Abstract
Background
Carbapenem-resistant Acinetobacter baumannii (CRAB) infection is associated with significant mortality, causing worldwide concern, yet there are limited data on contributing microbiological factors. This study aimed to identify the clinical and microbiologic risk factors for mortality in CRAB bacteremia.
Methods
Adult patients with monomicrobial CRAB bacteremia in a 2700-bed tertiary hospital between December 2012 and December 2018 were retrospectively enrolled. Risk factors for 30-day mortality were evaluated. All isolates collected on the first day of bacteremia were subjected to colistin susceptibility testing by broth microdilution and to genotyping by multilocus sequence typing.
Results
A total of 164 patients were enrolled, and 90 (55%) died within 30 days. The most common genotype among the isolates was ST191 (49%), and 12 isolates (7%) were resistant to colistin. Genotype, colistin minimum inhibitory concentration, and colistin resistance were not significantly associated with mortality, in contrast to several clinical factors. In multivariable analysis, ineradicable or not-eradicated focus (adjusted odds ratio [aOR], 4.92; 95% CI, 1.95–12.42; P = .001), septic shock (aOR, 4.72; 95% CI, 2.12–10.49; P < .001), and inappropriate antimicrobial therapy (aOR, 2.54; 95% CI, 1.05–6.16; P = .04) were independent risk factors for mortality. Among antibiotic strategies, colistin combined with tigecycline or other antibiotics were significantly associated with lower mortality after adjustment for confounding factors.
Conclusions
Clinical factors such as the nature of the infection source and source control, severity of bacteremia, and appropriateness of antibiotics, rather than microbiological factors, contribute to mortality in CRAB bacteremia. A specific antibiotic combination may help improve outcomes.
Keywords: Acinetobacter baumannii, bacteremia, carbapenems, mortality, multidrug-resistant, risk factors
Acinetobacter baumannii, an aerobic, catalase-positive, oxidase-negative, gram-negative coccobacillus, causes a variety of human infections in the health care setting [1]. Two major nosocomial manifestations are bacteremia and ventilator-associated pneumonia. Others include intravascular catheter-related infection, surgical site infection, and urinary tract infection [1]. Infections with this organism have unfavorable clinical outcomes and impose a considerable economic burden [2]. Acinetobacter species can readily have multiple resistance mechanisms. Carbapenem resistance is an emerging concern because it is associated with high mortality [3–5]. Carbapenem-resistant Acinetobacter baumannii (CRAB) was classified by the World Health Organization as one of the critical priority pathogens in the global priority list of antibiotic-resistant bacteria that was intended to guide research, discovery, and the development of new antibiotics [6]. In South Korea, the carbapenem resistance rate of A. baumannii identified in hospitals was reported to reach 90% [7, 8]. In a report from Kor-GLASS (the Korean part of the Global Antimicrobial Resistance Surveillance System), CRAB bacteremia occurred in 6.3 episodes per 10 000 patient-days in the intensive care unit and was highest among multidrug-resistant pathogens [8].
To improve the clinical outcome of CRAB bacteremia, identifying risk factors for mortality is crucial. The factors known to affect mortality are underlying medical condition, severity of bacteremia, and early appropriate antimicrobial therapy [9–13]. In a recent systematic review, inappropriate empirical antimicrobial treatment, septic shock, chronic liver disease, renal failure, neutropenia, immunosuppressant use, and a high Pitt bacteremia score were risk factors for mortality in CRAB bacteremia [10]. Although there have been studies on the effects of clinical factors on mortality, few studies have examined the impact of microbiological factors. Therefore, we aimed to investigate the effect of microbiological factors including multilocus sequence type (MLST) and colistin minimum inhibitory concentration (MIC), as well as clinical factors, on mortality.
METHODS
Patients and Study Design
All adult patients with monomicrobial CRAB bacteremia at the Asan Medical Center, a 2700-bed tertiary hospital, between December 2012 and December 2018 were retrospectively enrolled in the study. Patients were excluded if (1) they were <18 years old, (2) they had polymicrobial bacteremia, (3) they died within 24 hours of bacteremia, or (4) they had A. baumannii bacteremia within the previous 3 months. If patients had more than 1 episode of CRAB bacteremia, only the first episode was analyzed. Consecutive blood isolates that had been prospectively collected on the first day of bacteremia were used for further microbiological testing. Risk factors for 30-day mortality were evaluated through a detailed clinical and microbiological analysis of the study patients.
