Abstract
Open aorta and vena cava surgeries are usually associated with substantial blood loss which may result in postoperative acute kidney injury (AKI). The present study is designed to investigate the prevalence, outcome and risk factors of postoperative AKI associated with open aorta and vena cava surgeries, with a focus on the role of anemia in these conditions. A retrospective review of medical records of Peking Union Medical College Hospital was conducted. Patients who underwent open aorta and vena cava surgeries during January 1, 2010 and June 30, 2014 were included in this study. The primary analysis was between patients underwent open aorta and vena cava surgeryies, with or without postoperative AKI. Multivariable logistic regression models were used to determine risk factors of postoperative AKI. The study included 79 patients (63.3% male) with a mean age of 52.5±17.3 years (range, 17–81 years). Postoperative AKI occurred in 23/79 (29.1%) of the patients. Anemia was present in 11/79 (16%) at baseline, and increased to 45/79 (52%) postoperatively. After adjustment for various risk factors, postoperative anemia (OR, 5.202; 95% CI 1.403–19.285) was independently associated with postoperative AKI. AKI is a common complication in patients who undergo open aorta and vena cava surgeries, and postoperative anemia was the most relevant predictive factor of AKI. Strategies to minimize bleeding and anemia for all patients may be advisable. Further studies are needed to assess the impact of AKI on long term outcome and to examine preventive strategies to address potentially modifiable risk factors.
Introduction
Acute kidney injury (AKI) is a prevalent and prognostically significant complication in surgical patients. Depending on the AKI definitions adopted and the population studied, AKI occurs in up to 16% - 48% of surgical patients [1–3]. AKI after any type of surgery is independently associated with substantially worse outcomes, including higher short-term mortality, prolonged length of hospital stay, worsened long-term survival, and increased risk of end stage renal disease (ESRD) [4–8].
Despite advances of endograft techniques, open aorta and vena cava surgeries are still used in patients unsuitable for endovascular repair, and are associated with substantial incidence of postoperative AKI [9, 10]. These procedures are usually associated with significant blood loss, which may result in hypovolemia or hypoxemia. Regionally reduced oxygen supply lead to physiologically low oxygen tensions within the renal cortex and the medulla and may cause postoperative AKI [2, 3]. However, most previous researches focused on AKI after endovascular procedures which may be associated with contrast use, and the prevalence, risk factors and outcomes of AKI after open aorta and vena cava surgeries were poorly addressed in the literatures. Hence we performed an analysis in patients underwent open surgeries of major vasculatures including aorta and vena cava, to investigate the prevalence, outcome and risk factors of postoperative AKI, with a focus on the role of anemia in these conditions.
Materials and methods
This study was approved by the Institutional Review Board (IRB) of Peking Union Medical College Hospital the University (S-K048).
Database and participants
We conducted a retrospective review of the medical records of patients during September to November, 2015. The patients underwent vascular surgeries at Peking Union Medical College Hospital from January 1, 2010 to June 30, 2014. The medical records were accessed on patients who underwent vascular surgeries of all types, and cases involving open aorta and vena cava surgery were included in this study. Cases that involved contrast administration during the perioperative period, perioperative death, or with incomplete data were excluded from the analysis. Patient demographics and perioperative data were obtained from the chart review. Informed consents were obtained by the time of patient admission for later access of their medical records. Authors had access to information that could identify individual participants during or after data collection.
Covariates and definitions
The primary analysis was between patients underwent open surgeries of aorta or vena cava, with or without postoperative AKI, to evaluate the risk factors associated with postoperative AKI. AKI was defined as ≥26.5 μmol/L increase in serum creatine (SCr) level within the first 48 hours post-operation, or an increase in SCr levels from baseline by more than 50% during the first 7 postoperative days, according to the AKI criteria from the Kidney Disease: Improving Global Outcomes (KDIGO) Clinical Practice Guideline for Acute Kidney Injury, 2012 [11]. AKI was staged for severity according to the criteria presented in Table 1. Anemia: hemoglobin (Hb) (male) <120g / l, Hb (fmale) <110g / l. (Other definitions are included in Tables 2 and 3). Chronic kidney diseases (CKD): GFR<60ml/min/1.73m2. Hypoproteinemia: albumin <30g / l. Coronary heart disease: left ventricular ejection fraction (LVEF) <40%. Blood loss: anesthesiologist estimated with blood recovered intraoperative blood salvage. Using of vasopressors: using the vasopressors drugs during operation.
