Abstract
Inflammatory cardiomyopathy, characterized by inflammatory cell infiltration into the myocardium and a high risk of deteriorating cardiac function, has a heterogeneous aetiology. Inflammatory cardiomyopathy is predominantly mediated by viral infection, but can also be induced by bacterial, protozoal or fungal infections as well as a wide variety of toxic substances and drugs and systemic immune-mediated diseases. Despite extensive research, inflammatory cardiomyopathy complicated by left ventricular dysfunction, heart failure or arrhythmia is associated with a poor prognosis. At present, the reason why some patients recover without residual myocardial injury whereas others develop dilated cardiomyopathy is unclear. The relative roles of the pathogen, host genomics and environmental factors in disease progression and healing are still under discussion, including which viruses are active inducers and which are only bystanders. As a consequence, treatment strategies are not well established. In this Review, we summarize and evaluate the available evidence on the pathogenesis, diagnosis and treatment of myocarditis and inflammatory cardiomyopathy, with a special focus on virus-induced and virus-associated myocarditis. Furthermore, we identify knowledge gaps, appraise the available experimental models and propose future directions for the field. The current knowledge and open questions regarding the cardiovascular effects associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection are also discussed. This Review is the result of scientific cooperation of members of the Heart Failure Association of the ESC, the Heart Failure Society of America and the Japanese Heart Failure Society.
Subject terms: Inflammation, Cardiomyopathies, Infectious diseases, SARS-CoV-2
In this Review, Tschöpe and colleagues summarize and evaluate the available evidence on the pathogenesis, diagnosis and treatment of myocarditis and inflammatory cardiomyopathy, with special focus on virus-induced and virus-associated myocarditis. The authors also identify knowledge gaps, appraise available experimental models and propose future directions for the field.
Key points
The role of specific viruses, immune cells and autoimmunity in the pathogenesis of myocarditis and inflammatory cardiomyopathy is still incompletely understood, and advanced animal and cell models are required for future research.
Advanced animal models that take into account immune experience and exposure to environmental factors and in vitro models with immune cell interactions are needed to facilitate better clinical translation of the findings.
Improved standardization of available invasive and noninvasive diagnostic tools and a consensus on their specific use are needed to allow specific diagnosis and stratification of patient cohorts for the implementation of aetiology-based therapies.
To develop aetiology-based therapies, the efficacy of many existing, repurposed or emerging therapies needs to be evaluated in large, controlled, randomized trials.
Introduction
Inflammatory cardiomyopathy is defined as myocarditis in association with cardiac dysfunction and ventricular remodelling1,2. Despite extensive research and improved diagnosis and understanding of the pathogenesis of inflammatory cardiomyopathy, this disorder is still associated with a poor prognosis when complicated by left ventricular (LV) dysfunction, heart failure (HF) or arrhythmia3. Furthermore, fulminant myocarditis, a rare, sudden and severe cardiac inflammation, is one of the main causes of cardiogenic shock in young adults4,5. Prompt diagnosis and specific treatment strategies are needed to reduce mortality and the need for heart transplantation in these patients4,5. Many questions remain unanswered regarding the pathogenesis of inflammatory cardiomyopathy and the role of the viral infection, the immune system, the host genetic background and the environment in disease progression and prognosis. These gaps in knowledge highlight the need for advanced experimental systems that can better model the human immune system and the need to improve the characterization and classification of the patients, for example, with the use of phenomapping and phenomics, which involve detailed evaluation of immune status, viral presence and/or other biomarkers.
In this Review, we discuss the available evidence and identify the gaps in our understanding of the pathogenesis, diagnosis, treatment and prognosis of myocarditis and inflammatory cardiomyopathy, appraise the available animal and cell models of these conditions and propose future directions for the field. We discuss the role of viruses as active inducers or as potential bystanders of myocarditis and inflammatory cardiomyopathy. We assess the relevance of histology, immunohistology and molecular biology techniques for the analysis of endomyocardial biopsy (EMB) samples, as well as advanced imaging methods and the role of inflammatory and immune cell markers, immune cell ratios, microRNAs and antibodies for the diagnosis, guidance of therapeutic decisions and management in patients with myocarditis and inflammatory cardiomyopathy. We outline patient-specific therapeutic options that are based on an accurate diagnosis, covering current and novel strategies. The aim of the Review is to help clinicians and scientists apply the best diagnostic and therapeutic approaches to solve individual patient problems in clinical practice. This Review is the result of a scientific cooperation of members from the Heart Failure Association of the ESC, the Heart Failure Society of America and the Japanese Heart Failure Society.
Pathogenesis
The role of viruses
Myocarditis is an inflammatory cardiac disorder induced predominantly by viruses6,7 but also by other infectious agents including bacteria (such as Borrelia spp.), protozoa (such as Trypanosoma cruzi) and fungi. Myocarditis can also be induced by a wide variety of toxic substances and drugs (such as immune checkpoint inhibitors)8 and systemic immune-mediated diseases9. Importantly, the aetiopathogenesis, induction and course of myocarditis related to different infectious agents vary considerably. The most common viruses associated with inflammatory cardiomyopathy include: primary cardiotropic viruses that can be cleared from the heart, including adenoviruses and enteroviruses (such as coxsackie A viruses or coxsackie B viruses, and echoviruses); vasculotropic viruses that are likely to have lifelong persistence, including parvovirus B19 (B19V; from the erythrovirus family); lymphotropic viruses with lifelong persistence that belong to the Herpesviridae family (such as human herpesvirus 6 (HHV6), Epstein–Barr virus and human cytomegalovirus); viruses that indirectly trigger myocarditis by activating the immune system10,11, including human immunodeficiency virus (HIV), hepatitis C virus (HCV), influenza A virus and influenza B virus; and viruses from the Coronaviridae family, including Middle East respiratory syndrome coronavirus (MERS-CoV), severe acute respiratory syndrome coronavirus (SARS-CoV) and SARS-CoV-2, which have angiotensin-converting enzyme 2 (ACE2) tropism and can potentially mediate direct cardiac injury. These coronaviruses are also suggested to indirectly trigger myocarditis, in a similar manner to influenza A and B viruses, via cytokine-mediated cardiotoxicity or by triggering an autoimmune response against components of the heart12 (Table 1). The exact pathological mechanisms underlying SARS-CoV-2-associated heart disease are so far unknown and require in-depth investigation of EMB and autopsy samples from affected patients.
Table 1.
Viruses associated with myocarditis and inflammatory cardiomyopathy
| Viral tropism | Virus | Viral genome | Virulence | Treatment for associated heart disease |
|---|---|---|---|---|
| Cardiotropic | Adenoviruses | dsDNA | Virulent | IFNα or IFNβ (?); direct-acting antiviral therapy (?); intravenous immunoglobulins (?) |
| Enteroviruses (coxsackieviruses, echoviruses) | (+)ssRNA | |||
| Vasculotropic | Parvovirus B19 | ssDNA | Bystander: latent; low viral DNA copy numbers in cardiac tissue; with or without cardiac inflammation | No need for antiviral therapy |
| Virulent: high viral DNA copy numbers in cardiac tissue (>500 viral DNA copies per microgram cardiac DNA) with cardiac inflammation or systemic infection | Intravenous injection of immunoglobulins (?) | |||
| Lymphotropic | Cytomegalovirus; Epstein–Barr virus; human herpesvirus 6 | dsDNA | Bystander: latent; low DNA copy numbers in cardiac tissue; with or without cardiac inflammation | No need for antiviral therapy |
| Virulent: high cardiac DNA copy numbers; with cardiac inflammation | Anti-herpesvirus drugs | |||
| Cardiotoxic | Hepatitis C virus; HIV; Influenza viruses | (+)ssRNA | Virulent: cardiac inflammation with viraemia | Direct-acting antiviral therapy |
| ACE2-tropic; cardiotoxic (?) | Coronaviruses (MERS-CoV, SARS-CoV, SARS-CoV-2) | (+)ssRNA | Virulent: viraemia; cardiac inflammation (?) | Potential treatments currently under investigation: remdesivir; hydroxychloroquine and azithromycin; darunavir and cobicistat; lopinavir–ritonavir; favipiravir; ribavirin; IFNα; camostat mesylate |
‘(?)’ indicates unclear, needs further investigation; ACE2, angiotensin-converting enzyme 2; dsDNA, double-stranded DNA; HIV, human immunodeficiency virus; MERS-CoV, Middle East respiratory syndrome coronavirus; SARS-CoV, severe acute respiratory syndrome coronavirus; ssDNA, single-stranded DNA; (+)ssRNA, positive-sense single-stranded RNA.
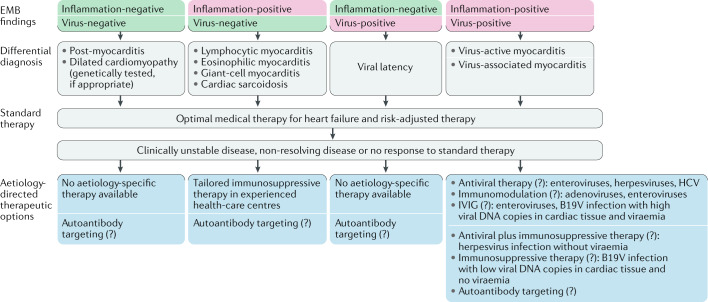
A distinction is needed between virus-induced inflammatory cardiomyopathy and virus-associated inflammatory cardiomyopathy (in case of viral latency), which depends on the causality between the virus and the pathogenesis of inflammatory cardiomyopathy. Furthermore, a clear classification should be made to distinguish between viruses that directly (cardiotropic and vasculotropic viruses) or indirectly (lymphotropic viruses) infiltrate the heart, and viruses that might not necessarily infect cardiac cells but indirectly induce cardiac injury and negative inotropy by triggering a cytokine storm or a cellular immune response by molecular mimicry.
The ESC guidelines require viral diagnostics2, involving viral genome analysis of EMB samples via quantitative PCR, to define the underlying aetiology of inflammatory cardiomyopathy. By contrast, the AHA does not recommend routine viral genome analysis for the diagnosis of (viral) inflammatory cardiomyopathy13, but this technique is discussed in a 2020 scientific statement as a potential option in cases of diagnostic uncertainty4. Further prospective studies are needed to determine and validate the role of viral genome detection in the heart in the diagnosis and management of inflammatory cardiomyopathy.
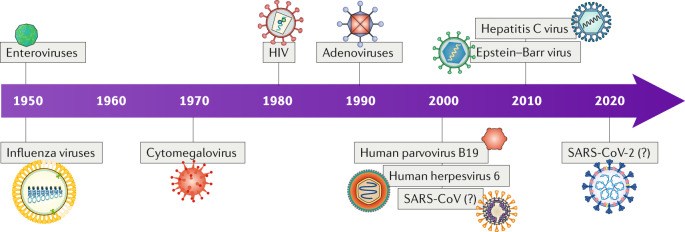
In the past two decades, B19V and HHV6 have been more frequently detected in EMB samples from patients with myocarditis than enteroviruses or adenoviruses, and approximately 30% of patients have multiple viral infections10,14. In infants, a high number of cases of acute enterovirus myocarditis has been observed in the past 5 years15–17. In general, the detection frequency of the viruses associated with inflammatory cardiomyopathy has changed over time, partly influenced by the evaluation of a broader repertoire of viruses (Fig. 1). Many viral infections have a characteristic seasonal distribution. For example, influenza viruses are prevalent during the winter months, whereas enteroviruses, including coxsackie A and B viruses, and echoviruses, are more frequently detected during summer and autumn18. However, regional climate differences can influence the seasonal variation of viral infections18. Enteroviral myocarditis predominates in male adolescent and adult patients19. Male sex is also a major risk factor for death in patients with coronavirus disease 2019 (COVID-19), which is caused by SARS-CoV-2 infection20, suggesting that the outcome of virus-associated heart disease might depend on differences in the immune responses between women and men21,22.
Fig. 1. Prominent viruses associated with inflammatory cardiomyopathy over time.
Over the years, the number of recognized viruses associated with inflammatory cardiomyopathy has grown. This evolution is partly influenced by the intentional detection of a broader repertoire of viruses over time as well as by the occurrence of novel viruses or virus genotypes in the heart. The association between severe acute respiratory syndrome coronavirus (SARS-CoV) and SARS-CoV-2 and inflammatory cardiomyopathy is not yet clear. ‘(?)’ denotes unclear, needing further investigation; HIV, human immunodeficiency virus. Based on data from ref.308.
Adenoviruses and enteroviruses
Enteroviruses, most commonly coxsackie B viruses, and some adenoviruses are established causes of acute myocarditis and inflammatory cardiomyopathy18. These viruses infect cardiomyocytes by binding to a common transmembrane receptor (the coxsackievirus and adenovirus receptor (CAR)23) and can thereby induce direct myocardial injury, including cytoskeletal disruption24, and trigger an uncontrolled immune response even after viral clearance. These viruses are examples of cytolytic viruses, which trigger myocarditis by inducing viral replication inside the host cell followed by lysis of the cell for viral release. Persistence of adenoviruses and enteroviruses in the myocardium leads to LV dysfunction, poor clinical outcomes and increased mortality in these patients10,25. However, about 50% of patients with enterovirus-induced or adenovirus-induced myocarditis completely recover without residual injuries, resulting in healed myocarditis26. Patients carrying the CCR5Δ32 deletion (which results in deficiency of CC-chemokine receptor 5 (CCR5)), either in heterozygosity or homozygosity, showed spontaneous clearance of enterovirus infection compared with patients carrying the wild-type CCR5 (ref.25), a finding that accentuates the importance of the genetic background on disease progression and outcome. Furthermore, mutant strains of coxsackievirus B3 (CVB3) with 5′-terminal deletions in their genomic RNA have been isolated from a patient with idiopathic dilated cardiomyopathy (DCM), a mutation that has been suggested as a possible mechanism of viral persistence in the heart27. Indeed, a later study showed that persistent forms of group B enteroviruses harbour a 5′-terminal deletion in their genomic RNAs and that these viruses can impair cardiomyocyte function through the proteolytic activity of viral proteinase 2A in patients with unexplained DCM28. Beyond providing a potential mechanism for viral persistence, this study suggests that conventional PCR might not be capable of detecting these mutant viruses29.
Parvovirus B19
B19V infection is associated with a broad range of clinical manifestations. B19V infections are usually mild and self-limiting, but can also induce severe septic and haematological complications. B19V can cause acute cardiac infection during severe viraemia30, but B19V infection can also persist in the heart with virus reactivation episodes6. This vasculotropic virus can enter endothelial cells and trigger the release of pro-inflammatory cytokines mediated by the toxic, non-structural viral protein NS1 (ref.31). B19V infection of endothelial cells can thereby induce cardiomyocyte apoptosis, as shown in vitro in B19V-infected endothelial cells co-cultured with cardiomyocytes32. Sustained severe cardiac inflammation, especially in the setting of acute B19V infection, might lead to cardiomyocyte necrosis33. How B19V persistence in the heart affects clinical outcomes is still under discussion34,35. The aetiological role of B19V infection in the development of myocarditis is also unclear, given that B19V is often found in myocardial autopsy samples from individuals without myocarditis or DCM36,37. This observation suggests that the presence of B19V DNA in cardiac tissue might indicate in most cases that B19V is a non-specific bystander of myocarditis rather than the main pathogen causing the disease38. Only the presence of high copy numbers of B19V DNA in cardiac tissue (>500 viral DNA copies per microgram of cardiac DNA) is currently considered to be related to myocarditis39. Beyond a high B19V DNA copy number in the heart40,41, the presence of active replicating B19V with detectable viral RNA42, as well as the co-presence of lymphotropic viruses (such as HHV6)40, is thought to be related to myocarditis, but this finding needs to be further investigated32. The majority of EMB samples from patients with acute myocarditis or inflammatory cardiomyopathy have low copy numbers of B19V DNA, which raises the question about the aetiopathogenic role of persistent B19V infection as a relevant trigger of chronic inflammatory heart disease.
Herpesviridae
Viruses belonging to the Herpesviridae family (such as Epstein–Barr virus, HHV6 and cytomegalovirus) can have lifelong persistence in the body. Epstein–Barr virus was found to induce a severe, chronic active infection of CD8+T cells in the myocardium in a patient with ongoing perimyocarditis43. The most prevalent cardiac herpesvirus, HHV6, which also infects T cells, is divided into the subgroups HHV6A and HHV6B. Interestingly, the HHV6 genome can be integrated into the DNA of somatic cells or germ line cells44. Whether the integrated HHV6 copies can be reactivated and induce myocarditis is still unclear.
HIV, hepatitis C virus and influenza A and B viruses
Myocarditis associated with HIV45, HCV46 or influenza virus47 infections has been suggested to result from immune-mediated effects. The persistence of HCV infection and development of DCM have been linked to the genetic background of the patients, with HLA-DPB1*0901 and HLA-DRB1*1201 alleles being more prevalent in these patients46.
Coronaviridae
Coronaviruses, belonging to the Coronaviridae family, are classified into four groups, Alphacoronavirus, Betacoronavirus, Gammacoronavirus and Deltacoronavirus, of which Alphacoronavirus and Betacoronavirus are known to cause infection in humans48. Different members of Coronaviridae constantly circulate in the human population, usually causing mild respiratory diseases49. By contrast, MERS-CoV, SARS-CoV and SARS-CoV-2 can be transmitted from animals to humans to cause severe respiratory diseases50. To date, older age (>60 years), male sex and presence of comorbidities, including hypertension and obesity, are known to be the major risk factors for death in patients with COVID-19 (refs51,52). Presence of cardiac injury (defined by elevated troponin levels in plasma), increased levels of d-dimer or IL-6 in plasma, and acute respiratory distress syndrome are other strong and independent factors associated with mortality in these patients20. The suggested mechanisms of myocardial injury in patients with COVID-19 include myocardial damage by a cytokine storm triggered by an imbalanced response of T helper 1 cells (TH1 cells) and T helper 2 cells (TH2 cells)53,54, and respiratory dysfunction and hypoxaemia caused by SARS-CoV-2 infection55. Myocardial injury might also be attributable to decreased activity of the ACE2–angiotensin (1–7) axis, which has cardiovascular protective effects as a counter-regulatory element of angiotensin II signalling56. ACE2 and angiotensin (1–7) levels have been shown to be reduced in autopsy heart samples from patients with a positive test for SARS-CoV57. In addition, ACE2 is the entry receptor for coronaviruses, including SARS-CoV58 and SARS-CoV-2 (ref.59), into host cells. SARS-CoV and SARS-CoV-2 entry into the host cell requires binding of the viral spike protein to ACE2 and spike protein priming mediated by the host cell serine proteases TMPRSS2, cathepsin B and cathepsin L59,60. TMPRSS2 is present on lung cells that express ACE2, and has been shown to be essential for viral entry59. Nicin and colleagues showed that cardiac cells including cardiomyocytes, pericytes, fibroblasts, endothelial cells and leukocytes from patients with HF with reduced ejection fraction or with aortic stenosis express ACE2 (ref.61). Similar to these findings, our group analysed a single EMB sample from a patient with DCM and found that ACE2 is mainly expressed in cardiomyocytes, pericytes and fibroblasts, although these cardiac cells did not express TMPRSS2 (N.H, H.M., C.T., S.V.L., unpublished observations). SARS-CoV-2 has also been detected in macrophages in cardiac tissue, which suggests that SARS-CoV-2 can reach the heart during transient viraemia or through infiltration of infected macrophages into the myocardium62. Furthermore, presence of viral elements within endothelial cells and an accumulation of inflammatory cells in the myocardium, with evidence of endothelial and inflammatory cell death indicative of endotheliitis, has been reported63. So far, the classic type of acute lymphocytic myocarditis or lymphocytic inflammatory cardiomyopathy has not been detected in patients with COVID-19 (ref.12). Further insights into SARS-CoV-2 infection and myocardial damage are needed for the appropriate classification of the associated heart disease.
Knowledge gaps and future directions
Improve viral detection methods, given that current diagnostic methods have low sensitivity for viral genome detection in heart samples.
Adopt next-generation sequencing (NGS) and metagenomics approaches that allow unbiased pathogen detection64 to improve the accuracy of diagnosis, given that knowledge about mutant viruses and ‘new’ viruses associated with inflammatory cardiomyopathy is lacking.
Understand the diagnostic distinction between active versus persistent and/or latent viral cardiac infection.
Understand the pathogenic and prognostic importance of viral load.
Understand the role of the patient genetic background and sex on the progression and outcome of viral myocarditis.
Develop registries to assess the presence and type of viruses in acute myocarditis versus chronic inflammatory cardiomyopathy and paediatric versus adult patient populations.
Develop vaccines against viruses related to myocarditis.
Understand the pathogenic mechanisms of SARS-CoV2-associated heart disease.
Role of immune cells
Our understanding of the role of immune cells in inflammatory cardiomyopathy is evolving. The importance of immune cells in the pathogenesis of viral myocarditis and viral inflammatory cardiomyopathy has been demonstrated in experimental mouse models65.The pathogenic process of viral inflammatory cardiomyopathy can conceptually be divided into three phases: an acute phase of viral entry into the cell and activation of the innate immune response (which can last 1–7 days), a subacute phase with activation of the adaptive immune response (which can last 1–4 weeks), and a chronic phase that can last from months to years, in which delayed or ineffective viral clearance together with chronic inflammation and cardiac remodelling can lead to DCM66.
Upon infection, the innate immune response is activated. Innate immune cells, as well as cardiac cells including cardiomyocytes, are activated via recognition by pattern recognition receptors, including Toll-like receptors (TLRs)67 and nucleotide-binding oligomerization domain-like receptors68,69, of specific molecular patterns of pathogens (termed pathogen-associated molecular patterns (PAMPs)) and patterns released from endogenous damaged cells (termed damage-associated molecular patterns (DAMPs)), such as released ATP, S100A8 and S100A9 (ref.70). The type of pattern recognition receptor and downstream signalling can differ depending on the pathogen or DAMP. The activated innate immune cells and cardiac cells release cytokines, chemokines, interferons and alarmins, leading to further activation and homing of innate immune cells to the heart, including mast cells, neutrophils, dendritic cells, monocytes and macrophages71. Monocytes and macrophages are the main inflammatory cell subsets found in human and experimental myocarditis71. Although activation of the innate immune response in the heart is beneficial to the host owing to its antiviral effects, excessive or persistent activation of the innate immune system can lead to an exaggerated and/or chronic inflammatory process that triggers myocardial destruction and remodelling, culminating in cardiac dysfunction72.