Definitions
CRAB was defined as A. baumannii resistant to imipenem and meropenem based on Clinical and Laboratory Standards Institute (CLSI) 2019 breakpoints [14]. CRAB bacteremia was defined as at least 1 blood culture positive for CRAB with clinical features compatible with infection. Empirical antimicrobial therapy was defined as treatment started no later than 24 hours after blood samples for the index blood culture had been drawn. Definitive therapy was defined as treatment that was continued or commenced on the day the antibiogram results were reported to the clinicians. Antimicrobial therapy was considered appropriate if at least 1 antimicrobial agent with in vitro activity against the causative pathogen was administered. Inappropriate antimicrobial therapy was defined as inappropriate empirical therapy in patients who died before susceptibility results were available and inappropriate definitive therapy in the other patients. Combination therapy was defined as the use of 1 in vitro active antimicrobial agent combined with ≥1 antimicrobial with gram-negative activity, regardless of in vitro susceptibility. Corticosteroid therapy was the use of systemic prednisolone ≥20 mg equivalent over 2 weeks. A diagnosis of septic shock was made when systolic blood pressure was <90 mmHg and there was clinical evidence of peripheral hypoperfusion and the need for vasopressors [15]. Bacteremia was considered high grade if ≥2 blood culture sets from ≥2 blood culture sets drawn at the initial CRAB episode were positive. Charlson’s comorbidity index was used to score the severity of underlying disease, and the severity of illness at the time of bacteremia was assessed by the Pitt bacteremia score [16, 17]. The time interval between bacteremia and the removal of an eradicable focus was limited to 7 days to reduce potential survival bias.
Identification and Susceptibility Testing and Treatment Regimens
All blood culture samples were processed by the hospital microbiology laboratory using a standard blood culture system (BACTEC FX; Becton Dickinson, NJ, USA). A. baumannii identification and antimicrobial susceptibility tests were performed using a MicroScan WalkAway 96 Plus system and Neg Combo Panel Type 72 (Beckman Coulter, Brea, CA, USA). Colistin susceptibility was tested in triplicate using the broth microdilution method according to CLSI guidelines [18]. The results were interpreted according to the CLSI susceptibility criteria [14]. Isolates with tigecycline MIC ≤2 mg/L were considered to be susceptible to tigecycline. The dose of colistin was a 5-mg/kg or 300-mg colistin base activity loading, followed by a 150-mg colistin base activity every 12 hours in patients with normal renal function [19]. The dose of tigecycline was a 100-mg loading, followed by 50 mg every 12 hours.
Genotyping by MLST
MLST was performed on 7 housekeeping genes (gltA, gyrB, gdhB, recA, cpn60, gpi, and rpoD) as described previously [20]. Isolates were assigned to sequence types (STs) using tools available on the A. baumannii MLST database (http://pubmlst.org/abaumannii/).
Statistical Analysis
Categorical variables were compared using the χ 2 test or Fisher exact test, as appropriate, and continuous variables were compared using the Student t test or Mann-Whitney U test, as appropriate. All tests of significance were 2-tailed, and P values <.05 were considered statistically significant. Risk factors for 30-day mortality of patients with CRAB bacteremia were analyzed by logistic regression analysis and Cox proportional hazards regression analysis. Variables that were significant in the univariate analysis and other variables of clinical importance were included in a multiple logistic regression model and multivariate Cox proportional hazards model. A linear-by-linear association test was used to analyze trends among multiple groups. All statistical analyses were performed using the SPSS for Windows software package, version 24 (SPSS Inc., Chicago, IL, USA).
Patient Consent Statement
This observational study was approved by the Institutional Review Board of the Asan Medical Center. To protect personal privacy, identifying information in the electronic database was encrypted. Informed consent was waived by the ethics committee because no intervention was involved and no patient-identifying information was included.
RESULTS
Patient Characteristics
A total of 298 patients were diagnosed with CRAB bacteremia during the study period. Patients under 18 years (n = 27) or who died within 24 hours of bacteremia (n = 12), as well as patients with polymicrobial bacteremia (n = 90), with A. baumannii bacteremia within the previous 3 months (n = 2), and for whom no corresponding blood isolate was collected (n = 3), were excluded. A total of 164 patients with CRAB bacteremia were included in the final analysis. The mean age of these patients (SD) was 63.7 (14.7) years, and 67% were male. Hematologic malignancy and solid tumor were frequent underlying diseases. Regarding treatment, 115 of the 164 patients (70%) received appropriate antimicrobial therapy. Thirty-day mortality was 55% (90/164), and there were no significant differences in age, gender, or Charlson comorbidity index between the surviving and deceased patients (Table 1). However, the deceased patients were significantly different in terms of underlying characteristics, site of infection, severity of infection, and management of bacteremia. Hematologic malignancy, corticosteroid use, neutropenia, pneumonia, primary bacteremia, ineradicable or not-eradicated focus, high-grade bacteremia, septic shock, higher Pitt bacteremia score, and inappropriate definitive therapy were significantly associated with 30-day mortality. Combination antimicrobial therapy was associated with a mortality benefit.
Table 1.
