Highlights
-
•
Photobiomodulation (PBM) can reduce lung edema, cytokines in bronchoalveolar parenchyma, neutrophil influx.
-
•
PBM reduces TNF-α, IL-1β, IL-6, ICAM-1, MIP-2 and Reactive oxygen species.
-
•
Transthoracic approach is the direct methods for reducing lung inflammation.
-
•
Intravenous approach increases the oxygenation of red blood cells.
Keywords: Photobiomodulation, COVID-19, SARS Cov2, Low-level laser therapy, Virus, Laser therapy
Abstract
Background
COVID-19, as a newly-emerged viral infection has now spread all over the world after originating in Wuhan, China. Pneumonia is the hallmark of the disease, with dyspnea in half of the patients and acute respiratory distress syndrome (ARDS) in up to one –third of the cases. Pulmonary edema, neutrophilic infiltration, and inflammatory cytokine release are the pathologic signs of this disease. The anti-inflammatory effect of the photobiomodulation (PBM) has been confirmed in many previous studies. Therefore, this review study was conducted to evaluate the direct effect of PBM on the acute lung inflammation or ARDS and also accelerating the regeneration of the damaged tissues. The indirect effects of PBM on modulation of the immune system, increasing the blood flow and oxygenation in other tissues were also considered.
Methodology
The databases of PubMed, Cochrane library, and Google Scholar were searched to find the relevant studies. Keywords included the PBM and related terms, lung inflammation, and COVID-19 -related signs. Studies were categorized with respect to the target tissue, laser parameters, and their results.
Results
Seventeen related papers were included in this review. All of them were in animal models. They showed that the PBM could significantly decrease the pulmonary edema, neutrophil influx, and generation of pro-inflammatory cytokines (tumor necrosis factor-α (TNF-α), interleukin 1 beta (IL-1β), interleukin 6 (IL-6), intracellular adhesion molecule (ICAM), reactive oxygen species (ROS), isoform of nitric oxide synthase (iNOS), and macrophage inflammatory protein 2 (MIP-2)).
Conclusion
Our findings revealed that the PBM could be helpful in reducing the lung inflammation and promoting the regeneration of the damaged tissue. PBM can increase the oxygenation indirectly in order to rehabilitate the affected organs. Thus, the infra-red lasers or light-emitting diodes (LEDs) are recommended in this regard.
1. Introduction
Coronaviridae is a family of the enveloped RNA viruses, which can infect the humans or other mammals. The two beta corona viruses known as SARS CoV and MERS CoV are among some of the well-known viruses within this family. In the recent decades, two epidemics of the acute respiratory syndrome (ARS) including severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS) have occurred. Mild to severe pneumonia was the main feature of these syndromes. Beginning in December 2019, a novel type of coronavirus was emerged in Wuhan, China that caused a disease known as corona virus disease 2019 (COVID-19) [1]. High transmissibility is the hallmark of this virus. The rapid worldwide distribution of the COVID-19 led to the declaration of a pandemic by the world health organization (WHO).
Epidemiologic data have shown that the median age of the patients is between 49 and 57 years old [2]. High fever, myalgia, dry cough, and dyspnea are the most common clinical symptoms of the disease [3]. Pneumonia is the hallmark of this infection. It can be distinguished from other probable types of pneumonia by the “crazy paving” lesions observed in the chest CT scans. They are the bilateral ground glass opacities in the parenchyma of the lungs [2]. Diarrhea, sputum production, headache, haemoptysis, rhinorrhea, sneezing and sore throat are other common symptoms of this disease [1], [3]. Clinical data have suggested that the pneumonia could cause dyspnea in roughly half of the patients (Liu, 2019). One -third of the patients may develop the acute respiratory distress syndrome (ARDS) and require admission to an intensive care unit (ICU). Invasive mechanical ventilation is needed in 17 and 4% of the patients who are candidates for the extracorporeal membrane oxygenation (ECMO). Older or smoker patients are more likely to require the invasive life support [2]. Lower blood oxygenation and tissue hypoxia can be expected following the damage to the respiratory system. Concomitant tissue hypoxia can lead to a subsequent damage to the heart or central nervous system, considering their sensitivity to the blood oxygen supply.
The paraclinical findings include the hypoalbuminemia, leukopenia, lymphopenia, and low platelet counts. Increased serum level of C-reactive protein, aminotransferase [2], and lactate dehydrogenase (LDH) can be seen in these patients. The number of cluster of differentiation 8(CD8) cells may be reduced during the course of the disease [3]. Levels of some inflammatory cytokines, such as IL-1B, IL-6, interferon-gamma (IFNγ), interferon-gamma-inducible protein-10 (IP10) and monocyte chemotactic protein-1 (MCP1) increase in the serum. These cytokines increase the number and function of T helper type 1 (Th1) lymphocytes. Meanwhile, activation of T helper type 2 (Th2) lymphocytes following the release of interleukin 4(IL-4) and interleukin 10(IL-10) could also be expected. In the patients with severe disease admitted to the ICU, high levels of granulocyte colony-stimulating factor (GCSF), IP10, MCP1, macrophage inflammatory protein 1 A (MIP1A), and TNFα have been commonly found [1]. In a recent study, it was suggested to consider a “cytokine storm” in the COVID-19. Cytokine storm results in an influx of a lot of immune cells into the infection site, damage of the vascular barrier or capillaries, and may also lead to the multi-organ failure. An increase was observed in the level of many cytokines in the COVID-19 disease including IL-1β, interleukin 7 (IL-7), interleukin 8 (IL-8), interleukin 9 (IL-9), IL-10, fibroblast growth factor (FGF), G-CSF, granulocyte macrophage colony-stimulating factor (GM-CSF), IFN-γ, IP-10, MCP-1, MIP-1A, macrophage inflammatory protein 1B (MIP1-B), platelet-derived growth factor (PDGF), TNF-α, and vascular endothelial growth factor (VEGF). IL-1, TNF-α, and IL-6 are the most important pro-inflammatory cytokines. It was found that the IL-6 has a pivotal role in the COVID-19 disease especially it is associated with higher mortality [4].
Although, the clinical features of the COVID-19 are less severe than the SARS or MERS, it is more easily transmitted. The mortality rate of the COVID-19 is equal to 2–3%, while it is equal to 10 and 37% in the SARS and MERS, respectively. Presently, there is no gold standard treatment protocol available for the COVID-19. Supportive protocols are recommended most often. There is a considerable controversy about the beneficial effects of the antiviral medications and corticosteroids, although dexamethasone has been shown to be effective in reducing the deaths recently. The efficacy of the lopinavir plus ritonavir [5] has also been reported in treating the MERS. In the meantime, remedesivir [6], [7] has been indicated to have a good antiviral effect in treatment of the SARS/MERS. Since, the corticosteroids can alleviate the inflammation and subsequent injury, they are also recommended for the severe cases in which standard supportive procedures were not adequate. However, there is no evidence stating that the steroids can reduce the mortality rate in ARDS in general [8]. Corticosteroids can prolong the virus clearance [1] and might cause immune suppression with concomitant susceptibility to the secondary infections [8], [9]. Hyperglycemia, heart attack, hypertension, and gastrointestinal bleeding are some important side effects of the corticosteroids [8]. It should also be mentioned that nearly half of the COVID-19 patients have underlying systemic diseases like diabetes, hypertension, and cardiovascular diseases. Therefore, corticosteroid prescription might worsen the patient’s underlying systemic disease. Another point is the existence of genetic polymorphisms, which can influence the patient’ response to the corticosteroids. This issue might explain why some patients with ARDS may experience harmful outcomes after steroid administration [8].