Table 1. Staging of acute kidney injury.
| Stage | Serum creatinine |
|---|---|
| 1 | 1.5 to 1.9 times baseline or≥0.3 mg/dl (≥26.5 μmol/l) increase |
| 2 | 2.0 to 2.9 times baseline |
| 3 | 3 times baseline or increase to ≥353.6 μmol/L in SCr or initiation of renal replacement therapy |
Table 2. Patients demographics and characteristics.
| All (N = 79) | AKI (N = 23) | No AKI (N = 56) | P | |
|---|---|---|---|---|
| Mean (± SD) or median (IQR) or N (%) | Mean (± SD) or median (IQR) or N (%) | Mean (± SD) or median (IQR) or N (%) | ||
| Demographics | ||||
| Age, years | 52.5(±17.3) | 55.8(±15.2) | 51.2(±18) | 0.25 |
| Male sex | 50(63.2%) | 14(60.9%) | 36(64.3%) | 0.82 |
| BMI, kg/m2 | 23.04±2.45 | 23.18±2.24 | 22.98±2.54 | 0.32 |
| Ace inhibitors/ ARBs use | 39(49.4%) | 14(60.9%) | 25(44.6%) | 0.20 |
| Comorbidities | ||||
| Hypertension | 38(48.1%) | 14(60.9%) | 24(42.9%) | 0.15 |
| Coronary heart disease | 6(7.5%) | 2(8.7%) | 4(7.1%) | 0.813 |
| Hypoproteinemia a | 9(14.3%) | 4(25%) | 5(10.6%) | 0.315 |
| CKD | 9(11.4%) | 3(13.0%) | 6(10.7%) | 0.12 |
| Diabetes | 2(2.5%) | |||
| Baseline Scr, μmol/l | 74.0(±24.8) | 75.4(±27.2) | 73.5(±23.9) | 0.758 |
| Peak Scr, μmol/l | 95.1(±38.0) | 124.2(±47.6) | 83.2(±25.0) | 0.001 |
| Surgery types | ||||
| Renal artery aneurysm repair | 8(10.1%) | 6(26.1%) | 2(3.6%) | 0.009 |
| Inferior vena cava leiomyosarcoma resection | 6(7.6%) | 3(13.0%) | 3(5.4%) | 0.48 |
| Aortic aneurysm repair | 31(39.2%) | 8(34.8%) | 23(41.1%) | 0.6 |
| Aortic bypass | 16(20.3%) | 3(13.0%) | 13(23.2%) | 0.48 |
| Other surgeries | 18(22.8%) | 3(13.0%) | 15(26.8%) | 0.3 |
| Intra-operative conditions and management | ||||
| Baseline MAP, mmHg | 99.0(±13.6) | 105(±14) | 97(±13) | 0.017 |
| Nadir of Intraoperative MAP, mmHg | 71.5(±9.5) | 72(±10) | 71(±9) | 0.963 |
| Vasopressors use | 12(15.2%) | 5(21.7%) | 7(12.5%) | 0.282 |
| Antibiotics use | 61(77.2%) | 19(82.6%) | 42(75%) | 0.47 |
| Fluid infusion, ml | 4314(3000,5100) | 4600(3700,6500) | 4150(3000,4775) | 0.094 |
| Suprarenal aortic cross clamp | 11(13.9%) | 5(21.7%) | 6(10.7%) | 0.316 |
| Surgical duration, min | 420.76±117.49 | 469.13±142.475 | 400.89±100.39 | 0.018 |
P value denotes comparison to AKI and non-AKI subgroups; AKI, acute kidney injury; CKD, chronic kidney diseases; SCr, serum creatinine; SD: standard deviation, IQR: interquartile range
aThe data is not complete.
Table 3. Perioperative anemia of patients with and without acute kidney injury (AKI).
| All (N = 79) | AKI (N = 23) | No AKI (N = 56) | P | |
|---|---|---|---|---|
| Mean (± SD) or median (IQR) or N (%) | Mean (± SD) or median (IQR) or N (%) | Mean (± SD) or median (IQR) or N (%) | ||
| Blood loss during operation, ml | 1000(700,2000) | 1200(800,2500) | 1000(600,1950) | 0.042 |
| Transfusion | 41(56.9%) | 15(69.6%) | 26(46.4%) | 0.162 |
| Baseline Hb, g/l | 130.7(±20.2) | 124.9(±20.5) | 132.8(±19.9) | 0.14 |
| Postoperative Hb, g/l | 112.4(±19.5) | 104.2(±16.3) | 115.4(±19.9) | 0.02 |
| Preoperative anemia | 11(13.9%) | 4(21.1%) | 7(13.0%) | 0.4 |
| Postoperative anemia | 46(58.2%) | 17(74.0%) | 29(51.8%) | 0.049 |
Hb: hemoglobin.
