Abstract
Cardiovascular involvement in COVID-19 has different features. Here we report the ominous fate of a neglected adolescent with Williams syndrome that was infected by SARS-CoV-2 and ended by acute aortic dissection.
Keywords: Williams syndrome, aortic dissection, COVID-19, aortitis
Patients with Williams syndrome have a great risk of sudden cardiac death. Bilateral outflow tract obstruction, coronary artery stenosis, long QT interval, and microvascular ischemia have been proposed as the etiology of sudden death in this population.1 Below we will describe the medical history of a teenager with Williams syndrome who had sudden death after SARS-CoV-19 infection.
Case report
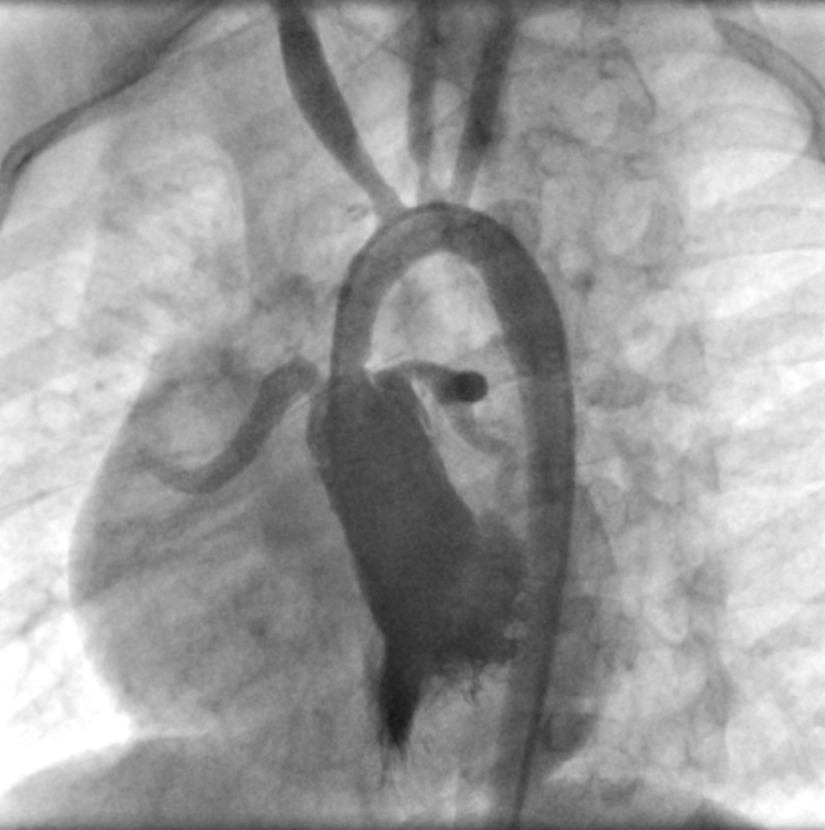
A 14-year-old boy, a neglected known case of Williams syndrome, was admitted to the emergency ward due to chest pain and shortness of breath for a couple of days ago (March 2020). Cardiology consultation was requested for acute coronary syndrome. He had a high grade fever of 39.8°C and suffered from generalised weakness, myalgia, and dry cough. On admission, he was ill, tachypneic, and mildly desaturated (O2 Saturation = 92%). Past medical history revealed a non-compliant family and only mild dyspnea on vigorous exertion. Except for a diagnostic angiographic study that showed a long segment supravalvar aortic stenosis with prominent coronary involvement (Fig 1), there was no history of intervention or surgical procedure.
Figure 1.
Left ventricular angiogram at the age of 8 years showed a long segment supravalvar aortic stenosis with coronary ostial narrowing. Proximal narrowing of brachiocephalic and left common carotid artery is seen too.