The pandemic of COVID-19 underlines the importance of supportive protocols, which can reduce the inflammation in the severe cases. As previously mentioned, comprehensive evaluation of the patient’s medical history is necessary to catalogue the underlying systemic diseases and the used medications. The situation is challenging in emergencies where there are many infected patients along with a shortage of medical services as observed in the COVID-19 pandemic. At the moment, there is no definitive treatment, so the therapeutic protocols should be confined to supportive treatments in order to alleviate the inflammation.
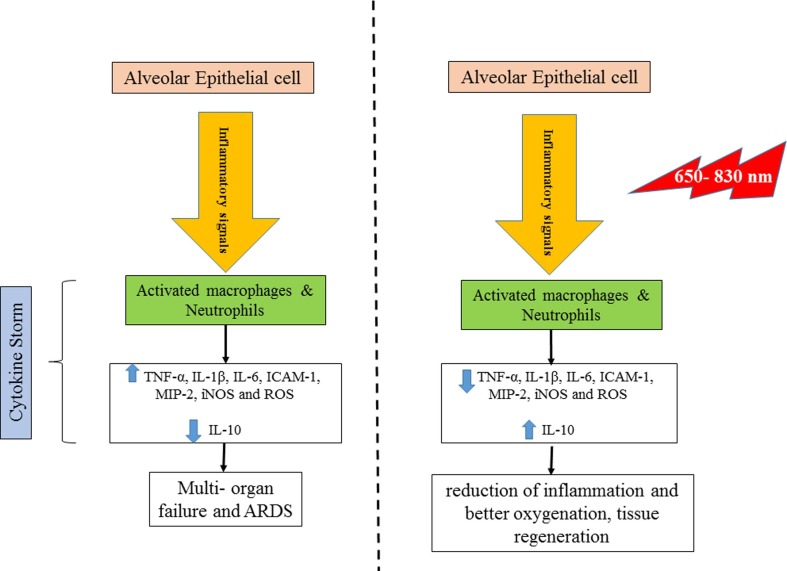
Photobiomodulation could be a promising novel treatment approach. In this non-invasive method, light-emitting diodes or low-level lasers are used to irradiate on the tissue in order to activate the cellular photo-acceptors. Irradiation is absorbed by the internal photo-acceptors like porphyrins, cytochrome C oxidase, and light -sensitive ion channels. Cytochrome C oxidase is unit IV of the mitochondrial respiratory chain, absorbing the red and near infrared wavelengths. This leads to higher electron transport, increased mitochondrial membrane potential and increased production of the adenosine triphosphate (ATP). Light-sensitive ion channels absorb the photons, which increases the concentration of the intracellular calcium (Ca2 + ) ions. These processes activate several signaling pathways via reactive oxygen species (ROS), cyclic adenosine monophosphate (cAMP), nitric oxide (NO), and Ca2+ [10], [11] (Fig. 1 ). These pathways influence the cellular processes like proliferation and differentiation, and can also influence other processes like inflammation as histamine release, prostaglandin production, or cyclo-oxygenase expression [10], [11], [12], [13]. In this way, PBM can be helpful in accelerating the regeneration processes like wound healing and reducing the inflammation [10]. PBM has mainly local effects and can be irradiated just to a target organ without any side effects on the distant areas. Considering the anti-inflammatory and biostimulatory effects of the PBM, it seems to be a reasonable approach to be applied in controlling the COVID-19 symptoms especially in the cases with ARDS.
Fig. 1.
Left) Cytokine storm produced by the corona virus Right) Photobiomodulation effects on the cytokines and inflammatory processes.
Therefore, this study was conducted to evaluate the direct effect of the PBM on the acute lung inflammation or ARDS and accelerating the regeneration of the damaged tissue. Moreover, the indirect effects of the PBM on modulation of the immune system or blood flow oxygenation within the tissues were also investigated.
2. Materials and methods
Our search was categorized based on the common COVID-19 symptoms including respiratory problems and also paraclinical changes influencing the blood cells or cytokines. The review strategy was constructed according to the preferred reporting items for systematic reviews and meta-analyses (PRISMA) checklist. The databases of Medline, PubMed, Cochrane library, and Google Scholar were comprehensively searched to find the relevant studies. Initially, there were no time or language limitations, in order to avoid missing any relevant papers. In the next step, the non-English papers were excluded from the study.
Search keywords included the combination of:
Medical Subject Heading (MeSH) Keywords: Low-Level Light Therapy [MeSH], PBM, Severe Acute Respiratory Syndrome Coronavirus 2 [Supplementary Concept], Viral Envelope Proteins [MeSH], SARS Virus [MeSH]), Respiratory Distress Syndrome, Adult [MeSH], Alveolar Epithelial Cells [MeSH], COVID-19 [Supplementary Concept], Hypoalbuminemia [MeSH], Lymphopenia [MeSH], C-Reactive Protein [MeSH], Blood Cells [MeSH], and neutrophils [MeSH].
Keywords: Light therapy, low -level laser, laser, phototherapy, PBM, photobiostimulation, photodynamic therapy, intravenous laser, corona virus, COVID-19, viral infection, virus, inflammation, lung inflammation, ARDS, acute respiratory distress syndrome, acute lung inflammation, SARS, MERS, alveolar epithelial cells, lung parenchyma, hypoalbuminemia, lymohopenia, C-reactive protein, lymphocyte, neutrophil, oxygenation, and lung regeneration.
The included papers were evaluated for the effect of light therapy, PBM, or low -level laser therapy on the lung inflammation, ARDS, lymphocytes, neutrophils, and lung parenchyma. Papers were categorized with respect to the type of light source, wavelength, target tissue/ organ, light source parameters, and PBM results. References in all of the included papers were also reviewed.
3. Results
Totally, 161 available papers were retrieved from the PubMed database. Twelve papers were excluded due to non-English language and three papers were duplicates. There were 99 available papers on the Cochrane library and none of them were related. Search in the Google scholar database yielded 3960 papers. Fig. 1 shows the final 17 included papers in the form of a diagram. There were some related studies evaluated the effect of the PBM on the chronic respiratory inflammation (such as chronic obstructive pulmonary disease (COPD) and asthma).
Table 1 shows the related papers included in this study. All the studies confirmed that the PBM can reduce the lung inflammation, neutrophil recruitment, and pro-inflammatory cytokine production.
Table 1.
Papers included in the review.
