Abstract
Coronavirus disease 2019 (COVID-19), like cancer, is a complex disease with clinical phases of progression. Initially conceptualized as a respiratory disease, COVID-19 is increasingly recognized as a multi-organ and heterogeneous illness. Disease staging is a method for measuring the progression and severity of an illness using objective clinical and molecular criteria. Integral to cancer staging is “metastasis,” defined as the spread of a disease-producing agent, including neoplastic cells and pathogens such as certain viruses, from the primary site to distinct anatomic locations. Staging provides valuable frameworks and benchmarks for clinical decision-making in patient management, improved prognostication, and evidence-based treatment selection.
Coronavirus disease 2019 (COVID-19), like cancer, is a complex disease with clinical phases of progression. Initially conceptualized as a respiratory disease, COVID-19 is increasingly recognized as a multi-organ and heterogeneous illness. Disease staging is a method for measuring the progression and severity of an illness using objective clinical and molecular criteria. Integral to cancer staging is “metastasis,” defined as the spread of a disease-producing agent, including neoplastic cells and pathogens such as certain viruses, from the primary site to distinct anatomic locations. Staging provides valuable frameworks and benchmarks for clinical decision-making in patient management, improved prognostication, and evidence-based treatment selection.
Main Text
Coronavirus disease 2019 (COVID-19), like cancer, is a complex disease with a natural history that manifests specific clinical phases of progression. Initially conceptualized as a primarily respiratory viral disease posing additional risk to elderly patients with co-morbidities, COVID-19 is now recognized as a more complex, multi-organ and heterogeneous illness than initially anticipated. Patients with COVID-19 display a diverse array of symptoms and complications, including a hyper-inflammatory state, endothelial dysfunction and thromboembolic disease, neuropsychiatric symptoms, and a clinical course that may be complicated by abrupt, unexpected deterioration during apparent recovery.
Disease staging is a method for measuring the progression and severity of an illness using objective clinical and molecular criteria. Through such a process, staging defines discrete points in the course of a particular disease that are clinically detectable and reflect present risk, potential long-term effects, and likelihood of death. Staging systems provide valuable frameworks and benchmarks for clinical decision-making in patient management, improved prognostication, and evidence-based treatment selection. Cancer has become the paradigm of disease staging, using alphanumeric codes (“TNM” for tumor, lymph nodes, and metastasis; “0–4” for absence [“0”] or presence and severity of the code [“1–4”]), and obtaining global acceptance for classifying the extent of any given malignancy. Integral to cancer staging is also metastasis, defined as the spread of a disease-producing agent, including transformed neoplastic cells and pathogens, from the original or primary site to a distinct anatomic location in the body. Although predominantly adjudicated for neoplastic progression, this biological phenomenon is also observed and recognized in other disease processes, such as certain viral illnesses, and should be applied when justified by clinical and molecular criteria. Increasing evidence indicates that vascular endothelial cells in many organs, including the central nervous system, heart, and lung, express ACE-2 and other severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) entry receptors and are a likely target of infection beyond the nasopharynx and lung, contributing to dissemination of the virus throughout the body (Libby and Lüscher, 2020). Furthermore, multiple studies report detectable blood viremia in severely ill patients (Prebensen et al., 2020), suggesting that viral shedding may occur intra-vascularly. These data warrant the consideration of COVID-19 as a metastatic disease, with local extension and hematogenous spread of SARS-CoV-2 from the primary point of entry in the upper respiratory track to neighboring and distant anatomic locations.
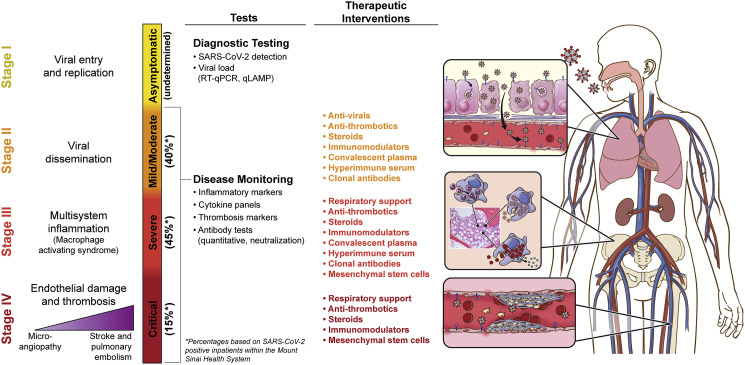
Here, we postulate an integrative clinicopathologic staging of COVID-19 that synthesizes the underlying mechanisms, clinical progression, and severity of disease. Our proposed staging is characterized by four phases, detailed in Figure 1 , acknowledging that the model is not necessarily linear and that some patients may present at an advanced stage.
