (RevMan). 5.3. Copenhagen: the Cochrane collaboration. [Google Scholar]
- 19.Sterne J.A.C., Savović J., Page M.J., Elbers R.G., Blencowe N.S., Boutron I. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019 Aug 28:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 20.Sterne J.A., Hernán M.A., Reeves B.C., Savović J., Berkman N.D., Viswanathan M. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016 Oct 12;355:i4919. doi: 10.1136/bmj.i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Orr N.W. Is a mask necessary in the operating theatre? Ann R Coll Surg Engl. 1981 Nov;63(6):390–392. [PMC free article] [PubMed] [Google Scholar]
- 22.Chamberlain G.V., Houang E. Trial of the use of masks in the gynaecological operating theatre. Ann R Coll Surg Engl. 1984 Nov;66(6):432–433. [PMC free article] [PubMed] [Google Scholar]
- 23.Webster J., Croger S., Lister C., Doidge M., Terry M.J., Jones I. Use of face masks by non-scrubbed operating room staff: a randomized controlled trial. ANZ J Surg. 2010 Mar;80(3):169–173. doi: 10.1111/j.1445-2197.2009.05200.x. [DOI] [PubMed] [Google Scholar]
- 24.Tunevall T.G. Postoperative wound infections and surgical face masks: a controlled study. World J Surg. 1991 May;15(3):383–387. doi: 10.1007/BF01658736. [DOI] [PubMed] [Google Scholar]
- 25.Singh B., Wani A.A., Malik A. Myth breaker about surgical face mask. JK Pract. 2000;7(2):129–130. [Google Scholar]
- 26.Kamalarajah S., Ling R., Silvestri G., Sharma N.K., Cole M.D., Cran G. Presumed infectious endophthalmitis following cataract surgery in the UK: a case-control study of risk factors. Eye. 2007;21(5):580–586. doi: 10.1038/sj.eye.6702368. 2006/07/07. [DOI] [PubMed] [Google Scholar]
- 27.Alwitry A., Jackson E., Chen H., Holden R. The use of surgical facemasks during cataract surgery: is it necessary? Br J Ophthalmol. 2002;86(9):975–977. doi: 10.1136/bjo.86.9.975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wright J.E., Henniessy E.J., Bissett R.L. Wound infection: experience with 12,000 sutured surgical wounds in a general hospital over a period of 11 years. Aust N Z J Surg. 1968;41(2):107–112. doi: 10.1111/j.1445-2197.1968.tb06270.x. [DOI] [PubMed] [Google Scholar]
- 29.Vincent M., Edwards P. Disposable surgical face masks for preventing surgical wound infection in clean surgery. Cochrane Database Syst Rev. 2016 Apr 26;2016(4) doi: 10.1002/14651858.CD002929.pub3. CD002929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Berger S.A., Kramer M., Nagar H., Finkelstein A., Frimmerman A., Miller H.I. Effect of surgical mask position on bacterial contamination of the operative field. J Hosp Infect. 1993;23(1):51–54. doi: 10.1016/0195-6701(93)90130-r. [DOI] [PubMed] [Google Scholar]
- 31.Tunevall T.G., Jorbeck H. Influence of wearing masks on the density of airborne bacteria in the vicinity of the surgical wound. Eur J Surgery, Acta Chir. 1992;158(5):263–266. [PubMed] [Google Scholar]
- 32.Schweizer R.T. Mask wiggling as a potential cause of wound contamination. Lancet. 1976;2(7995):1129–1130. doi: 10.1016/s0140-6736(76)91101-6. [DOI] [PubMed] [Google Scholar]
- 33.McLure H.A., Talboys C.A., Yentis S.M., Azadian B.S. Surgical face masks and downward dispersal of bacteria. Anaesthesia. 1998 Jul 6;53(7):624–626. doi: 10.1046/j.1365-2044.1998.435-az0528.x. [DOI] [PubMed] [Google Scholar]
- 34.Ha’eri G.B., Wiley A.M. The efficacy of standard surgical face masks: an investigation using “tracer particles”. Clin Orthop Relat Res. 1980;148:160–162. [PubMed] [Google Scholar]
- 35.Philips B.J., Fergusson S., Armstrong P., Anderson F.M., Wildsmith J.A. Surgical face masks are effective in reducing bacterial contamination caused by dispersal from the upper airway. Br J Anaesth. 1992 Oct;69(4):407–408. doi: 10.1093/bja/69.4.407. [DOI] [PubMed] [Google Scholar]
- 36.Mitchell N.J., Hunt S. Surgical face masks in modern operating rooms—a costly and unnecessary ritual? J Hosp Infect. 1991 Jul;18(3):239–242. doi: 10.1016/0195-6701(91)90148-2. [DOI] [PubMed] [Google Scholar]
- 37.Ritter M.A., Eitzen H., French M.L., Hart J.B. The operating room environment as affected by people and the surgical face mask. Clin Orthop Relat Res. 1975;(111):147–150. doi: 10.1097/00003086-197509000-00020. [DOI] [PubMed] [Google Scholar]