| No | Author | Year | Induction of lung inflammation | Target tissue/ organ | Light source | Light parameters | Energy density (J/cm2) | Power density (mW/cm2) | Evaluation method | Results |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Aimbire et al., [14] | 2005 | Airway and lung inflammation induced by the Gram negative bacterial lipopolysaccharide (LPS) through intravenous injection | Rat’s lung | Ga–Al–As diode laser | 685 nm,12 mW, irradiation time = 1 min 20 s, spot size = 0.08 cm2, Continuous wave |
2.5 | – | Rat’s trachea hyperreactivity (RTHR) Bronchoalveolar lavage (BAL) lung neutrophil influx |
↓ RTHR, ↓BAL ↓Lung neutrophils influx Anti- inflammatory effect was associated with inhibiting the COX-2-derived metabolites |
| 2 | Aimbire et al., (15) | 2006 | Immune complex formation by instillation of the ovalbumin intrabronchially followed by IV injection | Rat’s lung | Ga- -Al-As laser | 650 nm, 42 sec at 5 min after induction, continuous mode, 0.04, 0.11, and 0.22 Joules Spot size = 0.08 cm2 Irradiated through the skin over upper bronchus |
– | 12.5, 31.25, and 62.5 | Bronchoalveolar lavage (BAL) TNF-α level by Cytotoxicity assay through the use of L-929 tumor cells |
↓↓TNF-α expression LLLT reduced the TNF-α level in a dose- dependent manner. The irradiation energy of 0.11 J had the best efficacy. |
| 3 | Aimbire et al.,(16) | 2008 | Acute lung injury induced by IV injection of lipopolysaccharide (LPS) (5 mg/kg) |
Rat’s lung | Diode laser | 660 nm, 30 mW spot size = 0.785 cm2 skin over the upper bronchus |
7.5 | – | Lung permeability via assessing extravasated albumin concentration in the lung homogenate Inflammatory cell (neutrophil) influx by myeloperoxidase (MPO) activity IL-1β in BAL by the enzyme-linked immunosorbent assay (ELISA) IL-1β mRNA expression in the trachea by the real time polymerase chain reaction (RT-PCR) |
↓Lung permeability ↓ Neutrophils influx ↓ MPO activity ↓ IL-1β expression and its mRNA Laser showed an anti-inflammatory effect at 4, 12 and 24 h after LPS exposure. |
| 4 | Mafra de Lima et al.,(17) | 2009 | TNF-a-induced acute lung inflammation | Rat’s dissected bronchi with or without TNF-α | Ga- Al-As laser | 650 nm, 2.5 mW, spot size = 0.08 cm2, 0.44 J, irradiation time = 42 s | 1.3 J/cm2 in two sessions (total dose = 2.6 J/cm2) | 31.25 | Evaluation of the bronchi smooth muscles (BSM) by response to Acetylcholine and Isoproterenol cAMP concentration TNF-α mRNA expression in the BSM tissue by the RT-PCR |
PBM reduced the BSM hyperreactivity or relaxation after Acetylcholine and Isoproterenol application, respectively. ↓TNF-α mRNA expression |
| 5 | Mafra de Lima et al.,(18) | 2010 | Acute pulmonary inflammation induced by aerosol lipopolysaccharide from Escherichia coli (0.3 mg/ mL) |
Rat’s lung | Ga- -Al-As diode laser | 650 nm, 2.5 mW, spot size = 0.08 cm2, irradiation time = 42 s skin over the upper bronchus |
1.3 | 31.2 | Neutrophil recruitment and activation evaluated by edema. BAL fluid cellularity and MPO activity. mRNA expression of TNF-α, IL-1β, IL-10, cytokine -induced neutrophil chemoattractant-1 (CINC-1), MIP-2 and ICAM-1 by the RT-PCR Proteins levels by the ELISA |
↓ Pulmonary edema ↓ Neutrophil influx ↓ Endothelial cytoskeleton damage ↓TNF-α expression ↓IL-1β expression ↓ICAM-1 expression. Levels of CINC-1, MIP-2, and IL-10 were not influenced by the laser irradiation. |
| 6 | Mafra de Lima et al.,(19) | 2010 | 4 h incubation with LPS or H2O2 for inducing the acute lung inflammation | Rat’s AM cell line AMJ2-C11 | Diode laser | 660 nm, 30 mW, spot size = 0.785 cm2, irradiation time = 252 s | 4.5 | MIP-2 mRNA expression by the RT-PCR Intracellular ROS generation by 2′,7″ dichlorofluorescin diacetate (DCFH-DA) NF-kB protein by the ELISA |
↓MIP-2 mRNA expression ↓ Intracellular ROS generation |
|
| 7 | Mafra de Lima et al.,(20) | 2011 | Acute lung inflammation induced by the intestinal ischemia and reperfusion | Rat’s lung | Diode laser | 660 nm, 30 mW, 5.4 J, spot size = 0.08 cm2 Irradiation time = 3 min skin over the upper bronchus |
6.9 | 38.4 | Lung edema by Evans blue extravasation Pulmonary neutrophils influx by the MPO activity TNF and IL-10 expression in the lung by the RT-PCR TNF and IL-10 protein in the lung by the ELISA |
↓Lung edema ↓Neutrophils influx ↓MPO activity ↓TNF expression and production ↑ IL-10 production |
| 8 | Mafra de Lima et al.,(21) | 2011 | Acute lung inflammation induced by the LPS inhalation or TNF intra nasal instillation | Rat’s dissected bronchi | InGaAlP laser | 660 (685) nm, 8.82 J, irradiation time = 252 s On the skin over the right upper bronchus |
4.5 | 17.85 | TNF mRNA expression by the RT- PCR and protein levels by the ELISA cAMP levels by the ELISA |
PBM increased the cAMP level indirectly in the alveolar macrophages by a TNF-dependent mechanism. |
| 9 | Mafra de Lima et al.,(22) | 2013 | Acute lung injury induced by the gut ischemia and reperfusion |
Rat’s lung | Diode laser | 660 nm, 30 mW, 5.4 J, continuous mode, irradiation = 180 s, spot size = 0.08 cm2 skin over the bronchus in direction of the trachea distal two series of laser irradiation: a) 5 min after initial or 5 min before the end of the intestinal reperfusion b)30 min after the beginning of the reperfusion |
6.9 | 38.4 | Lung edema by Evans blue extravasation Bronchoalveolar lavage fluid neutrophils by the MPO activity Injury score by the lung histology ROS generation by fluorescence ICAM-1 and PPARy expression by the RT-PCR Lung HSP70 and glutathione (GSH) protein by the ELISA Brown stain for immunohistochemical localization of the ICAM-1 |
↓Lung edema ↓Neutrophils influx ↓MPO activity ↓ ICAM-1 mRNA expression ↓ROS formation ↑GSH concentration in the lung ↑HSP70 expression ↑peroxisome proliferator-activated receptor y (PPARy) expression PBM reduced the acute lung injury induced by the gut ischemia and reperfusion. |
| 10 | Mafra de Lima et al.,(23) | 2013 | ARDS induced by the intestinal ischemia and reperfusion | Rat’s lung | Diode laser | 660 nm, 30 mW, 5.4 J, continuous mode, irradiation = 3 min spot size = 0.08 cm2 skin over the upper bronchus |
7.5 | 31.25 | Lung edema by Evans blue extravasation Pulmonary neutrophils influx by the MPO activity mRNA expression of the TNF-α, IL-10, ICAM-1, and isoform of NO synthase (iNOS) by the RT-PCR TNF-α, IL-10, and iNOS proteins in the lung by the ELISA |
↓ Airway reactivity dysfunction by:↓lung edema ↓MPO activity ↓ TNF-α expression ↓ iNOS expression ↓ ICAM-1 expression ↑ IL-10. |
| 11 | Mafra de Lima et al.,(24) | 2014 | ARDS induced by the intestinal ischemia and reperfusion | Rat’s lung | Diode laser | 660 nm, 30 mW, 5.4 J, continuous mode, irradiation = 5 min spot size = 0.08 cm2 skin over the upper bronchus |
1, 3, 5 and 7,5 | – | Neutrophils influx by the MPO activity mRNA expression and protein concentration of the IL-1β, IL-6, IL-10, and TNF by the RT-PCR and ELISA, respectively |
↓↓IL-6 mRNA expression ↓↓ TNF mRNA expression However, IL-1β and MPO activity were reduced by all doses except 1 J/cm2. PBM increased the IL-10 protein in the irradiation energy of 1 J/cm2. |
