Abstract
The plant-specific TCP transcription factors are well-characterized in both monocots and dicots, which have been implicated in multiple aspects of plant biological processes such as leaf morphogenesis and senescence, lateral branching, flower development and hormone crosstalk. However, no systematic analysis of the petunia TCP gene family has been described. In this work, a total of 66 petunia TCP genes (32 PaTCP genes in P. axillaris and 34 PiTCP genes in P. inflata) were identified. Subsequently, a systematic analysis of 32 PaTCP genes was performed. The phylogenetic analysis combined with structural analysis clearly distinguished the 32 PaTCP proteins into two classes—class Ι and class Ⅱ. Class Ⅱ was further divided into two subclades, namely, the CIN-TCP subclade and the CYC/TB1 subclade. Plenty of cis-acting elements responsible for plant growth and development, phytohormone and/or stress responses were identified in the promoter of PaTCPs. Distinct spatial expression patterns were determined among PaTCP genes, suggesting that these genes may have diverse regulatory roles in plant growth development. Furthermore, differential temporal expression patterns were observed between the large- and small-flowered petunia lines for most PaTCP genes, suggesting that these genes are likely to be related to petal development and/or petal size in petunia. The spatiotemporal expression profiles and promoter analysis of PaTCPs indicated that these genes play important roles in petunia diverse developmental processes that may work via multiple hormone pathways. Moreover, three PaTCP-YFP fusion proteins were detected in nuclei through subcellular localization analysis. This is the first comprehensive analysis of the petunia TCP gene family on a genome-wide scale, which provides the basis for further functional characterization of this gene family in petunia.
Keywords: petunia, TCP transcription factors, expression patterns, petal development, floral organ size
1. Introduction
The final shape and size of lateral organs such as leaves and flowers are established mainly by the elaborate coordination of cell proliferation and cell expansion in response to internal developmental and external environmental cues. This coordination relies on the complex regulatory circuits in which many microRNAs (miRNAs) and transcription factors are recruited [1]. The TCP gene family, first described in 1999 and named after the initials of four characterized members, namely Teosinte Branched 1 (TB1) from maize (Zea mays), CYCLOIDEA (CYC) from snapdragon (Antirrhinum majus) and PROLIFERATING CELL FACTORS (PCF1 and PCF2) from rice (Oryza sativa), encompasses a group of phylogenetically related and plant-specific transcription factors (TFs). TCP proteins are featured by a highly conserved non-canonical basic helix–loop–helix (bHLH) DNA-binding domain referred to as the TCP domain, which is responsible for the mediation of DNA binding, nuclear localization as well as interactions with other proteins [2]. Based on the differential features within the conserved TCP domain, TCP proteins can be categorized into two classes: class I (or TCP-P clade) and class II (or TCP-C clade), and the latter can be further divided into the ubiquitous CINCINNATA (CIN-TCP subclade) and the angiosperm-specific CYC/TB1 subclade [3,4]. Different from class II TCPs, the class I TCP proteins possess a four-amino-acid deletion in the basic region within the TCP domain. Besides the conserved TCP domain, some class II members contain an arginine-rich R domain with unknown biological function, and the majority of the CYC/TB1 subclade members have a conserved ECE motif (a glutamic acid-cysteine-glutamic acid stretch) that remains functionally uncharacterized [3,5].
Accumulating evidence indicates that TCP transcription factors have versatile functions in a plethora of plant developmental processes, such as lateral branching, leaf development and senescence, flowering and flower development, gametophyte development, seed germination and defense responses [6,7,8,9,10,11,12,13,14,15,16]. The crosstalk between TCP transcription factors and hormone signaling as well as their roles in mediating the hormone activity in cell proliferation has been reviewed in detail [17]. As for class II TCP, five Arabidopsis CIN-like genes (TCP2-4, 10 and 24), which are targeted by miR319, have been shown to participate in the transcriptional control of cell cycle genes during leaf development and morphogenesis. Overexpression of miR319 or downregulation of TCP genes induce excess cell proliferation, thus leading to crinkled leaves [8,10,18]. Three nontargeted TCP5-like genes are demonstrated to promote the transition from cell division to post-mitotic cell expansion during petal development [19]. Moreover, Arabidopsis CIN-TCPs have been implicated in the regulation of floral organ development and flowering time [20,21]. Recent advances in the understanding of the function and regulation of CIN-like TCP genes have been summarized by Qin et al. [22]. To our knowledge, the CYC/TB1 clade mainly plays a prominent role in axillary meristem development [3]. AtTCP18 and AtTCP12, two homologs of maize TB1, are identified as integrators of branching signals and negatively regulate the axillary bud outgrowth [6,23]. AtTCP18 also engages in suppressing the precocious floral transition of the axillary meristems by interacting with florigen proteins [24]. AtTCP1, the homolog of CYC, is associated with brassinosteroid (BR) biosynthesis and the longitudinal elongation of leaves [25,26].
By contrast, class I TCPs are suggested to promote cell proliferation and plant growth [3]. Most loss-of-function mutants of class I TCP genes exhibit mild or no defective phenotypes due to genetic redundancy [9]. Even so, several class I TCP transcription factors have been integrated into multiple hormone-signaling pathways and participated in the regulation of inflorescences stem elongation [27], gynoecium development [28], seed germination [15], filament elongation of stamen [29] and flowering [13] in Arabidopsis. AtTCP20 binds to the GCCCR motif of the CYCB1 promoter and participates in the regulation of cell division, cell expansion and growth coordination [30]. Given that both classes of TCP proteins have distinct but overlapping DNA binding sites: GGNCCC [31], it has been reported that class I and II TCP proteins participate in antagonistic and synergistic biological interactions [32]. For example, AtTCP15 indirectly regulates the boundary-specific genes through the auxin pathway, which partly overlaps with the one affected by CIN-TCP genes [33,34]. AtTCP20 and its direct target AtTCP9 repress the jasmonate acid (JA) biosynthesis, which is antagonized by the CIN-TCP protein TCP4 [10,35].
To date, the TCP gene family has been identified and characterized in various plant species, such as Arabidopsis and rice [36], tomato [37], Prunus mume [38], watermelon [39], strawberry [40], poplar [41], turnips [42], Chinese cabbage [43], Medicago [44], chickpea [45], switchgrass [46] and grapevine [47]. Petunia is well-known as a popular and important ornamental plant belonging to the Solanaceae family. Considering the advantages of the highly efficient genetic transformation system, ease of cultivation and propagation as well as genetic diversity, petunia has been used as a genetic model system for a long history [48,49]. For ornamental plants, flower size is an important trait and is always the breeding objective of horticultural breeders. Recently, the whole-genome sequencing and assembly of two wild parents of petunia, P. axillaris and P. inflata, was achieved [50], which contribute significantly to identify and characterize TCP genes in petunia on a genome-wide scale. However, only three petunia CYC/TB1 subclade genes (PhTCP1-PhTCP3, isolated from Petunia hybrida inbred line V26) are functionally characterized as the regulators of the strigolactone (SL) signaling pathway [51]; no comprehensive analysis of the petunia TCP gene family was reported. In this study, a genome- and transcriptome-wide analysis of the TCP gene family in petunia was carried out for the first time. The present work identified 32 PaTCP genes from P. axillaris, together with analyzing their gene classification, phylogenetic relationships, conserved motifs, exon-intron organization, putative promoter cis-acting elements, as well as subcellular localization. In addition, the spatiotemporal expression profiles of PaTCP genes were investigated by real-time quantitative RT-PCR (qPCR). This study lays a good basis for further exploring the potential roles of PaTCP genes in plant growth and development.
2. Results
2.1. Identification of TCP Genes in Petunia Species
To identify the petunia TCP genes, Arabidopsis and tomato TCP protein sequences as well as the HMM profile of the TCP domain (PF03634) were used to search against the genome and transcriptome datasets of P. axillaris and P. inflata [50,52]. As a result, 32 PaTCP genes were identified in P. axillaris (Table 1, Table S1) and 34 PiTCP genes were identified in P. inflata (Table S2). We named these petunia TCP genes according to the closest homologs of them in Arabidopsis. The number of TCP genes in P. axillaris (32 members) and P. inflata (34 members) is almost 1.3 and 1.4 times more, respectively, than that in Arabidopsis (24 members), which is consistent with protein-coding genes in the P. axillaris genome (32,928) and P. inflata genome (36,697) compared to the Arabidopsis genome (25,498) [50,53].
Table 1.
TCP genes identified in Petunia axillaris.
| Gene Name | GenBank Accession Number | Genomic Location | Protein Length (aa) | MW (Da) | pI | Type |
|---|---|---|---|---|---|---|
| PaTCP2 | MT293300 | Peaxi162Scf00939: 373844-373149 | 231 | 25,346.90 | 4.49 | CIN |
| PaTCP3a | MT293301 | Peaxi162Scf00069: 1992329-1991073 | 418 | 45,694.00 | 6.27 | CIN |
| PaTCP3b | MT293302 | Peaxi162Scf00000: 2512603-2513853 | 416 | 45,483.18 | 6.11 | CIN |
| PaTCP4a | MT293303 | Peaxi162Scf00189: 194164-193223 | 313 | 35,529.20 | 6.59 | CIN |
| PaTCP4b | MT293304 | Peaxi162Scf00241: 1053171-1054422 | 314 | 35,195.08 | 6.46 | CIN |
| PaTCP5 | MT293305 | Peaxi162Scf00013: 698884-699834 | 316 | 35,316.19 | 8.67 | CIN |
| PaTCP10 | MT293310 | Peaxi162Scf00269: 345789-344431 | 452 | 50,862.17 | 5.87 | CIN |
| PaTCP13 | MT293314 | Peaxi162Scf00166: 407926-406976 | 316 | 35,253.97 | 7.95 | CIN |
| PaTCP17 | MT293318 | Peaxi162Scf00201: 332740-333747 | 335 | 37,477.28 | 5.61 | CIN |
| PaTCP24a | MT293328 | Peaxi162Scf00317: 44365-45591 | 408 | 45,311.45 | 5.83 | CIN |
| PaTCP24b | MT293329 | Peaxi162Scf00317: 35045-36322 | 425 | 46,728.30 | 6.61 | CIN |
| PaTCP1a | MT293298 | Peaxi162Scf00479: 76745-78241 | 362 | 40,884.15 | 9.75 | CYC |
| PaTCP1b | MT293299 | Peaxi162Scf00535: 12436-13766 | 333 | 37,208.83 | 9.65 | CYC |
| PaTCP12a | MT293312 | Peaxi162Scf00086: 565892-567112 | 406 | 46,671.52 | 6.48 | CYC |
| PaTCP12b | MT293313 | Peaxi162Scf00367: 862637-863788 | 383 | 43,907.64 | 6.33 | CYC |
| PaTCP18a | MT293319 | Peaxi162Scf00119: 242135-243400 | 343 | 39,094.66 | 9.07 | CYC |
| PaTCP18b | MT293320 | Peaxi162Scf00013: 930102-931799 | 365 | 41,213.33 | 5.97 | CYC |
| PaTCP18c | MT293321 | Peaxi162Scf00015: 735479-737065 | 341 | 38,603.76 | 7.25 | CYC |
| PaTCP6 | MT293306 | Peaxi162Scf00128: 1570847-1571590 | 247 | 27,160.02 | 8.76 | PCF |
| PaTCP7 | MT293307 | Peaxi162Scf00681: 139168-139935 | 255 | 26,952.13 | 9.24 | PCF |
| PaTCP8 | MT293308 | Peaxi162Scf00011: 881162-882733 | 523 | 56,100.68 | 6.82 | PCF |
| PaTCP9 | MT293309 | Peaxi162Scf00021: 216329-217791 | 309 | 33,695.65 | 10.01 | PCF |
| PaTCP11 | MT293311 | Peaxi162Scf00123: 1704660-1705283 | 207 | 22,320.35 | 8.86 | PCF |
| PaTCP14a | MT293315 | Peaxi162Scf00013: 171612-173102 | 332 | 36,667.84 | 9.44 | PCF |
| PaTCP14b | MT293316 | Peaxi162Scf00274: 795778-797097 | 413 | 44,322.68 | 6.86 | PCF |
| PaTCP15 | MT293317 | Peaxi162Scf00541: 598300-599367 | 355 | 38,885.66 | 7.02 | PCF |
| PaTCP19a | MT293322 | Peaxi162Scf00192: 477569-478627 | 352 | 37,603.70 | 5.12 | PCF |
| PaTCP19b | MT293323 | Peaxi162Scf00620: 802496-803548 | 350 | 37,896.90 | 9.23 | PCF |
| PaTCP20 | MT293324 | Peaxi162Scf00420: 631358-632224 | 288 | 31,161.43 | 7.94 | PCF |
| PaTCP21 | MT293325 | Peaxi162Scf00021: 2123474-2124109 | 211 | 22,362.10 | 9.46 | PCF |
| PaTCP22 | MT293326 | Peaxi162Scf00391: 454319-455563 | 414 | 44,056.39 | 7.40 | PCF |
| PaTCP23 | MT293327 | Peaxi162Scf00268: 644156-645127 | 323 | 34,471.24 | 7.89 | PCF |
aa: Amino acid; pI: Isoelectric point; MW: Molecular weight.
