Abstract
Older adults are far more vulnerable to adverse health outcomes and mortality after contracting COVID-19. There are likely multiple age-related biological, clinical, and environmental reasons for this increased risk, all of which are exacerbated by underlying age-associated changes to the immune system as well as increased prevalence of chronic disease states in older adults. Innate immune system overactivity, termed the cytokine storm, appears to be critical in the development of the worst consequences of COVID-19 infection. Pathophysiology suggests that viral stimulation of the innate immune system, augmented by inflammatory signals sent from dying cells, ramps up into a poorly controlled outpouring of inflammatory mediators. Other aging-related changes in cells such as senescence as well as higher prevalence of chronic disease states also likely ramp up inflammatory signaling. This in turn drives downstream pathophysiological changes to pulmonary, cardiovascular, skeletal muscle, and brain tissues that drive many of the adverse health outcomes observed in older adults. This article provides an overview of the underlying etiologies of innate immune system activation and adaptive immune system dysregulation in older adults and how they potentiate the consequences of the COVID-19-related cytokine storm, and possible uses of this knowledge to develop better risk assessment and treatment monitoring strategies.
Keywords: Adaptive immune system, Aging, COVID-19, Cytokine storm, Innate immune system
In the face of the current COVID-19 pandemic sweeping the globe, there has been a marked interest in understanding the etiology of vulnerability to severe COVID-19 disease faced by older and chronically ill adults (1,2). Early on in the COVID-19 pandemic, epidemiological data suggested that older adults were more susceptible to viral infection (1,3). Although more recent information suggests that all age groups are infected at equal rates, older adults, in particular the frail subset, and those with chronic disease states are more likely to develop severe COVID-19-related symptoms requiring hospitalization, ICU utilization, and ultimately death than younger age groups (4,5). The biology that drives these adverse outcomes is not well understood, but prior understanding of vulnerabilities present in the aging immune system and the disease states most associated with mortality provide important clues to the biology underlying severe COVID-19 disease (6). Immunosenescence, which is characterized by a reduced ability to mount an adequate immune response to infection and the predisposition to a proinflammatory state, will be discussed as a major contributor to this vulnerability. Beyond this, additional information concerning advanced vulnerability of older adults will be reviewed based on clues gleaned from the clinical course of the illness and from molecular, cellular, and tissue level studies.
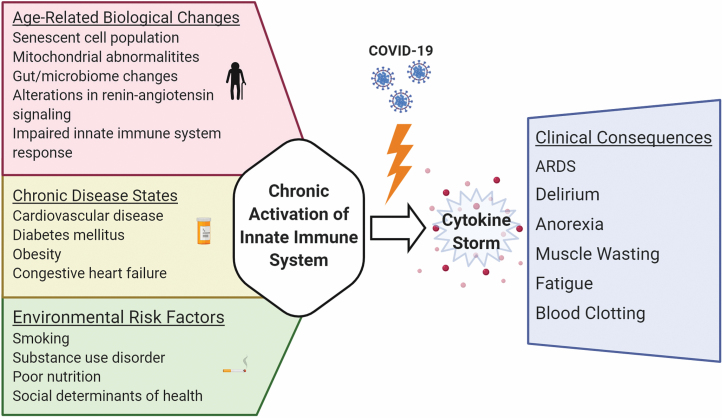
Figure 1 provides an overview of the possible connections between age-related increases in inflammatory signaling and the development of adverse health outcomes related to COVID-19 in older adults.
Figure 1.
Probable age-related contributions to COVID-19-related cytokine storm, as well as observed clinical consequences. Created with BioRender.com. ARDS = acute respiratory distress syndrome.
Immunosenescence and Ineffective Viral Responses
The innate immune system is the first responder system of most tissues and cells to bacterial and viral stressors. Antigenic particles of invading organisms are first detected, followed by ramped up inflammatory signaling. This activates an adaptive immune system response, which is meant to clear infections and quiet the innate immune system response (7). The result of this innate immune system signaling is the marked and rapid production of inflammatory stimulating molecules including interleukins (ILs), chemokines, interferons (IFNs), and tumor necrosis factor (TNF), which impact numerous additional cell types, including endothelial and adipose cells as well as fibroblasts.