| 12 | Oliveira et al., (25) | 2014 | Acute lung inflammation and ARDS induced by the LPS | Rat’s lung | Infra-red laser | 830 nm, 35 mW, 80 s per point, 3 points per application (total 240 s), continuous mode Direct contact to skin point 1 was in the end part of trachea, points 2 and 3 were in the right and left lungs respectively |
9 | – | Differential cell count in the BAL Levels of cytokines (IL-1beta, IL-6, KC, and TNF-alpha) in the BAL fluid |
PBM significantly reduced the inflammation in the LPS-induced ARDS. ↓Number of total cells ↓Neutrophils in BAL or lung parenchyma Levels in BAL fluid and in serum of: ↓ IL-1beta ↓IL-6 ↓ KC ↓TNF-α. |
| 13 | Miranda da Silva et al.,(26) | 2015 | Lung inflammation induced by the formaldehyde (1%) or vehicle inhalation (distillated water) | Rat’s lung | Diode laser | 660 nm, 30 mW, spot size = 0.14 cm2, 60 s/point, 1.8 J for 1 min | 12.86 | 210 | Blood cell count MPO activity for neutrophil influx Evans blue for lung vascular permeability ELISA for the level of IL-6, TNF-α, and IL-10 in the BAL RT-PCR assay for IL-6, IL-10, and HPRT for gene expression Histology for lung morphological changes |
↓Neutrophil influx (MPO activity) ↓ Leukocyte number ↓ Degranulation of the mast cells ↓ Lung Microvascular permeability. ↓IL-6 ↓ TNF-α ↑ IL-10 generation |
| 14 | Oliveira et al., (27) | 2015 | ARDS induced by the LPS intratracheally or intraperitoneally | Rat’s lung | Infra-red laser | 830 nm, 35mW, 80 s per point 3 points per application) direct contact with the skin |
3 | Total and differential cell counts in the BAL Levels of cytokines (IL-1beta, IL-6, KC, and TNF-alpha) in the BAL and serum Neutrophils count in the lung parenchyma |
PBM significantly reduced the ARDS in both inducing ways. ↓Total cells and neutrophils count in the BAL ↓ Neutrophil count in the lung parenchyma, Levels in the BAL and in serum of: ↓ IL-1beta ↓ IL-6, KC ↓TNF-α IL-10 level was not increased by the PBM. |
|
| 15 | da Silva Sergio et al.,(28) | 2018 | Acute lung injury induced by the intraperitoneal injection of LPS from Escherichia coli | Alveolar epithelial cells of the rat’s lung | -Ga-Al-As diode laser | 808 nm, 100mW, Spot size = 0.028 cm2, 2 and 5 J energy per point, four points of irradiation, time = 2 and 5 s per point Skin over the lung |
10 20 |
3.571 | caspase 3 and Bcl-2 mRNA levels by the RT-qPCR DNA fragmentation by the TUNEL (terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine, 5′-triphosphate nick-end-labeling) assay |
↑ Bcl-2 mRNA levels ↓ caspase-3 mRNA levels DNA fragmentation: ↓ in the alveolar cells but ↑ in polymorphonuclear cells, which induced the apoptosis in these inflammatory cells. |
| Indirect PBM | ||||||||||
| 16 | Fujimaki et al.,(29) | 2003 | Isolated neutrophils from the human’s peripheral blood samples | Human blood | Ga-Al-As laser | 830-nm continuous wave, 1000 mW, spot size 6.6 cm2, irradiation time = 30 or 60 sec | 9.5 19.0 J/cm2 |
150 mW/cm2 | Production of ROS by luminol-dependent chemiluminescence (LmCL) expression of CD11b and CD16 neutrophil surface by the flow cytometry |
PBM attenuated the ROS production in the neutrophils and reduced the oxidative tissue injury. |
| 17 | Al Musawi et al.,(30) | 2016 | Human’s whole blood sample | Human blood | Diode laser | Wavelengths of 405, 589, and 780 nm, 10 mW, spot size = 0.332 cm2, irradiation time = 20, 30, 40, and 50 min | 36, 54, 72, and 90 | 30 | Lymphocyte count Flow cytometry |
Photobiomodulatory effects were related to the laser parameter. Wavelength of 589 nm and fluence of 72 J/cm2 resulted in the best results. ↑↑ CD45 lymphocytes and natural killer (NK) (CD16, CD56) cells There was no significant change for CD3 T lymphocytes, T-suppressor (CD3, CD8) cells, T-helper (CD3, CD4) cells, and CD19 B lymphocytes |
↓ shows the reduction in the target variable. ↓↓ shows a significant reduction in the target variable. ↑ shows an increase in the target variable.↑↑ shows a significant increase in the target variable.
4. Discussion
During the clinical course of the COVID-19 disease, dyspnea can start as early as 5 days after the infection. Coughing is a major sign, indicating an inflammatory reaction in the respiratory system. Sputum production is another common sign of the disease. Ground glass opacities can be found in the chest radiographs and there are more of them in the severe cases. ARDS may occur after 8 days and it can be observed in one-third of the patients. It is a life-threatening condition, which might require invasive mechanical ventilation in 17% of the patients and extracorporeal membrane oxygenation (ECMO) in 4% of the patients. Elderly patients and smokers are more likely to require the invasive life support [2].
Although, there is no definititive treatment protocol for the COVID-19 disease yet, supportive care is the mainstay for management of the COVID-19 infection. Chloroquine, kaletra [31], remedesivir [6], [7], tocilizumab, Favipiravir [31], and a combination of lopinavir and ritonavir [5] are some of the treatment options in this regard, but no definite effect has been observed on the patient’s survival after treatment with these medications [31].
Corticosteroids are the most common anti-inflammatory option. Inhibiting the edema formation, leukocyte extravasation, fibrin deposition, capillary dilation, and phagocytosis are some of positive effects of the corticosteroids. Reduction of inflammation can also be induced by triggering the apoptosis of the eosinophils and lymphocytes [8], [32]. It should be considered that prescription of the corticosteroids should be confined to the ICU-admitted cases with ARDS [8]. They may lead to higher susceptibility to the secondary infections [8], [9] and delay the viral clearance [1].
Chan et al., suggested that the corticosteroids should only be used in the severe ARDS cases where standard supportive measures are insufficient. Corticosteroids should not be prescribed when there is a remaining infection [8]. Also, genetic polymorphisms play a pivotal role in the patient’s response to the corticosteroids. A wide range of adverse reactions make the use of corticosteroids questionable in the ARDS induced by the COVID-19. Other potential side effects of the corticosteroids may affect other organs especially the liver and kidneys as major organs for metabolism. Other side effects include the hyperglycemia, polyneuropathy in the patients staying in the ICU for a long time, higher risk of necrotizing myopathy, risk of secondary infections, delayed wound healing, suppression of calcium absorption leading to the osteoporosis and avascular necrosis of the hip, and gastrointestinal ulcers [8]. Also, the corticosteroids may have negative interactions with other medications taken by the patients. This situation is more noticeable in the COVID-19 patients where nearly half of them are older and usually have underlying systemic diseases like diabetes, hypertension, cardiovascular diseases, etc., requiring the additional medications or have compromised immune systems. Moreover, COVID-19 sharply lowers the blood oxygenation, which can directly damage the pulmonary tissues or indirectly damage the other organs like the heart or brain by inducing the hypoxia.