Clinical Characteristics and Management of CRAB Bacteremia, According to 30-Day Mortality
| Characteristic or Treatment | Deceased Patients (n = 90) | Surviving Patients (n = 74) | Total (n = 164) | P Value |
|---|---|---|---|---|
| Age, mean ± SD, y | 62.1 ± 16.0 | 65.6 ± 12.7 | 63.7 ± 14.7 | .12 |
| Male gender | 58 (64) | 53 (72) | 111 (67) | .33 |
| Site of acquisition | .01 | |||
| Community-onset health care–associated infection | 0 (0) | 6 (8) | 6 (4) | |
| Hospital-acquired infection | 90 (100) | 68 (92) | 158 (96) | |
| Underlying disease/condition | ||||
| Hematologic malignancy | 33 (37) | 13 (18) | 46 (28) | .01 |
| Solid tumor | 18 (20) | 26 (35) | 44 (27) | .03 |
| Chronic kidney disease | 10 (11) | 14 (19) | 24 (15) | .16 |
| Chronic obstructive pulmonary disease | 8 (9) | 4 (5) | 12 (7) | .39 |
| Recent chemotherapy | 30 (33) | 15 (20) | 45 (27) | .06 |
| Recent surgery | 20 (22) | 18 (24) | 38 (23) | .75 |
| Immunosuppressant use | 13 (14) | 15 (20) | 28 (17) | .46 |
| Corticosteroid use | 31 (34) | 15 (20) | 46 (28) | .04 |
| Neutropenia | 32 (36) | 7 (10) | 39 (24) | <.001 |
| Ventilator care | 46 (51) | 36 (49) | 82 (50) | .75 |
| Charlson comorbidity, median (IQR) | 3 (2–5) | 4 (2–7) | 3 (2–6) | .09 |
| Type of infection | ||||
| Catheter-related infection | 7 (8) | 16 (22) | 23 (14) | .01 |
| Intra-abdominal infection | 10 (11) | 8 (11) | 18 (11) | .95 |
| Biliary tract infection | 2 (2) | 19 (26) | 21 (13) | <.001 |
| Pneumonia | 49 (54) | 20 (27) | 69 (42) | <.001 |
| Skin & soft tissue infection | 4 (4) | 3 (4) | 7 (4) | .99 |
| Primary bacteremia | 18 (20) | 6 (8) | 24 (15) | .03 |
| Eradicable focus | 14 (16) | 41 (55) | 55 (34) | <.001 |
| Removal of eradicable focus within 7 d | 9/14 (64) | 38/41 (93) | 47/55 (85) | .02 |
| Days from diagnosis to focus removal, median (IQR) | 1 (0–2) | 2 (0–4) | 1 (0–4) | .33 |
| Ineradicable or not-eradicated focus | 81 (90) | 36 (49) | 117 (71) | <.001 |
| Intensive care unit | 70 (78) | 38 (51) | 108 (66) | <.001 |
| Septic shock | 67 (74) | 20 (27) | 87 (47) | <.001 |
| Pitt bacteremia score, median (IQR) | 4 (2–8) | 2 (0–3) | 3 (0–5) | <.001 |
| High-grade CRAB bacteremiaa | 65 (72) | 38 (51) | 103 (63) | .01 |
| Inappropriate empirical therapy | 61 (68) | 47 (64) | 108 (66) | .57 |
| Inappropriate antimicrobial therapyb | 36 (40) | 13 (18) | 49 (30) | .002 |
| Combination antimicrobial therapy | 43 (48) | 47 (64) | 90 (55) | .04 |
| Anti-CRAB strategiesc | 47 | 61 | 108 | |
| Tigecycline monotherapy | 3 (6) | 3 (5) | 6 (6) | >.99 |
| Tigecycline combinationd | 6 (13) | 5 (8) | 11 (10) | .53 |
| Tigecycline with colistin | 10 (21) | 20 (33) | 30 (28) | .19 |
| Colistin combinatione | 20 (43) | 20 (33) | 40 (37) | .30 |
| Colistin monotherapy | 7 (15) | 10 (16) | 17 (16) | .83 |
| Sulbactam-based therapyf | 1 (2) | 2 (3) | 3 (3) | >.99 |
Data are presented as number of patients (with the corresponding percentage in parentheses) unless otherwise specified.
Abbreviations: CRAB, carbapenem-resistant Acinetobacter baumannii; IQR, interquartile range.
aHigh-grade CRAB bacteremia was defined when 2 or more blood culture sets were positive from at least 2 blood culture sets drawn.
bDefined as not using at least 1 antibiotic susceptible to the causative isolate.
cAmong patients who received appropriate definitive therapy.
dTigecycline combination was defined as tigecycline combination therapy without colistin.
eColistin combination was defined as colistin combination therapy without tigecycline.
fSulbactam-based therapy was sulbactam monotherapy or sulbactam combination without tigecycline and colistin.