SD: standard deviation, IQR: interquartile range.
The baseline renal function was the last serum creatinine before surgery; the peak serum creatinine was the highest value of serum creatinine obtained during the first 7 days after surgery. The baseline levels of hemoglobin (Hb), hematocrit (HCT) and albumin (Alb) were obtained from the latest preoperative data, and the first test results after surgery were used as the postoperative results.
Data analyses
Data were expressed as a mean ± SD for continuous variables and as percentages for discrete variables. Continuous data were analyzed by Student’s t test for equal variance or the Mann-Whitney test for unequal variance, and categorical valuables were investigated by the Pearson χ2 or Fisher’s exact test. A two-sided P value < .05 was considered significant. Logistic regression (adjusted for multiple covariates) was used to determine the odds ratio (OR) and the 95% confidence intervals (CI) associated with and without AKI. To adjust for additional unmeasured confounding variables, we fit a logistic regression model using the aforementioned covariates with the procedure type as the dependent variable. All covariates with P < .3 from univariate analysis were put into logistic regression model. Statistical analyses were performed using SPSS (Version 20.0. for Windows; SPSS, Inc.), and significance was assigned at P values < .05.
Results
Clinical characteristics
A review of the medical records revealed 4926 cases of vascular surgery in Peking Union Medical College Hospital from January 1, 2010 to June 30, 2014. A total of 79 cases met the inclusion criteria, accounting for 1.6% of the population. The clinical features of the 79 cases are summarized in Table 2. The patient population included 50 males and 29 females, with a male predominance (63.3%). The mean age at the time of surgery was 52.5±17.3 years (range, 17–81 years). Among the patients, 48.1% (38) had a medical history of hypertension, and the prevalence of CKD, coronary heart disease and diabetes were 11.4% (9), 7.5% (6), and 2.5% (2), respectively. 31 patients (39.2%) underwent open aortic aneurysm repair, 16 patients (20.3%) underwent aortic bypass, 8 patients (10.1%) underwent renal artery aneurysm repair, 6 patients (7.6%) underwent inferior vena cava leiomyosarcoma resection, and 18 patients (22.8%) underwent other major vascular surgeries, including superior mesenteric artery aneurysm repair, splenic aneurysm repair, and common iliac artery aneurysm repair. AKI occurred in 23 (29.1%) of the patients, with stage 1 AKI in 19 patients (82.6%), stage 2 in 3 patients (13.0%), and Stage 3 in 1 patient (4.3%). The demographic characteristics and intraoperative data for the study participants are shown in Tables 2 and 3. No significant differences were observed between the 2 groups in age, sex, preoperative blood pressure, BMI, Ace inhibitors/ARBs use and preexisting comorbidities such as hypertension, hypoalbuminemia and CKD. The mean baseline serum creatinine in both the AKI and non-AKI groups was similar.