Our experience in the recent COVID-19 pandemic suggests that the clinicians should consider a comprehensive evaluation of the patient’s medical condition, medications, systemic diseases, genetic polymorphisms that may affect the drug reactions. These factors make it complex for the clinicians to manage an emergency like the COVID-19, involving large numbers of the infected people along with insufficient medical services. As mentioned above, there is not yet any definite treatment for the COVID-19 therefore, every possible intervention, which may help to reduce the inflammation and restore the respiratory system or other impaired tissues could be tried for managing the COVID-19 progression. This could be achieved by direct rehabilitation of the damaged tissue, or indirectly by increasing the oxygenation and blood flow. So, reducing the inflammation and aiding the tissue regeneration are the major goals of the clinicians. It is important for the clinicians to choose the treatment modalities with the least drug interactions or side effects.
Photobiomodulation is an innovative approach in this regard. It is a non-invasive approach in which the LEDs or low -level lasers are used to produce the red or near- infrared (NIR) light absorbed by the cellular photo-acceptors. This light absorption produces the ROS, such as singlet oxygen, hydrogen peroxide (H2O2), and superoxide [33], [34], [35]. These ROS affect many cellular processes, such as proliferation, differentiation [12], [13], [36], [37], [38], adenosine triphosphate (ATP) formation [39], [40] and also can reduce the inflammtion [10]. So, PBM is a helpful approach when the cellular function is impaired especially by the hypoxia [41]. Previous studies have shown the advantages of the PBM including anti-inflammatory effects and acceleration of the wound healing [10]. These features operate alongside the general effects of the PBM for balancing the metabolic, analgesic, and immunomodulatory conditions. One advantage of this approach is that it is applied locally without any systemic side effects on the other organs [31]. Based on the aforementioned advantages of the PBM and the current lack of established treatments for COVID-19 disease, it seems that the PBM could be helpful in controlling the COVID-19 disease as an alternative or adjunctive treatment, particularly in the severe cases with ARDS.
Table 1 shows the related papers used the PBM for treatment of lung inflammation or ARDS. The majority of studies have used the red diode lasers (650 and 660 nm) with an energy density within the range of 1–12.86 J/cm2 and power density within the range of 12.5–210 mW/cm2. Three papers [25], [27], [28] have used the infra-red lasers (808 and 830 nm) with an energy density within the range of 3–20 J/cm2 and there was not sufficient information about the power density except in one paper (3.571 mW/cm2 [28]. All the papers have shown the anti-inflammatory effects of the PBM including reducing the lung edema, cytokines in the bronchoalveolar lavage (BAL) fluid, neutrophil influx, myeloperoxidase (MPO) activity, and damage to the endothelial cytoskeleton.
The mechanisms of action of the PBM involve many factors related to the inflammation such as:
-
1-
Activating the interferons (IFNs) having a pivotal role in the defense against viruses and the modulation of the immune system. Huang et al., [1] found a higher level of IFN-γ in the COVID-19 patients. IFN-γ has antiviral and anti-tumor activity, and also increases the T lymphocytes and natural killer cells. It can regulate the immune system reactions. IFN-α and IFN-β stimulate the natural killer cells and macrophages, they can also stimulate the lymphocytes and macrophages in order to improve the antiviral or anti-tumor activity [42], [43], [44].
-
2-
PBM can also activate the phagocytes engulfing and removing the microorganisms (like bacteria or viruses) as well as apoptotic cells [27], [45].
-
3-
PBM can increase the micro and macro-circulation in order to increase the tissue or organ resistance against the external harmful factors [46].
-
4-
It also can increase the oxygen saturation of the tissues, which in turn increases the cell metabolism and capacity for proliferation or regeneration of the damaged tissue [47].
Based on Table 1, there are several cytokines influenced by the PBM.
-
1-
TNF-α
According to the literature, PBM can reduce the expression of TNF-α mRNA and its production levels [15], [17], [18], [20], [21], [23], [24], [25], [26], [27]. Neutrophil adhesion and activation can be influenced by the TNF-α and also IL-6 generation can be stimulated by the TNF-α. Moreover, TNF-α can promote the coagulation and edema in the acute lung inflammation [15], [48].
-
2-
Interleukin-1beta (IL-1β)
IL-1β, as the main inflammatory cytokine contributes to the initiation of inflammation. Neutrophils are the main source of this cytokine and it increases the survival rate of the neutrophils in a reciprocal manner. Severe cases of ARDS with poor prognosis show higher levels of IL-1β. Many studies have confirmed its effects in the acute lung inflammation. It has been found that the PBM can reduce the production of this cytokine [16], [18], [24], [25]. Mafra de Lima et al. [24] revealed that the IL-1β expression and concentration were not reduced by the energy dose of 1 J/cm2, but on the other hand, they were reduced by higher doses (3, 5 and 7.5 J/cm2). Therefore, it is necessary to provide a sufficient energy dose to the target tissue, especially the lung parenchyma. Laser irradiation will be attenuated by the passage through overlying tissue, such as intercostal muscles and skin.
-
3-
Interleukin-6 (IL-6)
PBM can reduce the IL-6 levels during the acute lung inflammation or ARDS [24], [25], [26], [27]. This cytokine prolongs the duration of inflammation [49], [50] and is related to the poor prognosis in ARDS [49], [51], [52], [53].
-
4-
Interleukin-10 (IL-10)
Studies have shown that the low -level laser can increase the IL-10 generation. IL-10, as an anti-inflammatory cytokine can modulate the production of other inflammatory cytokine or reduce the tissue injury. It has been suggested that there is a balance between the TNF and IL-10 production [54]. So, the TNF production can be reduced by promoting the IL_10 formation by the PBM. This protective effect has been demonstrated in several studies [20], [23], [24], [26]. It has been found that the PBM increases the IL-10 production. In contrast, two studies [18], [27] have shown that the low -level laser does not influence the IL-10 production.
-
5-
Intercellular Adhesion Molecule-1 (ICAM-1)
Neutrophil recruitment and their chemotaxis from the bloodstream to the lung parenchyma is one of the hallmarks of the acute lung inflammation or ARDS. There are some cytokine- induced neutrophil chemoattractants (CINCs) present on the neutrophils and endothelial cells. CD18 integrin is present on the polymorphonuclear leukocytes (PMNs). However, ICAM-1 is a related adhesion molecule on the endothelial cells. It has been shown that the PBM reduces the ICAM-1 expression [18], [22], [23]. It has been suggested that the inhibitory effect of the PBM on ICAM-1 expression may be related to the suppression of TNF-α and IL-1β production. This may be another mechanism by which the PBM reduces the lung inflammation.
-
6-
Macrophage Inflammatory Protein-2 (MIP-2)
MIP-2 is another neutrophil chemoattractant. It is secreted by the monocytes and neutrophils at sites of inflammation. It can attract the leukocytes and hematopoietic cells. Therefore, inhibition of MIP-2 generation by the PBM may have a pivotal role in reducing the lung edema [18], [19].
-
7-
ROS and Isoforms of NO Synthase (iNOS)
ROS can be produced by the laser irradiation of the cells. Superoxide or hydrogen peroxide (H2O2) are some examples in this regard [33], [34], [35]. ROS can change the cellular process as the secondary messengers at low concentrations. Nitric oxide (NO) is beneficial at low concentrations, but at high concentrations, it can react to produce the reactive nitrogen species (RNS), such as peroxynitrite. Both ROS and RNS are destructive at high doses. In the lung inflammation, the increased neutrophil influx produces high concentrations of ROS and RNS damaging the lung tissue. Some studies have shown that the PBM can reduce the ROS formation [19], [22] or lower the iNOS levels [23].
There are two approaches to deliver the light used in the PBM.