Sequence analysis demonstrated that the length of PaTCP genes varied from 624 bp (PaTCP11) to 1698 bp (PaTCP18b) and the ORF length of PaTCPs varied from 624 bp (PaTCP11) to 1572 bp (PaTCP8); the mean length of the 32 deduced PaTCP proteins was 346 amino acids (range 207-523 amino acids) (Table 1, Table S1). Similarly, the length of PiTCP genes varied from 624 bp (PiTCP11) to 1584 bp (PiTCP18c), the ORF length of PiTCPs varied from 624 bp (PiTCP11) to 1572 bp (PiTCP8), and the size of deduced PiTCP proteins ranged from 207 to 523 amino acids with an average of 350 amino acids (Table S2). More sequence characteristics of PaTCPs and PiTCPs, such as the isoelectric point (pI), molecular weight (MW) and the location at Petunia scaffolds, are listed in Table 1 and Table S2, respectively.
2.2. Phylogenetic Analysis of TCP Proteins from Petunia, Tomato and Arabidopsis
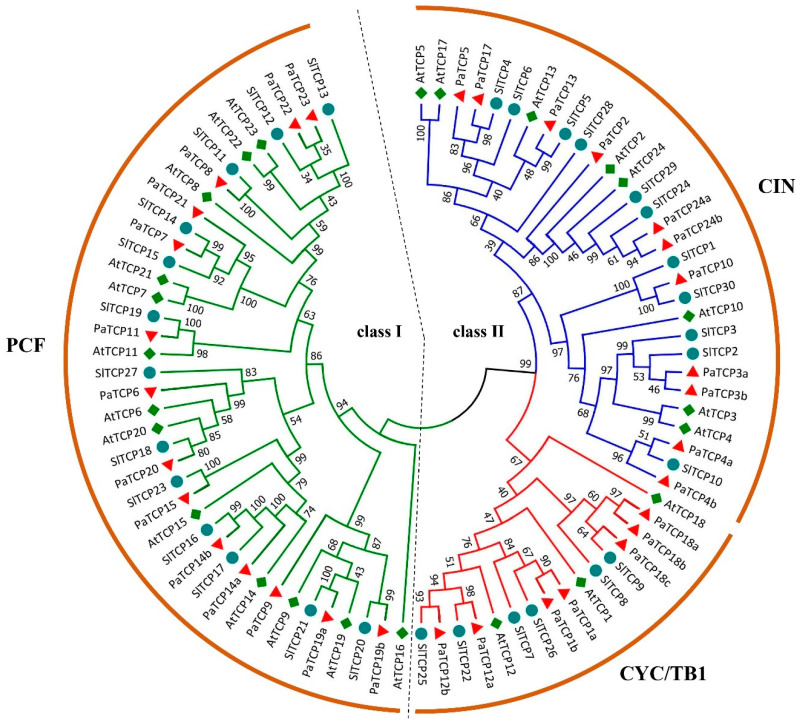
To clarify the evolutionary and phylogenetic relationships between the PaTCP proteins and other known TCPs, an unrooted phylogenetic tree was generated with the neighbor-joining (NJ) method using the full-length sequences of 32 PaTCP proteins together with 24 Arabidopsis TCP proteins (AtTCPs) and 30 tomato TCP proteins (SlTCPs) (Figure 1). Multiple alignments of the conserved TCP domains of 32 PaTCPs were also performed (Figure 2a). Both the phylogenetic analysis and multiple alignments of the TCP domains suggested that these PaTCP proteins were classified into two classes: 14 of 32 PaTCPs belong to class I (PCF/TCP-P clade) because of a deletion of four amino acids in the basic region, and 18 belong to class II (TCP-C clade) which could be further divided into the CYC/TB1 subclade and CIN subclade (Figure 1 and Figure 2a). The class I PaTCPs encompassed a group of relatively closely related proteins with extended homology from the TCP domain to C-terminal. In addition, the key residues in the basic region exhibited high conservation, while the residues in the loop and the hydrophilic residues of the helices were relatively less conserved in both classes I and II PaTCPs (Figure 2a). Notably, PaTCP1a protein lacked the conserved residues of the basic region at the N-terminus, suggesting that this protein may have a deficiency of DNA binding ability.
Figure 1.
Phylogenetic relationship of TCP transcription factors from petunia, Arabidopsis and tomato, based on the multiple alignments of the full-length amino acid sequences of TCP proteins from P. axillaris (PaTCP), tomato (SlTCP) and Arabidopsis (AtTCP). The neighbor-joining (NJ) algorithm-based phylogenetic tree was built using MEGA7.0 software with 500 bootstrap replicates. Green, red and blue lines represent the PCF, CYC/TB1 and CIN subclades, respectively.
Figure 2.
Multiple sequence alignments of PaTCP proteins and analysis of the putative miR319 binding sites. (a). Alignment of the TCP domain and adjoining sequence for the predicted PaTCP proteins. Overall conserved amino acids were shaded in black. The Basic, Helix I, Loop and Helix II regions are indicated. (b). Alignment of the R-domain of class II PaTCP members. (c). Alignment of putative target areas for PamiR319 (aligned in reverse).
Except for the TCP domain, eight class II proteins, PaTCP1a, PaTCP1b, PaTCP12a, PaTCP18a, PaTCP18b, PaTCP18c, PaTCP24a and PaTCP24b, shared the R domain (18-residues arginine-rich motif) at the C-terminus downstream of the TCP domain, and an R domain-like sequence was present in PaTCP12b, but less conserved (Figure 2b). Additionally, seven PaTCPs (PaTCP3a, 3b, 4a, 4b, 10, 24a and 24b) contained the putative miR319 target sites (Figure 2c). However, PaTCP2, the closest petunia homolog of AtTCP2, had no miR319 target site due to a nonsense mutation just after the TCP domain.
2.3. Gene Structure and Conserved Motif Analysis
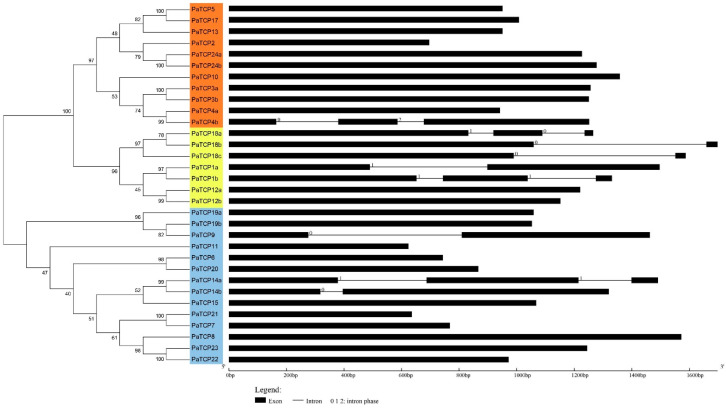
To further explore the structural features of petunia TCP genes, the exon-intron organization was analyzed. The class II PaTCP genes contained more introns than those of class Ι genes (Figure 3). Among the class I PaTCP genes, 11 of 14 had no intron and the other three genes had one or two introns. For CIN subclade genes, only PaTCP4b contained two introns and all the others had no intron. In contrast, most CYC/TB1 subclade genes possessed one or more intron, with the exception of PaTCP12a and PaTCP12b, which had no intron. Moreover, similar exon/intron structures were also observed in PiTCPs (Figure S1).
Figure 3.
Exon-intron structures of PaTCP genes. The cladogram was produced by MEGA 7.0 software with the NJ method using the full-length sequences of PaTCP proteins. The orange, yellow and blue rectangles were used to cluster the genes into the CIN, CYC/TB1 and PCF subclades, respectively. 5′UTR and 3′UTR were not shown. The exons and introns are represented by a black block and a thin gray line, respectively. The 0, 1 and 2 denote intron phases.