- 38.Zhiqing L., Yongyun C., Wenxiang C., Mengning Y., Yuanqing M., Zhenan Z. Surgical masks as source of bacterial contamination during operative procedures. J Orthop Transl. 2018;14:57–62. doi: 10.1016/j.jot.2018.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Whitehouse J.D., Friedman N.D., Kirkland K.B., Richardson W.J., Sexton D.J. The impact of surgical-site infections following orthopedic surgery at a community hospital and a university hospital: adverse quality of life, excess length of stay, and extra cost. Infect Control Hosp Epidemiol. 2002 Apr;23(4):183–189. doi: 10.1086/502033. [DOI] [PubMed] [Google Scholar]
- 40.Lerner R.K., Esterhai J.L., Polomano R.C., Cheatle M.D., Heppenstall R.B. Quality of life assessment of patients with posttraumatic fracture nonunion, chronic refractory osteomyelitis, and lower-extremity amputation. Clin Orthop Relat Res. 1993 Oct;(295):28–36. [PubMed] [Google Scholar]
- 41.Poulsen N.R., Mechlenburg I., Søballe K., Lange J. Patient-reported quality of life and hip function after 2-stage revision of chronic periprosthetic hip joint infection: a cross-sectional study. HIP Int. 2018 Jul 12;28(4):407–414. doi: 10.5301/hipint.5000584. [DOI] [PubMed] [Google Scholar]
- 42.Vanhegan I.S., Malik A.K., Jayakumar P., Ul Islam S., Haddad F.S. A financial analysis of revision hip arthroplasty: the economic burden in relation to the national tariff. Bone Joint Lett J. 2012;94-B(5):619–623. doi: 10.1302/0301-620X.94B5.27073. [DOI] [PubMed] [Google Scholar]
- 43.Cook C.R., Gaston T., Woods B., Orozco F., Ong A., Radcliff K. Operative field debris often rises to the level of the surgeon's face shield during spine surgery: are orthopedic space suits a reasonable solution? Internet J Spine Surg. 2019;13(6):501–506. doi: 10.14444/6067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wines M.P., Lamb A., Argyropoulos A.N., Caviezel A., Gannicliffe C., Tolley D. Blood splash injury: an underestimated risk in endourology. J Endourol. 2008;22(6):1183–1187. doi: 10.1089/end.2008.0052. [DOI] [PubMed] [Google Scholar]
- 45.Davies C.G., Khan M.N., Ghauri A.S.K., Ranaboldo C.J. Blood and body fluid splashes during surgery - the need for eye protection and masks. Ann R Coll Surg Engl. 2007;89(8):770–772. doi: 10.1308/003588407X209301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hardenstine H., Smith R.L., Wolfgang J., Rose L., Snedeker L. Implementation of a multidisciplinary action plan for the reduction of surgical site infection after hip replacement surgery. Am J Infect Contr. 2013;41(6 SUPPL. 1):S88–S89. [Google Scholar]
- 47.Gaillard T., Gaillard C., Martinaud C., Védy S., Pons S., Brisou P. Epidemic surgical site infections attributable to incorrect use of face masks. J Hosp Infect. 2009 Feb;71(2):192–193. doi: 10.1016/j.jhin.2008.10.004. [DOI] [PubMed] [Google Scholar]
- 48.Elbich J.D., Sydnor M.K., Jr., Komorowski D.J., Morano G., Strife B.J., Romano-Daniels A. Successful implementation of a prospectively designed QA protocol to reduce port infection rates. J Vasc Intervent Radiol. 2014;25(3 SUPPL. 1):S60. [Google Scholar]
- 49.Ollivere B.J., Ellahee N., Logan K., Miller-Jones J.C.A., Allen P.W. Asymptomatic urinary tract colonisation predisposes to superficial wound infection in elective orthopaedic surgery. Int Orthop. 2009 Jun;33(3):847–850. doi: 10.1007/s00264-008-0573-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Weigelt J.A., Lipsky B.A., Tabak Y.P., Derby K.G., Kim M., Gupta V. Surgical site infections: causative pathogens and associated outcomes. Am J Infect Contr. 2010 Mar;38(2):112–120. doi: 10.1016/j.ajic.2009.06.010. [DOI] [PubMed] [Google Scholar]
- 51.Leung N.H.L., Chu D.K.W., Shiu E.Y.C., Chan K.-H., McDevitt J.J., Hau B.J.P. Respiratory virus shedding in exhaled breath and efficacy of face masks. Nat Med. 2020 May 3;26(5):676–680. doi: 10.1038/s41591-020-0843-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ma Q.-X., Shan H., Zhang H.-L., Li G.-M., Yang R.-M., Chen J.-M. Potential utilities of mask-wearing and instant hand hygiene for fighting SARS-CoV-2. J Med Virol. 2020 Apr 8;92:1567–1571. doi: 10.1002/jmv.25805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Chu D.K., Akl E.A., Duda S., Solo K., Yaacoub S., Schünemann H.J. Physical distancing, face masks, and eye protection to prevent person-to-person transmission of SARS-CoV-2 and COVID-19: a systematic review and meta-analysis. Lancet. 2020 Jun;395(10242):1973–1987. doi: 10.1016/S0140-6736(20)31142-9. [DOI] [PMC free article] [PubMed] [Google Scholar]