4.1. Transthoracic approach
This involves a direct irradiation of the target organs [31]. In the COVID-19 disease, the lungs can be irradiated through the chest skin or through the back area when the patient is in the prone position. Studies on the transcranial irradiation have demonstrated that the light can penetrate through the scalp and skull to reach the brain [55]. As mentioned above, red and NIR wavelengths have been used in this regard. PBM should irradiate through the interchondral space so that, the laser clusters are applied over the entire surface of the thorax. Infrared wavelengths (e.g., 810 nm) are suggested for the PBM, because less laser irradiation will be absorbed by the overlying tissues. Infrared lasers have lower tissue absorption compared to the red lasers.
High-intensity laser therapy (HILT) may be helpful to achieve deeper penetration of the laser light to the underlying tissues, especially lung tissues. HILT has been shown to reduce the inflammation by a non-invasive approach [56].
The lack of any long-term toxicity, gene mutations, and damage to other organs are among the potential advantages of this approach. There has not been any bacteremia observed after irradiation, which is beneficial for the immune-suppressed patients.
4.2. Intravenous approach
This approach increases the oxygenation of the red blood cells, and influences the biomarkers in order to indirectly decrease the inflammation or regenerate the damaged tissues. This approach can be carried out intravenously, transmucosally, or transcutaneously over the superficial arteries. Blood vessels in the easily accessible areas like the nasal mucosa, under the tongue, behind the knee, or on the wrist can be irradiated with the laser or LEDs. Oxygenation can be increased by the green lasers however; viruses could be cleared by the blue laser irradiation. Red lasers improve the ATP production. [31], [57] Therefore, using visible lasers is recommended in this approach with an adjusted dosage.
Two studies have supported the use of this approach [29], [30]. They have shown that the laser parameters (wavelength, energy density, etc.) have a critical role in the therapeutic effects [30]. It has been shown that the PBM could reduce the ROS production in the neutrophils in peripheral blood [29], and PBM could regulate the immune system by increasing the lymphoctes and neutral killer cells involved in the defense against the viruses [30].
There was just one clinical study which successfully treated a female patient with ARDS following a secondary viral infection [58]. Clinicians used the PBM (820 nm and 50 mW) in order to reduce the lung inflammation. Laser was delivered to the skin between the ribs. This study was a case-report, so it was excluded from our study.
Other immunomodulatory effects of the PBM may be expected by irradiating the lymphoid tissues like the thymus, spleen, bone marrow, or the lymphatic system.
5. Conclusion
Despite the lack of available studies on the PBM effects on the COVID-19, but it is assumed that the PBM may be a helpful adjunctive treatment in management of the COVID-19 disease. PBM can reduce the lung edema, neutrophil influx and promote the regeneration of the lung tissue and better oxygenation for all the related organs. Infra-red lasers are recommended because of their higher ability for penetration into the lung tissue. Dosimetry of 6.5–7.5 J/cm2 energy density is suitable for the red lasers and an amount of 9.5–10.5 J/cm2 is a suitable dose for the infra-red lasers. Continous mode irradiation at different points of the respiratory system may be helpful in management of the COVID-19 pneumonia. PBM may be used as a preventive approach in the high -risk patients who could receive pre-treatment PBM while being still at a relatively mild stage of the disease. Also, PBM may be considered as a therapeutic approach in the hospitalized patients before their condition worsens sufficiently to require the ICU admission. Therefore, randomized clinical trials should be carried out on the PBM effects for the COVID-19 disease, and indeed some have already been started in various parts of the world.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y. Clinical features of patients infected with 2019 novel coronavirus in Wuhan. China. The lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Petrosillo N., Viceconte G., Ergonul O., Ippolito G., Petersen E.J.C.M. Infection. COVID-19, SARS and MERS: are they closely related? Clin. Microbiol. Infect. 2020 doi: 10.1016/j.cmi.2020.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Liu Y., Yang Y., Zhang C., Huang F., Wang F., Yuan J. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci. China Life Sci. 2020;63(3):364–374. doi: 10.1007/s11427-020-1643-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ragab D., Salah Eldin H., Taeimah M., Khattab R., Salem R. The COVID-19 Cytokine Storm. What We Know So Far. 2020;11(1446) doi: 10.3389/fimmu.2020.01446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Arabi Y.M., Alothman A., Balkhy H.H., Al-Dawood A., AlJohani S., Al Harbi S. Treatment of Middle East Respiratory Syndrome with a combination of lopinavir-ritonavir and interferon-β1b (MIRACLE trial): study protocol for a randomized controlled trial. Trials. 2018;19(1):81. doi: 10.1186/s13063-017-2427-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sheahan T.P., Sims A.C., Graham R.L., Menachery V.D., Gralinski L.E., Case J.B. Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses. Sci. Transl. Med. 2017;9(396) doi: 10.1126/scitranslmed.aal3653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sheahan T.P., Sims A.C., Leist S.R., Schäfer A., Won J., Brown A.J. Comparative therapeutic efficacy of remdesivir and combination lopinavir, ritonavir, and interferon beta against MERS-CoV. Nat. Commun. 2020;11(1):1–14. doi: 10.1038/s41467-019-13940-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chan E.D., Chan M.M., Chan M.M., Marik P.E.J.P. Therapeutics. Use of glucocorticoids in the critical care setting: Science and clinical evidence. Pharmacol. Ther. 2020;206:107428. doi: 10.1016/j.pharmthera.2019.107428. [DOI] [PubMed] [Google Scholar]
- 9.Cain D.W., Cidlowski J.A. Immune reulation by glucocorticoids. Nat. Rev. Immunol. 2017;17(4):233–247. doi: 10.1038/nri.2017.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tuner J., Biostimulation L.H. Prima Books AB; Grangesberg, Sweden: 2007. The Laser Therapy Handbook; pp. 61–94. [Google Scholar]
- 11.de Freitas L.F., Hamblin MRJIJostiqe Proposed mechanisms of photobiomodulation or low-level light therapy. IEEE J. Sel. Top. Quantum Electron. 2016;22(3):348–364. doi: 10.1109/JSTQE.2016.2561201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fekrazad R., Asefi S., Allahdadi M., Kalhori K.A.J.P. surgery l. Effect of photobiomodulation on mesenchymal stem cells. Photomed. Laser Surg. 2016;34(11):533–542. doi: 10.1089/pho.2015.4029. [DOI] [PubMed] [Google Scholar]