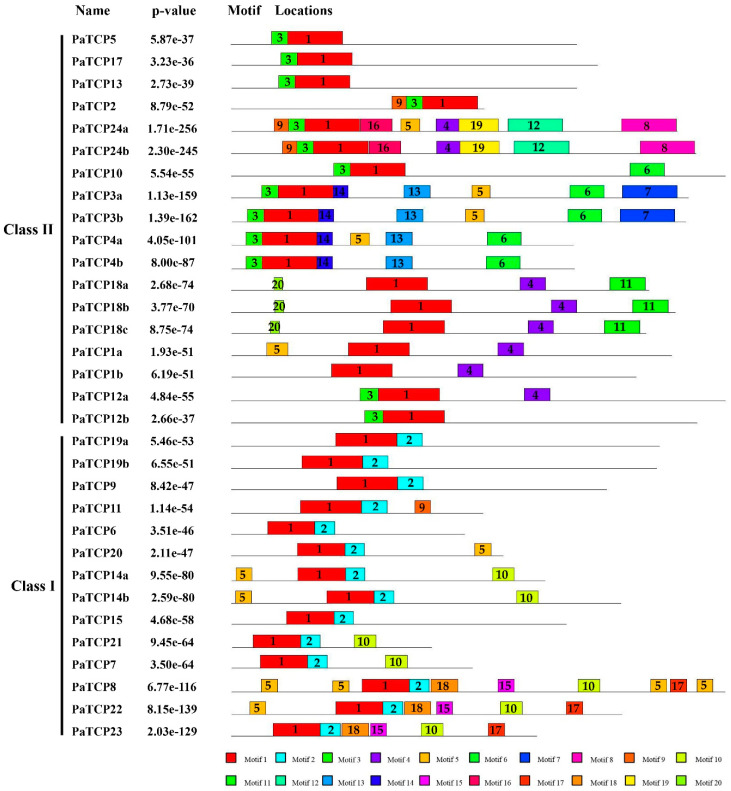
To get more understanding of the structural and functional divergences of the PaTCP proteins, the conserved motif compositions of PaTCPs were investigated using the online MEME tool. As shown in Figure 4, a total of 20 motifs were identified and designated as motif 1 to motif 20. The number of motifs varied from 2 to 10. Three motifs (motif 1, 2 and 3) were identified as the TCP domain, and motif 1 was uniformly observed in all PaTCP proteins. Some motifs only presented in a specific subclade. For instance, all class I proteins shared motif 2 in the C-terminal TCP domain, while motif 3 of the N-terminal TCP domain was conserved in all CIN subclade members. In addition to the TCP domain, motif 4, identified as the conserved R domain, was hit in six CYC/TB1 proteins and two CIN subclade proteins. Moreover, some motifs were exclusively present in specific subfamily members, such as motif 6, 7, 8, 12, 13 and 14 in the CIN-TCP subclade, motif 11 and 20 in CYC/TB1 subclade and motif 10, 15, 17 and 18 in class I members. Similar motif compositions were observed in PaTCP proteins, which were clustered into the same clade or subclade, while significant divergences were found in different subclades (Figure 4), suggesting that PaTCP proteins likely have functional divergence between subclades and functional redundancy within the subclade.
Figure 4.
Distribution of the conserved and potential motifs of PaTCP proteins. Each motif is represented by rectangular boxes with different colors and numbered from 1 to 20.
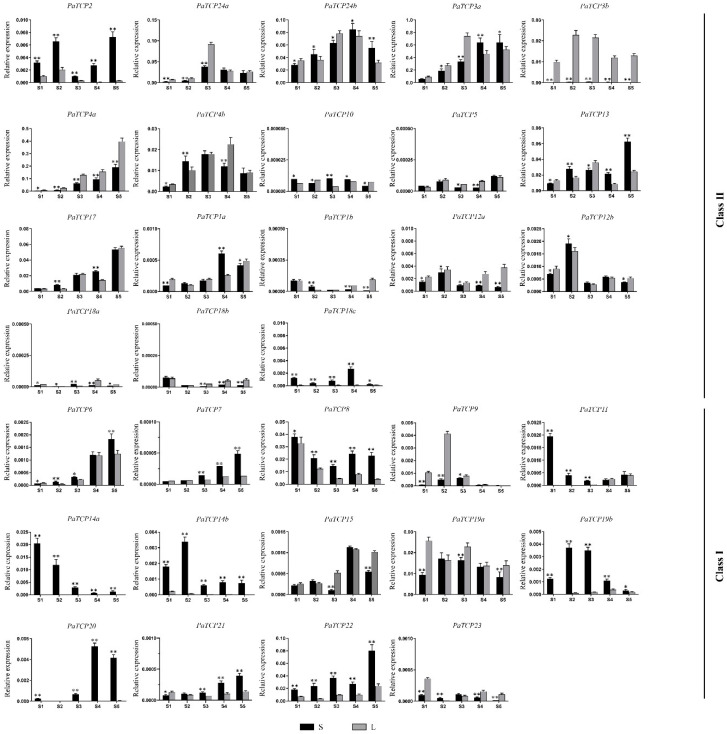
2.4. Expression Profiles of PaTCP Genes
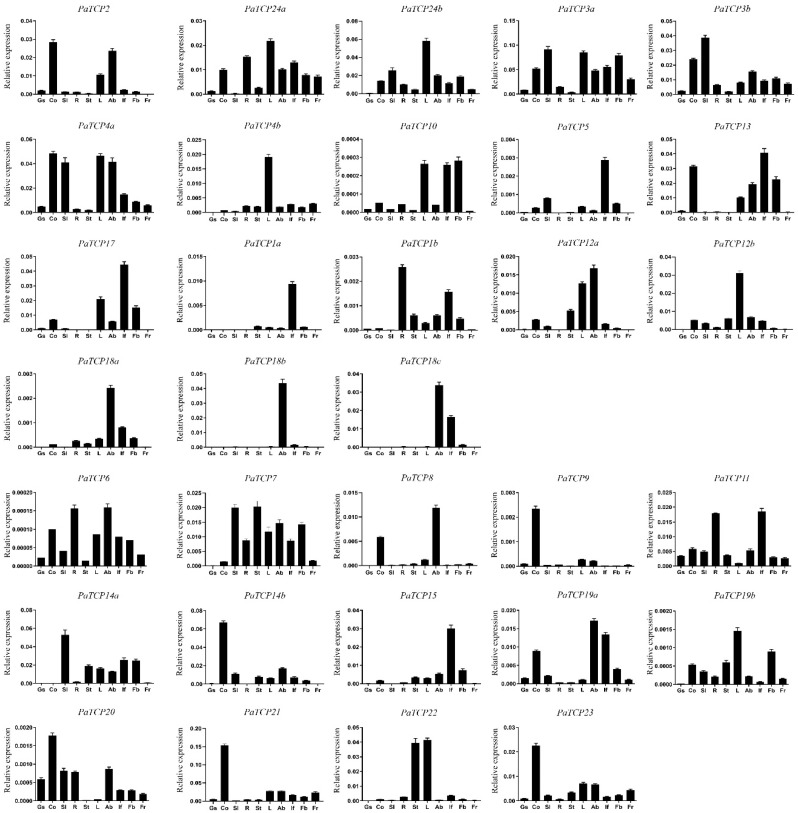
In order to predict the potential function for PaTCP genes, we examined the spatial expression profiles of 32 PaTCP genes in various tissues, including germinating seeds, cotyledons, seedlings, roots, stems, leaves, axillary buds, inflorescences, flower buds and fruits by qPCR. As indicated in Figure 5, the expression patterns of PaTCP genes could be classified into four groups. The first group was ubiquitously expressed in almost all tested tissues, including PaTCP3a, PaTCP3b, PaTCP4a, PaTCP7, PaTCP11, PaTCP19a, PaTCP21, PaTCP23, PaTCP24a and PaTCP24b. Of these, PaTCP3a, PaTCP3b, PaTCP4a, PaTCP24a and PaTCP24b showed relatively high levels of expression in most tissues with lower expression in germinating seeds, stems and roots or seedlings. PaTCP11 showed the highest expression in inflorescences and roots. PaTCP21 and PaTCP23 showed the highest expression in cotyledons. The second group, including PaTCP1b, PaTCP5, PaTCP9, PaTCP18a, PaTCP19b and PaTCP20, especially PaTCP6 and PaTCP10, showed a very low expression level (less than 0.005 relative to that of the reference gene PhEF1α) in all tissues (Figure 5). Among these genes, PaTCP5, PaTCP9 and PaTCP18a showed more predominant expression in inflorescences, cotyledons and axillary buds, respectively. The third group exhibited a tissue-preferential expression pattern, including PaTCP1a, PaTCP4b, PaTCP8, PaTCP12b, PaTCP14b, PaTCP15, PaTCP18b, PaTCP18c, PaTCP21 and PaTCP22. For example, PaTCP1a and PaTCP15 showed a significantly higher level of expression in inflorescences. PaTCP4b and PaTCP12b were predominantly expressed in leaves, while PaTCP8, PaTCP18b and PaTCP18c were mainly detected in axillary buds. The transcript of PaTCP22 was mostly detected in stems and leaves, while PaTCP14b was dominantly expressed in cotyledons. The fourth group, including PaTCP2, PaTCP12a, PaTCP13, PaTCP14a and PaTCP17, was expressed specifically in partial tissues. For instance, PaTCP2, PaTCP13, PaTCP14a and PaTCP17 showed higher expression in cotyledons, inflorescences, flower buds and axillary buds, with very weak or no expression in germinating seeds, seedlings, roots, stems and fruits. PaTCP12a had the highest expression in axillary buds followed by leaves and stems.
Figure 5.
Expression analysis of PaTCP genes by qPCR. Gs, germinating seeds; Co, cotyledons; Sl, young seedlings; R, roots; St, stems; L, leaves; Ab, axillary buds; If, inflorescences; Fb, flower buds (0.5 cm); Fr, fruits. The relative expression level was normalized to the petunia EF1α gene. For each tissue, three biological replicates were used to calculate the mean values ± SD (standard deviation) with the 2−ΔΔCT method.
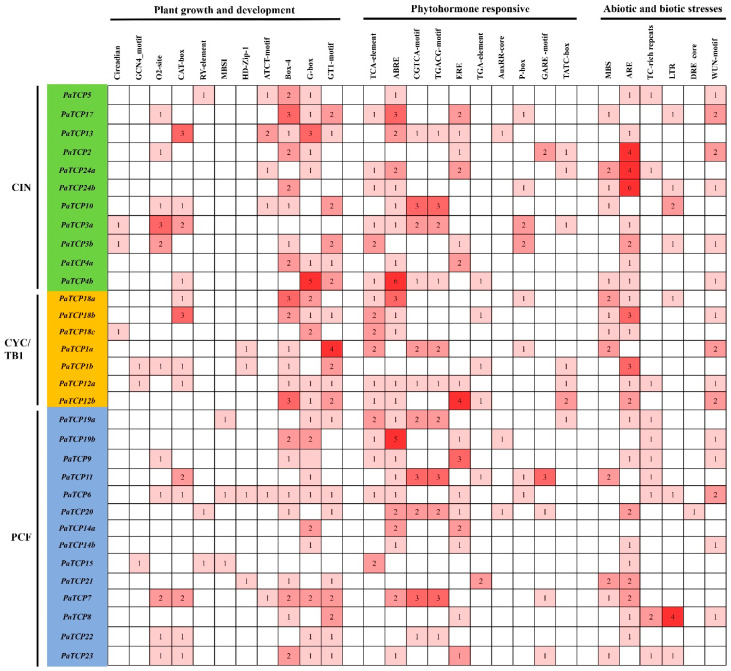
2.5. Identification of Cis-Acting Elements in the Promoter of PaTCP Genes
To investigate the potential regulatory mechanism of PaTCP genes, the promoter regions (1.5 kb of genomic DNA sequence upstream of the translational start site) of the PaTCPs were submitted to the PlantCARE database [54]. Apart from the common CAAT-box and TATA-box, a large number of cis-elements responsible for plant growth and development, phytohormone responses, as well as abiotic and biotic stress responses were identified (Figure 6). The zein-metabolism regulation element (O2 site) and meristem expression and specific activation element (CAT-box) were found in 11 and 13 PaTCP genes, respectively. Additionally, some cis-regulatory elements related to plant growth and development, including leaf development-related element (HD-Zip1), the circadian control element (circadian), the endosperm-specific expression element (GCN4_motif), the flavonoid biosynthetic (MBSI), as well as the seed-specific regulation element (RY element), were also identified in four, three, three, three and three PaTCP genes, respectively. Meanwhile, many regulatory elements involved in light responsiveness were detected, such as ATCT-motif, Box-4, G-box and GT1-motif. In hormone-related cis-acting elements, the ABA-responsive element (ABRE), the SA-responsive element (TCA element) and the ethylene-responsive element (ERE) were frequently identified in the promoter regions of 24, 17 and 16 PaTCP genes, respectively. The gibberellin-responsive elements (GARE-motif, P-box and TATC-box) and MeJA-responsive elements (CGTCA-motif and TGACG-motif) were observed in the promoter regions of 17 and 11 PaTCP genes, respectively. Auxin-responsive elements (TGA element and AuxRR-core) were also found in the promoter regions of nine PaTCP genes. Additionally, some stress-related cis-acting elements such as drought-inducibility (MBS), anaerobic induction (ARE), low-temperature (LTR) and wounds (WUN-motif) were also detected in the promoter regions of PaTCP genes, which indicated that these PaTCP genes were likely to participate in defense signaling.