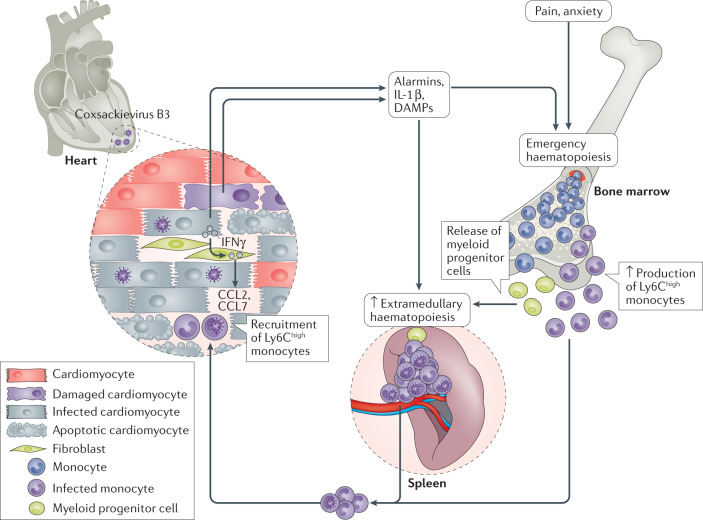
The pain, anxiety and released danger signals (including alarmins and IL-1β) triggered by the cardiac damage induce emergency haematopoiesis in the bone marrow, leading to medullary monocytopoiesis73 (Fig. 2). Monocytes and myeloid progenitor cells leave the bone marrow and myeloid progenitor cells migrate to the spleen, where extramedullary monocytopoiesis takes place74. Consequently, the pool of pro-inflammatory monocytes in the spleen is replenished and can be mobilized to the damaged heart. The homing of immune cells, mainly monocytes, from the spleen into the heart (the so-called cardiosplenic axis) has been particularly assessed in the context of ischaemic heart disease75,76. Our understanding of the importance of the cardiosplenic axis in inflammatory cardiomyopathy and as a target to modulate the trafficking of immune cells to the heart77,78 in inflammatory cardiomyopathy stems from findings in mice with CVB3-induced myocarditis. In this mouse model, blunting the CCR2–CCL2 axis, which is involved in the recruitment of pro-inflammatory Ly6Chigh monocytes to the heart, attenuates myocarditis77–80. By contrast, blockade of the CX3C chemokine receptor 1 (CX3CR1)–CX3C chemokine ligand 1 (CX3CL1) axis, which is involved in the recruitment of anti-inflammatory monocytes, worsens CVB3-induced myocarditis81. Of note, the spleen is a target organ of CVB3, with mouse splenic B cells, CD4+ TH cells and macrophages and monocytes expressing αMβ2 integrin (also known as Mac 1) as the target cells for CVB3 (ref.82). In humans, monocytes have also been identified as target cells of CVB3 (ref.83). Therefore, homing of CVB3-infected immune cells into the heart can further contribute to cardiac viral infection and chronic inflammation78,80,84. Modulation of the cardiosplenic axis might help to block this process; for example, with therapies that reduce monocytopoiesis, such as IL-1β inhibitors74, or with transfer of regulatory T cells (Treg cells)80 and mesenchymal stromal cells69,78,85, which block the recruitment of pro-inflammatory monocytes into the heart.
Fig. 2. Cardiosplenic axis in coxsackievirus B3-induced myocarditis.
In the heart, coxsackievirus B3 infection of cardiomyocytes leads to cell damage and death and the release of IL-1β and damage-associated molecular patterns (DAMPs), which trigger the recruitment and activation of cells from the innate immune system. Pain, anxiety and the release of danger signals into the systemic circulation trigger emergency haematopoiesis in the bone marrow, leading to medullary monocytopoiesis as well as release of myeloid progenitor cells into the circulation. Myeloid progenitor cells then migrate to the spleen, where extramedullary monocytopoiesis takes place to replenish the pool of pro-inflammatory Ly6Chigh monocytes, which can be rapidly mobilized to the damaged heart. In the heart, IFNγ released by infected cardiomyocytes boosts the production by fibroblasts of the pro-inflammatory C-C motif chemokines CCL2 and CCL7, which promote the homing of Ly6Chigh monocytes to the heart. Given that the spleen is a target organ of coxsackievirus B3 and monocytes target cells of coxsackievirus B3, the recruited Ly6Chigh monocytes might be infected with coxsackievirus B3 and thereby transport the virus into the heart, further contributing to the viral infection. Activation of the innate immune system in the heart is beneficial for its antiviral effects but excessive or persistent activation can lead to exaggerated and/or chronic inflammation that triggers myocardial destruction and remodelling, culminating in cardiac dysfunction.
Mast cells, natural killer cells and dendritic cells
Mast cells are among the first cells to respond to viral infection of the heart. Mast cells degranulate within 6 h of infection and produce pro-inflammatory cytokines such as tumour necrosis factor (TNF), IL-1β and IL-4. High numbers of mast cells are found in mice susceptible to autoimmune heart disease after CVB3 infection86. Viral infection also leads to the recruitment of natural killer cells87,88 and dendritic cells89, which prevent the development of myocarditis. In mice with enterovirus-induced myocarditis, signalling mediated by the activating receptor NKG2D in natural killer cells was found to protect against progression to inflammatory cardiomyopathy, leading to effective clearance of CVB3 from the heart87. Dendritic cells have been reported to accumulate in the mouse heart after viral infection of the myocardium, coincident with monocyte infiltration and loss of resident reparative, embryonic cardiac macrophages89. After ingesting dead and damaged cardiomyocytes, dendritic cells migrate to regional lymph nodes and the spleen to present antigens to naive B cells and T cells, which initiates the activation of the adaptive immune response. The relevance of dendritic cells as antigen-presenting cells has been demonstrated in mouse studies showing that cardiac dendritic cell depletion abrogated the generation of antigen-specific CD8+ T cells, promoting the progression of subclinical cardiac injury into overt HF89. Dendritic cells can also process endogenous antigens and, therefore, might also trigger autoimmune myocarditis. Indeed, dendritic cells loaded with myosin peptides have been used to develop an experimental model of myosin-induced autoimmune myocarditis90.
Neutrophils
Neutrophils are among the first lines of defence against infection. In a mouse model of CVB3 myocarditis, an early (2.5 days) and abundant mobilization and influx of neutrophils into the heart and the pancreas occur after CVB3 infection91. This mobilization occurs earlier than that of any other infiltrated innate immune cells. Depletion of neutrophils in CVB3-infected mice results in reduced myocarditis92. Neutrophils can maintain inflammation by a specific process called NETosis, which involves the formation of neutrophil extracellular traps. The role of NETosis in promoting cardiac inflammation has been shown in mice with experimental autoimmune myocarditis (EAM)93, and the severity of acute myocardial inflammation in these mice is strongly associated with neutrophil accumulation in the heart94. The alarmins S100A8 and S100A9 released by neutrophils (and monocytes) are also involved in promoting inflammatory cardiomyopathy, as demonstrated by a study showing that S100a9−/− mice were protected from the detrimental effects of CVB3 infection95. Neutrophil numbers in blood positively correlate with the level of cardiomyocyte necrosis (measured by troponin I levels in plasma) in patients with acute coronary syndrome96. Likewise, plasma levels of the S100A8–S100A9 heterodimer are increased in patients with acute myocarditis when myocytolysis occurs, compared with the levels in healthy individuals70.
Monocytes
Monocytes are crucial effector cells in myocarditis97, comprising a major proportion of infiltrating cells in the heart. Monocytes are a heterogeneous, multifunctional cell population with a critical role in the pathogenesis of myocarditis. The role of the different monocyte subsets has been investigated in mouse models of myocarditis but so far has not been addressed in patients with myocarditis. In mice, the pro-inflammatory CD115+CD11b+Ly6ChighCCR2highCX3CR1low and CD115+CD11b+Ly6CmiddleCCR2highCX3CR1low monocyte subsets (which are considered the counterparts of human classic CD14++CD16−CCR2highCX3CR1low and CD14++CD16+CCR2middleCX3CR1highCCR5+ monocytes98) infiltrate sites of cardiac inflammation and damage in response to chemokine signals99. The infiltrated monocytes differentiate into inflammatory macrophages that secrete pro-inflammatory cytokines, such as TNF and IL-6, and contribute to tissue degradation and T cell activation78,80. By contrast, CD115+CD11b+Ly6ClowCCR2lowCX3CR1high monocytes (corresponding to human non-classic CD14+CD16++CCR2lowCX3CR1high monocytes98) recruited to the inflamed cardiac tissue are more likely to differentiate into macrophages that secrete anti-inflammatory cytokines and contribute to tissue repair78,98. Cardiac fibroblasts secrete chemokines that promote the migration of monocyte subsets to the myocardium99 and also facilitate the differentiation of Ly6Chigh monocytes and Ly6Clow monocytes into macrophages in mice with myocarditis100.
T cells and B cells
T cells, and to a certain extent B cells specific for viral antigens, are critical mediators of cardiac damage in experimental models of myocarditis65,80,101–103. Activation of the T cell system is believed to be the major pathophysiological mechanism underlying autoimmune myocarditis and autoimmune inflammatory cardiomyopathy104. The presence of activated T cells is essential for the cardiac damage in virus-induced myocarditis, as shown by studies indicating that impaired T cell maturation protects against CVB3-induced myocarditis in mice101. In another study in mice with CVB3-induced myocarditis, disease severity increased in mice lacking the CD8 receptor and was attenuated in mice lacking the CD4 receptor compared with wild-type mice, suggesting that different T cell subsets have different functions in virus-induced myocarditis105. Similar findings were observed in another study in which deficiency of CD8+ T cells in mice led to increased CVB3-induced cardiac injury and chronic myocarditis, a process that was unrelated to perforin-mediated cytotoxicity106. Mice lacking T-box transcription factor TBX21 (T-bet) — which is essential for TH1 lineage differentiation and IFNγ production — are highly susceptible to autoimmune myocarditis owing to the induction of IL-17 production107. In mice with EAM, TH17 cells promote the progression to DCM102. By contrast, Treg cells, which are reduced in patients with myocarditis or DCM108, protect against the development of CVB3-induced myocarditis in mice by attenuating cardiac inflammation80,103. IL-23, which is primarily secreted by antigen-presenting cells, induces an increase in the ratio of TH17 cells to Treg cells by promoting the maturation of TH17 cells109, and is an important trigger for the initiation of autoimmune myocarditis in mice110.
Limited data are available on the role of B cells in the progression of myocarditis to DCM. Detection of infected activated B cells both in the heart tissue of CVB3-infected immunocompetent mice and in severe combined immunodeficient mice receiving splenocytes from CVB3-infected syngeneic donors84 supports the concept that B cell traffic might contribute to the maintenance of chronic inflammatory heart disease. B cells are a crucial link between the innate and adaptive immune system. In addition to antigen-specific B cell receptors, B cells also express TLRs. TLR signalling is associated with B cell activation and tolerance and with diverse pathological conditions, such as viral myocarditis and septic cardiomyopathy111. Most of the information on the role of B cells in inflammatory cardiomyopathy is derived from the identification of autoantibodies implicated in DCM111. Autoantibodies, such as those against β1-adrenergic receptor, mitochondrial components, cardiac myosin heavy chain isoforms, cardiac troponin, Na+/K+-ATPase and other heart-related proteins, might contribute to cardiac dysfunction111. Additionally, findings from a study in patients with subacute or chronic inflammatory myocarditis suggest that CD20+ B cells — which induce myocardial damage in mice by activating T cells112 and triggering monocyte mobilization113 — could have a pathophysiological role in inflammatory cardiomyopathy114.
Eosinophils
Patients with eosinophilia frequently develop cardiomyopathies115. Eosinophils are also implicated in parasite-mediated, drug-induced or hypersensitivity myocarditis with progression to DCM, as shown by studies in mice with EAM. Eosinophil-deficient mice with EAM were protected from developing DCM, whereas hypereosinophilic mice with EAM had a more rapid progression to DCM, mediated by eosinophil-derived IL-4 (ref.115). The eosinophil cationic protein derived from degranulation of eosinophils has an important role in the pathogenesis of eosinophilic myocarditis in mice116. Major basic protein, the most abundant protein in eosinophilic granules, is highly thrombogenic and contributes to the high rate of vascular thromboembolism in patients with eosinophilic myocarditis117. Treatment with mepolizumab, an antibody against IL-5 (a key mediator of eosinophil maturation and survival), was found to be effective in a patient with eosinophilic myocarditis118.
Knowledge gaps and future directions
Improve our understanding of how the immune cell response switches from host defence to host injury.
Generate data on ratios of Treg cells to TH17 cells in patients with myocarditis or inflammatory cardiomyopathy.
Understand why only some patients with viral myocarditis or inflammatory cardiomyopathy show autoimmunity or abnormal immune cell responses.
Understand why some patients with autoimmunity or abnormal immune cell responses do not develop myocarditis or inflammatory cardiomyopathy.
Improve our knowledge of the genetic and epigenetic factors involved in maladaptive immune cell responses.
Understand how to target autoimmunity or neutralize immune cell functions involved in the pathophysiology of inflammatory cardiomyopathy without an associated risk to the host.
Improve our knowledge of the role of neutrophils in viral myocarditis.
Assess whether genetic cardiomyopathies have an immune cell component.
Understand how research on cardiac immune cells from mouse models can be applied to humans.
Role of autoimmunity
The involvement of autoimmunity in inflammatory cardiomyopathy is well established. Inflammatory cardiomyopathy fulfils the Rose–Witebski diagnostic criteria for organ-specific autoimmune disease, including: presence of immune cell infiltrates and abnormal expression of HLA class II and/or adhesion molecules in the absence of viral genomes in EMB samples from both index patients and family members34,119; presence of circulating heart-specific autoantibodies in patients with inflammatory cardiomyopathy and their relatives2,120–122; availability of animal models of experimentally induced inflammatory cardiomyopathy, with or without a DCM phenotype, after immunization with specific autoantigen(s)2,123–125; and response to immunosuppression or immunomodulation in patients with virus-negative inflammatory cardiomyopathy2,126–128.
Heart-specific autoantibodies
Heart-specific autoantibodies are present in up to 60% of patients with inflammatory cardiomyopathy and their relatives2,129,130. These autoantibodies recognize many cardiac autoantigens, particularly cardiac α-myosin heavy chain (also known as myosin 6) and β-myosin heavy chain (also known as myosin 7) isoforms131. Some of these autoantibodies seem to have a direct pathogenic and/or prognostic role132,133. Immunization of animals with autoantigens that have been identified in patients with inflammatory cardiomyopathy, such as β1-adrenergic receptor, muscarinic acetylcholine receptor M2, cardiac myosin heavy chain isoforms and cardiac troponin123–125,134–136, leads to cardiac abnormalities that mimic the human disease phenotype. Passive transfer of antibodies purified from rats immunized with cardiac myosin leads to antibody deposition in the myocardium and myocyte apoptosis, producing cardiomyopathy in recipient animals125. Both antibody-mediated and cell-mediated autoimmune forms of inflammatory cardiomyopathy have been shown in animal models, but whether both forms can be found in patients is still unknown because most studies in patients with inflammatory cardiomyopathy to date have investigated humoral rather than cellular immune mechanisms.
Gene–environment interactions
Autoimmune inflammatory cardiomyopathy can occur in the context of systemic immune-mediated diseases9 or be iatrogenic (for example, induced by immune checkpoint inhibitor therapy8). Inflammatory cardiomyopathy might also have a hereditary component, as shown in a genome-wide association study in patients with DCM137. This study revealed a risk locus for idiopathic DCM encoding HLA class I and HLA class II proteins, suggesting a role for genetically driven, autoimmune inflammatory processes in the pathogenesis of idiopathic DCM137. After the MOGE(S) classification138,139, autoimmune inflammatory cardiomyopathy probably represents a common end-stage resulting from a combination of several aetiological factors in a multifactorial cascade involving gene–environment interactions140. For example, patients with myocarditis have detectable immune reactivity to both myosin 6 antigens and myosin peptide mimics derived from commensal Bacteroides species from the gut141. These findings fit with the gene–environment interaction model and suggest that targeting the microbiome of genetically predisposed patients with myocarditis might reduce disease severity and, therefore, might help prevent the potentially lethal consequences of inflammatory cardiomyopathy141.
Knowledge gaps and future directions
Understand how the genetic background influences the susceptibility to immune-mediated disease in patients with inflammatory cardiomyopathy, including the role of HLA genotyping for disease management.
Improve our understanding of autoimmunity triggers (for example, viruses, drugs and other environmental agents).
Improve the definition of potential humoral predictors (such as distinct autoantibody specificities, pathogenic immunoglobulin class and IgG subclass, autoantibody titre and new relevant autoantigens) and cellular predictors (immune phenotype of circulating T cells, in particular TH1, TH17 and Treg cells, and of myocardium-infiltrating cells, such as T cells, B cells and myeloid cells) of the risk of progression to HF, death or heart transplantation, and of spontaneous or immunosuppressive therapy-induced recovery in patients with inflammatory cardiomyopathy.
Assess the potential role of peripheral and myocardial levels of inflammatory and pro-fibrotic cytokines (for example, IL-1, IL-6, IL-17, IL-23 and TGFβ) in patient risk stratification.
Understand how to distinguish a beneficial immune reaction to clear a pathogen from a pathogenic autoimmune reaction.
Assess the effect of gut microbiome modulation on the course of inflammatory cardiomyopathy.
Translational research models
Several animal models of myocarditis have been developed and tested that cover different underlying aetiologies, including virus-induced myocarditis, Trypanosoma cruzi-induced myocarditis and autoimmune myocarditis93–95. The approaches include the use of pathogens as well as engineered models such as transgenic mice (for example, Il5-transgenic mice, which develop eosinophilic myocarditis)115. The advantage of infectious models is that they more closely reflect the physiological processes of the human disease than models of autoimmune myocarditis, given that infectious models couple the immune response involved in pathogen clearance with autoimmune responses. Conversely, autoimmune myocarditis models facilitate the investigation of the progression of myocarditis to inflammatory cardiomyopathy and DCM and the involvement of specific components of the immune system in the disease process142 (for example, use of Pd1−/− mice to assess the role of the immune checkpoint PD1 (ref.136)).
The mouse model of CVB3-induced myocarditis was established 60 years ago143 and has become the standard for the evaluation of virus-induced myocarditis given its similarity to the myocardial injury observed in humans144. However, these mice have severe pancreatitis and a high systemic inflammatory response and, therefore, mainly mimic CVB3 infection in infants rather than in adult patients145. A study in a mouse model of CVB3 infection targeted to the heart and with attenuated virulence in the pancreas indicates that the systemic inflammatory response, rather than the cardiac damage induced by the infection, underlies the cardiac dysfunction observed in the classical CVB3 myocarditis model. Further refinement of this new model is still needed to allow representation of CVB3-induced myocarditis in adult patients. In addition, to improve the clinical relevance of animal models of viral myocarditis, models of B19V-induced myocarditis need to be developed given that B19V is currently the most frequently detected virus in EMB samples from patients with myocarditis10. Data indicate that this vasculotropic virus33,39 induces endothelial damage32,146 and that patients with B19V infection have diastolic dysfunction147 and elevated levels of circulating endothelium-derived microparticles148. However, whether persistent B19V infection is a bystander or has a causal role in inflammatory cardiomyopathy is unclear32. Humanized mouse models of B19V infection will shed light on the aetiological role of B19V infection in the development of inflammatory cardiomyopathy.
The influence of the mouse strain149 and sex21 on the immune status and, consequently, on the model of myocarditis is well established. However, the role of housing conditions on the outcome of myocarditis in mice remains under-studied. Mice are kept in ‘pathogen-free’ conditions in animal facilities and, consequently, have a predominantly naive immune system, which contrasts with the experienced immune system of patients. Other housing factors can also affect the clinical relevance of the myocarditis model; for example, mice exposed to bisphenol A leached from plastic cages and water bottles had increased myocarditis and pericarditis compared with mice housed in glass cages that drunk out of glass water bottles150. This finding clearly highlights that to improve the translation of experimental results to the clinical setting, advanced animal models are needed that better represent the human conditions, and which take into account not only the immune experience of the animal model, but also the environmental factors common to a Western lifestyle, such as exposure to plastics, intake of processed foods, antibiotic usage, physical inactivity and HF medications150–152. The complexity of the immune system and its involvement in inflammatory cardiomyopathy further underscore that experiments in animal models are essential to understand the fundamental mechanisms underlying the pathogenesis of this disease. In vitro models, including inducible pluripotent stem cell (iPSC)-derived cardiomyocytes, have been developed as antiviral drug screening platforms153. These cells could be used to test the efficacy of antiviral agents in counteracting the direct cytotoxic effects of the virus. However, the use of iPSC-derived cardiomyocytes in vitro does not mimic the in vivo conditions for toxicity testing and does not take into account the systemic immune effects associated with inflammatory cardiomyopathy154.
Knowledge gaps and future directions
Develop advanced animal models that more closely represent the human disease process, such as immune experience and exposure to environmental factors.
Develop in vitro models that allow the study of immune cell interactions.
Diagnosis and prognosis
Clinical scenarios
The typical symptoms and signs at presentation in patients with acute myocarditis include chest pain, dyspnoea, fatigue, palpitations, syncope and cardiogenic shock4. Acute myocarditis can also present as sudden cardiac death, accounting for approximately 10% of deaths from sudden cardiac death in young individuals (aged <35 years)155. Prodromal manifestations, including fever, gastrointestinal disorders and influenza-like symptoms, are recorded in up to 80% of patients with acute myocarditis in the weeks preceding the acute phase3. A multicentre study showed that the type of presentation — specifically, complicated myocarditis (LV ejection fraction (LVEF) <50% on first echocardiography, sustained ventricular tachycardia or haemodynamic instability at presentation) versus uncomplicated myocarditis — is associated with outcomes in patients hospitalized with suspected acute myocarditis (cardiac death or heart transplantation at 5 years was 14.7% versus 0%)3. In patients with preserved LVEF, assessment of late gadolinium enhancement (LGE) distribution patterns on cardiac MRI can improve patient risk stratification156,157. Among patients with fulminant myocarditis (patients who present with cardiogenic shock needing inotropes and/or mechanical circulatory support (MCS)), the histological subtype subtending the myocarditis, including giant-cell and eosinophilic myocarditis, is independently associated with increased mortality158.
Finally, inflammatory cardiomyopathy can be the first presentation in patients with HF symptoms and can be the result of a delayed diagnosis of acute myocarditis. Therefore, defining the time of cardiac symptom onset is crucial. A mild elevation of troponin levels in plasma that is disproportionate to the severity of the LVEF impairment and associated with a dilated left ventricle at presentation is suggestive of inflammatory cardiomyopathy rather than acute myocarditis158. Patients presenting with inflammatory cardiomyopathy are often haemodynamically stable owing to a gradual and unrecognized attenuation of LV systolic dysfunction and remodelling. However, the recovery rate of patients with complicated myocarditis is only 50%159. Currently, no established clinical markers are available to characterize the prognosis of patients with inflammatory cardiomyopathy.