Perioperative characteristics of patients with AKI
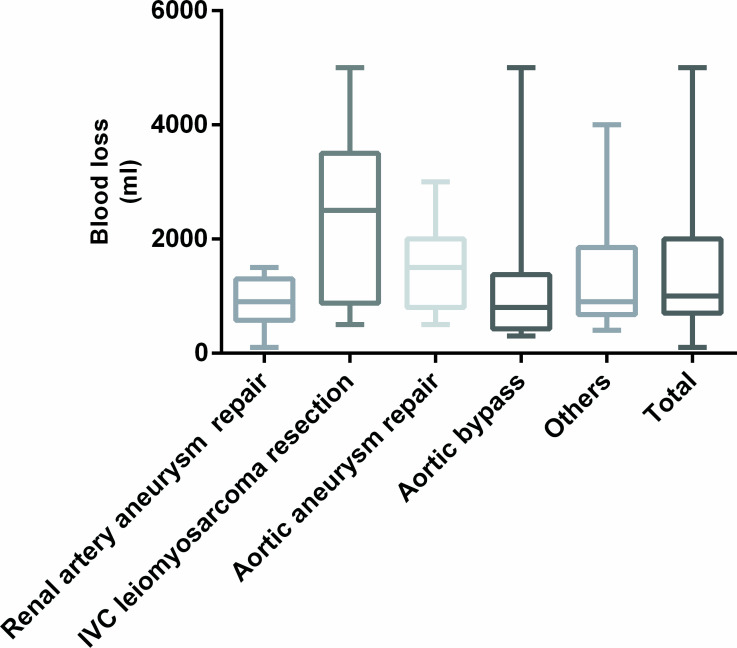
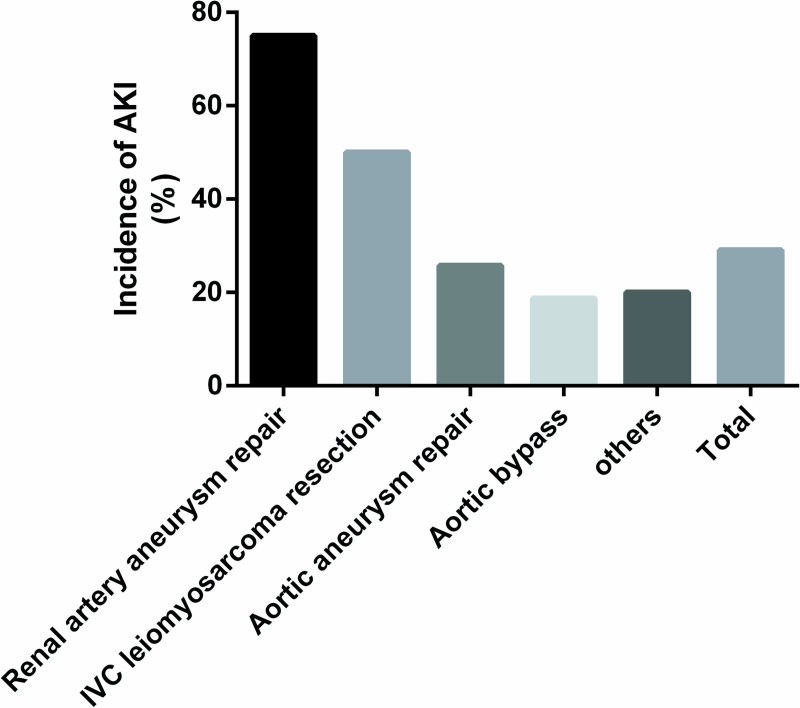
Among all of the cases studied, the surgical duration of AKI groups was longer than the non-AKI groups (469.13±142.475min vs 400.89±100.39 min, p = .018). The median blood loss (ml) during the operation was 1000 (700, 2000). The mean of intraoperative transfusion of packed red blood cells was 1.99 ± 2.86 units and intraoperative transfusion of plasma was 255.70 ± 430.78 ml A total of 11/79(16%) of the patients had preoperative anemia; postoperatively, the occurrence of anemia increased to 45/79(52%). There was a significant difference in intraoperative blood loss (ml) between the AKI (1200 [800, 2500]) and the non-AKI (1000 [600,1950]) patients. Transfusion rates in AKI and non-AKI was 65.2% (15/23) and 46.4% (26/56) respectively. In particular, the incidence of AKI increased in patients with postoperative anemia comparing to those without anemia (37.0% [17/46] vs 18.2% [6/33]). The proportion of patients that received vasopressors during operations were 21.7% (5/23) and 10.7% (7/56) among the AKI and non-AKI patients, respectively. The time of suprarenal aortic cross clamp was 41.67±22.39min. Use of suprarenal aortic cross clamp during the operation was associated with an increased incidence of AKI (45.5% [5/11] vs 26.5% [18/68]). The mean blood loss of the patients who received vasopressors was 1764 ml vs 1361 ml in those who did not. The mean blood loss in patients underwent renal artery aneurysm repair was 900±160 ml. The maximum amount transfusion of packed red blood cells was 2 units and the mean was 0.25±0.7 units. The maximum amount transfusion of plasma was 400ml and the mean was 50±141ml. 2 units and the mean was 0.25±0.7 units. The maximum amount of intraoperative salvage autotransfusion was 1907ml and the mean was 502.5±632.6ml. and 75.0% of these patients developed AKI. The mean blood loss of patients who underwent inferior vena cava leiomyosarcoma resection was 2400±650 ml. The maximum amount transfusion of packed red blood cells was 13 units and the mean was 7.17±4.83 units. The maximum amount transfusion of plasma was 1000ml and the mean was 533.33±467.62ml. The maximum amount of intraoperative salvage autotransfusion was 1037ml and the mean was 322.83±502ml. and 50% of these patients developed AKI. The mean blood loss in patients who underwent open aortic aneurysm repair was 1500±160 ml. The maximum amount transfusion of packed red blood cells was 10 units and the mean was 1.94±2.54 units. The maximum amount transfusion of plasma was 2000ml and the mean was 219.35±439.26ml. The maximum amount of intraoperative salvage autotransfusion was 2300ml and the mean was 559.43±480.69ml. and 25.8% of these patients developed AKI. The incidence of AKI among patients underwent aortic bypass surgery was 18.8%, and the incidence in patients underwent other major vascular surgeries was 20% (Figs 1 and 2).