Figure 6.
Cis-regulatory elements analysis of PaTCP promoters. Based on the functional annotation, the cis-acting elements were categorized into three major classes: plant growth and development, phytohormone responsive or abiotic and biotic stresses-related cis-acting elements (details are shown in Table S3).
2.6. Transcriptional Profiles of PaTCPs during Petal Development in Large- and Small-Flowered Lines
To gain an in-depth knowledge of the potential roles of PaTCP genes during petal development, we carried out qPCR to further investigate the transcript accumulation patterns of PaTCPs at five different petal developmental stages (S1–S5). These stages cover the developmental range from young flower bud to fully blooming flower. Samples were taken from the corollas of large- and small-flowered lines ‘L’ and ‘S,’ respectively, which originated from the self-cross separation of the same inbred line. The mean size of flower diameter of ‘L’ (8.53 ± 0.18 cm) is much larger than that of ‘S’ (3.54 ± 0.13 cm) (Figure S2). All 32 PaTCP genes were detected during petal development in both large- and small-flowered lines.
As shown in Figure 7, seven PaTCP genes were gradually upregulated during the petal development in ‘L’ and/or ‘S,’ showing the highest expression in fully blooming corollas (S5). Of these, three class II TCP genes (PaTCP3a, PaTCP4a and PaTCP17) and four class I TCP genes (PaTCP6, PaTCP7, PaTCP21 and PaTCP22) were included. On the contrary, several PaTCPs, such as PaTCP8, PaTCP11, PaTCP14a, PaTCP19a and PaTCP23, showed downregulated patterns, with the highest expression in young flower buds (S1 or S2). Moreover, the expression of PaTCP4b, PaTCP15, PaTCP20, PaTCP24a and PaTCP24b increased first and then dropped at later stages, with the highest expression in semi-open (S4) or fully blooming (S5) corollas. PaTCP3b, PaTCP9, PaTCP12b and PaTCP14b shared similar expression trends, but with the highest expression in young flower buds (S2). However, some PaTCP genes, including PaTCP1a/b, PaTCP5, PaTCP10, PaTCP12a and PaTCP18a/b/c, showed very low expression levels during all the petal developmental stages, and no regular trend was observed. These results suggested that most PaTCP genes should participate in the regulation of petal development in petunia.
Figure 7.
Transcriptional profiles of PaTCP genes in the large- and small-flowered lines, ‘L’ and ‘S,’ respectively, during different petal developmental stages: young flower buds (<0.5 cm, S1), extending flower buds (when flower buds just enclosed by sepals, S2), pre-anthesis (when flower buds extended to full length, S3), semi-open flowers (S4) and fully blooming flowers before the anthers dehisced (S5). The results were normalized to the expression of the petunia EF1α gene. The mean values ± SD (standard deviation) were calculated from three biological replicates with the 2−ΔΔCT method. The black and dark grey square represented the ‘S’ and ‘L,’ respectively. Asterisks denote statistically significant differences between ‘S’ and ‘L’ at each developmental stage, as determined by Student’s t-test (* p < 0.05, ** p < 0.01).
Some PaTCP genes showed differential expression patterns between the large- and small-flowered lines during petal development process (Figure 7, Figure S3). The expression levels of eight genes (PaTCP2, PaTCP8, PaTCP14a, PaTCP14b, PaTCP18c, PaTCP19b, PaTCP20 and PaTCP22) were significantly higher in ‘S’ than in ‘L’ during petal development. Among them, most genes belonged to the TCP-P clade. However, three genes (PaTCP3b, PaTCP4a and PaTCP9) exhibited a relatively higher expression in ‘L’ than in ‘S.’ For PaTCP3b, high levels of expression were observed in ‘L,’ while almost no transcript was detected in ‘S’ at various petal developmental stages.
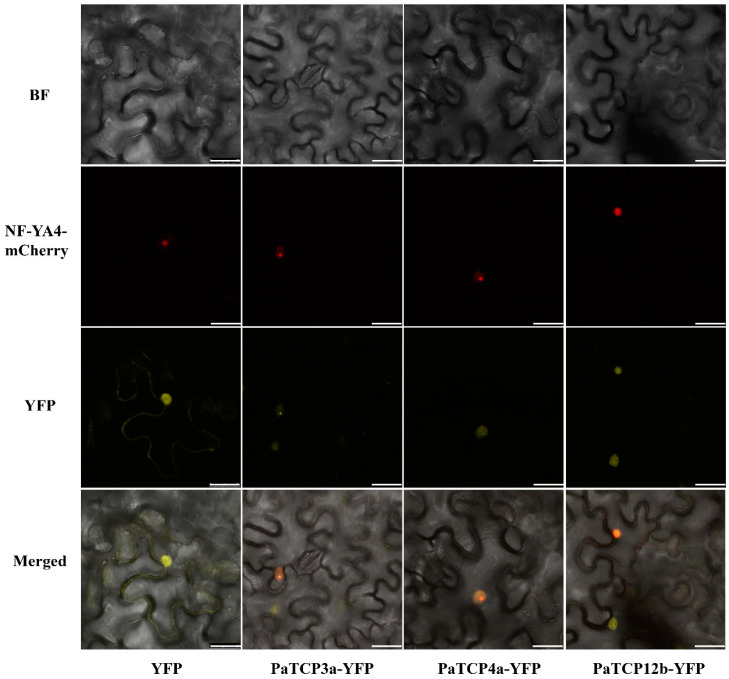
2.7. Subcellular Localization of PaTCP Proteins
It has been reported that TFs are usually involved in regulating the transcription of target genes by binding to specific cis-elements in their promoters. We further examined the subcellular localization of three PaTCP proteins. The full-length coding sequences without the stop codon of three cloned PaTCP genes (PaTCP3a, PaTCP4a and PaTCP12b) were inserted into a vector with yellow fluorescence protein (YFP) under the control of the CaMV 35S promoter. Yellow fluorescence signals of these PaTCP-YFP fusion proteins were detected in the nuclei, which was consistent with the putative prediction of transcription factor activities of the PaTCP proteins (Figure 8).
Figure 8.
Subcellular localization of three PaTCP-YFP fusion proteins in tobacco. Empty vector of yellow fluorescence (YFP) was used as control. The co-transformation of the nuclear marker (NF-YA4-mCherry) was used to visualize the nuclei. Merged panel showed the nuclear localization in tobacco epidermis cells. Scale bar, 25 μm.
3. Discussion
3.1. Evolution of TCP Genes in P. axillaris
The plant-specific TCP transcription factors have been implicated in exerting regulatory roles in multiple aspects of physiological and biological processes during plant growth and development [32]. In this study, we identified 32 PaTCPs from P. axillaris genome and 34 PiTCPs from P. inflata genome. The phylogenetic analysis and sequence alignment showed that all 32 PaTCPs were classified into three major groups: PCF-TCPs, CIN-TCPs and CYC-TCPs (Figure 1 and Figure 2a). The conserved TCP domain was shared by all PaTCP members, suggesting that PaTCP transcription factors possess the capacity of dimerization and DNA binding [2]. Moreover, all Arabidopsis and tomato TCPs were clustered into the same clade or subclade as reported previously [36,37]. Furthermore, most PaTCP members within the same clade and/or subclade display a similar exon-intron organization in terms of exon length and intron number (Figure 3), and relatively conserved motif compositions (Figure 4), which further support the close evolutionary relationships among PaTCPs and the reliability of our phylogenetic analysis.
The TCP gene family is expanded (32 PaTCPs) in petunia compared with that in Arabidopsis (24 AtTCPs) but comparable to the family in tomato (30 SlTCPs), which may be the consequences of more genome duplication events in the Solanaceae family [50]. In addition, PaTCP24a and PaTCP24b were clustered into the same branch and located closely in the same assembled scaffold (Table 1, Figure 1), suggesting that tandem duplication may be another reason for the expansion of the TCP gene family in petunia. Interestingly, the number of protein-coding genes in P. axillaris genome (32,928) and P. inflata genome (36,697) are in strong correspondence with the fact that the number of protein-coding genes is 25,498 in the Arabidopsis genome [50]. From the phylogenetic tree, most Arabidopsis class II TCP genes contain more than one counterpart in petunia and tomato; especially in the CYC/TB1 subclade, where each gene has two or more counterparts (Figure 1), such as the PaTCP1a/PaTCP1b and SlTCP7/SlTCP27 gene pairs vs. AtTCP1, PaTCP12a/PaTCP12b and SlTCP22/SlTCP25 vs. AtTCP12 and PaTCP18a/PaTCP18b/PaTCP18c and SlTCP8/SlTCP9 vs. AtTCP18. Furthermore, there are two orthologs in petunia corresponding to one SlTCP in tomato and vice versa two tomato orthologs corresponding to one petunia gene for several members, such as PaTCP24a and PaTCP24b vs. SlTCP24, PaTCP4a and PaTCP4b vs. SlTCP10, PaTCP19a and PaTCP19b vs. SlTCP20, SlTCP5 and SlTCP28 vs. PaTCP13, as well as SlTCP1 and SlTCP30 vs. PaTCP10 (Figure 1). It may be the consequence of the differential duplication events or frequencies of retaining copies after the divergence of tomato and petunia from a common ancestor. Only AtTCP16 is separate without a close homolog in petunia, and a similar result was also observed in tomato [37], implying a lineage-specific gene loss in petunia and tomato.
3.2. Expression Patterns and Potential Functions of PaTCP Genes
The TCP gene family has become the focus of functional studies at multiple levels after its initial definition in 1999 [32]. At present, however, very little is known about the function of TCP genes in petunia. It is widely recognized that the expression profile of a gene is tightly linked with its function to a large extent. In this study, the expression profiles of PaTCPs in various tissues were investigated by qPCR. The results indicate that the transcript accumulation pattern in different tissues varies widely among PaTCP genes (Figure 5), suggesting their functional diversification during plant growth and development.