Knowledge gaps and future directions
Determine the factors involved in the transition from acute myocarditis to chronic inflammatory cardiomyopathy in patients (an issue that has been addressed by few clinical studies160). Establish whether chronic inflammation, specific infection or both in the myocardium is critical in this transition or whether other mechanisms have a role, and determine the real incidence of the transition from acute myocarditis to chronic inflammatory cardiomyopathy among adults.
Develop clinical research tools, including prospective registries of diverse patient populations, aimed at identifying patients at low risk versus those at high risk of developing chronic inflammatory cardiomyopathy. The accuracy of the risk stratification should be confirmed on the basis of uncomplicated versus complicated clinical presentation at the time of hospitalization3 or on the basis of immunohistological34 or viral analysis data5,130 from EMB samples.
Improve EMB-based diagnosis and identify innovative, non-invasive approaches for the diagnosis of myocarditis.
Develop a multimarker approach including cardiac MRI to enable prognosis independently of the use of the LVEF.
Identify specific biomarkers to guide diagnosis and treatment decisions.
Imaging
Currently, the non-invasive gold-standard method for the diagnosis of myocarditis is cardiac MRI (class I recommendation, level of evidence C in the 2012 ESC guidelines161). In 2018, consensus guidelines recommended the addition of T2-weighted cardiac MRI to the pre-existing Lake Louise criteria (LLC) for the diagnosis of myocarditis162. The 2018 LLC showed better diagnostic performance than the original criteria owing to increased sensitivity163. T1 and T2 mapping have added further diagnostic information and accuracy to cardiac MRI164–169. In addition, parametric cardiac MRI modalities, such as T1 and T2 mapping, can facilitate the objective assessment of myocardial inflammation or diffuse myocardial fibrosis. However, the diagnostic accuracy of cardiac MRI might vary according to the clinical presentation and extent of cell necrosis in patients with EMB-proven acute myocarditis170. Cardiac MRI diagnostic sensitivity is high for infarct-like presentations, low for cardiomyopathy-like presentations and very low for arrhythmia presentations170. Furthermore, the type of myocarditis (that is, the specific immune cell infiltration and the underlying aetiology) cannot be established with the use of cardiac MRI. In routine clinical practice, many patients have borderline ‘normal’ T1 and T2 mapping values, which potentially leads to false-negative exclusion of myocarditis as a diagnosis. Data about the diagnostic and prognostic value of ‘borderline’ results are not available.
Cardiac MRI also allows the objective evaluation of myocardial deformation (strain), either with the use of new post-processing software tools on existing cine images or by acquisition of specific sequences (such as displacement encoding with stimulated echoes (DENSE) MRI or fast strain-encoded MRI). Strain can be evaluated in different layers of the myocardium and also in the right ventricle and left atrium171. Even years after acute myocardial inflammation, patients might present clinically with dyspnoea; by using strain analysis, diastolic impairment can be detected either by echocardiography or with a more complete assessment of the heart by cardiac MRI156,157,172. However, data on the combination of different cardiac MRI parameters and their added diagnostic and long-term prognostic value in patients with myocardial inflammation are still rare. In addition, current recommendations are based on classic assessment of cardiac MRI parameters and do not take into account advanced cardiac MRI parameters such as parametric mapping and strain. Moreover, cardiac MRI protocols for acute or chronic myocarditis specific for the different magnetic field strengths (1.5 T and 3.0 T) and for MRI scanners from different vendors need to be validated, and proof of the prognostic value of these protocols in large, multicentre trials is necessary to provide the basis for guideline recommendations. Cardiac MRI is also a potent tool for therapy monitoring in selected patients173 (Table 2). However, data on the best timing to perform follow-up cardiac MRI for therapy control in patients with myocardial inflammation are lacking.
Table 2.
Cardiac MRI versus endomyocardial biopsy in routine clinical practice
| Clinical scenario | Characteristics | Cardiac MRI | Endomyocardial biopsy |
|---|---|---|---|
| Suspected acute myocarditis (disease onset ≤30 days) | Cardiogenic shock | − | ++a,b |
| Complicated, impaired ejection fraction or arrhythmias | ++ | ||
| Uncomplicated, stable, preserved ejection fraction and no arrhythmias | + | −/(+)a,c | |
| Suspected inflammatory cardiomyopathy (disease onset > 30 days) | Persistent heart failure symptoms in preserved EF | ++c | +a,b |
| Persistent heart failure symptoms and reduced EF despite optimal medical therapy | ++c | ++a,b | |
| Significant arrhythmias despite optimal medical therapy | ++c | ++a,b | |
| Suspected acquired dilated cardiomyopathy with a disease onset of months | Impaired EF and dilated LV of unknown etiology | ++ | +/++a,b |
| Ongoing clinical management | NA | + | −/+d |
−, not recommended; +, recommended; ++, highly recommended; NA, not applicable. aFor differential diagnosis; specific therapy decision and selection. bCardiac MRI or electroanatomical mapping-guided endomyocardial biopsy in selected patients. cA negative result does not exclude the persistence of low-grade cardiac inflammation in patients with severe cardiac complaints, refractory to therapy. dOption in selected patients who do not respond to therapy, to re-evaluate the diagnosis or to exclude viral reactivation. Based on data from refs71,159,162,307.
Although the value of cardiac MRI in acute myocarditis has been widely proven, the technology continues to be under-used, partly owing to the limited availability of cardiac MRI in standard clinical practice174–176 (Table 2). Potential solutions to overcome this limitation might be the use of mobile cardiac MRI units associated with expert centres for interpretation, diagnosis and therapy recommendation. Additional essential factors to increase the use of cardiac MRI are appropriate training and increased awareness of the utility of cardiac MRI for the diagnosis of myocardial inflammation. Another factor limiting the widespread use of cardiac MRI in clinical practice are the difficulties for the reimbursement of cardiac MRI associated costs in many regions worldwide, even though cost-effectiveness and value for the health-care system have been proven for various clinical indications177–179. In addition, the utility of cardiac MRI is often limited in patients with haemodynamic instability owing to fast or irregular heart rates and mechanical ventilation2,162. In these patients and in patients with myocarditis presenting as acute HF with high-grade heart block, symptomatic ventricular tachycardia or shock, an EMB-guided approach is recommended (AHA and ESC class I recommendation, level of evidence B)2,13,180 (Table 2). New real-time cardiac MRI protocols for the assessment of anatomy, function and flow in these patients are under development181,182. Likewise, an EMB-based diagnostic approach is needed in patients in whom the onset of the disease occurred >3 months previously and the diagnostic accuracy of cardiac MRI is low162,168,183, such as in patients with HF of >3 months duration that is associated with a dilated left ventricle.
Patterns of LGE, the potential progression of LGE and the extent of focal fibrosis on cardiac MRI have been shown to predict the risk of hospitalization and adverse cardiovascular events in patients with suspected myocarditis184–186. Even in patients with myocarditis who seem to show clinical improvements, LGE can increase on imaging and should be considered as an indicator of the risk of adverse cardiovascular events187. Use of LGE parameters could also be an option for risk stratification in patients with cardiac sarcoidosis, independently of LVEF188. 18F-Fluorodeoxyglucose (18F-FDG) uptake is a quantifiable surrogate parameter of increased glucose metabolism, which is a hallmark of inflammation. Therefore, 18F-FDG PET is a valuable tool for diagnosis and monitoring treatment response in patients with cardiac sarcoidosis. In selected patients with myocarditis, the use of 18F-FDG PET in addition to cardiac MRI might provide complementary information on disease progression189.
New technologies to assess intraventricular pressure gradients or LV kinetic energy by 4D flow cardiac MRI are under development. Evaluation of LV kinetic energy or haemodynamic forces will potentially allow better characterization of patient populations at various stages of HF190 by providing quantitative measures, and might be of interest for diagnosis and therapy control in patients with myocardial inflammation.
The clinical value of vasodilator stress cardiac MRI for the quantitative assessment of microvascular disease has been proven. However, data about the use of this modality in patients with acute cardiac inflammation are sparse. Together with the quantitative assessment of myocardial strain, diffuse myocardial fibrosis and oedema by parametric mapping, cardiac MRI data might facilitate the generation of a potential objective cardiac MRI-based inflammation score.
Knowledge gaps and future directions
Current imaging methods have low predictive value in patients with chronic inflammatory cardiomyopathy and ongoing low-grade inflammation.
Current imaging methods cannot detect the underlying aetiology of myocardial inflammation (for example, viral persistence), with the exception of PET for cardiac sarcoidosis.
Develop standardized protocols specific for myocarditis across different cardiac MRI field strengths and scanners from different vendors.
Generate data on advanced cardiac MRI parameters and their predictive value (including myocardial strain, parametric T1 and T2 mapping, 4D flow and LV kinetic energy, and haemodynamic forces) in patients with myocarditis.
Assess for microvascular disease with the use of stress cardiac MRI in patients with suspected acute myocardial inflammation.
Perform studies to assess the value of combining different imaging modalities for diagnosis and therapy control in patients with acute or chronic myocarditis.
Evaluate cost–benefit, cost-effectiveness and budget-impact models that include appropriate imaging methods for the management of acute or chronic myocardial inflammation.
Develop a cardiac MRI inflammation score that is based on conventional and advanced cardiac MRI parameters.
Endomyocardial biopsy
EMB is the gold-standard method for the diagnosis of acute or chronic inflammatory heart diseases. Right ventricular (RV) and LV EMB are well accepted as standard procedures in the diagnostic work-up of patients with myocarditis, because biopsies are often the only method that allows the identification of the underlying aetiology of cardiac inflammation2,159,191–193. A 2013 ESC position paper advocates the characterization of cardiac inflammation using immunohistochemistry and viral genome analysis with quantitative PCR (real-time PCR and nested PCR with reverse transcription) for the diagnosis of myocarditis and the selection of therapeutic regimens2,159. Immunohistochemistry with the use of a panel of monoclonal and polyclonal antibodies (including anti-CD3, anti-CD68 and anti-HLA-DR antibodies) is recommended for the characterization of the inflammatory infiltrate194. Compared with the histological Dallas criteria, immunohistochemistry is more sensitive195 and has prognostic value34,191. Given that cardiac inflammation often has a patchy distribution, analysis of at least five or six tissue samples is suggested to reduce the EMB sampling error196 (Fig. 3). Furthermore, given the focal nature of many viral infections, two or three EMB samples are also recommended for the detection of viral nucleic acids to avoid false-negative results197,198 (Fig. 4). Despite this knowledge and the risk of under-diagnosing patients, the acceptance by clinicians of the need to take more than four EMB samples during a routine clinical procedure is often low, owing to the fear of complications such as ventricle perforation. Therefore, combined strategies involving EMB and imaging or electroanatomical mapping could be helpful in overcoming this problem. T2-mapping cardiac MRI might facilitate the identification of patients who would potentially benefit from undergoing EMB for therapeutic decision-making199,200 (Table 2). Moreover, use of 3D electroanatomical voltage mapping201–204, cardiac MRI200 or 18F-FDG PET205 to guide EMB has been found to be helpful for increasing the sensitivity and specificity of the conventional EMB approach, by reducing sampling errors and allowing a deeper insight into different (local) pathologies206 (Fig. 5).
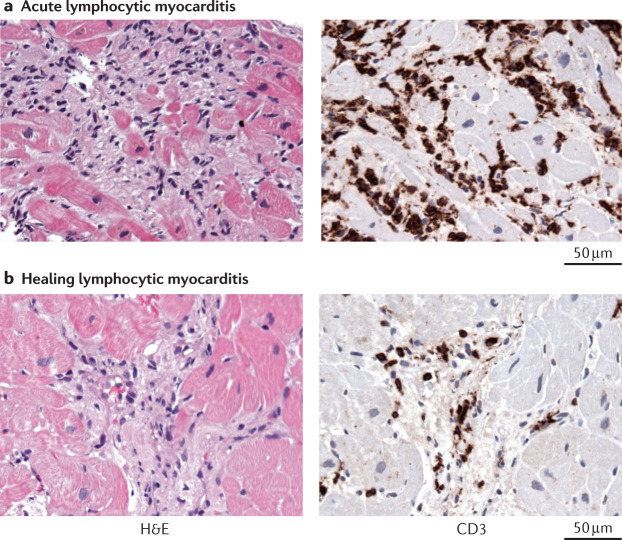
Fig. 3. Diagnosis of lymphocytic myocarditis.
Acute and healing lymphocytic myocarditis is diagnosed with histology and immunohistology of endomyocardial biopsy samples. a | Acute lymphocytic myocarditis caused by enterovirus A71 infection. Histology image showing cardiomyocyte necrosis (as revealed by the haematoxylin and eosin (H&E) staining in the left panel)) and immunohistology image showing diffuse infiltration of CD3+ T cells (as shown by anti-CD3 antibody staining (brown) in the right panel). b | Healing lymphocytic immune-mediated myocarditis. Histology image showing fibrosis but no cardiomyocyte necrosis (left panel) and immunohistology image showing the presence of infiltrated CD3+ T cells (right panel). All images ×400.
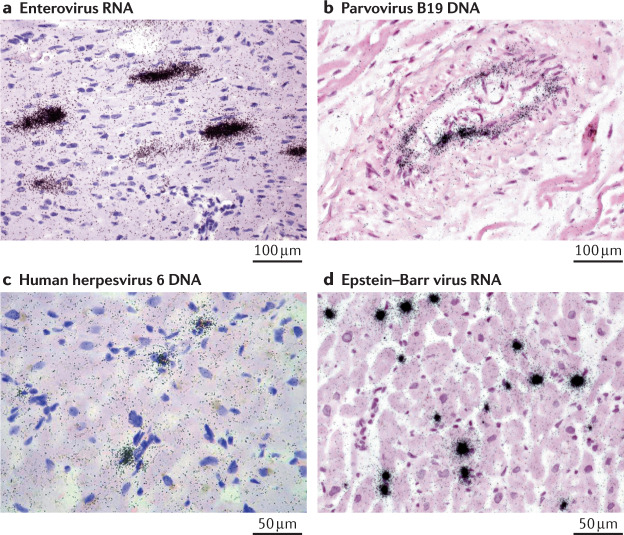
Fig. 4. Visualization of viral nucleic acids in acute myocarditis.
Viral nucleic acids in heart tissue samples from patients with acute myocarditis can be detected with radioactive in situ hybridization (black spots). Cell nuclei (purple) and cell cytoplasm and extracellular matrix (pink) are visualized with haematoxylin and eosin staining. Enteroviruses (panel a) infect and lyse cardiomyocytes, parvovirus B19 (panel b) infects endothelial cells, and human herpesviruses (panel c) and Epstein–Barr viruses (panel d) replicate in immune cells. Panels a and b ×400, panels c and d ×630.
Fig. 5. Electroanatomical voltage mapping to guide endomyocardial biopsy.
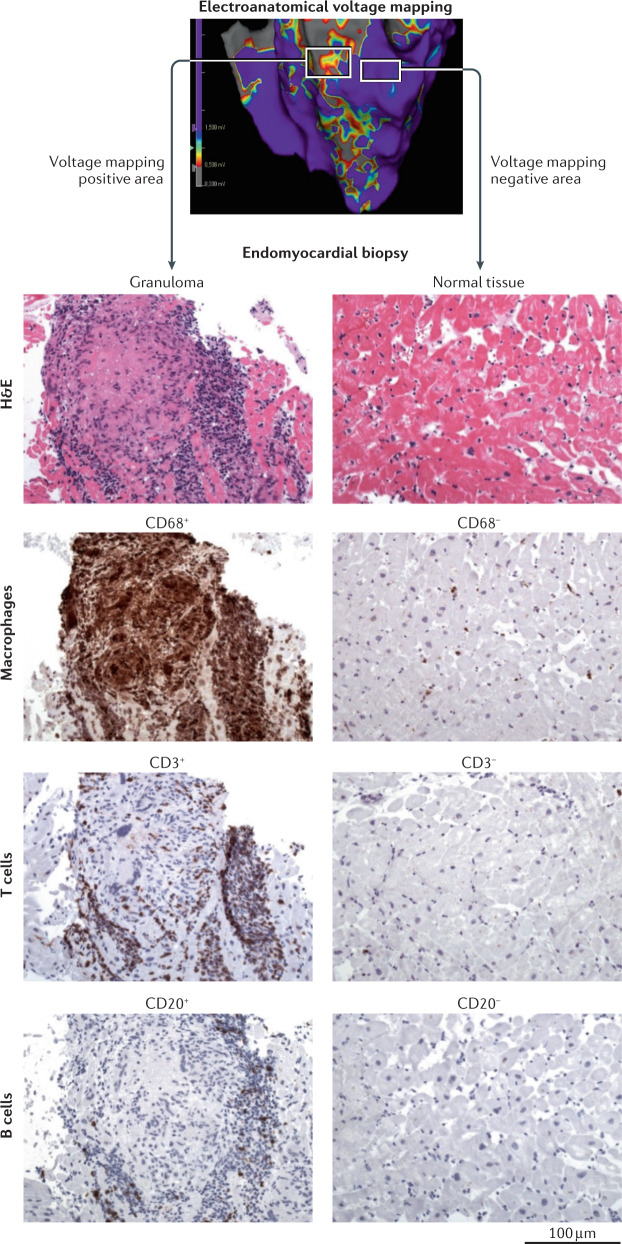
Identification of a myocardial scar area with the use of 3D electroanatomical voltage mapping in a patient with suspected cardiac sarcoidosis. The left square in the top panel marks a scar area identified by the presence of low voltages (red and yellow). Subsequent analysis of endomyocardial biopsy samples from this region identified the presence of a sarcoid granuloma in the scar area, visualized with haematoxylin and eosin staining (H&E), with the presence of CD68+ cells (macrophages), CD3+ cells (T cells) and CD20+ cells (B cells), as revealed by antibody staining (brown). By contrast, analysis of endomyocardial biopsy samples from a non-scar area, identified by high voltages (purple) in electroanatomical voltage mapping, showed normal tissue structure and no infiltration of immune cells. All histology images ×100.
At present, comprehensive datasets of the inflamed heart can be derived only from sophisticated molecular analyses of heart tissue samples, mainly obtained by EMB. Beyond the classic (immuno)histological and virological analyses, gene expression profiling has been suggested to contribute to the differential diagnosis of idiopathic giant-cell myocarditis and cardiac sarcoidosis207. Targeted biopsies of the inflamed heart are required to study the role of omics technologies, such as genomics, epigenomics, proteomics and metabolomics, in diagnosis and drug discovery, which have to be correlated with state-of-the-art methods including histology, immunohistochemistry and molecular virology. A study using global proteome profiling has shown that inflammatory heart disease is associated with extracellular matrix remodelling and a decrease in the levels of proteins involved in carbohydrate metabolism, the tricarboxylic acid cycle and oxidative phosphorylation208. Mass spectrometry analysis of EMB samples, which allows region-specific evaluation of protein profiles, allowed patient clustering to discriminate patients with from those without cardiac inflammation209. The characterization of patient epigenetic profiles combined with other genetic approaches including NGS can shed light on complex gene networks in patients with myocarditis who develop HF210. Use of NGS has led to the identification of an increasing number of gene variants and mutations associated with the risk of heart diseases such as DCM. With the expanded use of genome sequencing, the identification of further high-risk gene variants seems likely, which will thereby improve the clinical decision-making process and provide insights into the pathogenetic mechanisms of inflammatory cardiomyopathy. Successful integration of these omics techniques into existing diagnostic algorithms will contribute to a more sensitive, specific and cost-effective approach for the personalized treatment of patients with inflammatory cardiomyopathy211. Use of NGS will also facilitate the detection of so-far-unknown pathogenic, cardiotropic agents, such as DNA and RNA viruses, in myocarditis212.
Heart tissue samples have a considerable cell heterogeneity, which makes establishing the contribution of specific cell types to the pathogenesis of myocarditis difficult. New methods such as single-cell RNA sequencing of cardiomyocytes offer great opportunities for studying cardiac pathology at single-cell resolution213. Single-cell RNA sequencing also opens up the field of cardio-immunology, as shown by a study that mapped the immune cell activation profile in the heart in a mouse model of HF214.
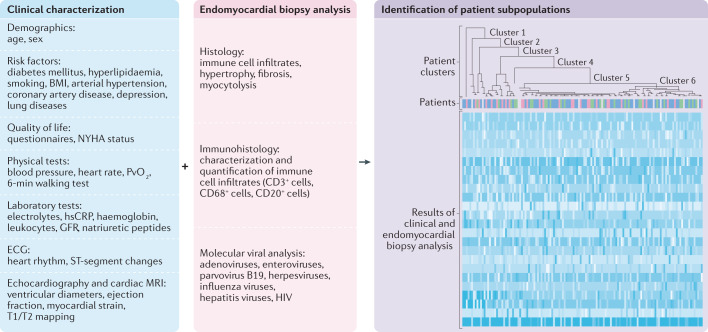
The profound characterization of EMB samples via phenomics (such as proteomics, viral diagnosis and immune cell profiling) together with the clinical characterization of the patient (known as phenomapping209,215, that includes, for example, electrocardiography, echocardiography and laboratory and physical tests) will allow improved diagnosis and differentiation of the type of inflammatory cardiomyopathy, with the ultimate goal of defining therapeutically homogeneous patient subpopulations to improve patient outcomes209 (Fig. 6).
Fig. 6. Improving the subclassification of patients with inflammatory cardiomyopathy.
Clinical characterization of the patient (using risk factors and demographic and quality of life parameters, in addition to basic, physical and laboratory tests, and electrocardiography (ECG), echocardiography and cardiac MRI measurements) — known as phenomapping — combined with histology, immunohistology and viral diagnosis of endomyocardial biopsy samples will allow the classification of patients with inflammatory cardiomyopathy into different clusters, with the ultimate goal of defining therapeutically homogeneous patient subpopulations to improve outcomes. The figure shows a schematic example of a heatmap with hierarchical clustering of the patients on the basis of clinical parameters and endomyocardial biopsy results. GFR, glomerular filtration rate; HIV, human immunodeficiency virus; hsCRP, high-sensitivity C-reactive protein; PvO2, mixed venous oxygen tension.
Knowledge gaps and future directions
Standardize the diverse immunohistochemistry markers and protocols for analysing EMB samples; for example, use of formalin-fixed tissue versus frozen tissue sections or the most appropriate antibodies to use195.