Figure 1.
COVID-19 Staging
Visual summary of COVID-19 stages, with their associated pathophysiology, and suggested testing and therapeutic interventions. Illustration by Jill Gregory, used with permission of ©Mount Sinai Health System. ∗, approximate distribution of primary staging of disease among symptomatic patients.
COVID-19 Stage 1: Viral Entry and Replication (Asymptomatic)
Asymptomatic disease has been well-described since the earliest days of the SARS-CoV-2 pandemic. Children and younger adults are more likely to be in this group, though asymptomatic nucleic acid amplification test (NAAT)-positive cases have been seen in all age and risk groups to varying degrees. Estimates of asymptomatic disease in environments where widespread population sample testing has been done have ranged from 10% to 60%, with 40% being the newest estimate reported by the Centers for Disease Control and Prevention (CDC) (https://www.cdc.gov/coronavirus/2019-ncov/hcp/planning-scenarios.html). A subset of asymptomatic carriers may become symptomatic over subsequent days. There have been varying reports of infectivity of asymptomatic carriers, but data increasingly indicate that asymptomatic carriers can transmit SARS-CoV-2 (Gandhi et al., 2020), highlighting the importance of frequent testing in people with exposures or high-risk work environments. Critical processes for detecting stage 1 COVID-19 include qualitative and quantitative testing, isolation, infection prevention, and monitoring for development of symptoms (Wajnberg et al., 2020). A high viral load, as measured by means of real-time quantitative polymerase chain reaction (RT-PCR), is a significant and independent biomarker associated with COVID-19 progression and death in patients without or with cancer (Pujadas et al., 2020; Westblade et al., 2020). Transforming qualitative into quantitative testing could assist clinicians in risk-stratifying patients and choosing among available patient management strategies. Widespread adoption of viral load measurements could be facilitated by rapid tests under development, such as quantitative loop-mediated isothermal amplification (qLAMP), enabling viral load assessment within an hour. Treatment is not recommended for this group, though individuals may be eligible for temporal surveillance studies and/or clinical trials focused on prevention of symptoms, reduction of infectivity, and disease progression.
COVID-19 Stage 2: Viral Dissemination (Mild or Moderate)
There have been multiple reports of variable presentations among this group, the consensus being that patients present one or more of the following symptoms: fever or chills, cough, shortness of breath, fatigue, muscle or body aches, headache, new loss of taste or smell, sore throat, congestion or runny nose, nausea or vomiting, and diarrhea. Stage 2 requires testing and confirmation of disease, isolation, and infection prevention precautions, as most people are likely highly infectious during their acute period of symptoms. Predictors of poor prognosis include male gender, increased age, high viral load, and presence of comorbidities, including hypertension, diabetes mellitus, and coronary artery disease. Laboratory data potentially helpful in predicting progress to more severe disease (see stages 3 and 4, below) include inflammatory markers, such as procalcitonin and C-reactive protein, and raised levels of byproducts of the coagulation cascade, including D-dimers and tissue factor. Recommended clinical monitoring includes intermittent pulse oximetry and vigilance for other symptoms or signs of progression toward complications, such as cardiac, neurologic, and hypercoagulable indicators. This is the stage that correlates with active viral replication and with treatment protocols including anti-viral drugs and steroids. Convalescent plasma, hyperimmune serum, clonal antibody therapies, and anti-thrombotics could be relevant in this phase of disease (Liu et al., 2020).
COVID-19 Stage 3: Multi-system Inflammation (Severe)
A subset of patients with COVID-19 progress to or present with a severe phase of the disease, requiring hospitalization. In this stage, often about 1–2 weeks after symptom onset, patients experience worsening dyspnea and hypoxia, along with subclinical elevations in indicators of organ damage (e.g., cardiac, kidney, and liver). Derangements of coagulation biomarkers can also be present (e.g., elevated D-dimer) and may represent sub-clinical progression toward stage 4. Neutrophils and macrophages have emerged as key players in COVID-19 disease progression. Neutrophil extracellular traps (NETs) containing complement, tissue factor, and oxidants have been reported as a mechanism connecting the hyper-inflammatory and hyper-coagulable states (Jose and Manuel, 2020). Studies have revealed extensive hemophagocytosis, consistent with a hyper-inflammatory state on a similar spectrum as macrophage activating syndrome (MAS), hemophagocytic lymphohistiocytosis (HLH), multisystem inflammatory syndrome in children (MIS-C), and canonical Kawasaki’s disease (Colafrancesco et al., 2020). These mechanisms point to additional potential biomarkers, such as elevated interleukins (mainly IL-6) (Del Valle et al., 2020), calprotectin, ferritin, and soluble CD163 (Leppkes et al., 2020). Depending on the time between onset of disease and entering stage 3, patients may have reduced benefit from antibody-based treatments, and instead therapy should focus on providing oxygen support, anti-inflammatory and immunomodulatory therapies, anti-thrombotics, and clinical trials, including experimental therapies such as mesenchymal stem cells (Golchin et al., 2020). Following the guiding principle from personalized cancer care of matching treatments to the right stage, prognostic group, and molecular subtype for each patient can help avoid side effects of treatments that are unlikely to help a particular patient and boost the power of clinical trials by conducting them in a more well-defined population.