Immunosenescence is most often described as an age-related change to the adaptive immune system that results in impaired ability to fight infections, blunted response to new antigens, increased incidence of autoimmunity, and contributes to the development of aging-associated chronic diseases (8,9). A constellation of maladaptive aging-related immune system changes occurs in immunosenescence, with age-related changes to T cells including CD4+ and CD8+ T-cell subtypes developing a limited ability to mount robust responses to new epitopes and a decreased pool of naive CD4+ and CD8+ T cells. In particular, increases in terminally differentiated CD8+ T cells (CD8+CD28−) and replicative-senescent T cells (CD8+CD57+) contribute to proinflammatory states, and also adversely affect antibody-producing B cells (10). CD28 is a co-stimulatory molecule critical for T-cell activation and is known to have an age-related decline in expression. Along with decreases in CD28, the marker CD57 typically increases in expression with age and is associated with non-proliferative T cells. Additionally, increased frequencies of clonal T-cell populations specific for chronic viral infections such as cytomegalovirus emerge in some older adults (11). This change is associated with poor CD4+ T-cell and antibody-generating responses to influenza vaccine, which in turn results in a decreased ability to clear viral infections and higher symptom burden in older adults (12,13). In sum, these changes would make older adults less likely to effectively and efficiently fight and resolve COVID-19 infections.
Aging-Related Changes to the Innate Immune System
Innate immune system changes with aging that may impact age-related vulnerability to COVID-19 include altered cytokine response to immune activators, impaired phagocytosis by macrophages and dendritic cells, and altered Toll-like receptor (TLR) expression (8). There is a shift from type 1 (cell-mediated) to type 2 (humoral-mediated) cytokine response in aging as well as chronic, low-grade proinflammatory cytokine expression due in part to the increased presence of senescent cells. Differential expression of TLRs, which detect components from pathogens and trigger host defense responses, are also observed in aging. These changes in TLR diversity are associated with decreased response to vaccines in older adults and paradoxical increases in IL-8 and IL-6, inflammatory cytokines that are important components of the cytokine storm discussed below (14). Indeed, age-altered TLR signaling declines provide important rationale for the inclusion of TLR agonists in influenza vaccines to appropriately stimulate dendritic cells and promote an adequate cytotoxic T lymphocyte response (10).
Impaired effector functions of neutrophils, dendritic cells and natural killer (NK) cells are also observed in the aging innate immune system. Neutrophils from older adults demonstrate increased spontaneous apoptosis compared to those from young adults which can further contribute to an abnormal innate immune response (15). Aging monocytes and dendritic cells demonstrate delayed response to stimulation by TLR4 due to inefficiencies in downstream signaling pathways (10). These alterations tilt the balance towards a proinflammatory state which may prime some older adults to develop an over exuberant, unregulated inflammatory response to virulent pathogens.
COVID-19-Induced Cytokine Storm in Older Adults and Its Consequences
The term “cytokine storm” was first used when observing the massive cytokine releases related to cancer or to cancer-related therapies (16,17). The term has come to be used in a host of clinical conditions, mostly related to cancer treatment responses or to infectious disease states (18,19).
In COVID-19, IL-6, IL-8, IL-1β, and TNF-α are among the early response molecules that rapidly rise in those at highest risk for severe disease and are key measurable components of the “cytokine storm” seen in a subset of patients with this infection (20–22). These molecules markedly amplify inflammatory pathway activation via nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling, which in turn sets off a chain reaction of pathophysiological damage and decline that contributes to increased mortality rates in those individuals who develop it. Indeed, circulating debris from infection and cytokine damaged cells as well as coronavirus accessory proteins further contribute to the storm (23). Circulating cell debris, termed damage-associated molecular patterns (DAMPs), can result in a highly activated innate immune system partially uncoupled from the viral infection itself (24–26). In a recent paper studying the type 1 IFN response in COVID-19 patients, it was hypothesized that the NF-κB pathway can be activated from excess stimulation via viral components and DAMPs which can occur in the setting of a severe viral infection (27). The impairments in type I IFN responses, specifically reduced IFN-β and IFN-α in severe COVID infections, were associated with reduced viral clearance. In this case, as the cytokine storm develops and is amplified, a host of pathophysiological consequences in multiple tissues induce adverse outcomes and mortality increases.
For example, COVID-19-related pneumonia is among the more common and well-described causes of mortality that is amplified by the cytokine storm. In addition to viral cytotoxicity causing direct tissue damage, high levels of inflammatory cytokines damage and create gaps between endothelial cells, which in turn drives fluid leakage into alveolar spaces, hypoxia, further amplifying tissue damage and localized inflammatory signaling (28). Many of these inflammatory mediators also activate clotting pathways, leading to localized and disseminated intravascular coagulation (DIC), and further hypoxia. As the chain of events continues, severe lung injury, vascular collapse, and death can result (20,29,30).