Define thresholds for the number of different cell types required for diagnosis and prognosis of myocarditis and inflammatory cardiomyopathy.
Standardize the number of biopsies required for histology, immunohistology and viral diagnostics.
Develop a more detailed classification of the specific immune cell subtypes (such as Treg cells, TH17 cells and pro-inflammatory and anti-inflammatory monocyte and macrophage subsets) involved in myocarditis and inflammatory cardiomyopathy.
Genetic testing and biomarkers
Genetic testing
Monogenetic familial forms of acute myocarditis or chronic inflammatory cardiomyopathies are rare. Nevertheless, arrhythmogenic cardiomyopathies, in particular those caused by heterozygous pathogenic variants in DSP, have been associated with increased cardiac inflammation and a clinical presentation of acute myocarditis with elevated plasma troponin levels in addition to typical cardiac MRI or PET-CT scan abnormalities216–220. A homozygous but not heterozygous carrier state of rare variants in genes associated with inherited arrhythmogenic cardiomyopathies is significantly more frequent in children with acute myocarditis than in healthy individuals221. The mechanisms underlying the increased cardiac inflammation in arrhythmogenic cardiomyopathies remain unclear. Nevertheless, genetic testing should be considered in all familial forms of myocarditis, in familial cardiomyopathy or when signs of arrhythmogenic cardiomyopathy are present in imaging or electrophysiological tests.
MicroRNA profiling in EMB samples and blood
Epigenetic factors influence the expression of different genes and the genetic susceptibility to developing myocarditis and inflammatory cardiomyopathy. MicroRNAs (miRNAs) have emerged as important epigenetic regulators of the immune response in the heart222. Therefore, miRNA profiling of EMB samples might help distinguish different forms of myocarditis. For example, 107 miRNAs were found to be differentially expressed in RV septal samples from patients with acute viral myocarditis compared with heart samples from control individuals223. Cardiac miRNA profiles also differ in patients with myocarditis with or without CVB3 persistence, whereby the expression of eight miRNAs was strongly increased in samples from patients with late viral persistence and progressive cardiac dysfunction compared with samples from patients with spontaneous virus clearance and cardiac recovery224. The expression of 113 of 641 miRNAs analysed was significantly altered in heart samples from mice with Trypanosoma cruzi-induced myocarditis compared with heart samples from control mice225. A study assessing heart-associated, fibrosis-associated and leukocyte-associated miRNAs in blood found that only miRNAs related to cardiomyocyte injury (including miR-208 and miR-499) were elevated in patients with acute myocarditis compared with control individuals226,227. However, these markers of cardiomyocyte injury are non-specific because they are also increased in patients with acute ischaemic or hypertensive cardiac events226,228. Interestingly, the plasma levels of miRNAs related to inflammation, including miR-21, miR-146b and miR-155, were not increased in patients with acute myocarditis compared with control individuals even though leukocyte counts were elevated226, which possibly reflects the absence of miRNA release by inflammatory cells. In general, the correlation between circulating and tissue miRNAs is not clear and needs further investigation. Future investigations to further profile miRNAs, mRNA and proteins should focus on both circulating leukocytes and EMB samples to provide a better reflection of disease pathophysiology. These studies might provide new ways to distinguish different forms of myocarditis as well as to differentiate between myocarditis and ischaemic injury, which is an important unmet medical need.
EMB transcriptome-based biomarker
In addition to miRNAs, a panel of mRNAs has been shown to be highly predictive of the presence or absence of lymphocytic myocarditis229. A microarray-derived, transcriptome-based biomarker had a 100% sensitivity and specificity for the detection of myocarditis in EMB samples229. The most parsimonious transcriptomic signature was highly enriched for immune markers, notably various members of the TLR family. This transcriptome-based biomarker effectively detected lymphocytic myocarditis and active cardiac inflammatory disease in EMB samples from patients with rheumatic disease or peripartum cardiomyopathy229. Work is ongoing to test this approach for liquid biopsy.
Blood biomarkers
In addition to EMB-based biomarkers, several blood biomarkers, including high-sensitivity C-reactive protein, N-terminal pro-B-type natriuretic peptide (NT-proBNP), troponin T and soluble IL-1 receptor-like 1 (IL1RL1; also known as ST2), have been studied in the context of myocarditis and inflammatory cardiomyopathy2,22,66. In men, but not in women, aged ≤50 years with clinically suspected or EMB-confirmed myocarditis, elevated serum levels of soluble ST2 are associated with an increased risk of more severe HF, as assessed by NYHA class22. This finding highlights the potential of using soluble ST2 as a biomarker to predict the risk of HF in men and the importance of analysing inflammatory biomarkers such as soluble ST2 according to sex and age, and indicates the need for biomarkers that predict the risk of HF in women with myocarditis22. Myocarditis-specific blood biomarkers that can inform the diagnosis in patients with suspected myocarditis and can determine the presence or absence of active myocardial inflammation have not been established so far2,66. Preliminary evidence indicates that plasma levels of the S100A8-S100A9 heterodimer, which is predominantly released by monocytes and neutrophils, accurately reflect disease activity in cardiac tissue samples from patients with recent-onset myocarditis70. In addition, these preliminary data suggest that S100A8-S100A9 could serve as a diagnostic and therapy-monitoring biomarker in patients with suspected acute myocarditis (≤30 days after myocarditis onset)70.
Patients with autoimmune myocarditis230 or idiopathic DCM231 have lower numbers of circulating Treg cells and greater response of circulating TH17 cells than healthy individuals. Therefore, measuring blood Treg cell and TH17 cell numbers in these patients might be beneficial in guiding therapeutic decisions and for therapy follow-up, given the availability of therapies that increase the Treg cell to TH17 cell ratio. Inflammatory cardiomyopathy has been shown to be driven, at least in part, by the activation of heart-specific CD4+ T cells induced by myosin peptide mimics derived from Bacteroides thetaiotaomicron, an intestinal commensal bacterium. This finding suggests that the analysis of IgG antibodies specific for this gut bacterium species might help guide antibiotic treatment decisions141.
In the future, analysis of liquid biopsies might help dissect the high heterogeneity of cardiac tissue by providing information on circulating cell types (such as immune cells) and their products at specific time points, thereby allowing real-time monitoring of disease evolution. Use of innovative NGS platforms to analyse blood samples might help identify novel circulating biomarkers, including DNA methylation, histone modification and miRNA makers, as crucial pathogenic determinants of inflammatory heart diseases232. Another interesting approach is to evaluate whether the analysis of the proteome of exosomes present in serum can help identify the different types of myocarditis and whether exosomal protein analysis can be used for the development of predictive and prognostic biomarkers233.
Knowledge gaps and future directions
Understand why variants in DSP that are associated with arrhythmogenic cardiomyopathies have been associated with a clinical presentation of myocarditis on cardiac MRI scans.
Develop myocarditis-specific blood biomarkers that can inform the diagnosis in patients with suspected myocarditis and help to determine the presence or absence of active myocarditis.
Develop markers for therapy monitoring.
Develop biomarkers that predict the risk of HF in women with myocarditis.
Establish the reason for the lack of correlation between miRNAs levels in blood and in EMB samples.
Determine whether potential markers that are developed on the basis of circulating cells would be more sensitive and specific in diagnosing and discriminating myocarditis from other causes than markers that are developed on the basis of EMB samples.
Therapy
Management of HF and arrhythmias
Patients with myocarditis and reduced LVEF are treated with optimal medical care, according to guidelines for the management of HF234. However, many patients with myocarditis have preserved LVEF. Whether early initiation of treatment with inhibitors of the renin–angiotensin–aldosterone system or with β-blockers can reduce inflammation, adverse remodelling and scar formation in these patients is questionable. In particular, the risk of arrhythmia is increased in patients with myocarditis independently of LVEF235.
In patients with myocarditis, life-threatening bradyarrhythmias and tachyarrhythmias can occur at any stage of the disease and lead to sudden cardiac death155,236. Ventricular arrhythmias (VA) are mostly reported in patients with giant-cell myocarditis or cardiac sarcoidosis, with a prevalence of 29%237 and 55%238, respectively. Supraventricular arrhythmias occur more frequently than VA in patients with myocarditis and can vary in prevalence depending on the type of myocarditis239. Atrioventricular block is less common in patients with acute or fulminant myocarditis than in patients with cardiac sarcoidosis, and has a variable, but mostly low prevalence in patients with giant-cell myocarditis9. The frequency of cardiac electrical conductance disturbances decreases from giant-cell myocarditis to eosinophilic myocarditis to lymphocytic myocarditis240. The high prevalence of cardiac electrical conductance disturbances in patients with myocarditis highlights a clinical need to identify patients with myocarditis at risk of arrhythmia, independently of LVEF and LGE.
Several pathogenic mechanisms have been postulated to explain the presence of different arrhythmias observed in patients with acute myocarditis, including electrical instability due to direct cytopathic effects, ischaemia due to coronary microvascular or macrovascular disease, gap junction dysfunction, abnormal calcium handling and involvement of the cardiac conduction system. The risk of sudden cardiac death in patients with acute myocarditis is not always associated with the severity of myocardial inflammation241, and can persist after the acute phase of myocarditis is resolved13. Post-inflammatory, scar-related VA can present as monomorphic ventricular tachycardia in patients with healed myocarditis242. Post-inflammatory scar-related VA occurs in regions of myocardial fibrosis, which appear as low-voltage regions on electroanatomical voltage mapping or as LGE on cardiac MRI. Although systolic dysfunction is a common finding in patients with myocarditis with VA, an arrhythmogenic scar can occur in patients with preserved LVEF13. EMB is recommended for the diagnosis of myocarditis in patients with VA and acute cardiomyopathy because the risk of VA is increased in patients with inflammation in EMB samples241 (Table 2). The presence of viral nucleic acids in EMB samples can also indicate an increased risk of VA and late myocardial damage with progressive electrical conduction defects. In a mouse model of CVB3-induced myocarditis, modulating, time-dependent effects of the CVB3 infection were found in the cardiac ion channels KCNQ1, hERG1 and Cav1.2 in heterologous expression, providing an explanation for the development of arrhythmias in enteroviral myocarditis243. Given that EMB for the diagnosis of cardiac inflammation in patients with VA can have a high sampling error in patients with focal myocarditis and especially in patients with cardiac sarcoidosis, electroanatomical voltage mapping can be used to target the bioptome (the instrument used to obtain EMB samples) to areas with <0.5 mV amplitude and fractionated electrogram signal201–204 (Fig. 5).
Symptomatic VA in patients with acute myocarditis is usually managed with antiarrhythmic drugs, but the efficacy of this approach has not been tested. Cardiac device implantation for the management of VA should be evaluated after the resolution of reversible acute myocarditis, generally 3–6 months after initiation of the acute phase244. However, the timings for the placement of an implantable cardioverter–defibrillator (ICD) remain unclear. Temporary pacing might be required on presentation, but decisions for chronic pacing typically require a period of observation, histological examination of EMB samples and assessment of the disease course. Early de novo ICD implantation in patients with reduced LVEF alone should be avoided, and the use of a wearable cardioverter–defibrillator (LifeVest, ZOLL) should be considered in patients at high risk of sudden cardiac death245, in patients with lymphocytic myocarditis and in patients with myocarditis and VA in the acute phase of disease246. Nevertheless, the best timing of wearable cardioverter–defibrillator use has not been prospectively investigated. Patients with giant-cell myocarditis and VA with a heart transplant-free life expectancy of >1 year should receive an ICD. Risk management in patients with cardiac sarcoidosis is most probably independent of LVEF, indicating that LVEF-independent markers need to be identified.
Knowledge gaps and future directions
Assess the role of classic HF medications for the primary prevention of HF in patients with myocarditis.
Characterize the role of LVEF for predicting the risk of sudden death in patients with myocarditis.
Assess the influence of the different forms of inflammatory cardiomyopathy on the risk of sudden cardiac death (for example, cardiac sarcoidosis versus lymphocytic myocarditis).
Assess the best timing for cardiac pacing device implantation.
Investigate the role of a wearable cardioverter–defibrillator in patients with myocarditis with preserved LVEF and with signs of clinically significant cardiac rhythm abnormalities.
Develop a patient stratification model for the risk of sudden death in patients with myocarditis.
Perform clinical trials to assess the efficacy of antiarrhythmic drugs in patients with cardiac inflammation.
Investigate the effect of (intensive) exercise on the propensity to sudden cardiac death and HF247.
Drugs and biologics
Several treatment options have been studied in specific cohorts of patients with inflammatory cardiomyopathy that have been developed on the basis of the EMB-defined presence of viral genome (virus type and viral load) and immune cell infiltrates.
Virus-negative inflammatory cardiomyopathy
Studies and registries of EMB samples from patients with virus-negative, chronic inflammatory cardiomyopathy suggest that the use of immunosuppressive therapy with prednisone and azathioprine can improve cardiac function127,128,248,249. These findings contrast with results from earlier studies in patients with acute cardiomyopathy in which viral pathogens were not assessed. A single-centre, observational study found that 53% of patients with inflammatory cardiomyopathy who do not respond to steroid-based therapy had CD20+ B cells in EMB samples114. In this subset of six patients with virus-negative inflammatory cardiomyopathy and CD20+ B cell-positive EMB results, treatment with rituximab (a chimaeric monoclonal antibody against the pan-B cell surface molecule CD20) improved cardiac function and alleviated signs and symptoms of HF from baseline114, suggesting that rituximab therapy has beneficial effects in this patient population. Alternative treatment regimens for patients with virus-negative or autoimmune inflammatory cardiomyopathy include steroid-based treatment combined with cyclosporine250 or mycophenolate mofetil251, or immunoadsorption with subsequent intravenous immunoglobulin (IVIG) therapy252–254 (immunoadsorption–IVIG). Removal of circulating antibodies by non-specific immunoadsorption has been successful in the treatment of several autoimmune diseases255,256. Pilot studies indicate that immunoadsorption–IVIG improves myocardial function in patients with DCM252 and reduces myocardial inflammation257. However, these novel findings should be viewed as hypothesis-generating and more data are required from randomized trials. Indeed, a large, placebo-controlled multicentre study to investigate the effects of immunoadsorption–IVIG on LV function in patients with DCM or inflammatory cardiomyopathy is ongoing258. The randomization phase of this study was completed in 2019. An alternative to immunoadsorption is the intravenous administration of small soluble molecules (such as peptides or aptamers) that specifically target and neutralize autoantibodies against the β1-adrenergic receptor259. Of note, the use of antibody-targeting approaches does not depend on the presence of cardiac inflammation (Fig. 7).
Fig. 7. Gaps in evidence for endomyocardial biopsy-guided therapy in myocarditis and inflammatory cardiomyopathy.
Patients with myocarditis or inflammatory cardiomyopathy can be classified into four groups on the basis of endomyocardial biopsy (EMB) results: inflammation-negative, virus-negative; inflammation-positive, virus-negative; inflammation-negative, virus-positive; and inflammation-positive, virus-positive. In patients with virus-positive inflammatory cardiomyopathy, a clear distinction between virus-active and virus-associated inflammatory cardiomyopathy is required. Given the different aetiologies and clinical presentations of the four groups, specific therapy regimens are suggested for each group (blue boxes), in addition to approved optimal medical therapy for heart failure and risk-adjusted therapy. Immunosuppressive therapy is mandatory for specific forms of virus-negative autoimmune myocarditis, such as eosinophilic myocarditis, giant-cell myocarditis and cardiac sarcoidosis. Immunosuppressive therapy is also safe and effective in clinically unstable or non-resolving lymphocytic virus-negative myocarditis and in lymphocytic virus-negative myocarditis refractory to standard heart failure therapy. Autoantibody targeting can be achieved with immunoadsorption or with newly developed small molecules (aptamers) that neutralize specific autoantibodies. Autoantibody targeting is also under investigation for the treatment of non-primary inflammatory heart diseases, in which autoimmunity could have a role in disease progression. However, knowledge gaps remain about the type and length of immunosuppression and on novel biological agents to target specific immune pathways or autoantibodies. Data from registries and large randomized clinical trials are needed to evaluate the efficacy of the different proposed regimens, which will contribute to improving the clinical value of EMB-guided diagnosis. The role of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in myocarditis and corresponding treatment options are still unclear; therefore, SARS-CoV-2 is not included in the figure. (?) denotes unclear, needs further investigation; B19V, parvovirus B19; HCV, hepatitis C virus; IVIG, intravenous immunoglobulins.
Virus-positive inflammatory cardiomyopathy
Differentiating between virus-induced active myocarditis (for example, caused by adenoviruses or enteroviruses) and virus-associated myocarditis (in which the viral genome is detected in EMB samples but whether the virus is a bystander is not clear; for example, caused by latent infections with herpesviruses or B19V) is important (Fig. 7). To date, the therapeutic efficacy of targeting the viral infection in acute viral myocarditis has not yet been established in randomized clinical trials. The phase II BICC trial260 investigated the effects of immunomodulation with IFNβ therapy on viral clearance in patients with inflammatory cardiomyopathy and myocardial viral persistence (adenoviruses, enteroviruses or B19V). Trial participants with enterovirus-positive myocarditis or adenovirus-positive myocarditis (as assessed with EMB) showed viral clearance after treatment with IFNβ26,260, but IFNβ therapy was not associated with viral DNA clearance in patients with B19V-positive myocarditis146,260. The antiviral drugs pocapavir and pleconaril as well as IVIG therapy are effective in neonates with enteroviral myocarditis15–17. In patients with latent infection with Epstein–Barr virus, cytomegalovirus or HHV6, the use of anti-herpesvirus drugs is an option to reduce viral copy numbers261. Whether a combination of antiviral and immunosuppressive drugs can be an option in selected patients with virus-positive inflammatory cardiomyopathy depending on the stage of the disease needs to be investigated.
IVIG is often used in patients with severe B19V viraemia and clinical complications. New antiviral strategies against B19V infections are under investigation30 and include the synthetic nucleotide analogues cidofovir and brincidofovir, flavonoid molecules, and hydroxyurea. However, no therapy options are so far available for B19V-associated inflammatory cardiomyopathy. The consensus is that no therapy is needed if low B19V copy numbers are detected in cardiac tissue samples in the absence of cardiac inflammation2. Evidence from small observational studies indicates that immunosuppressive treatment has beneficial effects in patients with low B19V DNA load in the myocardium and with cardiac inflammation (CaPACITY programme)262, and in patients with B19V RNA positivity, from treatment with the antiviral drug telbivudine, owing to its immunomodulatory properties32,159. However, placebo-controlled clinical trials are needed to validate these observations. Of note, immunoadsorption–IVIG was found to be safe and effective in improving clinical symptoms in patients with virus-positive inflammatory cardiomyopathy, regardless of B19V or HHV6 presence in EMB samples263. By contrast, IVIG did not show any beneficial effects in patients with DCM in a study that did not evaluate cardiac viral persistence264. Registry data indicate that IVIG is associated with clinical improvement in patients with B19V-associated inflammatory cardiomyopathy265. IVIG was associated with a reduction in cardiac inflammation whereas cardiac B19V eradication was limited, as assessed on EMB samples265.
Patients with HIV-associated, HCV-associated or influenza-associated myocarditis or inflammatory cardiomyopathy are treated with established antiviral drugs (Table 1), including antiretroviral therapy for patients with HIV-associated myocarditis266, a combination of ombitasvir, paritaprevir, ritonavir and dasabuvir for patients with HCV-associated myocarditis46, and neuraminidase inhibitors (peramivir and zanamivir) for patients with influenza-associated myocarditis267,268. For patients with COVID-19, several antiviral regimens are under investigation and include strategies to prevent viral entry into the host cell (such as chloroquine, hydroxychloroquine, camostat mesylate and umifenovir), protease inhibitors (lopinavir–ritonavir and darunavir), RNA polymerase inhibitors (remdesivir) and anti-cytokine agents (such as IL-6 receptor antagonists and IL-1β inhibitors)269.
Knowledge gaps and future directions
Perform large, prospective, randomized controlled studies to explore new or existing (repurposed) immunosuppressive or immunomodulatory regimens and antiviral regimens in patients with myocarditis or inflammatory cardiomyopathy.
Perform multicentre, EMB-guided or cardiac MRI-guided trials to assess optimal treatment duration with conventional HF drugs and, particularly, with treatment regimens that still need to be approved for clinical use.
Perform sex-matched clinical studies, given the well-known sex-related differences in immune responses21,22 and outcomes in patients with myocarditis270.
Novel therapeutic strategies
Patients who do not respond to guideline-directed neurohormonal inhibitor therapy and haemodynamic support might benefit from therapies that either inhibit one or more of the effector arms of the immune response or promote regulatory elements of the immune system. Insights from exploratory clinical trials suggest that multiple signalling pathways can be differentially activated in patients with myocarditis or inflammatory cardiomyopathy. Importantly, current clinical trials are designed on the basis of lessons learned from previous trials on high-dose TNF inhibitor therapy in patients with systolic HF, which did not show improvements in patient outcomes271. By taking a systematic approach to personalize targeted therapies, the current generation of therapeutic agents are aimed at minimizing toxicity and maximizing the likelihood of recovery in patients with specific phenotypes of inflammatory cardiomyopathy.
Soluble anti-CAR antibody
Treatment with an engineered soluble CAR fused to the carboxy terminus of human IgG, which reduces virus uptake into host cells, has been shown to limit the development of acute272 and chronic273 CVB3-induced myocarditis in mice. The potential of this approach still needs to be evaluated in humans.
Anti-IL-1β and anti-IL-1 receptor antibodies
Findings from studies in animal models of viral and autoimmune myocarditis support a central role for NLRP3 inflammasome activation and subsequent IL-1β production in the pathogenesis of myocarditis68,274. Treatment with an anti-mouse IL-1β antibody at different stages of enteroviral infection prevented the development of chronic viral myocarditis by reducing inflammation, interstitial fibrosis and adverse cardiac remodelling in mice274. One clinical trial275 and several case series276,277 support the use of an anti-IL-1β monoclonal antibody for the treatment of recurrent pericarditis. The ongoing ARAMIS278 and RHAPSODY279 trials are designed to assess the efficacy of IL-1β-blocking agents in patients with myocarditis and associated pericarditis.
Anti-IL-17 antibody
Increased IL-17-related responses and the activation of profibrotic pathways have been associated with a greater risk of death in mice with CVB3-induced myocarditis280 and with a lower rate of functional recovery in patients with myocarditis108. TH17 cells have been shown to promote the progression to DCM in mice102, whereas Treg cells protected against myocarditis in mice by attenuating inflammation80,103. A clinical trial of secukinumab, an anti-IL-17 monoclonal antibody, has been proposed.