COVID-19 Stage 4: Endothelial Damage, Thrombosis, and Multi-organ Dysfunction (Critical)
A minority of patients with COVID-19 will reach stage 4 disease, associated with higher risk of mortality. This stage is characterized by diffuse endothelial injury (characterized by elevated levels of von Willebrand factor and endothelialitis) and a severely hyperinflammatory and hypercoagulable state. These processes lead to dysregulated thrombo-inflammatory pathways, resulting in microthrombi formation and systemic microvascular dysfunction (Nadkarni et al., 2020). Clinical phenomena include severe hypoxemic respiratory failure associated with multi-organ failure, including myocardial injury as evidenced by troponemia, with cardiac structural abnormalities and arrhythmias, severe acute kidney injury requiring renal replacement therapy, acute neurological disease, venous and arterial thromboembolic events, and severe metabolic derangements, such as persistent hyperglycemia and ketosis. Post-mortem evaluations have revealed extensive presence of pulmonary embolisms and microthrombi across a variety of organs, including but not limited to lung, heart, and brain (Bryce et al., 2020). We have observed that some stage 4 patients have high levels of antibodies to SARS-CoV-2 virus without evidence of active viral infection (Wajnberg et al., 2020), suggesting that in this stage the underlying pathophysiology is primarily driven by the inflammatory response and coagulopathy rather than direct viral injury. End-organ damage can be very rapid and challenging to treat, but studies so far have shown some improvement with anti-thrombotics and anti-inflammatory protocols (Mehta et al., 2020). As is the case with cancer patients, those with late-stage disease often have very limited options and are refractory to most available treatments. Therefore, clinical trials should be considered for these patients.
In conclusion, what has been learned about COVID-19 highlights the complexity of this multi-system disease with a hyperinflammatory phenotype and endothelial damage. A staging system that integrates diagnostic testing, clinical data, and interventions with pathophysiologic mechanisms will assist in guiding clinical management and provide mechanistic insights, just as the TNM system first devised by Denoix in the 1940s has provided an invaluable framework for the conceptualization of cancer and evolution of its treatment. Important areas deserving further study include chronic symptoms in COVID-19 survivors, such as cardiac and renal disease, pulmonary fibrosis, reduced exercise tolerance, and overall functional impairment. More longitudinal data are required to determine the full extent of chronic injury and better define the needs of the COVID-19 survivor population. It will also be of interest to better characterize viral load dynamics and tropism for distinct organs as disease progresses, the exact routes and mechanisms of dissemination of the virus, associated antibody titers, and the challenging balance between the need to control the virus and the importance of mitigating associated inflammatory damage. Continued clinical trials, prospective and retrospective data analysis, and histopathologic and molecular studies will further our understanding of this complex disease.
Acknowledgments
We would like to acknowledge the generous support of the JPB Foundation, the Steven & Alexandra Cohen Foundation, and the Open Philanthropy Project (#2020-215611) as well as other philanthropic donations. CCC, AW, A.F.-B., F.K., and V.S. are recipients of a grant/contract from NCI; this effort was deliberated with Leidos Biomedical Research Inc./NCI over the last several months and is now going to be supported by the Serological Sciences Network (SeroNet). This project will be funded in whole or in part with Federal funds from the National Cancer Institute, National Institutes of Health, under Contract No. 75N91019D00024, Task Order No. 75N91020F00003. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products or organizations imply endorsement by the U.S. Government. We would like to thank Bethany Percha for help with epidemiologic estimates of SARS-CoV-2-positive patients within the Mount Sinai Health System.We would like to thank Patricia Spilman and Phil Yang for their helpful edits.
Declaration of Interests
C.C.-C., A.F.-B., F.K., and V.S., as part of Mount Sinai, report that the institution has licensed SARS-CoV-2 serological assays to commercial entities and have filed for patent protection for serological assays. R.S. reports to be associated with SEMA4. P.S.-S. is the chief executive officer and majority shareholder of both ImmunityBio, Inc., and NantKwest, Inc., and is developing a qLAMP assay.