Beyond contributing to pulmonary injury, the inflammatory molecules in the cytokine storm also trigger multiple and varied systemic signs and symptoms highly relevant to clinical outcomes observed in vulnerable older adults. Central nervous system signs and symptoms are very common in older adults with COVID-19, particularly headache and delirium (31,32). Additionally, severe anorexia and adipsia, known to be driven by cytokinemia, are common in older COVID-19 patients (33,34). Muscle wasting and fatigue are driven by elevated cytokines such as TNF, and satellite cell recovery is inhibited by IL-6, further supporting that the cytokine storm likely drives many additional COVID-19 signs and symptoms, as well as a slow functional recovery, in many older adults (35,36).
Short- and Long-term Consequences of COVID-19 to the Aging Immune System
In addition to direct viral-mediated inflammatory responses, the observed chronic, low-grade inflammation observed in aging has been proposed to contribute to a subset of older adults experiencing cytokine storm in severe COVID-19 infections (37). Insights to the immunopathology of COVID-19 and maladaptive immune responses that can contribute to cytokine storm can be found in the medical literature from the related coronavirus severe acute respiratory syndrome (SARS). Inefficient transitioning from innate to adaptive immunity was observed in individuals at highest risk of requiring mechanical ventilation from SARS infection. In particular, these patients had prolonged expression of IFN-γ-stimulated genes and decreased expression of antigen presentation genes compared to non-severe SARS patients (38). Recent evidence from COVID-19 literature suggests that an increased population of proinflammatory, activated macrophages can also contribute to increased pulmonary inflammation and cytokine storm (39). Furthermore, studies of the SARS virus demonstrated that direct viral protein–host protein interactions led to upregulation of host innate immune signaling, thereby increasing inflammation, which may also be occurring with COVID-19 (40). These innate immune system responses to infection directly contribute to an increased inflammatory state which, in conjunction with a restricted adaptive immune response in older adults, may increase risk of developing cytokine storm.
In addition to acute inflammatory responses from the host response to COVID-19 and the coronavirus itself causing direct cytotoxicity, it has been hypothesized that COVID-19 infection can lead to sustained proinflammatory responses that can lead to prolonged sequelae of the infection (41,42). Following the SARS epidemic, a post-SARS sickness syndrome consisting of malaise, anorexia, and myalgia was observed in a subset of patients, with some studies estimating approximately 30% of patients having post-infectious sickness syndromes, which are classically mediated by hypercytokinemia (43). It is not yet known whether the immune changes observed in acute COVID-19 infection that predispose older adults to adverse outcomes persist over time. If so, it may be that COVID-19 infection may in itself contribute to chronic inflammation that consequently leads to further morbidity. It will be of great interest to perform follow-up immunological phenotyping and measurement of cytokines on patients in the months following resolution of COVID-19 infection to see whether COVID-19 continues to affect host immune responses in the long term.
Age-Related Etiologies of the COVID-19 Cytokine Storm
Aging-specific drivers of chronic innate immune system activation include increased senescent immune and connective tissue cell populations known to secrete a broad array of inflammatory cytokines, an age-altered gastrointestinal (GI) tract and microbiome that enables the low-grade activation of the innate immune system, aging-related increases in the number of cytokine producing fat cells, poor clearance of damaged mitochondria (mitophagy) that contributes to ramping up of inflammatory signaling, and age-related shifts in the renin–angiotensin system (RAS) receptors towards increased inflammatory signaling (44–47). RAS is known to regulate inflammation and has both pro- and anti-inflammatory properties (48). Older animals have heightened responses to angiotensin 2 compared to younger counterparts, and this too, may be contributing to further parenchymal lung damage and vascular injury observed in patients with the most severe presentations of COVID-19 (49,50). Whether or not this chronically activated inflammatory state predisposes a subset of older adults to rapid development of a cytokine storm remains to be determined.