Previous studies have shown that the CIN subclade TCP genes mainly participate in the regulation of flowering time, floral organ development, leaf development and senescence, secondary cell wall thickening and morphogenesis of lateral organs [55,56]. In Arabidopsis, five CIN-like genes (TCP2, 3, 4, 10 and 24) are post-transcriptionally regulated by microRNA319 (miR319). High levels of miR319 or low miR319-regulated TCP activity induce serrated and crinkled leaves due to excessive cell proliferation and delay leaf senescence by inhibiting the biosynthesis of jasmonic acid [8,10,33]. Proper regulation of TCP4 by miR319a is critical for petal and stamen development [12]. In tomato, seven SlTCP genes are predicted to be the targets of miR319, and LANCEOLATE (LA, SlTCP2), regulated by miR319, is required for the formation of compound leaves [37,57]. In petunia, seven closest homologs of the CIN-like genes (PaTCP3a, 3b, 4a, 4b, 10, 24a and 24b) have the putative binding sites for PamiR319 (Figure 2c). High expression levels of PaTCP3a, PaTCP4a/PaTCP4b, PaTCP10 and PaTCP24a/PaTCP24b in leaves suggest their potential roles in leaf development, which may be conserved for the TCP genes targeted by miR319 [8,55,57,58,59]. Moreover, the transcripts of most putative miR319-regulated PaTCP genes are detected in most tested tissues, including seedlings, cotyledons, leaves, axillary buds, inflorescences and flower buds, suggesting that these PaTCPs may play diverse regulatory roles at multiple developmental processes in petunia. Recently, the TCP4-like genes were proved to play a positive role in light-induced cotyledon opening via directly targeting and transcriptionally activating several SAUR (SMALL AUXIN UPREGULATED RNA) genes in Arabidopsis [60]. AtTCP4 is predominantly expressed in cotyledons and young leaves [8] and regulates the morphogenesis of these organs [33]. Notably, PaTCP4a, as well as PaTCP2, shows the highest expression levels in the cotyledons, suggesting that they may have a similar role as AtTCP4 in regulating cotyledon development and/or opening. In contrast, PaTCP4b, the duplicated sister gene of PaTCP4a, appears to have lost its expression in the cotyledons and seedlings, implying a possible subfunctionalization for PaTCP4b after gene duplication. This subfunctionalization or even neofunctionalization may also have occurred in PaTCP1a/PaTCP1b, PaTCP12a/PaTCP12b and PaTCP1a/PaTCP1b gene pairs based on their differential expression patterns (Figure 5). However, PaTCP3a and PaTCP3b, as well as PaTCP24a and PaTCP24b, show almost identical expression patterns, suggesting these gene pairs may be resulted from more recent duplications and have redundant functions.
TCP5, TCP13 and TCP17 (TCP5-like) are the CIN-like genes not regulated by miR319 in Arabidopsis. It has been suggested that TCP5/13/17 play redundant regulatory roles in flowering time, leaf and petal growth and axillary branching [19,21,61,62]. Constitutive expression of TCP5, -13 or -17 accelerates flowering time [21], and TCP5 overexpression also leads to smaller petals [61], while knockdown of all three genes by the T-DNA insertion or artificial miRNA, as well as the tcp5/13/17 triple mutation, results in delayed flowering, with larger petals and leaves, although the single tcp5, tcp13 or tcp17 mutant shows no or weak phenotypic alterations [19,21,62]. In petunia, PaTCP5, PaTCP13 and PaTCP17 are the orthologs of AtTCP5, -13 and -17, respectively. There is no target site of PamiR319 in these genes, indicating they are not regulated by miR319. Interestingly, PaTCP5, PaTCP13 and PaTCP17 all exhibit the highest level of expression in the inflorescences (Figure 5), implying that these genes probably perform conserved roles in controlling flower development and/or flowering time.
In core eudicots, the CYC/TB1 subclade contains three main groups: CYC1, CYC2 and CYC3. Increasing functional analyses in recent years indicate that the CYC3, and particularly CYC1 genes, play a central and well-conserved regulatory role in the development of axillary meristems and shoot branching [63], whereas the CYC2 genes are key players in the control of floral symmetry, inflorescence architecture and reproductive organ development [64]. Our study identified seven genes in petunia belonging to the members of CYC/TB1 subclade, namely PaTCP1a/b, PaTCP12a/b and PaTCP18a/b/c, which are corresponding to the CYC2, CYC3 and CYC1 group, respectively. The three petunia CYC1 group of genes (PaTCP18a/b/c) are all expressed predominantly in vegetative axillary buds (Figure 5), which is consistent with the expression patterns of BRANCHED1-like genes in other species, such as AtBRC1 (AtTCP18) and AtBRC2 (AtTCP12) in Arabidopsis [6], PsBRC1 in pea [65], SlBRC1s in tomato [66] and CsBRC1 in cucumber [67], suggesting that PaTCP18a/b/c have a conserved role in regulating axillary bud outgrowth and shoot branching. This was supported by the recent study that identified PhTCP3 (corresponding to PaTCP18c in this study) acting downstream strigolactone (SL) signaling to regulate lateral branching in petunia [51]. In addition, downstream genes in SL signaling have been identified using a microarray in Arabidopsis [68]. It is interesting to further investigate the molecular mechanism for petunia CYC1 group genes in SL pathway. The two CYC3-group genes, PaTCP12a and PaTCP12b, have a litter different expression pattern, with PaTCP12a mainly expressing in axillary buds followed by leaves and stems while PaTCP12b predominantly expressed in leaves, suggesting their potentially divergent roles in the axillary bud and leaf development. AtTCP1, the CYC2 group member in Arabidopsis, exhibits strong expression in the dorsal (adaxial) region of the axillary shoot meristems and early-stage flower meristems, the petiole and midrib of leaves including cotyledons, the distal region of expanding rosette leaves, as well as the lower part of inflorescences stem, and play a role in the longitudinal elongation of leaves [26,69]. PaTCP1a and PaTCP1b, the two orthologs of AtTCP1, are transcribed at high levels in inflorescences that contain flower meristems and flower buds at an early stage of development, implying that these genes may participate in the regulation of inflorescence architecture and/or flower development. In addition, PaTCP1b is also highly expressed in roots, even higher than that in inflorescences, suggesting it has a potential function in root development. Taken together, the petunia CYC/TB1-like genes may have conserved roles in the control of lateral branching, inflorescence architecture and flower development, while some members may also have undergone functional divergence after gene duplication.
Class I PaTCP genes demonstrate two distinct expression patterns (Figure 5). Most genes, such as PaTC6, PaTCP7, PaTCP11, PaTCP14a, PaTCP14b, PaTCP20 and PaTCP23, show a more widespread and less tissue-specific expression profile, which is consistent with the previous results described in Arabidopsis [36], watermelon [39] and grapevine [47], and also coincide with the diverse roles of Class I TCP genes in various biological processes and stress adaptation [56]. While several genes display a tissue-preferential expression pattern, such as PaTC8, PaTCP9, PaTCP15 and PaTCP22. In Arabidopsis, the two best-characterized class I genes AtTCP14 and AtTCP15 are dynamically expressed in young proliferating tissues such as leaf primordia, young inflorescence stems, as well as young flower pedicels, and their expression gradually decreases as the tissues mature [70]. Functional analysis indicates AtTCP14 and AtTCP15 act redundantly to regulate internode elongation and plant stature by promoting cell proliferation in young stem internodes [70]. Furthermore, the ClTCP14a and ClTCP15 of watermelon, closely related to AtTCP14 and AtTCP15, were proven to associate with the internode elongation. Ectopic expression of either ClTCP14a or ClTCP15 resulted in an increase in inflorescence height of wild-type Arabidopsis and was sufficient to restore the stem internode length and inflorescence height of tcp14 tcp15 double-mutant [39]. In petunia, the transcripts of PaTCP14a and PaTCP14b (two orthologs of AtTCP14), as well as PaTCP11, PaTCP15 and PaTCP22, were markedly detected in the stems and/or inflorescences, implying that these genes may have similar roles in the regulation of internode elongation of stems and/or inflorescences. In tomato, three class I genes SlTCP12, SlTCP15 and SlTCP18 are predominantly expressed in developing fruits and were suggested to participate in the regulation of tomato fruit ripening [37]. Noteworthily, PaTCP21 and PaTCP23 represent a relatively high level of expression in developing fruits, suggesting they may play roles in fruit development. PaTCP9, PaTCP14b, PaTCP20, PaTCP21 and PaTCP23 are transcribed at the highest levels in cotyledons, suggesting their potential roles in cotyledon development, similar to the TCP4-like genes in Arabidopsis [8,33,60].
3.3. Potential Roles of PaTCP Genes during Petal Development
The TCP family has been reported to play key roles in the development of lateral organs, which mainly relies on direct transcriptional regulation of cell cycle genes and several hormone pathways [17]. In this study, we investigated the transcriptional profiles of PaTCPs during petal development in large- and small-flowered lines, respectively (Figure 7, Figure S3). As a result, most CIN-TCP genes such as PaTCP3a, PaTCP4a/b, PaTCP13, PaTCP17 and PaTCP24a/b were shown to be expressed at relatively high levels in developing petals of both large- and small-flowed lines (Figure 7), which is consistent with their high levels of expression in flower buds relative to the CYC-TCP members (Figure 5). In addition, these CIN-TCP genes mostly showed gradually increasing expression during the early petal development stages or throughout the whole petal development process, suggesting they function in petal development in petunia. In most species, CIN-TCPs show dynamic expression patterns in the leaves that are undergoing morphogenesis, which is spatiotemporally related to the arrest front during leaf development, indicating they function as repressors of cell division [55]. The upregulation of most petunia CIN-TCPs during petal development is consistent with their role as growth repressors to inhibit cell proliferation and promote post-mitotic differentiation at later stages of fetal development. In Arabidopsis, RABBIT EARS (RBE) inhibits the expression of TCP4 and TCP5 to promote cell proliferation and petal growth during early petal development. However, this repression is alleviated at late developmental stages, which is required for the proper transition from cell division to cell expansion [19,71]. However, it is worth mentioning that PaTCP3b and PaTCP4a showed significantly higher levels of expression in the petals of large flowers (‘L’) than that of small flowers (‘S’), implying that they may play a role in promoting petal growth, which is contradictory with the function of Arabidopsis TCP4 and TCP5 in petal growth, but similar to the role of CIN, a TCP4 homolog in Antirrhinum majus. CIN represses cell proliferation and promotes growth arrest in leaves, but it promotes growth in petals [72]. It is necessary to characterize the functions of petunia CIN-TCP genes, especially PaTCP3b and PaTCP4a, by further studies.
For class I TCPs, most PaTCPs showed very low levels of expression in flower buds and petals, except for PaTCP8, PaTCP14a, PaTCP19a and PaTCP22, which have relatively high expression (more than 0.005 relatives to PhEF1α) during petal development (Figure 5 and Figure 7). In contrast to the CIN-TCPs, these PaTCPs mostly showed decreasing expression trends during the petal growth process and have higher expression levels in ‘S’ than ‘L,’ suggesting they play opposite roles to the CIN-TCPs in petal development. Previously, class I TCP genes are suggested to promote cell proliferation and concomitant organ growth. For example, AtTCP20 is involved in stimulation of cell growth and division [73]. Overexpressing CmTCP20 in Arabidopsis and chrysanthemum plants exhibit larger inflorescences and flower buds and longer petal length [74]. Again, the expression patterns of PaTCP14a/b and PaTCP19b during the petal development support their functions to promote cell proliferation, while higher expression in the petals of small flowers is contrary to the promoting role in petal growth, which needs further study to be understood.