References
- Bryce C., Grimes Z., Pujadas E., Ahuja S., Beasley M.B., Albrecht R., Hernandez T., Stock A., Zhao Z., Al Rasheed M., et al. Pathophysiology of SARS-CoV-2: targeting of endothelial cells renders a complex disease with thrombotic microangiopathy and aberrant immune response. The Mount Sinai COVID-19 autopsy experience. medRxiv. 2020 doi: 10.1101/2020.05.18.20099960. [DOI] [Google Scholar]
- Colafrancesco S., Alessandri C., Conti F., Priori R. COVID-19 gone bad: a new character in the spectrum of the hyperferritinemic syndrome? Autoimmun. Rev. 2020;19:102573. doi: 10.1016/j.autrev.2020.102573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Del Valle D.M., Kim-Schulze S., Huang H.H., Beckmann N.D., Nirenberg S., Wang B., Lavin Y., Swartz T.H., Madduri D., Stock A., et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat. Med. 2020 doi: 10.1038/s41591-020-1051-9. Published online August 24, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gandhi M., Yokoe D.S., Havlir D.V. Asymptomatic transmission, the Achilles’ heel of current strategies to control Covid-19. N. Engl. J. Med. 2020;382:2158–2160. doi: 10.1056/NEJMe2009758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golchin A., Seyedjafari E., Ardeshirylajimi A. Mesenchymal stem cell therapy for COVID-19: present or future. Stem Cell Rev. Rep. 2020;16:427–433. doi: 10.1007/s12015-020-09973-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jose R.J., Manuel A. COVID-19 cytokine storm: the interplay between inflammation and coagulation. Lancet Respir. Med. 2020;8:e46–e47. doi: 10.1016/S2213-2600(20)30216-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leppkes M., Knopf J., Naschberger E., Lindemann A., Singh J., Herrmann I., Stürzl M., Staats L., Mahajan A., Schauer C., et al. Vascular occlusion by neutrophil extracellular traps in COVID-19. EBioMedicine. 2020;58:102925. doi: 10.1016/j.ebiom.2020.102925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Libby P., Lüscher T. COVID-19 is, in the end, an endothelial disease. Eur. Heart J. 2020;41:3038–3044. doi: 10.1093/eurheartj/ehaa623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S.T.H., Lin H.M., Baine I., Wajnberg A., Gumprecht J.P., Rahman F., Rodriguez D., Tandon P., Bassily-Marcus A., Bander J., et al. Convalescent plasma treatment of severe COVID-19: a propensity score-matched control study. Nat. Med. 2020 doi: 10.1038/s41591-020-1088-9. [DOI] [PubMed] [Google Scholar]
- Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J., HLH Across Speciality Collaboration, UK COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nadkarni G.N., Lala A., Bagiella E., Chang H.L., Moreno P., Pujadas E., Arvind V., Bose S., Charney A.W., Chen M.D., et al. Anticoagulation, mortality, bleeding and pathology among patients hospitalized with COVID-19: a single health system study. J. Am. Coll. Cardiol. 2020 doi: 10.1016/j.jacc.2020.08.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prebensen C., Hre P.L.M., Jonassen C., Rangberg A., Blomfeldt A., Svensson M., Omland T., Berdal J.E. SARS-CoV-2 RNA in plasma is associated with ICU admission and mortality in patients hospitalized with COVID-19. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa1338. Published online September 5, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pujadas E., Chaudhry F., McBride R., Richter F., Zhao S., Wajnberg A., Nadkarni G., Glicksberg B.S., Houldsworth J., Cordon-Cardo C. SARS-CoV-2 viral load predicts COVID-19 mortality. Lancet Respir. Med. 2020;8:e70. doi: 10.1016/S2213-2600(20)30354-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wajnberg A., Mansour M., Leven E., Bouvier N.M., Patel G., Firpo-Betancourt A., Mendu R., Jhang J., Arinsburg S., Gitman M., et al. Humoral response and PCR positivity in patients with COVID-19 in the New York City region, USA: an observational study. Lancet Microbe. 2020 doi: 10.1016/S2666-5247(20)30120-8. Published online September 25, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westblade L.F., Brar G., Pinheiro L.C., Paidoussis D., Rajan M., Martin P., Goyal P., Sepulveda J.L., Zhang L., George G., et al. SARS-CoV-2 viral load predicts mortality in patients with and without cancer who are hospitalized with COVID-19. Cancer Cell. 2020;38:661–671. doi: 10.1016/j.ccell.2020.09.007. this issue. [DOI] [PMC free article] [PubMed] [Google Scholar]