In addition, the same age-related biological changes that drive chronic activation of innate immune systems are also potential etiologies of the age-related vulnerability in developing the cytokine storm. Gerontologists have long highlighted the importance of elevated inflammatory mediators such as IL-6 and C-reactive protein (CRP) as hallmarks of vulnerability to adverse health outcomes. In fact, IL-6, one of the major contributors to the cytokine storm, was called a “Cytokine for Gerontologists” nearly 30 years ago by Ershler et al. for its role in driving adverse health outcomes (51). Although these findings were often dismissed as epidemiological phenomena, meaning that IL-6 was simply identifying individuals who were already sick and therefore more likely to have adverse health outcomes, subsequent biological evidence has demonstrated the pathophysiological potential of chronically elevated levels of IL-6 and other inflammatory mediators (52). More recently, the condition of chronic, age-related elevation of inflammatory mediators and the acceleration of related adverse health outcomes has been termed “inflammaging” by many authors, in part because of the evidence that chronic inflammatory pathway activation related to aging drives aging phenotypes and chronic disease states (44, 52–54).
Aging, Chronic Disease, and COVID-19 Adverse Outcomes
Older adults are far more likely than younger adults to have the chronic conditions type 2 diabetes mellitus, hypertension, cardiovascular disease, renal failure, and chronic obstructive pulmonary disease. These chronic conditions greatly increase the likelihood that those infected with COVID-19 experience more severe illness adverse outcomes, including acute respiratory distress syndrome (ARDS) and respiratory failure, sepsis, acute cardiac injury, and hypercoagulability (55,56). There is also growing evidence linking COVID-19 mortality with pre-existing metabolic syndrome, its tendency to induce inflammation, and its related comorbidities, which has been observed in adults of all ages (57). In particular, obesity and insulin resistance promote a proinflammatory state primed to release immunostimulatory factors (58).
Although there are likely many reasons for this observed relationship, a key biological driver of the propensity towards adverse outcomes arises from the interaction between the coronavirus and components of the RAS. A critical step in providing the virus access to the target cell is the viral spike protein binding with the cell membrane-bound angiotensin-converting enzyme 2 (ACE2) receptor (59). ACE2 receptors are widely expressed in lung tissue, in the GI tract, and in cardiovascular and endothelial tissues (60). Higher expression of ACE2 in individuals with chronic diseases, specifically hypertension and diabetes, is hypothesized to greatly facilitate viral entry after initial exposure to the COVID-19 virus, leading to a rapid increase in innate immune system/proinflammatory signaling. Furthermore, the accompanying microvascular disease and endothelial dysfunction observed in severe COVID-19 cases increase the likelihood for compromised blood flow and oxygenation at the tissue level, enhancing cell death and contributing to the development of DIC.
Importantly, RAS may be playing another crucial role in the development of adverse health outcomes in older adults and in those with chronic disease states. Prior studies suggest that aging and chronic disease states drive higher expression of angiotensin receptor type 1 (ATR1) and lower expression of ATR2 in mitochondria and in other tissues (61,62). The ATR1 receptor, when stimulated by angiotensin 2, contributes to increased mitochondrial free radical production, greater inflammatory signaling, and vasoconstriction, while ATR2 facilitates anti-inflammatory signaling and vascular relaxation (48). ATR1 activation also increases production of reactive oxygen species and increases proinflammatory cytokines, specifically IL-6, TNF-α, and IFN-γ, which can potentially further exacerbate the inflammation and cytotoxicity caused by host response to the virus (61). This aging-associated shift towards ATR1 signaling can also explain the propensity towards the development of the cytokine storm and related endothelial cell and vascular compromise observed in older adults with chronic disease states.
Finally, physical resiliency in the face of COVID-19 likely plays an important role in an individual’s ability to withstand the marked stressor of infection. Although the underlying mechanisms that drive physical resiliency have not been characterized to date, biological hypotheses have been developed that center around the physiological stress response systems including the hypothalamic pituitary adrenal axis (HPA axis), the innate immune system, and autonomic nervous system (63,64). These systems become increasingly dysregulated with age and with chronic disease states and are therefore less likely to function effectively in the face of a major stressor such as COVID-19. Indeed, some evidence suggests that autonomic nervous system function (respiratory rate, heart rate, etc.) cortisol, and innate immune system functions play important roles in COVID-19 pathophysiology and warrant further investigation (65).