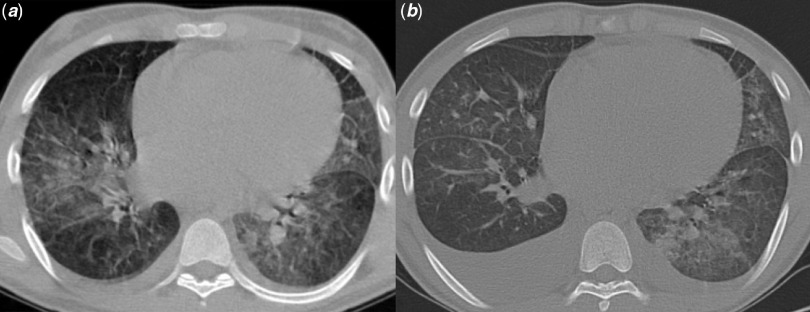
Labouratory findings showed white blood cell count of 12,890/µl (4000–11,000/µl) lymphocyte count of 940/µl, C-reactive protein of 520 mg/L (<6 mg/L), procalcitonin of 4 ng/ml (0.5–2 ng/ml), and lactate dehydrogenase of 816 U/L (5–615 U/L). Prothrombin time, partial thromboplastin time, and international normalized ratio were within the normal range. The liver enzymes were slightly elevated: serum glutamic oxaloacetic transaminase of 68 U/L (10–40 U/L) and serum glutamic pyruvic transaminase of 58 U/L (10–40 U/L). Cardiac troponin I was 0.7 mg/ml (0–0.3 mg/ml) with a declining trend to 0.4 mg/ml in a couple of weeks. The level of creatine phosphokinase-MB and total creatine phosphokinase were 17 U/L (0–24 U/L) and 252 U/L (24–195 U/L), respectively with a ratio of 7% that was within the normal limit (normal of <10%). Out of three samples of blood cultures, only one was positive for staphylococcus aureus. Echocardiography showed a systolic pressure gradient of more than 140 mmHg across the supravalvar aortic stenosis in addition to mild peripheral pulmonary artery narrowing and coronary ostial stenosis. Mild mitral regurgitation with prominent left ventricular hypertrophy and preserved left ventricular systolic function were other echocardiographic findings. No abnormal regional wall motion was detected either. Moreover, there was no evidence of vegetation on serial echo imaging. High resolution computed tomography scan findings included diffuse opacification in both lungs and bilateral air space involvement with mosaic pattern especially in the lower lobes, in addition to mild bilateral pleural effusion (Fig 2a). Reverse transcription-polymerase chain reaction for SARS-CoV-2 was positive. Combination therapy of oseltamivir, hydroxychloroquine, and azithromycin was started based on the guidelines of that time. The corrected QT interval was 452 ms. Vancomycin was started because of positive blood culture for staphylococcus aureus. However, follow-up blood cultures were negative. After 2 weeks of hospitalisation, he was discharged in good condition.
Figure 2.
Chest high resolution computed tomography scan in the first and second admission. (a) bilateral parenchymal involvement and mild pleural effusion. (b) Augmentation of pleural effusion in addition to ground-glass opacification of both lungs.
Three days later, he was again admitted because of exacerbated cough, recurrence of chest pain, and occasional abdominal pain. He was febrile and also mildly tachypneic and hypoxic (O2 Saturation = 91%). Thoracic high resolution computed tomography scan showed augmentation of bilateral pleural effusion and enlargement of mediastinal lymph nodes in multiple levels (Fig 2b). Cocktail of hydroxychloroquine, azithromycin, and Kaletra (lopinavir/ritonavir) was administered and diuretics were added. Combination therapy of cotrimoxazole and vancomycin was used to cover the possible bacterial infection especially his previous suspected bacteremia with staphylococcus aureus.
Lab examination in the second admission showed lactate dehydrogenase of 631 U/L (5–615 U/L), total creatine phosphokinase of 61 U/L (24–195 U/L), D-Dimer of 0.9 pg/ml (<0.5 pg/ml), Albumin of 3.3 g/dL (3.5–5.5 g/dl), and lymphocyte count of 390/µl. Prothrombin time, partial thromboplastin time, and international normalized ratio were within the normal range. Progressive desaturation (O2 Saturation = 82%) urged us to immediately treat the possible cytokine storm or multisystem inflammatory syndrome by methylprednisolone (loading dose 2 mg/kg over 30 minutes then 0.5 mg/kg every 6 hours) and intravenous immunoglobulin 1 g/kg (maximum 60 g). Three days after corticosteroid therapy, the patient’s condition dramatically improved with disappearance of dyspnea and cough. The results of both polymerase chain reaction tests in the second admission for SARS-CoV-2 were negative. Echocardiographic assessment for bacterial endocarditis was negative and no evidence of myocarditis or cardiac systolic dysfunction was found either. The abdominal sonography was normal. Ten days after the second discharge while he was in good condition, he was awakened by severe back pain at 4:00 o’clock in the morning. Unfortunately, he did not have any detectable vital signs on arrival to the nearest hospital. All attempts to resuscitate the patient failed. Autopsy findings revealed type A aortic dissection in addition to the evidence of aortitis with inflammatory cell infiltration mainly consists of mononuclear cells.