Microbiological Characteristics
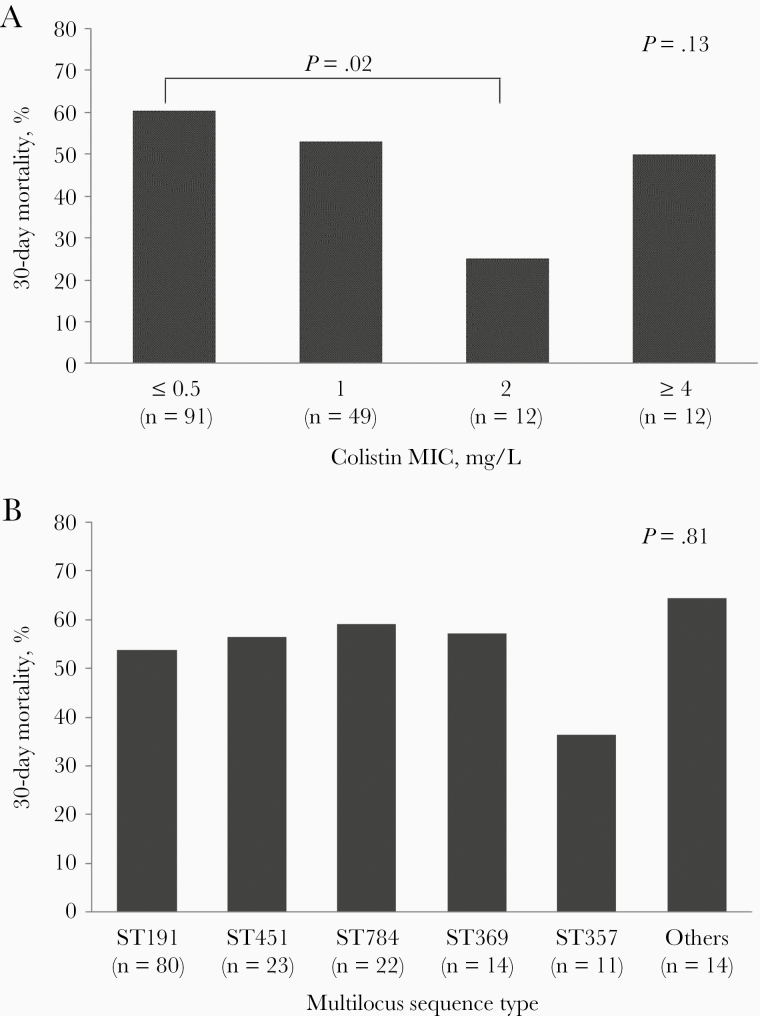
Of the 164 CRAB isolates, 152 (93%) were susceptible to colistin (MIC ≤2.0 mg/L) (Table 2). Six of the 12 patients with colistin-resistant isolates died. In the 152 patients with colistin-susceptible CRAB isolates, 30-day mortality decreased as the colistin MIC increased (60% vs 53% vs 25%; P = .02) (Figure 1). Similar trends were observed in the 93 patients treated with colistin (52% vs 48% vs 13%; P = .06) (Supplementary Figure 1). Susceptibility profiles to other antibiotics are provided in Supplementary Table 1. A total of 9 MLSTs were identified. ST191 was the dominant genotype, with 80 isolates (49%). Thirty-day mortality rates did not differ significantly between the major ST groups (P = .81) (Figure 1). However, there were significant differences in rates of septic shock between them (Supplementary Figure 2).
Table 2.
Microbiological Characteristics of CRAB bacteremia, According to 30-Day Mortality
| Charactristic of Blood Isolates | Deceased Patients (n = 90) | Surviving Patients (n = 74) | Total (n = 164) | P Value |
|---|---|---|---|---|
| Colistin susceptibility | ||||
| MIC ≤0.5 mg/L | 55 (61) | 36 (49) | 91 (56) | .11 |
| MIC 1.0 mg/L | 26 (29) | 23 (31) | 49 (30) | .76 |
| MIC 2.0 mg/L | 3 (3) | 9 (12) | 12 (7) | .03 |
| MIC ≥4.0 mg/L | 6 (7) | 6 (8) | 12 (7) | .72 |
| Multilocus sequence type | ||||
| ST191 | 43 (48) | 37 (50) | 80 (49) | .78 |
| ST451 | 13 (14) | 10 (14) | 23 (14) | .86 |
| ST784 | 13 (14) | 9 (12) | 22 (13) | .67 |
| ST369 | 8 (9) | 6 (8) | 14 (9) | .86 |
| Others | 13 (14) | 12 (16) | 25 (15) | .70 |
Data are presented as number of patients (with corresponding percentage in parentheses).
Abbreviations: CRAB, carbapenem-resistant Acinetobacter baumannii; MIC, minimum inhibitory concentration.
Figure 1.
Thirty-day mortality according to (A) colistin minimum inhibitory concentration and (B) genotype of carbapenem-resistant Acinetobacter baumannii blood isolates. Abbreviation: MIC, minimum inhibitory concentration.