General Risk Factors That May Impact Younger and Older Adults
Although age has emerged as the most important risk factor for adverse health outcomes related to the development of the cytokine storm and mortality, some younger individuals also fall gravely ill and develop a similar cytokine storm pathology with COVID-19. Chronic disease states and obesity appear to track closely with this, but there are likely important genetic and environmental reasons that are operant across age groups. For example, there are several polymorphisms in inflammatory cytokine-related genes that make individuals more or less susceptible to inflammatory pathway cascade signaling (66) and hence to development of or protection from cytokine storm. Environmental risk factors, such as smoking (tobacco, electronic-cigarette, or marijuana), chronic infections, such as CMV or HIV, and nutritional deficiencies, such as Vitamin D are known to activate inflammatory pathways, potentially driving differential inflammatory pathway activation across all age groups (67). Social determinants of health also appear to have inverse correlations with systemic inflammation, and a recent analysis of COVID-19 patients without comorbidities demonstrated that low literacy, education, and lack of access to water and electricity were associated with higher rates of mechanical ventilation and mortality (68).
Next Steps
The use of specific cytokine measures to identify older adults at highest risk of adverse health outcomes has previously been confined to the research domain (69). With the advent of COVID-19 and recognition of the potential importance of these measurements for risk assessment and possibly as measurement to guide cytokine storm treatments, research data from older adults will be increasingly important to generate and analyze. For example, the identification of “inflammaging” or chronic inflammatory measures to identify those most at risk of adverse COVID-19 outcomes may help to prioritize future vaccination strategies. Clinical measurement of IL-6 has already been incorporated into treatment algorithms for severely ill patients that may benefit from tocilizumab and other IL-6 blocking agents. Numerous clinical trials are ongoing in the United States regarding use of cytokine blocking agents such as tocilizumab, and Janus Kinase (JAK) inhibitors including baricitinib for patients with COVID-19. JAK inhibition targets cytokine signaling downstream of the cytokine binding to its receptor; one JAK inhibitor can potentially block signaling from several different cytokines. Several observational studies have been published using these agents (70,71).
Understanding the efficacy of targeted anti-inflammatory strategies including IL-6 blockers, JAK inhibitors, as well as patient selection and optimal timing of these therapies will be important steps in successfully attenuating cytokine storm in COVID-19 patients. A focus on mitigating adverse outcomes from the most severe presentations of COVID-19, including cytokine storm, can be more quickly implemented in the clinical setting while vaccine development and testing progress. Given the many similarities, it is important to continue to apply the lessons we have learned on the biology of aging and the development of frail and resilient phenotypes as we understand more about the biology of vulnerability in COVID-19. These approaches and others that build off of both prior evidence and emerging knowledge will be critical to mitigating the mortality and morbidity from COVID-19 in older adults.
Funding
This work was supported by the Johns Hopkins University Claude D. Pepper Older Americans Independence Center funded by the National Institute on Aging of the National Institutes of Health under award number P30AG021334 and the Translational Aging Research Training Program funded by the National Institute on Aging (T32AG058527) to L.S.N.
Conflict of Interest
None declared.
References
- 1. Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. J Am Med Assoc. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Goyal P, Choi JJ, Pinheiro LC, et al. Clinical characteristics of Covid-19 in New York City. N Engl J Med. 2020;382:2372–2374. doi: 10.1056/NEJMc2010419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lippi G, Mattiuzzi C, Sanchis-Gomar F, Henry BM. Clinical and demographic characteristics of patients dying from COVID-19 in Italy versus China. J Med Virol. 2020. doi: 10.1002/jmv.25860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area. J Am Med Assoc. 2020;323:2052–2059. doi: 10.1001/jama.2020.6775 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Maltese G, Corsonello A, Di Rosa M, et al. Frailty and COVID-19: a systematic scoping review. J Clin Med. 2020;9:2106. doi: 10.3390/jcm9072106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Mueller AL, McNamara MS, Sinclair DA. Why does COVID-19 disproportionately affect older people? Aging (Albany NY). 2020;12:9959–9981. doi: 10.18632/aging.103344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Jones SA. Directing transition from innate to acquired immunity: defining a role for IL-6. J Immunol. 2005;175:3463–3468. doi: 10.4049/jimmunol.175.6.3463 [DOI] [PubMed] [Google Scholar]