Our promoter analysis shows that most PaTCP genes harbor plenty of hormone-related cis-regulatory elements (Figure 6), implying that these genes may have profound effects on plant developmental processes downstream of hormone pathways. Previous genetic studies indicate that phytohormones and transcription factors have vital roles in different pathways responsible for the determination of flower size in Arabidopsis [75]. Cytokinin is reported to affect the duration of cell division in Arabidopsis developing floral organs [76]. Similarly, it has been reported that cytokinin application accelerates the cell proliferation rate and prolongs the duration of cell proliferation phase during corolla development in petunia, resulting in the formation of larger flowers [77]. The extending corollas of the large- and small-flowered lines ‘L’ and ‘S’ were harvested to measure the levels of auxin, cytokinin, brassinosteroids, ethylene and JA. The levels of IAA, BL, ACC and JA in the corollas of ‘S’ were significantly higher than those of ‘L,’ and the cytokinin level in ‘S’ was also higher than that in ‘L’ (unpublished), which is consistent with previous results [78]. Considering the distribution of a plentiful of hormone-responsive elements in the promoter regions of PaTCP genes (Figure 6), together with accumulating functional studies of the effects of TCPs on hormone biosynthesis, transport and signal transduction in tissues and organs, it could be postulated that PaTCP genes might function as key mediators of hormone-induced changes in plant growth and development. These results indicate that these phytohormone levels are tightly linked with petunia petal size. Taken together, PaTCP transcription factors and phytohormone levels may have an interactional and coefficient relationship in the development of floral organ size in petunia.
4. Materials and Methods
4.1. Plant Materials
Petunia axillaris (S26) and two petunia selfing lines ‘L’ and ‘S,’ with large and small flowers, respectively, were used in this study. The seeds of S26 were originally obtained from the Swammerdam Institute for Life Sciences, University of Amsterdam, The Netherlands (gifted by prof. Ronald Koes), and the two selfing lines were developed by our laboratory. Plant materials were grown in phytotron under a long-day condition (16 h light/8 h dark) at 22–25 °C with a luminous intensity of 12,000 lux and relative humidity of 70–80%.
4.2. Identification of TCP Genes from Petunia Species
Two different approaches were used to identify TCP genes in petunia genomes. Firstly, petunia TCP genes were retrieved from P. axillaris N and P. inflata S6 genome databases [50] in the Sol Genomics Network (SGN) (https://solgenomics.net/) using the TBLASTN algorithm with the hidden Markov model (HMM) for the TCP domain (PF03634) obtained from the Pfam database (http://pfam.sanger.ac.uk). Secondly, the TCP protein sequences of 24 Arabidopsis TCPs and 30 tomato TCPs, which were downloaded from the Arabidopsis Information Resource (TAIR) database (http://www.arabidopsis.org) and the Solanaceae Genomics Network (https://solgenomics.net/), respectively, were used as queries to search the genome datasets of P. axillaris and P. inflata [50]. Furthermore, all obtained TCP sequences from petunia were put in InterproScan (http://www.ebi.ac.uk/InterProScan) to confirm the presence of the TCP domain. Subsequently, the TSA (Transcriptome Shotgun Assembly) databases of P. axillaris (GBRU) and P. integrifolia (GBRV) as well as P. integrifolia subsp. inflata (GBDS) were searched for the transcripts of the identified PaTCPs and PiTCPs genes by nucleotide BLAST in the NCBI [52]. The molecular weight (MW) and isoelectric points (pI) of PaTCP and PiTCP proteins were computed by the ExPASy website (https://web.expasy.org/protparam/).
4.3. Multiple Sequence Alignments and Phylogenetic Analysis
TCP protein sequences of the 24 AtTCPs and 32 SlTCPs were retrieved from TAIR (http://www.arabidopsis.org/) and the tomato genome database in SGN, respectively. Multiple sequence alignments of the full-length protein sequences of TCP proteins of petunia, Arabidopsis and tomato were conducted using the Clustal W program of MEGA 7.0 software. An unrooted phylogenetic tree based on the alignments was constructed by the neighbor-joining (NJ) method using Jones-Taylor-Thornton (JTT) substitution model with Gamma-distributed rates (five categories) and bootstrap values were calculated with 500 replicates.
4.4. Gene Structure and Conserved Motif Analysis
The exon-intron structure of 66 petunia genes (32 PaTCPs from P. axillaris and 34 PiTCPs from P. inflata) was analyzed using the Gene Structure Display Server 2.0 (http://gsds.cbi.pku.edu.cn/).
The conserved TCP domains of PaTCP proteins were confirmed by the Conserved Domain Database (CDD) in NCBI with default parameters. The conserved and potential motifs of 32 PaTCP protein sequences were analyzed by the online Multiple Expectation Maximization for Motif Elicitation (MEME) (http://meme-suite.org/tools/meme) with the optimized parameter settings: repetition number, any; minimum motif width, 6; maximum motif width, 50; maximum number of motifs, 20.
4.5. PamiR319-Targeted Site Prediction
The mature sequences of PamiR319 were obtained from the miRbase Database (http://www.mirbase.org). The coding sequences of 32 PaTCP genes and mature sequences of PamiR319 (Table S4) were analyzed using the psRNATarget online application (http://plantgrn.noble.org/psRNATarget/) with default parameters.
4.6. Putative Cis-Element Analysis of PaTCP Promoters
The promoter sequences of 32 PaTCPs genes were retrieved from P. axillaris genome database and the promoter sequences of upstream 1.5 kb region from the start codon for each gene were used for putative promoter cis-acting element analysis in PlantCARE online program (http://bioinformatics.psb.ugent.be/webtools/plantcare/html/).
4.7. RNA Isolation and Real-Time Quantitative RT-PCR Analysis
For the investigation of spatial expression profiles of PaTCP genes, different vegetative and reproductive tissues were harvested from P. axillaris, including germinating seeds (3 d (day) after sowing), cotyledons (7 d after sowing), 4-euphylla seedlings, roots, stems, leaves, axillary buds of adult plants (after flowering), inflorescences, flower buds (0.5 cm) and immature fruits (7 d after pollination). Moreover, samples used in the analysis of the temporal expression patterns of PaTCP genes were taken from the large- and small-flowered lines of ‘L’ and ‘S’ at five different floral developmental stages, including young flower buds (petal length < 0.5 cm, S1), extending flower buds (when flower buds just enclosed by sepals, S2), pre-anthesis flowers (when flower buds extended to full length, S3), semi-open flowers (S4) and fully blooming flowers before the anthers dehisced (S5), respectively. Corollas were detached from flower buds at each development stages, with the sepals, stamens and pistils removed, and then frozen in liquid nitrogen immediately.
RNA extraction and qPCR were performed as described previously [79]. Gene-specific primers for PaTCPs were designed using Primer 5.0 and were listed in Table S4. The amplification efficiency of each primer pair was examined by melting curve analysis. For all qPCR experiments, three biological replicates were analyzed for each sample and the mean values ± SD (standard deviation) were calculated. The petunia EF1α gene was used as the internal control. The 2 −ΔΔCT method [80] was used for calculating the relative expression level of each gene.
4.8. Subcellular Localization of PaTCP Proteins
The full-length coding sequences of PaTCP3a, PaTCP4a and PaTCP12b, except for the stop codon, were amplified (primers are listed in Table S4) and inserted into the pMD18-T vector (Takara, Japan). Digestion of these vectors was conducted using KpnI and Sal I (Thermo Fisher Scientific, Waltham mass, MA, USA) restriction endonuclease enzymes. Then, the digested products were purified and immediately ligated into the appropriate sites of the 35S promoter-driven yellow fluorescent protein (YFP)-fusion vector (L101-YFP). All the constructed plasmids were confirmed by sequencing. The Agrobacterium tumefaciens EHA105 strains harboring fusion constructs were transformed into tobacco epidermal cells. Signals were detected by confocal laser microscopy (Leica SP8).
5. Conclusions
In this work, 32 PaTCP genes in P. axillaris and 34 PiTCP genes in P. inflata were identified, respectively. For PaTCP genes, we carried out a comprehensive analysis and characterization. Based on the multiple sequence alignments and phylogenetic analysis, 32 PaTCP genes were categorized into two classes, which was supported by the highly conserved exon-intron structure and motif compositions. The spatial-temporal expression analysis revealed that the PaTCP genes may be involved in multiple aspects of plant growth and development. Remarkably, some PaTCP genes showed significantly different expression levels between the small-flowered line ‘S’ and the large-flowered line ‘L,’ suggesting their potential roles in petal development and/or specification of petal size. Moreover, the number of cis-regulatory elements observed in the PaTCP promoter sequences are likely to be conducive to mediate hormone and/or stress responses. Taken together, the extensive genome-wide analysis of PaTCP genes lays a solid foundation for unraveling the potential functions of PaTCPs during plant growth and development.
Acknowledgments
We thank Ronald Koes in the Swammerdam Institute for Life Sciences, University of Amsterdam, The Netherlands for gifting the seeds of Petunia axillaris. Our thanks to all colleagues in our laboratory for helpful discussions and technical assistance.
Abbreviations
| ABA | Abscisic acid |
| ACC | 1-aminocyclopropane-1-carboxylic acid |
| bHLH | Basic helix-loop-helix |
| BR | Brassinosteroids |
| CYC | Cycloidea |
| IAA | Indole-3-acetic acid |
| JA | Jasmonic acid |
| MeJA | Methyl jasmonate |
| MW | Molecular weight |
| NJ | Neighbor-Joining |
| PCF | Proliferating cell factor |
| pI | Isoelectric point |
| qPCR | Real-time quantitative RT-PCR |
| SL | Strigolactones |
| TB1 | Teosinte branched1 |
| TFs | Transcription factors |
| UTR | Untranslated region |
| YFP | Yellow fluorescence protein |
Supplementary Materials
The following are available online at https://www.mdpi.com/1422-0067/21/18/6594/s1.
Author Contributions
S.Z. designed and performed the experiments, carried out the bioinformatics analyses and drafted the manuscript. Q.Z. and F.C. participated in guiding and carrying out some experiments. B.L., L.W. and F.L. participated in data analysis and taking care of the plant materials. M.B. and J.Z. participated in guiding and designing this project. G.L. provided the idea and designed the framework of this study, carried out the preliminary bioinformatics analyses and modified the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by the National Natural Science Foundation of China (grant nos. 31471914 and 31772345) and the Natural Science Foundation of Guangdong Province, China (grant no. 2019A1515011840).
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design and performance of the study nor in the writing of the manuscript.