Cell-based therapies
Clinical application of Treg cells281 or the use of IL-2 agonists282 (which promote Treg cell production and increase survival and suppressor function of mature Treg cells283) are alternative approaches to elevate the Treg cell to TH17 cell ratio. Another potential cell-based approach involves the use of mesenchymal stromal cells, which have been shown to increase the number of Treg cells85 and have immunomodulatory and cardioprotective effects in mouse models of myocarditis69,78,284, such as by modulating the cardiosplenic axis. Therapy with allogeneic mesenchymal stromal cells has also been shown to be safe and effective in patients with non-ischaemic DCM in the POSEIDON-DCM trial285. In this trial, a significant improvement in LVEF with autologous mesenchymal stromal cell therapy was observed only in patients who did not carry a pathogenic gene variant associated with DCM, indicating the relevance of the genetic profile of patients with non-ischaemic DCM in dictating responsiveness to mesenchymal stromal cell therapy286. This response was associated with a marked reduction in circulating TNF levels285, suggestive of a therapeutic effect governed by immunomodulation. Taken together, these findings indicate that cell-based therapy has a potential role in the treatment of patients with inflammatory cardiomyopathy. Future trials are warranted to test this hypothesis.
Aldosterone antagonists
Evidence shows that early blockade (starting at the acute phase of CVB3 infection) of the mineralocorticoid receptor with eplerenone has pleiotropic effects, including immunomodulatory, anti-oxidative and anti-apoptotic effects, and prevents adverse cardiac remodelling and dysfunction without affecting viral load in the heart in a mouse model of persistent viral myocarditis287. This finding suggests that eplerenone is an ideal candidate as an acute treatment of myocarditis, together with HF treatment. However, current guidelines do not consider aldosterone antagonist therapy for acute myocarditis, indicating the need to test this new therapeutic concept in clinical trials.
Cannabidiol and antagomirs
Interventions that primarily promote regulatory functions of the immune system for the treatment of myocarditis are under investigation in experimental models. The approaches include therapy with cannabidiol288 and therapy with antisense miRNA complements71 (known as antagomirs or anti-miRs). Antagomirs injected systemically or locally can be used as a therapeutic tool to reduce either inflammation or virus replication71.
Modulation of the gut microbiome
Accumulating findings demonstrate the contribution of the gut microbiome and its derived metabolites to the underlying inflammation associated with HF289. In addition, a gut microbiota-derived myosin-mimic peptide has been linked to inflammatory cardiomyopathy141. These findings suggest that modulation of the microbiome and its derived metabolites are potential preventive and therapeutic strategies for inflammatory heart diseases.
Mechanical circulatory support in patients with fulminant myocarditis
In patients with cardiogenic shock due to fulminant myocarditis, parenteral inotropes and short-term MCS systems are often required. MCS can be used regardless of initiation of immunosuppressive therapy. Different MCS devices are effective for temporary haemodynamic stabilization and can serve as a bridge-to-transplant in patients with fulminant myocarditis, including veno-arterial extracorporeal membrane oxygenation (V-A ECMO)290,291, intra-aortic balloon pumps (IABPs)291,292, the percutaneous ventricular assist devices TandemHeart291 and ProtekDuo293, and the Impella microaxial flow catheters291,292,294. These devices differ in their mode of action, especially their effect on afterload modification. The effect of these devices on afterload modification might be of special interest in fulminant myocarditis, because a rise in peripheral resistance can also further stimulate cardiac inflammatory responses owing to an increase in ventricular wall stress. The high flow of V-A ECMO is known to increase LV afterload, which has been suggested to trigger cardiac inflammatory responses and unfavourable cardiac remodelling over time. IABP and TandemHeart have a minimal effect on LV afterload reduction, whereas intravascular aortic catheter (iVAC) and, even more effective, LV Impella microaxial flow catheter systems (the Impella CP, 5.0 and 5.5) support peripheral circulation in combination with ventricular unloading295. Mechanical load has been shown to activate the cardiac mechanotransduction network296, which is associated with unfavourable cardiac remodelling and cardiac fibroblast activation, and further promotion of inflammatory processes297. Evidence from LV assist devices in patients with chronic HF indicates that mechanical unloading can lead to reverse remodelling involving immunomodulatory mechanisms298,299. According to first single-centre studies, prolonged use of microaxial flow catheters over weeks exerts unique anti-inflammatory, disease-modifying effects beyond circulatory support (PROPELLA concept)295,300.
In routine clinical practice, the choice of MCS device usually depends on its availability at the centre and on whether the left or right ventricle alone, or both, are compromised. Patients with primarily RV or biventricular failure are usually treated with an extracorporeal centrifugal flow-based regimen (V-A ECMO, iVAC, TandemHeart and ProtekDuo)291. Analyses of data from experienced centres show that the combination of a V-A ECMO with an IABP, an iVAC (EC-iVAC) or LV Impella (ECMELLA) is safe and probably more effective than a single V-A ECMO approach in patients with fulminant myocarditis292,301,302. Fewer data are available on the efficacy of the RP Impella system and/or its combination with the LV Impella in unloading both ventricles (BIPELLA-concept) in patients with fulminant myocarditis300,303. In patients with primarily LV failure and preserved RV function, use of the LV Impella system for unloading might be favourable159,294,295,300. More research is needed to understand the mechanisms underlying the anti-inflammatory effects of prolonged LV unloading (PROPELLA concept)295 with the use of percutaneous LV support devices, which can serve as a bridge-to-recovery or bridge-to-transplant. Furthermore, clinical trials are needed to validate the PROPELLA concept, preferably including EMB analysis. Furthermore, the question of whether pulsatile systems (such as iVAC and IABPs) differ from non-pulsatile systems needs to be answered.
Knowledge gaps and future directions
Generate prospective data from multicentre registries to assess standardized protocols that can guide the management of patients with fulminant myocarditis or acute myocarditis complicated by severe HF.
Identify the best MCS or combination of MCS that allow optimal perfusion and LV unloading.
Perform randomized clinical trials to assess the efficacy of immunosuppressive treatments for patients with fulminant myocarditis receiving MCS.
Conclusions
Myocarditis and inflammatory cardiomyopathy were first defined in the early 1900s304. Since then, milestone discoveries (including the technique of EMB305, the invention of PCR and the development of cardiac MRI306) and consensus statements have led to a refined definition of myocarditis and inflammatory cardiomyopathy, with the latest major update published in 2013 (ref.2). This timeline renders the field of myocarditis and inflammatory cardiomyopathy as a fairly young area in cardiology. Entering the 2020s, gaps remain in our understanding of the pathogenesis of, and the diagnostic and therapeutic options for, myocarditis and inflammatory cardiomyopathy. The availability of novel sophisticated techniques, emerging and existing (repurposed) therapies, computational modelling and novel insights will help address these knowledge gaps in the near future. Specific diagnostic approaches for different clinical scenarios will help improve the subclassification of patients with inflammatory cardiomyopathy, and improve the common terminology in a field that is not completely univocal. Finally, the efficacy of many existing, repurposed or emerging therapies needs to be evaluated in large, controlled, randomized trials to facilitate the development of aetiology-based therapies159 (Fig. 7).
Acknowledgements
C.T. acknowledges the support of the Federal Ministry of Education and Research (BMBF), Germany, for the CaPACITY (Cortisone in Parvovirus Inflammatory Cardiomyopathy) programme. A.L.P.C. acknowledges the support of Budget Integrato per la Ricerca dei Dipartimenti (BIRD, year 2019), Padova University, Padova, Italy (project title: Myocarditis: Genetic Background, Predictors of Dismal Prognosis and of Response to Immunosuppressive Therapy). S.H. acknowledges the support of the ERA-Net-CVD project MacroERA (01KL1706) and IMI2-CARDIATEAM (no. 821508); the support of the Netherlands Cardiovascular Research Initiative, an initiative with support of the Dutch Heart Foundation, CVON2016-Early HFPEF, 2015-10, CVON She-PREDICTS, grant 2017-21, CVON Arena-PRIME, 2017-18; and the support of the Flemish FWO G091018N and FWO G0B5930N. C.T. and S.V.L. acknowledge the support of the German Centre for Cardiovascular Research (DZHK) for the ‘Voltage-mapping-guided and MRI-guided endomyocardial biopsy in myocarditis and DCMi’ study.
Glossary
- CD8+ T cells
A subpopulation of MHC class I-restricted T cells and mediators of adaptive immunity. They include cytotoxic T cells, which are important for killing cancerous or virus-infected cells, and CD8+ suppressor T cells, which block certain types of immune response.
- T helper 1 cells
(TH1 cells). A subset of T helper cells that primarily secrete IFNγ, which is associated with protection against intracellular microbes (predominantly viruses) and the onset of antitumorigenic or pro-tumorigenic effects.
- T helper 2 cells
(TH2 cells). A subset of T helper cells that fight parasitic infections by secreting specific interleukins, including IL-4, IL-5 and IL-13.
- Regulatory T cells
(Treg cells). A subpopulation of CD4+ T cells, constituting 5–10% of the peripheral T cells, that have a pivotal role in the induction and maintenance of immune homeostasis and tolerance. Treg cells have multiple effector functions and execute their regulatory potency by directly suppressing T cells, B cells and antigen-presenting cells, and also by interacting with non-immune tissue cells.
- Antigen-presenting cells
(APCs). A heterogeneous group of immune cells that mediate the cellular immune response by processing and presenting antigens recognizable by T cells. Classic APCs include macrophages, dendritic cells, B cells and Langerhans cells.
- Neutrophil extracellular traps
Complexes of chromosomal DNA, histones and granule proteins that are released by neutrophils and can entangle bacteria, thereby limiting infection.
- Experimental autoimmune myocarditis
(EAM). EAM can be induced in susceptible mouse strains by immunization with self-peptides derived from the myosin H chain together with a strong adjuvant, or by injection of activated, myosin H chain-loaded dendritic cells.
- TH17 cells
A subset of T helper cells that fight microbial pathogens by secreting cytokines such as IL-17A, IL-17F and IL-22.
- Dallas criteria
The Dallas criteria were proposed in 1986 and provide a histopathological categorization for the diagnosis of myocarditis. According to the Dallas criteria, myocarditis requires an inflammatory infiltrate and associated myocyte necrosis or damage not characteristic of an ischaemic event, whereas in borderline myocarditis, a less intense inflammatory infiltrate and no light-microscopic evidence of myocytolysis (myocyte destruction) is evident.
- Immunoadsorption
Selective apheresis method for the removal of specific antibodies and immune complexes through high-affinity adsorbers, achieved by passing a patient’s plasma over columns that remove immunoglobulins. The adsorbed plasma is then reinfused into the patient.
- Intravenous immunoglobulin
(IVIG). Therapy based on the intravenous administration of a blood product prepared from the serum of 1,000–15,000 donors per batch. IVIG therapy is the treatment of choice for patients with antibody deficiencies and is commonly used after immunoadsorption.
Author contributions
C.T., N.H., H.M., K.K. and S.V.L. researched data for the article; C.T., E.A., K.K. and S.V.L. contributed to discussion of the content; C.T., E.A., B.B., A.L.P.C., L.T.C, S.B.F., J.M.H., B.H., S.H., S.K., K.K., H.M., A.S.P., F.S., R.C.S., H.T., P.S. and S.V.L. wrote the article and C.T., A.L.P.C., L.T.C., S.B.F., J.M.H., B.H., S.H., N.H., K.K. and S.V.L. reviewed and/or edited the article before submission.
Competing interests
C.T. is a consultant for Cardiotropic Labs, Miami, FL, USA. S.B.F. reports grants from Fresenius Medical Care and ENDI Foundation. J.M.H. holds equity in Heart Genomics. J.M.H. and B.H. are both inventors on a patent involving the use of RNA as a biomarker for myocarditis. The other authors declare no competing interests.
Footnotes
Peer review information
Nature Reviews Cardiology thanks D. Cihakova, D. Fairweather, A. Frustaci, G. Thiene and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Richardson P, et al. Report of the 1995 World Health Organization/International Society and Federation of Cardiology task force on the definition and classification of cardiomyopathies. Circulation. 1996;93:841–842. doi: 10.1161/01.CIR.93.5.841. [DOI] [PubMed] [Google Scholar]
- 2.Caforio AL, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2013;34:2636–2648. doi: 10.1093/eurheartj/eht210. [DOI] [PubMed] [Google Scholar]
- 3.Ammirati E, et al. Clinical presentation and outcome in a contemporary cohort of patients with acute myocarditis. Circulation. 2018;138:1088–1099. doi: 10.1161/CIRCULATIONAHA.118.035319. [DOI] [PubMed] [Google Scholar]
- 4.Kociol RD, et al. Recognition and initial management of fulminant myocarditis: a scientific statement from the American Heart Association. Circulation. 2020;141:e69–e92. doi: 10.1161/CIR.0000000000000745. [DOI] [PubMed] [Google Scholar]
- 5.Ammirati E, et al. Fulminant versus acute nonfulminant myocarditis in patients with left ventricular systolic dysfunction. J. Am. Coll. Cardiol. 2019;74:299–311. doi: 10.1016/j.jacc.2019.04.063. [DOI] [PubMed] [Google Scholar]
- 6.Dominguez F, Kuhl U, Pieske B, Garcia-Pavia P, Tschöpe C. Update on myocarditis and inflammatory cardiomyopathy: reemergence of endomyocardial biopsy. Rev. Esp. Cardiol. 2016;69:178–187. doi: 10.1016/j.recesp.2015.10.018. [DOI] [PubMed] [Google Scholar]
- 7.Trachtenberg BH, Hare JM. Inflammatory cardiomyopathic syndromes. Circ. Res. 2017;121:803–818. doi: 10.1161/CIRCRESAHA.117.310221. [DOI] [PubMed] [Google Scholar]
- 8.Hu JR, et al. Cardiovascular toxicities associated with immune checkpoint inhibitors. Cardiovasc. Res. 2019;115:854–868. doi: 10.1093/cvr/cvz026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Caforio ALP, et al. Diagnosis and management of myocardial involvement in systemic immune-mediated diseases: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Disease. Eur. Heart J. 2017;38:2649–2662. doi: 10.1093/eurheartj/ehx321. [DOI] [PubMed] [Google Scholar]
- 10.Kuhl U, et al. High prevalence of viral genomes and multiple viral infections in the myocardium of adults with “idiopathic” left ventricular dysfunction. Circulation. 2005;111:887–893. doi: 10.1161/01.CIR.0000155616.07901.35. [DOI] [PubMed] [Google Scholar]
- 11.Ukimura A, Satomi H, Ooi Y, Kanzaki Y. Myocarditis associated with influenza A H1N1pdm2009. Influenza Res. Treat. 2012;2012:351979. doi: 10.1155/2012/351979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Van Linthout S, Klingel K, Tschope C. SARS-CoV2-related myocarditis-like syndroms: Shakespeare’s question: What’s in a name? Eur. J. Heart Fail. 2020;22:922–925. doi: 10.1002/ejhf.1899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bozkurt B, et al. Current diagnostic and treatment strategies for specific dilated cardiomyopathies: a scientific statement from the American Heart Association. Circulation. 2016;134:e579–e646. doi: 10.1161/CIR.0000000000000455. [DOI] [PubMed] [Google Scholar]
- 14.Pauschinger M, et al. Detection of adenoviral genome in the myocardium of adult patients with idiopathic left ventricular dysfunction. Circulation. 1999;99:1348–1354. doi: 10.1161/01.CIR.99.10.1348. [DOI] [PubMed] [Google Scholar]
- 15.Yen MH, et al. Effect of intravenous immunoglobulin for neonates with severe enteroviral infections with emphasis on the timing of administration. J. Clin. Virol. 2015;64:92–96. doi: 10.1016/j.jcv.2015.01.013. [DOI] [PubMed] [Google Scholar]
- 16.Abzug MJ, et al. A randomized, double-blind, placebo-controlled trial of pleconaril for the treatment of neonates with enterovirus sepsis. J. Pediatric Infect. Dis. Soc. 2016;5:53–62. doi: 10.1093/jpids/piv015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Amdani SM, et al. Successful treatment of fulminant neonatal enteroviral myocarditis in monochorionic diamniotic twins with cardiopulmonary support, intravenous immunoglobulin and pocapavir. BMJ Case Rep. 2018;2018:bcr-2017-224133. doi: 10.1136/bcr-2017-224133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Woodruff JF. Viral myocarditis. A review. Am. J. Pathol. 1980;101:425–484. [PMC free article] [PubMed] [Google Scholar]
- 19.Smith WG. Coxsackie B myopericarditis in adults. Am. Heart J. 1970;80:34–46. doi: 10.1016/0002-8703(70)90035-9. [DOI] [PubMed] [Google Scholar]
- 20.Zhou F, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Frisancho-Kiss S, et al. Cutting edge: cross-regulation by TLR4 and T cell Ig mucin-3 determines sex differences in inflammatory heart disease. J. Immunol. 2007;178:6710–6714. doi: 10.4049/jimmunol.178.11.6710. [DOI] [PubMed] [Google Scholar]
- 22.Coronado MJ, et al. Elevated sera sST2 is associated with heart failure in men ≤50 years old with myocarditis. J. Am. Heart Assoc. 2019;8:e008968. doi: 10.1161/JAHA.118.008968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.He Y, et al. Interaction of coxsackievirus B3 with the full length coxsackievirus-adenovirus receptor. Nat. Struct. Biol. 2001;8:874–878. doi: 10.1038/nsb1001-874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Badorff C, et al. Enteroviral protease 2A cleaves dystrophin: evidence of cytoskeletal disruption in an acquired cardiomyopathy. Nat. Med. 1999;5:320–326. doi: 10.1038/6543. [DOI] [PubMed] [Google Scholar]
- 25.Lassner D, et al. CCR5del32 genotype in human enteroviral cardiomyopathy leads to spontaneous virus clearance and improved outcome compared to wildtype CCR5. J. Transl Med. 2018;16:249. doi: 10.1186/s12967-018-1610-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kuhl U, Lassner D, von Schlippenbach J, Poller W, Schultheiss HP. Interferon-beta improves survival in enterovirus-associated cardiomyopathy. J. Am. Coll. Cardiol. 2012;60:1295–1296. doi: 10.1016/j.jacc.2012.06.026. [DOI] [PubMed] [Google Scholar]
- 27.Leveque, N. et al. Functional consequences of RNA 5′-terminal deletions on coxsackievirus B3 RNA replication and ribonucleoprotein complex formation. J. Virol. 91, e00423-17 (2017). [DOI] [PMC free article] [PubMed]
- 28.Bouin A, et al. Enterovirus persistence in cardiac cells of patients with idiopathic dilated cardiomyopathy is linked to 5′ terminal genomic RNA-deleted viral populations with viral-encoded proteinase activities. Circulation. 2019;139:2326–2338. doi: 10.1161/CIRCULATIONAHA.118.035966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Maisch B. Cardio-immunology of myocarditis: focus on immune mechanisms and treatment options. Front. Cardiovasc. Med. 2019;6:48. doi: 10.3389/fcvm.2019.00048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Manaresi E, Gallinella G. Advances in the development of antiviral strategies against parvovirus B19. Viruses. 2019;11:659. doi: 10.3390/v11070659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Duechting A, et al. Human parvovirus B19 NS1 protein modulates inflammatory signaling by activation of STAT3/PIAS3 in human endothelial cells. J. Virol. 2008;82:7942–7952. doi: 10.1128/JVI.00891-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Van Linthout S, et al. Telbivudine in chronic lymphocytic myocarditis and human parvovirus B19 transcriptional activity. ESC. Heart Fail. 2018;5:818–829. doi: 10.1002/ehf2.12341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bultmann BD, Sotlar K, Klingel K. Parvovirus B19. N. Engl. J. Med. 2004;350:2006–2007. doi: 10.1056/NEJM200405063501920. [DOI] [PubMed] [Google Scholar]
- 34.Kindermann I, et al. Predictors of outcome in patients with suspected myocarditis. Circulation. 2008;118:639–648. doi: 10.1161/CIRCULATIONAHA.108.769489. [DOI] [PubMed] [Google Scholar]
- 35.Hjalmarsson C, et al. Parvovirus B19 in endomyocardial biopsy of patients with idiopathic dilated cardiomyopathy: foe or bystander? J. Card. Fail. 2019;25:60–63. doi: 10.1016/j.cardfail.2018.07.466. [DOI] [PubMed] [Google Scholar]