Fig 1. Blood loss of different surgery types.
IVC, Inferior vena cava.
Fig 2. Incidence of post-operative acute kidney injury of different surgery types.
IVC, Inferior vena cava.
Risk factor of AKI
Open aorta and vena cava surgery, postoperative anemia, renal artery aneurysm repair, blood loss, and the use of intraoperative vasopressors, were all associated with AKI on univariate analysis (Tables 2 and 3). In the multivariate analysis, postoperative anemia (OR, 5.202; 95% CI, 1.403~19.285) was an independent predictor of postoperative AKI, but not age (OR, 1.015; 95% CI, 0.981~1.049), hypertension (OR, 1.586; 95% CI, 0.511~4.919), CKD (OR, 0.126; 95% CI, 0.011~1.391), or vasopressor use (OR, 4.577; 95% CI, 0.988~21.194) (Table 4).
Table 4. Risk factors of postoperative acute kidney injury.
| P | OR | 95% CI | |
|---|---|---|---|
| Age, years | 0.396 | 1.015 | 0.981–1.049 |
| Hypertension | 0.425 | 1.586 | 0.511–4.919 |
| CKD | 0.091 | 0.126 | 0.011–1.391 |
| Vasopressors use | 0.052 | 4.577 | 0.988–21.194 |
| Postoperative Anemia | 0.014 | 5.202 | 1.403–19.285 |
CI, confidence interval; OR, odds ratio; CKD, chronic kidney diseases.
Discussion
Despite the development of endovascular procedures, open surgeries of aorta and vena cava are still used, especially in patients who are not suitable for endovascular procedures, and are associated with substantial mortality and complicated courses [1–3]. AKI in a common complication associated with open surgeries of aorta and vena cava. However, the incidence and risk factors of AKI in these conditions have not been adequately addressed.
Ischemic-reperfusion injury is common following surgeries and causes AKI [12–14]. The incidence of AKI after surgery was different in different studies, and it was mainly related to the observation from different patients [3, 15]. In our study, we assessed the incidence and risk factors of AKI in patients underwent open surgery of aorta and vena cava. The prevalence of postoperative AKI was as high as 29.1%, indicating that AKI is a common complication in these patients. The patients included in this study underwent open surgery of aorta and vena cava, which may be extremely difficult due to the complicated anatomic structures and abundant collateral circulations [9, 16]. These procedures are usually accompanied with prolonged length of surgery, higher bleeding risk, and more complicated perioperative course, placing the patients at increased risk of AKI [3, 8, 17]. In our study, the surgical duration of postoperative AKI groups was longer than that of no-AKI groups. The importance of AKI as a risk factor for mortality and we need to focus on high-risk patients for prevention and early identification of AKI and prompt treatment. However, we adopted the 2012 KIDIGO definition of AKI in our study, which is still based on increases in serum creatinine concentrations. New biomarkers of kidney injury, such as serum cystatin C (ScysC) Neutrophil Gelatinase-Associated Lipocalin (NGAL) and brain natriuretic peptide (BNP) may facilitate earlier detection of renal injury, and thus early intervention aimed at limiting the associated morbidity and mortality [18–20].
Postoperative AKI has been shown to be associated with early and long-term mortality and long-term risk of ESRD [4, 5, 21]. Thus adequate attentions are needed pertaining AKI following these surgeries.