- 8. Oh SJ, Lee JK, Shin OS. Aging and the immune system: the impact of immunosenescence on viral infection, immunity and vaccine immunogenicity. Immune Netw. 2019;19:e37. doi: 10.4110/in.2019.19.e37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Jenny NS, French B, Arnold AM, et al. Long-term assessment of inflammation and healthy aging in late life: the cardiovascular health study all stars. J Gerontol A Biol Sci Med Sci. 2012;67:970–976. doi: 10.1093/gerona/glr261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. McElhaney JE, Verschoor CP, Andrew MK, Haynes L, Kuchel GA, Pawelec G. The immune response to influenza in older humans: beyond immune senescence. Immun Ageing. 2020;17:10. doi: 10.1186/s12979-020-00181-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Yoshida K, Cologne JB, Cordova K, et al. Aging-related changes in human T-cell repertoire over 20 years delineated by deep sequencing of peripheral T-cell receptors. Exp Gerontol. 2017;96:29–37. doi: 10.1016/j.exger.2017.05.015 [DOI] [PubMed] [Google Scholar]
- 12. Derhovanessian E, Maier AB, Hähnel K, McElhaney JE, Slagboom EP, Pawelec G. Latent infection with cytomegalovirus is associated with poor memory CD4 responses to influenza A core proteins in the elderly. J Immunol. 2014;193:3624–3631. doi: 10.4049/jimmunol.1303361 [DOI] [PubMed] [Google Scholar]
- 13. Yao X, Hamilton RG, Weng NP, et al. Frailty is associated with impairment of vaccine-induced antibody response and increase in post-vaccination influenza infection in community-dwelling older adults. Vaccine. 2011;29:5015–5021. doi: 10.1016/j.vaccine.2011.04.077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Pinti M, Appay V, Campisi J, et al. Aging of the immune system: focus on inflammation and vaccination. Eur J Immunol. 2016;46:2286–2301. doi: 10.1002/eji.201546178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Shaw AC, Goldstein DR, Montgomery RR. Age-dependent dysregulation of innate immunity. Nat Rev Immunol. 2013;13:875–887. doi: 10.1038/nri3547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Clark IA. The advent of the cytokine storm. Immunol Cell Biol. 2007;85:271–273. doi: 10.1038/sj.icb.7100062 [DOI] [PubMed] [Google Scholar]
- 17. Ferrara JL, Abhyankar S, Gilliland DG. Cytokine storm of graft-versus-host disease: a critical effector role for interleukin-1. Transplant Proc. 1993;25:1216–1217. [PubMed] [Google Scholar]
- 18. Guo XJ, Thomas PG. New fronts emerge in the influenza cytokine storm. Semin Immunopathol. 2017;39:541–550. doi: 10.1007/s00281-017-0636-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Ahmed R, Oldstone MB, Palese P. Protective immunity and susceptibility to infectious diseases: lessons from the 1918 influenza pandemic. Nat Immunol. 2007;8:1188–1193. doi: 10.1038/ni1530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Liu J, Li S, Liu J, et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. doi: 10.1016/j.ebiom.2020.102763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Chen G, Wu D, Guo W, et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130:2620–2629. doi: 10.1172/JCI137244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Giamarellos-Bourboulis EJ, Netea MG, Rovina N, et al. Complex immune dysregulation in COVID-19 patients with severe respiratory failure. Cell Host Microbe. 2020;27:992–1000.e3. doi: 10.1016/j.chom.2020.04.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Salimi S, Hamlyn JM. COVID-19 and crosstalk with the hallmarks of aging. J Gerontol A Biol Sci Med Sci. 2020. doi: 10.20944/preprints202004.0182.v1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Shi Y, Evans JE, Rock KL. Molecular identification of a danger signal that alerts the immune system to dying cells. Nature. 2003;425:516–521. doi: 10.1038/nature01991 [DOI] [PubMed] [Google Scholar]
- 25. Newton K, Dixit VM. Signaling in innate immunity and inflammation. Cold Spring Harb Persp Biol. 2012;4:a006049. doi: 10.1101/cshperspect.a006049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. La Gruta NL, Kedzierska K, Stambas J, Doherty PC. A question of self-preservation: immunopathology in influenza virus infection. Immunol Cell Biol. 2007;85:85–92. doi: 10.1038/sj.icb.7100026 [DOI] [PubMed] [Google Scholar]