References
- 1.Ingram G.C., Waites R. Keeping it together: Co-ordinating plant growth. Curr. Opin. Plant Biol. 2006;9:12–20. doi: 10.1016/j.pbi.2005.11.007. [DOI] [PubMed] [Google Scholar]
- 2.Cubas P., Lauter N., Doebley J., Coen E. The TCP domain: A motif found in proteins regulating plant growth and development. Plant. J. 1999;18:215–222. doi: 10.1046/j.1365-313X.1999.00444.x. [DOI] [PubMed] [Google Scholar]
- 3.Martin-Trillo M., Cubas P. TCP genes: A family snapshot ten years later. Trends Plant. Sci. 2010;15:31–39. doi: 10.1016/j.tplants.2009.11.003. [DOI] [PubMed] [Google Scholar]
- 4.Navaud O., Dabos P., Carnus E., Tremousaygue D., Hervé C. TCP transcription factors predate the emergence of land plants. J. Mol. Evol. 2007;65:23–33. doi: 10.1007/s00239-006-0174-z. [DOI] [PubMed] [Google Scholar]
- 5.Howarth D.G., Donoghue M.J. Phylogenetic analysis of the “ECE” (CYC/TB1) clade reveals duplications predating the core eudicots. Proc. Natl. Acad. Sci. USA. 2006;103:9101–9106. doi: 10.1073/pnas.0602827103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Aguilar-Martinez J.A., Poza-Carrion C., Cubas P. Arabidopsis BRANCHED1 acts as an integrator of branching signals within axillary buds. Plant Cell. 2007;19:458–472. doi: 10.1105/tpc.106.048934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Koyama T., Furutani M., Tasaka M., Ohme-Takagi M. TCP transcription factors control the morphology of shoot lateral organs via negative regulation of the expression of boundary-specific genes in Arabidopsis. Plant Cell. 2007;19:473–484. doi: 10.1105/tpc.106.044792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Palatnik J.F., Allen E., Wu X., Schommer C., Schwab R., Carrington J.C., Weigel D. Control of leaf morphogenesis by microRNAs. Nature. 2003;425:257–263. doi: 10.1038/nature01958. [DOI] [PubMed] [Google Scholar]
- 9.Aguilar-Martinez J.A., Sinha N. Analysis of the role of Arabidopsis class I TCP genes AtTCP7, AtTCP8, AtTCP22, and AtTCP23 in leaf development. Front. Plant Sci. 2013;4:406. doi: 10.3389/fpls.2013.00406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schommer C., Palatnik J.F., Aggarwal P., Chetelat A., Cubas P., Farmer E.E., Nath U., Weigel D. Control of jasmonate biosynthesis and senescence by miR319 targets. PLoS Biol. 2008;6:e230. doi: 10.1371/journal.pbio.0060230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Luo D., Carpenter R., Vincent C., Copsey L., Coen E. Origin of floral asymmetry in Antirrhinum. Nature. 1996;383:794–799. doi: 10.1038/383794a0. [DOI] [PubMed] [Google Scholar]
- 12.Nag A., King S., Jack T. miR319a targeting of TCP4 is critical for petal growth and development in Arabidopsis. Proc. Natl. Acad. Sci. USA. 2009;106:22534–22539. doi: 10.1073/pnas.0908718106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lucero L.E., Manavella P.A., Gras D.E., Ariel F.D., Gonzalez D.H. Class I and Class II TCP transcription factors modulate SOC1-dependent flowering at multiple levels. Mol. Plant. 2017;10:1571–1574. doi: 10.1016/j.molp.2017.09.001. [DOI] [PubMed] [Google Scholar]
- 14.Takeda T., Amano K., Ohto M.-A., Nakamura K., Sato S., Kato T., Tabata S., Ueguchi C. RNA interference of the Arabidopsis putative transcription factor TCP16 gene results in abortion of early pollen development. Plant Mol. Biol. 2006;61:165–177. doi: 10.1007/s11103-006-6265-9. [DOI] [PubMed] [Google Scholar]
- 15.Resentini F., Felipo-Benavent A., Colombo L., Blazquez M.A., Alabadi D., Masiero S. TCP14 and TCP15 mediate the promotion of seed germination by gibberellins in Arabidopsis thaliana. Mol. Plant. 2015;8:482–485. doi: 10.1016/j.molp.2014.11.018. [DOI] [PubMed] [Google Scholar]
- 16.Kim S.H., Son G.H., Bhattacharjee S., Kim H.J., Nam J.C., Nguyen P.D.T., Hong J.C., Gassmann W. The Arabidopsis immune adaptor SRFR 1 interacts with TCP transcription factors that redundantly contribute to effector-triggered immunity. Plant. J. 2014;78:978–989. doi: 10.1111/tpj.12527. [DOI] [PubMed] [Google Scholar]
- 17.Nicolas M., Cubas P. TCP factors: New kids on the signaling block. Curr. Opin. Plant Biol. 2016;33:33–41. doi: 10.1016/j.pbi.2016.05.006. [DOI] [PubMed] [Google Scholar]
- 18.Bresso E.G., Chorostecki U., Rodriguez R.E., Palatnik J.F., Schommer C. Spatial control of gene expression by miR319-regulated TCP transcription factors in leaf development. Plant Physiol. 2018;176:1694–1708. doi: 10.1104/pp.17.00823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Huang T., Irish V.F. Temporal control of plant organ growth by TCP transcription factors. Curr. Biol. 2015;25:1765–1770. doi: 10.1016/j.cub.2015.05.024. [DOI] [PubMed] [Google Scholar]
- 20.Kubota A., Ito S., Shim J.S., Johnson R.S., Song Y.H., Breton G., Goralogia G.S., Kwon M.S., Laboy Cintron D., Koyama T., et al. TCP4-dependent induction of CONSTANS transcription requires GIGANTEA in photoperiodic flowering in Arabidopsis. PLoS Genet. 2017;13:e1006856. doi: 10.1371/journal.pgen.1006856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Li D., Zhang H., Mou M., Chen Y., Xiang S., Chen L., Yu D. Arabidopsis Class II TCP transcription factors integrate with the FT–FD module to control flowering. Plant Physiol. 2019;181:97–111. doi: 10.1104/pp.19.00252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lan J., Qin G. The regulation of CIN-like TCP transcription factors. Int. J. Mol. Sci. 2020;21:4498. doi: 10.3390/ijms21124498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Poza-Carrión C., Aguilar-Martínez J.A., Cubas P. Role of TCP gene BRANCHED1 in the control of shoot branching in Arabidopsis. Plant Signal. Behav. 2007;2:551–552. doi: 10.4161/psb.2.6.4811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Niwa M., Daimon Y., Kurotani K., Higo A., Pruneda-Paz J.L., Breton G., Mitsuda N., Kay S.A., Ohme-Takagi M., Endo M., et al. BRANCHED1 interacts with FLOWERING LOCUS T to repress the floral transition of the axillary meristems in Arabidopsis. Plant Cell. 2013;25:1228–1242. doi: 10.1105/tpc.112.109090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guo Z., Fujioka S., Blancaflor E.B., Miao S., Gou X., Li J. TCP1 modulates brassinosteroid biosynthesis by regulating the expression of the key biosynthetic gene DWARF4 in Arabidopsis thaliana. Plant Cell. 2010;22:1161–1173. doi: 10.1105/tpc.109.069203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Koyama T., Sato F., Ohme-Takagi M. A role of TCP1 in the longitudinal elongation of leaves in Arabidopsis. Biosci. Biotechnol. Biochem. 2010;74:2145–2147. doi: 10.1271/bbb.100442. [DOI] [PubMed] [Google Scholar]
- 27.Daviere J.M., Wild M., Regnault T., Baumberger N., Eisler H., Genschik P., Achard P. Class I TCP-DELLA interactions in inflorescence shoot apex determine plant height. Curr. Biol. 2014;24:1923–1928. doi: 10.1016/j.cub.2014.07.012. [DOI] [PubMed] [Google Scholar]
- 28.Lucero L.E., Uberti-Manassero N.G., Arce A.L., Colombatti F., Alemano S.G., Gonzalez D.H. TCP15 modulates cytokinin and auxin responses during gynoecium development in Arabidopsis. Plant J. 2015;84:267–282. doi: 10.1111/tpj.12992. [DOI] [PubMed] [Google Scholar]
- 29.Gastaldi V., Lucero L.E., Ferrero L.V., Ariel F.D., Gonzalez D.H. Class I TCP transcription factors activate the SAUR63 gene subfamily in gibberellin-dependent stamen filament elongation. Plant Physiol. 2020;182:2096–2110. doi: 10.1104/pp.19.01501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Herve C., Dabos P., Bardet C., Jauneau A., Auriac M.C., Ramboer A., Lacout F., Tremousaygue D. In vivo interference with AtTCP20 function induces severe plant growth alterations and deregulates the expression of many genes important for development. Plant Physiol. 2009;149:1462–1477. doi: 10.1104/pp.108.126136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shunichi K., Yuko O. DNA binding and dimerization specificity and potential targets for the TCP protein family. Plant J. 2002;30:337–348. doi: 10.1046/j.1365-313x.2002.01294.x. [DOI] [PubMed] [Google Scholar]
- 32.Li S. The Arabidopsis thaliana TCP transcription factors: A broadening horizon beyond development. Plant Signal. Behav. 2015;10:e1044192. doi: 10.1080/15592324.2015.1044192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Koyama T., Mitsuda N., Seki M., Shinozaki K., Ohme-Takagi M. TCP transcription factors regulate the activities of ASYMMETRIC LEAVES1 and miR164, as well as the auxin response, during differentiation of leaves in Arabidopsis. Plant Cell. 2010;22:3574–3588. doi: 10.1105/tpc.110.075598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Uberti-Manassero N.G., Lucero L.E., Viola I.L., Vegetti A.C., Gonzalez D.H. The class I protein AtTCP15 modulates plant development through a pathway that overlaps with the one affected by CIN-like TCP proteins. J. Exp. Bot. 2012;63:809–823. doi: 10.1093/jxb/err305. [DOI] [PubMed] [Google Scholar]
- 35.Danisman S., van der Wal F., Dhondt S., Waites R., de Folter S., Bimbo A., van Dijk A.D., Muino J.M., Cutri L., Dornelas M.C., et al. Arabidopsis class I and class II TCP transcription factors regulate jasmonic acid metabolism and leaf development antagonistically. Plant Physiol. 2012;159:1511–1523. doi: 10.1104/pp.112.200303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Xuan Y., Hong M., Jian W., Dabing Z. Genome-wide comparative analysis and expression pattern of TCP gene families in Arabidopsis thaliana and Oryza sativa. J. Integr. Plant Biol. 2007;49:885–897. [Google Scholar]