It is noteworthy that AKI is more prevalent among those patients with more blood loss in our study. The mean blood loss during leiomyosarcoma and aortic aneurysm surgeries were 2400 ± 650 ml and 1500 ± 160 ml, and incidences of AKI after these procedures were up to 50% and 25.8%, respectively. Bleeding and postoperative anemia were associated with postoperative AKI, and multivariate analysis showed that postoperative anemia was an independent risk factor for AKI (OR, 5.202; 95% CI, 1.403–19.285). And vasopressors use during surgeries, which probably indicate more blood loss during the procedures, is associated with higher risk of postoperative AKI.
However, even without massive hemorrhage, the incidence of AKI after open renal artery aneurysm repair was higher comparing to other surgical procedures of the aorta and vena cava. Aortic cross clamping was a renal hazard for this group of patients, and AKI occurred in 21.5% of patients who underwent surgery with a suprarenal aortic cross clamp compared to 12.5% in those who didn’t. Aortic cross clamping blocks effective renal perfusion and exposes the renal parenchyma to reduced oxygen tension, contributing to ischemia/reperfusion injury [14].
The kidney is especially vulnerable to hypoxic injury. Acute blood loss, hypovolemia, hemodilution or renal artery blocking during surgical processes may reduce regional oxygen supply, leading to physiological low oxygen tensions within the renal cortex and the medulla and microvascular dysfunction, and may result in acute kidney injury [12–14]. Furthermore, these conditions are associated with increased renal tubular oxygen consumption and oxidative stress, further confounding the imbalance of oxygen supply and demandz [13, 14].
Postoperative anemia is the main predictor of postoperative AKI in our study, and other baseline characteristics that have been traditionally associated with AKI after other surgery types including age, baseline renal function, hypertension, and diabetes were not associated with postoperative AKI in our study. This may be attributed to the patient population selected, and limited case number in our study. Larger, multicenter, prospective studies may be needed to verify these conclusions.
Therefore, among patients undergoing open vascular surgery of aorta or vena cava, prevention and treatment may include avoiding unnecessary blood loss, ensuring optimal renal perfusion and ameliorating postoperative anemia. However, as shown in previous studies, treating perioperative anemia with blood transfusion didn’t improve renal outcomes of patients undergoing surgery [22–24]. Furthermore, receiving intraoperative blood transfusion was associated with a higher risk of postoperative mortality, pulmonary complications, renal failure and infectious complications. Transfusions were associated with cytokine release, bacterial contamination, sepsis, metabolic disturbances, circulatory overload, hemolytic reactions, and risk of old blood [25]. Given these findings, other means to alleviate perioperative anemia are needed to consider. Erythropoietin (EPO) is a natural hormone produced in human body, and is a regulator of erythroid precursor cells. Evidences suggest that EPO has anti-apoptotic, anti-oxidative and anti-inflammatory properties, and is able to reduce the injury caused by ischemia/reperfusion of kidney in animal model [26, 27]. Thus, EPO might be considered as a possible prophylactic intervention to prevent postoperative AKI.
This study has several limitations that need to be considered. First, the effects of unmeasured confounders on these relationships we describe cannot be dismissed because this is an observational study. Potentially important unmeasured confounders include the use of perioperative medications that may influence kidney function. Second, this is a single-center study with limited cases, and larger, multicenter, and prospective studies may be needed to further validate our finding.
Conclusions
In our study, AKI is a common complication in patients who undergo open aorta and vena cava surgeries, and postoperative anemia is the most relevant predictive factor of AKI. Strategies to minimize bleeding and anemia for all patients may be advisable. Further studies are needed to assess the impact of AKI on long term outcome and to examine preventive strategies to address potentially modifiable risk factors.