- 13.Khorsandi, K., Hosseinzadeh, R., Abrahamse, H., Fekrazad, R., 2020. Biological responses of stem cells to photobiomodulation therapy, Curr. Stem Cell Res. Ther. [DOI] [PubMed]
- 14.Aimbire F., Albertine R., de Magalhães R., Lopes-Martins R., Castro-Faria-Neto H., Zângaro R. Effect of LLLT Ga–Al–As (685 nm) on LPS-induced inflammation of the airway and lung in the rat. Lasers Med. Sci. 2005;20(1):11–20. doi: 10.1007/s10103-005-0339-9. [DOI] [PubMed] [Google Scholar]
- 15.Aimbire F., Albertini R., Pacheco M., Castro-Faria-Neto H.C., Leonardo P., Iversen V.V. Low-level laser therapy induces dose-dependent reduction of TNFα levels in acute inflammation. Photomed. Laser Surg. 2006;24(1):33–37. doi: 10.1089/pho.2006.24.33. [DOI] [PubMed] [Google Scholar]
- 16.Aimbire F., De Oliveira A.L., Albertini R., Correa J., De Campos C.L., Lyon J. Low level laser therapy (LLLT) decreases pulmonary microvascular leakage, neutrophil influx and IL-1β levels in airway and lung from rat subjected to LPS-induced inflammation. Inflammation. 2008;31(3):189. doi: 10.1007/s10753-008-9064-4. [DOI] [PubMed] [Google Scholar]
- 17.Mafra de Lima F., Costa M., Albertini R., Silva J., Jr, Aimbire FJLiS, Medicine MTOJotASfL Low level laser therapy (LLLT): Attenuation of cholinergic hyperreactivity, β2-adrenergic hyporesponsiveness and TNF-α mRNA expression in rat bronchi segments in E. coli lipopolysaccharide-induced airway inflammation by a NF-κB dependent mechanism. Lasers Surg. Med. 2009;41(1):68–74. doi: 10.1002/lsm.20735. [DOI] [PubMed] [Google Scholar]
- 18.de Lima F.M., Villaverde A., Salgado M., Castro-Faria-Neto H., Munin E., Albertini R. Low intensity laser therapy (LILT) in vivo acts on the neutrophils recruitment and chemokines/cytokines levels in a model of acute pulmonary inflammation induced by aerosol of lipopolysaccharide from Escherichia coli in rat. J. Photochem. Photobiol. B. 2010;101(3):271–278. doi: 10.1016/j.jphotobiol.2010.07.012. [DOI] [PubMed] [Google Scholar]
- 19.de Lima F.M., Villaverde A.B., Albertini R., de Oliveira A.L., Neto H.C.F., Aimbire F.J.P. Low-level laser therapy associated to N-acetylcysteine lowers macrophage inflammatory protein-2 (MIP-2) mRNA expression and generation of intracellular reactive oxygen species in alveolar macrophages. Photomed. Laser Surg. 2010;28(6):763–771. doi: 10.1089/pho.2009.2638. [DOI] [PubMed] [Google Scholar]
- 20.de Lima F.M., Villaverde A., Albertini R., Corrêa J., Carvalho R., Munin E. Dual Effect of low-level laser therapy (LLLT) on the acute lung inflammation induced by intestinal ischemia and reperfusion: Action on anti-and pro-inflammatory cytokines. Lasers Surg. Med. 2011;43(5):410–420. doi: 10.1002/lsm.21053. [DOI] [PubMed] [Google Scholar]
- 21.de Lima F.M., Moreira L.M., Villaverde A., Albertini R., Castro-Faria-Neto H.C., Aimbire FJLims Low-level laser therapy (LLLT) acts as cAMP-elevating agent in acute respiratory distress syndrome. Lasers Med. Sci. 2011;26(3):389–400. doi: 10.1007/s10103-010-0874-x. [DOI] [PubMed] [Google Scholar]
- 22.de Lima F.M., Albertini R., Dantas Y., Maia-Filho A.L., de Loura Santana C, Castro-Faria-Neto H.C. Low-level laser therapy restores the oxidative stress balance in acute lung injury induced by gut ischemia and reperfusion. Photochem. Photobiol. 2013;89(1):179–188. doi: 10.1111/j.1751-1097.2012.01214.x. [DOI] [PubMed] [Google Scholar]
- 23.de Lima F.M., Vitoretti L., Coelho F., Albertini R., Breithaupt-Faloppa A.C., de Lima W.T. Suppressive effect of low-level laser therapy on tracheal hyperresponsiveness and lung inflammation in rat subjected to intestinal ischemia and reperfusion. Lasers Med. Sci. 2013;28(2):551–564. doi: 10.1007/s10103-012-1088-1. [DOI] [PubMed] [Google Scholar]
- 24.de Lima F.M., Aimbire F., Miranda H., de Paula Vieira R, de Oliveira A.P.L., Albertini RJJolims Low-level laser therapy attenuates the myeloperoxidase activity and inflammatory mediator generation in lung inflammation induced by gut ischemia and reperfusion: a dose-response study. J. Lasers Med. Sci. 2014;5(2):63. [PMC free article] [PubMed] [Google Scholar]
- 25.Oliveira M.C., Jr, Greiffo F.R., Rigonato-Oliveira N.C., Custódio R.W.A., Silva V.R., Damaceno-Rodrigues N.R. Low level laser therapy reduces acute lung inflammation in a model of pulmonary and extrapulmonary LPS-induced ARDS. J. Photochem. Photobiol. B. 2014;134:57–63. doi: 10.1016/j.jphotobiol.2014.03.021. [DOI] [PubMed] [Google Scholar]
- 26.da Silva C.M., Leal M.P., Brochetti R.A., Braga T., Vitoretti L.B., Camara N.O.S. Low level laser therapy reduces the development of lung inflammation induced by formaldehyde exposure. PLoS ONE. 2015;10(11) doi: 10.1371/journal.pone.0142816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.de Oliveira Junior M.C., Brito A.A., Oliveira N.C.R., Oliveira A.R.A., Junior J.C.J.A., Souza A.S.A. Low level laser therapy (830nm) decreases LPS-induced acute respiratory distress syndrome independent of IL-10 release. PLoS ONE. 2015;10(11) [Google Scholar]
- 28.da Silva Sergio L.P., Thomé A.M.C., Trajano L.Ad.S.N., Mencalha A.L., da Fonseca Ad.S., de Paoli F.J.P. Photobiomodulation prevents DNA fragmentation of alveolar epithelial cells and alters the mRNA levels of caspase 3 and Bcl-2 genes in acute lung injury. Photochem. Photobiol. Sci. 2018;17(7):975–983. doi: 10.1039/c8pp00109j. [DOI] [PubMed] [Google Scholar]
- 29.Fujimaki Y., Shimoyama T., Liu Q., Umeda T., Nakaji S., Sugawara KJJoclm Low-level laser irradiation attenuates production of reactive oxygen species by human neutrophils. J. Clin. Laser Med. Surg. 2003;21(3):165–170. doi: 10.1089/104454703321895635. [DOI] [PubMed] [Google Scholar]
- 30.Musawi M.S.A., Jafar M.S., Al-Gailani B.T., Ahmed N.M., Suhaimi F.M., Suardi N.J.P. In vitro mean red blood cell volume change induced by diode pump solid state low-level laser of 405 nm. Photomed. Laser Surg. 2016;34(5):211–214. doi: 10.1089/pho.2015.4043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fekrazad R. Photobiomodulation and Antiviral Photodynamic Therapy as a Possible Novel Approach in COVID-19 Management. Photobiomodul. Photomed. Laser Surg. 2020 doi: 10.1089/photob.2020.4868. [DOI] [PubMed] [Google Scholar]
- 32.Walsh G.M., Sexton D.W., Blaylock MGJTJoe Corticosteroids, eosinophils and bronchial epithelial cells: new insights into the resolution of inflammation in asthma. J. Endocrinol. 2003;178(1):37–43. doi: 10.1677/joe.0.1780037. [DOI] [PubMed] [Google Scholar]
- 33.V. Derr, S. Fine, editors. FREE RADICAL OCCURRENCE IN SOME LASER-IRRADIATED BIOLOGIC MATERIALS. Federation proceedings; 1965. [PubMed]
- 34.Lubart R., Malik Z., Rochkind S., Fisher T.J.L.T. A possible mechanism of low level laser-living cell interaction. Laser Therapy. 1990;2(2):65–68. [Google Scholar]
