Here we report the case of a patient who presented a severe acute dysimmune peripheral neuropathy in the context of SARS-Cov-2 infection. A 76-year-old man was admitted to our hospital in March 2020 for dyspnea, with oxygen saturation on room air of 91%. He had a history of ischemic cardiomyopathy (2016) and abdominal aortic aneurysm (2015), and was being treated for dyslipidemia and hypertension. He complained of asthenia for 10 days prior to admission and fever for 7 days. He also suffered from a mild cough. A few hours after admission, the patient reported distal paresthesia of all four limbs. One day after admission, he had a flaccid quadriplegia with acute respiratory failure, requiring mechanical ventilation. Clinical examination revealed complete facial diplegia, ophthalmoplegia, quadriplegia and lack of response to sensory stimulation in all four limbs. There was no evidence of dysautonomia. Deep tendon reflexes were all abolished. Chest computed tomography found minimal ground-glass lesions and bilateral basal lung consolidative opacities suggestive of SARS-Cov-2 infection. The nasal and tracheal SARS-Cov2 RT-qPCR were both positive (RdRp IP2 and IP4). Nerve conduction studies performed 7 days after onset of neurological symptoms revealed abolition of all sensory and motor responses in four limbs, as well as blink-reflex and facial muscle responses. Needle electrode examination (tibialis anterior and deltoid) showed no abnormal spontaneous activity (see details and evolution in Supplemental Table 1). Short latency auditory evoked potentials were depressed with impairment at the peripheral level and mid-latency auditory evoked potentials were present. Mismatch negativity occurred after an oddball sequence. Electroencephalography showed normal background activity and was reactive. Brain magnetic resonance imaging (MRI) did not show any brain lesion but demonstrated contrast enhancement of oculomotor, facial and vestibulocochlear cranial nerves.
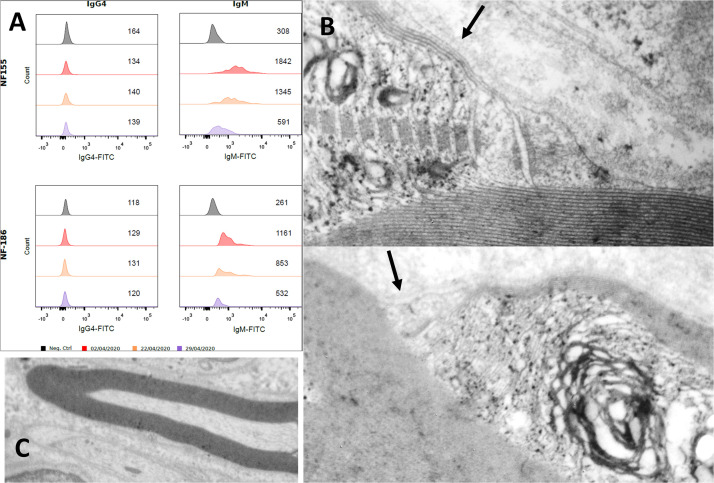
Cerebrospinal fluid analysis (CSF) revealed increased protein levels progressively increasing over time (0.37, 0.56 and 1.1 g/L respectively at days 1, 3 and 9) with no cellular reaction. RT-qPCR was negative in CSF. No antiganglioside antibodies were detectable, but the patient tested positive for autoantibodies to neurofascin-155 IgM (NF155) and neurofascin-186 IgM (NF186). We did not detect any IgG antibodies nor observe further seroconversion of IgG (Fig. 1 A).
Figure 1.
(A) Detection and monitoring of anti-NF-155 and NF-186 antibodies in flow cytometry using transfected cells according to the method described previously (1). Mean fluorescence intensity is reported in the figure (number per row) and the presence of antibodies is also visible by the decrease in the area under the curve. In black, you can see a control subject (without IgG4 nor IgM antibodies). Each color corresponds to a date (reported below). On the left, you can see the IgG4 serotype with no specific antibodies. On the right, the IgM serotype was reported, for NF155 (up) and NF186 (down). The intensity of fluorescence intensity for anti-NF155 and NF186 IgM antibodies decreased after plasmapharesis (in purple), but not after immunoglobulins therapy (in orange), compared to the initial examination on the day of Guillain-Barré syndrome onset (in red). (B) Longitudinal ultrastructure of left peroneal nerve: node of Ranvier slightly wide. In the paranodal region: loss of cohesion of the outermost myelin sheath. Terminal loops are partially disrupted and distant from the axolemma (arrows), whereas myelin is compacta and not disorganized (C).