Risk Factors for 30-Day Mortality
Significant variables in the univariate analysis and other variables of clinical importance were included in a logistic regression model to identify independent risk factors for mortality. Because there were significant correlations between the variables that reflected the severity of bacteremia, we retained only septic shock. For the same reason, ineradicable or not-eradicated focus was retained, and type of infection was excluded. Because combination therapy was inversely correlated with inappropriate definitive treatment and its benefit on mortality disappeared when patients with inappropriate definitive treatment were excluded, only the latter was retained. Finally, age, hematologic malignancy, corticosteroid use, ineradicable or not-eradicated focus, septic shock, high-grade bacteremia, and inappropriate definitive therapy were included in the logistic regression. Multivariate analysis indicated that ineradicable or not-eradicated focus (adjusted odds ratio [aOR], 4.92; 95% CI, 1.95 –12.42; P = .001), septic shock (aOR, 4.72; 95% CI, 2.12–10.49; P < .001), and inappropriate antimicrobial therapy (aOR, 2.54; 95% CI, 1.05–6.16; P = .04) were independent risk factors for mortality (Table 3). Cox proportional hazards regression analysis also showed similar results indicating that ineradicable or not-eradicated focus (adjusted hazard ratio [aHR], 2.50; 95% CI, 1.18–4.85; P = .02), septic shock (aHR, 2.67; 95% CI, 1.52–4.72; P = .001), and inappropriate antimicrobial therapy (aHR, 3.75; 95% CI, 2.89–6.17; P < .001) were independent risk factors for mortality.
Table 3.
Univariate and Multivariate Analysis of Risk Factors for Mortality in Patients With CRAB Bacteremia
| Risk Factor | Univariate Analysis Result | Multivariate Analysis Result | ||
|---|---|---|---|---|
| OR (95% CI) | P Value | Adjusted OR (95% CI) | P Value | |
| Age | 0.98 (0.96–1.01) | .13 | ||
| Hematologic malignancy | 2.72 (1.30–5.67) | .01 | ||
| Steroid use | 2.07 (1.01–4.22) | .05 | ||
| Ineradicable or not-eradicated focus | 9.50 (4.16–21.70) | <.001 | 4.92 (1.95–12.42) | .001 |
| Septic shock | 7.87 (3.91–15.81) | <.001 | 4.72 (2.12–10.49) | <.001 |
| High-grade CRAB bacteremia | 1.57 (1.14–2.17) | .01 | ||
| Inappropriate antimicrobial therapy | 3.13 (1.50–6.51) | .002 | 2.54 (1.05–6.16) | .04 |
The model fitted the data well in terms of discrimination (C-statistic, 0.84) and calibration (Hosmer-Lemeshow goodness of fit statistic, 9.62; P = .29).
Abbreviations: CRAB, carbapenem-resistant Acinetobacter baumannii; OR, odds ratio.
Anti-CRAB Strategies
We classified all anti-CRAB treatments as appropriate definitive therapy into 6 strategies. Of the groups with >10 patients, tigecycline combination without colistin had the highest 30-day mortality (6/11, 55%), followed by colistin combination without tigecycline (20/40, 50%) and colistin monotherapy (7/17, 41%). Tigecycline with colistin had the lowest 30-day mortality (10/30, 33%) (Table 1). Of 30 CRAB isolates in patients treated with tigecycline with colistin, 27 (90%) were susceptible to both antimicrobial agents, 2 (6%) were colistin-resistant but susceptible to tigecycline, and 1 was colistin-susceptible and tigecycline-resistant. Different anti-CRAB strategies among patients who received appropriate definitive therapy were compared. When compared with monotherapy (using only 1 antibiotic except colistin), tigecycline and colistin combination (aHR, 0.26; 95% CI, 0.08–0.92; P = .04) and colistin combination therapy without tigecycline (aHR, 0.28; 95% CI, 0.09–0.89; P = .03) were significantly associated lower 30-day mortality after adjustment for confounding factors (Table 4). In addition, compared with patients receiving 1 in vitro active agent against CRAB, patients receiving 2 or more in vitro active agents tended to have lower mortality (aHR, 0.40; 95% CI, 0.16–1.04; P = .06).
Table 4.
Univariate and Multivariate Analysis of Associations Between Different Definitive Antibiotic Strategies and 30-Day Mortality in Patients With CRAB Bacteremia (n = 108)
| Risk Factor | Univariate Analysis | Multivariate Analysisa | ||
|---|---|---|---|---|
| HR (95% CI) | P Value | Adjusted HR (95% CI) | P Value | |
| Monotherapy other than colistinb (ref) | – | – | – | – |
| Tigecycline combinationc | 0.30 (0.09–1.05) | .06 | 0.28 (0.08–1.04) | .06 |
| Tigecycline with colistin | 0.22 (0.07–0.72) | .01 | 0.26 (0.08–0.92) | .04 |
| Colistin combinationd | 0.25 (0.09–0.76) | .01 | 0.28 (0.09–0.89) | .03 |
| Colistin monotherapy | 0.25 (0.07–0.95) | .04 | 0.28 (0.06–1.36) | .12 |
Abbreviations: CRAB, carbapenem-resistant Acinetobacter baumannii; HR, hazard ratio.
aAdjusted HRs and 95% CIs were calculated by Cox proportional hazards regression among patients receiving appropriate definitive therapy, adjusted for age, hematologic malignancy, ineradicable or not-eradicated focus, septic shock, and CRAB isolation from more than 2 bottles.
bMonotherapy was defined as using only 1 antibiotic except colistin.
cTigecycline combination was defined as tigecycline combination therapy without colistin.
dColistin combination was defined as colistin combination therapy without tigecycline.