- 35.Mrowiec J., Sieron A., Plech A., Cieslar G., Biniszkiewicz T., Brus R. Analgesic effect of low-power infrared laser radiation in rats. Proc. SPIE. 1997;3198:83–89. [Google Scholar]
- 36.Nasu F., Tomiyasu K., Inomata K., Calderhead R.J.L.T. Cytochemical effects of GaAlAs diode laser radiation on rat saphenous artery calcium ion dependent adenosine triphosphatase activity. Laser Therapy. 1989;1(2):89. [Google Scholar]
- 37.AlGhamdi K.M., Kumar A., Moussa NAJLims Low-level laser therapy: a useful technique for enhancing the proliferation of various cultured cells. Lasers Med. Sci. 2012;27(1):237–249. doi: 10.1007/s10103-011-0885-2. [DOI] [PubMed] [Google Scholar]
- 38.Eduardo Fd.P., Bueno D.F., de Freitas P.M., Marques M.M., Passos-Bueno M.R., Eduardo Cd.P. Stem cell proliferation under low intensity laser irradiation: a preliminary study. Lasers Surg. Med. 2008;40(6):433–438. doi: 10.1002/lsm.20646. [DOI] [PubMed] [Google Scholar]
- 39.Passarella S., Casamassima E., Molinari S., Pastore D., Quagliariello E., Catalano I. Increase of proton electrochemical potential and ATP synthesis in rat liver mitochondria irradiated in vitro by helium-neon laser. FEBS Lett. 1984;175(1):95–99. doi: 10.1016/0014-5793(84)80577-3. [DOI] [PubMed] [Google Scholar]
- 40.Hu W.-P., Wang J.-J., Yu C.-L., Lan C.-C.E., Chen G.-S., Yu H-SJJoID Helium–neon laser irradiation stimulates cell proliferation through photostimulatory effects in mitochondria. J. Invest. Dermatol. 2007;127(8):2048–2057. doi: 10.1038/sj.jid.5700826. [DOI] [PubMed] [Google Scholar]
- 41.Tezel A., Kara C., Balkaya V., Orbak R.J.P. surgery l. An evaluation of different treatments for recurrent aphthous stomatitis and patient perceptions: Nd: YAG laser versus medication. Photomed. Laser Surg. 2009;27(1):101–106. doi: 10.1089/pho.2008.2274. [DOI] [PubMed] [Google Scholar]
- 42.Funk J.O., Kruse A., Kirchner H. Cytokine production after helium-neon laser irradiation in cultures of human peripheral blood mononuclear cells. J. Photochem. Photobiol. B. 1992;16(3–4):347–355. doi: 10.1016/1011-1344(92)80022-n. [DOI] [PubMed] [Google Scholar]
- 43.Huang T.J., MacAry P.A., Wilke T., Kemeny D.M., Chung K.F. Inhibitory effects of endogenous and exogenous interferon-gamma on bronchial hyperresponsiveness, allergic inflammation and T-helper 2 cytokines in Brown-Norway rats. Immunology. 1999;98(2):280–288. doi: 10.1046/j.1365-2567.1999.00870.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tau G., Rothman P. Biologic functions of the IFN-gamma receptors. Allergy. 1999;54(12):1233–1251. doi: 10.1034/j.1398-9995.1999.00099.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Maldaner D.R., Azzolin V.F., Barbisan F., Mastela M.H., Teixeira C.F., Dihel A. In vitro effect of low-level laser therapy on the proliferative, apoptosis modulation, and oxi-inflammatory markers of premature-senescent hydrogen peroxide-induced dermal fibroblasts. Lasers Med. Sci. 2019;34(7):1333–1343. doi: 10.1007/s10103-019-02728-1. [DOI] [PubMed] [Google Scholar]
- 46.Safavi S.M., Kazemi B., Esmaeili M., Fallah A., Modarresi A., Mir M. Effects of low-level He-Ne laser irradiation on the gene expression of IL-1beta, TNF-alpha, IFN-gamma, TGF-beta, bFGF, and PDGF in rat's gingiva. Lasers Med. Sci. 2008;23(3):331–335. doi: 10.1007/s10103-007-0491-5. [DOI] [PubMed] [Google Scholar]
- 47.Wang X.Y., Ma W.J., Liu C.S., Li Y.X. Effect of low-level laser therapy on allergic asthma in rats. Lasers Med. Sci. 2014;29(3):1043–1050. doi: 10.1007/s10103-013-1456-5. [DOI] [PubMed] [Google Scholar]
- 48.Souza D.G., Soares A.C., Pinho V., Torloni H., Reis L.F., Martins M.T. Increased mortality and inflammation in tumor necrosis factor-stimulated gene-14 transgenic mice after ischemia and reperfusion injury. Am. J. Pathol. 2002;160(5):1755–1765. doi: 10.1016/s0002-9440(10)61122-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Meduri G.U., Headley S., Kohler G., Stentz F., Tolley E., Umberger R. Persistent elevation of inflammatory cytokines predicts a poor outcome in ARDS: plasma IL-1β and IL-6 levels are consistent and efficient predictors of outcome over time. Chest. 1995;107(4):1062–1073. doi: 10.1378/chest.107.4.1062. [DOI] [PubMed] [Google Scholar]
- 50.Fu P.-K., Wu C.-L., Tsai T.-H., Hsieh C.-L., Medicine A. Anti-inflammatory and anticoagulative effects of paeonol on LPS-induced acute lung injury in rats. Evid. Based Complement. Alternat. Med. 2012;2012 doi: 10.1155/2012/837513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cho J.S., Guo Y., Ramos R.I., Hebroni F., Plaisier S.B., Xuan C. Neutrophil-derived IL-1β is sufficient for abscess formation in immunity against Staphylococcus aureus in mice. PLoS Pathog. 2012;8(11) doi: 10.1371/journal.ppat.1003047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Rojas M., Parker R.E., Thorn N., Corredor C., Iyer S.S., Bueno M. Infusion of freshly isolated autologous bone marrow derived mononuclear cells prevents endotoxin-induced lung injury in an ex-vivo perfused swine model. Stem Cell Res. Ther. 2013;4(2):26. doi: 10.1186/scrt174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sharifov O.F., Xu X., Gaggar A., Grizzle W.E., Mishra V.K., Honavar J. Anti-inflammatory mechanisms of apolipoprotein AI mimetic peptide in acute respiratory distress syndrome secondary to sepsis. PLoS ONE. 2013;8(5) doi: 10.1371/journal.pone.0064486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.de Perrot M., Fischer S., Liu M., Imai Y., Martins S., Sakiyama S. Impact of human interleukin-10 on vector-induced inflammation and early graft function in rat lung transplantation. Am. J. Respir. Cell Mol. Biol. 2003;28(5):616–625. doi: 10.1165/rcmb.2002-0109OC. [DOI] [PubMed] [Google Scholar]
- 55.Xuan W., Huang L., Hamblin MRJJob Repeated transcranial low-level laser therapy for traumatic brain injury in mice: biphasic dose response and long-term treatment outcome. J. Biophotonics. 2016;9(11–12):1263–1272. doi: 10.1002/jbio.201500336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ezzati K., Laakso E.L., Salari A., Hasannejad A., Fekrazad R., Aris A. The Beneficial Effects of High-Intensity Laser Therapy and Co-Interventions on Musculoskeletal Pain Management: A Systematic Review. J. Lasers Med. Sci. 2020;11(1):81–90. doi: 10.15171/jlms.2020.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Elblbesy M.A.J.P. photomedicine, surgery l. Comparative In Vitro Study: Examining 635 nm Laser and 265 nm Ultraviolet Interaction with Blood. Photobiomodul. Photomed. Laser Surg. 2019;37(6):342–348. doi: 10.1089/photob.2018.4611. [DOI] [PubMed] [Google Scholar]
- 58.C.J.J.O.C. Gunn, I. Medicine, Acute respiratory distress syndrome successfully treated with low level laser therapy, J. Complement. Integrative Med., 2 (1) (2005).