- 37.Parapunova V., Busscher M., Busscher-Lange J., Lammers M., Karlova R., Bovy A.G., Angenent G.C., de Maagd R.A. Identification, cloning and characterization of the tomato TCP transcription factor family. BMC Plant Biol. 2014;14:157. doi: 10.1186/1471-2229-14-157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhou Y., Xu Z., Zhao K., Yang W., Cheng T., Wang J., Zhang Q. Genome-wide Identification, Characterization and Expression Analysis of the TCP Gene Family in Prunus mume. Front. Plant Sci. 2016;7:1301. doi: 10.3389/fpls.2016.01301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Shi P., Guy K.M., Wu W., Fang B., Yang J., Zhang M., Hu Z. Genome-wide identification and expression analysis of the ClTCP transcription factors in Citrullus lanatus. BMC Plant Biol. 2016;16:85. doi: 10.1186/s12870-016-0765-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wei W., Hu Y., Cui M., Han Y., Gao K., Feng J. Identification and transcript analysis of the TCP transcription factors in the diploid woodland strawberry Fragaria vesca. Front. Plant Sci. 2016;7:1937. doi: 10.3389/fpls.2016.01937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ma X., Ma J., Fan D., Li C., Jiang Y., Luo K. Genome-wide identification of TCP family transcription factors from Populus euphratica and their involvement in leaf shape regulation. Sci. Rep. 2016;6:32795. doi: 10.1038/srep32795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Du J., Hu S., Yu Q., Wang C., Yang Y., Sun H., Yang Y., Sun X. Genome-wide identification and characterization of BrrTCP transcription factors in Brassica rapa ssp. rapa. Front. Plant Sci. 2017;8:1588. doi: 10.3389/fpls.2017.01588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Liu Y., Guan X., Liu S., Yang M., Ren J., Guo M., Huang Z., Zhang Y. Genome-wide identification and analysis of TCP transcription factors involved in the formation of leafy head in Chinese cabbage. Int. J. Mol. Sci. 2018;19:847. doi: 10.3390/ijms19030847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang H., Wang H., Liu R., Xu Y., Lu Z., Zhou C. Genome-wide identification of TCP family transcription factors in Medicago truncatula reveals significant roles of miR319-targeted TCPs in nodule development. Front. Plant Sci. 2018;9:774. doi: 10.3389/fpls.2018.00774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tran C.D., Chu H.D., Nguyen K.H., Watanabe Y., La H.V., Tran K.D., Tran L.-S.P. Genome-wide identification of the TCP transcription factor family in chickpea (Cicer arietinum L.) and their transcriptional responses to dehydration and exogenous abscisic acid treatments. J. Plant Growth Regul. 2018;37:1286–1299. doi: 10.1007/s00344-018-9859-y. [DOI] [Google Scholar]
- 46.Huo Y., Xiong W., Su K., Li Y., Yang Y., Fu C., Wu Z., Sun Z. Genome-wide analysis of the TCP gene family in switchgrass (Panicum virgatum L.) Int. J. Genom. 2019;2019:8514928. doi: 10.1155/2019/8514928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Leng X., Wei H., Xu X., Ghuge S.A., Jia D., Liu G., Wang Y., Yuan Y. Genome-wide identification and transcript analysis of TCP transcription factors in grapevine. BMC Genom. 2019;20:786. doi: 10.1186/s12864-019-6159-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gerats T., Vandenbussche M. A model system for comparative research: Petunia. Trends Plant Sci. 2005;10:251–256. doi: 10.1016/j.tplants.2005.03.005. [DOI] [PubMed] [Google Scholar]
- 49.Vandenbussche M., Chambrier P., Rodrigues Bento S., Morel P. Petunia, Your Next Supermodel? Front. Plant Sci. 2016;7:72. doi: 10.3389/fpls.2016.00072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bombarely A., Moser M., Amrad A., Bao M., Bapaume L., Barry C.S., Bliek M., Boersma M.R., Borghi L., Bruggmann R., et al. Insight into the evolution of the Solanaceae from the parental genomes of Petunia hybrida. Nat. Plants. 2016;2:16074. doi: 10.1038/nplants.2016.74. [DOI] [PubMed] [Google Scholar]
- 51.Drummond R.S., Janssen B.J., Luo Z., Oplaat C., Ledger S.E., Wohlers M.W., Snowden K.C. Environmental control of branching in petunia. Plant Physiol. 2015;168:735–751. doi: 10.1104/pp.15.00486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Guo Y., Wiegert-Rininger K.E., Vallejo V.A., Barry C.S., Warner R.M. Transcriptome-enabled marker discovery and mapping of plastochron-related genes in Petunia spp. BMC Genom. 2015;16:726. doi: 10.1186/s12864-015-1931-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Arabidopsis Genome Initiative Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature. 2000;408:796–815. doi: 10.1038/35048692. [DOI] [PubMed] [Google Scholar]
- 54.Lescot M., Déhais P., Thijs G., Marchal K., Moreau Y., Van de Peer Y., Rouzé P., Rombauts S. PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res. 2002;30:325–327. doi: 10.1093/nar/30.1.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sarvepalli K., Nath U. CIN-TCP transcription factors: Transiting cell proliferation in plants. IUBMB Life. 2018;70:718–731. doi: 10.1002/iub.1874. [DOI] [PubMed] [Google Scholar]
- 56.Dhaka N., Bhardwaj V., Sharma M.K., Sharma R. Evolving tale of TCPs: New paradigms and old lacunae. Front. Plant Sci. 2017;8:479. doi: 10.3389/fpls.2017.00479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ori N., Cohen A.R., Etzioni A., Brand A., Yanai O., Shleizer S., Menda N., Amsellem Z., Efroni I., Pekker I., et al. Regulation of LANCEOLATE by miR319 is required for compound-leaf development in tomato. Nat. Genet. 2007;39:787–791. doi: 10.1038/ng2036. [DOI] [PubMed] [Google Scholar]
- 58.Nath U., Crawford B.C., Carpenter R., Coen E. Genetic control of surface curvature. Science. 2003;299:1404–1407. doi: 10.1126/science.1079354. [DOI] [PubMed] [Google Scholar]
- 59.Seki K., Komatsu K., Tanaka K., Hiraga M., Kajiya-Kanegae H., Matsumura H., Uno Y. A CIN-like TCP transcription factor (LsTCP4) having retrotransposon insertion associates with a shift from Salinas type to Empire type in crisphead lettuce (Lactuca sativa L.) Hortic. Res. 2020;7:15. doi: 10.1038/s41438-020-0241-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Dong J., Sun N., Yang J., Deng Z., Lan J., Qin G., He H., Deng X.W., Irish V.F., Chen H., et al. The transcription factors TCP4 and PIF3 antagonistically regulate organ-specific light induction of SAUR genes to modulate cotyledon opening during de-etiolation in Arabidopsis. Plant Cell. 2019;31:1155–1170. doi: 10.1105/tpc.18.00803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Van Es S.W., Silveira S.R., Rocha D.I., Bimbo A., Martinelli A.P., Dornelas M.C., Angenent G.C., Immink R.G.H. Novel functions of the Arabidopsis transcription factor TCP5 in petal development and ethylene biosynthesis. Plant J. 2018;94:867–879. doi: 10.1111/tpj.13904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Efroni I., Blum E., Goldshmidt A., Eshed Y. A protracted and dynamic maturation schedule underlies Arabidopsis leaf development. Plant Cell. 2008;20:2293–2306. doi: 10.1105/tpc.107.057521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wang M., Le Moigne M.A., Bertheloot J., Crespel L., Perez-Garcia M.D., Oge L., Demotes-Mainard S., Hamama L., Daviere J.M., Sakr S. BRANCHED1: A key hub of shoot branching. Front. Plant Sci. 2019;10:76. doi: 10.3389/fpls.2019.00076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nicolas M., Cubas P. Plant Transcription Factors. Academic Press; Boston, MA, USA: 2016. The role of tcp transcription factors in shaping flower structure, leaf morphology, and plant architecture; pp. 249–267. [Google Scholar]
- 65.Braun N., de Saint Germain A., Pillot J.P., Boutet-Mercey S., Dalmais M., Antoniadi I., Li X., Maia-Grondard A., Le Signor C., Bouteiller N., et al. The pea TCP transcription factor PsBRC1 acts downstream of strigolactones to control shoot branching. Plant Physiol. 2012;158:225–238. doi: 10.1104/pp.111.182725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Martin-Trillo M., Grandio E.G., Serra F., Marcel F., Rodriguez-Buey M.L., Schmitz G., Theres K., Bendahmane A., Dopazo H., Cubas P. Role of tomato BRANCHED1-like genes in the control of shoot branching. Plant J. 2011;67:701–714. doi: 10.1111/j.1365-313X.2011.04629.x. [DOI] [PubMed] [Google Scholar]
- 67.Shen J., Zhang Y., Ge D., Wang Z., Song W., Gu R., Che G., Cheng Z., Liu R., Zhang X. CsBRC1 inhibits axillary bud outgrowth by directly repressing the auxin efflux carrier CsPIN3 in cucumber. Proc. Natl. Acad. Sci. USA. 2019;116:17105–17114. doi: 10.1073/pnas.1907968116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kumar M., Kim I., Kim Y.-K., Hero J.B., Suh M.C., Kim H.U. Strigolactone signaling genes showing differential expression patterns in Arabidopsis max mutants. Plants. 2019;8:352. doi: 10.3390/plants8090352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Cubas P., Coen E., Zapater J.M. Ancient asymmetries in the evolution of flowers. Curr. Biol. 2001;11:1050–1052. doi: 10.1016/S0960-9822(01)00295-0. [DOI] [PubMed] [Google Scholar]
- 70.Kieffer M., Master V., Waites R., Davies B. TCP14 and TCP15 affect internode length and leaf shape in Arabidopsis. Plant J. 2011;68:147–158. doi: 10.1111/j.1365-313X.2011.04674.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Jing L., Wang Y., Zhang Y., Wang W., Irish V.F., Huang T. RABBIT EARS regulates the transcription of TCP4 during petal development in Arabidopsis. J. Exp. Bot. 2016;67:6473–6480. doi: 10.1093/jxb/erw419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Crawford B.C., Nath U., Carpenter R., Coen E.S. CINCINNATA controls both cell differentiation and growth in petal lobes and leaves of Antirrhinum. Plant Physiol. 2004;135:244–253. doi: 10.1104/pp.103.036368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Li C., Potuschak T., Colon-Carmona A., Gutierrez R.A., Doerner P. Arabidopsis TCP20 links regulation of growth and cell division control pathways. Proc. Natl. Acad. Sci. USA. 2005;102:12978–12983. doi: 10.1073/pnas.0504039102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Wang J., Guan Y., Ding L., Li P., Zhao W., Jiang J., Chen S., Chen F. The CmTCP20 gene regulates petal elongation growth in Chrysanthemum morifolium. Plant Sci. 2019;280:248–257. doi: 10.1016/j.plantsci.2018.12.008. [DOI] [PubMed] [Google Scholar]
- 75.Krizek B.A., Anderson J.T. Control of flower size. J. Exp. Bot. 2013;64:1427–1437. doi: 10.1093/jxb/ert025. [DOI] [PubMed] [Google Scholar]
- 76.Bartrina I., Otto E., Strnad M., Werner T., Schmulling T. Cytokinin regulates the activity of reproductive meristems, flower organ size, ovule formation, and thus seed yield in Arabidopsis thaliana. Plant Cell. 2011;23:69–80. doi: 10.1105/tpc.110.079079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Takaaki N., Miyaki H., Sasaki K., Okazawa T. Cultivar and anatomical analysis of corolla enlargement of petunia (Petunia hybrida Vilm.) by cytokinin application. Sci. Hortic. 2006;111:49–55. [Google Scholar]
- 78.Takaaki N., Tomoya N., Tomoko N. Corolla of the large-flowered petunia (Petunia hybrida Vilm.) cultivars exhibit low endogenous cytokinin concentration through enhanced expression of the genes encoding cytokinin oxidases. J. Jpn. Soc. Hortic. Sci. 2011;80:334–342. [Google Scholar]
- 79.Zhou Q., Zhang S., Chen F., Liu B., Wu L., Li F., Zhang J., Bao M., Liu G. Genome-wide identification and characterization of the SBP-box gene family in Petunia. BMC Genom. 2018;19:193. doi: 10.1186/s12864-018-4537-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Livak K.J., Schmittgen T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.