DISCUSSION
In this retrospective study, microbiological factors such as colistin MIC and genotype in addition to clinical factors were investigated to evaluate the risk factors for mortality in CRAB bacteremia. Ineradicable or not-eradicated infection focus, severity of bacteremia, and appropriate antibiotic therapy, rather than microbiological factors, were independently associated with mortality. Colistin-based combination therapy may have improved outcomes.
Previous studies reported that underlying medical conditions, severity of bacteremia, and use of early appropriate antimicrobial agents were predictors of mortality in patients with CRAB bacteremia [9–13]. Surgery before bacteremia, chronic liver disease, chronic renal disease, neutropenia, and use of corticosteroid or immune suppressants are medical conditions that also had adverse effects on clinical outcome [9, 10]. Our study yielded similar results. In addition, inappropriate definitive therapy was an independent risk factor for mortality. Of importance, a relatively large proportion (29%) of our study patients did not receive appropriate antibiotic treatment, and in those patients, 30-day mortality was very high (75%). Therefore, antimicrobial stewardship for patients with CRAB bacteremia should be reinforced. Contrary to a meta-analysis emphasizing the importance of empirical therapy [10], the appropriateness of empirical antibiotics was not associated with mortality in our study. A possible explanation is that the effect of inappropriate empirical therapy is short lived (ie, until susceptibility results are available) and can be corrected by subsequent appropriate definitive therapy. Ineradicable or not-eradicated focus was also an important predictor of mortality. Infection source control is a variable associated with the site of infection, and pneumonia and undrainable complicated intraabdominal collections are situations in which the site of infection cannot be eliminated. A previous study also reported that the mortality rate was higher in cases where the respiratory tract was the source of bacteremia [11]. In addition, we found that the mortality rate increased significantly when an eradicable focus was for any reason not removed. To reduce potential survival bias (patients who survive longer have a greater chance of receiving source control), we limited the time interval between bacteremia and the removal of an eradicable focus to 7 days in our analysis. Therefore, in patients whose infection focus is ineradicable or challenging to eradicate, more aggressive treatment should be considered.
The optimal antibiotic choice for CRAB bacteremia is controversial. Colistin, tigecycline, and sulbactam, as monotherapy or combination therapy, are the most commonly used options. Colistin is generally considered the backbone of treatment [21]. Some studies have suggested the use of tigecycline combination therapy [22–24]. On the other hand, other studies have not supported the effectiveness of combination therapy [21, 25]. In our study, after adjusting for potential confounders, the combination of colistin with tigecycline or other antibiotics, such as carbapenem or sulbactam increased survivial. However, given the high overall mortality of CRAB bacteremia (55%), colistin combination therapy was not strikingly effective (mortality rate, 43%). This suggests that colistin-based therapy may still be suboptimal. In view of the retrospective design of the study, it cannot be concluded that colistin combination therapy is the best treatment option. A future well-designed study is warranted.
In the present study, colistin MIC was not associated with a significant difference in mortality. However, mortality rates declined with increasing colistin MIC within the susceptible range (≤2 mg/L), and a similar trend was evident within the colistin-treated group. However, the number of isolates with high colistin MIC was too small to conclude that high colistin MIC contributes to lower mortality. On the other hand, in a previous study of CRAB pneumonia, mortality was significantly lower when colistin MIC was below 1 mg/L [26]. However, that study differed from ours in 2 ways. First, it included cases of CRAB pneumonia that were nonbacteremic. Second, colistin MIC was determined using an automated system that may have yielded results that differed from the reference method. For these reasons, our study may better reflect the relationship between clinical outcome of CRAB bacteremia and colistin MIC. We infer that the tendency of mortality to decrease with increasing colistin MIC may be due to weakened virulence as CRAB gains colistin tolerance [27].
The distribution of sequence types (STs) varies regionally [28–32]. ST191 is known as the dominant ST in South Korea [30, 31]. In our case, ST191 was also the most common type and accounted for about half of the total. Certain clonal types were found to be associated with poor outcome in previous studies [9, 28]. Mortality did not differ between STs in our study, but rates of septic shock did differ significantly; thus, genotype may influence the early clinical manifestations of bacteremia. However, further research is needed to elucidate the effect of the genotype of CRAB on clinical outcomes.