Left superficial fibular nerve biopsy was performed on day 8, and neuropathology revealed myelin damage as well as infiltration of lymphocytes and macrophages within the endoneurium. Endoneurial inflammatory cells predominantly consisted of CD8 T-lymphocytes; deposition of C4d on myelin was also observed. Electronic microscopy showed myelinic alterations including disruption of myelin loops but no axonal loss was observed (Fig. 1B).
First-line therapy with intravenous immunoglobulin (IVIG) (2 g/kg in five days) was administered without any clinical (Table 1) or biological (IgM titer, see Fig. 1A) improvement two weeks later. He then underwent 4 rounds of plasma exchange over a period of 10 days (albumin and gelofusine initially then albumin and fresh frozen plasma due to the decrease of coagulation factors), in addition to corticosteroids (intravenous methylprednisolone 500 mg daily for 5 consecutive days followed by 0.5 mg/kg oral prednisone).
Following IVIG and steroids, a partial clinical improvement was seen with better head movements and proximal movements of the right upper limb (MRC score deltoid 2/5, biceps 1/5). The patient complained of numbness in both legs, below the knee. Two months after onset, facial diplegia was still severe but improvements in muscle strength continued in axial, proximal and distal segments (Supplemental Table 1). Repeated conduction studies still showed no motor and sensory responses. Needle EMG showed voluntary activity in deltoid, brachial biceps and forearm muscles, with a rapid firing rate of a reduced number of recruited motor units; fibrillations were present in forearm, hand, and lower limb muscles. Clinical and electrophysiological improvement continued over 4 months without relapse. Anti-NF155 and anti-NF186 IgM disappeared and no IgG seroconversion occurred. Corticosteroid therapy was progressively tapered.
Here, we describe a case of COVID-19-related Guillain-Barré syndrome in which clinical, pathobiological and neurophysiological evidence converged to suggest a paranodopathy with nodal involvement. Anti-neurofascin and anti-contactin antibodies were tested because of the absence of antiganglioside antibodies and according to the clinical phenotype (severe course, cranial nerve involvement and respiratory failure) [1], [2], [3], [6].
The serological profile is original in the reported case. Usually, anti-neurofascin antibodies targeting the NF-155 (paranodal) are associated with a chronic-onset neuropathy and are IgG4 or IgG3 isotypes directed to its Fn3 domain [2]. Here, the patient was seropositive for both NF-155 (located in the paranodes) and NF-186 (nodal isoform), corresponding to pan-neurofascin antibodies, and only had the IgM isotype without further IgG seroconversion. Occurrence of IgM isotype only against the NF-155 was also previously described [4]. The clinical picture (acute onset of neuropathy without further clinical relapse) is in line with the absence of NF-155 or NF-186 IgG seroconversion. The rapid decrease of the NF-155 and NF-186 IgM observed after the second round of plasma exchange was concomitant with the beginning of clinical improvement, supporting that these antibodies were clinically significant rather than coincidental. It is worth noting that immunoglobulin administration (IgG) resulted in no clinical improvement or biological effect. In Guillain-Barré syndrome, antiganglioside antibodies are predominantly IgG, but IgM and IgA antibodies have also been demonstrated.
The pathological data were compatible with previous reports concerning neuropathy with anti-NF antibodies [5], [7], [10]. Within the first month after onset, there was evidence of absence of axonal degeneration or loss on needle EMG (absence of spontaneous activity) and also on nerve biopsy (Fig. 1). Therefore, the clinical deficit and the absence of distal CMAPs/SNAPs were consistent with a process of conduction block. This functional alternation was likely due to nodo-paranodopathy rather than to segmental demyelination [9] according to nerve biopsy findings, although myelin changes were observed.
Needle EMG performed about 2 months after the onset showed widespread fibrillation potentials, consistent with secondary axonal degeneration. In addition, there were signs of collateral sprouting (polyphasic MUPs) on voluntary activity. Therefore, we can conclude in favor of a severe denervation-reinnervation process occurring in a second time. However, the conflicting result between absent CMAPs and recovery of voluntary muscle contraction, as evidenced by needle EMG, could be in favor of additional mechanisms of altered nerve fibre excitability (due to persistent nodo-paranodopathy) [8].