Supporting information
(SAV)
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
This study was supported by the Development of Medical Scientific Research Fund of the Capital (2011⁃4001⁃08) and the National Key Technology Support Program (20llBAll0802, 2012BAJl8803). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Handa N, Onohara T, Okamoto M, Yamamoto T, Shimoe Y, Okada M, et al. Early outcomes of open abdominal repair versus endovascular repair for abdominal aortic aneurysm: report from national hospital organization network study in Japan. Annals of vascular diseases. 2012;5(2):172–9. Epub 2012/01/01. 10.3400/avd.oa.12.00010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Piffaretti G, Mariscalco G, Bonardelli S, Sarcina A, Gelpi G, Bellosta R, et al. Predictors and outcomes of acute kidney injury after thoracic aortic endograft repair. Journal of vascular surgery. 2012;56(6):1527–34. Epub 2012/10/13. 10.1016/j.jvs.2012.05.106 . [DOI] [PubMed] [Google Scholar]
- 3.Ghincea CV, Reece TB, Eldeiry M, Roda GF, Bronsert MR, Jarrett MJ, et al. Predictors of Acute Kidney Injury Following Aortic Arch Surgery. The Journal of surgical research. 2019;242:40–6. Epub 2019/05/08. 10.1016/j.jss.2019.03.055 . [DOI] [PubMed] [Google Scholar]
- 4.Ishani A, Xue JL, Himmelfarb J, Eggers PW, Kimmel PL, Molitoris BA, et al. Acute kidney injury increases risk of ESRD among elderly. Journal of the American Society of Nephrology: JASN. 2009;20(1):223–8. Epub 2008/11/21. 10.1681/ASN.2007080837 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. Journal of the American Society of Nephrology: JASN. 2005;16(11):3365–70. Epub 2005/09/24. 10.1681/ASN.2004090740 . [DOI] [PubMed] [Google Scholar]
- 6.Bravi CA, Vertosick E, Benfante N, Tin A, Sjoberg D, Hakimi AA, et al. Impact of Acute Kidney Injury and Its Duration on Long-term Renal Function After Partial Nephrectomy. European urology. 2019. Epub 2019/05/14. 10.1016/j.eururo.2019.04.040 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Waskowski J, Pfortmueller CA, Erdoes G, Buehlmann R, Messmer AS, Luedi MM, et al. Mannitol for the Prevention of Peri-Operative Acute Kidney Injury: A Systematic Review. European journal of vascular and endovascular surgery: the official journal of the European Society for Vascular Surgery. 2019. Epub 2019/05/13. 10.1016/j.ejvs.2019.02.003 . [DOI] [PubMed] [Google Scholar]
- 8.Sueyoshi K, Watanabe Y, Inoue T, Ohno Y. Predictors of long-term prognosis in acute kidney injury survivors who require continuous renal replacement therapy after cardiovascular surgery. 2019;14(1):e0211429 10.1371/journal.pone.0211429 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ren H, Li F, Tian J, Zheng Y. An infrarenal abdominal aortic aneurysm with severe neck stenosis. Journal of vascular surgery. 2015;62(1):229–30. Epub 2015/06/28. 10.1016/j.jvs.2014.04.010 . [DOI] [PubMed] [Google Scholar]
- 10.van Lammeren GW, Unlu C, Verschoor S, van Dongen EP, Wille J, van de Pavoordt ED, et al. Results of open pararenal abdominal aortic aneurysm repair: single centre series and pooled analysis of literature. Vascular. 2017;25(3):234–41. Epub 2016/08/28. 10.1177/1708538116665268 . [DOI] [PubMed] [Google Scholar]
- 11.Kasiske BL, Wheeler DC. Kidney Disease: Improving Global Outcomes—an update. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association—European Renal Association. 2014;29(4):763–9. Epub 2013/11/30. 10.1093/ndt/gft441 . [DOI] [PubMed] [Google Scholar]
- 12.Bian A, Shi M, Flores B, Gillings N, Li P, Yan SX, et al. Downregulation of autophagy is associated with severe ischemia-reperfusion-induced acute kidney injury in overexpressing C-reactive protein mice. 2017;12(9):e0181848 10.1371/journal.pone.0181848 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Brezis M, Rosen S. Hypoxia of the renal medulla—its implications for disease. The New England journal of medicine. 1995;332(10):647–55. Epub 1995/03/09. 10.1056/NEJM199503093321006 . [DOI] [PubMed] [Google Scholar]