- 36.Schenk T, Enders M, Pollak S, Hahn R, Huzly D. High prevalence of human parvovirus B19 DNA in myocardial autopsy samples from subjects without myocarditis or dilative cardiomyopathy. J. Clin. Microbiol. 2009;47:106–110. doi: 10.1128/JCM.01672-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lotze U, et al. Low level myocardial parvovirus B19 persistence is a frequent finding in patients with heart disease but unrelated to ongoing myocardial injury. J. Med. Virol. 2010;82:1449–1457. doi: 10.1002/jmv.21821. [DOI] [PubMed] [Google Scholar]
- 38.Koepsell SA, Anderson DR, Radio SJ. Parvovirus B19 is a bystander in adult myocarditis. Cardiovasc. Pathol. 2012;21:476–481. doi: 10.1016/j.carpath.2012.02.002. [DOI] [PubMed] [Google Scholar]
- 39.Bock CT, Klingel K, Kandolf R. Human parvovirus B19-associated myocarditis. N. Engl. J. Med. 2010;362:1248–1249. doi: 10.1056/NEJMc0911362. [DOI] [PubMed] [Google Scholar]
- 40.Bock CT, et al. Molecular phenotypes of human parvovirus B19 in patients with myocarditis. World J. Cardiol. 2014;6:183–195. doi: 10.4330/wjc.v6.i4.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Dennert R, et al. Differences in virus prevalence and load in the hearts of patients with idiopathic dilated cardiomyopathy with and without immune-mediated inflammatory diseases. Clin. Vaccine Immunol. 2012;19:1182–1187. doi: 10.1128/CVI.00281-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kuhl U, et al. A distinct subgroup of cardiomyopathy patients characterized by transcriptionally active cardiotropic erythrovirus and altered cardiac gene expression. Basic. Res. Cardiol. 2013;108:372. doi: 10.1007/s00395-013-0372-y. [DOI] [PubMed] [Google Scholar]
- 43.Richter J, et al. An unusual presentation of a common infection. Infection. 2013;41:565–569. doi: 10.1007/s15010-012-0321-y. [DOI] [PubMed] [Google Scholar]
- 44.Kaufer BB, Flamand L. Chromosomally integrated HHV-6: impact on virus, cell and organismal biology. Curr. Opin. Virol. 2014;9:111–118. doi: 10.1016/j.coviro.2014.09.010. [DOI] [PubMed] [Google Scholar]
- 45.Barbaro G. HIV-associated cardiomyopathy etiopathogenesis and clinical aspects. Herz. 2005;30:486–492. doi: 10.1007/s00059-005-2728-z. [DOI] [PubMed] [Google Scholar]
- 46.Sanchez MJ, Bergasa NV. Hepatitis C associated cardiomyopathy: potential pathogenic mechanisms and clinical implications. Med. Sci. Monit. 2008;14:RA55–RA63. [PubMed] [Google Scholar]
- 47.Kumar K, et al. Influenza myocarditis and myositis: case presentation and review of the literature. Can. J. Cardiol. 2011;27:514–522. doi: 10.1016/j.cjca.2011.03.005. [DOI] [PubMed] [Google Scholar]
- 48.Zhang SF, et al. Epidemiology characteristics of human coronaviruses in patients with respiratory infection symptoms and phylogenetic analysis of HCoV-OC43 during 2010-2015 in Guangzhou. PLoS ONE. 2018;13:e0191789. doi: 10.1371/journal.pone.0191789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cui J, Li F, Shi ZL. Origin and evolution of pathogenic coronaviruses. Nat. Rev. Microbiol. 2019;17:181–192. doi: 10.1038/s41579-018-0118-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhou P, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang D, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zheng YY, Ma YT, Zhang JY, Xie X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020;17:259–260. doi: 10.1038/s41569-020-0360-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Huang C, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Moore BJB, June CH. Cytokine release syndrome in severe COVID-19. Science. 2020;368:473–474. doi: 10.1126/science.abb8925. [DOI] [PubMed] [Google Scholar]
- 55.Guo J, Huang Z, Lin L, Lv J. Coronavirus disease 2019 (COVID-19) and cardiovascular disease: a viewpoint on the potential influence of angiotensin-converting enzyme inhibitors/angiotensin receptor blockers on onset and severity of severe acute respiratory syndrome coronavirus 2 infection. J. Am. Heart Assoc. 2020;9:e016219. doi: 10.1161/JAHA.120.016219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Santos RAS, et al. The ACE2/angiotensin-(1-7)/MAS axis of the renin-angiotensin system: focus on angiotensin-(1-7) Physiol. Rev. 2018;98:505–553. doi: 10.1152/physrev.00023.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Oudit GY, et al. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur. J. Clin. Invest. 2009;39:618–625. doi: 10.1111/j.1365-2362.2009.02153.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Li W, et al. Receptor and viral determinants of SARS-coronavirus adaptation to human ACE2. EMBO J. 2005;24:1634–1643. doi: 10.1038/sj.emboj.7600640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hoffmann M, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Glowacka I, et al. Evidence that TMPRSS2 activates the severe acute respiratory syndrome coronavirus spike protein for membrane fusion and reduces viral control by the humoral immune response. J. Virol. 2011;85:4122–4134. doi: 10.1128/JVI.02232-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Nicin L, et al. Cell type-specific expression of the putative SARS-CoV-2 receptor ACE2 in human hearts. Eur. Heart J. 2020;41:1804–1806. doi: 10.1093/eurheartj/ehaa311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Tavazzi G, et al. Myocardial localization of coronavirus in COVID-19 cardiogenic shock. Eur. J. Heart Fail. 2020;22:911–915. doi: 10.1002/ejhf.1828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Varga Z, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Forbes JD, Knox NC, Peterson CL, Reimer AR. Highlighting clinical metagenomics for enhanced diagnostic decision-making: a step towards wider implementation. Comput. Struct. Biotechnol. J. 2018;16:108–120. doi: 10.1016/j.csbj.2018.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Swirski FK, Nahrendorf M. Cardioimmunology: the immune system in cardiac homeostasis and disease. Nat. Rev. Immunol. 2018;18:733–744. doi: 10.1038/s41577-018-0065-8. [DOI] [PubMed] [Google Scholar]
- 66.Pollack A, Kontorovich AR, Fuster V, Dec GW. Viral myocarditis-diagnosis, treatment options, and current controversies. Nat. Rev. Cardiol. 2015;12:670–680. doi: 10.1038/nrcardio.2015.108. [DOI] [PubMed] [Google Scholar]
- 67.Alexopoulou L, Holt AC, Medzhitov R, Flavell RA. Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature. 2001;413:732–738. doi: 10.1038/35099560. [DOI] [PubMed] [Google Scholar]
- 68.Tschope C, et al. NOD2 (nucleotide-binding oligomerization domain 2) is a major pathogenic mediator of coxsackievirus B3-induced myocarditis. Circ. Heart Fail. 2017;10:e003870. doi: 10.1161/CIRCHEARTFAILURE.117.003870. [DOI] [PubMed] [Google Scholar]
- 69.Miteva K, et al. Mesenchymal stromal cells inhibit NLRP3 inflammasome activation in a model of coxsackievirus B3-induced inflammatory cardiomyopathy. Sci. Rep. 2018;8:2820. doi: 10.1038/s41598-018-20686-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Muller I, et al. Serum alarmin S100A8/S100A9 levels and its potential role as biomarker in myocarditis. ESC Heart Fail. 2020;7:1442–1451. doi: 10.1002/ehf2.12760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Heymans S, Eriksson U, Lehtonen J, Cooper LT., Jr The quest for new approaches in myocarditis and inflammatory cardiomyopathy. J. Am. Coll. Cardiol. 2016;68:2348–2364. doi: 10.1016/j.jacc.2016.09.937. [DOI] [PubMed] [Google Scholar]
- 72.Huang CH, Vallejo JG, Kollias G, Mann DL. Role of the innate immune system in acute viral myocarditis. Basic. Res. Cardiol. 2009;104:228–237. doi: 10.1007/s00395-008-0765-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Libby P, Nahrendorf M, Swirski FK. Leukocytes link local and systemic inflammation in ischemic cardiovascular disease: an expanded “cardiovascular continuum”. J. Am. Coll. Cardiol. 2016;67:1091–1103. doi: 10.1016/j.jacc.2015.12.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Leuschner F, et al. Rapid monocyte kinetics in acute myocardial infarction are sustained by extramedullary monocytopoiesis. J. Exp. Med. 2012;209:123–137. doi: 10.1084/jem.20111009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Swirski FK, et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science. 2009;325:612–616. doi: 10.1126/science.1175202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ismahil MA, et al. Remodeling of the mononuclear phagocyte network underlies chronic inflammation and disease progression in heart failure: critical importance of the cardiosplenic axis. Circ. Res. 2014;114:266–282. doi: 10.1161/CIRCRESAHA.113.301720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Leuschner F, et al. Silencing of CCR2 in myocarditis. Eur. Heart J. 2015;36:1478–1488. doi: 10.1093/eurheartj/ehu225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Miteva K, et al. Mesenchymal stromal cells modulate monocytes trafficking in coxsackievirus B3-induced myocarditis. Stem Cell Transl Med. 2017;6:1249–1261. doi: 10.1002/sctm.16-0353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Cooper LT, Jr, Fairweather D. Nano-scale treatment for a macro-scale disease: nanoparticle-delivered siRNA silences CCR2 and treats myocarditis. Eur. Heart J. 2015;36:1434–1436. doi: 10.1093/eurheartj/ehu302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pappritz K, et al. Immunomodulation by adoptive regulatory T-cell transfer improves Coxsackievirus B3-induced myocarditis. FASEB J. 2018;32:6066–6078. doi: 10.1096/fj.201701408R. [DOI] [PubMed] [Google Scholar]
- 81.Muller I, et al. CX3CR1 knockout aggravates Coxsackievirus B3-induced myocarditis. PLoS ONE. 2017;12:e0182643. doi: 10.1371/journal.pone.0182643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Klingel K, et al. Pathogenesis of murine enterovirus myocarditis: virus dissemination and immune cell targets. J. Virol. 1996;70:8888–8895. doi: 10.1128/JVI.70.12.8888-8895.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Hofmann P, Schmidtke M, Stelzner A, Gemsa D. Suppression of proinflammatory cytokines and induction of IL-10 in human monocytes after Coxsackievirus B3 infection. J. Med. Virol. 2001;64:487–498. doi: 10.1002/jmv.1076. [DOI] [PubMed] [Google Scholar]
- 84.Kandolf R, et al. Mechanisms and consequences of enterovirus persistence in cardiac myocytes and cells of the immune system. Virus Res. 1999;62:149–158. doi: 10.1016/S0168-1702(99)00041-6. [DOI] [PubMed] [Google Scholar]
- 85.Savvatis K, et al. Mesenchymal stromal cells but not cardiac fibroblasts exert beneficial systemic immunomodulatory effects in experimental myocarditis. PLoS ONE. 2012;7:e41047. doi: 10.1371/journal.pone.0041047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Fairweather D, et al. Mast cells and innate cytokines are associated with susceptibility to autoimmune heart disease following coxsackievirus B3 infection. Autoimmunity. 2004;37:131–145. doi: 10.1080/0891693042000196200. [DOI] [PubMed] [Google Scholar]
- 87.Klingel K, et al. The activating receptor NKG2D of natural killer cells promotes resistance against enterovirus-mediated inflammatory cardiomyopathy. J. Pathol. 2014;234:164–177. doi: 10.1002/path.4369. [DOI] [PubMed] [Google Scholar]
- 88.Yuan J, et al. CXCL10 inhibits viral replication through recruitment of natural killer cells in coxsackievirus B3-induced myocarditis. Circ. Res. 2009;104:628–638. doi: 10.1161/CIRCRESAHA.108.192179. [DOI] [PubMed] [Google Scholar]
- 89.Clemente-Casares X, et al. A CD103(+) conventional dendritic cell surveillance system prevents development of overt heart failure during subclinical viral myocarditis. Immunity. 2017;47:974–989 e978. doi: 10.1016/j.immuni.2017.10.011. [DOI] [PubMed] [Google Scholar]
- 90.Eriksson U, et al. Dendritic cell-induced autoimmune heart failure requires cooperation between adaptive and innate immunity. Nat. Med. 2003;9:1484–1490. doi: 10.1038/nm960. [DOI] [PubMed] [Google Scholar]
- 91.Xu D, et al. Gr-1+ cells other than Ly6G+ neutrophils limit virus replication and promote myocardial inflammation and fibrosis following coxsackievirus B3 infection of mice. Front. Cell Infect. Microbiol. 2018;8:157. doi: 10.3389/fcimb.2018.00157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Rivadeneyra L, et al. Role of neutrophils in CVB3 infection and viral myocarditis. J. Mol. Cell Cardiol. 2018;125:149–161. doi: 10.1016/j.yjmcc.2018.08.029. [DOI] [PubMed] [Google Scholar]
- 93.Weckbach LT, et al. Midkine drives cardiac inflammation by promoting neutrophil trafficking and NETosis in myocarditis. J. Exp. Med. 2019;216:350–368. doi: 10.1084/jem.20181102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Afanasyeva M, et al. Quantitative analysis of myocardial inflammation by flow cytometry in murine autoimmune myocarditis: correlation with cardiac function. Am. J. Pathol. 2004;164:807–815. doi: 10.1016/S0002-9440(10)63169-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Muller I, et al. Pathogenic role of the damage-associated molecular patterns S100A8 and S100A9 in coxsackievirus B3-induced myocarditis. Circ. Heart Fail. 2017;10:e004125. doi: 10.1161/CIRCHEARTFAILURE.117.004125. [DOI] [PubMed] [Google Scholar]
- 96.Tahto E, Jadric R, Pojskic L, Kicic E. Neutrophil-to-lymphocyte ratio and its relation with markers of inflammation and myocardial necrosis in patients with acute coronary syndrome. Med. Arch. 2017;71:312–315. doi: 10.5455/medarh.2017.71.312-315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Nahrendorf M, Swirski FK. Monocyte and macrophage heterogeneity in the heart. Circ. Res. 2013;112:1624–1633. doi: 10.1161/CIRCRESAHA.113.300890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Yang J, Zhang L, Yu C, Yang XF, Wang H. Monocyte and macrophage differentiation: circulation inflammatory monocyte as biomarker for inflammatory diseases. Biomark Res. 2014;2:1. doi: 10.1186/2050-7771-2-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Pappritz K, et al. Cardiac (myo)fibroblasts modulate the migration of monocyte subsets. Sci. Rep. 2018;8:5575. doi: 10.1038/s41598-018-23881-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Hou X, et al. The cardiac microenvironment instructs divergent monocyte fates and functions in myocarditis. Cell Rep. 2019;28:172–189. doi: 10.1016/j.celrep.2019.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Liu P, et al. The tyrosine kinase p56lck is essential in coxsackievirus B3-mediated heart disease. Nat. Med. 2000;6:429–434. doi: 10.1038/74689. [DOI] [PubMed] [Google Scholar]
- 102.Baldeviano GC, et al. Interleukin-17A is dispensable for myocarditis but essential for the progression to dilated cardiomyopathy. Circ. Res. 2010;106:1646–1655. doi: 10.1161/CIRCRESAHA.109.213157. [DOI] [PubMed] [Google Scholar]
- 103.Shi Y, et al. Regulatory T cells protect mice against coxsackievirus-induced myocarditis through the transforming growth factor β-coxsackie-adenovirus receptor pathway. Circulation. 2010;121:2624–2634. doi: 10.1161/CIRCULATIONAHA.109.893248. [DOI] [PubMed] [Google Scholar]
- 104.Anzai A, et al. Self-reactive CD4(+) IL-3(+) T cells amplify autoimmune inflammation in myocarditis by inciting monocyte chemotaxis. J. Exp. Med. 2019;216:369–383. doi: 10.1084/jem.20180722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Opavsky MA, et al. Susceptibility to myocarditis is dependent on the response of alphabeta T lymphocytes to coxsackieviral infection. Circ. Res. 1999;85:551–558. doi: 10.1161/01.RES.85.6.551. [DOI] [PubMed] [Google Scholar]
- 106.Klingel K, Schnorr JJ, Sauter M, Szalay G, Kandolf R. β2-Microglobulin-associated regulation of interferon-γ and virus-specific immunoglobulin G confer resistance against the development of chronic coxsackievirus myocarditis. Am. J. Pathol. 2003;162:1709–1720. doi: 10.1016/S0002-9440(10)64305-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Rangachari M, et al. T-bet negatively regulates autoimmune myocarditis by suppressing local production of interleukin 17. J. Exp. Med. 2006;203:2009–2019. doi: 10.1084/jem.20052222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Myers JM, et al. Cardiac myosin-Th17 responses promote heart failure in human myocarditis. JCI Insight. 2016;1:e85851. doi: 10.1172/jci.insight.85851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Ahern PP, et al. Interleukin-23 drives intestinal inflammation through direct activity on T cells. Immunity. 2010;33:279–288. doi: 10.1016/j.immuni.2010.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Wu L, et al. Pathogenic IL-23 signaling is required to initiate GM-CSF-driven autoimmune myocarditis in mice. Eur. J. Immunol. 2016;46:582–592. doi: 10.1002/eji.201545924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kaya Z, Leib C, Katus HA. Autoantibodies in heart failure and cardiac dysfunction. Circ. Res. 2012;110:145–158. doi: 10.1161/CIRCRESAHA.111.243360. [DOI] [PubMed] [Google Scholar]
- 112.Weber MS, et al. B-cell activation influences T-cell polarization and outcome of anti-CD20 B-cell depletion in central nervous system autoimmunity. Ann. Neurol. 2010;68:369–383. doi: 10.1002/ana.22081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Zouggari Y, et al. B lymphocytes trigger monocyte mobilization and impair heart function after acute myocardial infarction. Nat. Med. 2013;19:1273–1280. doi: 10.1038/nm.3284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Tschope C, et al. Targeting CD20+ B-lymphocytes in inflammatory dilated cardiomyopathy with rituximab improves clinical course: a case series. Eur. Heart J. Case Rep. 2019;3:ytz131. doi: 10.1093/ehjcr/ytz131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Diny NL, et al. Eosinophil-derived IL-4 drives progression of myocarditis to inflammatory dilated cardiomyopathy. J. Exp. Med. 2017;214:943–957. doi: 10.1084/jem.20161702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Tai PC, et al. Deposits of eosinophil granule proteins in cardiac tissues of patients with eosinophilic endomyocardial disease. Lancet. 1987;1:643–647. doi: 10.1016/S0140-6736(87)90412-0. [DOI] [PubMed] [Google Scholar]
- 117.Thambidorai SK, Korlakunta HL, Arouni AJ, Hunter WJ, Holmberg MJ. Acute eosinophilic myocarditis mimicking myocardial infarction. Tex. Heart Inst. J. 2009;36:355–357. [PMC free article] [PubMed] [Google Scholar]
- 118.Song T, Jones DM, Homsi Y. Therapeutic effect of anti-IL-5 on eosinophilic myocarditis with large pericardial effusion. BMJ Case Rep. 2017;2017:bcr2016218992. doi: 10.1136/bcr-2016-218992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Mahon NG, et al. Immunohistologic evidence of myocardial disease in apparently healthy relatives of patients with dilated cardiomyopathy. J. Am. Coll. Cardiol. 2002;39:455–462. doi: 10.1016/S0735-1097(01)01762-4. [DOI] [PubMed] [Google Scholar]
- 120.Caforio AL, et al. Evidence from family studies for autoimmunity in dilated cardiomyopathy. Lancet. 1994;344:773–777. doi: 10.1016/S0140-6736(94)92339-6. [DOI] [PubMed] [Google Scholar]
- 121.Caforio AL, et al. Prospective familial assessment in dilated cardiomyopathy: cardiac autoantibodies predict disease development in asymptomatic relatives. Circulation. 2007;115:76–83. doi: 10.1161/CIRCULATIONAHA.106.641472. [DOI] [PubMed] [Google Scholar]
- 122.Mestroni L, et al. Familial dilated cardiomyopathy: evidence for genetic and phenotypic heterogeneity. Heart Muscle Disease Study Group. J. Am. Coll. Cardiol. 1999;34:181–190. doi: 10.1016/S0735-1097(99)00172-2. [DOI] [PubMed] [Google Scholar]
- 123.Neu N, et al. Cardiac myosin induces myocarditis in genetically predisposed mice. J. Immunol. 1987;139:3630–3636. [PubMed] [Google Scholar]
- 124.Smith SC, Allen PM. Myosin-induced acute myocarditis is a T cell-mediated disease. J. Immunol. 1991;147:2141–2147. [PubMed] [Google Scholar]
- 125.Li Y, Heuser JS, Cunningham LC, Kosanke SD, Cunningham MW. Mimicry and antibody-mediated cell signaling in autoimmune myocarditis. J. Immunol. 2006;177:8234–8240. doi: 10.4049/jimmunol.177.11.8234. [DOI] [PubMed] [Google Scholar]
- 126.Frustaci A, et al. Immunosuppressive therapy for active lymphocytic myocarditis: virological and immunologic profile of responders versus nonresponders. Circulation. 2003;107:857–863. doi: 10.1161/01.CIR.0000048147.15962.31. [DOI] [PubMed] [Google Scholar]
- 127.Frustaci A, Russo MA, Chimenti C. Randomized study on the efficacy of immunosuppressive therapy in patients with virus-negative inflammatory cardiomyopathy: the TIMIC study. Eur. Heart J. 2009;30:1995–2002. doi: 10.1093/eurheartj/ehp249. [DOI] [PubMed] [Google Scholar]
- 128.Escher F, et al. Long-term outcome of patients with virus-negative chronic myocarditis or inflammatory cardiomyopathy after immunosuppressive therapy. Clin. Res. Cardiol. 2016;105:1011–1020. doi: 10.1007/s00392-016-1011-z. [DOI] [PubMed] [Google Scholar]
- 129.Caforio AL, et al. Novel organ-specific circulating cardiac autoantibodies in dilated cardiomyopathy. J. Am. Coll. Cardiol. 1990;15:1527–1534. doi: 10.1016/0735-1097(90)92821-I. [DOI] [PubMed] [Google Scholar]
- 130.Caforio AL, et al. A prospective study of biopsy-proven myocarditis: prognostic relevance of clinical and aetiopathogenetic features at diagnosis. Eur. Heart J. 2007;28:1326–1333. doi: 10.1093/eurheartj/ehm076. [DOI] [PubMed] [Google Scholar]
- 131.Caforio AL, et al. Identification of alpha- and beta-cardiac myosin heavy chain isoforms as major autoantigens in dilated cardiomyopathy. Circulation. 1992;85:1734–1742. doi: 10.1161/01.CIR.85.5.1734. [DOI] [PubMed] [Google Scholar]