Given that Guillain-Barré syndrome is a typical post-infectious disorder, we hypothesize that the presence of anti-pan-neurofascin IgM antibodies may have been induced by COVID-19 infection. However, they may also have been triggered by tissue damage related to Guillain-Barré syndrome. Either way, it is likely that these antibodies were involved in the pathophysiology of this case. Further investigations are needed to determine whether nodal damage is a feature of severe Guillain-Barré syndrome associated with COVID-19. In particular, a severe phenotype with facial diplegia, respiratory failure and abolition of all electrical responses seems frequent with COVID-19 and these antibodies should be tested in this infectious context.
Conflict of interest
None.
Acknowledgement
The authors thank Frédéric London for his contribution to the final version of the manuscript.
References
- 1.Burnor E., Yang L., Zhou H., Patterson K.R., Quinn C., Reilly M.M. Neurofascin antibodies in autoimmune, genetic, and idiopathic neuropathies. Neurology. 2018;90:e31–e38. doi: 10.1212/WNL.0000000000004773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Delmont E., Manso C., Querol L., Cortese A., Berardinelli A., Lozza A. Autoantibodies to nodal isoforms of neurofascin in chronic inflammatory demyelinating polyneuropathy. Brain. 2017;140:1851–1858. doi: 10.1093/brain/awx124. [DOI] [PubMed] [Google Scholar]
- 3.Delmont E., Brodovitch A., Kouton L., Allou T., Beltran S., Brisset M. Antibodies against the node of Ranvier: a real-life evaluation of incidence, clinical features and response to treatment based on a prospective analysis of 1500 sera. J Neurol. 2020 doi: 10.1007/s00415-020-10041-z. (in press) [DOI] [PubMed] [Google Scholar]
- 4.Doppler K., Stengel H., Appeltshauser L., Grosskreutz J., Man Ng J.K., Meinl E. Neurofascin-155 IgM autoantibodies in patients with inflammatory neuropathies. J Neurol Neurosurg Psychiatry. 2018;89:1145–1151. doi: 10.1136/jnnp-2018-318170. [DOI] [PubMed] [Google Scholar]
- 5.Koike H., Kadoya M., Kaida K., Ikeda S., Kawagashira Y., Iijima M. Paranodal dissection in chronic inflammatory demyelinating polyneuropathy with anti-neurofascin-155 and anti-contactin-1 antibodies. J Neurol Neurosurg Psychiatry. 2017;88:465–473. doi: 10.1136/jnnp-2016-314895. [DOI] [PubMed] [Google Scholar]
- 6.Kouton L., Boucraut J., Devaux J., Rajabally Y.A., Adams D., Antoine J.C. Electrophysiological features of chronic inflammatory demyelinating polyradiculoneuropathy associated with IgG4 antibodies targeting neurofascin 155 or contactin 1 glycoproteins. Clin Neurophysiol. 2020;131:921–927. doi: 10.1016/j.clinph.2020.01.013. [DOI] [PubMed] [Google Scholar]
- 7.Kuwahara M., Suzuki H., Oka N., Ogata H., Yanagimoto S., Sadakane S. Electron microscopic abnormality and therapeutic efficacy in chronic inflammatory demyelinating polyneuropathy with anti-neurofascin155 immunoglobulin G4 antibody. Muscle Nerve. 2018;57:498–502. doi: 10.1002/mus.25757. [DOI] [PubMed] [Google Scholar]
- 8.Ratcliffe C.F., Westenbroek R.E., Curtis R., Catterall W.A. Sodium channel beta1 and beta3 subunits associate with neurofascin through their extracellular immunoglobulin-like domain. J Cell Biol. 2001;154:427–434. doi: 10.1083/jcb.200102086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Uncini A., Susuki K., Yuki N. Nodo-paranodopathy: beyond the demyelinating and axonal classification in anti-ganglioside antibody-mediated neuropathies. Clin Neurophysiol. 2013;124:1928–1934. doi: 10.1016/j.clinph.2013.03.025. [DOI] [PubMed] [Google Scholar]
- 10.Vallat J.M., Mathis S., Magy L., Bounolleau P., Skarzynski M., Heitzmann A. Subacute nodopathy with conduction blocks and anti-neurofascin 140/186 antibodies: an ultrastructural study. Brain. 2018;141 doi: 10.1093/brain/awy134. e56–e56. [DOI] [PubMed] [Google Scholar]