Discussion
Our patient’s clinical manifestation and labouratory findings in the first and second admission were correlated with COVID-19 pneumonia and multisystem inflammatory syndrome in children respectively.2 His aggravating condition in the multisystem inflammatory syndrome in children phase was dramatically improved after corticosteroid and intravenous immunoglobulin therapy. Although our patient’s level of troponin I was higher than normal, the ratio of creatine phosphokinase-MB to total creatine phosphokinase was less than 10%. Moreover, we could not find any evidence of apparent myocarditis either in echocardiographic parameters or clinical manifestations. Myocardial damage in COVID-19 might occur as a result of cytokine storm, hypoxia of acute respiratory distress syndrome or direct virus invasion. Stenosis of coronary ostia in Williams syndrome will much more threaten the compromised circulation of a hypertrophic myocardium.
One might have the concern of life-threatening arrhythmia in patients with Williams syndrome and concomitant prolonged QTc interval while receiving drugs such as hydroxychloroquine, Kaletra, and azithromycin. However, our patient was monitored according to the risk score for drug associated QTc prolongation.3 It is important to emphasise that although the possibility of sudden death in Williams syndrome with concomitant prolonged QTc interval has been addressed in literature, administration of anti-COVID-19 medications with adverse effect of QT interval prolongation is not an absolute contraindication.4
None of the aforementioned mechanism was the cause of sudden death in our patient. Instead, aortic dissection was found in post mortem examination. Aortitis associated with virus-related cryoglobulinemia has been reported in acute aortic dissection.5 Elastin aortopathy due to ELN haploinsufficiency has been proposed as the etiology of aortic dissection in cases with Williams syndrome.6 There is still debate regarding the balance of benefit and hazard in corticosteroid therapy of COVID-19 patients. However, it has been shown that corticosteroid therapy particularly its long-term use might inhibit the formation of granulation tissue. Hence corticosteroids contribute to aneurysm formation by interfering with the repair of intimal injury.7 It is also noteworthy to declare that we could not assess the possible biochemical adverse effect of the medication cocktail other than methylprednisolone in the formation of aortic aneurysm and dissection in our patient.8
By identifying the unknown aspects of the COVID-19 disease in vascular endothelial damage and thrombus formation in arteries of different calibers, vascular events might occur either due to cytokine storm or hypoxia induced apoptosis (ceramide pathway). We assume that aortitis and endothelial cell damage by COVID-19 induced inflammatory responses have led to acute aortic dissection on the background of elastin arteriopathy and negative effects of corticosteroids.
Acknowledgements
We would like to appreciate Dr Maryam Rostamyan and Dr Minoo Dadkhah for their help to prepare the manuscript.
Financial support
This research received no specific grant from any funding agency, commercial, or not-for-profit sectors.
Conflicts of interest
None.
Ethical standards
Not applicable.
References
- 1. Collins RT. Cardiovascular disease in Williams syndrome. Circulation 2013; 127: 2125–2134. [DOI] [PubMed] [Google Scholar]
- 2. Riphagen S, Gomez X, Gonzalez-Martinez C, Wilkinson N, Theocharis P. Hyperinflammatory shock in children during COVID-19 pandemic. Lancet 2020; 395: 1607–1608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Simpson TF, Kovacs RJ, Stecker EC. Ventricular arrhythmia risk due to hydroxychloroquine-azithromycin treatment for COVID-19. Cardiol Mag 2020; 29: 29. [Google Scholar]
- 4. Collins RT. Clinical significance of prolonged QTc interval in Williams syndrome. Am J Cardiol 2011; 108: 471–473. [DOI] [PubMed] [Google Scholar]
- 5. Fukunaga N, Fujiwara H, Nasu M, Okada Y. Aortic dissection caused by aortitis associated with hepatitis C virus-related cryoglobulinemia. J Thorac Cardiovasc Surg 2010; 140: e81–e82. [DOI] [PubMed] [Google Scholar]
- 6. Vanacker P, Thijs V. Spontaneous cervical artery dissection in adult Williams syndrome. Cerebrovasc Dis 2009; 27: 309–310. [DOI] [PubMed] [Google Scholar]
- 7. Wei HY, Chung HT, Wu CT, Huang JL. Aortic dissection complicated with hemothorax in an adolescent patient with systemic lupus erythematosus: case report and review of literature. Semin Arthritis Rheum 2011; 41: 12–18. [DOI] [PubMed] [Google Scholar]
- 8. Spigset O. Drug-induced aortic aneurysms, ruptures and dissections In: Grundmann R (ed). Etiology, Pathogenesis and Pathophysiology of Aortic Aneurysms and Aneurysm Rupture. InTech, Croatia, 2011; 159–174. [Google Scholar]