- 132.Schulze K, Becker BF, Schultheiss HP. Antibodies to the ADP/ATP carrier, an autoantigen in myocarditis and dilated cardiomyopathy, penetrate into myocardial cells and disturb energy metabolism in vivo. Circ. Res. 1989;64:179–192. doi: 10.1161/01.RES.64.2.179. [DOI] [PubMed] [Google Scholar]
- 133.Caforio AL, et al. Passive transfer of affinity-purified anti-heart autoantibodies (AHA) from sera of patients with myocarditis induces experimental myocarditis in mice. Int. J. Cardiol. 2015;179:166–177. doi: 10.1016/j.ijcard.2014.10.165. [DOI] [PubMed] [Google Scholar]
- 134.Zwacka RM, et al. Redox gene therapy for ischemia/reperfusion injury of the liver reduces AP1 and NF-κB activation. Nat. Med. 1998;4:698–704. doi: 10.1038/nm0698-698. [DOI] [PubMed] [Google Scholar]
- 135.Jahns R, et al. Direct evidence for a β1-adrenergic receptor-directed autoimmune attack as a cause of idiopathic dilated cardiomyopathy. J. Clin. Invest. 2004;113:1419–1429. doi: 10.1172/JCI200420149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Nishimura H, et al. Autoimmune dilated cardiomyopathy in PD-1 receptor-deficient mice. Science. 2001;291:319–322. doi: 10.1126/science.291.5502.319. [DOI] [PubMed] [Google Scholar]
- 137.Meder B, et al. A genome-wide association study identifies 6p21 as novel risk locus for dilated cardiomyopathy. Eur. Heart J. 2014;35:1069–1077. doi: 10.1093/eurheartj/eht251. [DOI] [PubMed] [Google Scholar]
- 138.Arbustini E, et al. The MOGE(S) classification for a phenotype-genotype nomenclature of cardiomyopathy: endorsed by the World Heart Federation. J. Am. Coll. Cardiol. 2013;62:2046–2072. doi: 10.1016/j.jacc.2013.08.1644. [DOI] [PubMed] [Google Scholar]
- 139.Hazebroek MR, et al. Prognostic relevance of gene-environment interactions in patients with dilated cardiomyopathy: applying the MOGE(S) classification. J. Am. Coll. Cardiol. 2015;66:1313–1323. doi: 10.1016/j.jacc.2015.07.023. [DOI] [PubMed] [Google Scholar]
- 140.Pinto YM, et al. Proposal for a revised definition of dilated cardiomyopathy, hypokinetic non-dilated cardiomyopathy, and its implications for clinical practice: a position statement of the ESC working group on myocardial and pericardial diseases. Eur. Heart J. 2016;37:1850–1858. doi: 10.1093/eurheartj/ehv727. [DOI] [PubMed] [Google Scholar]
- 141.Gil-Cruz C, et al. Microbiota-derived peptide mimics drive lethal inflammatory cardiomyopathy. Science. 2019;366:881–886. doi: 10.1126/science.aav3487. [DOI] [PubMed] [Google Scholar]
- 142.Blyszczuk P. Myocarditis in humans and in experimental animal models. Front. Cardiovasc. Med. 2019;6:64. doi: 10.3389/fcvm.2019.00064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Grodums EI, Dempster G. Myocarditis in experimental coxsackie B-3 infection. Can. J. Microbiol. 1959;5:605–615. doi: 10.1139/m59-074. [DOI] [PubMed] [Google Scholar]
- 144.Klingel K, et al. Ongoing enterovirus-induced myocarditis is associated with persistent heart muscle infection: quantitative analysis of virus replication, tissue damage, and inflammation. Proc. Natl Acad. Sci. USA. 1992;89:314–318. doi: 10.1073/pnas.89.1.314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Tracy S, et al. Group B coxsackievirus myocarditis and pancreatitis: connection between viral virulence phenotypes in mice. J. Med. Virol. 2000;62:70–81. doi: 10.1002/1096-9071(200009)62:1<70::AID-JMV11>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- 146.Schmidt-Lucke C, et al. Interferon beta modulates endothelial damage in patients with cardiac persistence of human parvovirus b19 infection. J. Infect. Dis. 2010;201:936–945. doi: 10.1086/650700. [DOI] [PubMed] [Google Scholar]
- 147.Tschope C, et al. High prevalence of cardiac parvovirus B19 infection in patients with isolated left ventricular diastolic dysfunction. Circulation. 2005;111:879–886. doi: 10.1161/01.CIR.0000155615.68924.B3. [DOI] [PubMed] [Google Scholar]
- 148.Bachelier K, et al. Parvovirus B19-induced vascular damage in the heart is associated with elevated circulating endothelial microparticles. PLoS ONE. 2017;12:e0176311. doi: 10.1371/journal.pone.0176311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Huber SA, Lodge PA. Coxsackievirus B-3 myocarditis. Identification of different pathogenic mechanisms in DBA/2 and Balb/c mice. Am. J. Pathol. 1986;122:284–291. [PMC free article] [PubMed] [Google Scholar]
- 150.Bruno KA, et al. BPA alters estrogen receptor expression in the heart after viral infection activating cardiac mast cells and T cells leading to perimyocarditis and fibrosis. Front. Endocrinol. 2019;10:598. doi: 10.3389/fendo.2019.00598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Bucher CH, et al. Experience in the adaptive immunity impacts bone homeostasis, remodeling, and healing. Front. Immunol. 2019;10:797. doi: 10.3389/fimmu.2019.00797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Andreadou I, et al. Immune cells as targets for cardioprotection: new players and novel therapeutic opportunities. Cardiovasc. Res. 2019;115:1117–1130. doi: 10.1093/cvr/cvz050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Sharma A, et al. Human induced pluripotent stem cell-derived cardiomyocytes as an in vitro model for coxsackievirus B3-induced myocarditis and antiviral drug screening platform. Circ. Res. 2014;115:556–566. doi: 10.1161/CIRCRESAHA.115.303810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Van Linthout S, Tschope C, Schultheiss HP. Lack in treatment options for virus-induced inflammatory cardiomyopathy: can iPS-derived cardiomyocytes close the gap? Circ. Res. 2014;115:540–541. doi: 10.1161/CIRCRESAHA.114.304951. [DOI] [PubMed] [Google Scholar]
- 155.Corrado D, Basso C, Thiene G. Sudden cardiac death in young people with apparently normal heart. Cardiovasc. Res. 2001;50:399–408. doi: 10.1016/S0008-6363(01)00254-1. [DOI] [PubMed] [Google Scholar]
- 156.Aquaro GD, et al. Cardiac MR with late gadolinium enhancement in acute myocarditis with preserved systolic function: ITAMY study. J. Am. Coll. Cardiol. 2017;70:1977–1987. doi: 10.1016/j.jacc.2017.08.044. [DOI] [PubMed] [Google Scholar]
- 157.Kasner M, et al. Multimodality imaging approach in the diagnosis of chronic myocarditis with preserved left ventricular ejection fraction (MCpEF): the role of 2D speckle-tracking echocardiography. Int. J. Cardiol. 2017;243:374–378. doi: 10.1016/j.ijcard.2017.05.038. [DOI] [PubMed] [Google Scholar]
- 158.Ammirati E, et al. Acute and fulminant myocarditis: a pragmatic clinical approach to diagnosis and treatment. Curr. Cardiol. Rep. 2018;20:114. doi: 10.1007/s11886-018-1054-z. [DOI] [PubMed] [Google Scholar]
- 159.Tschope C, Cooper LT, Torre-Amione G, Van Linthout S. Management of myocarditis-related cardiomyopathy in adults. Circ. Res. 2019;124:1568–1583. doi: 10.1161/CIRCRESAHA.118.313578. [DOI] [PubMed] [Google Scholar]
- 160.Merlo M, et al. Persistent left ventricular dysfunction after acute lymphocytic myocarditis: frequency and predictors. PLoS ONE. 2019;14:e0214616. doi: 10.1371/journal.pone.0214616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.McMurray JJ, et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2012;33:1787–1847. doi: 10.1093/eurheartj/ehs104. [DOI] [PubMed] [Google Scholar]
- 162.Ferreira VM, et al. Cardiovascular magnetic resonance in nonischemic myocardial inflammation: expert recommendations. J. Am. Coll. Cardiol. 2018;72:3158–3176. doi: 10.1016/j.jacc.2018.09.072. [DOI] [PubMed] [Google Scholar]
- 163.Luetkens JA, et al. Comparison of original and 2018 Lake Louise criteria for diagnosis of acute myocarditis: results of a validation cohort. Radiol. Cardiothorac. Imaging. 2019 doi: 10.1148/ryct.2019190010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Thavendiranathan P, et al. Improved detection of myocardial involvement in acute inflammatory cardiomyopathies using T2 mapping. Circ. Cardiovasc. Imaging. 2012;5:102–110. doi: 10.1161/CIRCIMAGING.111.967836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Messroghli DR, et al. Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2* and extracellular volume: A consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imaging (EACVI) J. Cardiovasc. Magn. Reson. 2017;19:75. doi: 10.1186/s12968-017-0389-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Bohnen S, et al. Performance of T1 and T2 mapping cardiovascular magnetic resonance to detect active myocarditis in patients with recent-onset heart failure. Circ. Cardiovasc. Imaging. 2015;8:e003073. doi: 10.1161/CIRCIMAGING.114.003073. [DOI] [PubMed] [Google Scholar]
- 167.Radunski UK, et al. T1 and T2 mapping cardiovascular magnetic resonance imaging techniques reveal unapparent myocardial injury in patients with myocarditis. Clin. Res. Cardiol. 2017;106:10–17. doi: 10.1007/s00392-016-1018-5. [DOI] [PubMed] [Google Scholar]
- 168.Lurz P, et al. Comprehensive cardiac magnetic resonance imaging in patients with suspected myocarditis: the MyoRacer-trial. J. Am. Coll. Cardiol. 2016;67:1800–1811. doi: 10.1016/j.jacc.2016.02.013. [DOI] [PubMed] [Google Scholar]
- 169.Puntmann VO, Zeiher AM, Nagel E. T1 and T2 mapping in myocarditis: seeing beyond the horizon of Lake Louise criteria and histopathology. Expert Rev. Cardiovasc. Ther. 2018;16:319–330. doi: 10.1080/14779072.2018.1455499. [DOI] [PubMed] [Google Scholar]
- 170.Francone M, et al. CMR sensitivity varies with clinical presentation and extent of cell necrosis in biopsy-proven acute myocarditis. JACC Cardiovasc. Imaging. 2014;7:254–263. doi: 10.1016/j.jcmg.2013.10.011. [DOI] [PubMed] [Google Scholar]
- 171.Tanacli R, et al. Range variability in CMR feature tracking multilayer strain across different stages of heart failure. Sci. Rep. 2019;9:16478. doi: 10.1038/s41598-019-52683-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Escher F, et al. Development of diastolic heart failure in a 6-year follow-up study in patients after acute myocarditis. Heart. 2011;97:709–714. doi: 10.1136/hrt.2010.199489. [DOI] [PubMed] [Google Scholar]
- 173.Bohnen S, et al. Tissue characterization by T1 and T2 mapping cardiovascular magnetic resonance imaging to monitor myocardial inflammation in healing myocarditis. Eur. Heart J. Cardiovasc. Imaging. 2017;18:744–751. doi: 10.1093/ehjci/jex007. [DOI] [PubMed] [Google Scholar]
- 174.Heidecker B, et al. Systematic use of cardiac magnetic resonance imaging in MINOCA led to a five-fold increase in the detection rate of myocarditis: a retrospective study. Swiss Med. Wkly. 2019;149:w20098. doi: 10.4414/smw.2019.20098. [DOI] [PubMed] [Google Scholar]
- 175.Patriki D, et al. Approximation of the incidence of myocarditis by systematic screening with cardiac magnetic resonance imaging. JACC Heart Fail. 2018;6:573–579. doi: 10.1016/j.jchf.2018.03.002. [DOI] [PubMed] [Google Scholar]
- 176.Jessup M, Lindenfeld J. Light at the end of the myocarditis tunnel. JACC Heart Fail. 2018;6:580–582. doi: 10.1016/j.jchf.2018.04.015. [DOI] [PubMed] [Google Scholar]
- 177.Schneider JE, Stojanovic I. Economic evaluation of cardiac magnetic resonance with fast-SENC in the diagnosis and management of early heart failure. Health Econ. Rev. 2019;9:13. doi: 10.1186/s13561-019-0229-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Ge Y, et al. Cost-effectiveness analysis of stress cardiovascular magnetic resonance imaging for stable chest pain syndromes. JACC Cardiovasc. Imaging. 2020;13:1505–1517. doi: 10.1016/j.jcmg.2020.02.029. [DOI] [PubMed] [Google Scholar]
- 179.Petrov G, Kelle S, Fleck E, Wellnhofer E. Incremental cost-effectiveness of dobutamine stress cardiac magnetic resonance imaging in patients at intermediate risk for coronary artery disease. Clin. Res. Cardiol. 2015;104:401–409. doi: 10.1007/s00392-014-0793-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Cooper LT, et al. The role of endomyocardial biopsy in the management of cardiovascular disease: a scientific statement from the American Heart Association, the American College of Cardiology, and the European Society of Cardiology. Circulation. 2007;116:2216–2233. doi: 10.1161/CIRCULATIONAHA.107.186093. [DOI] [PubMed] [Google Scholar]
- 181.Backhaus SJ, et al. Real-time cardiovascular magnetic resonance T1 and extracellular volume fraction mapping for tissue characterisation in aortic stenosis. J. Cardiovasc. Magn. Reson. 2020;22:46. doi: 10.1186/s12968-020-00632-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Zhang S, et al. Real-time magnetic resonance imaging of cardiac function and flow–recent progress. Quant. Imaging Med. Surg. 2014;4:313–329. doi: 10.3978/j.issn.2223-4292.2014.06.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Lurz P, et al. Diagnostic performance of CMR imaging compared with EMB in patients with suspected myocarditis. JACC Cardiovasc. Imaging. 2012;5:513–524. doi: 10.1016/j.jcmg.2011.11.022. [DOI] [PubMed] [Google Scholar]
- 184.Grani C, et al. Prognostic value of cardiac magnetic resonance tissue characterization in risk stratifying patients with suspected myocarditis. J. Am. Coll. Cardiol. 2017;70:1964–1976. doi: 10.1016/j.jacc.2017.08.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Schelbert EB, et al. Myocardial fibrosis quantified by extracellular volume is associated with subsequent hospitalization for heart failure, death, or both across the spectrum of ejection fraction and heart failure stage. J. Am. Heart Assoc. 2015;4:e002613. doi: 10.1161/JAHA.115.002613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Mewton N, Liu CY, Croisille P, Bluemke D, Lima JA. Assessment of myocardial fibrosis with cardiovascular magnetic resonance. J. Am. Coll. Cardiol. 2011;57:891–903. doi: 10.1016/j.jacc.2010.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Berg J, et al. Cardiac magnetic resonance imaging in myocarditis reveals persistent disease activity despite normalization of cardiac enzymes and inflammatory parameters at 3-month follow-up. Circ. Heart Fail. 2017;10:e004262. doi: 10.1161/CIRCHEARTFAILURE.117.004262. [DOI] [PubMed] [Google Scholar]
- 188.Murtagh G, et al. Prognosis of myocardial damage in sarcoidosis patients with preserved left ventricular ejection fraction: risk stratification using cardiovascular magnetic resonance. Circ. Cardiovasc. Imaging. 2016;9:e003738. doi: 10.1161/CIRCIMAGING.115.003738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Nensa F, et al. Hybrid cardiac imaging using PET/MRI: a joint position statement by the European Society of Cardiovascular Radiology (ESCR) and the European Association of Nuclear Medicine (EANM) Eur. Radiol. 2018;28:4086–4101. doi: 10.1007/s00330-017-5008-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Lapinskas T, et al. The Intraventricular hemodynamic forces estimated using routine CMR cine images: a new marker of the failing heart. JACC Cardiovasc. Imaging. 2019;12:377–379. doi: 10.1016/j.jcmg.2018.08.012. [DOI] [PubMed] [Google Scholar]
- 191.Frey N, Meder B, Katus HA. Left ventricular biopsy in the diagnosis of myocardial diseases. Circulation. 2018;137:993–995. doi: 10.1161/CIRCULATIONAHA.117.030834. [DOI] [PubMed] [Google Scholar]
- 192.Nakayama T, Murai S, Ohte N. Dilated cardiomyopathy with eosinophilic granulomatosis with polyangiitis in which active myocardial inflammation was only detected by endomyocardial biopsy. Intern. Med. 2018;57:2675–2679. doi: 10.2169/internalmedicine.0330-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Cooper LT, et al. The role of endomyocardial biopsy in the management of cardiovascular disease: a scientific statement from the American Heart Association, the American College of Cardiology, and the European Society of Cardiology Endorsed by the Heart Failure Society of America and the Heart Failure Association of the European Society of Cardiology. Eur. Heart J. 2007;28:3076–3093. doi: 10.1093/eurheartj/ehm456. [DOI] [PubMed] [Google Scholar]
- 194.Leone O, et al. 2011 consensus statement on endomyocardial biopsy from the Association for European Cardiovascular Pathology and the Society for Cardiovascular Pathology. Cardiovasc. Pathol. 2012;21:245–274. doi: 10.1016/j.carpath.2011.10.001. [DOI] [PubMed] [Google Scholar]
- 195.Katzmann JL, et al. Meta-analysis on the immunohistological detection of inflammatory cardiomyopathy in endomyocardial biopsies. Heart Fail. Rev. 2019;25:277–294. doi: 10.1007/s10741-019-09835-9. [DOI] [PubMed] [Google Scholar]
- 196.Baughman KL. Diagnosis of myocarditis: death of Dallas criteria. Circulation. 2006;113:593–595. doi: 10.1161/CIRCULATIONAHA.105.589663. [DOI] [PubMed] [Google Scholar]
- 197.Andreoletti L, Leveque N, Boulagnon C, Brasselet C, Fornes P. Viral causes of human myocarditis. Arch. Cardiovasc. Dis. 2009;102:559–568. doi: 10.1016/j.acvd.2009.04.010. [DOI] [PubMed] [Google Scholar]
- 198.Badorff C, Knowlton KU. Dystrophin disruption in enterovirus-induced myocarditis and dilated cardiomyopathy: from bench to bedside. Med. Microbiol. Immunol. 2004;193:121–126. doi: 10.1007/s00430-003-0189-7. [DOI] [PubMed] [Google Scholar]
- 199.Spieker M, et al. Abnormal T2 mapping cardiovascular magnetic resonance correlates with adverse clinical outcome in patients with suspected acute myocarditis. J. Cardiovasc. Magn. Reson. 2017;19:38. doi: 10.1186/s12968-017-0350-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Unterberg-Buchwald C, et al. Targeted endomyocardial biopsy guided by real-time cardiovascular magnetic resonance. J. Cardiovasc. Magn. Reson. 2017;19:45. doi: 10.1186/s12968-017-0357-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Casella M, et al. Feasibility of combined unipolar and bipolar voltage maps to improve sensitivity of endomyocardial biopsy. Circ. Arrhythm. Electrophysiol. 2015;8:625–632. doi: 10.1161/CIRCEP.114.002216. [DOI] [PubMed] [Google Scholar]
- 202.Liang JJ, et al. Electrogram guidance: a method to increase the precision and diagnostic yield of endomyocardial biopsy for suspected cardiac sarcoidosis and myocarditis. JACC Heart Fail. 2014;2:466–473. doi: 10.1016/j.jchf.2014.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Konecny T, et al. Endomyocardial biopsy-integrating electrode at the bioptome tip. Ther. Adv. Cardiovasc. Dis. 2015;9:66–69. doi: 10.1177/1753944715574660. [DOI] [PubMed] [Google Scholar]
- 204.Vaidya VR, et al. The efficacy and safety of electroanatomic mapping-guided endomyocardial biopsy: a systematic review. J. Interv. Card. Electrophysiol. 2018;53:63–71. doi: 10.1007/s10840-018-0410-7. [DOI] [PubMed] [Google Scholar]
- 205.Omote K, et al. (18)F-FDG uptake of the right ventricle is an important predictor of histopathologic diagnosis by endomyocardial biopsy in patients with cardiac sarcoidosis. J. Nucl. Cardiol. 2019 doi: 10.1007/s12350-018-01541-7. [DOI] [PubMed] [Google Scholar]
- 206.Van Linthout S, Tschope C. Viral myocarditis: a prime example for endomyocardial biopsy-guided diagnosis and therapy. Curr. Opin. Cardiol. 2018;33:325–333. doi: 10.1097/HCO.0000000000000515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Lassner D, et al. Improved diagnosis of idiopathic giant cell myocarditis and cardiac sarcoidosis by myocardial gene expression profiling. Eur. Heart J. 2014;35:2186–2195. doi: 10.1093/eurheartj/ehu101. [DOI] [PubMed] [Google Scholar]
- 208.Hammer E, Darm K, Volker U. Characterization of the human myocardial proteome in dilated cardiomyopathy by label-free quantitative shotgun proteomics of heart biopsies. Methods Mol. Biol. 2013;1005:67–76. doi: 10.1007/978-1-62703-386-2_6. [DOI] [PubMed] [Google Scholar]
- 209.Van Linthout S, Tschope C. Lost in markers? Time for phenomics and phenomapping in dilated cardiomyopathy. Eur. J. Heart Fail. 2017;19:499–501. doi: 10.1002/ejhf.744. [DOI] [PubMed] [Google Scholar]
- 210.Soler-Botija C, Galvez-Monton C, Bayes-Genis A. Epigenetic biomarkers in cardiovascular diseases. Front. Genet. 2019;10:950. doi: 10.3389/fgene.2019.00950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Halliday BP, Cleland JGF, Goldberger JJ, Prasad SK. Personalizing risk stratification for sudden death in dilated cardiomyopathy: the past, present, and future. Circulation. 2017;136:215–231. doi: 10.1161/CIRCULATIONAHA.116.027134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Takeuchi S, et al. Identification of potential pathogenic viruses in patients with acute myocarditis using next-generation sequencing. J. Med. Virol. 2018;90:1814–1821. doi: 10.1002/jmv.25263. [DOI] [PubMed] [Google Scholar]
- 213.Kannan S, et al. Large particle fluorescence-activated cell sorting enables high-quality single-cell RNA sequencing and functional analysis of adult cardiomyocytes. Circ. Res. 2019;125:567–569. doi: 10.1161/CIRCRESAHA.119.315493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Martini E, et al. Single cell sequencing of mouse heart immune infiltrate in pressure overload-driven heart failure reveals extent of immune activation. Circulation. 2019;140:2089–2107. doi: 10.1161/CIRCULATIONAHA.119.041694. [DOI] [PubMed] [Google Scholar]
- 215.Shah SJ, et al. Phenomapping for novel classification of heart failure with preserved ejection fraction. Circulation. 2015;131:269–279. doi: 10.1161/CIRCULATIONAHA.114.010637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Reichl K, Kreykes SE, Martin CM, Shenoy C. Desmoplakin variant-associated arrhythmogenic cardiomyopathy presenting as acute myocarditis. Circ. Genom. Precis. Med. 2018;11:e002373. doi: 10.1161/CIRCGEN.118.002373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Calabrese F, Basso C, Carturan E, Valente M, Thiene G. Arrhythmogenic right ventricular cardiomyopathy/dysplasia: is there a role for viruses? Cardiovasc. Pathol. 2006;15:11–17. doi: 10.1016/j.carpath.2005.10.004. [DOI] [PubMed] [Google Scholar]