- 27. Hadjadj J, Yatim N, Barnabei L, et al. Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients. Science. 2020;369:718–724. doi: 10.1126/science.abc6027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Channappanavar R, Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin Immunopathol. 2017;39:529–539. doi: 10.1007/s00281-017-0629-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844–847. doi: 10.1111/jth.14768 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ; HLH Across Speciality Collaboration, UK . COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Prencipe M, Casini AR, Ferretti C, et al. Prevalence of headache in an elderly population: attack frequency, disability, and use of medication. J Neurol Neurosurg Psychiatry. 2001;70:377–381. doi: 10.1136/jnnp.70.3.377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Inouye SK, Westendorp RG, Saczynski JS. Delirium in elderly people. Lancet. 2014;383:911–922. doi: 10.1016/S0140-6736(13)60688-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Kent S, Bluthe RM, Dantzer R, et al. Different receptor mechanisms mediate the pyrogenic and behavioral effects of interleukin 1. Proc Natl Acad Sci USA. 1992;89:9117–9120. doi: 10.1073/pnas.89.19.9117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Mo P, Xing Y, Xiao Y, et al. Clinical characteristics of refractory COVID-19 pneumonia in Wuhan, China. Clin Infect Dis. 2020. doi: 10.1093/cid/ciaa270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Thoma A, Lightfoot AP. NF-kB and inflammatory cytokine signalling: role in skeletal muscle atrophy. Adv Exp Med Biol. 2018;1088:267–279. doi: 10.1007/978-981-13-1435-3_12 [DOI] [PubMed] [Google Scholar]
- 36. Belizário JE, Fontes-Oliveira CC, Borges JP, Kashiabara JA, Vannier E. Skeletal muscle wasting and renewal: a pivotal role of myokine IL-6. SpringerPlus. 2016;5:619. doi: 10.1186/s40064-016-2197-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Meftahi GH, Jangravi Z, Sahraei H, Bahari Z. The possible pathophysiology mechanism of cytokine storm in elderly adults with COVID-19 infection: the contribution of “inflame-aging.” Inflamm Res. 2020;69(9):825–839. doi: 10.1007/s00011-020-01372-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cameron MJ, Bermejo-Martin JF, Danesh A, Muller MP, Kelvin DJ. Human immunopathogenesis of severe acute respiratory syndrome (SARS). Virus Res. 2008;133:13–19. doi: 10.1016/j.virusres.2007.02.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Liao M, Liu Y, Yuan J, et al. Single-cell landscape of bronchoalveolar immune cells in patients with COVID-19. Nat Med. 2020;26:842–844. doi: 10.1038/s41591-020-0901-9 [DOI] [PubMed] [Google Scholar]
- 40. Zhao C, Zhao W. NLRP3 inflammasome—a key player in antiviral responses. Front Immunol. 2020;11:211. doi: 10.3389/fimmu.2020.00211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Galeotti C, Bayry J. Autoimmune and inflammatory diseases following COVID-19. Nat Rev Rheumatol. 2020;16:413–414. doi: 10.1038/s41584-020-0448-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Batisse D, Benech N, Botelho-Nevers E, et al. Clinical recurrences of COVID-19 symptoms after recovery: viral relapse, reinfection or inflammatory rebound? J Infect. 2020. doi: 10.1016/j.jinf.2020.06.073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Chrousos GP, Kaltsas G. Post-SARS sickness syndrome manifestations and endocrinopathy: how, why, and so what? Clin Endocrinol (Oxf). 2005;63:363–365. doi: 10.1111/j.1365-2265.2005.02361.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Coppé J, Desprez P, Krtolica A, Campisi J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol. 2010;5:99–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Kim S, Jazwinski SM. The gut microbiota and healthy aging: a mini-review. Gerontology. 2018;64:513–520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Tchkonia T, Morbeck DE, Zglinicki Tv, et al. Fat tissue, aging, and cellular senescence. Aging Cell. 2010;9:667–684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Jang JY, Blum A, Liu J, Finkel T. The role of mitochondria in aging. J Clin Invest. 2018;128:3662–3670. doi: 10.1172/JCI120842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Fyhrquist F, Saijonmaa O. Renin-angiotensin system revisited. J Intern Med. 2008;264:224–236. doi: 10.1111/j.1365-2796.2008.01981.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Dinh QN, Drummond GR, Kemp-Harper BK, et al. Pressor response to angiotensin II is enhanced in aged mice and associated with inflammation, vasoconstriction and oxidative stress. Aging. 2017;9:1595–1606. doi: 10.18632/aging.101255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Wichmann D, Sperhake J, Lütgehetmann M, et al. Autopsy findings and venous thromboembolism in patients with COVID-19: a prospective cohort study. Ann Intern Med. 2020. doi: 10.7326/M20-2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Ershler WB. Interleukin-6: a cytokine for gerontologists. J Am Geriatr Soc. 1993;41:176–181. doi: 10.1111/j.1532-5415.1993.tb02054.x [DOI] [PubMed] [Google Scholar]