This study has several limitations. First, it was a single-center study, and clinical data were retrospectively collected. Unrecognized clinical factors may have resulted in biases. However, a thorough chart review was done, and we included a relatively large number of patients to compensate for the limited study design. Second, the small number of patients who received tigecycline monotherapy and sulbactam-based therapy made it difficult to evaluate the effectiveness of those treatments. Third, because of the confusing nature of kidney injury due to decreased organ perfusion during bacteremia and the adverse effect of colistin, we could not evaluate nephrotoxicity during colistin use. However, despite such limitations, our data have important clinical implications because we analyzed the effect of microbiological factors such as colistin MIC and STs on mortality in CRAB bacteremia, which has rarely been done before.
In conclusion, ineradicable or not-eradicated focus, septic shock, and inappropriate definitive therapy are independent risk factors for mortality in CRAB bacteremia. There was no significant mortality difference according to microbiological factors. In the treatment of CRAB bacteremia, it is important to eliminate eradicable foci such as catheters and intraabdominal complicated fluid collections and to administer timely appropriate antibiotics. Colistin combination therapy may contribute to a better treatment outcome.
Supplementary Data
Supplementary materials are available at Open Forum Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author.
Acknowledgments
Financial support. This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI17C2052).
Potential conflicts of interest. All authors: no reported conflicts of interest. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
Author contributions. H.J.S., E.B.C., and Y.P.C. conceived the ideas; H.J.S., E.B.C., and S.C.L. collected and analyzed the data; H.J.S. and Y.P.C. wrote the manuscript and created tables; H.J.S., E.B.C., Y.P.C., M.B., H.S.S., M.N.K., J.J., M.J.K., S.H.K., S.O.L., S.H.C., Y.S.K., and J.H.W. reviewed, revised, and approved the manuscript.
References
- 1. Hochman S, Phillips M. Acinetobacter species. In: Bennett JE, Dolin R, Blaser MJ, eds. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 9th ed. Philadelphia: Elsevier; 2020:2718–24. [Google Scholar]
- 2. Nelson RE, Schweizer ML, Perencevich EN, et al. Costs and mortality associated with multidrug-resistant healthcare-associated Acinetobacter infections. Infect Control Hosp Epidemiol 2016; 37:1212–8. [DOI] [PubMed] [Google Scholar]
- 3. Lemos EV, de la Hoz FP, Einarson TR, et al. Carbapenem resistance and mortality in patients with Acinetobacter baumannii infection: systematic review and meta-analysis. Clin Microbiol Infect 2014; 20:416–23. [DOI] [PubMed] [Google Scholar]
- 4. Sheng WH, Liao CH, Lauderdale TL, et al. A multicenter study of risk factors and outcome of hospitalized patients with infections due to carbapenem-resistant Acinetobacter baumannii. Int J Infect Dis 2010; 14:e764–9. [DOI] [PubMed] [Google Scholar]
- 5. Park SY, Choo JW, Kwon SH, et al. Risk factors for mortality in patients with Acinetobacter baumannii bacteremia. Infect Chemother 2013; 45:325–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.World Health Organization. Global priority list of antibiotic-resistant bacteria to guide research, discovery, and development of new antibiotics 2017. Available at: https://www.who.int/medicines/publications/global-priority-list-antibiotic-resistant-bacteria/en/. Accessed 28 February 2020.
- 7. Kim D, Ahn JY, Lee CH, et al. Increasing resistance to extended-spectrum cephalosporins, fluoroquinolone, and carbapenem in gram-negative bacilli and the emergence of carbapenem non-susceptibility in Klebsiella pneumoniae: analysis of Korean Antimicrobial Resistance Monitoring System (KARMS) data from 2013 to 2015. Ann Lab Med 2017; 37:231–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Lee H, Yoon EJ, Kim D, et al. Antimicrobial resistance of major clinical pathogens in South Korea, May 2016 to April 2017: first one-year report from Kor-GLASS. Euro Surveill 2018; 23:1800047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Nutman A, Glick R, Temkin E, et al. A case-control study to identify predictors of 14-day mortality following carbapenem-resistant Acinetobacter baumannii bacteraemia. Clin Microbiol Infect 2014; 20:O1028–34. [DOI] [PubMed] [Google Scholar]
- 10. Du X, Xu X, Yao J, et al. Predictors of mortality in patients infected with carbapenem-resistant Acinetobacter baumannii: a systematic review and meta-analysis. Am J Infect Control 2019; 47:1140–5. [DOI] [PubMed] [Google Scholar]
- 11. Liu CP, Shih SC, Wang NY, et al. Risk factors of mortality in patients with carbapenem-resistant Acinetobacter baumannii bacteremia. J Microbiol Immunol Infect 2016; 49:934–40. [DOI] [PubMed] [Google Scholar]