- 218.Lopez-Ayala JM, et al. Genetics of myocarditis in arrhythmogenic right ventricular dysplasia. Heart Rhythm. 2015;12:766–773. doi: 10.1016/j.hrthm.2015.01.001. [DOI] [PubMed] [Google Scholar]
- 219.Protonotarios A, et al. Prevalence of (18)F-fluorodeoxyglucose positron emission tomography abnormalities in patients with arrhythmogenic right ventricular cardiomyopathy. Int. J. Cardiol. 2019;284:99–104. doi: 10.1016/j.ijcard.2018.10.083. [DOI] [PubMed] [Google Scholar]
- 220.Hata Y, et al. Minimal inflammatory foci of unknown etiology may be a tentative sign of early stage inherited cardiomyopathy. Mod. Pathol. 2019;32:1281–1290. doi: 10.1038/s41379-019-0274-0. [DOI] [PubMed] [Google Scholar]
- 221.Belkaya S, et al. Autosomal recessive cardiomyopathy presenting as acute myocarditis. J. Am. Coll. Cardiol. 2017;69:1653–1665. doi: 10.1016/j.jacc.2017.01.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Small EM, Olson EN. Pervasive roles of microRNAs in cardiovascular biology. Nature. 2011;469:336–342. doi: 10.1038/nature09783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Corsten MF, et al. MicroRNA profiling identifies microRNA-155 as an adverse mediator of cardiac injury and dysfunction during acute viral myocarditis. Circ. Res. 2012;111:415–425. doi: 10.1161/CIRCRESAHA.112.267443. [DOI] [PubMed] [Google Scholar]
- 224.Kuehl U, et al. Differential cardiac microRNA expression predicts the clinical course in human enterovirus cardiomyopathy. Circ. Heart Fail. 2015;8:605–618. doi: 10.1161/CIRCHEARTFAILURE.114.001475. [DOI] [PubMed] [Google Scholar]
- 225.Navarro IC, et al. MicroRNA transcriptome profiling in heart of Trypanosoma cruzi-infected mice: parasitological and cardiological outcomes. PLoS Negl. Trop. Dis. 2015;9:e0003828. doi: 10.1371/journal.pntd.0003828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Corsten MF, et al. Circulating microRNA-208b and microRNA-499 reflect myocardial damage in cardiovascular disease. Circ. Cardiovasc. Genet. 2010;3:499–506. doi: 10.1161/CIRCGENETICS.110.957415. [DOI] [PubMed] [Google Scholar]
- 227.Goldberg L, et al. Circulating microRNAs: a potential biomarker for cardiac damage, inflammatory response, and left ventricular function recovery in pediatric viral myocarditis. J. Cardiovasc. Transl Res. 2018;11:319–328. doi: 10.1007/s12265-018-9814-0. [DOI] [PubMed] [Google Scholar]
- 228.Devaux Y, et al. Use of circulating microRNAs to diagnose acute myocardial infarction. Clin. Chem. 2012;58:559–567. doi: 10.1373/clinchem.2011.173823. [DOI] [PubMed] [Google Scholar]
- 229.Heidecker B, et al. Transcriptomic biomarkers for the accurate diagnosis of myocarditis. Circulation. 2011;123:1174–1184. doi: 10.1161/CIRCULATIONAHA.110.002857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Chen P, et al. Susceptibility to autoimmune myocarditis is associated with intrinsic differences in CD4(+) T cells. Clin. Exp. Immunol. 2012;169:79–88. doi: 10.1111/j.1365-2249.2012.04598.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Li J, et al. The Treg/Th17 imbalance in patients with idiopathic dilated cardiomyopathy. Scand. J. Immunol. 2010;71:298–303. doi: 10.1111/j.1365-3083.2010.02374.x. [DOI] [PubMed] [Google Scholar]
- 232.Benincasa G, Mansueto G, Napoli C. Fluid-based assays and precision medicine of cardiovascular diseases: the ‘hope’ for Pandora’s box? J. Clin. Pathol. 2019;72:785–799. doi: 10.1136/jclinpath-2019-206178. [DOI] [PubMed] [Google Scholar]
- 233.Kennel PJ, et al. Serum exosomal protein profiling for the non-invasive detection of cardiac allograft rejection. J. Heart Lung Transpl. 2018;37:409–417. doi: 10.1016/j.healun.2017.07.012. [DOI] [PubMed] [Google Scholar]
- 234.Ponikowski P, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 2016;18:891–975. doi: 10.1002/ejhf.592. [DOI] [PubMed] [Google Scholar]
- 235.Peretto G, et al. Ventricular arrhythmias in myocarditis: characterization and relationships with myocardial inflammation. J. Am. Coll. Cardiol. 2020;75:1046–1057. doi: 10.1016/j.jacc.2020.01.036. [DOI] [PubMed] [Google Scholar]
- 236.Baksi AJ, Kanaganayagam GS, Prasad SK. Arrhythmias in viral myocarditis and pericarditis. Card. Electrophysiol. Clin. 2015;7:269–281. doi: 10.1016/j.ccep.2015.03.009. [DOI] [PubMed] [Google Scholar]
- 237.Cooper LT, Jr., Berry GJ, Shabetai R. Idiopathic giant-cell myocarditis–natural history and treatment. Multicenter Giant Cell Myocarditis Study Group investigators. N. Engl. J. Med. 1997;336:1860–1866. doi: 10.1056/NEJM199706263362603. [DOI] [PubMed] [Google Scholar]
- 238.Birnie DH, et al. HRS expert consensus statement on the diagnosis and management of arrhythmias associated with cardiac sarcoidosis. Heart Rhythm. 2014;11:1305–1323. doi: 10.1016/j.hrthm.2014.03.043. [DOI] [PubMed] [Google Scholar]
- 239.Imazio M, Trinchero R. Myopericarditis: etiology, management, and prognosis. Int. J. Cardiol. 2008;127:17–26. doi: 10.1016/j.ijcard.2007.10.053. [DOI] [PubMed] [Google Scholar]
- 240.Adegbala O, et al. Predictors, burden, and the impact of arrhythmia on patients admitted for acute myocarditis. Am. J. Cardiol. 2019;123:139–144. doi: 10.1016/j.amjcard.2018.09.017. [DOI] [PubMed] [Google Scholar]
- 241.Maron BJ, et al. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: Task Force 3: Hypertrophic cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy and other cardiomyopathies, and myocarditis: a scientific statement from the American Heart Association and American College of Cardiology. J. Am. Coll. Cardiol. 2015;66:2362–2371. doi: 10.1016/j.jacc.2015.09.035. [DOI] [PubMed] [Google Scholar]
- 242.Zorzi A, et al. Nonischemic left ventricular scar as a substrate of life-threatening ventricular arrhythmias and sudden cardiac death in competitive athletes. Circ. Arrhythm. Electrophysiol. 2016;9:e004229. doi: 10.1161/CIRCEP.116.004229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Steinke K, et al. Coxsackievirus B3 modulates cardiac ion channels. FASEB J. 2013;27:4108–4121. doi: 10.1096/fj.13-230193. [DOI] [PubMed] [Google Scholar]
- 244.Priori SG, et al. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: The Task Force for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death of the European Society of Cardiology (ESC). Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC) Europace. 2015;17:1601–1687. doi: 10.1093/europace/euv319. [DOI] [PubMed] [Google Scholar]
- 245.Sheppard R, et al. Implantable cardiac defibrillators and sudden death in recent onset nonischemic cardiomyopathy: results from IMAC2. J. Card. Fail. 2012;18:675–681. doi: 10.1016/j.cardfail.2012.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Chung MK. The role of the wearable cardioverter defibrillator in clinical practice. Cardiol. Clin. 2014;32:253–270. doi: 10.1016/j.ccl.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 247.Halle, M. et al. Myocarditis in athletes: a clinical perspective. Eur. J. Prev. Cardiol. 10.1177/2047487320909670 (2020). [DOI] [PubMed]
- 248.Wojnicz R, et al. Randomized, placebo-controlled study for immunosuppressive treatment of inflammatory dilated cardiomyopathy: two-year follow-up results. Circulation. 2001;104:39–45. doi: 10.1161/01.CIR.104.1.39. [DOI] [PubMed] [Google Scholar]
- 249.Merken J, et al. Immunosuppressive therapy improves both short- and long-term prognosis in patients with virus-negative nonfulminant inflammatory cardiomyopathy. Circ. Heart Fail. 2018;11:e004228. doi: 10.1161/CIRCHEARTFAILURE.117.004228. [DOI] [PubMed] [Google Scholar]
- 250.Kleinert S, Weintraub RG, Wilkinson JL, Chow CW. Myocarditis in children with dilated cardiomyopathy: incidence and outcome after dual therapy immunosuppression. J. Heart Lung Transpl. 1997;16:1248–1254. [PubMed] [Google Scholar]
- 251.De Luca G, et al. Efficacy and safety of mycophenolate mofetil in patients with virus-negative lymphocytic myocarditis: a prospective cohort study. J. Autoimmun. 2020;106:102330. doi: 10.1016/j.jaut.2019.102330. [DOI] [PubMed] [Google Scholar]
- 252.Felix SB, et al. Hemodynamic effects of immunoadsorption and subsequent immunoglobulin substitution in dilated cardiomyopathy: three-month results from a randomized study. J. Am. Coll. Cardiol. 2000;35:1590–1598. doi: 10.1016/S0735-1097(00)00568-4. [DOI] [PubMed] [Google Scholar]
- 253.Trimpert C, et al. Immunoadsorption in dilated cardiomyopathy: long-term reduction of cardiodepressant antibodies. Eur. J. Clin. Invest. 2010;40:685–691. doi: 10.1111/j.1365-2362.2010.02314.x. [DOI] [PubMed] [Google Scholar]
- 254.Dandel M, et al. Long-term benefits of immunoadsorption in β(1)-adrenoceptor autoantibody-positive transplant candidates with dilated cardiomyopathy. Eur. J. Heart Fail. 2012;14:1374–1388. doi: 10.1093/eurjhf/hfs123. [DOI] [PubMed] [Google Scholar]
- 255.Kronbichler A, Brezina B, Quintana LF, Jayne DR. Efficacy of plasma exchange and immunoadsorption in systemic lupus erythematosus and antiphospholipid syndrome: a systematic review. Autoimmun. Rev. 2016;15:38–49. doi: 10.1016/j.autrev.2015.08.010. [DOI] [PubMed] [Google Scholar]
- 256.Yamaji K. Immunoadsorption for collagen and rheumatic diseases. Transfus. Apher. Sci. 2017;56:666–670. doi: 10.1016/j.transci.2017.08.012. [DOI] [PubMed] [Google Scholar]
- 257.Staudt A, et al. Immunohistological changes in dilated cardiomyopathy induced by immunoadsorption therapy and subsequent immunoglobulin substitution. Circulation. 2001;103:2681–2686. doi: 10.1161/01.CIR.103.22.2681. [DOI] [PubMed] [Google Scholar]
- 258.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT00558584 (2018).
- 259.Dungen HD, et al. β1-Adrenoreceptor autoantibodies in heart failure: physiology and therapeutic implications. Circ. Heart Fail. 2020;13:e006155. doi: 10.1161/CIRCHEARTFAILURE.119.006155. [DOI] [PubMed] [Google Scholar]
- 260.Schultheiss HP, et al. Betaferon in chronic viral cardiomyopathy (BICC) trial: effects of interferon-β treatment in patients with chronic viral cardiomyopathy. Clin. Res. Cardiol. 2016;105:763–773. doi: 10.1007/s00392-016-0986-9. [DOI] [PubMed] [Google Scholar]
- 261.Kuhl U, et al. Chromosomally integrated human herpesvirus 6 in heart failure: prevalence and treatment. Eur. J. Heart Fail. 2015;17:9–19. doi: 10.1002/ejhf.194. [DOI] [PubMed] [Google Scholar]
- 262.Tschope C, Elsanhoury A, Schlieker S, Van Linthout S, Kuhl U. Immunosuppression in inflammatory cardiomyopathy and parvovirus B19 persistence. Eur. J. Heart Fail. 2019;21:1468–1469. doi: 10.1002/ejhf.1560. [DOI] [PubMed] [Google Scholar]
- 263.Ameling S, et al. Changes of myocardial gene expression and protein composition in patients with dilated cardiomyopathy after immunoadsorption with subsequent immunoglobulin substitution. Basic Res. Cardiol. 2016;111:53. doi: 10.1007/s00395-016-0569-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.McNamara DM, et al. Controlled trial of intravenous immune globulin in recent-onset dilated cardiomyopathy. Circulation. 2001;103:2254–2259. doi: 10.1161/01.CIR.103.18.2254. [DOI] [PubMed] [Google Scholar]
- 265.Maisch B, et al. Treatment of inflammatory dilated cardiomyopathy and (peri)myocarditis with immunosuppression and i.v. immunoglobulins. Herz. 2004;29:624–636. doi: 10.1007/s00059-004-2628-7. [DOI] [PubMed] [Google Scholar]
- 266.Sudano I, et al. Cardiovascular disease in HIV infection. Am. Heart J. 2006;151:1147–1155. doi: 10.1016/j.ahj.2005.07.030. [DOI] [PubMed] [Google Scholar]
- 267.Baik SH, et al. A case of influenza associated fulminant myocarditis successfully treated with intravenous peramivir. Infect. Chemother. 2015;47:272–277. doi: 10.3947/ic.2015.47.4.272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Ito N, et al. Influenza A H1N1 pdm09-associated myocarditis during zanamivir therapy. Pediatr. Int. 2015;57:1172–1174. doi: 10.1111/ped.12712. [DOI] [PubMed] [Google Scholar]
- 269.Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;323:1824–1836. doi: 10.1001/jama.2019.20153. [DOI] [PubMed] [Google Scholar]
- 270.McNamara DM, et al. Clinical and demographic predictors of outcomes in recent onset dilated cardiomyopathy: results of the IMAC (Intervention in Myocarditis and Acute Cardiomyopathy)-2 study. J. Am. Coll. Cardiol. 2011;58:1112–1118. doi: 10.1016/j.jacc.2011.05.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Mann DL, et al. Targeted anticytokine therapy in patients with chronic heart failure: results of the Randomized Etanercept Worldwide Evaluation (RENEWAL) Circulation. 2004;109:1594–1602. doi: 10.1161/01.CIR.0000124490.27666.B2. [DOI] [PubMed] [Google Scholar]
- 272.Pinkert S, et al. Prevention of cardiac dysfunction in acute coxsackievirus B3 cardiomyopathy by inducible expression of a soluble coxsackievirus-adenovirus receptor. Circulation. 2009;120:2358–2366. doi: 10.1161/CIRCULATIONAHA.108.845339. [DOI] [PubMed] [Google Scholar]
- 273.Pinkert S, et al. Early treatment of coxsackievirus B3-infected animals with soluble coxsackievirus-adenovirus receptor inhibits development of chronic coxsackievirus B3 cardiomyopathy. Circ. Heart Fail. 2019;12:e005250. doi: 10.1161/CIRCHEARTFAILURE.119.005250. [DOI] [PubMed] [Google Scholar]
- 274.Kraft L, Erdenesukh T, Sauter M, Tschope C, Klingel K. Blocking the IL-1β signalling pathway prevents chronic viral myocarditis and cardiac remodeling. Basic Res. Cardiol. 2019;114:11. doi: 10.1007/s00395-019-0719-0. [DOI] [PubMed] [Google Scholar]
- 275.Brucato A, et al. Effect of anakinra on recurrent pericarditis among patients with colchicine resistance and corticosteroid dependence: the AIRTRIP randomized clinical trial. JAMA. 2016;316:1906–1912. doi: 10.1001/jama.2016.15826. [DOI] [PubMed] [Google Scholar]
- 276.Scott IC, Hajela V, Hawkins PN, Lachmann HJ. A case series and systematic literature review of anakinra and immunosuppression in idiopathic recurrent pericarditis. J. Cardiol. Cases. 2011;4:e93–e97. doi: 10.1016/j.jccase.2011.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Rodriguez-Gonzalez M, Ruiz-Gonzalez E, Castellano-Martinez A. Anakinra as rescue therapy for steroid-dependent idiopathic recurrent pericarditis in children: case report and literature review. Cardiol. Young. 2019;29:241–243. doi: 10.1017/S1047951118002020. [DOI] [PubMed] [Google Scholar]
- 278.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT03018834 (2020).
- 279.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT03737110 (2020).
- 280.Li Z, Yue Y, Xiong S. Distinct Th17 inductions contribute to the gender bias in CVB3-induced myocarditis. Cardiovasc. Pathol. 2013;22:373–382. doi: 10.1016/j.carpath.2013.02.004. [DOI] [PubMed] [Google Scholar]
- 281.Abou-El-Enein M, Volk HD, Reinke P. Clinical development of cell therapies: setting the stage for academic success. Clin. Pharmacol. Ther. 2017;101:35–38. doi: 10.1002/cpt.523. [DOI] [PubMed] [Google Scholar]
- 282.Koch M, et al. Immunosuppression with an interleukin-2 fusion protein leads to improved LV function in experimental ischemic cardiomyopathy. Int. Immunopharmacol. 2010;10:207–212. doi: 10.1016/j.intimp.2009.11.001. [DOI] [PubMed] [Google Scholar]
- 283.Fan MY, et al. Differential roles of IL-2 signaling in developing versus mature Tregs. Cell Rep. 2018;25:1204–1213. doi: 10.1016/j.celrep.2018.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Van Linthout S, et al. Mesenchymal stem cells improve murine acute coxsackievirus B3-induced myocarditis. Eur. Heart J. 2011;32:2168–2178. doi: 10.1093/eurheartj/ehq467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Hare JM, et al. Randomized comparison of allogeneic versus autologous mesenchymal stem cells for nonischemic dilated cardiomyopathy: POSEIDON-DCM trial. J. Am. Coll. Cardiol. 2017;69:526–537. doi: 10.1016/j.jacc.2016.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Rieger AC, et al. Genetic determinants of responsiveness to mesenchymal stem cell injections in non-ischemic dilated cardiomyopathy. EBioMedicine. 2019;48:377–385. doi: 10.1016/j.ebiom.2019.09.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Tschöpe C, et al. Modulation of the acute defence reaction by eplerenone prevents cardiac disease progression in viral myocarditis. ESC Heart Fail. 2020 doi: 10.1002/ehf2.12887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Lee WS, et al. Cannabidiol limits T cell-mediated chronic autoimmune myocarditis: implications to autoimmune disorders and organ transplantation. Mol. Med. 2016;22:136–146. doi: 10.2119/molmed.2016.00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Branchereau M, Burcelin R, Heymes C. The gut microbiome and heart failure: a better gut for a better heart. Rev. Endocr. Metab. Disord. 2019;20:407–414. doi: 10.1007/s11154-019-09519-7. [DOI] [PubMed] [Google Scholar]
- 290.Lorusso R, et al. Venoarterial extracorporeal membrane oxygenation for acute fulminant myocarditis in adult patients: a 5-year multi-institutional experience. Ann. Thorac. Surg. 2016;101:919–926. doi: 10.1016/j.athoracsur.2015.08.014. [DOI] [PubMed] [Google Scholar]
- 291.Kapur NK, Davila CD, Jumean MF. Integrating interventional cardiology and heart failure management for cardiogenic shock. Interv. Cardiol. Clin. 2017;6:481–485. doi: 10.1016/j.iccl.2017.03.014. [DOI] [PubMed] [Google Scholar]
- 292.Li S, et al. A life support-based comprehensive treatment regimen dramatically lowers the in-hospital mortality of patients with fulminant myocarditis: a multiple center study. Sci. China Life Sci. 2019;62:369–380. doi: 10.1007/s11427-018-9501-9. [DOI] [PubMed] [Google Scholar]
- 293.Kapur NK, et al. Mechanical circulatory support devices for acute right ventricular failure. Circulation. 2017;136:314–326. doi: 10.1161/CIRCULATIONAHA.116.025290. [DOI] [PubMed] [Google Scholar]
- 294.Annamalai SK, et al. The Impella microaxial flow catheter is safe and effective for treatment of myocarditis complicated by cardiogenic shock: an analysis from the global cVAD registry. J. Card. Fail. 2018;24:706–710. doi: 10.1016/j.cardfail.2018.09.007. [DOI] [PubMed] [Google Scholar]
- 295.Spillmann F, et al. Mode-of-action of the PROPELLA concept in fulminant myocarditis. Eur. Heart J. 2019;40:2164–2169. doi: 10.1093/eurheartj/ehz124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Sun M, et al. Experimental right ventricular hypertension induces regional β1-integrin-mediated transduction of hypertrophic and profibrotic right and left ventricular signaling. J. Am. Heart Assoc. 2018;7:e007928. doi: 10.1161/JAHA.117.007928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Lindner D, et al. Cardiac fibroblasts support cardiac inflammation in heart failure. Basic Res. Cardiol. 2014;109:428. doi: 10.1007/s00395-014-0428-7. [DOI] [PubMed] [Google Scholar]
- 298.Levin HR, et al. Reversal of chronic ventricular dilation in patients with end-stage cardiomyopathy by prolonged mechanical unloading. Circulation. 1995;91:2717–2720. doi: 10.1161/01.CIR.91.11.2717. [DOI] [PubMed] [Google Scholar]
- 299.Hata JA, et al. Lymphocyte levels of GRK2 (βARK1) mirror changes in the LVAD-supported failing human heart: lower GRK2 associated with improved β-adrenergic signaling after mechanical unloading. J. Card. Fail. 2006;12:360–368. doi: 10.1016/j.cardfail.2006.02.011. [DOI] [PubMed] [Google Scholar]
- 300.Tschope C, et al. Mechanical unloading by fulminant myocarditis: LV-IMPELLA, ECMELLA, BI-PELLA, and PROPELLA concepts. J. Cardiovasc. Transl. Res. 2019;12:116–123. doi: 10.1007/s12265-018-9820-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Chaparro SV, et al. Combined use of Impella left ventricular assist device and extracorporeal membrane oxygenation as a bridge to recovery in fulminant myocarditis. ASAIO J. 2012;58:285–287. doi: 10.1097/MAT.0b013e31824b1f70. [DOI] [PubMed] [Google Scholar]
- 302.Pappalardo F, et al. Concomitant implantation of Impella((R)) on top of veno-arterial extracorporeal membrane oxygenation may improve survival of patients with cardiogenic shock. Eur. J. Heart Fail. 2017;19:404–412. doi: 10.1002/ejhf.668. [DOI] [PubMed] [Google Scholar]
- 303.Pappalardo F, Scandroglio AM, Latib A. Full percutaneous biventricular support with two Impella pumps: the Bi-Pella approach. ESC Heart Fail. 2018;5:368–371. doi: 10.1002/ehf2.12274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Fiedler A. Uber akute interstitielle Myokarditis. Zentralblatt für Innere Med. 1900;21:212–213. [Google Scholar]
- 305.Sakakibara S, Konno S. Endomyocardial biopsy. Jpn. Heart J. 1962;3:537–543. doi: 10.1536/ihj.3.537. [DOI] [PubMed] [Google Scholar]
- 306.Zanatta A, Carturan E, Rizzo S, Basso C, Thiene G. Story telling of myocarditis. Int. J. Cardiol. 2019;294:61–64. doi: 10.1016/j.ijcard.2019.07.046. [DOI] [PubMed] [Google Scholar]
- 307.Ammirati E, Sormani P, Moroni F, Camici PG, Pedrotti P. Changes of late gadolinium enhancement extension compared with native T1 mapping early after acute myocarditis. Int. J. Cardiol. 2018;257:227. doi: 10.1016/j.ijcard.2017.12.056. [DOI] [PubMed] [Google Scholar]
- 308.Schultz JC, Hilliard AA, Cooper LT, Jr, Rihal CS. Diagnosis and treatment of viral myocarditis. Mayo Clin. Proc. 2009;84:1001–1009. doi: 10.1016/S0025-6196(11)60670-8. [DOI] [PMC free article] [PubMed] [Google Scholar]