- 52. Furman D, Campisi J, Verdin E, et al. Chronic inflammation in the etiology of disease across the life span. Nat Med. 2019;25:1822–1832. doi: 10.1038/s41591-019-0675-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018;15:505–522. doi: 10.1038/s41569-018-0064-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Gross AL, Walker KA, Moghekar AR, et al. Plasma markers of inflammation linked to clinical progression and decline during preclinical AD. Front Aging Neurosci. 2019;11:229. doi: 10.3389/fnagi.2019.00229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Yang J, Zheng Y, Gou X, et al. Prevalence of comorbidities and its effects in coronavirus disease 2019 patients: a systematic review and meta-analysis. Int J Infect Dis. 2020;94:91–95. doi: 10.1016/j.ijid.2020.03.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Chen T, Wu D, Chen H, et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. Br Med J (Clinical research ed.). 2020;368:m1091. doi: 10.1136/bmj.m1091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Palmieri L, Vanacore N, Donfrancesco C, et al. Clinical characteristics of hospitalized individuals dying with COVID-19 by age group in Italy. J Gerontol A Biol Sci Med Sci . 2020. doi: 10.1093/gerona/glaa146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Saltiel AR, Olefsky JM. Inflammatory mechanisms linking obesity and metabolic disease. J Clin Invest. 2017;127:1–4. doi: 10.1172/JCI92035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Groß S, Jahn C, Cushman S, Bär C, Thum T. SARS-CoV-2 receptor ACE2-dependent implications on the cardiovascular system: from basic science to clinical implications. J Mol Cell Cardiol. 2020;144:47–53. doi: 10.1016/j.yjmcc.2020.04.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631–637. doi: 10.1002/path.1570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Abadir PM. The frail renin-angiotensin system. Clin Geriatr Med. 2011;27:53–65. doi: 10.1016/j.cger.2010.08.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Hsueh WA, Wyne K. Renin‐angiotensin‐aldosterone system in diabetes and hypertension. J Clin Hypertens. 2011;13:224–237. doi: 10.1111/j.1751-7176.2011.00449.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. McEwen BS. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev. 2007;87:873–904. doi: 10.1152/physrev.00041.2006 [DOI] [PubMed] [Google Scholar]
- 64. Epel ES, Lithgow GJ. Stress biology and aging mechanisms: toward understanding the deep connection between adaptation to stress and longevity. J Gerontol A Biol Sci Med Sci. 2014;69(Suppl 1):S10–S16. doi: 10.1093/gerona/glu055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Pal R. COVID-19, hypothalamo-pituitary-adrenal axis and clinical implications. Endocrine. 2020;68:251–252. doi: 10.1007/s12020-020-02325-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Qi Y, Chen X, Wu N, Ma C, Cui X, Liu Z. Identification of risk factors for sepsis-associated mortality by gene expression profiling analysis. Mol Med Rep. 2018;17:5350–5355. doi: 10.3892/mmr.2018.8491 [DOI] [PubMed] [Google Scholar]
- 67. Alqahtani JS, Oyelade T, Aldhahir AM, et al. Prevalence, severity and mortality associated with COPD and smoking in patients with COVID-19: a rapid systematic review and meta-analysis. PLoS ONE. 2020;15:e0233147. doi: 10.1371/journal.pone.0233147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Bello-Chavolla OY, González-Díaz A, Antonio-Villa NE, et al. Unequal impact of structural health determinants and comorbidity on COVID-19 severity and lethality in older Mexican adults: considerations beyond chronological aging. J Gerontol A Biol Sci Med Sci. 2020. doi: 10.1093/gerona/glaa163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Varadhan R, Yao W, Matteini A, et al. Simple biologically informed inflammatory index of two serum cytokines predicts 10 year all-cause mortality in older adults. J Gerontol A Biol Sci Med Sci. 2014;69:165–173. doi: 10.1093/gerona/glt023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Alzghari SK, Acuña VS. Supportive treatment with tocilizumab for COVID-19: a systematic review. J Clin Virol. 2020;127:104380. doi: 10.1016/j.jcv.2020.104380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Cantini F, Niccoli L, Matarrese D, Nicastri E, Stobbione P, Goletti D. Baricitinib therapy in COVID-19: A pilot study on safety and clinical impact. J Infect. 2020;81:318–356. doi: 10.1016/j.jinf.2020.04.017 [DOI] [PMC free article] [PubMed] [Google Scholar]