- 12. Kim T, Park KH, Yu SN, et al. Early intravenous colistin therapy as a favorable prognostic factor for 28-day mortality in patients with CRAB bacteremia: a multicenter propensity score-matching analysis. J Korean Med Sci 2019; 34:e256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Park SY, Lee EJ, Kim T, et al. Early administration of appropriate antimicrobial agents to improve the outcome of carbapenem-resistant Acinetobacter baumannii complex bacteraemic pneumonia. Int J Antimicrob Agents 2018; 51:407–12. [DOI] [PubMed] [Google Scholar]
- 14.Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing: 29th Informational Supplement M100-S25. Wayne, PA: Clinical and Laboratory Standards Institute; 2015. [Google Scholar]
- 15. Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 2016; 315:801–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987; 40:373–83. [DOI] [PubMed] [Google Scholar]
- 17. Chow JW, Fine MJ, Shlaes DM, et al. Enterobacter bacteremia: clinical features and emergence of antibiotic resistance during therapy. Ann Intern Med 1991; 115:585–90. [DOI] [PubMed] [Google Scholar]
- 18. Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard-Tenth Edition, CLSI Document M07-A10. Wayne, PA: CLSI; 2015. [Google Scholar]
- 19. Garonzik SM, Li J, Thamlikitkul V, et al. Population pharmacokinetics of colistin methanesulfonate and formed colistin in critically ill patients from a multicenter study provide dosing suggestions for various categories of patients. Antimicrob Agents Chemother 2011; 55:3284–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Bartual SG, Seifert H, Hippler C, et al. Development of a multilocus sequence typing scheme for characterization of clinical isolates of Acinetobacter baumannii. J Clin Microbiol 2005; 43:4382–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Giannella M, Bartoletti M, Gatti M, Viale P. Advances in the therapy of bacterial bloodstream infections. Clin Microbiol Infect 2020; 26:158–67. [DOI] [PubMed] [Google Scholar]
- 22. Kim NH, Hwang JH, Song KH, et al. Tigecycline in carbapenem-resistant Acinetobacter baumannii bacteraemia: susceptibility and clinical outcome. Scand J Infect Dis 2013; 45:315–9. [DOI] [PubMed] [Google Scholar]
- 23. Piperaki ET, Tzouvelekis LS, Miriagou V, Daikos GL. Carbapenem-resistant Acinetobacter baumannii: in pursuit of an effective treatment. Clin Microbiol Infect 2019; 25:951–7. [DOI] [PubMed] [Google Scholar]
- 24. Chusri S, Singkhamanan K, Wanitsuwan W, et al. Adjunctive therapy of intravenous colistin to intravenous tigecycline for adult patients with non-bacteremic post-surgical intra-abdominal infection due to carbapenem-resistant Acinetobacter baumannii. J Infect Chemother 2019; 25:681–6. [DOI] [PubMed] [Google Scholar]
- 25. Amat T, Gutiérrez-Pizarraya A, Machuca I, et al. The combined use of tigecycline with high-dose colistin might not be associated with higher survival in critically ill patients with bacteraemia due to carbapenem-resistant Acinetobacter baumannii. Clin Microbiol Infect 2018; 24:630–4. [DOI] [PubMed] [Google Scholar]
- 26. Choi IS, Lee YJ, Wi YM, et al. Predictors of mortality in patients with extensively drug-resistant Acinetobacter baumannii pneumonia receiving colistin therapy. Int J Antimicrob Agents 2016; 48:175–80. [DOI] [PubMed] [Google Scholar]
- 27. Beceiro A, Tomás M, Bou G. Antimicrobial resistance and virulence: a successful or deleterious association in the bacterial world? Clin Microbiol Rev 2013; 26:185–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Liu CP, Lu HP, Luor T. Clonal relationship and the association of the ST218 strain harboring blaOXA-72 gene to mortality in carbapenem-resistant Acinetobacter baumannii bacteremia. J Microbiol Immunol Infect 2019; 52:297–303. [DOI] [PubMed] [Google Scholar]
- 29. Tavares LCB, de Vasconcellos FM, de Sousa WV, et al. Emergence and persistence of high-risk clones among MDR and XDR A. baumannii at a Brazilian teaching hospital. Front Microbiol 2018; 9:2898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Gurung M, Rho JS, Lee YC, et al. Emergence and spread of carbapenem-resistant Acinetobacter baumannii sequence type 191 in a Korean hospital. Infect Genet Evol 2013; 19:219–22. [DOI] [PubMed] [Google Scholar]
- 31. Selasi GN, Nicholas A, Jeon H, et al. Genetic basis of antimicrobial resistance and clonal dynamics of carbapenem-resistant Acinetobacter baumannii sequence type 191 in a Korean hospital. Infect Genet Evol 2015; 36:1–7. [DOI] [PubMed] [Google Scholar]
- 32. Huang G, Yin S, Gong Y, et al. Multilocus sequence typing analysis of carbapenem-resistant Acinetobacter baumannii in a Chinese Burns Institute. Front Microbiol 2016; 7:1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.