- 14.Eltzschig HK, Eckle T. Ischemia and reperfusion—from mechanism to translation. Nature medicine. 2011;17(11):1391–401. Epub 2011/11/09. 10.1038/nm.2507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Huber M, Ozrazgat-Baslanti T, Thottakkara P, Efron PA, Feezor R, Hobson C, et al. Mortality and Cost of Acute and Chronic Kidney Disease after Vascular Surgery. Annals of vascular surgery. 2016;30:72–81.e1-2. Epub 2015/07/19. 10.1016/j.avsg.2015.04.092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Liu D, Ren HL, Liu B, Shao J, Chen YX, Song XJ, et al. Renal Function Preservation in Surgical Resection of Primary Inferior Vena Cava Leiomyosarcoma Involving the Renal Veins. European journal of vascular and endovascular surgery: the official journal of the European Society for Vascular Surgery. 2018;55(2):229–39. Epub 2018/01/07. 10.1016/j.ejvs.2017.11.031 . [DOI] [PubMed] [Google Scholar]
- 17.Piffaretti G, Mariscalco G, Riva F, Fontana F, Carrafiello G, Castelli P. Abdominal aortic aneurysm repair: long-term follow-up of endovascular versus open repair. Archives of medical science: AMS. 2014;10(2):273–82. Epub 2014/06/07. 10.5114/aoms.2014.42579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Guo X, Qin Y, Zheng K, Gong M, Wu J, Shou W, et al. Improved glomerular filtration rate estimation using new equations combined with standardized cystatin C and creatinine in Chinese adult chronic kidney disease patients. Clinical biochemistry. 2014;47(13–14):1220–6. Epub 2014/06/03. 10.1016/j.clinbiochem.2014.05.060 . [DOI] [PubMed] [Google Scholar]
- 19.McIlroy DR, Wagener G, Lee HT. Neutrophil gelatinase-associated lipocalin and acute kidney injury after cardiac surgery: the effect of baseline renal function on diagnostic performance. Clinical journal of the American Society of Nephrology: CJASN. 2010;5(2):211–9. Epub 2010/01/09. 10.2215/CJN.04240609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chae MS, Park H, Choi HJ, Park M, Chung HS, Hong SH, et al. Role of serum levels of intraoperative brain natriuretic peptide for predicting acute kidney injury in living donor liver transplantation. 2018;13(12):e0209164 10.1371/journal.pone.0209164 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Alkandari O, Eddington KA, Hyder A, Gauvin F, Ducruet T, Gottesman R, et al. Acute kidney injury is an independent risk factor for pediatric intensive care unit mortality, longer length of stay and prolonged mechanical ventilation in critically ill children: a two-center retrospective cohort study. Critical care (London, England). 2011;15(3):R146 Epub 2011/06/15. 10.1186/cc10269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Smilowitz NR, Oberweis BS, Nukala S, Rosenberg A, Zhao S, Xu J, et al. Association Between Anemia, Bleeding, and Transfusion with Long-term Mortality Following Noncardiac Surgery. The American journal of medicine. 2016;129(3):315–23.e2. Epub 2015/11/03. 10.1016/j.amjmed.2015.10.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Karkouti K, Wijeysundera DN, Yau TM, McCluskey SA, Chan CT, Wong PY, et al. Influence of erythrocyte transfusion on the risk of acute kidney injury after cardiac surgery differs in anemic and nonanemic patients. Anesthesiology. 2011;115(3):523–30. Epub 2011/07/22. 10.1097/ALN.0b013e318229a7e8 . [DOI] [PubMed] [Google Scholar]
- 24.Newland RF, Baker RA. Low Oxygen Delivery as a Predictor of Acute Kidney Injury during Cardiopulmonary Bypass. The journal of extra-corporeal technology. 2017;49(4):224–30. Epub 2018/01/06. [PMC free article] [PubMed] [Google Scholar]
- 25.O'Keeffe SD, Davenport DL, Minion DJ, Sorial EE, Endean ED, Xenos ES. Blood transfusion is associated with increased morbidity and mortality after lower extremity revascularization. Journal of vascular surgery. 2010;51(3):616–21, 21.e1-3. Epub 2010/01/30. 10.1016/j.jvs.2009.10.045 . [DOI] [PubMed] [Google Scholar]
- 26.Shema-Didi L, Kristal B, Eizenberg S, Marzuq N, Sussan M, Feldman-Idov Y, et al. Prevention of contrast-induced nephropathy with single bolus erythropoietin in patients with diabetic kidney disease: A randomized controlled trial. 2016;21(4):295–300. 10.1111/nep.12609 . [DOI] [PubMed] [Google Scholar]
- 27.Coupes B, de Freitas DG, Roberts SA, Read I, Riad H, Brenchley PE, et al. rhErythropoietin-b as a tissue protective agent in kidney transplantation: a pilot randomized controlled trial. Nephrology (Carlton, Vic). 2015;8:21 Epub 2015/09/16. 10.1111/nep.1260910.1186/s13104-014-0964-0 . [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(SAV)
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.